Abstract
Foxp3+ regulatory T (Treg) cells contribute to the local dysfunctional immune environment in endometriosis, an estrogen-dependent gynecological disease, which affects the function of ectopic endometrial tissue clearance by the immune system. The reason for the high percentage of peritoneal Treg in endometriosis patients is unknown. Here, we show that the proportion of peritoneal Treg cells increases as endometriosis progresses. To determine the probable mechanism, we established a naive T cell-macrophage-endometrial stromal cell (ESC) co-culture system to mimic the peritoneal cavity microenvironment. After adding 1-methyl-tryptophan (1-MT), a specific inhibitor of indoleamine 2,3-dioxygenase-1 (IDO1), to the co-culture system, we found that the differentiation of Treg cells, mainly IL-10+ Treg cells, decreased. Therefore, 1-MT-pretreated ESCs-educated Treg cells performed impaired suppressive function. Moreover, estrogen promoted the differentiation of Treg cells by elevating IDO1 expression in the ectopic lesion. Subsequently, we examined mannose receptor C, type 2 (MRC2), which is an up-stream molecule of IL-10, by bioinformatics analysis and real-time PCR validation. MRC2 expression in ectopic ESCs was notably lower than that in normal ESCs, which further negatively regulated the expression of IDO1 and Ki-67 in ESCs. Furthermore, MRC2 is required for Treg differentiation in the ectopic lesion, especially that for CD4high Treg. Therefore, MRC2-silenced ESCs-educated Treg manifested a stronger suppressive function in vitro. Consistently, the percentage of Treg increased when MRC2-shRNA was administered in the peritoneal cavity of endometriosis-disease mice model. Besides, 1-MT improved the condition of endometriosis, in terms of reducing the number and weight of total ectopic lesions in vivo. These results indicate that the estrogen-IDO1-MRC2 axis participates in the differentiation and function of Treg and is involved in the development of endometriosis. Thus, blockage of IDO1 in the ectopic lesion, which does not influence physiological functions of estrogen, may be considered a potential therapy for endometriosis.
Under the influence of various factors, sloughed endometrial-like tissue in retrograde menstruation reaches the peritoneal cavity and adheres to endoabdominal structures to form ectopic lesions, resulting in dysmenorrhea, chronic pelvic pain and infertility, referred to as endometriosis (EMS).1, 2 At first, endometriosis was considered a benign, estrogen-dependent gynecological disease. However, it has been subsequently recognized as not only an endocrine disorder, but also a chronic inflammatory condition.
To date, three main aspects have been noted in the pathogenesis of endometriosis. Firstly, with regard to endometrial tissue, a lot of genes are differentially expressed in the ectopic endometrium compared with that in the eutopic and normal endometrium,3, 4 which may play pivotal roles in the development of endometriosis. We previously showed that the expression of indoleamine 2,3-dioxygenase-1 (IDO1), a rate-limiting enzyme that catalyzes the synthesis of tryptophan, is higher in ectopic endometrial stromal cells (ESCs) than that in normal ESCs. Additionally, IDO1 suppresses T-cell responses, promotes immune tolerance, and influences the differentiation of regulatory T (Treg) cells.5 We found that IDO1 promotes survival, proliferation, and invasion of ESCs via the JNK signaling pathway, but inhibits apoptosis of ESCs.6 The second aspect involved is abnormal endocrine function. High expression of mitochondrial cholesterol side-chain cleavage enzyme (CYP11A1) and hydroxysteroid (17 beta) dehydrogenase (HSD17B) in ectopic endometrial tissue increases local estrogen levels, which influences biological activities of ESCs,7, 8 leading to the development of endometriosis. Finally, the immunological aspect has been implicated, owing to evidence of immune tolerance in the endometriosis microenvironment, which is affected by changes in the proportion of Treg cells,9 which in turn plays an important role in the maintenance of immune homeostasis to prevent potentially severe autoimmunity.10, 11 It has been reported that the percentage of Treg cells in the peritoneal fluid of patients with endometriosis is higher than that in healthy women,12 and that Foxp3 expression by Treg cells and Treg function is increased in estrogen-treated mice.13 However, the exact mechanisms are unknown.
Considering the higher peritoneal Treg cell percentage in EMS patients than that in healthy women, as well as findings that IDO1 controls Treg cell function in response to inflammatory stimuli,14, 15 and higher expression of IDO1 in ectopic lesion regulates biological activities of ESCs in endometriosis,6 we reasoned that excessive estrogen may regulate IDO1 expression in the ectopic lesion to induce Treg cell differentiation. In this study, we explored the origin of excessive Treg cells in the peritoneal fluid of patients with endometriosis. To our knowledge, this is the first report showing that mannose receptor C, type 2 (MRC2), which is related to collagen turnover 16 and cancer prognosis,17, 18 plays a vital role in Treg cell differentiation and function in endometriosis. Clinically, our findings may provide evidence that 1-methyl-tryptophan (1-MT) has potential applications in the treatment of endometriosis, retaining the physiological functions of estrogen.
Results
Peritoneal Treg increases as endometriosis progresses
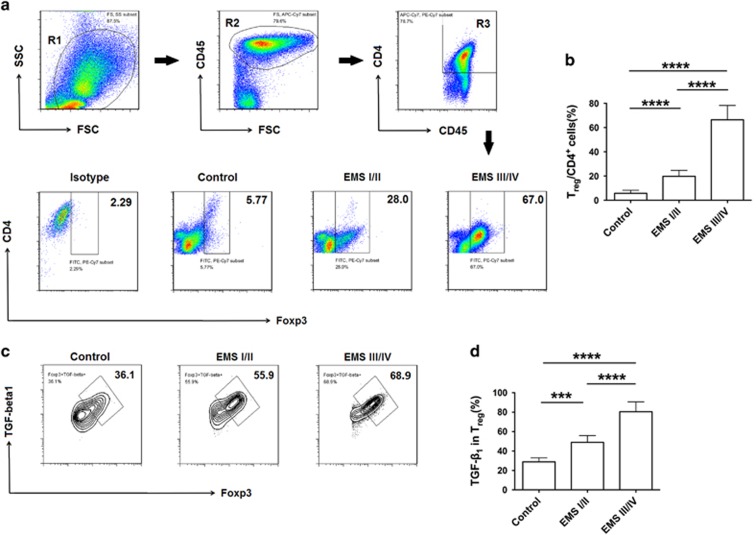
The percentage of Treg cells in peritoneal fluid is higher in patients with endometriosis than in healthy women.12 As shown in Figure 1a and b, the proportion of peritoneal Treg cells in EMS stage III/IV patients was significantly higher than that in EMS stage I/II patients and healthy women. TGF-β1 expression in peritoneal Treg cells also showed a similar trend (Figure 1c and d); however, IL-10 expression in Treg cells did not (data not shown). These data demonstrated that the percentage of peritoneal Treg cells increases with exacerbation of endometriosis.
Figure 1.
Percentage of Treg cells in peritoneal fluid increases as endometriosis progresses. (a) Complete gating strategy for peritoneal Treg cells. Gate R2 is inclusive of gate R1; cells of gate R2 represent CD45+ cells. Gate R3 is inclusive of gate R2; cells of gate R3 represent CD4+CD45+ cells. Peritoneal fluid from a patient with EMS stage III/IV is represented above. Flow cytometric analysis was used to determine the percentage of Treg cells in peritoneal fluid of endometriosis patients in different stages. Numbers in quadrants indicate the percentage of cells. (b) Quantification of the percentage of Treg cells in a. Values indicate mean±S.D., n (Control)=6, n (EMS stage I/II)=6, n (EMS stage III/IV)=6, ****P<0.0001, two-tailed, unpaired t-test. (c) Flow cytometric analysis was used to determine the expression of TGF- β1 in peritoneal Treg cells of endometriosis patients in different stages. Numbers in quadrants indicate the percentage of cells. (d) Quantification of TGF-β1 expression of Treg cells in c. Values indicate mean±S.D., n (Control)=6, n (EMS stage I/II)=6, n (EMS stage III/IV)=6, ***P<0.001, ****P<0.0001, two-tailed, unpaired t-test. EMS I/II, stage I and II of endometriosis; EMS III/IV, stage III and IV of endometriosis
IDO1 participates in the differentiation of Treg cells in endometriosis
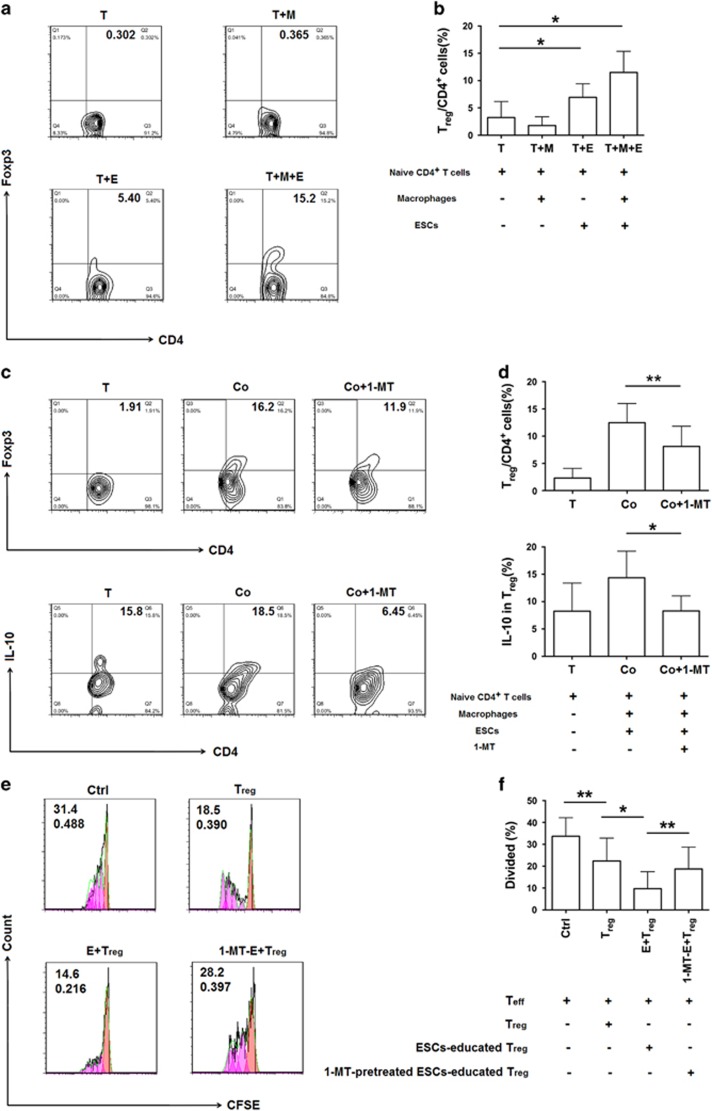
To verify whether IDO1 influences the differentiation of Treg cells in the ectopic lesion, we established co-culture systems to mimic the microenvironment of the peritoneal cavity (Supplementary Figure 1) and detected Treg cell differentiation and expression of functional molecules in Treg cells when treated with 1-MT or not. The highest percentage of Treg was found in the naive T cell-macrophage-ESC co-culture system, compared with that in two other co-culture systems (Figure 2a and b). Therefore, we used this co-culture system for further analysis.
