Abstract
Traumatic brain injury (TBI) is a major public health issue, with recent increased awareness of the potential long-term sequelae of repetitive injury. While TBI is common, objective diagnostic tools with sound neurobiological predictors of outcome are lacking. Indeed, such tools could help identify those at risk for more severe outcomes after repetitive injury and improve understanding of biological underpinnings to provide important mechanistic insights. We tested the hypothesis that acute and subacute pathological injury, including the microgliosis that results from repeated, mild closed head injury, is reflected in susceptibility-weighted magnetic resonance imaging (SWI) and diffusion-tensor imaging (DTI) microstructural abnormalities. Using a combination of high-resolution magnetic resonance imaging (MRI), stereology and qPCR, we studied the pathophysiology of male mice that sustained 7 consecutive mild traumatic brain injuries over 9 days in acute (24 hour) and subacute (1 week) time periods. Repetitive mild closed head injury induced focal cortical microhemorrhages and impaired axial diffusivity at one week post-injury. These microstructural abnormalities were associated with a significant increase in microglia. Notably, microgliosis was accompanied by a change in inflammatory microenvironment, defined by robust spatiotemporal alterations in tumor necrosis factor alpha (TNFα) receptor mRNA. Together, these data contribute novel insight on the fundamental biological processes associated with repeated mild brain injury concomitant with subacute imaging abnormalities in a clinically relevant animal model of repeated mild TBI. These findings suggest new diagnostic techniques can be used as biomarkers to guide the use of future protective or reparative interventions.
Keywords: DTI, Microhemorrhage, Inflammation, SWI, Axial Diffusion, RRID:AB_2314667, RRID:AB_323909, RRID:AB_2144905
Graphical Abstract

Using a combination of high-resolution magnetic resonance imaging (MRI), stereology and qPCR, we studied the pathophysiology of mice that sustained 7 consecutive mild traumatic brain injuries over 9 days. Repetitive mild closed head injury induced focal cortical microhemorrhages and impaired axial diffusivity at 1 week post-injury. These microstructural abnormalities were associated with a significant increase in microglia at 24h and 1 week post-injury.
INTRODUCTION
Every year in the United States, approximately 2.5 million people seek emergency medical treatment for a traumatic brain injury (TBI) (Faul and Coronado 2015). While a growing major public health issue (Marin et al. 2014), diagnostic tools and readily available neurobiological predictors of outcomes for TBI are lacking (Bruce et al. 2015). The need for objective assessment criteria is amplified by the increasing concern over the consequences of repetitive trauma, especially if a second injury is sustained before complete recovery. Prior studies have suggested a temporal window of vulnerability after TBI, but without empirical measures of injury, clinicians have limited tools to help guide critical management decisions aimed at preventing the sequelae of repetitive injury. Currently, computerized tomography (CT) scans and magnetic resonance imaging (MRI) with standard T1 and T2 sequences are the most frequently employed TBI diagnostic modalities, but the diagnostic and prognostic yield of these standard techniques is increasingly minor in the context of mild TBI. In order to stratify appropriate clinical resources and ensure the best treatment and follow-up care, objective diagnostic studies and biomarkers of injury must be identified and validated (Bruce et al. 2015). This is especially vital in the case of mild TBI, where clinical management is often based on subjective symptom reporting.
The first step to achieving objective diagnostic approaches is the development of appropriate preclinical models where pathophysiological mechanisms are similar to those observed in humans and can be studied in depth. Previously, we have shown graded levels of neuropathology in mice subjected to repetitive mild closed head injury (rmCHI) as a result of varying the frequency, quantity, and severity of injuries, with more frequent injuries resulting in long-term cognitive deficits (Kondo et al. 2015; Mannix et al. 2013; Meehan et al. 2012). Additionally, we demonstrated that mice subjected to rmCHI have impaired balance and spatial memory deficits that persist 3 months following injury concomitant with chronic astrocytosis and microgliosis (Mannix et al. 2014).
Glial activation and upregulation of inflammatory mediators such as tumor necrosis factor alpha (TNFα) have been directly linked to the pathophysiology and progression of TBI (Webster et al. 2015). Together with brain microstructure, regional and phenotypic glial activation data following repetitive mild TBI may yield clinically relevant biomarkers and predictors of outcome. Furthermore, detailed analysis of complete sets of diffusion metrics (mean diffusivity (MD), axial diffusivity (AD) and radial diffusivity (RD), fractional anisotropy (FA)) suggests that gliosis, rather than cytotoxic edema, is most consistent with changes in these parameters after acute, mild TBI in humans (Croall et al. 2014). Thus, we undertook the present investigation using a combination of high-resolution susceptibility-weighted (SWI) and diffusion tensor imaging (DTI), in tandem with a temporal and regional evaluation of microglia and common molecular mediators of a neuroinflammatory microenvironment. We tested the hypothesis that rmCHI in mice leads to significant diffusion abnormalities, concomitant with anomalous white and gray matter microstructure, microgliosis, and increased cytokine production both in the acute and sub-acute phases post-injury
MATERIALS & METHODS
All experiments were approved by the Institutional Animal Care and Use Committee at Boston Children’s Hospital and the University of New Mexico, and in compliance with the NIH Guide for the Care and Use of Laboratory Animals.
Experimental Design
Sample size estimates were developed a priori based on prior studies (Jantzie et al. 2014b; Mannix et al. 2014). Pilot studies of DTI in rodents following controlled cortical impact (CCI) using the same imaging sequences, MRI scanner, and software analysis revealed that significant differences in fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD) and radial diffusivity (RD) were detected with a sample size of 6–7 (Robinson et al. 2016). A sample size of 10 was used for the DTI studies because we hypothesized the effect size of microstructural changes after rmCHI would be smaller than those observed after CCI (Robinson et al. 2016). We previously detected significant differences in stereological estimates of microgliosis with sample size of 3–5 (Jantzie et al. 2014b; Mannix et al. 2014). Here, we used a sample size of 6–7 for stereological estimates of microglial immunolabeling. For PCR analyses, a sample of size of 5–8 was used based on prior studies (Jantzie et al. 2014b). Animals from at least two cohorts were used in all experiments.
rmCHI
Two-month-old male wild type C57BL/6 mice were randomized to undergo rmCHI or sham procedure, as previously described (Mannix et al. 2014; Mannix et al. 2013). Briefly, anesthesia of 4% isoflurane in a 70:30 mixture of nitrogen and oxygen was induced. Anesthesia exposure for each mouse was strictly controlled to 45 seconds. Mice were placed on a delicate task wiper (Kimwipe; Kimberly-Clark, Irving, TX) and grasped by the tail. The head was placed directly under a hollow, 28-inch tall guide tube. A 54 g metal bolt was used to deliver the impact to the dorsal aspect of the head. At impact, the mouse head readily penetrated the Kimwipe, documenting in a rotational acceleration of the head. All mice recovered in room air. Injured mice underwent 7 consecutive injuries over 9 days. Specifically, mice were injured daily for 5 days, followed by 2 days without injury, and then 2 additionally daily injuries consistent with prior reports (Mannix et al. 2014). Sham mice underwent anesthesia only, at the same frequency and duration as injured animals.
