Abstract
Background
Fruits and vegetables are rich in compounds with proposed antioxidant, anti-allergic and anti-inflammatory properties, which could contribute to reduce the prevalence of asthma and allergic diseases.
Objective
We investigated the association between asthma, and chronic rhino-sinusitis (CRS) with intake of fruits and vegetables in European adults.
Methods
A stratified random sample was drawn from the Global Allergy and Asthma Network of Excellence (GA2LEN) screening survey, in which 55,000 adults aged 15–75 answered a questionnaire on respiratory symptoms. Asthma score (derived from self-reported asthma symptoms) and CRS were the outcomes of interest. Dietary intake of 22 subgroups of fruits and vegetables was ascertained using the internationally validated GA2LEN Food Frequency Questionnaire. Adjusted associations were examined with negative binomial and multiple regressions. Simes procedure was used to control for multiple testing.
Results
A total of 3206 individuals had valid data on asthma and dietary exposures of interest. 22.8% reported having at least 1 asthma symptom (asthma score ≥1), whilst 19.5% had CRS. After adjustment for potential confounders, asthma score was negatively associated with intake of dried fruits (β-coefficient −2.34; 95% confidence interval [CI] −4.09, −0.59), whilst CRS was statistically negatively associated with total intake of fruits (OR 0.73; 95% CI 0.55, 0.97). Conversely, a positive association was observed between asthma score and alliums vegetables (adjusted β-coefficient 0.23; 95% CI 0.06, 0.40). None of these associations remained statistically significant after controlling for multiple testing.
Conclusion and clinical relevance
There was no consistent evidence for an association of asthma or CRS with fruit and vegetable intake in this representative sample of European adults.
Keywords: Fruits, Vegetables, Asthma, Chronic rhino-sinusitis, Adults, Europe, Meta-analysis, GA2LEN
Background
Fruits and vegetables are rich sources of nutrients and compounds with antioxidant, anti-allergic and anti-inflammatory properties, which could modulate the expression of asthma and allergic diseases [1]. A recent systematic review suggested an overall reduced risk of wheeze or self-reported Dr diagnosed asthma in adults and children with higher intakes of fruits and vegetables [2]. Several observational studies in adults have shown a negative association between various asthma prevalence outcomes, and intake of apples [3], citrus fruits [4], tomatoes or leafy vegetables [4]. Smaller studies in asthmatic adults with a dietary pattern mainly comprised of fruits and vegetables have also been shown to have a lower risk of severe asthma [2]. The current evidence on a possible protective effect of fruits and vegetables on allergic diseases is mixed, with some studies showing a negative association between intake of vegetables [5] or food groups that contain them [6] and a lower asthma prevalence, whilst several population-based studies have reported no association between allergic symptoms and fruits or vegetables when measured individually [7, 8] or as part of a dietary pattern [9, 10].
Epidemiological studies use different operational definitions to assess asthma, as well as different instruments to ascertain usual dietary intake. These issues may make it more difficult to ascribe a consistent interpretation on their relationship. The current observational evidence in European adults is inconclusive, with very few multi-national studies examining in some standardised fashion, the association between asthma and diet [10]. Within the Global Allergy and Asthma Network of Excellence (GA2LEN), we designed and piloted a single, common, food frequency questionnaire (FFQ) [11], which was used to estimate usual dietary intake of over 3500 adults from 10 European countries participating in the GA2LEN Follow-up survey. In this analysis, we investigate the cross-sectional association between asthma and chronic rhino-sinusitis (CRS), with dietary intake of fruits and vegetables in these adults.
Methods
The GA2LEN study—screening and clinical surveys
The core protocol for the GA2LEN survey required 18 European participating centres to identify a random sample of at least 3000 adults aged 15–74 years from an available population-based sampling frame. A stratified random sample was drawn, in which 55,000 adults aged 15–75 answered a questionnaire on respiratory symptoms. The following countries (and cities) were included in this cross-sectional analysis: Belgium (Ghent), Denmark (Odense), Finland (Helsinki), Germany (Duisberg, Brandenburg), The Netherlands (Amsterdam), Poland (Krakow, Lodz, Katowice), Portugal (Coimbra), Sweden (Gothenburg, Stockholm, Umea, Uppsala), and the UK (Southampton, London). In 2008–2009, potential participants were sent a short questionnaire by mail, and at least three attempts were made to elicit a response [12]. The questionnaire collected information on age, gender, smoking and the presence of symptoms of asthma (including age of onset), and CRS. Four sub-samples were selected to define cases and controls: (1) those with self-reported asthma and at least one respiratory symptom reported in the last 12 months (‘asthma’), (2) those having chronic sinusitis (defined following the EP3OS criteria, that is, the presence of at least two of the following symptoms for at least 12 weeks in the past year: (i) nasal blockage, (ii) nasal discharge, (iii) facial pain or pressure or (iv) reduction in sense of smell with at least one of the symptoms being nasal blockage or nasal discharge), (3) those who had both ‘asthma’ and ‘chronic sinusitis’, and those who had none of these conditions. [13] Five questions on symptoms in the last 12 months (breathless when wheezing, woken with tightness in chest, shortness of breath while at rest, shortness of breath after exercise, woken by shortness of breath) were used to construct an asthma symptom score on a five-point scale [14].
Dietary intake
The GA2LEN food frequency questionnaire (FFQ) was designed to assess usual dietary intake across countries, using a single, common, and standardised instrument. The FFQ was validated in a random sample of adults from 5 participant centres in GA2LEN, namely Finland, Portugal, Germany, Greece, and Poland, each representing a different European Region [11]. All centres adhered to the same standard operational procedure (SOP) to translate the questionnaires and the same procedure was used to translate and standardise all other questionnaires in the GA2LEN survey. The GA2LEN FFQ has been translated into more than 25 languages for use in several single and multi-national epidemiological studies [15]. To facilitate international food comparisons, the FFQ was organised into 32 sections of food groups [16]. The FFQ collected data on a wide range of foods, including 43 vegetables and 25 fruits (Table 1). Total energy intake (TEI) was calculated using the latest available food composition estimates from the British Food Composition Table [17].