Figure 2.
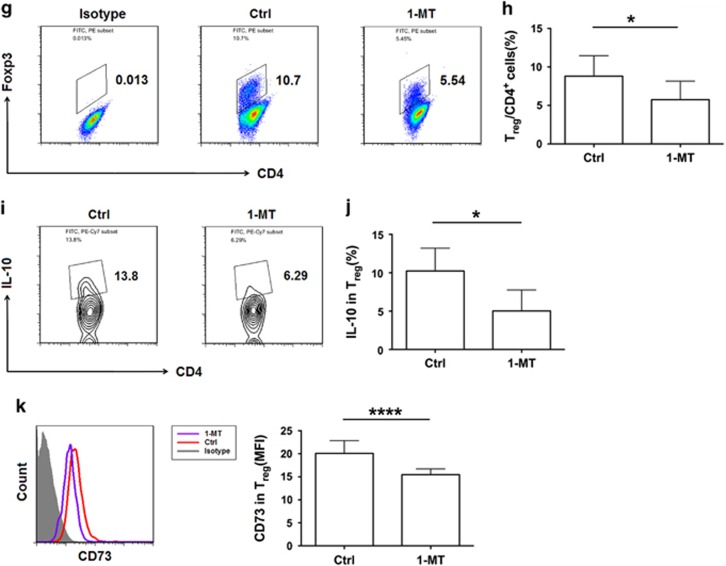
IDO1 is involved in the differentiation of Treg in endometriosis. (a) Ectopic ESCs were incubated alone or co-cultured with positively sorted CD14+ cells from human peripheral blood, while negatively sorted naive CD4+ T cells from peripheral blood were cultured in 24-well plates that coated with 5 μg/ml monoclonal anti-CD3 and 1 μg/ml monoclonal anti-CD28, in the presence of 50 ng/ml recombinant human IL-2. After 48 hours, macrophages and naive CD4+ T cells were collected to establish co-culture systems. The percentage of CD4+Foxp3+ T cells in co-culture systems were determined by flow cytometric analysis after 5 days. Numbers in quadrants indicate the percentage of cells. (b) Quantification of Treg cells in a. Values indicate mean±S.D., n=7, *P<0.05, two-tailed, paired t-test. (c) Flow cytometric analysis was used to determine the percentage of Treg cells and the expression of IL-10 of Treg cells in the co-culture system with or without 1-MT. Numbers in quadrants indicate the percentage of cells. (d) Quantification of the percentage of Treg cells and the expression of IL-10 of Treg cells in c. Values indicate mean±S.D., n=7, *P<0.05, **P<0.01, two-tailed, paired t-test. (e) Flow cytometric analysis was performed to determine the proliferation of Teff cells (CFSE-labeled) from cultured with non-treated-, ESCs-educated-, 1-MT-pretreated ESCs-educated-Treg cells or not. Numbers in quadrants indicate the percentage of divided cells and division index. Ctrl group contains Teff cells alone; Treg group contains Teff cells and non-treated Treg cells; E+Treg group contains Teff cells and ESCs-educated Treg cells; 1-MT-E+Treg group contains Teff cells and 1-MT-pretreated ESCs-educated Treg cells. E, ESCs. Peaks represent generations of cells. Salmon peak represents parental cells, magenta peaks represent daughter cells of Teff cells. (f) Quantification of the divided percentage shown in (e). Values indicate mean±S.D., n=10, *P<0.05, **P<0.01, two-tailed, paired t-test. (g) Flow cytometric analysis was used to determine the percentage of CD4+Foxp3+ T cells in peritoneal fluid of mice with endometriosis in PBS- (Ctrl) and 1-MT- administered groups. Numbers in quadrants indicate the percentage of cells. (h) Quantification of the percentage of CD4+Foxp3+ T cells in (g). Values indicate mean±S.D., n=8, *P<0.05, two-tailed, unpaired t-test. (i) Flow cytometric analysis was used to determine the expression of IL-10 in peritoneal Treg cells in vivo. Numbers in quadrants indicate the percentage of cells. (j) Quantification of IL-10 expression in Treg cells shown in (i). Values indicate mean±S.D., n=8, *P<0.05, two-tailed, paired t-test. (k) MFI of the expression of CD73 in Treg of peritoneal fluid from endometriosis-disease mice model in PBS (Ctrl) and 1-MT administration groups. Values are means±S.D., n=8, ****P<0.0001, two-tailed, unpaired t-test. T, naive CD4+ T cells; M, macrophages; E, ESCs; Co group in (c and d), the naive T cell-macrophage-endometrial stromal cell (ESC) co-culture system
After adding 1-MT, a specific inhibitor of IDO1, to the co-culture system, the differentiation of Treg cells decreased significantly, especially that of IL-10+ Treg cells (Figure 2c and d), whereas, TGF-β1+ Treg cell remained unchanged (data not shown). Moreover, 1-MT-pretreated ESCs-educated Treg performed less suppressive function, as the divided percentage of CD4+CD25− (Teff) cells was higher than that of E+Treg group, while ESCs-educated Treg cells owned a more suppressive function compared with non-pretreated-Treg (Figure 2e and f). These results indicate that IDO1 is involved in the differentiation and suppressive function of Treg cells in endometriosis. To investigate whether IDO1 participates in the differentiation of Treg cells in endometriosis in vivo, we established an endometriosis-disease mouse model (Supplementary Figure 2). The results showed that 1-MT distinctly inhibited the percentage of Treg cells in peritoneal fluid of mice (Figures 2g and h), especially IL-10 and CD 73 expression in peritoneal Treg cells (Figures 2i and k), which were consistent with the in vitro results.
IDO1 is up-regulated by estrogen in the ectopic lesion
Patients with endometriosis show high local estrogen levels.7 Additionally, IDO1 expression in ectopic ESCs is higher than that in normal ESCs,6 leading us to consider that estrogen may regulate the expression of IDO1 in the ectopic lesion. We found that IDO1 expression in estrogen-conditioned ESCs and estrogen-conditioned macrophages were obviously higher than that in the control groups (Figures 3c–f). Besides, the effect of ESCs on up-regulating the expression of IDO1 in macrophages was more significant than that with estrogen alone (Figures 3e and f), which indicates a crosstalk between ESCs and macrophages that related to IDO1 expression.
Figure 3.
Expression of IDO1 is up-regulated by estrogen in the ectopic lesion. (a) Complete gating strategy of ectopic ESCs. Gate R2 is inclusive of gate R1; cells of gate R2 represent ESCs. (b) Complete gating strategy of monocytes. Gate R2 is inclusive of gate R1; cells of gate R2 represent CD14+ cells. (c) Flow cytometric analysis was used to determine the expression of IDO1 in ESCs (Ctrl), estrogen-treated ESCs (E2), monocyte-treated ESCs (M), and monocyte-treated ESCs in the presence of estrogen (M+E2). (d) MFI of the expression of IDO1 in groups shown in (c). Values indicate mean±S.D., n=5, *P<0.05, two-tailed, paired t-test. (e) Flow cytometric analysis was used to determine the expression of IDO1 in monocytes (Ctrl), estrogen-treated monocytes (E2), ESCs-treated monocytes (e), and ESCs-treated monocytes in the presence of estrogen (E+E2). (f) MFI of the expression of IDO1 in groups shown in (e). Values indicate mean±S.D., n=13, *P<0.05, two-tailed, paired t-test. (g) ESCs were pretreated with estrogen receptor-α inhibitor (ERαi), estrogen receptor-β inhibitor (ERβi), or estrogen receptors inhibitor (ERi) for 24 h, washed and then estrogen was added to each group, except control group. Control (Ctrl) group contained untreated ESCs. Flow cytometric analysis was used to determine the expression of IDO1 in ESCs from these groups. (h) MFI of the expression of IDO1 shown in (g). Values indicate mean±S.D., n=4, *P<0.05, **P<0.01, two-tailed, paired t-test
To identify which sub-unit estrogen receptor (ER) that allows estrogen to promote the expression of IDO1 in ESCs, we blocked ERα, ERβ, and ER respectively. We found that IDO1 expression in ESCs was down-regulated regardless of whether a single or both sub-unit ERs were blocked. This suggests that both sub-units of ER are involved in this activity, especially ERβ (Figures 3g and h). Although the percentage of Treg cells in ectopic lesions of the estrogen receptor inhibitor (ERi) group showed little changes in vivo (data not shown), the percentage of TGF-β1+, IL-10+, CD73+, and CTLA-4+ Treg cells decreased (Supplementary Figure 3).
