Abstract
Renal inflammation has a key role in the onset and progression of immune– and nonimmune–mediated renal diseases. Therefore, the search for novel anti–inflammatory pharmacologic targets is of great interest in renal pathology. JQ1, a small molecule inhibitor of bromodomain and extraterminal (BET) proteins, was previously found to preserve renal function in experimental polycystic kidney disease. We report here that JQ1–induced BET inhibition modulated the in vitro expression of genes involved in several biologic processes, including inflammation and immune responses. Gene silencing of BRD4, an important BET protein, and chromatin immunoprecipitation assays showed that JQ1 alters the direct association of BRD4 with acetylated histone–packaged promoters and reduces the transcription of proinflammatory genes (IL-6, CCL-2, and CCL-5). In vivo, JQ1 abrogated experimental renal inflammation in murine models of unilateral ureteral obstruction, antimembrane basal GN, and infusion of Angiotensin II. Notably, JQ1 downregulated the expression of several genes controlled by the NF-κB pathway, a key inflammatory signaling pathway. The RelA NF-κB subunit is activated by acetylation of lysine 310. In damaged kidneys and cytokine–stimulated renal cells, JQ1 reduced the nuclear levels of RelA NF-κB. Additionally, JQ1 dampened the activation of the Th17 immune response in experimental renal damage. Our results show that inhibition of BET proteins reduces renal inflammation by several mechanisms: chromatin remodeling in promoter regions of specific genes, blockade of NF-κB pathway activation, and modulation of the Th17 immune response. These results suggest that inhibitors of BET proteins could have important therapeutic applications in inflammatory renal diseases.
Keywords: transcription factors, Cell Signaling, chemokine
All cells in an organism contain the same genetic information, but the gene expression pattern varies between cell types. Epigenetics involves heritable changes in gene expression that are not associated with DNA sequence modifications. Epigenetic mechanisms, especially DNA methylation and histone modifications, are dynamic processes and mediate the diversified gene expression profiles in normal and diseased states. Some of these epigenetic marks are being characterized in acute and chronic renal disease and could help explain gene expression regulation patterns in injured kidney. Oxidative stress, proinflammatory cytokines, and uremic toxins might induce epigenetic modifications involved in alterations of immune response, fibrosis, and cardiovascular disease in patients with renal disease.1–4 These modifications are potentially reversible, presenting an opportunity for identifying novel therapeutic targets.
The bromodomain and extraterminal (BET) protein family (BRD2, BRD3, BRD4, and BRDT) has two conserved N–terminal bromodomains (BD1 and BD2) and participates in tumor development, infections, autoimmunity, and inflammation.5–7 Bromodomains in BET proteins bind to acetylated lysine residues on histones and other nuclear proteins to regulate the transcriptional program.8 The extraterminal domain in BET proteins interacts with histone modifiers, such as the arginine demethylase JMJD6 and the lysine methyltransferase NSD3, or ATP–dependent nucleosome–remodeling enzymes SWI/SNF, causing changes in chromatin structure.9,10 Additionally, BET proteins form part of the mammalian mediator complex that facilitates binding between transcription factors and the transcriptional machinery.11,12
Among BET proteins, BRD4 is the best studied and participates in several steps of the transcription hierarchy. BRD4 recruits the positive transcription elongation factor b (P-TEFb) complex (heterodimer of CDK9 and cyclin T1, T2, or K) to the promoter region and stimulates RNA polymerase II–dependent transcription through its kinase activity.13,14 The oncogene c-myc was the first BRD4 target gene to be described.15,16 Subsequent studies reported that BRD4 binds to acetylated histones in the enhancers or promoter regions of inflammatory genes.17 BRD4 interacts with specific acetylated residues of transcription factors. One of the best characterized is NF-κB, a key player in regulating immune and inflammatory responses.18 BRD4, through its two bromodomains, specifically recognizes the acetylated lysine-310 residue of the RelA NF-κB subunit.19 In tumor cells, BRD4/RelA binding avoids RelA ubiquitinization and its proteasome-mediated degradation, thereby maintaining persistent activation of the nuclear NF-κB complex and promoting cancer cell proliferation.20,21
Two potent BET inhibitors, I-BET and JQ1, have recently been developed.5,22 These small molecules compete for the bromodomain binding pocket and displace BET proteins from binding to acetylated lysine residues. Several studies have shown that these compounds strongly inhibit malignant cell growth by targeting cancer-promoting genes.23–27 Initial studies in midline carcinoma and hematologic malignances showed that BET inhibitors induce release of the BRD4-NUT oncoprotein and the transcriptional suppression of MYC and BCL2 proto-oncogenes.28 Two human phase 1 clinical trials in patients with cancer are currently in progress.8,29 In vitro evidence in nonmalignant cells suggests that BET inhibitors could be a potential therapeutic option for inflammatory diseases. In activated macrophages, JQ1 and I-BET reduced inflammatory gene production in response to LPS.22,30 In synovial fibroblasts of patients with rheumatoid arthritis, I-BET suppressed proinflammatory factors and diminished their capacity to recruit immune cells.31 In a murine model of collagen-induced arthritis, JQ1 reduced inflammation and joint damage.32 JQ1 also ameliorated bleomycin–induced murine lung fibrosis33 and cardiac damage in a mouse model of pressure overload–induced left ventricular hypertrophy.34 A recent study reported that JQ1 delayed cyst growth and preserved renal function in a murine model of polycystic kidney disease by inhibiting c-Myc gene and cystic epithelial cell proliferation.35 Our aim was to investigate whether BET inhibition could modulate experimental inflammation in the kidney, investigating potential mechanisms involved.
Results
JQ1 Modulates Inflammatory-Related Genes in Cultured Renal Cells
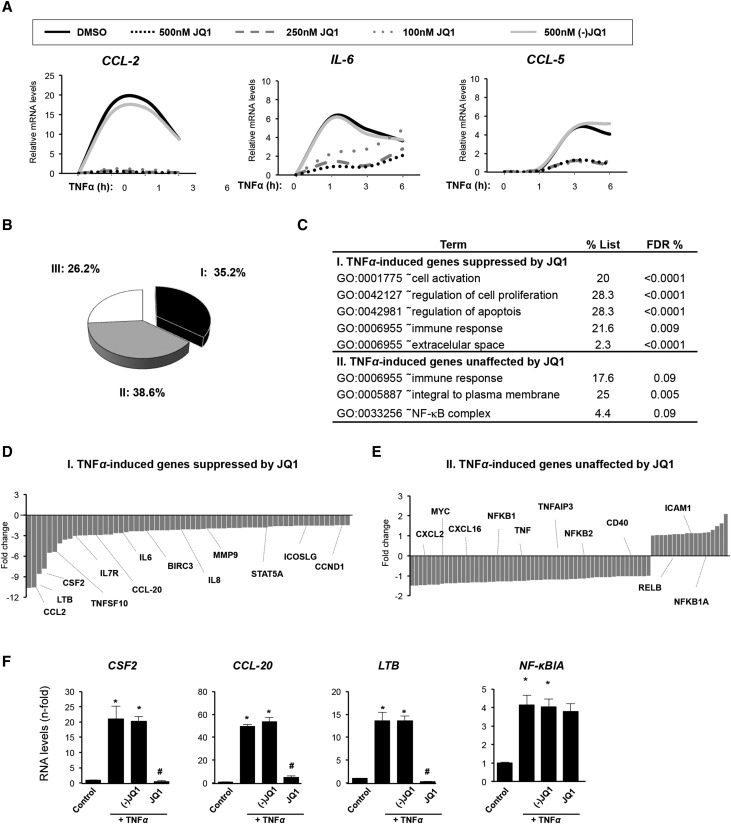
The anti-inflammatory properties of the BET inhibitor JQ1 were evaluated in vitro in human tubular epithelial cells (HK2 cell line) after TNF-α stimulation. This well characterized inflammatory cytokine regulates many proinflammatory genes and is involved in experimental and human kidney diseases.36 In HK2 cells, TNF-α stimulation upregulated gene expression of the proinflammatory factors CCL-2, CCL-5, and IL-6 for up to 6 hours, peaking around 3 hours. JQ1 dose dependently diminished overexpression of these genes (Figure 1A). The enantiomer (−)JQ1 had no effect, indicating the specificity of the response to active JQ1 (Figure 1A). Interestingly, BRD4 gene or protein levels were not altered in cells treated with JQ1 compounds, suggesting that BET proteins activity was independent of changes in their expression (Supplemental Figure 1A and B). In addition, similar results were observed with another BET inhibitor (I-BET 762) (Supplemental Figure 2).