Table 1.
Fruit and vegetable subgroup classification in the GA2LEN Follow-up study
| Food group | Food items included |
|---|---|
| Vegetables | |
| Leafy vegetables | Lettuce, spinach, chard, fenugreek, wild greens |
| Fruit vegetables | Capers, tomatoes, aubergine, courgette, sweet peppers, pumpkin, artichoke, okra, mushroom |
| Cucurbitacea | Cucumber, melon, watermelon, bitter melon |
| Apiaceae | Celery, carrot, herbs (coriander, parsley, chervil, dill), parsnip |
| Other root vegetables | Turnip or swede, radish, beetroot, ginger, taro |
| Maiz/Corn | Sweet corn |
| Alliums | Onion, garlic, leek |
| Brassicaceae | Brussels sprouts, broccoli, cabbage, cauliflower, coleslaw |
| Potatoes | Mashed potatoes, baked/roasted/casserole potatoes, chips/french fries, potatoes in salad, potato dumping/bread dumpling/gnocchi, potato tortilla |
| Pickled vegetables | Cucumber, radish, cabbage |
| All vegetables | Average intake of all above |
| Fruits | |
| Hard fruits | Apple, pear |
| Citrus fruits | Lemon, orange, mandarin/tangerine, grape-fruit, kiwi |
| Oily fruits | Olives, avocado |
| Fruit juice | Freshly squeezed fruits |
| Berries | Blueberries, strawberries, raspberries (‘forest berries’) |
| Nectarines | Nectarine, apricot, peach |
| Dried fruits | Raisin, prune |
| Tropical fruits | Mango, pineapple (banana assessed individually) |
| Canned fruits | Any canned fruits |
| Dark pigmented fruit | Cherries, rhubarb, grape, fig, plum |
| All fruits | Average intake of all above |
Statistical analyses
Sampling probability weights were used to standardise prevalences by gender and age to a European Standard Population.
Multivariable logistic regression was used to assess the relationship between food consumption and CRS within each country, controlling for education, employment, smoking status (never, ex-smoker, current smoker), BMI, age, gender, supplement use and TEI. The country level logistic analyses were weighted to take into account the case–control sampling selection. Negative binomial regression was used to assess the relationship between food consumption and asthma score within each country. This analysis controlled for the same variables and used the same sampling weights as in the logistic regression described above. There was only weak collinearity between the variables when we tested this in each of the multivariable models. The regression coefficients from the country level analyses were meta-analysed to give an overall coefficient. The I2 statistic was used to assess heterogeneity between countries. Simes procedure was used to correct statistical estimates derived from multiple testing [18].
All analyses were run using Stata 13.1 (StataCorp, 4905 Lakeway Drive, College Station, Texas 77845 USA).
Results
The main characteristics of the 3202 participants with valid data on diet and asthma score are summarised in Table 2. Of these, 22.8% reported having at least 1 symptom of asthma (asthma score = 1) whereas 9.3% had 3 or more symptoms. CRS was reported by 23.4% of individuals. Over half of all participants reported eating fruits or vegetables 5 times a week, with Portugal and Poland having the highest intake of these food groups.
Table 2.
General characteristics of the study population (based on individuals with complete data on dietary exposures and asthma score)
| Variables | Countries | |||||
|---|---|---|---|---|---|---|
| Denmark | Finland | Sweden | United Kingdom | Germany | The Netherlands | |
| Odense (359) | Helsinki (160) | Total (1261) | Total (173) | Total (376) | Amsterdam (215) | |
| Age, years; mean (SD) | 48.1 (14.5) | 46.8 (15.1) | 45.7 (15.1) | 51.6 (13.2) | 48.8 (15.6) | 52.6 (13.9) |
| Males, n (%) | 162 (45.1) | 62 (38.8) | 556 (44.1) | 70 (40.5) | 152 (403) | 111 (51.6) |
| BMI (kg/m2) | 27.4 (14.8) | 26.5 (4.6) | 25.9 (7.2) | 27.1 (5.6) | 26.3 (4.8) | 25.7 (3.7) |
| Age at completing full-time education; years (SD) | 23.4 (5.5) | 23.5 (5.5) | 24.5 (7.7) | 18.1 (3.6) | 20.6 (5.2) | 20.2 (4.6) |
| Employment status | ||||||
| Employed | 188 (52.7) | 94 (58.9) | 737 (58.5) | 85 (49.7) | 196 (52.0) | 103 (47.9) |
| Retired | 82 (23.0) | 32 (20.0) | 199 (15.8) | 39 (22.8) | 88 (23.3) | 56 (21.1) |
| Unemployed | 11 (3.1) | 3 (1.9) | 38 (3.0) | 4 (2.3) | 12 (3.2) | 5 (2.3) |
| Other | 76 (21.5) | 31 (19.4) | 286 (22.7) | 43 (25.1) | 81 (21.5) | 51 (23.7) |
| Smoking | ||||||
| Never smokers | 155 (43.4) | 83 (51.9) | 672 (53.3) | 77 (44.5) | 183 (48.4) | 84 (39.1) |