Estrogen induces the differentiation of Treg cells via IDO1
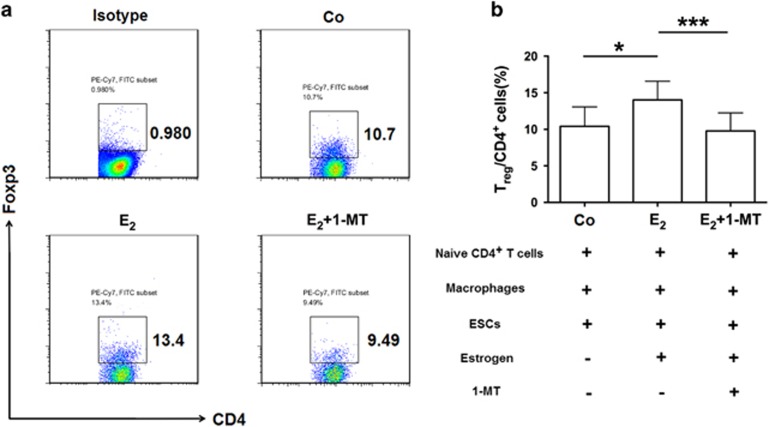
Considering that endometriosis is an estrogen-dependent disease, and that estrogen enhances Foxp3 expression and Treg cell function,13 we explored whether estrogen regulates the differentiation of Treg cells in endometriosis. After adding estrogen to the co-culture system, the differentiation of Treg cells notably increased (Figures 4a and b), which indicates that local high estrogen level participates in inducing the differentiation of Treg cells in the peritoneal fluid of patients with endometriosis.
Figure 4.
Estrogen promotes the differentiation of Treg cells through elevated IDO1 levels in the ectopic lesion. (a) Flow cytometric analysis was used to determine the percentage of Treg cells in the co-culture system obtained with estrogen, estrogen plus 1-MT, or neither. Numbers in quadrants indicate the percentage of cells. (b) Quantification of Treg cells shown in a. Values indicate mean±S.D., n=12, *P<0.05, ***P<0.001, two-tailed, paired t-test. Co, the naive T cells-macrophage-endometrial stromal cell (ESC) co-culture system. E2, estrogen
As mentioned above, estrogen promotes the expression of IDO1 in the ectopic lesion, and IDO1 influences the differentiation of Treg cells,5 raising the possibility that estrogen may promote the differentiation of Treg cells via up-regulation of IDO1 expression. Compared with that in the estrogen group, the differentiation of Treg cells in estrogen plus 1-MT group was lower (Figures 4a and b), which manifested that 1-MT inhibits Treg cell differentiation induced by estrogen.
MRC2 is a downstream molecule of IDO1 and negatively regulates IDO1
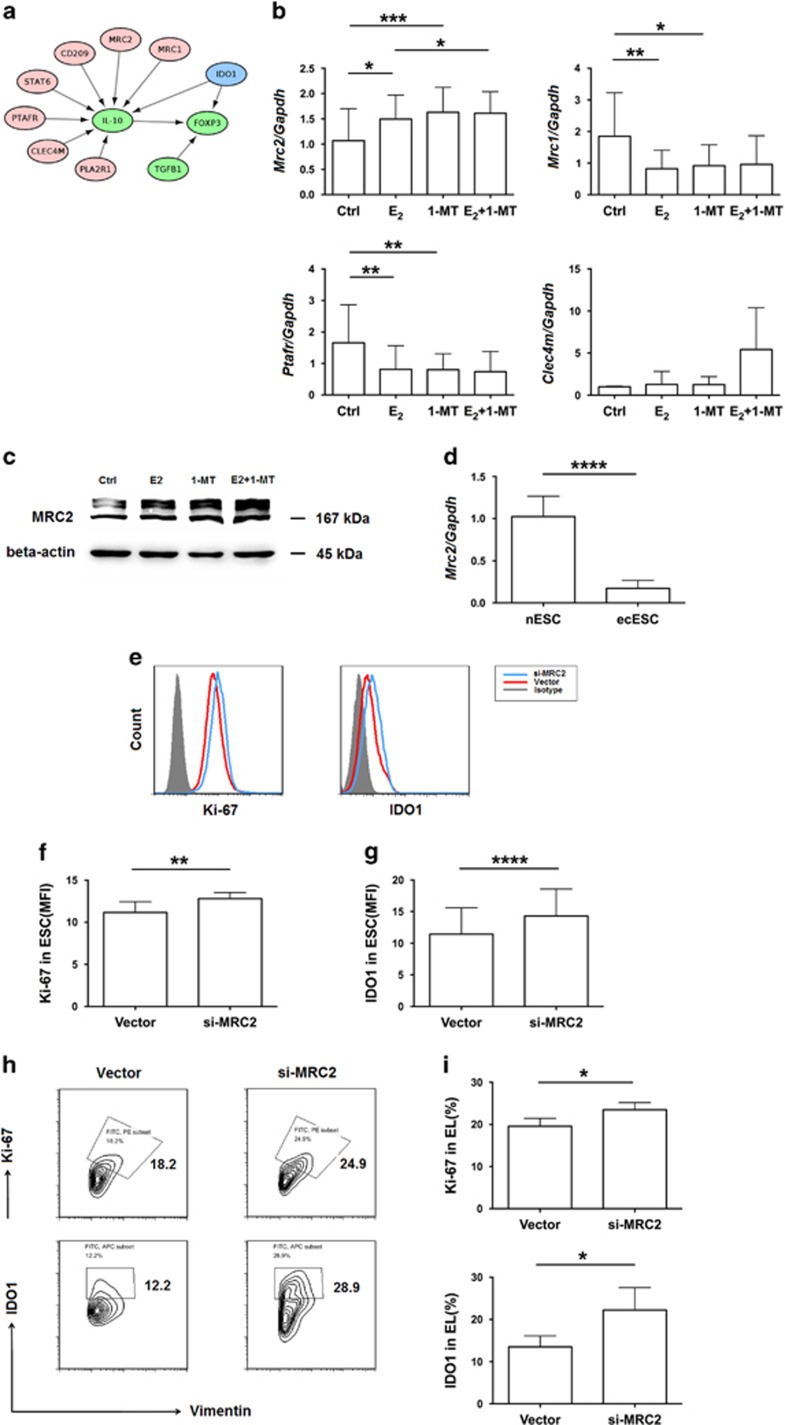
As outlined above, IDO1 influences the differentiation of Treg cells, especially IL-10+ Treg cells. To identify factors that possibly connect IDO1 and IL-10, and are involved in interactions of IL10, IDO1, Foxp3, and TGF-β, we performed a search of the KEGG database as part of the bioinformatics analysis. We found that molecules up-stream of IL-10 include C-type lectin domain family 4 member M (CLEC4M), mannose receptor C, type 1 (MRC1), mannose receptor C, type 2 (MRC2), STAT6, phospholipase A2 receptor1 (PLA2R1), platelet-activating factor receptor (PTAFR), and CD209 (Figure 5a). After treating ESCs with estrogen, 1-MT, or estrogen plus 1-MT, only MRC2 expression increased in the estrogen-treated group compared with that in the control group (Figure 5b), which was similar to our previous result where estrogen up-regulated IDO1 expression in ESCs (Figures 3c and d). Thus, the results demonstrate that 1-MT also promotes the expression of MRC2 in ESCs to a greater extent than estrogen (Figures 5b and c), which means that MRC2 is downstream to IDO1 and estrogen.
Figure 5.
MRC2 is a downstream molecule of IDO1 and negatively regulates IDO1 expression. (a) Interactions of IDO1, IL-10, TGF-β1, and Foxp3. (b) mRNA expression of MRC2 (n=13), MRC1 (n=9), PTAFR (n=9), CLEC4M (n=6) in ESCs (Ctrl group), estrogen-treated ESCs (E2 group), 1-MT-treated ESCs (1-MT group), and estrogen plus 1-MT-treated ESCs (E2+1-MT group) was determined. Values indicate mean±S.D., *P<0.05, **P<0.01, ***P<0.001, two-tailed, paired t-test. (c) Protein expression of MRC2 shown in (b) analyzed by western blot. (d) mRNA expression of MRC2 in normal ESCs (nESC, n=9) and ectopic ESCs (ecESC, n=15). Values indicate mean±S.D., ****P<0.0001, two-tailed, unpaired t-test. (e) Flow cytometric analysis was used to determine the expression of IDO1 and Ki-67 in MRC2-silenced (si-MRC2) ESCs and ESCs of vector group. (f, g) MFI of the expression of IDO1 and Ki-67 shown in (e), n=8, **P<0.01, ****P<0.0001, two-tailed, paired t-test. (h) Flow cytometric analysis was used to determine the expression of Ki-67 and IDO1 in ectopic lesions of vector- and MRC2 shRNA-administered groups in vivo. Numbers in quadrants indicate the percentage of cells. (i) Quantification of the expression of Ki-67 and IDO1 in ectopic lesions in (h). Values indicate mean±S.D., n=6, *P<0.05, two-tailed, unpaired t-test
MRC2 is a constitutively recycling endocytic receptor belonging to the mannose receptor family.19 The expression of MRC2 in ectopic ESCs was significantly lower than that in normal ESCs (Figure 5d). Combined with evidence that the expression of IDO1 in ectopic ESCs is higher than that in normal ESCs,6 and 1-MT up-regulates the expression of MRC2, we reasoned that high levels of IDO1 might lead to low expression of MRC2 in ectopic ESCs. Besides, after silencing MRC2 in ESCs, the expression of IDO1 increased (Figures 5e and g), which indicates a negative feedback between MRC2 and IDO1. Therefore, the level of Ki-67 in MRC2-silenced ESCs increased compared with that in the vector group (Figures 5e and f), which is consistent with the notion that ectopic ESCs have a stronger ability for proliferation.6 Similarly, the expression of Ki-67 and IDO1 also increased in ectopic lesions after MRC2 shRNA was intraperitoneal injected to the peritoneal cavity of endometriosis mice model in vivo (Figures 5h and i).