Magnetic Resonance Imaging (MRI)
One week following the 7th hit, mice were perfused with 4% paraformaldehyde (PFA). Brains were post-fixed in 4% PFA for 1 week and embedded in 2% agarose containing 3 mM sodium azide for ex vivo MR imaging. Scanning was performed on a Bruker 4.7T BioSpec 47/40 Ultra-Shielded Refrigerated nuclear MRI system equipped with a quadrature RF coil (72mm I.D.) and a small-bore (12cm I.D.) gradient set with a maximum gradient strength of 50 Gauss/cm. MR protocols consisted of multi-slice multi-echo (MSME) spin-echo and rapid acquisition with relaxation enhancement (RARE) sequences for T2, echo-planar diffusion tensor imaging (EP-DTI), and susceptibility-weighted imaging (SWI-FLASH). Images of twelve contiguous coronal 1mm slices were obtained with a 2.00 cm field of view, a 3000 ms TR and a 12 ms TE. EP-DTI sequences used a 3000 ms TR, a 40 ms TE, and a 2000 mm2/s b-value with 30 diffusion gradient directions. SWI parameters were a 700 ms TR, 40 ms TE, 40° flip angle with 10 averages, including a 256×256 acquisition matrix and 30 minute acquisition time. Brain regions of interest (ROIs), including major white matter tracts such as the corpus callosum, fimbria and capsular white matter, together with hippocampus and sensory cortex, were analyzed using Bruker’s Paravision 5.1 imaging software. Tractography, diffusion-weighted images, susceptibility weighted images, and fractional anisotropy (FA) maps were generated. Mean Diffusivity (MD), Axial (λ1, AD) and Radial Diffusivity ((λ2+λ3)/2, RD) eigenvectors were calculated. Two observers blinded to treatment conditions evaluated animals from both treatment groups, with sham animals included in analysis to control for potential perfusion or fixation artifacts occasionally associated with SWI performed ex vivo. The numbers of micro-hemorrhages were counted through 12 contiguous coronal slices.
Prussian Blue Staining
A series of representative 6 μm coronal, paraffin embedded brain sections (average of 7 sections per mouse) encompassing the anterior to posterior regions of the cortex, including the level of the dorsal hippocampus, were cut on a vibratome. Following de-paraffinization, sections were washed and staining in freshly prepared aqueous hydrochloric acid/potassium ferrocyanide solution per standard protocols (Kallakuri et al. 2015; Liu et al. 2014b). Sections were then rinsed, counter stained, and washed thoroughly to remove staining artifacts, dehydrated, cleared and coverslipped with organic mounting media.
Immunohistochemistry
Serial 20 μm coronal brain sections were cut on a Leica cryostat from the anterior frontal lobes through the posterior extent of the dorsal hippocampus using stereology protocols.(Jantzie et al. 2014b; Mannix et al. 2014) Sections were treated with 0.03% hydrogen peroxide, blocked with 10% normal goat serum and 0.1% tritonX-100 in phosphate-buffered saline (PBS), incubated with primary antibodies overnight at 4°C, rinsed, and incubated sequentially with appropriate biotinylated secondary antibodies (1:200, Vector, Burlingame, CA), Vectastain (Vector), and diaminobenzadine (DAB), and mounted with Permount. Primary antibodies used for microglia were Iba1 (ionized calcium-binding adapter molecule 1, Wako, 1:500, RRID:AB_2314667) and for activated microglia/macrophages were CD68 (cluster of differentiation 68, Serotec, Raleigh, NC, 1:100, RRID:AB_323909) (Table 1). For double labeling, Iba1 was stained as above, followed by antibodies against CD206 (Santa Cruz, 1:100, RRID:AB_2144905), and species appropriate fluorescent secondary antibodies (Table 1).
Table 1.
Primary Antibodies Used
| Antigen | Description of Immunogen | Source, Host Species, Catalog No. RRID | Concentration Used (IHC) |
|---|---|---|---|
| Iba1 | Synthetic peptide corresponding to C-terminus of ionized calcium-binding adapter molecule 1 | Wako, rabbit polyclonal, #019-19741, AB_2314667 | 1:500 |
| CD68 | Unconjugated antibody raised against CD68 of mouse origin, clone FA-11. Purified Concanavalin A acceptor glycoprotein from P815 cell line. | Serotec, rat monoclonal MCA1957XZ, AB_323909 | 1:100 |
| CD206 | Uncongugated antibody raised against CD206 of mouse origin, clone MR5D3. Purified Mrc1. | Santa Cruz, rat monoclonal, sc-58987, AB_2144905 | 1:100 |
Stereological Estimates
All sections were coded prior to blinding. The observer was blinded to injury group. A Leica microscope with a motorized stage and electronic microcator was used with Stereologer software (Stereology Resource Center, St. Petersburg, FL) to perform the analyses. Object area fraction and volume probes were used to quantify the amount of Iba1-positive immunolabeling of microglia, and CD68-positive immunolabeling of microglia/macrophages (n=6–7/group) (Jantzie et al. 2014b). Estimates of immunolabeling of each antigen were obtained using a thin section modification of the optical fractionator method (Gundersen et al. 1988; Mouton 2014). Regions of interest (ROI) at 24h and 1 week following injury were defined in the hippocampus and fimbria using anterior, posterior, medial, and lateral anatomical landmarks and the amount of immunolabeling within each systematically-spaced, unbiased sampling frame was estimated. Coefficients of error were calculated and all were <0.10. At the completion of the stereological analyses, samples were decoded, and the means and SEM of the immunolabeling were calculated.
Enzyme-linked Immunosorbent Assay (ELISA)
Quantitative determination of mouse cortical and hippocampal TNF-α levels at 24 h and 1 week post-injury (n=6–8/group/time point) was performed using a Quantikine sandwich ELISA per manufacturer’s specifications (R&D Systems, Minneapolis, MN)
Quantitative PCR
Hippocampus and cortex from mice at 24 h and 1 week post-injury (n=5–8/group/time point) were rapidly dissected for RNA isolation (RNeasy mini kit, Qiagen, Germantown, MD). Genes investigated represented pro- and anti-inflammatory ligands and receptors including CD86 (cluster of differentiation), iNOS (inducible nitric oxide synthase), CD206, TNFα (tumor necrosis factor-α), IL-1β (interleukin 1β), IL-1βR, TNFR1 and TNFR2. Primer sequences were verified with the NCBI (National Center for Biotechnology Information) nucleotide Basic Local Alignment Search Tool (BLAST) and are listed in Table 2. Gene-of-interest primers and cDNA synthesized from 0.9 μg RNA were added to Power Sybr green MasterMix (Life Technologies, Grand Island, NY) or Taqman probes (Invitrogen, Grand Island, NY), and run in triplicate on a Step-One Plus qPCR instrument (Life Technologies)(Jantzie et al. 2014b). To standardize transcripts between experiments, cycle thresholds (Ct) from samples of interest were compared to Ct values from pooled cortical or hippocampal samples from naïve mice, with gene-of-interest transcription normalized to an 18s ribosomal endogenous control consistent with standard qPCR methods (Jantzie et al. 2014a; Jantzie et al. 2014c). Experimental replicates that varied by more than 0.25 Ct values were excluded from all experiments.
TABLE 2.
PRIMER SEQUENCES
| Name | Forward | Reverse |
|---|---|---|
| IL1β | CGC AGC AGC ACA TCA ACA AGA GC | TGT CCT CAT CCT GGA AGG TCC ACG |
| IL-1βR | ACC CCC ATA TCA GCG GAC CG | TTG CTT CCC CCG GAA CGT AT |
| TNFα | CGG GGT GAT CGG TCC CCA AAG | GGA GGG CGT TGG CGC GCT GG |
| TNFR1 | CTC AGG TAC TGC GGT GCT GTT | TCG GCA CAT TAA ACT GAT GAA GAT |
| TNFR2 | GGA ACC TGG GTA CGA GTG CCA | GCG GAT CTC CAC CTG GTC AGT |
| iNOS | CCC TTC AAT GGT TGG TAC ATG | ACA TTG ATC TCC GTG ACA GCC |
| CD86 | GAG CGG GAT AGT AAC GCT GA | GGC TCT CAC TGC CTT CAC TC |
| CD206 | CTT CGG GCC TTT GGA ATA AT | TAG AAG AGC CCT TGG GTT GA |
Statistical Analysis
Observers blinded to injury status performed quantification of MRI and immunolabeling. Differences between two groups were compared with Student’s t test or Mann Whitney U-test, and differences between more than two groups were compared with two-way ANOVA with Bonferonni correction. Data are represented mean ± SEM with p<0.05 considered significant.