Figure 1.
JQ1 downregulates proinflammatory gene expression in cultured renal cells. (A) Renal tubular epithelial cells (HK2 cell line) were preincubated with vehicle (DMSO), different concentrations of the BET proteins inhibitor JQ1 (100, 250, or 500 nM), or the nonactive control (−)JQ1 (500 nM) for 1 hour, and then, cells were stimulated with TNF-α (5 ng/ml) for the indicated times. Gene expression levels of CCL-2, CCL-5, and IL-6 were analyzed by real–time quantitative PCR. Results of six independent experiments are presented. In vehicle-treated cells, there were no changes in gene expression compared with untreated cells (considered as control). (B–F) BET bromodomain inhibition regulates the transcriptional program in renal tubular epithelial cells under proinflammatory conditions. HK2 cells were stimulated with TNF-α (5 ng/ml) for 3 hours in the presence of JQ1 or the nonactive control (−)JQ1 (500 nM) and analyzed by (B–E) whole–genome gene expression arrays or (F) real–time quantitative PCR. (B) Pie chart of the percentages of differentially expressed probes in each cluster: I, upregulated probes in TNF-α–stimulated cells versus untreated cells showing only those suppressed by JQ1; II, upregulated probes in TNF-α that were unchanged after JQ1 treatment; and III, downregulated probes in TNF-α–stimulated cells. (C) Functional analysis pathways (gene ontology [GO] analysis) of genes differentially expressed in clusters I and II. D and E show representative genes of I and II clusters. Genes with >1.5-fold change were considered to be differentially expressed. (F) Data validation by real–time quantitative PCR. Data are expressed as means±SEM of three independent experiments. *P<0.05 versus control (vehicle-treated cells); #P<0.05 versus TNF-α–treated cells.
Next, we investigated the transcriptional pathways altered by BET inhibition using a whole–genome gene expression array. Global gene expression analysis showed that 210 probes (corresponding to 193 genes) were regulated after TNF-α stimulation, of which 155 (73.8%; 139 genes) were upregulated and 55 were downregulated (26.2%; 54 genes) (Figure 1B). Among TNF-α–inducible genes, 74 probes (35.2%; 62 genes) were diminished by JQ1, whereas 81 probes (38.6%; 77 genes) were unaffected (Figure 1B). Functional pathway analysis revealed that JQ1 modulated the expression of genes related to cell activation (CSF2, IL-6, IL-8, LTB, and STAT5A), cell proliferation (SMAD3, CCND1, and ICOSLG), apoptosis (BIRC3, BCL3, and TNFSF10), and immune response (CCL-2, CCL-20, and IL-7R) (Figure 1, C and D). Conversely, the expression of some chemokines (CXCL-2), adhesion molecules (ICAM1), and immune-associated genes (CD40 and TNF-α) was unchanged after JQ1 treatment (Figure 1E). In contrast to observations in cancer cells and cystic epithelial cells,15,35,37 c-Myc expression was not changed by JQ1 in renal cells as described in the heart or the hematopoietic differentiation of stem cells.38,39 JQ1 did not modify certain genes of the NF-κB pathway (NF-κB1A, RelB, NF-κB2, and NF-κB1). Some of these data were corroborated by quantitative RT-PCR (Figure 1, A and F).
BRD4, through Bromodomains, Binds to Inflammatory Gene Promoters and Regulates Transcription
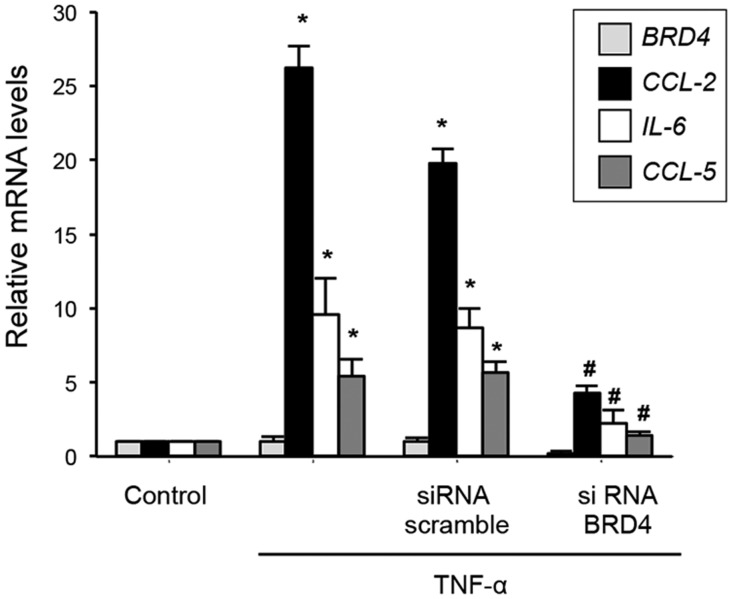
The role of BRD4 in TNF-α–mediated proinflammatory gene regulation was evaluated by gene silencing. Transfection of HK2 cells with small interfering RNA (siRNA) specifically targeting BRD4 but not with scramble siRNA significantly reduced gene upregulation of IL-6, CCL-2, and CCL-5 caused by TNF-α (Figure 2).
Figure 2.
BRD4 disruption inhibits TNF-α–induced upregulation of proinflammatory genes in renal tubular epithelial cells. HK2 cells were transfected with a pool of BRD4 siRNA or nonspecific scramble siRNA (40 nM) for 48 hours. Cells were then treated with TNF-α (5 ng/ml) for 3 hours more. Gene expression of BRD4 and the proinflammatory genes CCL-2, CCL-5 and IL-6 was measured by real–time quantitative PCR. Data are representative of four independent experiments, and results are expressed as the means±SEM. *P<0.05 versus control untransfected cells; #P<0.05 versus nonspecific scramble siRNA.
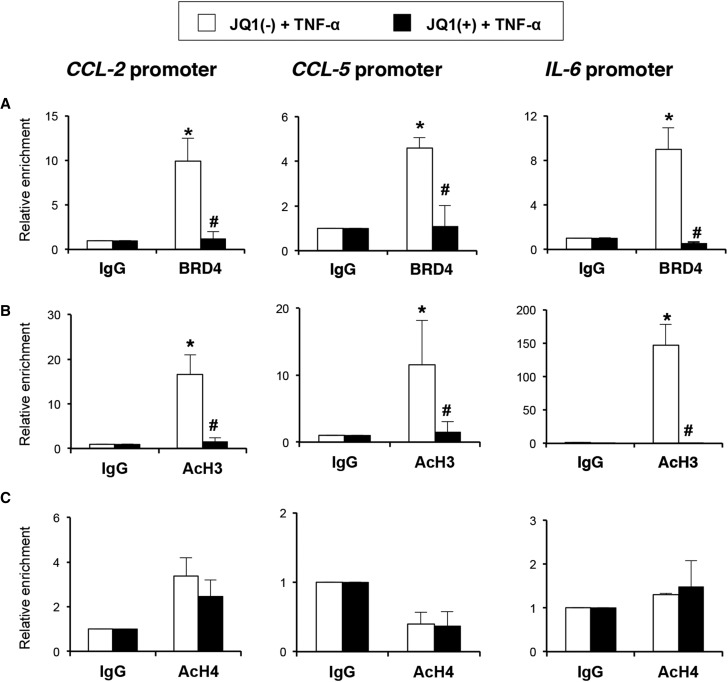
To determine whether JQ1 alters the direct association of BRD4 with acetylated histone-packaged promoters, chromatin immunoprecipitation (ChIP) assays with an anti-BRD4 antibody were performed. In the presence of the nonactive control (−)JQ1, BRD4 is robustly associated with the CCL-2, CCL-5, and IL-6 promoters in TNF-α–stimulated cells (Figure 3A). However, in the presence of JQ1, the binding of BRD4 to the regulatory regions was significantly reduced, indicating that BRD4 directly regulates the expression of these genes.
Figure 3.