MRC2 is required for the differentiation of Treg cells in endometriosis
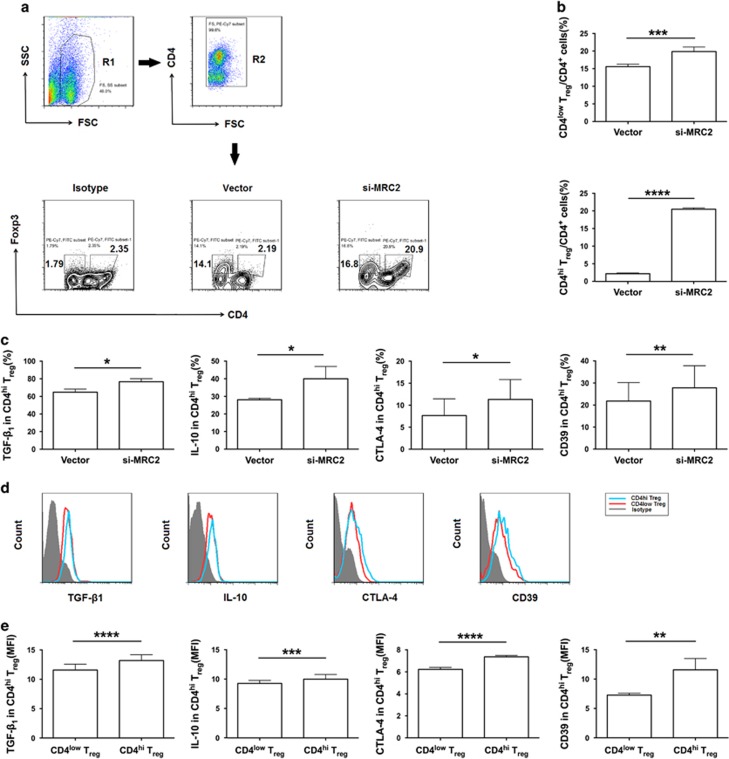
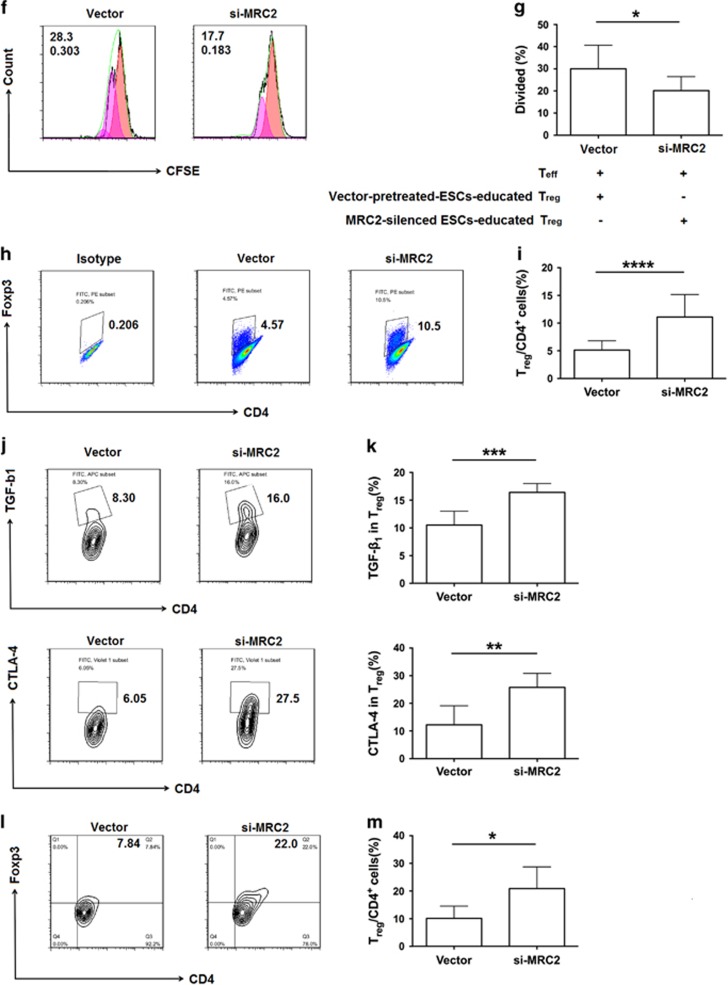
According to the findings above, MRC2 is downstream to IDO1, and IDO1 is involved in the differentiation of Treg in ectopic lesion, hinting the possibility that MRC2 may participate in the activity that IDO1 regulates the differentiation of Treg in endometriosis. When MRC2-silenced ESCs were co-cultured with naive CD4+ T cells and monocytes-derived macrophages, the percentage of CD4low Treg and CD4high Treg cells were higher in the MRC2-silenced group than that in the vector group, especially CD4high Treg cells (Figures 6a and b). Moreover, CD4high Treg cells from the MRC2-silenced group showed a more immunosuppressive phenotype, with higher expression of TGF-β1, IL-10, CD39 and CTLA-4, than that in CD4high Treg cells in the vector group and CD4low Treg cells in MRC2-silenced group (Figures 6c and e). Furthermore, MRC2-silenced ESCs-educated Treg performed stronger suppressive function, as divided percentage of Teff cells was lower than that of vector group, which indicated that MRC2 is involved in Treg suppressive function (Figures 6f and g). In vivo, at two weeks after administrating MRC2-shRNA via the peritoneal cavity of the endometriosis-disease mouse model, the percentage of peritoneal Treg cells and Treg cells in ectopic lesions were significantly higher than that in the vector group (Figures 6h, i, l and m), and mainly comprised TGF-β1+ and CTLA-4+ Treg cells in the peritoneal fluid (Figures 6j and k). These results demonstrate that MRC2 is not only responsible for the differentiation of Treg cells, mainly that of CD4high Treg cells; but also promotes Treg immunosuppressive function in endometriosis, which may exacerbate the development of endometriosis.
Figure 6.
MRC2 is required for the differentiation and immunosuppressive function of Treg cells in endometriosis. (a) Complete gating strategy of Treg cells from the co-culture system. Gate R2 is inclusive of gate R1; cells of gate R2 represent CD4+ cells. Flow cytometric analyses CD4+Foxp3+ T cells in the co-culture system contained MRC2-silenced ESCs or vector-treated ESCs. Numbers in quadrants indicate the percentage of Treg cells. (b) Quantification of CD4low and CD4high Treg cells in a. Values indicate mean±S.D., n=6, ***P<0.001, ****P<0.0001, two-tailed, paired t-test. (c) Expression of TGF-β1 (n=5), IL-10 (n=4), CTLA-4 (n=7), and CD39 (n=6) in CDhigh Treg cells shown in (a). Values indicate mean±S.D., *P<0.05, **P<0.01, two-tailed, paired t-test. (d) Flow cytometric analysis was performed to determine the expression of TGF-β1, IL-10, CTLA-4, and CD39 in CD4high Treg cells of si-MRC2 group shown in (a). (e) MFI of TGF-β1 (n=6), IL-10 (n=8), CTLA-4 (n=7) and CD39 (n=7) in CD4high Treg cells of si-MRC2 group shown in a. Values indicate mean±S.D., **P<0.01, ***P<0.001, ****P<0.0001, two-tailed, paired t-test. (f) Flow cytometric analysis was performed to determine the proliferation of Teff cells (CFSE-labeled) from cultured with vector-pretreated-ESCs-educated-, or MRC2-silenced ESCs-educated Treg cells. Numbers in quadrants indicate the percentage of divided cells and division index. Vector group contains Teff cells and vector-pretreated-ESCs-educated Treg cells; si-MRC2 group contains Teff cells and MRC2-silenced ESCs-educated Treg cells. Peaks represent generations of cells. Salmon peak represents parental cells, magenta peaks represent daughter cells of Teff cells. (g) Quantification of the divided percentage of Teff cells shown in (f). Values indicate mean±S.D., n=6, *P<0.05, two-tailed, paired t-test. (h) Flow cytometric analyses peritoneal CD4+Foxp3+ T cells in vivo from vector-administered or MRC2 shRNA-administered group. Numbers in quadrants indicate the percentage of Treg cells. (i) Flow cytometric analysis was performed to determine the percentage of peritoneal CD4+Foxp3+ T cells of vector group or si-MRC2 group in vivo. Values indicate mean±S.D., n=13, ****P<0.0001, two-tailed, unpaired t-test. (j) Flow cytometric analyses expression of TGF-β1 and CTLA-4 of peritoneal CD4+Foxp3+ T cells in vivo from vector-administered or MRC2 shRNA-administered group. Numbers in quadrants indicate the percentage of cells. (k) Expression of TGF-β1 and CTLA-4 in peritoneal Treg cells in vivo shown in (j). Values indicate mean±S.D., n=7, ***P<0.001, two-tailed, unpaired t-test. (l) Flow cytometric analyses the percentage of CD4+Foxp3+ T cells in ectopic lesions in vivo from vector-administered or MRC2 shRNA-administered group. Numbers in quadrants indicate the percentage of cells. (m) Quantification of Treg cells shown in (l). Values indicate mean±S.D., n=6, *P<0.05, two-tailed, unpaired t-test. si-MRC2, silenced-MRC2; CD4hi Treg, CD4high Treg
1-MT reverses the development of endometriosis in vivo
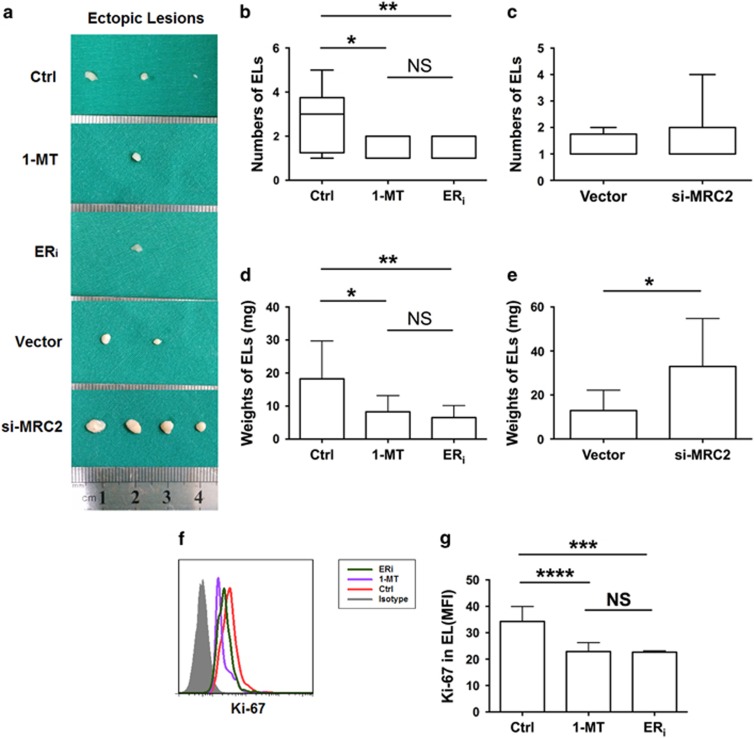
To determine whether 1-MT improves the condition of endometriosis in vivo, we measured the total number and weight of ectopic lesions and found that both parameters decreased notably in the 1-MT administered group compared with that in the control group, as similar as ERi administered group (Figures 7a, b and d). In the MRC2-shRNA administered group, the total number of ectopic lesions did not significantly increased (Figure 7c), but total weight of ectopic lesions was obviously higher than that in the vector group (Figure 7e), which indicates that MRC2 is responsible for ectopic lesion growth while not for the spread of the implant. Moreover, 1-MT notably restrained the proliferation of ectopic lesions in vivo, in terms of lower expression of Ki-67 compared with that in the control group, as seen with the ERi group (Figures 7f and g). These results suggest that 1-MT reverses the condition of endometriosis in vivo.
