Abstract
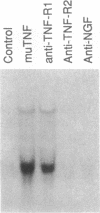
The individual roles of the murine type 1 and type 2 tumor necrosis factor (TNF) receptors (TNF-R1 and TNF-R2) were investigated utilizing (i) the strong species specificity of TNF-R2 for murine TNF compared to human TNF and (ii) agonistic rabbit polyclonal antibodies directed against the individual TNF receptors. Proliferation of mouse thymocytes and the murine cytotoxic T-cell line CT-6 is stimulated by murine TNF but not by human TNF. Consistent with this observation, polyclonal antibodies directed against TNF-R2 induced proliferation in both of these cell types, whereas polyclonal antibodies directed against TNF-R1 had no effect. In contrast, cytotoxicity in murine LM cells (which are sensitive to murine and human TNF) was induced by antibodies against TNF-R1 but not by antibodies against TNF-R2. Also, the steady-state level of manganous superoxide dismutase mRNA in the murine NIH 3T3 cell line was induced by murine TNF, human TNF, and anti-TNF-R1 but not by anti-TNF-R2. These results suggest that TNF-R2 initiates signals for the proliferation of thymocytes and cytotoxic T cells, whereas TNF-R1 initiates signals for cytotoxicity and the induction of the protective activity, manganous superoxide dismutase. The nonredundant signaling observed for the two TNF receptors cannot be explained simply by the differential expression of the two TNF receptors in the various cell types, because LM cells express on their surface higher levels of TNF-R2 than TNF-R1, and LM cells, NIH 3T3 cells, and thymus cells all express mRNA corresponding to both receptor types. It is therefore likely that the two receptors initiate distinct signaling pathways that result in the induction of different cellular responses.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baglioni C., McCandless S., Tavernier J., Fiers W. Binding of human tumor necrosis factor to high affinity receptors on HeLa and lymphoblastoid cells sensitive to growth inhibition. J Biol Chem. 1985 Nov 5;260(25):13395–13397. [PubMed] [Google Scholar]
- Beutler B., Cerami A. Tumor necrosis, cachexia, shock, and inflammation: a common mediator. Annu Rev Biochem. 1988;57:505–518. doi: 10.1146/annurev.bi.57.070188.002445. [DOI] [PubMed] [Google Scholar]
- Brockhaus M., Schoenfeld H. J., Schlaeger E. J., Hunziker W., Lesslauer W., Loetscher H. Identification of two types of tumor necrosis factor receptors on human cell lines by monoclonal antibodies. Proc Natl Acad Sci U S A. 1990 Apr;87(8):3127–3131. doi: 10.1073/pnas.87.8.3127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carswell E. A., Old L. J., Kassel R. L., Green S., Fiore N., Williamson B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3666–3670. doi: 10.1073/pnas.72.9.3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creasey A. A., Yamamoto R., Vitt C. R. A high molecular weight component of the human tumor necrosis factor receptor is associated with cytotoxicity. Proc Natl Acad Sci U S A. 1987 May;84(10):3293–3297. doi: 10.1073/pnas.84.10.3293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decker T., Lohmann-Matthes M. L., Gifford G. E. Cell-associated tumor necrosis factor (TNF) as a killing mechanism of activated cytotoxic macrophages. J Immunol. 1987 Feb 1;138(3):957–962. [PubMed] [Google Scholar]
- Ehrke M. J., Ho R. L., Hori K. Species-specific TNF induction of thymocyte proliferation. Cancer Immunol Immunother. 1988;27(2):103–108. doi: 10.1007/BF00200012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelmann H., Holtmann H., Brakebusch C., Avni Y. S., Sarov I., Nophar Y., Hadas E., Leitner O., Wallach D. Antibodies to a soluble form of a tumor necrosis factor (TNF) receptor have TNF-like activity. J Biol Chem. 1990 Aug 25;265(24):14497–14504. [PubMed] [Google Scholar]
- Engelmann H., Novick D., Wallach D. Two tumor necrosis factor-binding proteins purified from human urine. Evidence for immunological cross-reactivity with cell surface tumor necrosis factor receptors. J Biol Chem. 1990 Jan 25;265(3):1531–1536. [PubMed] [Google Scholar]
- Espevik T., Brockhaus M., Loetscher H., Nonstad U., Shalaby R. Characterization of binding and biological effects of monoclonal antibodies against a human tumor necrosis factor receptor. J Exp Med. 1990 Feb 1;171(2):415–426. doi: 10.1084/jem.171.2.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goeddel D. V., Aggarwal B. B., Gray P. W., Leung D. W., Nedwin G. E., Palladino M. A., Patton J. S., Pennica D., Shepard H. M., Sugarman B. J. Tumor necrosis factors: gene structure and biological activities. Cold Spring Harb Symp Quant Biol. 1986;51(Pt 1):597–609. doi: 10.1101/sqb.1986.051.01.072. [DOI] [PubMed] [Google Scholar]
- Hohmann H. P., Brockhaus M., Baeuerle P. A., Remy R., Kolbeck R., van Loon A. P. Expression of the types A and B tumor necrosis factor (TNF) receptors is independently regulated, and both receptors mediate activation of the transcription factor NF-kappa B. TNF alpha is not needed for induction of a biological effect via TNF receptors. J Biol Chem. 1990 Dec 25;265(36):22409–22417. [PubMed] [Google Scholar]
