Abstract
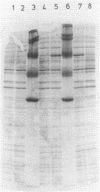
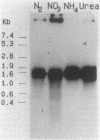
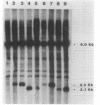
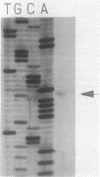
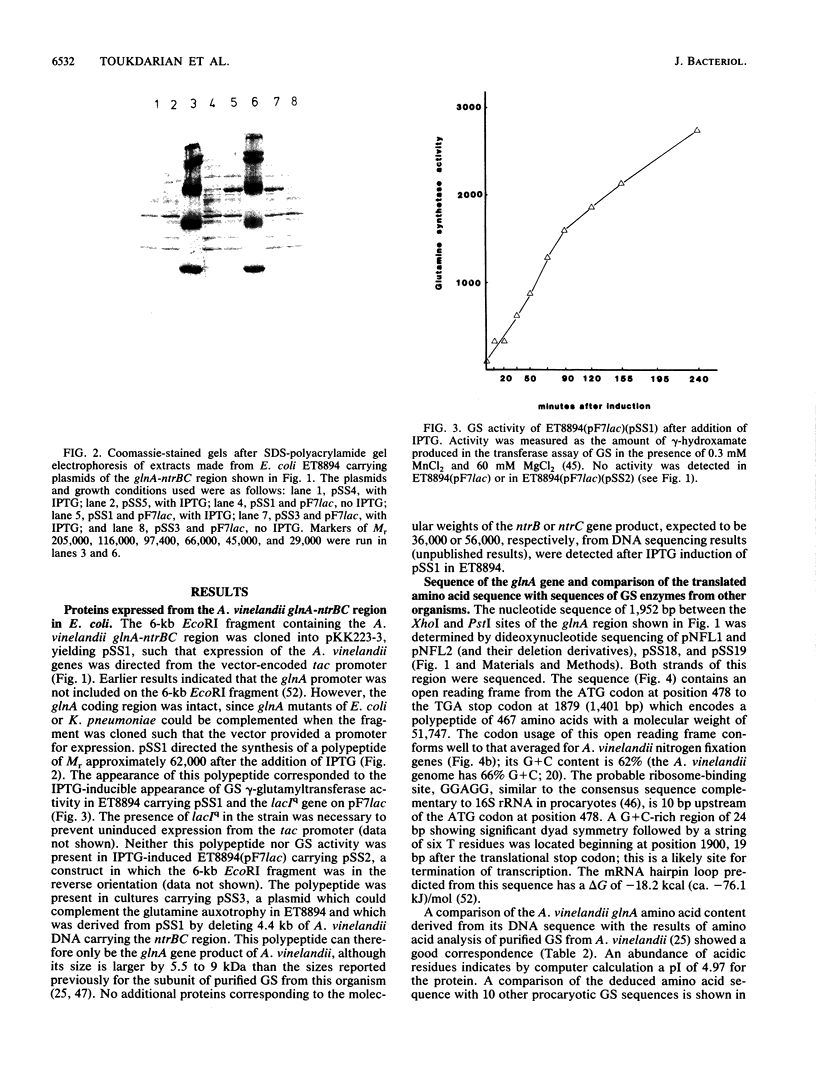
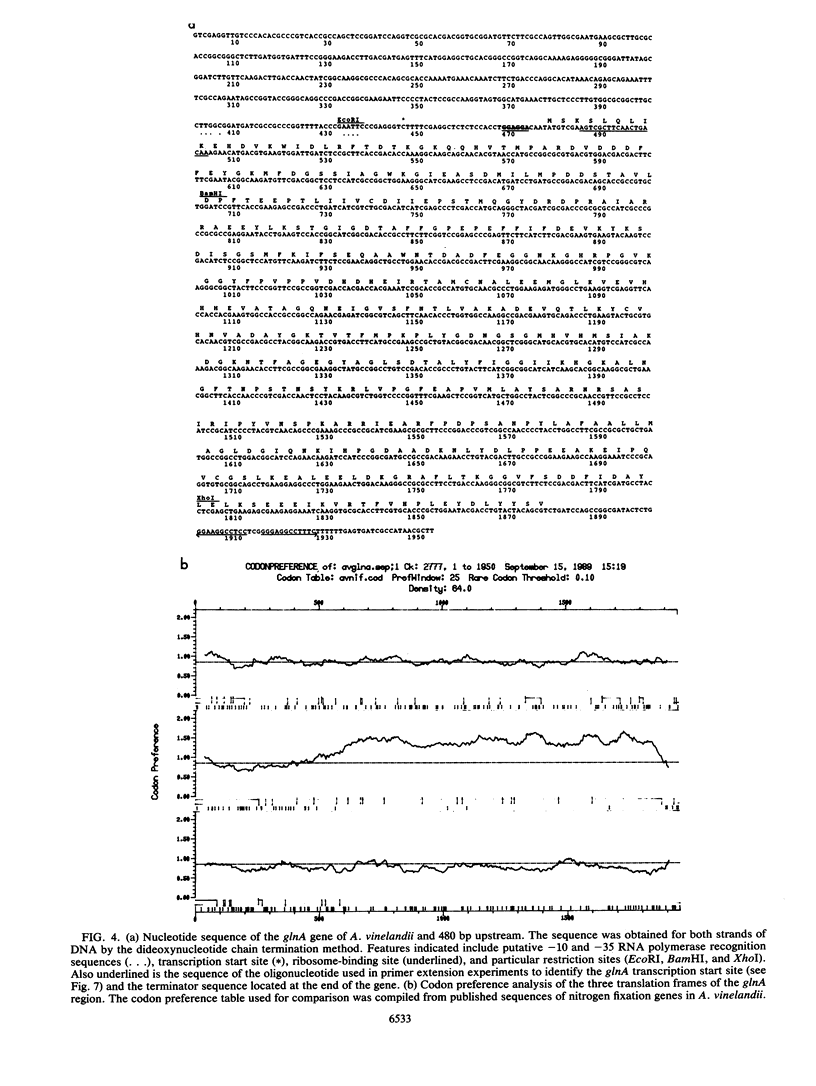
The gene encoding glutamine synthetase (GS), glnA, was cloned from Azotobacter vinelandii on a 6-kb EcoRI fragment that also carries the ntrBC genes. The DNA sequence of 1,952 bp including the GS-coding region was determined. An open reading frame of 467 amino acids indicated a gene product of Mr 51,747. Transcription of glnA occurred from a C residue located 32 bases upstream of an ATG considered to be the initiator codon because (i) it had a nearby potential ribosome-binding site and (ii) an open reading frame translated from this site indicated good N-terminal homology to 10 other procaryotic GSs. Sequences similar to the consensus RNA polymerase recognition sites at -10 and -35 were present at the appropriate distance upstream of the transcription initiation site. As expected from earlier genetic studies indicating that expression of A. vinelandii glnA did not depend on the rpoN (ntrA; sigma 54) gene product, no sigma 54 recognition sequences were present, nor was there significant regulation of glnA expression by fixed nitrogen. Repeated attempts to construct glutamine auxotrophs by recombination of glnA insertion mutations were unsuccessful, Although the mutated DNA could be found by hybridization experiments in drug-resistant A. vinelandii transformants, the wild-type glnA region was always present. These results suggest that glnA mutations are lethal in A. vinelandii. In [14C]glutamine uptake experiments, very little glutamine was incorporated into cells, suggesting that glutamine auxotrophs are nonviable because they cannot be supplied with sufficient glutamine to support growth.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Almassy R. J., Janson C. A., Hamlin R., Xuong N. H., Eisenberg D. Novel subunit-subunit interactions in the structure of glutamine synthetase. 1986 Sep 25-Oct 1Nature. 323(6086):304–309. doi: 10.1038/323304a0. [DOI] [PubMed] [Google Scholar]
- Barnes E. M., Jr, Zimniak P., Jayakumar A. Role of glutamine synthetase in the uptake and metabolism of methylammonium by Azotobacter vinelandii. J Bacteriol. 1983 Nov;156(2):752–757. doi: 10.1128/jb.156.2.752-757.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop P. E., Brill W. J. Genetic analysis of Azotobacter vinelandii mutant strains unable to fix nitrogen. J Bacteriol. 1977 May;130(2):954–956. doi: 10.1128/jb.130.2.954-956.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bozouklian H., Elmerich C. Nucleotide sequence of the Azospirillum brasilense Sp7 glutamine synthetase structural gene. Biochimie. 1986 Oct-Nov;68(10-11):1181–1187. doi: 10.1016/s0300-9084(86)80062-1. [DOI] [PubMed] [Google Scholar]