RESULTS
rmCHI induces focal cortical micro-hemorrhage and impairs axial diffusion
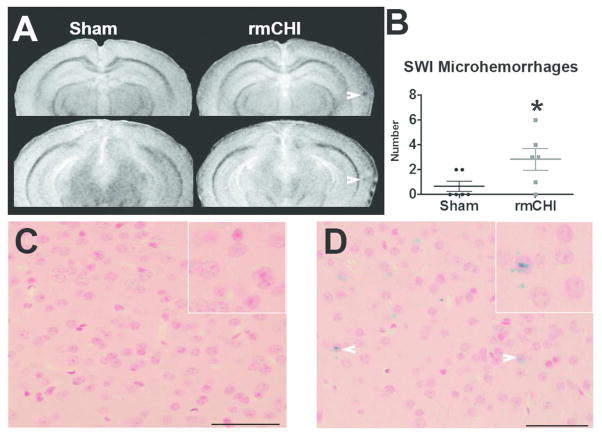
No animal had prolonged (>90s) loss of consciousness or any seizure activity following CHI. No focal lesions on standard T2-weighted images were observed for any animal (data not shown). Accordingly, as SWI exploits the distinct magnetic-susceptibility effects of extracellular and extravascular blood products in the brain, we used a susceptibility-weighted sequence to evaluate miniscule hemorrhages. Microhemorrhages were defined a-priori as foci with increased SWI signal not associated with bone, vessel or MRI artifact, consistent with clinical literature (Goos et al. 2011; Liu et al. 2014a; Liu et al. 2016; Mori et al. 2014). In injured animals, the median numbers of focal hypointensitites were 3, and as expected, there were significantly more throughout the brain, but primarily diffusely throughout the cortical mantle, in rmCHI animals compared to sham (Fig. 1, median sham animals=0, n=6, Mann Whitney U-test, p=0.045). Presence of microhemorrhage was confirmed histologically by evaluating Prussian blue stained sections (Fig. 1). Compared to sham (Fig. 1C), which had no Prussian blue reaction products, rmCHI animals had focal microhemorrhages identified by blue deposits throughout the neuropil consistent with previous reports (Kallakuri et al. 2015). Prussian blue streaks were observed diffusely primarily throughout the cortex, with additional presence detected in the subcortical structures of the hippocampus and thalamus. These SWI and histological data are consistent with diffuse microhemorrhage detected throughout the brain one week following rmCHI.
Figure 1.
(b&w). Susceptibility-weighted imaging (SWI) 1 week after rmCHI reveals micro-hemorrhages. A. Coronal SWI shows more hypodensities consistent with micro-hemorrhage 1 week after rmCHI (arrows). B. More micro-hemorrhages per brain were observed following rmCHI (n=6, Mann-Whitney U test, p<0.05). C. Representative photomicrograph shown sham animals are negative for Prussian blue in the cortex. D. Representative photomicrograph shown Prussian blue reaction products (arrows) in cortical neuropil 1 week following rmCHI. (Scale bar = 100μm)
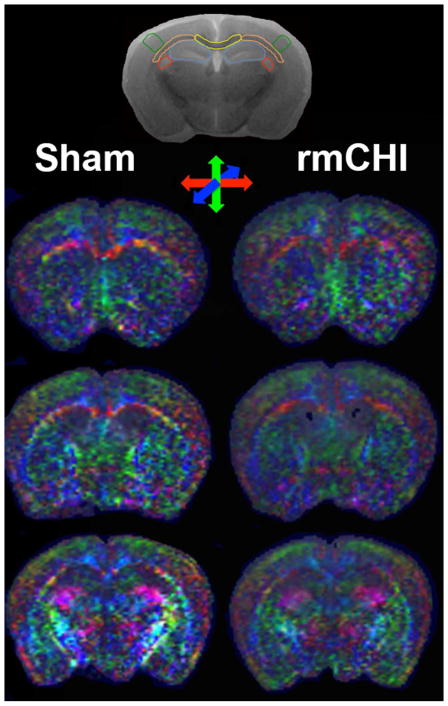
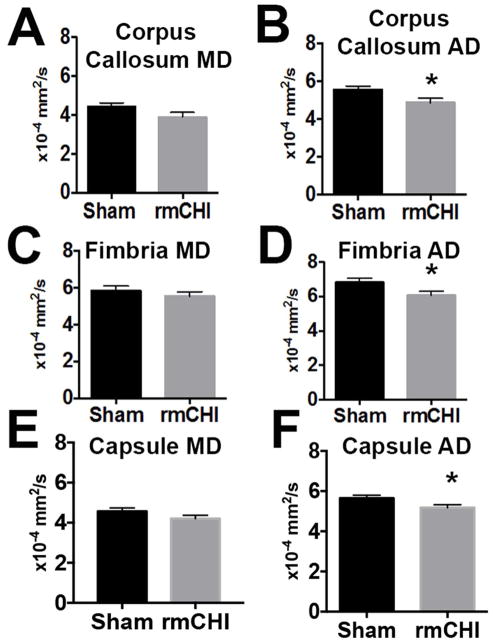
To investigate microstructural abnormalities in detail, we performed a thorough ex vivo multi-regional, multi-directional DTI analysis, including FA, MD, AD and RD in the corpus callosum, fimbria, sensory cortex, and hippocampus at 1 week post-rmCHI (Fig. 2, 3 and Table 3). Despite finding that there were no significant differences in MD in either the corpus callosum or fimbria, a reduced AD (λ1) was observed in the white matter 1 week following rmCHI (Fig. 2). Specifically, rmCHI animals had reduced AD at 1 week in the corpus callosum compared to sham animals (Fig. 3, all n=10, two-tailed Student’s t-test, p=0.03). Similarly, AD was lower in the fimbria (n=10, two-tailed Student’s t-test, p = 0.04) and in the capsular white matter (n=10, two tailed Student’s t-test, p=0.03) following rmCHI, suggesting that the injury to the white matter was widespread (Fig. 3). No change in AD was apparent in the gray matter structures of the sensory cortex and hippocampus (Table 3). Additionally, no differences in FA or RD were noted in any brain regions examined (Table 3). Together, these data suggest significantly abnormal diffusion parallel to axons following rmCHI, and are consistent with subacute white matter microstructural abnormalities.
Figure 2.
(color) Diffusion maps illustrate the directionality of diffusion with colors: red – transverse, green – vertical and blue – orthogonal to the plane. After rmCHI, subtle loss of color and directionality is evident in multiple regions, including white matter, consistent with microstructural injury and abnormal diffusion. Manual drawings of regions of interest on representative T2 image in the coronal view demarcating the corpus callosum (yellow), capsular white matter (orange), fimbria (red), hippocampus (blue) and sensory cortex (green).
Figure 3.
(b&w) Mean diffusivity (MD) quantifies the mean of the three eigenvectors, and axial diffusivity (AD) quantifies changes in the primary eigenvector. A. One week after rmCHI, no change in MD is present in white matter corpus callosum. B. Significant reduction of AD is observed one week after rmCHI in white matter corpus callosum. C. Similarly, no change in fimbria MD is evident one week after rmCHI. D. Reduced AD is also present in the fimbria one week after rmCHI. E. Consistent with the other white matter regions, MD is unchanged in the capsular white matter after rmCHI. F. AD is significantly reduced in capsular white matter 1 week following rmCHI (n=10, Student’s two-tailed t test, *p<0.05).
Table 3.