Figure 7.
1-MT reverses the development of endometriosis in vivo. (a) Represent of ectopic lesions from PBS- (Ctrl), 1-MT-, ERi-, Vector-, or MRC2 sh-RNA-administered group. (b and c) Numbers of total ectopic lesions in mice administered PBS (n=8), 1-MT (n=9), estrogen receptor inhibitor (n=10), vector (n=8), and MRC2-shRNA (n=11). Values indicate mean±S.D., *P<0.05, **P<0.01, two-tailed, unpaired t-test. (d, e) Weights of total ectopic lesions from PBS (n=8), 1-MT (n=9), estrogen receptor inhibitor (n=10), vector (n=8) and MRC2-shRNA (n=11) administered groups in vivo. Values indicate mean±S.D., *P<0.05, two-tailed, unpaired t-test. (f and g) Flow cytometric analysis was used to determine Ki-67 expression (MFI) in ectopic lesions shown in PBS (n=8), 1-MT (n=9), estrogen receptor inhibitor (n=10) administration group in vivo. Values indicate mean±S.D., ***P<0.001, ****P<0.0001, two-tailed, unpaired t-test. ERi, estrogen receptor inhibitor; si-MRC2, silenced-MRC2; Ctrl group, PBS administration group
Discussion
Although endometriosis is an estrogen-dependent disease, it is not enough to determine the pathogenesis of endometriosis from the endocrine perspective alone. Multiple factors participate in the development and maintenance of endometriosis, including immunological dysfunction, genetic susceptibility, psychological factors, and environment factors. Among these, the immunological aspect of endometriosis has recently been extensively studied. Up to 90% of women of reproductive age exhibit retrograde menstruation, whereas only 6–10% of these women develop endometriosis.20 This indicates that a microenvironment of immune tolerance is formed within the ectopic lesion in patients with endometriosis, where the function of menstrual debris clearance by the local immune responses is ineffectively.21
Several types of immune cells are involved in the formation of the local immune tolerance environment in endometriosis, such as Treg cells, macrophages, and natural-killer (NK) cells. Disturbance of Treg cells, which are responsible for self-tolerance, maintenance of immune homeostasis, and immunosuppressive functions,22 aggravates the condition of endometriosis. In this study, we have demonstrated not only that the percentage of Treg cells in the peritoneal fluid of endometriosis patients higher than that in healthy women, which is consistent with results of previous published studies,23 but also that the augmentation of Treg cell percentage, especially that of TGF-β1+ Treg cells, occurs in parallel with endometriosis exacerbation. This suggests that the detection of peritoneal Treg cells may be used as an indicator to assess the severity of endometriosis. However, the origin of excessive Treg cells in the peritoneal fluid of patients with endometriosis is yet unclear. Three assumptions may elucidate this clinic phenomenon. One is homing of peripheral Treg cells to the peritoneal fluid as the percentage of Treg cells decreases in the peripheral blood of EMS patients.23 The second is that local naive T cells differentiate into Treg cells under the influence of various factors involved; and the third is self-proliferation of local Treg cells. In fact, all of these assumptions may be involved in the increase of peritoneal Treg cell percentage in endometriosis.
IDO1 is essential for the generation and function of Treg cells.14, 24, 25 Initially, IDO1 was thought as the first line of host defense against infectious pathogens, as it causes tryptophan shortage, which in turn restricts mammalian cell growth,26 especially that of T cells.27 Subsequent studies revealed that IDO1 has multiple immunological functions, such as suppressing T-cell responses, regulating functions of Treg cells,14 and promoting immune tolerance.15 Evidence of higher IDO1 expression in ectopic ESCs than in normal ESCs6 and high percentage of peritoneal Treg cells in endometriosis23 demonstrates that IDO1 may play an important role in inducing the differentiation of Treg cells in the ectopic lesion, which is a probable reason for excessive Treg cells in the peritoneal fluid of patients with endometriosis.
In this study, we established a naive T cell-macrophage-ESC co-culture system to mimic the local microenvironment of the peritoneal cavity. The results of our study show that IDO1 participates in the differentiation of Treg cells in the ectopic lesion, evidenced by inhibition of Treg cell differentiation, especially that of IL-10+ Treg cells, by 1-MT. In addition, 1-MT-pretreated ESCs-educated Treg cells suppressed the proliferation of Teff cells less effectively compared with non-treated ESCs-educated Treg cells, which indicates that 1-MT is involved in Treg cell suppressive function. Moreover, estrogen promoted the expression of IDO1 in both ESCs and macrophages, which, on a side-note, explains that ectopic ESCs have high expression of IDO1 in endometriosis. Therein, the expression of IDO1 in ESCs-educated macrophages was notably higher than that in both estrogen-treated and untreated monocytes-derived macrophages. This demonstrates a crosstalk between ESCs and macrophages, involving IDO1. Besides, estrogen was found to induce Treg cell differentiation in the ectopic lesion and IDO1 was involved in this process, wherein, 1-MT down-regulated Treg cell differentiation induced by estrogen. These results indicate that estrogen regulates the expression of IDO1 in the ectopic lesion to induce the Treg cell differentiation.
Based on the finding that 1-MT mainly influences the differentiation of IL-10+ Treg cells; we performed a search of the KEGG database to identify possible molecules that connect IDO1 and IL-10. The results show that MRC2, which is an upstream molecule of IL-10, is essential for the differentiation and function of Treg cells. Most researches on MRC2 to date have focused on its role in the development of cancer, as it can promote breast tumor growth,17 co-operate with the matrix metalloproteinase to remodel of extracellular matrix that attenuate renal fibrosis,28 and predict prognosis of hepatocellular carcinoma18 and prostate cancer.29 Besides, MRC2 is also closely related to collagen turnover.30 In our study, we found that both estrogen and 1-MT promoted the expression of MRC2 in ESCs, which indicates that MRC2 is downstream to estrogen and IDO1. Therein, the regulatory ability of 1-MT was stronger than that of estrogen. Combined with the low expression of MRC2 by ectopic ESCs and MRC2 expression promoted by estrogen, we reasoned that other factors might be responsible for decreased MRC2 expression by ESCs. As shown in Figures 5b and c, we observed that 1-MT promoted the expression of MRC2 by ESCs to a greater extent than estrogen, whereas, decreased IDO1 levels caused an increase in MRC2 expression. Therefore, it is a possibility that increased IDO1 levels down-regulate the expression of MRC2 by ESCs. In addition, negative feedback was noted between IDO1 and MRC2, evidenced by increase in IDO1 expression when MRC2 in ESCs was silenced. Meanwhile, MRC2 negatively regulated proliferation of ESCs as the expression of Ki-67 increased in si-MRC2 ESCs compared with vector group. To explore the possibility that MRC2 influences the differentiation of Treg cells, we co-cultured MRC2-silenced ESC with macrophages and naive CD4+ T cells. Compared with that in the vector group, the percentage of Treg cells increased in the MRC2-silenced group, especially CD4high Treg cells, which demonstrate that MRC2 plays a key role in the differentiation of Treg cells in endometriosis. Therefore, MRC2-silenced ESCs-educated Treg cells acquired stronger suppressive function than vector-pretreated ESCs-educated Treg cells, which indicates that MRC2 negatively regulates Treg cell suppressive function. To our knowledge, this is the first report demonstrating the immunological functions of MRC2 as an important regulator of Treg cell differentiation and function, and showing that estrogen and IDO1 are up-stream to MRC2. These results illustrate that estrogen-IDO1-MRC2 axis is involved in the differentiation of Treg cells in endometriosis.
