Abstract
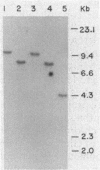
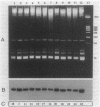
In the cyanobacterium Synechocystis sp. strain PCC 6803 (Synechocystis 6803) delta-aminolevulinic acid (ALA), the sole precursor for the synthesis of the porphyrin rings of heme and chlorophyll, is formed from glutamate activated by acylation to tRNA(Glu) (G. P. O'Neill, D. M. Peterson, A. Schön, M. W. Chen, and D. Söll, J. Bacteriol. 170:3810-3816, 1988; S. Rieble and S. I. Beale, J. Biol. Chem. 263:8864-8871, 1988). We report here that Synechocystis 6803 possesses a single tRNA(Glu) gene which was transcribed as monomeric precursor tRNA and matured into the two tRNA(Glu) species. They differed in the extent of modification of the first anticodon base, 5-methylaminomethyl-2-thiouridine (O'Neill et al., 1988). The two tRNA species had equivalent capacities to stimulate the tRNA-dependent formation of ALA in Synechocystis 6803 and to provide glutamate for protein biosynthesis in an Escherichia coli-derived translation system. These results are in support of a dual role of tRNA(Glu). The levels of tRNA(Glu) were examined by Northern (RNA) blot analysis of cellular RNA and by aminoacylation assays in cultures of Synechocystis 6803 in which the amount of chlorophyll synthesized was modulated over a 10-fold range by various illumination regimens or by the addition of inhibitors of chlorophyll and ALA biosynthesis. In these cultures, the level of tRNA(Glu) was always a constant fraction of the total tRNA population, suggesting that tRNA(Glu) and chlorophyll levels are regulated independently. In addition, the tRNA(Glu) was always fully aminoacylated in vivo.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andachi Y., Yamao F., Muto A., Osawa S. Codon recognition patterns as deduced from sequences of the complete set of transfer RNA species in Mycoplasma capricolum. Resemblance to mitochondria. J Mol Biol. 1989 Sep 5;209(1):37–54. doi: 10.1016/0022-2836(89)90168-x. [DOI] [PubMed] [Google Scholar]
- Avissar Y. J., Beale S. I. Identification of the enzymatic basis for delta-aminolevulinic acid auxotrophy in a hemA mutant of Escherichia coli. J Bacteriol. 1989 Jun;171(6):2919–2924. doi: 10.1128/jb.171.6.2919-2924.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avissar Y. J., Ormerod J. G., Beale S. I. Distribution of delta-aminolevulinic acid biosynthetic pathways among phototrophic bacterial groups. Arch Microbiol. 1989;151(6):513–519. doi: 10.1007/BF00454867. [DOI] [PubMed] [Google Scholar]
- Beale S. I., Gough S. P., Granick S. Biosynthesis of delta-aminolevulinic acid from the intact carbon skeleton of glutamic acid in greening barley. Proc Natl Acad Sci U S A. 1975 Jul;72(7):2719–2723. doi: 10.1073/pnas.72.7.2719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beale S. I. The biosynthesis of delta-aminolevulinic acid in Chlorella. Plant Physiol. 1970 Apr;45(4):504–506. doi: 10.1104/pp.45.4.504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett A., Bogorad L. Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol. 1973 Aug;58(2):419–435. doi: 10.1083/jcb.58.2.419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brendel V., Trifonov E. N. A computer algorithm for testing potential prokaryotic terminators. Nucleic Acids Res. 1984 May 25;12(10):4411–4427. doi: 10.1093/nar/12.10.4411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H. Z., Zubay G. Prokaryotic coupled transcription-translation. Methods Enzymol. 1983;101:674–690. doi: 10.1016/0076-6879(83)01047-2. [DOI] [PubMed] [Google Scholar]