Diffusion Tensor Imaging (DTI) Measures
|
|
|||
|---|---|---|---|
| Fractional Anisotropy (FA) | Axial Diffusion (AD) | Radial Diffusion (RD) | |
|
|
|||
| Corpus Callosum | 0.269±0.016 vs. 0.268±0.020, p=0.3 | 5.6±0.2 vs. 4.9±0.2 ×10−4 mm2/s, *p=0.03 | 3.6±0.3 vs. 3.5±0.2 ×10−4mm2/s, p=0.8 |
|
|
|||
| Fimbria | 0.172±0.014 vs. 0.177±0.011, p=0.8 | 6.8±0.2 vs. 6.1±0.2 ×10−4 mm2/s, *p=0.03 | 5.4±0.3 vs. 4.9±0.3 ×10−4mm2/s, p=0.2 |
|
|
|||
| Capsular White Matter | 0.246±0.018 vs. 0.243±0.015, p=0.2 | 5.7±0.1 vs. 5.2±0.1 ×10−4 mm2/s, *p=0.04 | 4.1±0.2 vs. 3.7±0.2 ×10−4mm2/s, p=0.2 |
|
|
|||
| Hippocampus | 0.149±0.011 vs. 0.015±0.013, p=0.1 | 6.5±.0.2 vs. 6.2±0.2 ×10−4mm2/s, p=0.2 | 5.3±0.2 vs. 5.2±0.2 ×10−4mm2/s, p=0.4 |
|
|
|||
| Sensory Cortex | 0.156±0.011 vs. 0.160±0.009, p=0.8 | 5.7±0.1 vs. 5.6±0.1 ×10−4 mm2/s, p=0.3 | 4.6±0.1 vs. 4.7±0.4 ×10−4mm2/s, p=0.7 |
|
|
|||
rmCHI induces acute and chronic regional microgliosis
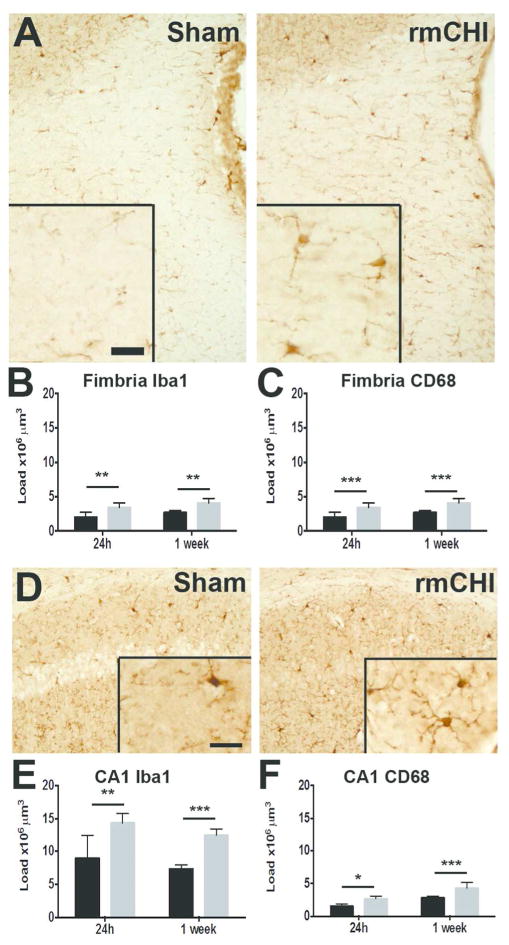
To determine if a microglial reaction occurred along with the microstructural changes on DTI, we estimated the amount of Iba1-immunolabeled microglia and CD68-immunolabeled microglia/macrophages using stereology at 24h and 1 week post-injury in both white and gray matter (Fig. 4). Twenty-four hours after the final hit, Iba-positive immunolabeling increased 1.7-fold in fimbria white matter (n=6–7, two-way ANOVA, p<0.01, Fig. 4B). This increase was maintained 1 week post-injury (n=6–7, two-way ANOVA, p<0.01, Fig. 4B). The Iba1-positive immunlabeling was accompanied by parallel increases in CD68-positive activated microglia/macrophages at both time points (n=6–7, two-way ANOVA, both p<0.001, Fig. 4C). These results show rmCHI causes an early white matter microgliosis that is sustained into the subacute period.
Figure 4.
(b&w) Immunolabeling of Iba1+ microglia reveals microgliosis following rmCHI. A. More Iba1+ immunolabeling of microglia is present in the fimbria one week after rmCHI. Bar = 20 μm. B. Stereological estimates of Iba1+ immunolabeling reveal more microglial labeling is present in the fimbria at both early and subacute intervals after rmCHI. C. More CD68+ activated microglia/macrophage immunolabeling is also more prevalent in the fimbria at both acute and subacute intervals following rmCHI. D. In the hippocampal CA1 subfield, more Iba1+ immunolabeling is present one week after rmCHI. E. Stereological estimates confirm markedly more Iba+1 immunolabeled microglia are present in the CA1 one week after rmCHI. F. More CD68-immunolabeled activated microglia/macrophages are also present in CA1 one week after the rmCHI. (n=6–7, two way ANOVA, *p < 0.05, **p < 0.01, ***p ≤ 0.001).
Similar to the pattern observed in the white matter, more immunolabeled microglia were present in gray matter at both 24h and 1 week after rmCHI (Fig. 4D). Interestingly, regional analysis revealed that the baseline population of Iba1-immunolabeled microglia was approximately 4-fold greater in the hippocampal CA1 subfield compared to the nearby fimbria. Similar to the severity and time course of microgliosis noted after injury in the white matter, Iba1-immunolabeling in rmCHI CA1 was elevated 1.6-fold compared to sham animals at both 24h and 1 week (two-way ANOVA, both p<0.001, Fig. 4E). Changes in CA1 subfield CD68-immunolabeling mirrored the increases in Iba1-postive labeling at both 24h and 1 week (two-way ANOVA, both p<0.001, Fig. 4F), consistent with increased microglial/macrophage activation following rmCHI. Thus, rmCHI induces sustained microgliosis in both gray and white matter that persists into the subacute period.
Inflammatory microenvironment changes occur following rmCHI
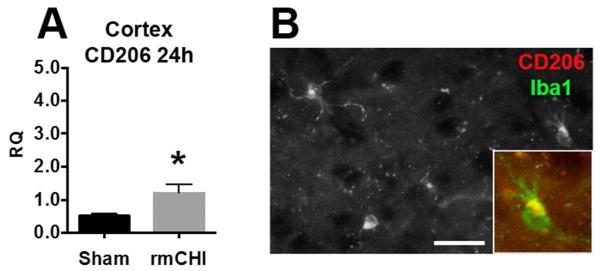
Given the microgliosis following rmCHI suggested an robust inflammatory phenotype, and inflammation is known to promote secondary injury after TBI (Hinson et al. 2015), we investigated molecules implicated in immune cell polarity, putative inflammatory signal transduction, and inflammatory microenvironment at discrete time points following injury. First, we assessed mRNA levels of CD86, iNOS, IL-1β and CD206, genes representing the extremes of classical pro-inflammatory and anti-inflammatory immune cell polarity/microenvironment, respectively (Wu et al. 2014). Interestingly, no changes in CD86, iNOS or IL-1βR mRNA levels were detected in either cortex or hippocampus at both the 24 h and 1 week time points (data not shown). However, CD206 mRNA, was increased in the cortex of rmCHI animals at 24 h (n=5–8, two-tailed Student’s t test, p=0.03, Fig. 5A), and observed on Iba1-positive microglia following rmCHI (Fig. 5B). No difference in CD206 levels was present at 1 week (data not shown).
Figure 5.