- Hohmann H. P., Remy R., Brockhaus M., van Loon A. P. Two different cell types have different major receptors for human tumor necrosis factor (TNF alpha). J Biol Chem. 1989 Sep 5;264(25):14927–14934. [PubMed] [Google Scholar]
- Kramer S. M., Carver M. E. Serum-free in vitro bioassay for the detection of tumor necrosis factor. J Immunol Methods. 1986 Nov 6;93(2):201–206. doi: 10.1016/0022-1759(86)90189-4. [DOI] [PubMed] [Google Scholar]
- Kull F. C., Jr, Jacobs S., Cuatrecasas P. Cellular receptor for 125I-labeled tumor necrosis factor: specific binding, affinity labeling, and relationship to sensitivity. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5756–5760. doi: 10.1073/pnas.82.17.5756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis M., Tartaglia L. A., Lee A., Bennett G. L., Rice G. C., Wong G. H., Chen E. Y., Goeddel D. V. Cloning and expression of cDNAs for two distinct murine tumor necrosis factor receptors demonstrate one receptor is species specific. Proc Natl Acad Sci U S A. 1991 Apr 1;88(7):2830–2834. doi: 10.1073/pnas.88.7.2830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loetscher H., Pan Y. C., Lahm H. W., Gentz R., Brockhaus M., Tabuchi H., Lesslauer W. Molecular cloning and expression of the human 55 kd tumor necrosis factor receptor. Cell. 1990 Apr 20;61(2):351–359. doi: 10.1016/0092-8674(90)90815-v. [DOI] [PubMed] [Google Scholar]
- Naume B., Shalaby R., Lesslauer W., Espevik T. Involvement of the 55- and 75-kDa tumor necrosis factor receptors in the generation of lymphokine-activated killer cell activity and proliferation of natural killer cells. J Immunol. 1991 May 1;146(9):3045–3048. [PubMed] [Google Scholar]
- Old L. J. Tumor necrosis factor. Sci Am. 1988 May;258(5):59-60, 69-75. doi: 10.1038/scientificamerican0588-59. [DOI] [PubMed] [Google Scholar]
- Ranges G. E., Bombara M. P., Aiyer R. A., Rice G. G., Palladino M. A., Jr Tumor necrosis factor-alpha as a proliferative signal for an IL-2-dependent T cell line: strict species specificity of action. J Immunol. 1989 Feb 15;142(4):1203–1208. [PubMed] [Google Scholar]
- Ranges G. E., Zlotnik A., Espevik T., Dinarello C. A., Cerami A., Palladino M. A., Jr Tumor necrosis factor alpha/cachectin is a growth factor for thymocytes. Synergistic interactions with other cytokines. J Exp Med. 1988 Apr 1;167(4):1472–1478. doi: 10.1084/jem.167.4.1472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schall T. J., Lewis M., Koller K. J., Lee A., Rice G. C., Wong G. H., Gatanaga T., Granger G. A., Lentz R., Raab H. Molecular cloning and expression of a receptor for human tumor necrosis factor. Cell. 1990 Apr 20;61(2):361–370. doi: 10.1016/0092-8674(90)90816-w. [DOI] [PubMed] [Google Scholar]
- Shalaby M. R., Sundan A., Loetscher H., Brockhaus M., Lesslauer W., Espevik T. Binding and regulation of cellular functions by monoclonal antibodies against human tumor necrosis factor receptors. J Exp Med. 1990 Nov 1;172(5):1517–1520. doi: 10.1084/jem.172.5.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith C. A., Davis T., Anderson D., Solam L., Beckmann M. P., Jerzy R., Dower S. K., Cosman D., Goodwin R. G. A receptor for tumor necrosis factor defines an unusual family of cellular and viral proteins. Science. 1990 May 25;248(4958):1019–1023. doi: 10.1126/science.2160731. [DOI] [PubMed] [Google Scholar]
- Spriggs D. R., Imamura K., Rodriguez C., Sariban E., Kufe D. W. Tumor necrosis factor expression in human epithelial tumor cell lines. J Clin Invest. 1988 Feb;81(2):455–460. doi: 10.1172/JCI113341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugarman B. J., Aggarwal B. B., Hass P. E., Figari I. S., Palladino M. A., Jr, Shepard H. M. Recombinant human tumor necrosis factor-alpha: effects on proliferation of normal and transformed cells in vitro. Science. 1985 Nov 22;230(4728):943–945. doi: 10.1126/science.3933111. [DOI] [PubMed] [Google Scholar]
- Tsujimoto M., Yip Y. K., Vilcek J. Tumor necrosis factor: specific binding and internalization in sensitive and resistant cells. Proc Natl Acad Sci U S A. 1985 Nov;82(22):7626–7630. doi: 10.1073/pnas.82.22.7626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner M., Londei M., Feldmann M. Human T cells from autoimmune and normal individuals can produce tumor necrosis factor. Eur J Immunol. 1987 Dec;17(12):1807–1814. doi: 10.1002/eji.1830171220. [DOI] [PubMed] [Google Scholar]
- Wong G. H., Elwell J. H., Oberley L. W., Goeddel D. V. Manganous superoxide dismutase is essential for cellular resistance to cytotoxicity of tumor necrosis factor. Cell. 1989 Sep 8;58(5):923–931. doi: 10.1016/0092-8674(89)90944-6. [DOI] [PubMed] [Google Scholar]
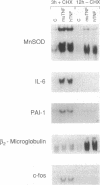
- Wong G. H., Goeddel D. V. Induction of manganous superoxide dismutase by tumor necrosis factor: possible protective mechanism. Science. 1988 Nov 11;242(4880):941–944. doi: 10.1126/science.3263703. [DOI] [PubMed] [Google Scholar]