- Chua B., Elson C., Shrago E. Growth-dependent factors in the regulation of aminoacyl-tRNA synthetase activities of Tetrahymena pyriformis. Biochim Biophys Acta. 1977 Oct 18;478(4):474–485. doi: 10.1016/0005-2787(77)90102-2. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dower W. J., Miller J. F., Ragsdale C. W. High efficiency transformation of E. coli by high voltage electroporation. Nucleic Acids Res. 1988 Jul 11;16(13):6127–6145. doi: 10.1093/nar/16.13.6127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dzelzkalns V. A., Bogorad L. Stable transformation of the cyanobacterium Synechocystis sp. PCC 6803 induced by UV irradiation. J Bacteriol. 1986 Mar;165(3):964–971. doi: 10.1128/jb.165.3.964-971.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evrard J. L., Kuntz M., Straus N. A., Weil J. H. A class-I intron in a cyanelle tRNA gene from Cyanophora paradoxa: phylogenetic relationship between cyanelles and plant chloroplasts. Gene. 1988 Nov 15;71(1):115–122. doi: 10.1016/0378-1119(88)90083-2. [DOI] [PubMed] [Google Scholar]
- Fournier M. J., Ozeki H. Structure and organization of the transfer ribonucleic acid genes of Escherichia coli K-12. Microbiol Rev. 1985 Dec;49(4):379–397. doi: 10.1128/mr.49.4.379-397.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garel J. P. Functional adaptation of tRNA population. J Theor Biol. 1974 Jan;43(1):211–225. doi: 10.1016/s0022-5193(74)80054-8. [DOI] [PubMed] [Google Scholar]
- Garel J. Quantitative adaptation of isoacceptor tRNAs to mRNA codons of alanine, glycine and serine. Nature. 1976 Apr 29;260(5554):805–806. doi: 10.1038/260805a0. [DOI] [PubMed] [Google Scholar]
- Goldsmith M. R., Kafatos F. C. Developmentally regulated genes in silkmoths. Annu Rev Genet. 1984;18:443–487. doi: 10.1146/annurev.ge.18.120184.002303. [DOI] [PubMed] [Google Scholar]
- Hentzen D., Chevallier A., Garel J. P. Differential usage of iso-accepting tRNASer species in silk glands of Bombyx mori. Nature. 1981 Mar 19;290(5803):267–269. doi: 10.1038/290267a0. [DOI] [PubMed] [Google Scholar]
- Hollingsworth M. J., Hallick R. B. Euglena gracilis chloroplast transfer RNA transcription units. Nucleotide sequence analysis of a tRNATyr-tRNAHis-tRNAMet-tRNATrp-tRNAGlu-tRNAGly gene cluster. J Biol Chem. 1982 Nov 10;257(21):12795–12799. [PubMed] [Google Scholar]
- Huang D. D., Wang W. Y. Chlorophyll biosynthesis in Chlamydomonas starts with the formation of glutamyl-tRNA. J Biol Chem. 1986 Oct 15;261(29):13451–13455. [PubMed] [Google Scholar]
- Huang D. D., Wang W. Y., Gough S. P., Kannangara C. G. delta-Aminolevulinic acid-synthesizing enzymes need an RNA moiety for activity. Science. 1984 Sep 28;225(4669):1482–1484. doi: 10.1126/science.6206568. [DOI] [PubMed] [Google Scholar]
- Jahn D., Kim Y. C., Ishino Y., Chen M. W., Söll D. Purification and functional characterization of the Glu-tRNA(Gln) amidotransferase from Chlamydomonas reinhardtii. J Biol Chem. 1990 May 15;265(14):8059–8064. [PubMed] [Google Scholar]
- Kannangara C. G., Gough S. P., Bruyant P., Hoober J. K., Kahn A., von Wettstein D. tRNA(Glu) as a cofactor in delta-aminolevulinate biosynthesis: steps that regulate chlorophyll synthesis. Trends Biochem Sci. 1988 Apr;13(4):139–143. doi: 10.1016/0968-0004(88)90071-0. [DOI] [PubMed] [Google Scholar]