(color) Early and subacute alterations in inflammatory gene expression following rmCHI in cortex and hippocampus. A. Cortical anti-inflammatory marker CD206 mRNA levels are elevated at 24 hours after rmCHI compared to sham B. CD206 (red, punctate) is primarily colocalized (yellow) with Iba1-positive microglia (green, cell bodies) 24h following rmCHI. (Scale bar = 20μm)
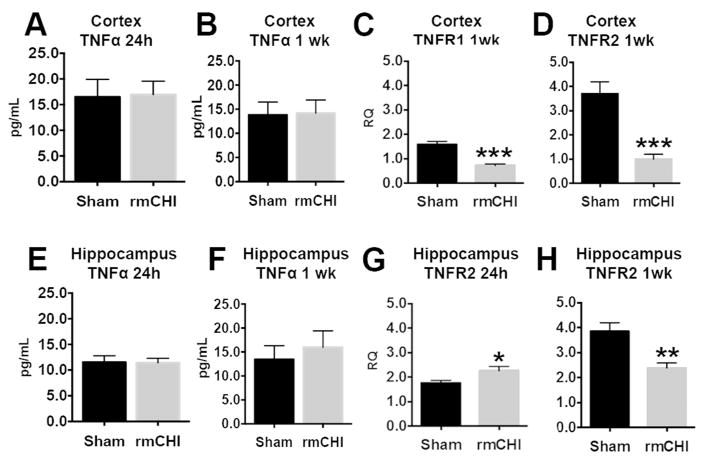
To further expand our assessment of putative inflammatory mediators, we complimented the above qPCR data by assessing regional changes in TNFα following rmCHI via ELISA and qPCR at 24 h and 1 week following injury (Figure 6). Interestingly, we found no statistically significant changes in TNFα protein levels in either region, at either time point (Figure 6A,B,E,F). Given cytokine signaling is highly dependent on function of cognate receptors and ultimately determines the nature of signal transduction, we measured TNFR mRNA to begin understand the temporal nature of TNFR expression (Erickson and Banks 2011). Interestingly, alterations in TNF-α inflammatory signaling pathway mRNA were detected in the cortex and hippocampus following rmCHI. Specifically, in the cortex at 24 hours after injury, no changes in cortical TNFR1 or TNFR2 mRNA were observed (data not shown). One week after injury, however, both cortical TNFR1 and TNFR2 mRNA levels were reduced in rmCHI cortex compared to shams (two-tailed Student’s t test, both p<0.001, Fig. 6C,D), consistent with altered cortical TNFα signal transduction 1 week following injury (Naude et al. 2011). However, a different pattern of changes in TNFR mRNA was observed in the hippocampus. No differences in hippocampal TNFR1 mRNA levels were observed at either 24 hours or 1 week (data not shown), but, TNFR2 mRNA levels were increased in rmCHI hippocampi at 24h compared to sham (two-tailed Student’s t test, p=0.03, Fig 6G). At 1 week, this TNFR2 pattern reversed and became similar to the cortex, with the hippocampus showing reduced TNFR2 mRNA levels after rmCHI compared to sham (two-tailed Student’s t test, p=0.004, Fig. 6H).
Figure 6.
(b&w) A. TNFα protein expression is unchanged in the cortex 24 h after rmCHI. B. Similarly, TNFα levels are consistent across groups at 1 week. C. However, loss of TNFR1 mRNA occurs 1 week after rmCHI in the cortex. D. Similarly, cortical TNFR2 expression is also decreased at 1 week following rmCHI. E. Consistent with the cortex, hippocampal TNFα levels are unchanged at 24 h and F. 1 week following rmCHI. G. However, TNFR2 mRNA expression is significantly increased in the hippocampus at 24h. H. This pattern reverses at 1 week with TNFR2 mRNA expression significantly decreased similar to the cortex at this subacute interval. (n=5–8, Student’s two-tailed t test, *p < 0.05, **p < 0.01, ***p ≤ 0.001)
DISCUSSION
Traumatic brain injury is a critical public health issue affecting over 2.5 million people worldwide (Budde et al. 2011; Cox et al. 2011). Recovery is often patient specific, and can be limited given the lack of effective treatment strategies (Budde et al. 2011). With these clinical issues in mind, we developed a mouse model to study the functional outcome and pathophysiology of rmTBI (Mannix et al. 2014; Mannix et al. 2013). Previously, we reported that 7 hits in 9 days, as used in the present study, results in loss of consciousness on days 1 and 2, concomitant with acute, subacute and chronic deficits in spatial memory, as well as hyperactivity and compromised motor performance (Mannix et al. 2014; Mannix et al. 2013). Collectively, these data offer a behavioral phenotype that has correlates in the clinical entity of concussion and provide a translational platform for mechanistic studies. Having established that this model produces a sustained functional deficit, we extend these investigations here by demonstrating that rmCHI yields subacute cortical microhemorrhage and reduced axial diffusivity at 1 week, together with increased microgliosis, and regional and temporal dysregulation of TNFR1 and TNFR2 gene expression.
Recently, sensitive imaging techniques have been increasingly utilized both experimentally and clinically to assess TBI. These techniques incorporate multi-modal MRI information on network functionality, microstructural integrity, tissue perfusion, hemorrhage and iron deposition, inflammation, edema and regional volume loss (Benson et al. 2012; Kallakuri et al. 2015; Mannix et al. 2013). Consistent with this concept, we performed a sophisticated multi-modal MRI investigation in mice 1-week post rmTBI using DTI and SWI. We assessed FA, MD, AD, and RD in our mice to examine specific, direction and orientation-dependent variation in water diffusivity (Mukherjee and McKinstry 2006; Mukherjee et al. 2001; Owen et al. 2013). We found significantly decreased AD in multiple regions, including the corpus callosum, fimbria and capsular white matter, consistent with impaired diffusion parallel to axons and microstructural aberrations. Indeed, the mechanical forces of rmTBI may result in stretching and disruption of subcellular and extracellular compartments resulting in the dysregulated diffusion observed here (Browne et al. 2011; Mayer et al. 2012; Povlishock and Katz 2005). Previously, we reported that H&E and SMI31-immunostained sections failed to reveal significant numbers of spheroids in major white matter tracts in injured mice 6 months after rmCHI (Mannix et al. 2013). However, the MRI analysis of white matter diffusivity encompassing directional diffusion measures, including AD and RD, performed here provides thorough microstructural information on axonal status in the subacute phase (Budde et al. 2009; Fox et al. 2008; Mac Donald et al. 2007; Mukherjee and McKinstry 2006; Mukherjee et al. 2001; Owen et al. 2013). Indeed, neurofilaments can be damaged without physical beading, with altered transport processes along the axon and at the cellular level. Typically, this injury manifests as increased membrane permeability, resulting in a decrease in the diffusion metric parallel to axonal plane (AD) as shown here (Mac Donald et al. 2007). Importantly, our data is consistent with and supportive of compelling clinical literature that includes athletes and patients with poor neuropsychological outcome who show similar AD changes in the corpus callosum and subcortical white matter, (Benson et al. 2012; Kumar et al. 2009; Lancaster et al. 2016; Mac Donald et al. 2007; Pasternak et al. 2014).
The utility of multi-modal MRI after rmTBI is best supported by its sensitivity to multiple pathophysiological processes and markers missed by conventional MRI, without requiring neuropathological assessment. For example, SWI exploits the magnetic susceptibility differences in blood, iron and calcium. Here, we report diffuse cortical microhemorrhages on SWI 1 week post-rmCHI, confirmed histopathologically with positive Prussian blue reaction products in the cortical neuropil of injured animals. Clinically, the number, localization, type and magnitude of microhemorrhage are associated with severity of injury and prognostication of functional outcome at 6–12 months (Liu et al. 2014a; Liu et al. 2016; Toth et al. 2016). The number and extent of microbleeds is dynamic however (Toth et al. 2016), emphasizing the importance of longitudinal and non-invasive SWI assessment of hemorrhage concomitant with clinical signs. With respect to our data, Prussian blue detects hemosiderin deposits at a cellular level in sections 4–6 microns thin, allowing for much greater resolution of reactive blood products. However, direct numerical comparisons between pathological and neuroradiological microbleeds must be made cautiously, as it is difficult to compare Prussian blue reaction products as observed under the microscope directly to the paramagnetic product of heme degradation resulting in homogenous signal loss on MRI (Greenberg et al. 2009; Toth et al. 2016). Indeed, bleeds are often smaller and more numerous than they appear on SWI (Greenberg et al. 2009; Kallakuri et al. 2015; Liu et al. 2014a; Liu et al. 2016; Liu et al. 2014b; Toth et al. 2016).