JQ1 inhibits the direct binding of BRD4 at the CCL-2, CCL-5, and IL-6 promoters through histone acetylation in renal tubular epithelial cells. HK2 cells were pretreated with (−)JQ1 or JQ1 (500 nM) for 1 hour and further stimulated with TNF-α (5 ng/ml) for 3 hours more. ChIP assays were performed using specific antibodies against (A) BRD4, (B) AcH3, and (C) AcH4. Normal rabbit IgG was used as a negative control. Enrichment of BRD4–specific DNA sequences around the transcriptional sites of the human CCL-2, CCL-5, and IL-6 promoters was measured by real–time quantitative PCR using specific primers. Data from three independent experiments are shown, in which each quantitative PCR was run in triplicate. Results are represented as the relative enrichment of BRD4 binding compared with the negative control antibody (IgG). *P<0.05 versus corresponding IgG; #P<0.05 versus (−)JQ1-treated cells.
BET proteins recognize acetylated residues in histones, facilitating recruitment of the transcriptional activation machinery.28 Because active promoters are closely related to the enrichment of acetylated histones, the presence of acetylated histone H3 (AcH3) or AcH4 in the promoter region of these proinflammatory genes was investigated. AcH3 was more enriched in TNF-α–treated cells, consistent with the active transcription of these genes, and AcH3 enrichment was significantly reduced by JQ1 (Figure 3B). However, AcH4 levels were not enhanced in TNF-α–treated cells and were unchanged by JQ1 (Figure 3C).
JQ1 Ameliorates Experimental Renal Inflammation by the Inhibition of BRD4 Binding to Proinflammatory Genes Promoters
The JQ1 effect in renal inflammation was investigated in the unilateral ureteral obstruction (UUO) model. Inflammatory cell infiltration is one of the earliest characteristics of renal damage after UUO.40,41 The presence of infiltrating of monocytes/macrophages in obstructed kidneys was observed after 2 days and increased at 5 days (Figure 4). Treatment with JQ1 diminished the presence of infiltrating monocytes/macrophages, but only the dose of 100 mg/kg per day exerted a significant inhibitory effect (Figure 4, A and C). The immunohistochemical characterization of immune cells revealed that JQ1 significantly reduced the number of monocytes/macrophages, neutrophils, dendritic cells, and T lymphocytes in obstructed kidneys to levels similar to those of controls (Figure 4, B and D). In obstructed kidneys, renal lesions and gene upregulation of Ngal and Kim1, two biomarkers of kidney injury,40,42 were diminished by JQ1 (Supplemental Figure 3).
Figure 4.
JQ1 reduces renal inflammation in the experimental models of UUO. The UUO model was done in mice and studied after (A) 2 or (B) 5 days. Some mice were treated with JQ1 (60 or 100 mg/kg per day intraperitoneally as indicated) or vehicle (10% hydroxypropyl β-cyclodextrin) starting 24 hours before UUO. In paraffin–embedded kidney sections, inflammatory cell infiltration was evaluated using antibodies against F4/80 (monocytes/macrophages/dendritic cells), myeloperoxidase (neutrophils), CD3 (T lymphocytes), and CD4 (T helper lymphocytes). A representative animal from each group is shown. Magnification, ×200×. C and D show the quantification of immunohistochemistry. Data are expressed as the means±SEM of six to eight animals per group. *P<0.05 versus contralateral (C); #P<0.05 versus vehicle–treated obstructed (Ob) kidneys.
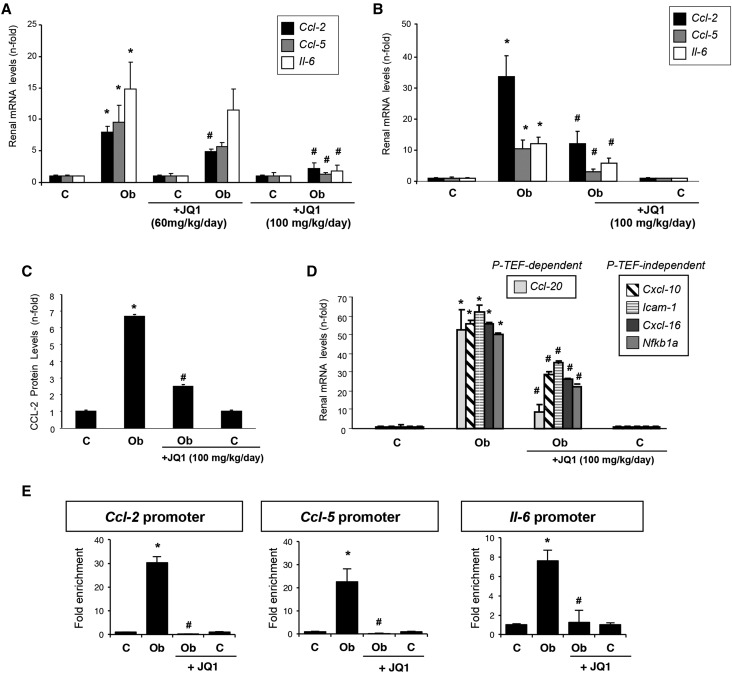
The recruitment of inflammatory cells into the kidney is mediated by proinflammatory mediators.43 Two days after UUO, gene upregulation of Ccl-2, Ccl-5, and Il-6 was found. JQ1 treatment at 100 mg/kg per day significantly diminished proinflammatory gene overexpression (Figure 5, A and B) and protein levels as exemplified in CCL-2 (Figure 5C). Other inflammation/immune-related genes upregulated in the obstructed kidneys were also inhibited by JQ1, including chemokines (Ccl-20 and Cxcl-16), adhesion molecules (Icam-1), and components of the NF-κB pathway (NF-κB1a) (Figure 5D). ChIP assays showed that BRD4 binding to the promoter regions of Ccl-2, Ccl-5, and Il-6 genes was significantly greater in obstructed than in contralateral kidneys. However, this BRD4 enrichment was significantly less in obstructed kidneys from JQ1-treated mice (Figure 5E). Our results show that direct binding of BRD4 to proinflammatory gene promoters and the consequent chromatin remodeling comprise a potential mechanism explaining the renal anti–inflammatory effects of BET inhibition.
Figure 5.
JQ1 attenuates gene expression of proinflammatory factors in obstructed kidneys. Mice were treated with JQ1 (100 or 60 mg/kg per day) or vehicle (10% cyclodextran solution) starting 24 hours before UUO and studied after 2 or 5 days. (A and B) RNA was isolated from frozen samples of whole kidney, and gene expression levels were evaluated by real–time quantitative PCR. (C) CCL-2 protein levels were evaluated by ELISA. Results are means±SEM of six to eight animals per group. Data are normalized versus contralateral untreated kidney (considered as one). (D) BRD4 downregulates the proinflammatory gene expression in the damaged kidney. (E) A ChIP assay was carried out in renal samples from UUO mice treated or not treated with JQ1 using an antibody specific for BRD4 or normal rabbit IgG, the latter being a negative control. Enrichment of BRD4 binding regions in the promoters of mouse Ccl-2, Ccl-5, and Il-6 genes was quantified by quantitative PCR using specific primers. Data are from two independent experiments, and each quantitative PCR was run in triplicate. Results are expressed as the n-fold enrichment of anti-BRD4 antibody relative to a negative control antibody (normal rabbit IgG) and further normalized versus contralateral kidney (considered as one). *P<0.05 versus contralateral (C); #P<0.05 versus vehicle–treated obstructed (Ob) kidneys.
JQ1 Ameliorates Inflammation and Kidney Injury in the Antimembrane Basement Membrane GN Mice Model
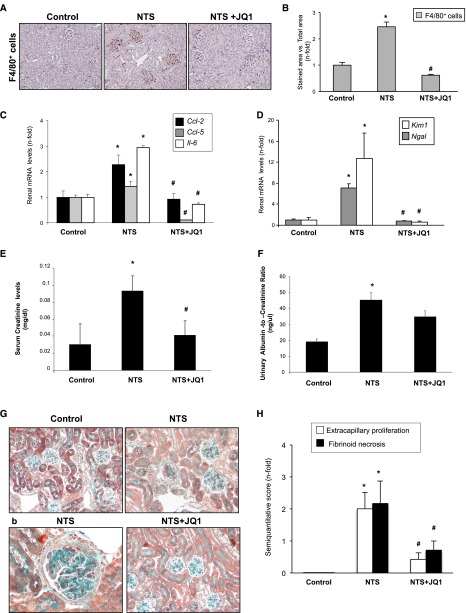
The experimental antiglomerular basement membrane nephritis induced by nephrotoxic serum (NTS) administration is a model commonly used to study mechanisms of human crescentic GN.44 In NTS-challenged mice, JQ1 diminished inflammatory cell infiltration (Figure 6, A and B), overexpression of proinflammatory genes (Ccl-2, Ccl-5, and Il-6) (Figure 6C), and biomarkers of renal injury (Ngal and Kim1) (Figure 6D). NTS-injected mice presented proliferation of intrinsic glomerular cells in the Bowman’s space, resulting in the rapid deterioration of renal function, resembling human pathology.44 JQ1 restored changes in renal function (as serum creatinine and urinary albumin) (Figure 6, E and F) and ameliorated glomerular lesions (Figure 6, G and H).