- Komine Y., Adachi T., Inokuchi H., Ozeki H. Genomic organization and physical mapping of the transfer RNA genes in Escherichia coli K12. J Mol Biol. 1990 Apr 20;212(4):579–598. doi: 10.1016/0022-2836(90)90224-A. [DOI] [PubMed] [Google Scholar]
- Koontz S. W., Jakubowski H., Goldman E. Control of RNA and protein synthesis by the concentration of Trp-tRNATrp in Escherichia coli infected with bacteriophage MS2. J Mol Biol. 1983 Aug 25;168(4):747–763. doi: 10.1016/s0022-2836(83)80073-4. [DOI] [PubMed] [Google Scholar]
- Kuntz M., Weil J. H., Steinmetz A. Nucleotide sequence of a 2 kbp BamH I fragment of Vicia faba chloroplast DNA containing the genes for threonine, glutamic acid and tyrosine transfer RNAs. Nucleic Acids Res. 1984 Jun 25;12(12):5037–5047. doi: 10.1093/nar/12.12.5037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapointe J., Delcuve G. Thermosensitive mutants of Escherichia coli K-12 altered in the catalytic Subunit and in a Regulatory factor of the glutamy-transfer ribonucleic acid synthetase. J Bacteriol. 1975 May;122(2):352–358. doi: 10.1128/jb.122.2.352-358.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapointe J., Söll D. Glutamyl transfer ribonucleic acid synthetase of Escherichia coli. I. Purification and properties. J Biol Chem. 1972 Aug 25;247(16):4966–4974. [PubMed] [Google Scholar]
- Li J. M., Brathwaite O., Cosloy S. D., Russell C. S. 5-Aminolevulinic acid synthesis in Escherichia coli. J Bacteriol. 1989 May;171(5):2547–2552. doi: 10.1128/jb.171.5.2547-2552.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClure W. R. Mechanism and control of transcription initiation in prokaryotes. Annu Rev Biochem. 1985;54:171–204. doi: 10.1146/annurev.bi.54.070185.001131. [DOI] [PubMed] [Google Scholar]
- Neihardt F. C., Parker J., McKeever W. G. Function and regulation of aminoacyl-tRNA synthetases in prokaryotic and eukaryotic cells. Annu Rev Microbiol. 1975;29:215–250. doi: 10.1146/annurev.mi.29.100175.001243. [DOI] [PubMed] [Google Scholar]
- Normanly J., Ogden R. C., Horvath S. J., Abelson J. Changing the identity of a transfer RNA. Nature. 1986 May 15;321(6067):213–219. doi: 10.1038/321213a0. [DOI] [PubMed] [Google Scholar]
- O'Neill G. P., Chen M. W., Söll D. delta-Aminolevulinic acid biosynthesis in Escherichia coli and Bacillus subtilis involves formation of glutamyl-tRNA. FEMS Microbiol Lett. 1989 Aug;51(3):255–259. doi: 10.1016/0378-1097(89)90406-0. [DOI] [PubMed] [Google Scholar]
- O'Neill G. P., Peterson D. M., Schön A., Chen M. W., Söll D. Formation of the chlorophyll precursor delta-aminolevulinic acid in cyanobacteria requires aminoacylation of a tRNAGlu species. J Bacteriol. 1988 Sep;170(9):3810–3816. doi: 10.1128/jb.170.9.3810-3816.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohme M., Kamogashira T., Shinozaki K., Sugiura M. Structure and cotranscription of tobacco chloroplast genes for tRNAGlu(UUC), tRNATyr(GUA) and tRNAAsp(GUC). Nucleic Acids Res. 1985 Feb 25;13(4):1045–1056. doi: 10.1093/nar/13.4.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quigley F., Weil J. H. Organization and sequence of five tRNA genes and of an unidentified reading frame in the wheat chloroplast genome: evidence for gene rearrangements during the evolution of chloroplast genomes. Curr Genet. 1985;9(6):495–503. doi: 10.1007/BF00434054. [DOI] [PubMed] [Google Scholar]