Undoubtedly, the cellular composition of and contribution to diffusivity is multifaceted, and the contribution of glia to pathophysiological processes following TBI cannot be underestimated (Budde et al. 2011). Neuroinflammation, including microglia and macrophage proliferation, is a robust response to TBI, and likely also plays a role in changes in diffusivity seen in both humans and animals following injury (Budde et al. 2011). Thus, we investigated microglia in the first week following rmCHI, temporally consistent with our imaging investigations. Here, we show significant increases in Iba1 and CD68 immunolabeling in major white and gray matter structures following rmCHI beginning 24 hours after injury and persisting through the first injury week. These data are consistent with our prior data showing microgliosis and astrocytosis 3 months following rmCHI (Mannix et al. 2014; Mannix et al. 2013).
Just as in the peripheral immune system, CNS microglia and macrophages are highly plastic and assume diverse phenotypes in response to specific changes in the local microenvironment. Indeed, a number of genes are associated with different microglial activation states and disease specific molecular inflammatory signatures (Butovsky et al. 2014; Chiu et al. 2013; Hickman et al. 2013). Therefore, after establishing increases in gray and white matter microglial load with corresponding increases in activation state (increased CD68 immunoreactivity), we performed qPCR in discrete brain regions for genes classically associated with pro-inflammatory states (iNOS, CD86, TNFα, and IL-1β) and anti-inflammatory microenvironments (CD206, TNFR2). We consistently observed changes in TNFR gene expression in the absence of overt injury induced alterations in TNFα protein itself, defined by significant decreases in both TNFR1 and TNFR2 mRNA expression in the cortex 1 week following injury. Additionally, we observed upregulated TNFR2 mRNA in the hippocampus of rmCHI mice at 24h, which was subsequently downregulated after 1 week. Importantly, cytokine signaling is highly dependent on the function of cognate receptors, mRNA expression, receptor cross-talk signaling kinetics, and pro-and anti-inflammatory signals in the microenvironment (Erickson and Banks 2011; Naude et al. 2011; Opal and DePalo 2000). Thus, measuring numerous cytokines together with their receptors provides a snapshot of their influence on physiology and captures the temporal nature of their expression (Erickson and Banks 2011). TNFα is one of the prominent mediators following TBI, driving the activation, proliferation and hypertrophy of mononuclear and phagocytic cells, including microglial and astrocytes. Other investigators have shown that increased TNFα signaling through TNFR1 exacerbates functional deficits following TBI (Longhi et al. 2013), while signaling through TNFR2 is neuroprotective (Yang et al. 2010). Indeed, TNFR2 deletion impairs novel object recognition, spatial memory recognition, contextual fear conditioning, motor performance, and can increase anxiety-like behavior in young adult mice (Naude et al. 2014), a behavioral phenotype consistent with our findings in this model of rmCHI. We also found CD206 mRNA acutely increased in the cortex following rmCHI, a molecule previously documented to be central to microglial response following TBI (Wang et al. 2013). Taken together with increased Iba1 and CD68 immunolabeling, our results indicate that microglia/macrophages respond dynamically to rmCHI and likely influence overall pro- and anti-inflammatory signal transduction driven primarily through cytokine and chemokine receptors (i.e. TNFR1 and TNFR2) following rmTBI and may contribute to changes in diffusion consistent with prior reports (Budde et al. 2011).
The limitations of the current study are that the MRI was performed ex vivo, and we are unable to provide in vivo evidence for the temporal relationship and longitudinal evolution of diffusion changes and microhemorrhage in this model. Future studies aimed at uncovering the real time evolution of diffusion metrics and structural coherence in this model are warranted through acute, subacute and long-term recovery phases. Similarly, traumatic microbleeds are not static, and change in number and extent. Thus, imaging at multiple time points following injury is important and relevant for optimizing the clinical prognostic utility of this biomarker and detecting the full burden of hemorrhagic and non-hemorrhagic diffuse axonal injury after repeated insult. Indeed, visibility of microbleeds on SWI attenuates longitudinally due to hemosiderin absorption, a similar mechanisms that has limited Prussian blue staining to detecting chronic hemorrhages via histology (Liu et al. 2014a; Liu et al. 2016; Liu et al. 2014b; Toth et al. 2016). Furthermore, rmCHI as described herein produces loss of consciousness in the initial injury days, which is not common in sports-related concussions. Repetition of injury is a critical variable for preclinical modeling and assessment of the cumulative effect of rmTBI on cognitive function and molecular pathophysiology, and the importance of dose response to this end cannot be underemphasized, considering the diverse mechanisms and severity of human rmTBI. Finally, the qPCR data from cortical and hippocampal samples shown here represent expression on total CNS cells, including neurons and infiltrating immune cells, and are indicative of regional changes in mRNA in the cerebral microenvironment, not cell specific changes in mRNA expression. Future studies in knockout mice (i.e. TNFR1 or TNFR2) and expanded longitudinal investigations of multiple cytokines in in wild-type animals are needed to provide conclusive evidence for the contribution of microglia and their polarization following rmCHI, including detailed analysis of protein expression and function.
In conclusion, rmCHI induced focal cortical microhemorrhages and impaired axial diffusivity at one week post-injury, significant increases in Iba1 and CD68-positive microglia/macrophages throughout vulnerable gray and white matter regions. Notably, microgliosis was accompanied by a change in inflammatory microenvironment, defined by robust spatiotemporal alterations in tumor necrosis factor alpha (TNFα) receptor mRNA. Together, these data contribute relavent neuropathological substrates of repeated mild brain injury and support new diagnostic techniques that can be used as biomarkers to guide the use of future protective or reparative interventions.
SIGNIFICANCE STATEMENT.
Understanding the pathophysiologic mechanisms of repetitive mild traumatic brain injury and the subsequent deficits is necessary for accurate diagnosis, prognosis, and assessment of treatment. In this preclinical investigation, we show repetitive mild closed head injuries induce focal cortical microhemorrhages and impair axial diffusion. These microstructural abnormalities are concomitant with significant increases in microglial load. Together, these data emphasize underpinnings of repeated mild brain injury that are consistent with subacute high-resolution diffusion tensor imaging, and reveal multiple avenues for future mechanistic investigations.
Acknowledgments
Support and Grant Information: This research was supported by funds from the P30 Centers for Biomedical Research Excellence Pilot Award to LLJ (Jantzie/CoBRE P30GM103400/Pi:Liu) and the National Hockey League Alumni Association through the Corey C. Griffin Pro-Am Tournament.
The authors are very grateful to Lindsay Chan, Sasha Alcon, and Jumana Hashim for technical assistance, for the funding provided by the P30 Centers for Biomedical Research Excellence Pilot Award to LLJ (Jantzie/CoBRE P30GM103400/Pi:Liu), and the philanthropic support from the National Hockey League Alumni Association through the Corey C. Griffin Pro-Am Tournament.
Footnotes
Conflict of Interest Statement
The authors declare no conflicts of interest related to this work.
Author Contributions
Jantzie, Robinson, Mannix, and Meehan conceived and designed the project. Jantzie and Robinson structured experimental design, critically reviewed all data and analysis, and wrote the manuscript. Berglass performed the stereology, and conducted all animal experimentation and preparations for immunohistochemistry and qPCR. All authors refined protocols, and contributed to data collection and analysis. Jantzie supervised all elements of data collection and analysis. Each author edited the manuscript and reviewed the final version.