Consistent with the in vitro results above, 1-MT dramatically reversed the condition of endometriosis in vivo, irrespective of total number or weight of ectopic lesions, expression of Ki-67 in total ectopic lesions, or the percentage of peritoneal Treg cells. These data suggest that locally applied 1-MT may clinically relieve sufferings of patients with endometriosis. Besides, MRC2 significantly induced the differentiation of Treg cells, increased weight of total ectopic lesions, and promoted the expression of Ki-67 in ectopic lesions in vivo, which is consistent with the in vitro results, suggesting that MRC2 is involved in the growth of ectopic lesions and Treg differentiation in endometriosis (Supplementary Figure 4).
Although endometriosis is a benign gynecological disease, its biological activities are similar to that of cancer, including metastasis, plantation, angiogenesis, immune tolerance, and recurrence.31, 32 In the present study, we focused on IDO1, MRC2, and Treg cells, which have been reported in previous cancer studies,17, 33–35 to identify the endocrine-immune mechanism for the high percentage of peritoneal Treg cells in patients with endometriosis. The results revealed that the percentage of Treg cells in the peritoneal fluid increases as endometriosis progresses. Therein, we found that the lower expression of MRC2 in ectopic ESCs significantly promoted the differentiation and function of Treg cells. This is a novel function of MRC2, identified in this study. Future work would involve identifying probable mechanisms by which MRC2 influences the differentiation of Treg cells, including the transcription factors involved in this process and interactions of MRC2 and Treg functional molecules. Moreover, the possibility that MRC2 influences the biological activities of ESCs in endometriosis remains to be explored. Common treatment options for endometriosis, including progestogens, ovulation induction, GnRH analog and surgery, however, typically do not provide long-term relief.36 Pharmacotherapy for endometriosis affects the physiological functions of estrogen and this limits its long-term use. In addition, high recurrence rate renders endometriosis clinically intractable. Therefore, to treat endometriosis, it is essential to identify possible targets that do not influence the physiological functions of estrogen. The in vivo results from this study showed that 1-MT, a specific inhibitor of IDO1, can notably improve the condition of endometriosis, as similar as ER inhibitor, which provide evidences that blocking IDO1 in ectopic lesions may be a novel treatment option for endometriosis, not only by virtue of its effects on biological activities of ESCs, but also on local immune-tolerance environment effected by inhibition of the differentiation and functions of Treg cells. To date, 1-MT is undergoing clinical phase II and I trials for application in cancer immunotherapy.15 Intrauterine devices containing 1-MT can be considered for the treatment of endometriosis. These data indicate that endometriosis is more than a disease involving endocrine disorders, and also involves immunological factors. Moreover, it should be taken into consideration that assessment of the local immunological status of patients with endometriosis is essential for the treatment choice to be made.
Materials and Methods
Ethics statement
The human ethics committee of Gynecology and Obstetrics Hospital, Shanghai Medical School, Fudan University, People's Republic of China approved this study. Written informed consent was obtained from each patient enrolled.
Patients
Endometriotic tissues were acquired from premenopausal patients who had undergone laparoscopic surgery for the treatment of ovary endometriosis cysts (n=55; aged 22–46 years) diagnosed by pathological examination. Control endometrial tissues were obtained from premenopausal patients who had undergone panhysterectomy surgery for multiple myoma (n=15; aged 46–52 years). Peritoneal fluid was aspirated from the cul de sac at the beginning of the laparoscopic procedure under general anesthesia, obtained from patients with ovary endometriosis (n=16; aged 25–46 years), or patients who had laparoscopic surgery for uterine fibroid without evidence of endometriosis (n=12; aged 29–41 years) at the Obstetrics and Gynecology Hospital of Fudan University, between August 2014 and September 2016. Samples of peritoneal fluid contaminated by blood were excluded from the study. None of women had received hormonal therapy within 6 months prior to tissue collection. The stage of endometriosis was diagnosed according to the revised American Society for Reproductive Medicine staging (1997). Peripheral blood samples (5–15 ml) were obtained from healthy volunteers (n=35; aged 25–38 years old). All samples were collected under sterile conditions.
Peritoneal Treg detected by flow cytometry
All collected peritoneal fluid samples were centrifuged at 1600 rpm, at 4 °C for 9 min, then supernatant was discarded, and cells were fixed with 4% par formaldehyde (Sheng Gong, Shanghai, China) for 35 min, at 4 °C in the dark. Then cells were washed twice with phosphate-buffered saline (PBS; Hyclone, Logan, UT, USA). After centrifugation and removal of the supernatant, the fixed cells were resuspended in Foxp3 Perm Buffer (10 × , Biolegend, San Diego, USA) according to the manufacturer protocol. Finally, these cells were labeled with flow cytometry antibodies according to the manufacturer protocol and detected by flow cytometry.
ESCs isolation
Endometrial stromal cells (ESCs) were purified as previously described.37 Endometriotic tissues were cut into<1mm-thick sections and digested with Dulbecco's Modified Eagle Medium (DMEM)/F12 (Hyclone, Logan, UT, USA) containing collagenase type IV (0.1% Sigma, San Francisco, CA, USA) with constant agitation for 40 min at 37 °C. The resulting suspension was then filtered through 100- to 70-μm nylon strainers (Becton Dickinson, Franklin Lakes, NJ, USA). After the filtrate was centrifuged at 1600 r.p.m. for 9 min at 4 °C, the supernatant was discarded. Finally, ESCs were resuspended in DMEM/F-12 containing 10% fetal bovine serum (FBS; Hyclone) in the presence of 100 U/ml penicillin and 100 mg/ml streptomycin, and placed in culture flasks at 37 °C under 5% CO2. The culture medium was replaced with fresh medium every 3 days.
Generation of human naive CD4+ T cells and monocytes
Peripheral blood mononuclear cells (PBMCs) were isolated by Lymphoprep (Stemcell Technologies Inc., Vancouver, BC, Canada) density gradient centrifugation. CD14+ cells were obtained by positively selection using CD14+ cells micro-magnetic beads according to the manufacturer instructions (Miltenyi Biotec, Bergisch Gladbach, Germany). Naïve CD4+ T cells were obtained by negatively selection from the remaining immune cells according to the manufacturer instructions (Miltenyi Biotec). The purity of CD14+ cells was confirmed by flow cytometry, using PE/Cy7 anti-human CD14 monoclonal antibody (mAb) (Biolegend, San Diego, USA, clone: HCD14), was found to be>95%.
Cell co-culture systems
Ectopic ESCs were cultured in 24-well plates (Corning, Steuben County, NY, USA) at a density of 1 × 105 cells/well. The co-culture systems were established by incubating 2 × 105 monocytes with ESCs or alone, adding of estrogen (10−8 M; Sigma), 1-MT (0.05 mM; Sigma), or estrogen (10−8 M; Sigma) plus 1-MT (0.05 mM; Sigma). Meanwhile, naive CD4+ T cells were cultured in 24-well plates that coated with monoclonal anti-CD3 (5 μg/ml; Biolegend) and monoclonal anti-CD28 (1 μg/ml; Biolegend), in the presence of recombinant human IL-2 (50 ng/ml; Biolegend). The monocytes-derived macrophages and 2 × 105 naive CD4+ T cells were collected to co-culture with ESCs in 1 ml medium/well for 5 days (Supplementary Figure 1).
Treg cell suppression assay
Peripheral blood mononuclear cells (PBMCs) were isolated by Lymphoprep (Stemcell Technologies Inc.) density gradient centrifugation. CD4+CD25+ T cells were obtained by positively selection using CD4+CD25+ T cells (Regulatory T cells) micro-magnetic beads according to the manufacturer instructions (Miltenyi Biotec). CD4+CD25− cells were obtained by negatively selection according to the manufacturer instructions (Miltenyi Biotec). CD4+CD25+ T cells were cultured in 96-well plates that coated with monoclonal anti-human CD3 (5 μg/ml; Biolegend, clone: OKT3) and monoclonal anti-human CD28 (1 μg/ml; Biolegend, clone: CD28.2), in the presence of recombinant human IL-2 (50 ng/ml; Biolegend), while CD4+CD25− cells were cultured in 24-well plates that coated with monoclonal anti-CD3 (5 μg/ml; Biolegend) and monoclonal anti-CD28 (1 μg/ml; Biolegend). Treg cells were collected to culture with non-treated ESCs, 1-MT-pretreated ESCs, vector-pretreated ESCs, and MRC2-silenced ESCs or not for 48 h after Treg cells proliferation for two weeks. After that, Treg cells from these groups respectively cultured with paired CFSE-labeled CD4+CD25− T cells (Teff) for 48 h and then detected by flow cytometry.
