Abstract
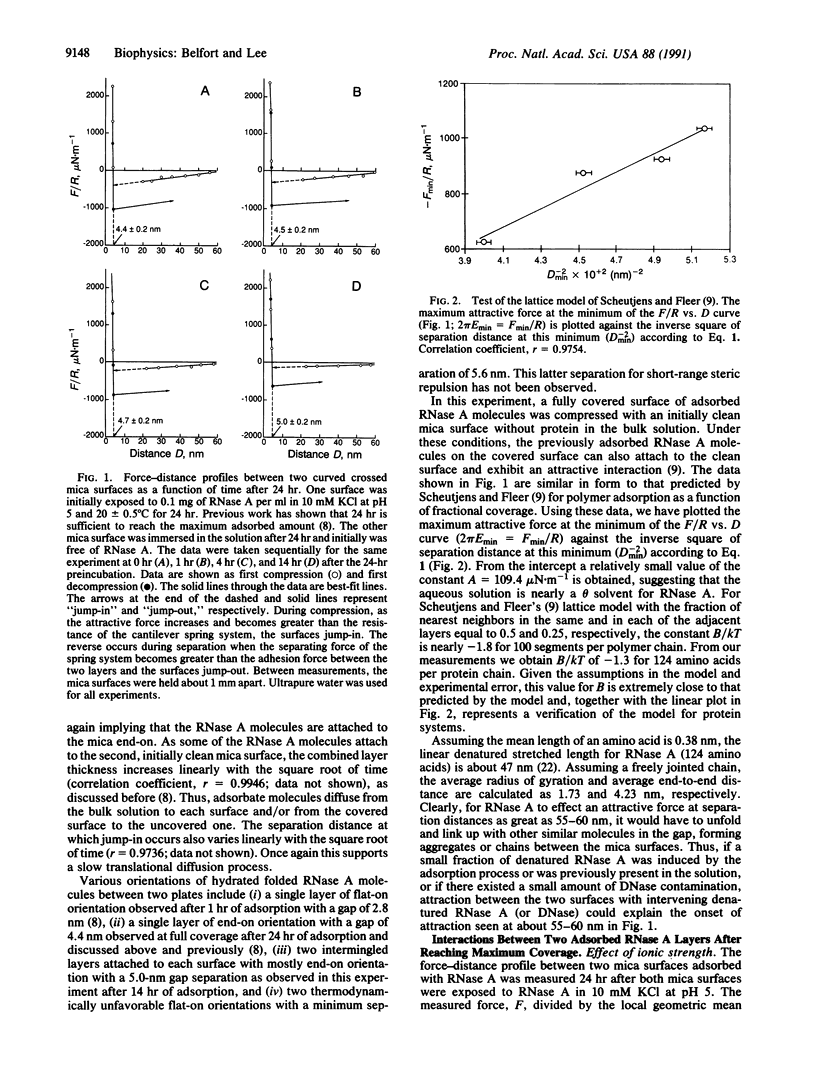
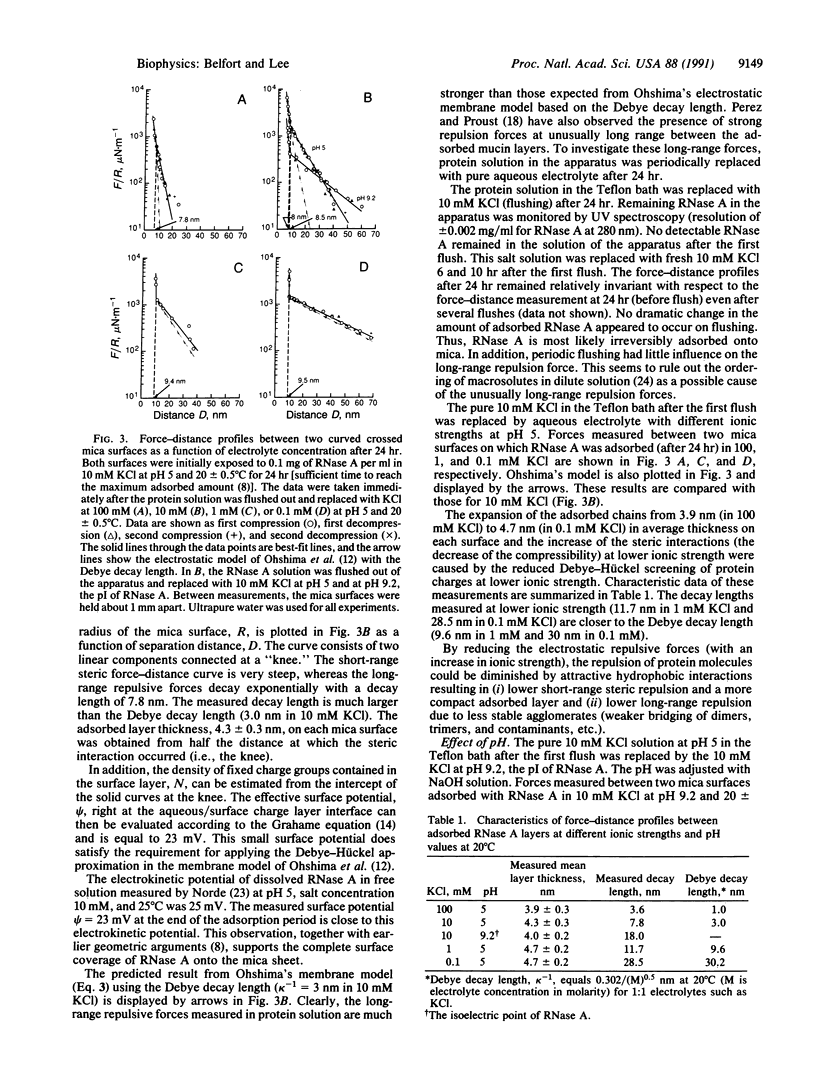
Adsorbed layers of pancreatic RNase A on molecularly smooth mica in aqueous solution attract inorganic mica surfaces whereas they repel similarly adsorbed RNase A layers. As the clean mica surface is covered with RNase A, the attractive interaction slowly diminishes with time and eventually converts to a purely repulsive interaction. Solvent is squeezed out of the solution in the gap during compression of the two surfaces so that the adsorbed protein concentration, as measured directly by the refractive index, increases significantly. The kinetics of this process is analyzed using surface force-distance measurements. All these results are predicted for constrained equilibrium by a discrete lattice model [Scheutjens, J. M. H. M. & Fleer, G. J. (1985) Macromolecules 18, 1882-1900]. Reasonable values are obtained for the constants of the model. We also report on the equilibrium behavior and interaction of densely adsorbed RNase A layers in aqueous solutions of varying ionic strength and pH. With increasing ionic strength, intramolecular forces dominate with diminished electrostatic repulsion. Thus, the adsorbed protein layer becomes more compact while unattached protein molecules coil and fold, making them less likely to form strong intermolecular bridges. Only at very low ionic strength (0.1 mM KCl), when electrostatic forces dominate, does the membrane potential model come close to predicting the long-distance repulsive behavior. Thus, at higher ionic strengths, other non-electrostatic interactions (such as hydrophobic interactions) possibly dominate. An increase in the pH of the solution from 5 to 9.2, the pI of RNase A, significantly reduces the electrostatic repulsion between protein molecules in favor of hydrophobic attractive interactions. This results in lower short-range steric repulsion. However, in contrast to the ionic-strength effect, an increased long-range repulsive force with a much longer decay length is observed. This may be due to contaminants such as DNase that have