References
- Benson RR, Gattu R, Sewick B, Kou Z, Zakariah N, Cavanaugh JM, Haacke EM. Detection of hemorrhagic and axonal pathology in mild traumatic brain injury using advanced MRI: implications for neurorehabilitation. NeuroRehabilitation. 2012;31(3):261–279. doi: 10.3233/NRE-2012-0795. [DOI] [PubMed] [Google Scholar]
- Browne KD, Chen XH, Meaney DF, Smith DH. Mild traumatic brain injury and diffuse axonal injury in swine. Journal of neurotrauma. 2011;28(9):1747–1755. doi: 10.1089/neu.2011.1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruce ED, Konda S, Dean DD, Wang EW, Huang JH, Little DM. Neuroimaging and traumatic brain injury: State of the field and voids in translational knowledge. Mol Cell Neurosci. 2015;66(Pt B):103–113. doi: 10.1016/j.mcn.2015.03.017. [DOI] [PubMed] [Google Scholar]
- Budde MD, Janes L, Gold E, Turtzo LC, Frank JA. The contribution of gliosis to diffusion tensor anisotropy and tractography following traumatic brain injury: validation in the rat using Fourier analysis of stained tissue sections. Brain: a journal of neurology. 2011;134(Pt 8):2248–2260. doi: 10.1093/brain/awr161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Budde MD, Xie M, Cross AH, Song SK. Axial diffusivity is the primary correlate of axonal injury in the experimental autoimmune encephalomyelitis spinal cord: a quantitative pixelwise analysis. J Neurosci. 2009;29(9):2805–2813. doi: 10.1523/JNEUROSCI.4605-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butovsky O, Jedrychowski MP, Moore CS, Cialic R, Lanser AJ, Gabriely G, Koeglsperger T, Dake B, Wu PM, Doykan CE, Fanek Z, Liu L, Chen Z, Rothstein JD, Ransohoff RM, Gygi SP, Antel JP, Weiner HL. Identification of a unique TGF-beta-dependent molecular and functional signature in microglia. Nat Neurosci. 2014;17(1):131–143. doi: 10.1038/nn.3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu IM, Morimoto ET, Goodarzi H, Liao JT, O’Keeffe S, Phatnani HP, Muratet M, Carroll MC, Levy S, Tavazoie S, Myers RM, Maniatis T. A neurodegeneration-specific gene-expression signature of acutely isolated microglia from an amyotrophic lateral sclerosis mouse model. Cell reports. 2013;4(2):385–401. doi: 10.1016/j.celrep.2013.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox CS, Jr, Baumgartner JE, Harting MT, Worth LL, Walker PA, Shah SK, Ewing-Cobbs L, Hasan KM, Day MC, Lee D, Jimenez F, Gee A. Autologous bone marrow mononuclear cell therapy for severe traumatic brain injury in children. Neurosurgery. 2011;68(3):588–600. doi: 10.1227/NEU.0b013e318207734c. [DOI] [PubMed] [Google Scholar]
- Croall ID, Cowie CJ, He J, Peel A, Wood J, Aribisala BS, Mitchell P, Mendelow AD, Smith FE, Millar D, Kelly T, Blamire AM. White matter correlates of cognitive dysfunction after mild traumatic brain injury. Neurology. 2014;83(6):494–501. doi: 10.1212/WNL.0000000000000666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson MA, Banks WA. Cytokine and chemokine responses in serum and brain after single and repeated injections of lipopolysaccharide: multiplex quantification with path analysis. Brain, behavior, and immunity. 2011;25(8):1637–1648. doi: 10.1016/j.bbi.2011.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faul M, Coronado V. Epidemiology of traumatic brain injury. Handbook of clinical neurology. 2015;127:3–13. doi: 10.1016/B978-0-444-52892-6.00001-5. [DOI] [PubMed] [Google Scholar]
- Fox RJ, McColl RW, Lee JC, Frohman T, Sakaie K, Frohman E. A preliminary validation study of diffusion tensor imaging as a measure of functional brain injury. Arch Neurol. 2008;65(9):1179–1184. doi: 10.1001/archneur.65.9.1179. [DOI] [PubMed] [Google Scholar]
- Goos JD, van der Flier WM, Knol DL, Pouwels PJ, Scheltens P, Barkhof F, Wattjes MP. Clinical relevance of improved microbleed detection by susceptibility-weighted magnetic resonance imaging. Stroke; a journal of cerebral circulation. 2011;42(7):1894–1900. doi: 10.1161/STROKEAHA.110.599837. [DOI] [PubMed] [Google Scholar]
- Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, Launer LJ, Van Buchem MA, Breteler MM Microbleed Study G. Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol. 2009;8(2):165–174. doi: 10.1016/S1474-4422(09)70013-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gundersen HJ, Bagger P, Bendtsen TF, Evans SM, Korbo L, Marcussen N, Moller A, Nielsen K, Nyengaard JR, Pakkenberg B, et al. The new stereological tools: disector, fractionator, nucleator and point sampled intercepts and their use in pathological research and diagnosis. APMIS: acta pathologica, microbiologica, et immunologica Scandinavica. 1988;96(10):857–881. doi: 10.1111/j.1699-0463.1988.tb00954.x. [DOI] [PubMed] [Google Scholar]
- Hickman SE, Kingery ND, Ohsumi TK, Borowsky ML, Wang LC, Means TK, El Khoury J. The microglial sensome revealed by direct RNA sequencing. Nat Neurosci. 2013;16(12):1896–1905. doi: 10.1038/nn.3554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinson HE, Rowell S, Schreiber M. Clinical evidence of inflammation driving secondary brain injury: a systematic review. The journal of trauma and acute care surgery. 2015;78(1):184–191. doi: 10.1097/TA.0000000000000468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Corbett CJ, Berglass J, Firl DJ, Flores J, Mannix R, Robinson S. Complex pattern of interaction between in utero hypoxia-ischemia and intra-amniotic inflammation disrupts brain development and motor function. J Neuroinflammation. 2014a;11:131. doi: 10.1186/1742-2094-11-131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Corbett CJ, Berglass J, Firl DJ, Flores J, Mannix R, Robinson S. Complex pattern of interaction between in utero hypoxia-ischemia and intra-amniotic inflammation disrupts brain development and motor function. J Neuroinflammation. 2014b;11(1):131. doi: 10.1186/1742-2094-11-131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Getsy PM, Firl DJ, Wilson CG, Miller RH, Robinson S. Erythropoietin attenuates loss of potassium chloride co-transporters following prenatal brain injury. Molecular and cellular neurosciences. 2014c;61:152–162. doi: 10.1016/j.mcn.2014.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallakuri S, Bandaru S, Zakaria N, Shen Y, Kou Z, Zhang L, Haacke EM, Cavanaugh JM. Traumatic Brain Injury by a Closed Head Injury Device Induces Cerebral Blood Flow Changes and Microhemorrhages. J Clin Imaging Sci. 2015;5:52. doi: 10.4103/2156-7514.166354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondo A, Shahpasand K, Mannix R, Qiu J, Moncaster J, Chen CH, Yao Y, Lin YM, Driver JA, Sun Y, Wei S, Luo ML, Albayram O, Huang P, Rotenberg A, Ryo A, Goldstein LE, Pascual-Leone A, McKee AC, Meehan W, Zhou XZ, Lu KP. Antibody against early driver of neurodegeneration cis P-tau blocks brain injury and tauopathy. Nature. 2015;523(7561):431–436. doi: 10.1038/nature14658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar R, Gupta RK, Husain M, Chaudhry C, Srivastava A, Saksena S, Rathore RK. Comparative evaluation of corpus callosum DTI metrics in acute mild and moderate traumatic brain injury: its correlation with neuropsychometric tests. Brain injury. 2009;23(7):675–685. doi: 10.1080/02699050903014915. [DOI] [PubMed] [Google Scholar]