- Bozouklian H., Fogher C., Elmerich C. Cloning and characterization of the glnA gene of Azospirillum brasilense Sp7. Ann Inst Pasteur Microbiol. 1986 Jul-Aug;137B(1):3–18. doi: 10.1016/s0769-2609(86)80089-8. [DOI] [PubMed] [Google Scholar]
- Cardy D. L., Murrell J. C. Cloning, sequencing and expression of the glutamine synthetase structural gene (glnA) from the obligate methanotroph Methylococcus capsulatus (Bath). J Gen Microbiol. 1990 Feb;136(2):343–352. doi: 10.1099/00221287-136-2-343. [DOI] [PubMed] [Google Scholar]
- Carlson T. A., Martin G. B., Chelm B. K. Differential transcription of the two glutamine synthetase genes of Bradyrhizobium japonicum. J Bacteriol. 1987 Dec;169(12):5861–5866. doi: 10.1128/jb.169.12.5861-5866.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colonna-Romano S., Riccio A., Guida M., Defez R., Lamberti A., Iaccarino M., Arnold W., Priefer U., Pühler A. Tight linkage of glnA and a putative regulatory gene in Rhizobium leguminosarum. Nucleic Acids Res. 1987 Mar 11;15(5):1951–1964. doi: 10.1093/nar/15.5.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Contreras A, Casadesús J. Tn10 mutagenesis in Azotobacter vinelandii. Mol Gen Genet. 1987 Sep;209(2):276–282. doi: 10.1007/BF00329654. [DOI] [PubMed] [Google Scholar]
- Darrow R. A., Knotts R. R. Two forms of glutamine synthetase in free-living root-nodule bacteria. Biochem Biophys Res Commun. 1977 Sep 23;78(2):554–559. doi: 10.1016/0006-291x(77)90214-5. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. "A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity". Addendum. Anal Biochem. 1984 Feb;137(1):266–267. doi: 10.1016/0003-2697(84)90381-6. [DOI] [PubMed] [Google Scholar]
- Filser D. M., Moscatelli C., Lamberti A., Vincze E., Guida M., Salzano G., Iaccarino M. Characterization and cloning of two Rhizobium leguminosarum genes coding for glutamine synthetase activities. J Gen Microbiol. 1986 Sep;132(9):2561–2569. doi: 10.1099/00221287-132-9-2561. [DOI] [PubMed] [Google Scholar]
- Fuchs R. L., Keister D. L. Comparative properties of glutamine synthetases I and II in Rhizobium and Agrobacterium spp. J Bacteriol. 1980 Nov;144(2):641–648. doi: 10.1128/jb.144.2.641-648.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawley D. K., McClure W. R. Compilation and analysis of Escherichia coli promoter DNA sequences. Nucleic Acids Res. 1983 Apr 25;11(8):2237–2255. doi: 10.1093/nar/11.8.2237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobson M. R., Brigle K. E., Bennett L. T., Setterquist R. A., Wilson M. S., Cash V. L., Beynon J., Newton W. E., Dean D. R. Physical and genetic map of the major nif gene cluster from Azotobacter vinelandii. J Bacteriol. 1989 Feb;171(2):1017–1027. doi: 10.1128/jb.171.2.1017-1027.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janson C. A., Kayne P. S., Almassy R. J., Grunstein M., Eisenberg D. Sequence of glutamine synthetase from Salmonella typhimurium and implications for the protein structure. Gene. 1986;46(2-3):297–300. doi: 10.1016/0378-1119(86)90415-4. [DOI] [PubMed] [Google Scholar]