Figure 6.
JQ1 diminishes renal inflammation in the NTS nephritis. Accelerated NTS nephritis was done in mice by administration of an antimurine glomerular basement membrane rabbit antiserum, and mice were studied 10 days after immunization. Mice were treated or not treated with JQ1 (100 mg/kg per day) starting before the first NTS injection. (A) In paraffin–embedded kidney sections of NTS mice, glomerular infiltration of monocytes/macrophages was found (detected using an antibody against F4/80). B shows the quantification of immunohistochemistry. (C and D) RNA was isolated from frozen samples of whole kidney, and gene expression levels were evaluated by real–time quantitative PCR. (E) JQ1 ameliorates renal damage in the NTS nephritis. Data on (E) serum creatinine levels and (F) the ratio of albumin to creatinine in the urine are shown. Data are expressed as the means±SEM of five to seven animals per group. Morphologic lesions were evaluated by Masson trichrome staining. Treated mice with JQ1 (100 mg/kg per day) displayed fewer and less severe glomerular lesions. G shows a representative animal from each group, and the quantification of the lesion is shown in H. Magnification, ×200 in G; ×400 in G, b. *P<0.05 versus control; #P<0.05 versus NTS.
JQ1 Diminishes Renal Inflammation in the Angiotensin II Infusion Model
The model of systemic administration of Angiotensin II (AngII) is a nonimmune model of renal damage.42,45 As described, AngII induced an early inflammatory response; at 3 days, increased infiltrating interstitial monocytes/macrophages and proinflammatory mediators were found that were diminished in JQ1-treated mice (Figure 7, A–C). In AngII-infused mice, there was no change in renal function (evaluated by serum creatinine levels) (Figure 7D) when inflammation appeared. In contrast, Ngal gene expression and protein levels in the kidney were significantly increased in AngII-infused mice and normalized by JQ1 treatment (Figure 7, E and F).
Figure 7.
JQ1 diminishes renal inflammation in the model of AngII infusion in mice. Systemic infusion of AngII (1000 ng/kg per minute; subcutaneous osmotic mini-pumps) was done for 3 days. Mice were treated or not treated with JQ1 (100 mg/kg per day) starting 24 hours before infusion. (A) In paraffin–embedded kidney sections of AngII-infused mice, some interstitial infiltrating monocytes/macrophages were found (detected using an antibody against F4/80; marked by arrows). A representative animal from each group is shown. (B) Shows the quantification of immunohistochemistry. (C) RNA was isolated from frozen samples of whole kidney, and proinflammatory gene expression levels were evaluated by real–time quantitative PCR. (D) Data of serum creatinine levels. (E) Renal Ngal gene expression and (F) protein levels are shown. Data are expressed as the means±SEM of four to six animals per group. *P<0.05 versus control; #P<0.05 versus AngII-infused mice.
JQ1 Modulates the Canonical NF-κB Pathway Activation in the Kidney In Vivo and In Vitro
BRD4 recognizes and binds to the acetylated lysine-310 of the RelA NF-κB subunit.19,20 We hypothesized that blockade of this interaction by JQ1 might suppress the inflammatory response mediated by NF-κB pathway activation in kidney damage.
NF-κB remains in the cytosol of resting cells as an inactive complex formed by p50 and RelA/p65 subunits bound to the inhibitory subunit IκBα. One of the earliest steps in NF-κB activation is the phosphorylation of IκBα and RelA.18 In HK2 cells, TNF-α increased RelA and IκBα phosphorylation, but in the presence of JQ1, there were no changes in their phosphorylation levels (Figure 8, A and B). Interestingly, the nuclear levels of RelA were reduced in JQ1–treated TNF-α–stimulated cells (Figure 8C).
Figure 8.
JQ1 inhibits NF-κB pathway activation in renal cells and murine damaged kidney. HK2 cells were pretreated with 500 nM JQ1 for 1 hour and stimulated with 5 ng/ml TNF-α for 20 minutes. (A) Activation of NF-κB was evaluated in terms of changes in phosphorylation levels of IκBα (p-IκBα) and the p65 NF-κB subunit (p-RelA) in total protein extracts by Western blot. GAPDH was used as the loading control. (B) Quantification. Means±SEM of three independent experiments. *P<0.05 versus control; #P<0.05 versus TNF-α. (C) Activation of NF-κB was evaluated by p65 NF-κB subunit (RelA) in nuclear extracts by Western blot. Histone H1 was used as the loading control. Means±SEM of three independent experiments. *P<0.05 versus control; #P<0.05 versus TNF-α. (D–F) In nuclear proteins isolated from different mice models treated or not treated with JQ1, p65 NF-κB levels were evaluated by Western blot. Ponceau Red was used as the loading control. *P<0.05 versus control mice; #P<0.05 versus injured kidneys (obstructed;NTS;AngII respectively). Data are expressed as the means±SEM of six to eight animals per group.
Previous work by our group showed that activation of NF-κB contributes to the progression of renal damage in UUO and AngII infusion models.40,42,45 Blockade of RelA NF-κB activation diminished renal inflammation in those models, reducing inflammatory cell infiltration mainly by downregulating proinflammatory genes. Treatment with JQ1 produced significantly lower renal RelA nuclear levels in obstructed kidneys than in untreated obstructed ones (Figure 8D). Similarly, JQ1 diminished renal RelA nuclear levels in the injured kidneys by NTS (Figure 8E) or AngII infusion (Figure 8F). These data show that BET inhibition blocks NF-κB activation by lowering nuclear RelA levels and subsequent functional activation.
JQ1 Inhibits Th17 Immune Response in Experimental Renal Inflammation
Growing evidence suggests that the Th17 immune response and its effector cytokine IL-17A contribute to the pathogenesis of renal diseases, including immune and nonimmune nephropathies.46–48 In UUO kidneys, IL-17A gene and protein levels were increased and significantly downregulated by JQ1 (Figure 9, A–C, Supplemental Figure 4). Similarly, JQ1 diminished renal IL-17A production in the injured kidneys by NTS (Figure 9D).
Figure 9.
JQ1 diminishes the Th17 immune response in experimental renal damage. Renal IL-17A (A) gene or (B) protein levels were evaluated in the UUO model by real time or Western blot, respectively; GAPDH or Tubulin was used as the loading control, respectively. Data are expressed as the means±SEM of six to eight animals per group. C, control; Ob, obstructed. *P<0.05 versus control mice; #P<0.05 versus injured kidney. Immunolocalization of IL-17A in (C) UUO or (D) NTS mice treated or not treated with JQ1. The figure shows a representative image of five mice per group.
Discussion
Our studies show that the specific BET inhibitor JQ1 reduces renal inflammation in different models of renal damage, including UUO and NTS–and AngII–induced renal damage. We have described that JQ1 reduces renal inflammation by several mechanisms: (1) direct binding of BET proteins to acetylated residues of H3 histone in the promoter regions of proinflammatory genes (shown in CCL-2, IL-6, and CCL-5), (2) blockade of NF-κB pathway activation by reducing RelA nuclear levels, and (3) modulation of the Th17 immune response. These results suggest that BET inhibitors could have important therapeutic applications in inflammatory renal diseases.