| Ex-smokers | 102 (28.6) | 37 (23.1) | 428 (33.9) | 70 (40.5) | 131 (34.7) | 88 (40.9) |
| Current smokers | 100 (28.0) | 40 (25.0) | 162 (12.8) | 26 (15.0) | 64 (16.9) | 43 (20.0) |
| Asthma score; N (%) | ||||||
| 0 | 145 (40.4) | 96 (59.6) | 583 (46.2) | 66 (38.2) | 161 (42.6) | 100 (41.0) |
| 1 | 85 (23.7) | 31 (19.3) | 276 (21.9) | 37 (21.4) | 107 (28.3) | 40 (18.6) |
| 2 | 50 (13.9) | 15 (9.3) | 195 (15.5) | 22 (12.7) | 47 (12.4) | 37 (17.2) |
| 3 | 47 (13.1) | 10 (6.2) | 114 (9.0) | 17 (11.5) | 35 (9.3) | 23 (10.7) |
| 4 | 24 (6.7) | 7 (4.4) | 61 (4.8) | 26 (15.0) | 16 (4.2) | 12 (5.6) |
| 5 | 8 (2.2) | 2 (1.2) | 33 (2.6) | 5 (2.9) | 12 (3.2) | 3 (1.4) |
| Chronic rhino-sinusitis; n (%) | 63 (17.6) | 29 (17.8) | 234 (18.3) | 22 (12.6) | 62 (16.2) | 52 (23.9) |
| Asthma ever (n; %) | 115 (32.0) | 44 (27.0) | 510 (39.8) | 80 (45.7) | 83 (21.7) | 44 (20.2) |
| CRS only (n; %) | 42 (11.7) | 17 (10.4) | 102 (8.0) | 10 (5.7) | 38 (9.9) | 40 (18.4) |
| Both asthma ever and CRS (n; %) | 21 (5.9) | 12 (7.4) | 132 (10.3) | 12 (6.9) | 23 (6.0) | 12 (5.5) |
| Total Energy Intake (TEI) | 2577 (761) | 3197 (1140) | 3110 (978) | 2833 (889.6) | 2821 (1049) | 2817 (827) |
| Use of nutritional supplements, n (%) | 143 (40.4) | 70 (43.5) | 325 (26.0) | 58 (33.7) | 102 (27.1) | 88 (41.0) |
| % people eating fruits (all types) ≥5 times/week | 202 (56.4) | 93 (57.1) | 717 (56.0) | 101 (57.7) | 213 (55.8) | 114 (52.3) |
| % people eating total vegetables (all types) ≥5 times/week | 224 (62.4) | 128 (78.5) | 906 (70.7) | 92 (52.6) | 194 (50.7) | 78 (35.8) |
| Variables | Countries | |||
|---|---|---|---|---|
| Portugal | Belgium | Poland | Total | |
| Coimbra (266) | Ghent (148) | Total (244) | 3202 | |
| Age, years; mean (SD) | 47.1 (15.0) | 45.7 (15.1) | 49.7 (15.7) | 47.6 (15.1) |
| Males, n (%) | 93 (35.0) | 71 (48.0) | 104 (42.6) | 1381 (43.1) |
| BMI, kg/m2 (SD) | 25.9 (5.1) | 24.9 (4.4) | 27.4 (5.2) | 26.3 (5.2) |
| Age at completing full-time education; years (SD) | 20.1 (4.6) | 20.6 (6.6) | 20.4 (3.4) | 22.4 (6.6) |
| Employment status | ||||
| Employed | 140 (52.6) | 75 (51.0) | 89 (38.0) | 1707 (53.6) |
| Retired | 56 (26.5) | 26 (17.7) | 86 (36.8) | 664 (20.8) |
| Unemployed | 11 (4.1) | 3 (2.0) | 12 (5.1) | 99 (3.1) |
| Other | 59 (22.2) | 30 (22.4) | 47 (20.0) | 717 (22.5) |
| Smoking | ||||
| Never smokers | 172 (64.7) | 75 (50.7) | 111 (45.7) | 1612 (50.4) |
| Ex-smokers | 56 (21.1) | 45 (30.4) | 78 (32.1) | 1035 (32.2) |
| Current smokers | 38 (14.3) | 28 (18.9) | 54 (22.2) | 555 (17.3) |
| Asthma score | ||||
| 0 | 109 (41.0) | 57 (38.5) | 78 (32.0) | 1395 (43.5) |
| 1 | 49 (18.4) | 34 (23.0) | 73 (29.9) | 732 (22.8) |
| 2 | 41 (15.4) | 22 (14.9) | 34 (13.9) | 463 (14.4) |
| 3 | 27 (910.2) | 17 (11.5) | 28 (11.5) | 318 (9.9) |
| 4 | 23 (8.7) | 12 (8.1) | 17 (7.0) | 198 (6.2) |
| 5 | 17 (6.4) | 6 (4.1) | 14 (5.7) | 100 (3.1) |
| Chronic rhino-sinusitis; n (%) | 78 (29.2) | 43 (29.1) | 50 (20.2) | 633 (19.5) |
| Asthma ever (n; %) | 59 (22.1) | 23 (15.5) | 37 (15.0) | 995 (30.7) |
| CRS only (n; %) | 44 (16.5) | 28 (18.9) | 39 (15.8) | 360 (11.1) |
| Both asthma ever and CRS (n; %) | 34 (12.7) | 15 (10.1) | 11 (4.5) | 272 (8.4) |
| Total Energy Intake (TEI); mean (SD) | 3195 (1296) | 2937 (885) | 3211 (1661) | 2993 (1072) |
| Use of nutritional supplements, n (%) | 16 (6.0) | 50 (33.8) | 53 (22.0) | 905 (28.4) |
| % people eating fruits (all types) ≥5 times/week | 189 (70.8) | 80 (54.1) | 158 (64.0) | 1867 (57.6) |
| % people eating total vegetables (all types) ≥5 times/week | 206 (77.2) | 77 (52.0) | 182 (73.7) | 2087 (64.4) |
The association between asthma score and fruit and vegetable intake is illustrated in Table 3. After controlling for potential confounders, a statistically significant negative association was observed between having an increasing asthma score and eating dried fruits (β-coefficient −2.34; 95% CI −4.09, −0.59; P value = 0.009). No other fruit groups were associated with asthma. Intake of fruity vegetables (which included capers, tomatoes, aubergine, courgette, sweet peppers, pumpkin, artichoke, okra, and mushroom) was positively associated with asthma score (β-coefficient 0.17; 95% CI 0.04, 0.30). Similarly, a higher asthma score was related to intake of alliums vegetables (onion, garlic, leek) (β-coefficient 0.23; 95% CI 0.06, 0.40). Figure 1 illustrates the per-country associations between asthma score and total fruit intake and fruity vegetables. There was no heterogeneity across countries (I2 = 0%).