- Janssen P. J., Jones W. A., Jones D. T., Woods D. R. Molecular analysis and regulation of the glnA gene of the gram-positive anaerobe Clostridium acetobutylicum. J Bacteriol. 1988 Jan;170(1):400–408. doi: 10.1128/jb.170.1.400-408.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayakumar A., Barnes E. M., Jr The role of glutamine in regulation of ammonium transport in Azotobacter vinelandii. Arch Biochem Biophys. 1984 May 15;231(1):95–101. doi: 10.1016/0003-9861(84)90366-7. [DOI] [PubMed] [Google Scholar]
- Kennedy C., Eady R. R., Kondorosi E., Rekosh D. K. The molybdenum--iron protein of Klebsiella pneumoniae nitrogenase. Evidence for non-identical subunits from peptide 'mapping'. Biochem J. 1976 May 1;155(2):383–389. doi: 10.1042/bj1550383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy C., Toukdarian A. Genetics of azotobacters: applications to nitrogen fixation and related aspects of metabolism. Annu Rev Microbiol. 1987;41:227–258. doi: 10.1146/annurev.mi.41.100187.001303. [DOI] [PubMed] [Google Scholar]
- Kleiner D., Paul W., Merrick M. J. Construction of multicopy expression vectors for regulated over-production of proteins in Klebsiella pneumoniae and other enteric bacteria. J Gen Microbiol. 1988 Jul;134(7):1779–1784. doi: 10.1099/00221287-134-7-1779. [DOI] [PubMed] [Google Scholar]
- Kleinschmidt J. A., Kleiner D. The glutamine synthetase from Azotobacter vinelandii: purification, characterization, regulation and localization. Eur J Biochem. 1978 Aug 15;89(1):51–60. doi: 10.1111/j.1432-1033.1978.tb20895.x. [DOI] [PubMed] [Google Scholar]
- Krakow J. S. On the role of sulfhydryl groups in the structure and function of the Azotobacter vinelandii RNA polymerase. Biochemistry. 1975 Oct 7;14(20):4522–4527. doi: 10.1021/bi00691a029. [DOI] [PubMed] [Google Scholar]
- MacNeil D. General method, using Mu-Mud1 dilysogens, to determine the direction of transcription of and generate deletions in the glnA region of Escherichia coli. J Bacteriol. 1981 Apr;146(1):260–268. doi: 10.1128/jb.146.1.260-268.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miranda-Ríos J., Sánchez-Pescador R., Urdea M., Covarrubias A. A. The complete nucleotide sequence of the glnALG operon of Escherichia coli K12. Nucleic Acids Res. 1987 Mar 25;15(6):2757–2770. doi: 10.1093/nar/15.6.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NEWTON J. W., WILSON P. W., BURRIS R. H. Direct demonstration of ammonia as an intermediate in nitrogen fixation by Azotobacter. J Biol Chem. 1953 Sep;204(1):445–451. [PubMed] [Google Scholar]
- Rawlings D. E., Jones W. A., O'Neill E. G., Woods D. R. Nucleotide sequence of the glutamine synthetase gene and its controlling region from the acidophilic autotroph Thiobacillus ferrooxidans. Gene. 1987;53(2-3):211–217. doi: 10.1016/0378-1119(87)90009-6. [DOI] [PubMed] [Google Scholar]