- Rasmussen O. F., Stummann B. M., Henningsen K. W. Nucleotide sequence of a 1.1 kb fragment of the pea chloroplast genome containing three tRNA genes, one of which is located within an open reading frame of 91 codons. Nucleic Acids Res. 1984 Dec 11;12(23):9143–9153. doi: 10.1093/nar/12.23.9143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rieble S., Beale S. I. Transformation of glutamate to delta-aminolevulinic acid by soluble extracts of Synechocystis sp. PCC 6803 and other oxygenic prokaryotes. J Biol Chem. 1988 Jun 25;263(18):8864–8871. [PubMed] [Google Scholar]
- Russell R. R., Pittard A. J. Mutants of Escherichia coli unable to make protein at 42 C. J Bacteriol. 1971 Nov;108(2):790–798. doi: 10.1128/jb.108.2.790-798.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samuelsson T., Axberg T., Borén T., Lagerkvist U. Unconventional reading of the glycine codons. J Biol Chem. 1983 Nov 10;258(21):13178–13184. [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneegurt M. A., Rieble S., Beale S. I. The tRNA Required for in Vitro delta-Aminolevulinic Acid Formation from Glutamate in Synechocystis Extracts : Determination of Activity in a Synechocystis in Vitro Protein Synthesizing System. Plant Physiol. 1988 Dec;88(4):1358–1366. doi: 10.1104/pp.88.4.1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schön A., Kannangara C. G., Gough S., Söll D. Protein biosynthesis in organelles requires misaminoacylation of tRNA. Nature. 1988 Jan 14;331(6152):187–190. doi: 10.1038/331187a0. [DOI] [PubMed] [Google Scholar]
- Shioi Y., Doi M., Tanabe K., Shimokawa K. Inhibition of porphyrin biosynthesis by exogenous 5-aminolevulinic acid in an aerobic photosynthetic bacterium, Erythrobacter sp. OCh 114. Arch Biochem Biophys. 1988 Nov 1;266(2):478–485. doi: 10.1016/0003-9861(88)90280-9. [DOI] [PubMed] [Google Scholar]
- Smith D. R., Calvo J. M. Nucleotide sequence of the E coli gene coding for dihydrofolate reductase. Nucleic Acids Res. 1980 May 24;8(10):2255–2274. doi: 10.1093/nar/8.10.2255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprinzl M., Hartmann T., Weber J., Blank J., Zeidler R. Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res. 1989;17 (Suppl):r1–172. doi: 10.1093/nar/17.suppl.r1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomale J., Nass G. Alteration of the intracellular concentration of aminoacyl-tRNA synthetases and isoaccepting tRNAs during amino-acid limited growth in Escherichia coli. Eur J Biochem. 1978 Apr 17;85(2):407–418. doi: 10.1111/j.1432-1033.1978.tb12253.x. [DOI] [PubMed] [Google Scholar]
- Vieira J., Messing J. The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene. 1982 Oct;19(3):259–268. doi: 10.1016/0378-1119(82)90015-4. [DOI] [PubMed] [Google Scholar]
- Vold B. S. Structure and organization of genes for transfer ribonucleic acid in Bacillus subtilis. Microbiol Rev. 1985 Mar;49(1):71–80. doi: 10.1128/mr.49.1.71-80.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson S. E., Doolittle W. F. Genes for tRNAIle and tRNAAla in the spacer between the 16S and 23S rRNA genes of a blue-green alga: strong homology to chloroplast tRNA genes and tRNA genes of the E. coli rrnD gene cluster. Nucleic Acids Res. 1983 Jan 11;11(1):225–235. doi: 10.1093/nar/11.1.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young L. S., Takahashi N., Sprague K. U. Upstream sequences confer distinctive transcriptional properties on genes encoding silkgland-specific tRNAAla. Proc Natl Acad Sci U S A. 1986 Jan;83(2):374–378. doi: 10.1073/pnas.83.2.374. [DOI] [PMC free article] [PubMed] [Google Scholar]