Table 3.
Association between severity of asthma (asthma score) and fruit and vegetable intake in adults from GA2LEN
| Fruit and vegetable groups | Asthma score Effect size (β-coefficient (95% confidence intervals) |
|
|---|---|---|
| Unadjusted (n = 3206) | Adjusted (n = 2945) | |
| Fruits | ||
| Hard fruits | 0.01 (−0.11, 0.14) n = 3196 | −0.02 (0.15, 0.11) n = 2940 |
| Bananas | 0.03 (−0.14, 0.21) n = 3187 | 0.04 (−0.19, 0.27) n = 2934 |
| Citrus fruits | −0.05 (−0.19, 0.09) n = 3196 | −0.03 (−0.18, 0.12) n = 2938 |
| Oily fruits | 0.25 (0.02, 0.48) n = 3196 | 0.24 (0.01, 0.46) n = 2942 |
| Freshly squeezed fruit | 0.16 (−0.03, 0.36) n = 3184 | 0.18 (−0.01, 0.38) n = 2930 |
| Berries | −0.07 (−0.32, 0.19) n = 3159 | −0.12 (−0.37, 0.13) n = 2907 |
| Nectarines | 0.26 (−0.10, 0.62) n = 3197 | 0.16 (−0.33, 0.65) n = 2942 |
| Dried fruits | −1.89 (−3.36, −0.42) n = 3190 | −2.34 (−4.09, −0.59) n = 2937 |
| Tropical fruits | 0.13 (−0.31, 0.56) n = 3194 | 0.21 (−0.15, 0.55) n = 2940 |
| Canned fruits | −4.62 (−6.50, −2.74) n = 3181 | −5.66 (−11.4, 0.07) n = 2930 |
| Dark pigmented fruits | −0.11 (−0.41, 0.19) n = 3201 | −0.09 (−0.37, 0.19) n = 2944 |
| All fruits | −0.03 (−0.16, 0.10) n = 3203 | 0.04 (−0.09, 0.17) n = 2944 |
| Nuts | 0.21 (−0.12, 0.54) n = 3192 | 0.20 (−0.21, 0.61) n = 2935 |
| Vegetables | ||
| Leafy vegetables | 0.11 (−0.04, 0.26) n = 3195 | 0.03 (−0.15, 0.22) n = 2937 |
| Fruity vegetables | 0.16 (0.04, 0.28) n = 3202 | 0.17 (0.04, 0.30) n = 2942 |
| Cucurbitacea | 0.07 (−0.10, 0.24) n = 3202 | −0.02 (−0.22, 0.18) n = 2943 |
| Apiaceae | 0.05 (−0.12, 0.21) n = 3204 | 0.05 (−0.09, 0.19) n = 2943 |
| Other root vegetables | 0.13 (−0.08, 0.33) n = 3200 | 0.12 (−0.13, 0.37) n = 2942 |
| Maize/corn | 0.41 (−0.12, 0.93) n = 3189 | 0.47 (−0.04, 0.98) n = 2936 |
| Alliums | 0.27 (0.15, 0.39) n = 3203 | 0.23 (0.06, 0.40) n = 2944 |
| Brassicaceae | 0.30 (0.01. 0.59) n = 3202 | 0.20 (−0.02, 0.41) n = 2943 |
| Potatoes | 0.09 (−0.21, 0.38) n = 3194 | 0.002 (−0.24, 0.24) n = 2937 |
| Pickled vegetables | −2.32 (−4.17, −0.47) n = 3175 | −1.90 (−3.94, 0.14) n = 2924 |
| Legumes | −2.10 (−3.65, −0.45) n = 3196 | −1.98 (−4.13, 0.18) n = 2939 |
| All vegetables | 0.12 (−0.001, 0.25) n = 3206 | 0.11 (−0.03, 0.25) n = 2945 |
Italics indicate a statistically significant effect size
Fig. 1.
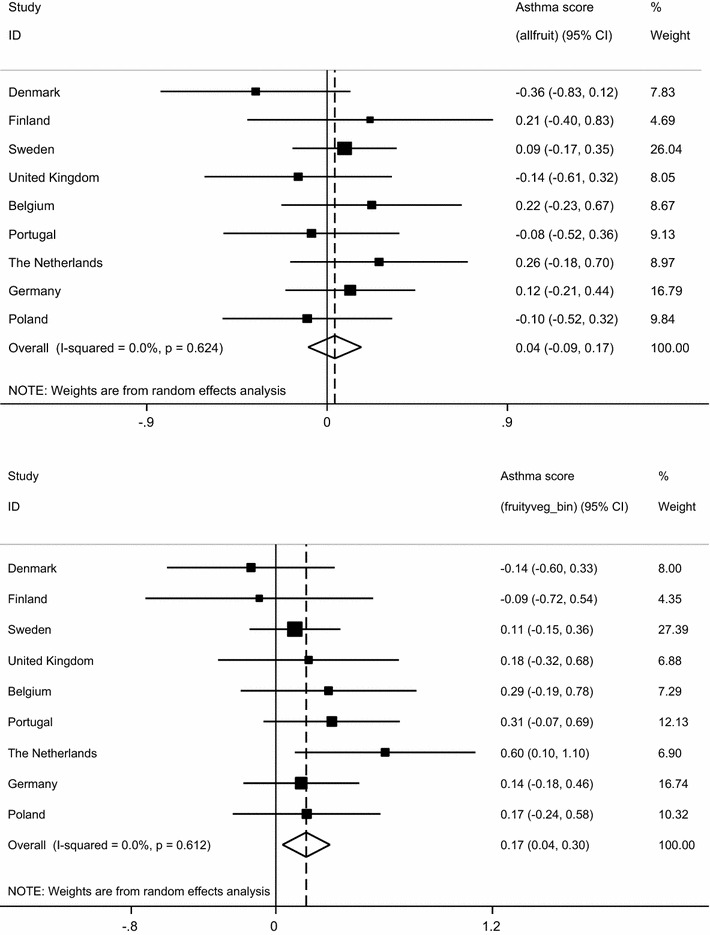
Weighted adjusted negative binomial regressions of asthma score association with total intake of fruits (top) and fruity vegetables (below) (per centre, and meta-analysis of pooled results)
Table 4 shows the associations found between CRS and fruit and vegetable intake. A 27% lower risk of disease was observed in those with a total intake of fruit ≥5 versus those who ate fruit below this cut-off point (OR 0.23; 95% CI 0.55, 0.97). As illustrated in Fig. 2, there was no evidence of heterogeneity between the estimates across countries (I2 = 0.0%; P value = 0.62).