Evidence is emerging that epigenetic mechanisms control acute inflammation. Histone acetylation and/or deacetylation are involved in NF-κB–mediated inflammation.49 Decreased H3 lysine-9 trimethylation in inflammatory gene promoters upregulates inflammatory gene expression in cells under hyperglycemic conditions.50 Several studies suggest that BET proteins regulate the inflammatory process. In activated macrophages, BET inhibitors suppress the expression of cytokines (TNF-α and IL-6) and chemokines (CCL-2), protecting against endotoxic shock.22 Moreover, BRD2 gene disruption in bone marrow–derived macrophages blunts proinflammatory cytokine (IL-6 and TNF-α) production.30 In experimental renal damage and cultured tubular epithelial cells under inflammatory conditions, we found that JQ1 reduces the expression of many proinflammatory mediators by modulating BRD4 actions. Gene silencing and ChIP assays show a key role for BRD4 in regulating the proinflammatory genes CCL-2, IL-6, and CCL-5 at the promoter level. JQ1 targeting the interaction between BRD4 and acetylated histones reduces proinflammatory gene transcription, showing that chromatin remodeling is a mechanism by which BET inhibition might impair the renal inflammatory response. JQ1 treatment reduced histone H3 acetylation without affecting histone H4, suggesting that BRD4 binding to the promoter region prevents recruitment of H3–specific histone deacetylases.
Many experimental studies have shown that NF-κB blockade attenuates renal inflammation and ameliorates disease progression, including our studies in the UUO and AngII infusion model.18,41 We found in vivo and in vitro that JQ1 reduced RelA nuclear levels and thereby, NF-κB transcriptional activation, revealing a chromatin-independent mechanism involved in the anti-inflammatory effect of BET inhibitors in injured kidneys. Intensive research has unraveled the role of BET proteins in NF-κB pathway regulation, showing that BRD4 is a coactivator of NF-κB.20,51,52 BRD4 binding to acetylated lysine-310 of RelA is essential for recruiting BRD4 and CDK9 to the promoters of specific NF-κB target genes.19 Our data suggest that inhibition of BRD4 binding to acetylated RelA and subsequent NF-κB–mediated proinflammatory gene expression downregulation are other mechanisms contributing to the anti-inflammatory effects of JQ1 in renal damage.
Whole–genome expression arrays in TNF-α–stimulated tubular epithelial cells showed that some NF-κB–regulated genes were downregulated by JQ1, whereas others were unchanged. Many components of the NF-κB pathway, including NF-κB1/p50, NF-κB2/RelA/p65, NF-κBIA/IκBα, RelB, A20/TNFAIP3, and TNIP1, were among the genes unaffected by this inhibitor. Previous studies have shown that JQ1 displaces BRD4 and consequently, P-TEFb from promoter regions, leading to a decrease in Pol II elongation at active genes.17 P-TEFb participates in the transcriptional elongation of a subset of NF-κB genes, whereas others are P-TEFb independent.52–54 Amir-Zilberstein et al.52 reported that NF-κB–controlled genes lacking a TATA-box sequence in their core promoters are regulated independently of the P-TEFb elongation factor. Some of our results support this hypothesis; all of the aforementioned JQ1–unaffected, NF-κB–regulated genes are TATA less and therefore, not regulated by P-TEFb and BRD4. Amir-Zilberstein et al.52 also suggested that a TATA box is a prerequisite for P-TEFb to bind to NF-κB–regulated gene promoters. However, we observed that, although several JQ1–downregulated, NF-κB–regulated genes (e.g., CCL-5, CCL-20, and IL-8) have a TATA–box core promoter, many others are TATA less (e.g., LTB, CSF2, MMP9, and CCND1). Our data suggest that JQ1 inhibited those P-TEFb–dependent, NF-κB–controlled genes by a chromatin-independent process but mediated them by direct effect on RelA/p65 levels.
Our in vitro data showed that JQ1 only reduces RelA nuclear levels, without affecting the basal or phosphorylated levels of cytosolic NF-κB proteins. Moreover, JQ1 preferentially inhibits the upregulation of proinflammatory genes under NF-κB control, whereas key NF-κB pathway components, such as NF-κB1/p50, NF-κB2/RelA/p65, NF-κBIA/IκBα, RelB, and A20/TNFAIP3, were unaffected. These data are consistent with the observation that TNF-α stimulation induced acetylated lysine-310–dependent recruitment of BRD4 to the promoter of proinflammatory genes, with no effect on A20/TNFAIP3 expression.19 Our results suggest that inhibiting the acetylated RelA/Brd4 interaction with BET-bromodomain inhibitors may provide a novel pharmacologic strategy to block NF-κB activation in inflammatory renal disease. This strategy may have more selective anti–inflammatory effects than using NF-κB kinase inhibitors or proteasome-targeted drugs.
Studies of genetically modified mice or using neutralizing antibodies against IL-17A or its receptor have shown the involvement of Th17 immune responses in chronic inflammatory diseases,55–60 including immune and nonimmune experimental renal damage.47,48 The presence of Th17 cells in UUO-damaged kidneys has been described.47,48 We have confirmed this observation, detecting IL-17A+ cells by immunohistochemistry and upregulation of IL-17A mRNA and protein. Treatment with JQ1 practically abolished kidney IL-17A levels in UUO and NTS-induced nephritis. BRD4 and BRD2 bind directly to the chromatin in the CNS2 regulatory region of the IL-17a gene controlling its expression.59 In vitro, other Th17–related cytokines, such as CCL-20 and CSF-1, were downregulated by JQ1. Moreover, BET inhibition suppresses Th17 differentiation from human and mouse naive CD4+ T cells and the activation of previously differentiated Th17 cells.16 Accordingly, BET inhibition protects against development of collagen-induced arthritis and experimental autoimmune encephalomyelitis caused by the impaired generation and function of Th17 cells.59 Our results expand the strategies for neutralizing IL-17A–mediated inflammatory effects in the kidney by using bromodomain inhibitors.
During renal damage, various extracellular signals promote rapid induction of proinflammatory gene expression by activating and recruiting transcription factors and/or transcriptional coactivators to the promoter regions. Our findings highlight the importance of BET proteins in regulating the inflammatory response in the kidney, where they act as epigenetic readers that recognize acetylation marks in histones and other proteins, including transcription factors (as described for NF-κB). Additionally, BET proteins help regulate the mammalian transcription complex that initiates the transcription of proinflammatory genes. Our data show that blocking BET protein bromodomains with small molecule inhibitors, such as JQ1, might be a powerful therapeutic strategy for inflammatory renal diseases. These findings identify BRD4 as a critical genomic regulator of renal inflammation and provide mechanistic insights into the intrinsic epigenetic vulnerabilities of inflammatory disease that can be exploited for pharmacologic intervention.
Concise Methods
Experimental Models
All animal procedures were performed according to the guidelines of animal research for the European Community and with prior approval by the Ethics Committee of the Health Research of the IIS-Fundación Jiménez Díaz. The BET bromodomain inhibitor JQ1, a thieno-triazolo-1,4-diazepine, was synthesized and provided collaboratively by James Bradner (Dana-Farber Cancer Institute, Boston, MA).5 For in vivo studies, JQ1 was dissolved in 10% hydroxypropyl β-cyclodextrin and used at a therapeutic dose (100 or 60 mg/kg per day intraperitoneally) as previously described.31
The model of UUO was established in male C57BL/6 mice under isoflurane-induced anesthesia as described; the left ureter was ligated with silk (5/0) at two locations and cut between ligatures to prevent urinary tract infection (obstructed kidney).40,47 Some animals were treated daily with JQ1 from 1 day before UUO and studied after 2 and 5 days (n=6–8 mice per group). At the time of euthanasia, animals were anesthetized with 5 mg/kg xylazine (Rompun; Bayer HealthCare, Whippany, NJ) and 35 mg/kg ketamine (Ketolar; Pfizer), and the kidneys were perfused in situ with cold saline before removal. Studies compared both kidneys (contralateral versus obstructed) in each mouse. Kidneys were further processed for immunohistochemistry (fixed and paraffin embedded) or ChIP assays (fixed in 1% formaldehyde followed by quenching with 0.125 M glycine) or snap frozen in liquid nitrogen for RNA and protein studies.
The model of systemic infusion of AngII was done in C57BL/6 mice subcutaneously (ALZET Osmotic mini-pumps Model 2001; Alza Corp., Palo Alto, CA) at the dose of 1000 ng/kg per minute for 3 days as described.42,45 Some animals were also daily treated with JQ1 from 1 day before AngII infusion. As control, saline infusion was done.