Antibodies and flow cytometry
Fluorochrome-conjugated antibodies the following human antigens were used for flow cytometry analysis: Alexa Fluor 488 anti-human Foxp3 mAb (Biolegend, clone: 206D), PE anti-human LAP (TGF-β1) mAb (Biolegend, clone: TW4-6H10), PE/Cy7 anti-human CD4 mAb (Biolegend, clone: RPA-T4), APC anti-human IL-10 mAb (Biolegend, clone: JES3-9D7), PerCP/Cy5.5 anti-human CD73 (Ecto-5'-nucleotidase) mAb (Biolegend, clone: AD2), Brilliant Violet 421TM anti-human CD152 (CTLA-4) mAb (BD Biosciences, clone: BNI3), Brilliant Violet 510TM anti-human CD39 mAb (Biolegend, clone: A1), PE/Cy7 anti-human CD14 mAb (Biolegend, clone:HCD14), APC/Cy7 anti-human CD45 (Biolegend, clone: HI30), Alexa Fluor 488 anti-human Vimentin mAb (BD Biosciences, clone: RV202), PE anti-human Ki-67 mAb (Biolegend, clone: Ki-67), Anti-human Indoleamine2,3-dioxygenase/IDO APC-conjugated mAb (R&D System, Inc., Minneapolis, USA, clone: 700838), CFSE Cell Division Tracker Kit (Biolegend). Additionally, fluorochrome-conjugated antibodies against the following mouse antigens were used for flow cytometry analysis: anti-mouse CD4 fluorescein isothiocyanate (FITC) mAb (eBioscience, clone: GK1.5), FITC anti-mouse Vimentin mAb (BD Biosciences), anti-mouse/rat Foxp3 PE mAb (eBioscience, clone: FJK-16 s), anti-mouse/rat Ki-67 PE mAb (eBioscience, clone: P46013), PE/Cy7 anti-mouse IL-10 mAb (Biolegend, clone: JES5-16E3), PerCP/Cy5.5 anti-mouse CD73 mAb (Biolegend, clone: TY/11.8), APC anti-mouse LAP(TGF-β1) mAb (Biolegend, clone: TW7-16B4), Brilliant Violet 421TM anti-mouse CD152 mAb (Biolegend, clone: UC10-4B9), and anti-mouse IDO eFluor® 660 (eBioscience, clone: mIDO-48). Surface staining was performed at 4 °C for 30 min. Intracellular staining was performed using Foxp3 Fix/Perm Buffer Set (4 × , Biolegend) according to the manufacturer protocol. The resulting data were analyzed using the LYSYS II software program (Becton Dickson, Franklin Lakes, NJ, USA).
The effect of estrogen on the expression of IDO1 in the ectopic lesion
2 × 105 monocytes were co-cultured with ectopic ESCs on 24-well plates (Corning) or alone, adding estrogen (10−8 M; Sigma) or not. After 48 h, ESCs and monocytes-derived macrophages from different groups were collected, and then detected the expression of IDO1 by flow cytometry.
Treatment with estrogen receptor inhibitors in ESCs
Ectopic ESCs were cultured in 24-well plates (Corning) at a density of 1 × 105 cells/well. Pretreated ESCs with estrogen receptor α inhibitor (ERαi, 10−6 M; Sigma), estrogen receptor β inhibitor (ERβi, 10−6 M; Sigma), estrogen receptors inhibitor (ERi, 10−6 M; Sigma) or not. After 24 h, inhibitors were washed off by PBS (Ji-Nuo, Hangzhou, China), and then added estrogen (10−8 M; Sigma) to these groups except control group. After 48 h, the expression of IDO1 in ESCs from different groups was detected by flow cytometry.
Quantitative real-time polymerase chain reaction
Total RNA from ESCs treated with estrogen (10−8 M; Sigma), 1-MT (0.05 mM; Sigma), estrogen (10−8 M; Sigma) along with 1-MT (0.05 mM; Sigma), or knockdown of MRC2 with shRNA for 48 h was extracted using the Trizol reagent (Life Technologies, Carlsbad, CA, USA), according to the manufacturer instructions. Total RNA (2 μg) was reverse transcribed into first-stand cDNA (TaKaRa Bio Inc., Japan) following the manufacturer protocol, which was then used as a template for polymerase chain reaction (PCR) amplification. Real-time PCR was performed using ABI PRISMTM 7900 Sequence Detector (Applied Biosystems, Warrington, UK). The primers sequences used are listed in Table 1. PCRs was carried out for 40 cycles using the following conditions: denaturizing at 95 °C for 30 s, annealing at 95 °C for 5 s, and elongation at 60 °C for 34 s. The expression levels of the samples were expressed as arbitrary units defined by the 2−ΔΔCT method. All measurements were performed in triplicate. The specificity of the product was assessed by melting curve analysis.
Table 1. Characteristics of primers used for qRT-PCR.
| Gene | Orientation | Primer seq | Product size |
|---|---|---|---|
| MRC1 | FORWARD | GGAGGATTGTGTGGTGATGA | 104 |
| REVERSE | GACCTTGGCTTCGTGATTTC | ||
| MRC2 | FORWARD | TGGGAGAAGACCAAGTGACC | 117 |
| REVERSE | TGTAGATGAGGCTGCTGACG | ||
| CLEC4M | FORWARD | TGGATGGGACTTTCAGACCT | 114 |
| REVERSE | CCCGCTATTGTTGGGTTCT | ||
| PTAFR | FORWARD | CTGGAGTCTGGGATGGTAGC | 112 |
| REVERSE | TCAGCAGGAAATGACACAGC | ||
| GAPDH | FORWARD | AGAAGGCTGGGGCTCATTTG | 258 |
| REVERSE | AGGGGCCATCCACAGTCTTC |
Western blot
Protein expression level of MRC2 and β-actin were measured by western blot as previously described.38 The primary antibodies used were anti-MRC2 mAb (1:1000; Abcam Cambridge, MA, USA), and β-actin rabbit mAb (1:1000; Cell Signaling Technology, Inc., Danvers, MA, USA). Horseradish peroxidase-conjugated goat anti-rabbit IgG (1:2000; Arigobio, Taiwan, ROC) was used as the secondary antibody. Quantitative analysis of the relative density of the bands obtained by western blot was performed using Image J (W.S. Rasband, National Institutes of Health, Bethesda, MD, USA).
shRNA knockdown assay
Ectopic ESCs were cultured on 24-well plates with DMEM/F-12 (Hyclone) plus 10% FBS (Hyclone) in the presence of 100 U/ml penicillin and 100 mg/ml streptomycin, at 37 °C under 5% CO2. Lipofectamine 3000 (Invitrogen; Life Technologies, Carlsbad, CA, USA), OPTI-MEM (Gibco BRL, Gaithersburg, MD, USA), and the pGPU6/GFP/Neo-MRC2 short hairpin RNA (shRNA) (GenePharma, Shanghai, China) were used to transfected ESCs according to the manufacturer protocol.
Mice
We used seven-week-old female C57B/L6 mice (Slaccas Animal Laboratory, Shanghai, China) to establish an endometriosis-disease model as previously described.39, 40 Each donor mouse was administered 200 μl estrogen (10−8 M; Sigma) via the peritoneal cavity. One week later, donor mice were sacrificed and the uterine horn were collected and minced. Every two recipient mice were then intraperitoneally injected with minced uterine horn tissue from one donor mouse equally. All procedures were performed under aseptic conditions. One week after uterine tissue injection, mice were randomly divided into five groups and each received an intraperitoneal injection of 200 μl PBS (Ji-Nuo), estrogen receptors blockers (10−6 M; Sigma), 1-MT (0.05 mM; Sigma), vector (GenePharma, Shanghai, China), or mouse MRC2 shRNA (GenePharma, Shanghai, China) every week. Vector and mouse MRC2 shRNA were mixed with Lipofectamine 3000 (Invitrogen) and OPTI-MEM (Gibco BRL), according to the manufacturer protocol. Two weeks later, we measured the total number and weight of ectopic lesions, the expression of Ki-67 and IDO1 within ectopic lesions, the percentage and the expression of functional molecules of Treg in total ectopic lesions and peritoneal fluid of mice from groups administered PBS, estrogen receptor blockers, 1-MT, vector and MRC2 shRNA (Supplementary Figure 2).
Statistics
All studies were set up to include three wells per condition, and each experiment was independently repeated more than three times. Data collected from each independent experiment were analyzed using the Graphpad Prism (Graphpad software Inc., La Jolla, CA, USA) statistical package. Paired t-test of variance was performed when appropriate. Differences were considered statistically significant at ±S.D.<0.05.
Acknowledgments
We thank NH Jiang, YB Du, XX Hou and Y Zhang from Hospital and Institute of Obstetrics and Gynecology, Shanghai Medical School, Fudan University, and B Zhang from department of Obstetrics and Gynecology, the affiliated hospital of Jiangnan University for technical assistance; CY Wang from Bioinformatics Department, UCLA, XL Hu from School of Life Sciences, Tinghua University, and D Li from School of Life Science and Technology, Tongji University for bioinformatics analysis. This study supported by the National Basic Research Program of China (No. 2015CB943304 to XY Zhu), the National Natural Science Foundation of China (No. 81270677 and No. 81671457 to XY Zhu; No. 91542108 and No. 81471513 to MQ Li; No. 81601354 to J Mei), Shanghai Rising-Star Program (No. 16QA1400800 to MQ Li), the Development Fund of Shanghai Talents (No. 201557 to MQ Li), the Training Program for Young Talents of Shanghai Health System (No. XYQ2013104 to MQ Li) and the Program for Zhuoxue of Fudan University to MQ Li.