their pI at a pH other than 9.2. Thus, as with the changing-ionic-strength study, thinner and denser adsorbed layers are formed. Finally, for the kinetic studies, two characteristic length scales--the thickness of the adsorbed layer and the "jump-in" distance--vary linearly with the square root of time. This is consistent with our earlier results and once again implies a diffusion-driven process.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Afshar-Rad T., Bailey A. I., Luckham P. F., MacNaughtan W., Chapman D. Forces between proteins and model polypeptides adsorbed on mica surfaces. Biochim Biophys Acta. 1987 Sep 2;915(1):101–111. doi: 10.1016/0167-4838(87)90129-4. [DOI] [PubMed] [Google Scholar]
- Dill K. A. Dominant forces in protein folding. Biochemistry. 1990 Aug 7;29(31):7133–7155. doi: 10.1021/bi00483a001. [DOI] [PubMed] [Google Scholar]
- Getzoff E. D., Tainer J. A., Lerner R. A., Geysen H. M. The chemistry and mechanism of antibody binding to protein antigens. Adv Immunol. 1988;43:1–98. doi: 10.1016/s0065-2776(08)60363-6. [DOI] [PubMed] [Google Scholar]
- Hammer D. A., Lauffenburger D. A. A dynamical model for receptor-mediated cell adhesion to surfaces. Biophys J. 1987 Sep;52(3):475–487. doi: 10.1016/S0006-3495(87)83236-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janda K. D. New strategies for the design of catalytic antibodies. Biotechnol Prog. 1990 May-Jun;6(3):178–181. doi: 10.1021/bp00003a002. [DOI] [PubMed] [Google Scholar]
- Norde W. Adsorption of proteins from solution at the solid-liquid interface. Adv Colloid Interface Sci. 1986 Sep;25(4):267–340. doi: 10.1016/0001-8686(86)80012-4. [DOI] [PubMed] [Google Scholar]
- Silberberg A., Klein J. Structure and properties of surface adsorbed biopolymer layers - soluble collagen adsorbed to glass. Biorheology. 1981;18(3-6):589–599. doi: 10.3233/bir-1981-183-622. [DOI] [PubMed] [Google Scholar]
- Suck D., Oefner C. Structure of DNase I at 2.0 A resolution suggests a mechanism for binding to and cutting DNA. Nature. 1986 Jun 5;321(6070):620–625. doi: 10.1038/321620a0. [DOI] [PubMed] [Google Scholar]