The accelerated NTS nephritis was produced in male C57BL/6 mice using antimurine glomerular basement membrane rabbit antiserum as described.44 Mice were injected with 50 μl NTS diluted 1:10 in sterile saline on day 1. Then, mice were injected with 4 μl/g body wt on days 2 and 3, and they were studied 10 days later. Some animals were also daily treated with JQ1 starting 1 day before the first NTS administration. The renal lesions were evaluated by Masson staining. Extracapillary proliferation ratio was calculated by counting injury and normal glomeruli (60–70 glomeruli for each renal sample). The proportion of pathologic glomeruli was evaluated by examination of at least 50 glomeruli per section by an examiner masked to the experimental conditions.
Cell Cultures and Treatments
Human renal proximal tubular epithelial cells (HK2 cell line; ATCC CRL-2190; American Type Culture Collection, Manassas, VA) were grown in RPMI 1640 medium (Gibco, Carlsbad, CA) as described elsewhere.42 JQ1 and its enantiomer (−)JQ1 (Selleckchem), the latter used as the nonactive control, were dissolved in DMSO (0.25%). Cells were maintained in serum-free medium for 24–48 hours before experiments.
Cell Transfection and Gene Silencing
Subconfluent HK2 cells were transfected with 40 nM Dharmacon ON-TARGETplus Human BRD4 (L-004937) SMARTpool siRNA using DharmaFECT 1 Transfection Reagent (Dharmacon). As control, a nonspecific scrambled siRNA was used. Cells were then incubated in serum-free medium for 48 hours before stimulation. Knockdown of BRD4 was validated by quantitative RT-PCR.
Histology and Immunohistochemistry
Paraffin–embedded kidney sections (5 μm) were stained using standard histology procedures as described elsewhere.47 Briefly, sections were incubated for antigen retrieval (PTlink System; Dako) followed by blockade of nonspecific bindings, incubated with primary antibodies and biotinylated secondary antibodies, and developed by standard procedures. The specificity was checked by omission of primary antibodies. The positive-stained areas were assessed in 10 randomly chosen fields (×200 magnification) using Image-Pro Plus software. Data are expressed as the positive-stained area relative to the total area. The primary antibodies used were myeloperoxidase, CD3, CD4 (ready to use; Dako), and F4/80 (1:50; Serotec).
Protein Studies
Total protein samples for frozen renal tissue were isolated in lysis buffer as described45 or NE-PER Reagent (Pierce, Rockford, IL) to obtain nuclear and cytoplasmic fractions. Proteins (20–100 μg per lane; quantified using a BCA protein assay kit) were separated on 8%–12% polyacrylamide-SDS gels under reducing conditions as described.47 The quality of proteins and the efficacy of protein transfer were evaluated by Ponceau Red staining (not shown). The following primary antibodies were used: p-p65 (Ser536; 1:500; Cell Signaling Technology, Danvers, MA), p65 (Sc-372) (1:500; Santa Cruz Biotechnology, Santa Cruz, CA), p-IκBα (Sc-8404) (Ser32; 1:500; Santa Cruz Biotechnology), BRD4 (1:2000; A301–985A; Bethyl), tubulin (1:5000; Sigma-Aldrich, St. Louis, MO), histone H1 (Sc-8030) (1:500; Santa Cruz Biotechnology), β-actin (1:10,000; A5441; Sigma-Aldrich), and GAPDH (1:5000; Chemicon International).
Gene Expression Studies
Total RNA from cells or frozen renal tissue was isolated with TriPure Reagent (Roche, Basel, Switzerland). cDNA was synthesized with a High-Capacity cDNA Archive Kit (Applied Biosystems, Foster City, CA) using 2 μg total RNA primed with random hexamer primers following the manufacturer’s instructions. Next, quantitative gene expression analysis was performed by real-time PCR on an AB7500 Fast Real-Time PCR System (Applied Biosystems) using fluorogenic TaqMan MGB Probes and primers designed by Assay-on-Demand gene expression products (Table 1). Data were normalized to GAPDH RNA expression. The mRNA copy numbers were calculated for each sample by the instrument software using the Ct value (arithmetic fit point analysis for the lightcycler).
Table 1.
Gene assays
| Gene | Mouse or Human Assay Identification |
|---|---|
| Mouse assay identification (FAM) | |
| Ccl-2 | Mm00441242_m1 |
| Ccl-5 | Mm01302428_m1 |
| Il-6 | Mm00446190_m1 |
| Ngal | Mm01324470_m1 |
| Kim-1 | Mm00506686_m1 |
| Mouse assay identification (VIC) | |
| gapdh | Mm99999915_g1 |
| Human assay identification (FAM) | |
| CCL-2 | Hs00234140_m1 |
| CCL-5 | Hs00174575_m1 |
| IL-6 | Hs00174131_m1 |
| BRD2 | Hs01121986_g1 |
| BRD4 | Hs04188087_m1 |
| LTB | Hs00242739_m1 |
| CSF2 | Hs00929873_m1 |
| CCL-20 | Hs01011368_m1 |
| NFKB1A (IκBα) | Hs00355671_g1 |
| CXCL-16 | Hs00222859_m1 |
| Human assay identification (VIC) | |
| 18s eukaryotic ribosomal | 4210893E |
| GAPDH | Hs02758991_g1 |
ChIP
Cells (107) were fixed with 1% formaldehyde (Sigma-Aldrich) followed by quenching. DNA fragments 500–1000 bp long were generated by shearing chromatin on a BioRuptor (Diagenode). The shared chromatin was incubated with BRD4 antibody (A301–985A; Bethyl) or normal rabbit IgG (negative control; Upstate Biotechnol-ogies Inc.) and recovered with protein A-agarose/salmon sperm DNA (Upstate Biotechnologies Inc.). After reversal, crosslinked, and proteinase K treatment, DNA was extracted and analyzed by quantitative RT-PCR with specific primers (Table 2). Relative enrichment was calculated as the percentage of input DNA for each sample using the formula % input =2 exp[(Ct [unbound] − log2 [unbound dilution factor]) − Ct (bound)] ×100 and normalized to normal rabbit IgG antibody (considered as one).
Table 2.
ChIP primers
| Gene | Forward primers (5′-3′) | Reverse primers (5′-3′) |
|---|---|---|
| Human | ||
| CCL-2 | CCCTCTGCCCGCTTTCAATA | CTTGGGGAATGAAGGTGGCT |
| CCL-5 | ATACCGGCCAATGCTTGGTT | TCCACGTGCTGTCTTGATCC |
| IL-6 | CACCCTCACCCTCCAACAAA | TTCTCTTTCGTTCCCGGTGG |
| Mouse | ||
| Ccl-2 | TGATGCTACTCCTTGGCACC | GGCTTCAGTGAGAGTTGGCT |
| Ccl-5 | CTGCTACCCTGGCTCCCTAT | TGAGGATGATGGTGAGGGCA |
| Il-6 | TGTGGGATTTTCCCATGAGT | TGCCTTCACTTACTTGCAGAGA |
ChIP assays in kidney samples were produced using the High Cell ChIP Kit (Diagenode) following the manufacturer’s instructions using specific primers (Table 2). Results were shown as the n-fold enrichment of anti-BRD4 antibody over a negative control antibody (normal rabbit IgG).
Human Gene Expression Arrays/Profiles
Total RNA was isolated using the RNAqueous Micro Kit (Ambion) according to the manufacturer’s instructions. RNA quality and quantity were assessed using an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA) and a fluorometric Qubit 2.0 Assay (Invitrogen, Carlsbad, CA), respectively. RNA was hybridized to an Illumina Expression Array HumanHT-12, version 3.
Expression data were analyzed using R packages from the Bioconductor project (www.bioconductor.org) and lumi packages.61 For the detection of differentially expressed genes, a linear model was fitted to the microarray data, and empiric Bayes moderated t statistics were calculated using the limma package of Bioconductor.62 Genes with a >1.5-fold change and adjusted P<0.05 were considered significant according to the technical specifications of Illumina. For gene functional analysis of the differentially expressed genes, we used the DAVID Go Web–based tool.63 The raw gene expression data have been submitted to the NCBI Gene Expression Omnibus under accession number GSE71602.
Biochemical Analyses
Serum creatinine levels were determined by the Biochemical Analysis Service at Fundacion Jimenez Diaz Hospital, and data are expressed as milligrams per deciliter. Urinary concentrations of albumin were normalized by creatinine concentrations in individual mice samples. Urinary creatinine levels were determined by the Creatinine Assay Kit (ab65340; Abcam, Inc., Cambridge, MA). Albumin excretion levels were determined by using a mouse Albumin ELISA Kit (ab108792; Abcam, Inc.). Albumin-to-creatinine ratio was expressed as nanograms per microliter.