Table 4.
Association between CRS and fruit and vegetable intake in adults from GA2LEN
| Fruit and vegetable groups | Effect size (odds ratio (95% confidence intervals) | |
|---|---|---|
| Unadjusted (n = 3242) | Adjusted (2970) | |
| Fruit group | ||
| Hard fruit | 0.83 (0.64–1.06) n = 3232 | 0.82 (0.62–1.09) n = 2965 |
| Bananas | 1.04 (0.78–1.40) n = 3223 | 0.99 (0.68–1.44) n = 2959 |
| Citrus fruit | 0.78 (0.48–1.26) n = 3232 | 0.87 (0.52–1.46) n = 2963 |
| Oily fruits | 1.40 (0.91–2.16) n = 3232 | 1.67 (0.91–3.06) n = 2967 |
| Freshly squeezed fruit | 0.73 (0.44–1.20) n = 3219 | 0.74 (0.44–1.24) n = 2954 |
| Berries | 1.08 (0.61–1.94) n = n = 3195 | 1.23 (0.55–2.76) n = 2932 |
| Nectarines | 1.42 (0.84–2.41) n = 3233 | 1.57 (0.79–3.11) n = 2967 |
| Dried fruits | 0.95 (0.42–2.14) n = 3226 | 0.98 (0.42–2.32) n = 2962 |
| Tropical fruits | 2.14 (1.10–4.16) n = 3230 | 2.50 (0.91–6.92) n = 2965 |
| Canned fruitsa | – | – |
| Dark pigmented fruits | 1.01 (0.71–1.45) n = 3237 | 1.11 (0.75–1.64) n = 2969 |
| All fruits | 0.75 (0.58–0.96) n = 3239 | 0.73 (0.55–0.97) n = 2969 |
| Nuts | 0.47 (0.21–1.06) n = 3227 | 0.64 (0.23–1.80) n = 2960 |
| Vegetables | ||
| Leafy vegetables | 1.15 (0.86–1.53) n = 3229 | 1.22 (0.86–1.71) n = 2961 |
| Fruity vegetables | 1.16 (0.87–1.53) n = 3237 | 1.22 (0.81–1.85) n = 2967 |
| Cucurbitacea | 1.15 (0.85–1.56) n = 3238 | 1.03 (0.73–1.44) n = 2968 |
| Apiaceae | 1.22 (0.93–1.62) n = 3239 | 1.22 (0.90–1.64) n = 2968 |
| Other root vegetables | 1.63 (0.98–2.70) n = 3235 | 1.77 (0.89–3.53) n = 2967 |
| Maize/corn | 1.64 (0.55–4.87) n = 3224 | 1.74 (0.42–7.22) n = 2961 |
| Alliums | 1.19 (0.91–1.55) n = 3238 | 0.99 (0.68–1.42) n = 2969 |
| Brassicaceae | 1.09 (0.73–1.62) n = 3237 | 1.05 (0.67–1.65) n = 2968 |
| Potatoes | 2.27 (1.47–3.52) n = 3229 | 1.82 (1.03–3.23) n = 2962 |
| Pickled vegetables | 1.73 (0.88–3.4) n = 3210 | 1.61 (0.72–3.59) n = 2949 |
| All vegetables | 1.11 (0.80–1.54) n = 3242 | 1.09 (0.67–1.77) n = 2970 |
| Legumes | 1.54 (0.51–4.64) n = 3231 | 1.24 (0.30–5.10) n = 2964 |
Italics indicate a statistically significant effect size
aNot enough people with data on this exposure to carry out analyses
Fig. 2.
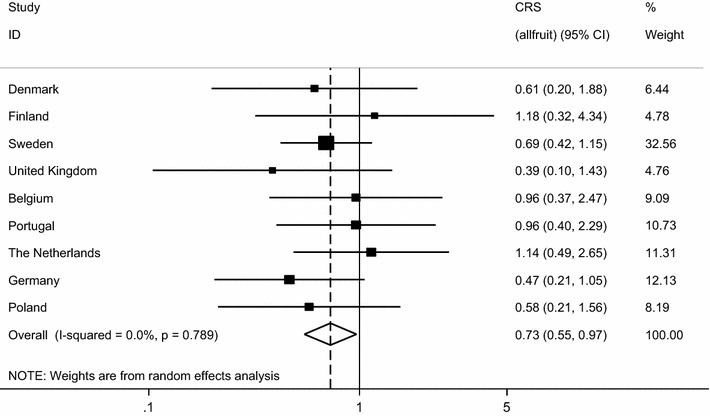
Weighted multivariable analyses of association between CRS with total intake of fruits (per centre, and meta-analysis of pooled results)
After applying Simes procedure, the statistical significance of the association between asthma score and dried fruits was attenuated (P value = 0.05), and all the other associations were no longer statistically significant (>0.15).