- Robson R. L., Chesshyre J. A., Wheeler C., Jones R., Woodley P. R., Postgate J. R. Genome size and complexity in Azotobacter chroococcum. J Gen Microbiol. 1984 Jul;130(7):1603–1612. doi: 10.1099/00221287-130-7-1603. [DOI] [PubMed] [Google Scholar]
- Sadoff H. L., Shimel B., Ellis S. Characterization of Azotobacter vinelandii deoxyribonucleic acid and folded chromosomes. J Bacteriol. 1979 Jun;138(3):871–877. doi: 10.1128/jb.138.3.871-877.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santero E., Luque F., Medina J. R., Tortolero M. Isolation of ntrA-like mutants of Azotobacter vinelandii. J Bacteriol. 1986 May;166(2):541–544. doi: 10.1128/jb.166.2.541-544.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro B. M., Stadtman E. R. 5'-adenylyl-O-tyrosine. The novel phosphodiester residue of adenylylated glutamine synthetase from Escherichia coli. J Biol Chem. 1968 Jul 10;243(13):3769–3771. [PubMed] [Google Scholar]
- Shine J., Dalgarno L. Determinant of cistron specificity in bacterial ribosomes. Nature. 1975 Mar 6;254(5495):34–38. doi: 10.1038/254034a0. [DOI] [PubMed] [Google Scholar]
- Siedel J., Shelton E. Purification and properties of Azotobacter vinelandii glutamine synthetase. Arch Biochem Biophys. 1979 Jan;192(1):214–224. doi: 10.1016/0003-9861(79)90086-9. [DOI] [PubMed] [Google Scholar]
- Simon R., Quandt J., Klipp W. New derivatives of transposon Tn5 suitable for mobilization of replicons, generation of operon fusions and induction of genes in gram-negative bacteria. Gene. 1989 Aug 1;80(1):161–169. doi: 10.1016/0378-1119(89)90262-x. [DOI] [PubMed] [Google Scholar]
- Strauch M. A., Aronson A. I., Brown S. W., Schreier H. J., Sonenhein A. L. Sequence of the Bacillus subtilis glutamine synthetase gene region. Gene. 1988 Nov 30;71(2):257–265. doi: 10.1016/0378-1119(88)90042-x. [DOI] [PubMed] [Google Scholar]
- Tinoco I., Jr, Borer P. N., Dengler B., Levin M. D., Uhlenbeck O. C., Crothers D. M., Bralla J. Improved estimation of secondary structure in ribonucleic acids. Nat New Biol. 1973 Nov 14;246(150):40–41. doi: 10.1038/newbio246040a0. [DOI] [PubMed] [Google Scholar]
- Toukdarian A., Kennedy C. Regulation of nitrogen metabolism in Azotobacter vinelandii: isolation of ntr and glnA genes and construction of ntr mutants. EMBO J. 1986 Feb;5(2):399–407. doi: 10.1002/j.1460-2075.1986.tb04225.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene. 1982 Oct;19(3):259–268. doi: 10.1016/0378-1119(82)90015-4. [DOI] [PubMed] [Google Scholar]
- Willis R. C., Iwata K. K., Furlong C. E. Regulation of Glutamine Transport in Escherichia coli. J Bacteriol. 1975 Jun;122(3):1032–1037. doi: 10.1128/jb.122.3.1032-1037.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray L. V., Jr, Fisher S. H. Cloning and nucleotide sequence of the Streptomyces coelicolor gene encoding glutamine synthetase. Gene. 1988 Nov 30;71(2):247–256. doi: 10.1016/0378-1119(88)90041-8. [DOI] [PubMed] [Google Scholar]