- Lancaster MA, Olson DV, McCrea MA, Nelson LD, LaRoche AA, Muftuler LT. Acute white matter changes following sport-related concussion: A serial diffusion tensor and diffusion kurtosis tensor imaging study. Hum Brain Mapp. 2016 doi: 10.1002/hbm.23278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Kou Z, Tian Y. Diffuse axonal injury after traumatic cerebral microbleeds: an evaluation of imaging techniques. Neural Regen Res. 2014a;9(12):1222–1230. doi: 10.4103/1673-5374.135330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Buch S, Chen Y, Choi HS, Dai Y, Habib C, Hu J, Jung JY, Luo Y, Utriainen D, Wang M, Wu D, Xia S, Haacke EM. Susceptibility-weighted imaging: current status and future directions. NMR in biomedicine. 2016 doi: 10.1002/nbm.3552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Grigoryan MM, Vasilevko V, Sumbria RK, Paganini-Hill A, Cribbs DH, Fisher MJ. Comparative analysis of H&E and Prussian blue staining in a mouse model of cerebral microbleeds. J Histochem Cytochem. 2014b;62(11):767–773. doi: 10.1369/0022155414546692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longhi L, Perego C, Ortolano F, Aresi S, Fumagalli S, Zanier ER, Stocchetti N, De Simoni MG. Tumor necrosis factor in traumatic brain injury: effects of genetic deletion of p55 or p75 receptor. J Cereb Blood Flow Metab. 2013;33(8):1182–1189. doi: 10.1038/jcbfm.2013.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mac Donald CL, Dikranian K, Bayly P, Holtzman D, Brody D. Diffusion tensor imaging reliably detects experimental traumatic axonal injury and indicates approximate time of injury. J Neurosci. 2007;27(44):11869–11876. doi: 10.1523/JNEUROSCI.3647-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannix R, Berglass J, Berkner J, Moleus P, Qiu J, Andrews N, Gunner G, Berglass L, Jantzie LL, Robinson S, Meehan WP., 3rd Chronic gliosis and behavioral deficits in mice following repetitive mild traumatic brain injury. Journal of neurosurgery. 2014;121(6):1342–1350. doi: 10.3171/2014.7.JNS14272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannix R, Meehan WP, Mandeville J, Grant PE, Gray T, Berglass J, Zhang J, Bryant J, Rezaie S, Chung JY, Peters NV, Lee C, Tien LW, Kaplan DL, Feany M, Whalen M. Clinical correlates in an experimental model of repetitive mild brain injury. Annals of neurology. 2013;74(1):65–75. doi: 10.1002/ana.23858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marin JR, Weaver MD, Yealy DM, Mannix RC. Trends in visits for traumatic brain injury to emergency departments in the United States. JAMA. 2014;311(18):1917–1919. doi: 10.1001/jama.2014.3979. [DOI] [PubMed] [Google Scholar]
- Mayer AR, Ling JM, Yang Z, Pena A, Yeo RA, Klimaj S. Diffusion abnormalities in pediatric mild traumatic brain injury. J Neurosci. 2012;32(50):17961–17969. doi: 10.1523/JNEUROSCI.3379-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meehan WP, 3rd, Zhang J, Mannix R, Whalen MJ. Increasing recovery time between injuries improves cognitive outcome after repetitive mild concussive brain injuries in mice. Neurosurgery. 2012;71(4):885–891. doi: 10.1227/NEU.0b013e318265a439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori Y, Murakami M, Arima Y, Zhu D, Terayama Y, Komai Y, Nakatsuji Y, Kamimura D, Yoshioka Y. Early pathological alterations of lower lumbar cords detected by ultrahigh-field MRI in a mouse multiple sclerosis model. Int Immunol. 2014;26(2):93–101. doi: 10.1093/intimm/dxt044. [DOI] [PubMed] [Google Scholar]
- Mouton. Neurostereology: Unbiased stereology of neural systems. Iowa USA: John Wiley & Sons; 2014. [Google Scholar]
- Mukherjee P, McKinstry RC. Diffusion tensor imaging and tractography of human brain development. Neuroimaging clinics of North America. 2006;16(1):19–43. vii. doi: 10.1016/j.nic.2005.11.004. [DOI] [PubMed] [Google Scholar]
- Mukherjee P, Miller JH, Shimony JS, Conturo TE, Lee BC, Almli CR, McKinstry RC. Normal brain maturation during childhood: developmental trends characterized with diffusion-tensor MR imaging. Radiology. 2001;221(2):349–358. doi: 10.1148/radiol.2212001702. [DOI] [PubMed] [Google Scholar]
- Naude PJ, den Boer JA, Luiten PG, Eisel UL. Tumor necrosis factor receptor cross-talk. FEBS J. 2011;278(6):888–898. doi: 10.1111/j.1742-4658.2011.08017.x. [DOI] [PubMed] [Google Scholar]
- Naude PJ, Dobos N, van der Meer D, Mulder C, Pawironadi KG, den Boer JA, van der Zee EA, Luiten PG, Eisel UL. Analysis of cognition, motor performance and anxiety in young and aged tumor necrosis factor alpha receptor 1 and 2 deficient mice. Behavioural brain research. 2014;258:43–51. doi: 10.1016/j.bbr.2013.10.006. [DOI] [PubMed] [Google Scholar]
- Opal SM, DePalo VA. Anti-inflammatory cytokines. Chest. 2000;117(4):1162–1172. doi: 10.1378/chest.117.4.1162. [DOI] [PubMed] [Google Scholar]
- Owen JP, Marco EJ, Desai S, Fourie E, Harris J, Hill SS, Arnett AB, Mukherjee P. Abnormal white matter microstructure in children with sensory processing disorders. NeuroImage Clinical. 2013;2:844–853. doi: 10.1016/j.nicl.2013.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasternak O, Koerte IK, Bouix S, Fredman E, Sasaki T, Mayinger M, Helmer KG, Johnson AM, Holmes JD, Forwell LA, Skopelja EN, Shenton ME, Echlin PS. Hockey Concussion Education Project, Part 2. Microstructural white matter alterations in acutely concussed ice hockey players: a longitudinal free-water MRI study. Journal of neurosurgery. 2014;120(4):873–881. doi: 10.3171/2013.12.JNS132090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Povlishock JT, Katz DI. Update of neuropathology and neurological recovery after traumatic brain injury. The Journal of head trauma rehabilitation. 2005;20(1):76–94. doi: 10.1097/00001199-200501000-00008. [DOI] [PubMed] [Google Scholar]
- Robinson S, Winer JL, Berkner J, Chan LA, Denson JL, Maxwell JR, Yang Y, Sillerud LO, Tasker RC, Meehan WP, 3rd, Mannix R, Jantzie LL. Imaging and serum biomarkers reflecting the functional efficacy of extended erythropoietin treatment in rats following infantile traumatic brain injury. Journal of neurosurgery Pediatrics. 2016:1–17. doi: 10.3171/2015.10.PEDS15554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toth A, Kovacs N, Tamas V, Kornyei B, Nagy M, Horvath A, Rostas T, Bogner P, Janszky J, Doczi T, Buki A, Schwarcz A. Microbleeds may expand acutely after traumatic brain injury. Neurosci Lett. 2016;617:207–212. doi: 10.1016/j.neulet.2016.02.028. [DOI] [PubMed] [Google Scholar]
- Wang G, Zhang J, Hu X, Zhang L, Mao L, Jiang X, Liou AK, Leak RK, Gao Y, Chen J. Microglia/macrophage polarization dynamics in white matter after traumatic brain injury. J Cereb Blood Flow Metab. 2013;33(12):1864–1874. doi: 10.1038/jcbfm.2013.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster SJ, Van Eldik LJ, Watterson DM, Bachstetter AD. Closed head injury in an age-related Alzheimer mouse model leads to an altered neuroinflammatory response and persistent cognitive impairment. J Neurosci. 2015;35(16):6554–6569. doi: 10.1523/JNEUROSCI.0291-15.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J, Zhao Z, Sabirzhanov B, Stoica BA, Kumar A, Luo T, Skovira J, Faden AI. Spinal cord injury causes brain inflammation associated with cognitive and affective changes: role of cell cycle pathways. J Neurosci. 2014;34(33):10989–11006. doi: 10.1523/JNEUROSCI.5110-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, You Z, Kim HH, Hwang SK, Khuman J, Guo S, Lo EH, Whalen MJ. Genetic analysis of the role of tumor necrosis factor receptors in functional outcome after traumatic brain injury in mice. Journal of neurotrauma. 2010;27(6):1037–1046. doi: 10.1089/neu.2009.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]