Author contributions
CY Wei conducted all experiments and prepared the figures and the manuscript. J Mei, LL Tang and YK Liu assisted with sample collection. DJ Li assisted with language editing. MQ Li and XY Zhu initiated and supervised the project and edited the manuscript.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on Cell Death and Disease website (http://www.nature.com/cddis)
Edited by M Herold
Supplementary Material
References
- Giudice LC, Evers JLH, Healy DL. Endometriosis: science and practice. In: Parazzini F, Vercellini P. Endometriosis: Epidemiology, and Etiological Factors, 1st edn. Blackwell Publishing Ltd: Malden, MA, USA, 2012, pp 19–26. [Google Scholar]
- Bulun SE. Endometriosis. N Engl J Med 2009; 360: 268–279. [DOI] [PubMed] [Google Scholar]
- Matsuzaki S, Canis M, Pouly J-L, Botchorishvili R, Déchelotte PJ, Mage G. Differential expression of genes in eutopic and ectopic endometrium from patients with ovarian endometriosis. Fertil Steril 2006; 86: 548–553. [DOI] [PubMed] [Google Scholar]
- Sun PR, Jia SZ, Lin H, Leng JH, Lang JH. Genome-wide profiling of long noncoding ribonucleic acid expression patterns in ovarian endometriosis by microarray. Fertil Steril 2014; 101: 1038–1046. [DOI] [PubMed] [Google Scholar]
- Puccetti P, Grohmann U. IDO and regulatory T cells: a role for reverse signaling and non-canonical NF-kappaB activation. Nat Rev Immunol 2007; 7: 817–823. [DOI] [PubMed] [Google Scholar]
- Mei J, Li MQ, Ding D, Li DJ, Jin LP. Indoleamine 2,3-dioxygenase-1 (IDO1) enhances survival and invasiveness of endometrial stromal cells via the activation of JNK signaling pathway. Int J Clin Exp Pathol 2013; 6: 431–444. [PMC free article] [PubMed] [Google Scholar]
- Huhtinen K, Ståhle M, Perheentupa A, Poutanen M. Estrogen biosynthesis and signaling in endometriosis. Mol Cell Endocrinol 2012; 358: 146–154. [DOI] [PubMed] [Google Scholar]
- Bulun SE, Zeitoun KM, Takayama K, Sasano H. Estrogen biosynthesis in endometriosis: molecular basis and clinical relevance. J Mol Endocrinol 2000; 25: 35–42. [DOI] [PubMed] [Google Scholar]
- Berbic M, Fraser IS. Regulatory T cells and other leukocytes in the pathogenesis of endometriosis. J Reprod Immunol 2011; 88: 149–155. [DOI] [PubMed] [Google Scholar]
- Cretney E, Xin A, Shi W, Minnich M, Masson F, Miasari M et al. The transcription factors Blimp-1 and IRF4 jointly control the differentiation and function of effector regulatory T cells. Nat Immunol 2011; 12: 304–311. [DOI] [PubMed] [Google Scholar]
- Liston A, Gray DHD. Homeostatic control of regulatory T cell diversity. Nat Rev Immunol 2014; 14: 154–165. [DOI] [PubMed] [Google Scholar]
- Podgaec S, Rizzo LV, Fernandes LFC, Baracat EC, Abrao MS. CD4(+) CD25(high) Foxp3(+) cells increased in the peritoneal fluid of patients with endometriosis. Am J Reprod Immunol 2012; 68: 301–308. [DOI] [PubMed] [Google Scholar]
- Polanczyk MJ, Hopke C, Huan J, Vandenbark AA, Offner H. Enhanced FoxP3 expression and Treg cell function in pregnant and estrogen-treated mice. J Neuroimmunol 2005; 170: 85–92. [DOI] [PubMed] [Google Scholar]
- Baban B, Chandler PR, Sharma MD, Pihkala J, Koni PA, Munn DH et al. IDO activates regulatory T cells and blocks their conversion into Th17-like T cells. J Immunol 2009; 183: 2475–2483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barth H, Raghuraman S. Persistent infectious diseases say - IDO. Role of indoleamine-2,3-dioxygenase in disease pathogenesis and implications for therapy. Crit Rev Microbiol 2014; 40: 360–368. [DOI] [PubMed] [Google Scholar]
- Madsen DH, Leonard D, Masedunskas A, Moyer A, Jürgensen HJ, Peters DE et al. M2-like macrophages are responsible for collagen degradation through a mannose receptor-mediated pathway. J Cell Biol 2013; 202: 951–966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wienke D, Davies GC, Johnson DA, Sturge J, Lambros MBK, Savage K et al. The collagen receptor Endo180 (CD280) is expressed on basal-like breast tumor cells and promotes tumor growth in vivo. Cancer Res 2007; 67: 10230–10240. [DOI] [PubMed] [Google Scholar]
- Gai X, Tu K, Lu Z, Zheng X. MRC2 expression correlates with TGFβ1 and survival in hepatocellular carcinoma. Int J Mol Sci 2014; 15: 15011–15025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelholm LH, Ingvarsen S, Jurgensen HJ, Hillig T. The collagen receptor uPARAP/Endo180. Front Biosci 2009; 14: 2103–2114. [DOI] [PubMed] [Google Scholar]
- Yuan Z, Wang L, Wang Y, Zhang T, Li L, Cragun JM et al. Tubal origin of ovarian endometriosis. Mod Pathol 2014; 27: 1154–1162. [DOI] [PubMed] [Google Scholar]
- Kráĺičková M, Vetvicka V. Immunological aspects of endometriosis: a review. Ann Transl Med 2015; 10: 2305–5839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan YY. Regulatory T cells: immune suppression and beyond. Cell Mol Immunol 2010; 7: 204–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olkowska-Truchanowicz J, Bocian K, Maksym RB, Bialoszewska A, Wlodarczyk D, Baranowski W et al. CD4+ CD25+ FOXP3+ regulatory T cells in peripheral blood and peritoneal fluid of patients with endometriosis. Hum Reprod 2012; 28: 119–124. [DOI] [PubMed] [Google Scholar]
- Söderlund J, Erhardt S, Kast RE. Acyclovir inhibition of IDO to decrease Tregs as a glioblastoma treatment adjunct. J Neuroinflamm 2009; 7: 44–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W, Liang X, Peterson AJ, Munn DH, Blazar BR. The Indoleamine 2,3-Dioxygenase pathway is essential for human plasmacytoid dendritic cell-induced adaptive t regulatory cell generation. J Immunol 2008; 181: 5396–5404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takikawa O, Kuroiwa T, Yamazaki F, Kido R. Mechanism of Interferon-γ Action. J Biol Chem 2001; 4: 2041–2048. [PubMed] [Google Scholar]
- Munn DH, Sharma MD, Baban B, Harding HP, Zhang Y, Ron D et al. GCN2 Kinase in T cells mediates proliferative arrest and anergy induction in response to Indoleamine 2,3-Dioxygenase. Immunity 2005; 22: 633–642. [DOI] [PubMed] [Google Scholar]
- López-Guisa JM, Cai X, Collins SJ, Yamaguchi I, Okamura DM, Bugge TH et al. Mannose receptor 2 attenuates renal fibrosis. J Am Soc Nephrol 2012; 23: 236–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Teja M, Gronau JH, Minamidate A, Darby S, Gaughan L, Robson C et al. Survival Outcome and EMT Suppression Mediated by a Lectin Domain Interaction of Endo180 and CD147. Mol Cancer Res 2015; 13: 538–547. [DOI] [PubMed] [Google Scholar]
- Shim JH, Shin DW, Noh MS, Lee TR. Reduced collagen internalization via down-regulation of MRC2 expression by UVA irradiation and its recovery by all-trans retinoic acid. J Dermatol Sci 2014; 73: 163–166. [DOI] [PubMed] [Google Scholar]
- Ahn SH, Monsanto SP, Miller C, Singh SS, Thomas R, Tayade C. Pathophysiology and Immune Dysfunction in Endometriosis. BioMed Res Int 2015; 10: 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fazleabas AT, Braundmeier A, Parkin K. Endometriosis-induced changes in regulatory T cells — insights towards developing permanent contraception. Contraception 2015; 92: 116–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friberg M, Jennings R, Alsarraj M, Dessureault S, Cantor A, Extermann M et al. Indoleamine 2,3-dioxygenase contributes to tumor cell evasion of T cell-mediated rejection. Int J Cancer 2002; 101: 151–155. [DOI] [PubMed] [Google Scholar]
- Wainwright DA, Dey M, Chang A, Lesniak MS. Targeting tregs in malignant brain cancer: overcoming IDO. Front Immunol 2013; 4: 116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Zhang H, Jia LQ, Sun H. Effects of Treg cells and IDO on human epithelial ovarian cancer cells under hypoxic conditions. Mol Med Rep 2014; 10: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young K, Fisher J, Kirkman M. Women's experiences of endometriosis: a systematic review and synthesis of qualitative research. J Fam Plann Reprod Health Care 2014; 0: 1–10. [DOI] [PubMed] [Google Scholar]
- Mei J, Jin L-P, Ding D, Li M-Q, Li D-J, Zhu X-Y. Inhibition of IDO1 suppresses cyclooxygenase-2 and matrix metalloproteinase-9 expression and decreases proliferation, adhesion and invasion of endometrial stromal cells. Mol Hum Reprod 2012; 18: 467–476. [DOI] [PubMed] [Google Scholar]
- Mei J, Zhu XY, Jin LP, Duan ZL, Li DJ, Li MQ. Estrogen promotes the survival of human secretory phase endometrial stromal cells via CXCL12/CXCR4 up-regulation-mediated autophagy inhibition. Hum Reprod 2015; 30: 1677–1689. [DOI] [PubMed] [Google Scholar]
- Ding D, Liu X, Duan J, Guo SW. Platelets are an unindicted culprit in the development of endometriosis: clinical and experimental evidence. Hum Reprod 2015; 30: 812–832. [DOI] [PubMed] [Google Scholar]
- Ruiz A, Rockfield S, Taran N, Haller E, Engelman RW, Flores I et al. Effect of hydroxychloroquine and characterization of autophagy in a mouse model of endometriosis. Cell Death Dis 2016; 7: e2059–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.