Discussion
In this multi-national study of adults participating in the GA2LEN Follow-up survey, asthma symptom score and CRS were negatively associated with dietary intake of dried fruits and total fruit intake, respectively. Asthma symptom score was also positively associated with a higher intake of fruity vegetables and alliums. These associations were observed after adjusting for several potential confounders, which included socio-economic, smoking, and lifestyle-related variables (including BMI, TEI, and nutritional supplement use). After controlling for multiple comparisons, the statistical significance of these associations was lost.
To our knowledge, this is the first multi-national population-based study to examine the association between asthma, CRS and allergic rhinitis, with fruit and vegetable intake, using a standardised method to ascertain both respiratory outcomes and dietary exposures. The results of this study were weighted to make results generalizable to the European adult population. We used an asthma score to ascertain individuals with a variety of symptoms, for its good predictability to ascertain outcomes related to asthma [14, 19]. Asthma is characterised for its clinical phenotypic heterogeneity and temporal phenotypic variability. Being a multi-categorical measure, the score provides more power to detect risk factors for asthma [19].
The GA2LEN FFQ was translated into each of the participant countries’ languages following international guidelines, and was previously piloted and validated in a subsample of 5 participating countries [11]. The FFQ uses a semi-quantitative approach to enquiring about the frequency of intake of 250 food items, which includes staple foods representative of each nation, but also foods that are commonly consumed in all these countries. The GA2LEN FFQ is being used in several other multi-national countries and appears to be a functional and accurate tool to ascertain usual dietary intake [15]. Given the large number of dietary exposure studied, we used Simes procedure to adjust the P values for multiple testing. This method has more power to identify true associations and its use is helpful when there are several highly correlated variables, as it is the case of dietary exposures [18].
The absence of robust evidence suggesting an association between dietary intake of fruits and vegetables with respiratory outcomes in this study has been confirmed in other population-based observational studies. Several authors have reported no association between asthma risk and intake of citrus fruits. As reported in other studies, we did not observe an association between the outcomes studies and citrus fruits [3, 20–22] nor with vitamin C, for which observational studies show mixed evidence of a beneficial effect [23].
We did find a negative association between dried fruit intake and asthma score, which remained statistically significant after controlling for multiple comparisons. Recent experimental evidence has demonstrated in an asthma-induced model in rats, that administering V. vinifera dried fruits inhibited the recruitment of inflammatory cytokines (IL)-4, IL-5, IL-1β, tumour necrosis factor, as well as IgE levels, and circulating levels of eosinophils in blood/serum and broncho-alveolar fluid [24]. Treatment with raisin extract also normalised lung function and histamine levels compared to control animals. Although no experimental evidence has demonstrated that prunes might exert similar effects, it has been proposed that the potential beneficial role of prunes on asthma might be mediated through their role in maintaining the gut microbiota balance [25]. Our findings of a negative association between dried fruits (raisins and prunes) might be explained at least partly by these biological mechanisms.
Several other studies have used a more integrative approach to elucidate the association between asthma and dietary exposures using dietary patterns, derived from Principal Component or Factor analysis, or through other indexes. However, dietary patterns that include fruits and vegetables as main food contributors have so far been unrelated to prevalence [9] or risk of adult asthma [26]. The uniformity of the associations observed per country in our study, and the absence of heterogeneity observed in most analyses, would lend further support to the notion that in general intakes of fruits and vegetables are not strongly associated with adult asthma.
Fruits and vegetables are also rich in various subclasses of flavonoids, for which strong anti-oxidant, anti-inflammatory and anti-allergic properties have been demonstrated in experimental studies of induced asthma [27]. These results have been echoed in some observational studies in adults showing a reduced risk of BHR [7] or asthma incidence [28], though others have reported no association with current asthma or allergic symptoms [29]. This is partly explained by the differences in the subclasses studied. In our study, we found some evidence that a lower risk of CRS was associated with a higher intake of fruits, which could partly be explained by the high content of vitamin C and flavonoids in them. We err on the cautious side though as this association was no longer statistically significant after controlling for multiple testing.
Due to the cross-sectional nature of our analysis, we cannot ascribe causality (or lack of) in the association between asthma, CRS, and allergic rhinitis with dietary intake of fruits and vegetables. Although we adjusted for several important potential confounders, there are likely to be other unmeasured confounders involved in the complex association between asthma and diet.
In conclusion, we found no consistent evidence for an association of asthma and allergic rhino-sinusitis with fruit and vegetable intake. The overall effect size observed for CRS and total fruit intake is suggestive of a protective effect, but this needs to be taken with caution given the multiple comparisons carried out in the study.
Authors’ contributions
VGL and PGB conceived the hypothesis for this analysis. VGL wrote the first draft of manuscript. VGL designed the GA2LEN FFQ which was used to collect data on dietary intake in the GA2LEN participants. PGB led the research efforts to make possible the international validation of the GA2LEN FFQ. RA and JFP contributed with statistical analyses. RA helped to interpret and classify the nutritional variables used in the study. All co-authors listed in the manuscript contributed to and approved the final version of the manuscript and led the research efforts to assess dietary intake in their centres. All authors read and approved the final manuscript.
Acknowledgements
We are indebted to the participants of the GA2LEN Follow-up survey across Europe.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
Data requests can be submitted to the GA2LEN Network of Excellence for the consideration and approval of the Scientific Steering Committee. Informal enquiries about data access and material availability can be sent to Dr Vanessa Garcia Larsen (v.garcialarsen@imperial.ac.uk). The Ga2len FFQ is free to use for academic and research purposes.
Consent for publication
All co-authors have read and approved the final version of the manuscript and gave their consent to publish it.