Statistical Analyses
Results are expressed as the n-fold increase with respect to the control as the mean±SEM. In mouse studies, data were obtained comparing both kidneys (contralateral versus obstructed) in each mouse. Mean results from untreated contralateral kidneys were taken as reference values (control). Differences between groups were assessed by the Mann–Whitney test. Statistical significance was concluded for values of P<0.05. These analyses were done using SPSS, version 16.0 (IBM SPSS, Chicago, IL).
Disclosures
None.
Supplementary Material
Acknowledgments
The bromodomain and extraterminal inhibitor JQ1 was provided collaboratively by Dr. James Bradner (Dana-Farber Cancer Institute, Boston, MA).
This work was supported by the Instituto de Salud Carlos III and Fondo Europeo de Desarrollo Regional grants PI014/0041, PI13/00047, and PI12/02587; Red de Investigación Renal grants RD12/0021/0002, RD12/0021/0021, RD12/0021/001, and RD16/009; Comunidad de Madrid Fibroteam grants S2010/BMD-2321 and S2010/BMD-2378; Plan de Ciencia, Tecnología e Innovación 2013–2017 del Principado de Asturias, grant GRUPIN-14-030; and the Instituto Reina Sofía de Investigación en Nefrología. B.S.-A. was supported by Sara Borrell, A.B.S. was supported by Miguel Servet, and A.O. and C.L.-L. were supported by Intensificación Instituto de Salud Carlos III.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2015080910/-/DCSupplemental.
References
- 1.Dwivedi RS, Herman JG, McCaffrey TA, Raj DS: Beyond genetics: Epigenetic code in chronic kidney disease. Kidney Int 79: 23–32, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Smyth LJ, McKay GJ, Maxwell AP, McKnight AJ: DNA hypermethylation and DNA hypomethylation is present at different loci in chronic kidney disease. Epigenetics 9: 366–376, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Reddy MA, Natarajan R: Recent developments in epigenetics of acute and chronic kidney diseases. Kidney Int 88: 250–261, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dressler GR, Patel SR: Epigenetics in kidney development and renal disease. Transl Res 165: 166–176, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Filippakopoulos P, Picaud S, Mangos M, Keates T, Lambert JP, Barsyte-Lovejoy D, Felletar I, Volkmer R, Müller S, Pawson T, Gingras AC, Arrowsmith CH, Knapp S: Histone recognition and large-scale structural analysis of the human bromodomain family. Cell 149: 214–231, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Boehm D, Calvanese V, Dar RD, Xing S, Schroeder S, Martins L, Aull K, Li PC, Planelles V, Bradner JE, Zhou MM, Siliciano RF, Weinberger L, Verdin E, Ott M: BET bromodomain-targeting compounds reactivate HIV from latency via a Tat-independent mechanism. Cell Cycle 12: 452–462, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu Y, Vakoc CR: Brd4 is on the move during inflammation. Trends Cell Biol 24: 615–616, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shi J, Vakoc CR: The mechanisms behind the therapeutic activity of BET bromodomain inhibition. Mol Cell 54: 728–736, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rahman S, Sowa ME, Ottinger M, Smith JA, Shi Y, Harper JW, Howley PM: The Brd4 extraterminal domain confers transcription activation independent of pTEFb by recruiting multiple proteins, including NSD3. Mol Cell Biol 31: 2641–2652, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shi J, Whyte WA, Zepeda-Mendoza CJ, Milazzo JP, Shen C, Roe JS, Minder JL, Mercan F, Wang E, Eckersley-Maslin MA, Campbell AE, Kawaoka S, Shareef S, Zhu Z, Kendall J, Muhar M, Haslinger C, Yu M, Roeder RG, Wigler MH, Blobel GA, Zuber J, Spector DL, Young RA, Vakoc CR: Role of SWI/SNF in acute leukemia maintenance and enhancer-mediated Myc regulation. Genes Dev 27: 2648–2662, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yang Z, Yik JH, Chen R, He N, Jang MK, Ozato K, Zhou Q: Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol Cell 19: 535–545, 2005 [DOI] [PubMed] [Google Scholar]
- 12.Yang Z, He N, Zhou Q: Brd4 recruits P-TEFb to chromosomes at late mitosis to promote G1 gene expression and cell cycle progression. Mol Cell Biol 28: 967–976, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Devaiah BN, Lewis BA, Cherman N, Hewitt MC, Albrecht BK, Robey PG, Ozato K, Sims RJ 3rd, Singer DS: BRD4 is an atypical kinase that phosphorylates serine2 of the RNA polymerase II carboxy-terminal domain. Proc Natl Acad Sci U S A 109: 6927–6932, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jang MK, Mochizuki K, Zhou M, Jeong HS, Brady JN, Ozato K: The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol Cell 19: 523–534, 2005 [DOI] [PubMed] [Google Scholar]
- 15.Delmore JE, Issa GC, Lemieux ME, Rahl PB, Shi J, Jacobs HM, Kastritis E, Gilpatrick T, Paranal RM, Qi J, Chesi M, Schinzel AC, McKeown MR, Heffernan TP, Vakoc CR, Bergsagel PL, Ghobrial IM, Richardson PG, Young RA, Hahn WC, Anderson KC, Kung AL, Bradner JE, Mitsiades CS: BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146: 904–917, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bandukwala HS, Gagnon J, Togher S, Greenbaum JA, Lamperti ED, Parr NJ, Molesworth AM, Smithers N, Lee K, Witherington J, Tough DF, Prinjha RK, Peters B, Rao A: Selective inhibition of CD4+ T-cell cytokine production and autoimmunity by BET protein and c-Myc inhibitors. Proc Natl Acad Sci U S A 109: 14532–14537, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lovén J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR, Bradner JE, Lee TI, Young RA: Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 153: 320–334, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sanz AB, Sanchez-Niño MD, Ramos AM, Moreno JA, Santamaria B, Ruiz-Ortega M, Egido J, Ortiz A: NF-kappaB in renal inflammation. J Am Soc Nephrol 21: 1254–1262, 2010 [DOI] [PubMed] [Google Scholar]
- 19.Huang B, Yang XD, Zhou MM, Ozato K, Chen LF: Brd4 coactivates transcriptional activation of NF-kappaB via specific binding to acetylated RelA. Mol Cell Biol 29: 1375–1387, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zou Z, Huang B, Wu X, Zhang H, Qi J, Bradner J, Nair S, Chen LF: Brd4 maintains constitutively active NF-κB in cancer cells by binding to acetylated RelA. Oncogene 33: 2395–2404, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu X, Qi J, Bradner JE, Xiao G, Chen LF: Bromodomain and extraterminal (BET) protein inhibition suppresses human T cell leukemia virus 1 (HTLV-1) Tax protein-mediated tumorigenesis by inhibiting nuclear factor κB (NF-κB) signaling. J Biol Chem 288: 36094–36105, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nicodeme E, Jeffrey KL, Schaefer U, Beinke S, Dewell S, Chung CW, Chandwani R, Marazzi I, Wilson P, Coste H, White J, Kirilovsky J, Rice CM, Lora JM, Prinjha RK, Lee K, Tarakhovsky A: Suppression of inflammation by a synthetic histone mimic. Nature 468: 1119–1123, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Asangani IA, Dommeti VL, Wang X, Malik R, Cieslik M, Yang R, Escara-Wilke J, Wilder-Romans K, Dhanireddy S, Engelke C, Iyer MK, Jing X, Wu YM, Cao X, Qin ZS, Wang S, Feng FY, Chinnaiyan AM: Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate cancer. Nature 510: 278–282, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Baratta