Ethics approval and consent to participate
All participant centres were granted ethical approval to take part in the GA2LEN Follow-up survey from which this analysis was done. Belgium: Committee for Medical Ethics, University of Ghent. Denmark: Den Videnskabsetiske Komite for Region Syddanmark. Finland: Helsingin Ja Uudenmaan Saira Anhoitopiirin Kuntayhtyma, Eetiset toimukunnat. Germany (both centres): Commission of Ethic, Faculty of Medicine, Heirich Heine Universitat Dusseldorf. Poland: Katowice: Biotethics Commision University of Katowice; Krakow (Commision of Bioethics University of Jagiellonskiego; Lodz (Committee of Bioethics University of Lodz. Portugal: Commission of Ethics for Health, Hospital of the University of Coimbra. Sweden (Joint ethical approval for all four participant centres) Karolinska Institute Ethics Committee. The Netherlands: Medical Ethics Committee, Academic Medical Centre, University of Amsterdam. United Kingdom: National Health Ethics Committee (NRES).
Funding
The GA2LEN study was supported by EU Framework programme for research; contract no. FOOD-CT-2004-506378.
Abbreviations
- GA2LEN
The Global Asthma and Allergy Network of Excellence
- FFQ
food frequency questionnaire
- CRS
chronic rhino-sinusitis
- TEI
total energy intake
- BMI
body mass index
Contributor Information
Vanessa Garcia-Larsen, Phone: (0044) 020 7594 9574, Email: v.garcialarsen@imperial.ac.uk.
Rhonda Arthur, Email: Rhonda.arthur@kcl.ac.uk.
James F. Potts, Email: j.potts@imperial.ac.uk
Peter H. Howarth, Email: p.h.howarth@soton.ac.uk
Matti Ahlström, Email: matti.ahlstrom@helsinki.fi.
Tari Haahtela, Email: tari.haahtela@helsinki.fi.
Carlos Loureiro, Email: acnl@sapo.pt.
Ana Todo Bom, Email: flcosta@netcabo.pt.
Grzegorz Brożek, Email: brozekg@mp.pl.
Joanna Makowska, Email: joanna.makowska@umed.lodz.pl.
Marek L. Kowalski, Email: kowalsml@csk.umed.lodz.pl
Trine Thilsing, Email: Trine.Thilsing@rsyd.dk.
Thomas Keil, Email: thomas.keil@charite.de.
Paolo M. Matricardi, Email: paolo.matricardi@charite.de
Kjell Torén, Email: kjell.toren@amm.gu.se.
Thibaut van Zele, Email: thibaut.vanzele@ugent.be.
Claus Bachert, Email: claus.bachert@ugent.be.
Barbara Rymarczyk, Email: b.rymarczyk@interia.pl.
Christer Janson, Email: Christer.Janson@medsci.uu.se.
Bertil Forsberg, Email: bertil.forsberg@envmed.umu.se.
Ewa Niżankowska-Mogilnicka, Email: ewan@ghml.pl.
Peter G. J. Burney, Email: p.burney@imperial.ac.uk
References
- 1.Julia V, Macia L, Dombrowicz D. The impact of diet on asthma and allergic diseases. Nat Rev Immunol. 2015;15:308–322. doi: 10.1038/nri3830. [DOI] [PubMed] [Google Scholar]
- 2.Seyedrezazadeh E, Moghaddam MP, Ansarin K, Vafa MR, Sharma S, Kolahdooz F. Fruit and vegetable intake and risk of wheezing and asthma: a systematic review and meta-analysis. Nutr Rev. 2014;72:411–428. doi: 10.1111/nure.12121. [DOI] [PubMed] [Google Scholar]
- 3.Shaheen SO, Sterne JA, Thompson RL, Songhurst CE, Margetts BM, Burney PG. Dietary antioxidants and asthma in adults: population-based case–control study. Am J Respir Crit Care Med. 2001;164:1823–1828. doi: 10.1164/ajrccm.164.10.2104061. [DOI] [PubMed] [Google Scholar]
- 4.Patel BD, Welch AA, Bingham SA, Luben RN, Day NE, Khaw KT, Lomas DA, Wareham NJ. Dietary antioxidants and asthma in adults. Thorax. 2006;61:388–393. doi: 10.1136/thx.2004.024935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Romieu I, Varraso R, Avenel V, Leynaert B, Kauffmann F, Clavel-Chapelon F. Fruit and vegetable intakes and asthma in the E3N study. Thorax. 2006;61:209–215. doi: 10.1136/thx.2004.039123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barros R, Moreira A, Padrão P, Teixeira VH, Carvalho P, Delgado L, Lopes C, Severo M, Moreira P. Dietary patterns and asthma prevalence, incidence and control. Clin Exp Allergy. 2015;45:1673–1680. doi: 10.1111/cea.12544. [DOI] [PubMed] [Google Scholar]
- 7.Garcia-Larsen V, Chinn S, Arts IC, Amigo H, Rona RJ. Atopy, wheeze and bronchial responsiveness in young Chilean adults. Do dietary antioxidants matter? Allergy. 2007;62:714–715. doi: 10.1111/j.1398-9995.2007.01357.x. [DOI] [PubMed] [Google Scholar]
- 8.Liang W, Chikritzhs T, Lee AH. Lifestyle of young Australian adults with asthma. Asia Pac J Public Health. 2015;27:NP248–NP254. doi: 10.1177/1010539512439229. [DOI] [PubMed] [Google Scholar]
- 9.Lv N, Xiao L, Ma J. Dietary pattern and asthma: a systematic review and meta-analysis. J Asthma Allergy. 2014;7:105–121. doi: 10.2147/JAA.S49960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hooper R, Heinrich J, Omenaas E, Sausenthaler S, Garcia-Larsen V, Bakolis I, Burney P. Dietary patterns and risk of asthma: results from three countries in European Community Respiratory Health Survey-II. Br J Nutr. 2010;103:1354–1365. doi: 10.1017/S0007114509993266. [DOI] [PubMed] [Google Scholar]
- 11.Garcia-Larsen V, Luczynska M, Kowalski ML, et al. Use of a common food frequency questionnaire (FFQ) to assess dietary patterns and their relation to allergy and asthma in Europe: pilot study of the GA2LEN FFQ. Eur J Clin Nutr. 2011;65:750–756. doi: 10.1038/ejcn.2011.15. [DOI] [PubMed] [Google Scholar]
- 12.Bousquet J, et al. GA2LEN (Global Allergy and Asthma European Network) addresses the allergy and asthma ‘epidemic’. Allergy. 2009;64:969–977. doi: 10.1111/j.1398-9995.2009.02059.x. [DOI] [PubMed] [Google Scholar]
- 13.Tomassen P, Newson RB, Hoffmans R, et al. Reliability of EP3OS symptom criteria and nasal endoscopy in the assessment of chronic rhinosinusitis: a GA2LEN study. Allergy. 2011;66:556–561. doi: 10.1111/j.1398-9995.2010.02503.x. [DOI] [PubMed] [Google Scholar]
- 14.Sunyer J, Pekkanen J, Garcia-Esteban R, Svanes C, Künzli N, Janson C, de Marco R, Antó JM, Burney P. Asthma score: predictive ability and risk factors. Allergy. 2007;62:142–148. doi: 10.1111/j.1398-9995.2006.01184.x. [DOI] [PubMed] [Google Scholar]
- 15.Palmer SC, Ruospo M, Campbell KL, et al. DIET-HD Study investigators. Nutrition and dietary intake and their association with mortality and hospitalisation in adults with chronic kidney disease treated with haemodialysis: protocol for DIET-HD, a prospective multinational cohort study. BMJ Open. 2015;5:e006897. doi: 10.1136/bmjopen-2014-006897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ireland J, van Erp-Baart AM, Charrondière UR, Møller A, Smithers G. Trichopoulou A; EFCOSUM Group. Selection of a food classification system and a food composition database for future food consumption surveys. Eur J Clin Nutr. 2002;56:S33–S45. doi: 10.1038/sj.ejcn.1601427. [DOI] [PubMed] [Google Scholar]
- 17.FSA (Food Standards Agency) McCance and Widdowson’s the composition of foods. Seventh Summary edn. Royal Society of Chemistry: Cambridge; 2002. [Google Scholar]
- 18.Simes RJ. An improved Bonferroni procedure for multiple tests of significance. Biometrika. 1986;73:751–754. doi: 10.1093/biomet/73.3.751. [DOI] [Google Scholar]
- 19.Pekkanen J, Sunyer J, Anto JM, Burney P, European Community Respiratory Survey Operational definitions of asthma in studies on its aetiology. Eur Respir J. 2005;26:28–35. doi: 10.1183/09031936.05.00120104. [DOI] [PubMed] [Google Scholar]
- 20.Kelly Y, Sacker A, Marmot M. Nutrition and respiratory health in adults: findings from the health survey for Scotland. Eur Respir J. 2003;21:664–671. doi: 10.1183/09031936.03.00055702. [DOI] [PubMed] [Google Scholar]
- 21.Troisi RJ, Willett WC, Weiss ST, et al. A prospective study of diet and adult-onset asthma. Am J Respir Crit Care Med. 1995;151:1401–1408. doi: 10.1164/ajrccm.151.5.7735592. [DOI] [PubMed] [Google Scholar]
- 22.Soutar A, Seaton A, Brown K. Bronchial reactivity and dietary antioxidants. Thorax. 1997;52:166–170. doi: 10.1136/thx.52.2.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moreno-Macias H, Romieu I. Effects of antioxidant supplements and nutrients on patients with asthma and allergies. J Allergy Clin Immunol. 2014;133:1237–1244. doi: 10.1016/j.jaci.2014.03.020. [DOI] [PubMed] [Google Scholar]
- 24.Arora P, Ansari SH, Najmi AK, et al. Investigation of anti-asthmatic potential of dried fruits of Vitis vinifera L. in animal model of bronchial asthma. Allergy Asthma Clin Immunol. 2016;12:42. doi: 10.1186/s13223-016-0145-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Anhê FF, Varin TV, Le Barz M, et al. Gut microbiota dysbiosis in obesity-linked metabolic diseases and prebiotic potential of polyphenol-rich extracts. Curr Obes Rep. 2015;4:389–400. doi: 10.1007/s13679-015-0172-9. [DOI] [PubMed] [Google Scholar]
- 26.Bédard A, Garcia-Aymerich J, Sanchez M, et al. Confirmatory factor analysis compared with principal component analysis to derive dietary patterns: a longitudinal study in adult women. J Nutr. 2015;145:1559–1568. doi: 10.3945/jn.114.204479. [DOI] [PubMed] [Google Scholar]
- 27.Tanaka T, Takahashi R. Flavonoids and asthma. Nutrients. 2013;5:2128–2143. doi: 10.3390/nu5062128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Garcia V, Arts IC, Sterne JA, et al. Dietary intake of flavonoids and asthma in adults. Eur Respir J. 2005;26:449–452. doi: 10.1183/09031936.05.00142104. [DOI] [PubMed] [Google Scholar]
- 29.Knekt P, Kumpulainen J, Jarvinen R, et al. Flavonoid intake and risk of chronic diseases. Am J Clin Nutr. 2002;76:560–568. doi: 10.1093/ajcn/76.3.560. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data requests can be submitted to the GA2LEN Network of Excellence for the consideration and approval of the Scientific Steering Committee. Informal enquiries about data access and material availability can be sent to Dr Vanessa Garcia Larsen (v.garcialarsen@imperial.ac.uk). The Ga2len FFQ is free to use for academic and research purposes.