MG, Schinzel AC, Zwang Y, Bandopadhayay P, Bowman-Colin C, Kutt J, Curtis J, Piao H, Wong LC, Kung AL, Beroukhim R, Bradner JE, Drapkin R, Hahn WC, Liu JF, Livingston DM: An in-tumor genetic screen reveals that the BET bromodomain protein, BRD4, is a potential therapeutic target in ovarian carcinoma. Proc Natl Acad Sci U S A 112: 232–237, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dawson MA, Prinjha RK, Dittmann A, Giotopoulos G, Bantscheff M, Chan WI, Robson SC, Chung CW, Hopf C, Savitski MM, Huthmacher C, Gudgin E, Lugo D, Beinke S, Chapman TD, Roberts EJ, Soden PE, Auger KR, Mirguet O, Doehner K, Delwel R, Burnett AK, Jeffrey P, Drewes G, Lee K, Huntly BJ, Kouzarides T: Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 478: 529–533, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Knoechel B, Roderick JE, Williamson KE, Zhu J, Lohr JG, Cotton MJ, Gillespie SM, Fernandez D, Ku M, Wang H, Piccioni F, Silver SJ, Jain M, Pearson D, Kluk MJ, Ott CJ, Shultz LD, Brehm MA, Greiner DL, Gutierrez A, Stegmaier K, Kung AL, Root DE, Bradner JE, Aster JC, Kelliher MA, Bernstein BE: An epigenetic mechanism of resistance to targeted therapy in T cell acute lymphoblastic leukemia. Nat Genet 46: 364–370, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shimamura T, Chen Z, Soucheray M, Carretero J, Kikuchi E, Tchaicha JH, Gao Y, Cheng KA, Cohoon TJ, Qi J, Akbay E, Kimmelman AC, Kung AL, Bradner JE, Wong KK: Efficacy of BET bromodomain inhibition in Kras-mutant non-small cell lung cancer. Clin Cancer Res 19: 6183–6192, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O, Morse EM, Keates T, Hickman TT, Felletar I, Philpott M, Munro S, McKeown MR, Wang Y, Christie AL, West N, Cameron MJ, Schwartz B, Heightman TD, La Thangue N, French CA, Wiest O, Kung AL, Knapp S, Bradner JE: Selective inhibition of BET bromodomains. Nature 468: 1067–1073, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chaidos A, Caputo V, Karadimitris A: Inhibition of bromodomain and extra-terminal proteins (BET) as a potential therapeutic approach in haematological malignancies: Emerging preclinical and clinical evidence. Ther Adv Hematol 6: 128–141, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Belkina AC, Nikolajczyk BS, Denis GV: BET protein function is required for inflammation: Brd2 genetic disruption and BET inhibitor JQ1 impair mouse macrophage inflammatory responses. J Immunol 190: 3670–3678, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Klein K, Kabala PA, Grabiec AM, Gay RE, Kolling C, Lin LL, Gay S, Tak PP, Prinjha RK, Ospelt C, Reedquist KA: The bromodomain protein inhibitor I-BET151 suppresses expression of inflammatory genes and matrix degrading enzymes in rheumatoid arthritis synovial fibroblasts. Ann Rheum Dis 75: 422–429, 2016 [DOI] [PubMed]
- 32.Zhang QG, Qian J, Zhu YC: Targeting bromodomain-containing protein 4 (BRD4) benefits rheumatoid arthritis. Immunol Lett 166: 103–108, 2015 [DOI] [PubMed] [Google Scholar]
- 33.Tang X, Peng R, Phillips JE, Deguzman J, Ren Y, Apparsundaram S, Luo Q, Bauer CM, Fuentes ME, DeMartino JA, Tyagi G, Garrido R, Hogaboam CM, Denton CP, Holmes AM, Kitson C, Stevenson CS, Budd DC: Assessment of Brd4 inhibition in idiopathic pulmonary fibrosis lung fibroblasts and in vivo models of lung fibrosis. Am J Pathol 183: 470–479, 2013 [DOI] [PubMed] [Google Scholar]
- 34.Spiltoir JI, Stratton MS, Cavasin MA, Demos-Davies K, Reid BG, Qi J, Bradner JE, McKinsey TA: BET acetyl-lysine binding proteins control pathological cardiac hypertrophy. J Mol Cell Cardiol 63: 175–179, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhou X, Fan LX, Peters DJ, Trudel M, Bradner JE, Li X: Therapeutic targeting of BET bromodomain protein, Brd4, delays cyst growth in ADPKD. Hum Mol Genet 24: 3982–3993, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Al-Lamki RS, Mayadas TN: TNF receptors: Signaling pathways and contribution to renal dysfunction. Kidney Int 87: 281–296, 2015 [DOI] [PubMed] [Google Scholar]
- 37.Zuber J, Shi J, Wang E, Rappaport AR, Herrmann H, Sison EA, Magoon D, Qi J, Blatt K, Wunderlich M, Taylor MJ, Johns C, Chicas A, Mulloy JC, Kogan SC, Brown P, Valent P, Bradner JE, Lowe SW, Vakoc CR: RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 478: 524–528, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Anand P, Brown JD, Lin CY, Qi J, Zhang R, Artero PC, Alaiti MA, Bullard J, Alazem K, Margulies KB, Cappola TP, Lemieux M, Plutzky J, Bradner JE, Haldar SM: BET bromodomains mediate transcriptional pause release in heart failure. Cell 154: 569–582, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rodriguez RM, Suarez-Alvarez B, Salvanés R, Huidobro C, Toraño EG, Garcia-Perez JL, Lopez-Larrea C, Fernandez AF, Bueno C, Menendez P, Fraga MF: Role of BRD4 in hematopoietic differentiation of embryonic stem cells. Epigenetics 9: 566–578, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ucero AC, Benito-Martin A, Izquierdo MC, Sanchez-Niño MD, Sanz AB, Ramos AM, Berzal S, Ruiz-Ortega M, Egido J, Ortiz A: Unilateral ureteral obstruction: Beyond obstruction. Int Urol Nephrol 46: 765–776, 2014 [DOI] [PubMed] [Google Scholar]
- 41.Esteban V, Lorenzo O, Rupérez M, Suzuki Y, Mezzano S, Blanco J, Kretzler M, Sugaya T, Egido J, Ruiz-Ortega M: Angiotensin II, via AT1 and AT2 receptors and NF-kappaB pathway, regulates the inflammatory response in unilateral ureteral obstruction. J Am Soc Nephrol 15: 1514–1529, 2004 [DOI] [PubMed] [Google Scholar]
- 42.Alique M, Civantos E, Sanchez-Lopez E, Lavoz C, Rayego-Mateos S, Rodrigues-Díez R, García-Redondo AB, Egido J, Ortiz A, Rodríguez-Puyol D, Rodríguez-Puyol M, Ruiz-Ortega M: Integrin-linked kinase plays a key role in the regulation of angiotensin II-induced renal inflammation. Clin Sci (Lond) 127: 19–31, 2014 [DOI] [PubMed] [Google Scholar]
- 43.Chung AC, Lan HY: Chemokines in renal injury. J Am Soc Nephrol 22: 802–809, 2011 [DOI] [PubMed] [Google Scholar]
- 44.Bollée G, Flamant M, Schordan S, Fligny C, Rumpel E, Milon M, Schordan E, Sabaa N, Vandermeersch S, Galaup A, Rodenas A, Casal I, Sunnarborg SW, Salant DJ, Kopp JB, Threadgill DW, Quaggin SE, Dussaule JC, Germain S, Mesnard L, Endlich K, Boucheix C, Belenfant X, Callard P, Endlich N, Tharaux PL: Epidermal growth factor receptor promotes glomerular injury and renal failure in rapidly progressive crescentic glomerulonephritis. Nat Med 17: 1242–1250, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Esteban V, Ruperez M, Vita JR, López ES, Mezzano S, Plaza JJ, Egido J, Ruiz-Ortega M: Effect of simultaneous blockade of AT1 and AT2 receptors on the NFkappaB pathway and renal inflammatory response. Kidney Int Suppl 86: S33–S38, 2003 [DOI] [PubMed] [Google Scholar]
- 46.Kitching AR, Holdsworth SR: The emergence of TH17 cells as effectors of renal injury. J Am Soc Nephrol 22: 235–238, 2011 [DOI] [PubMed] [Google Scholar]
- 47.Rodrigues-Díez R, Rodrigues-Díez RR, Rayego-Mateos S, Suarez-Alvarez B, Lavoz C, Stark Aroeira L, Sánchez-López E, Orejudo M, Alique M, Lopez-Larrea C, Ortiz A, Egido J, Ruiz-Ortega M: The C-terminal module IV of connective tissue growth factor is a novel immune modulator of the Th17 response. Lab Invest 93: 812–824, 2013 [DOI] [PubMed] [Google Scholar]
- 48.Pindjakova J, Hanley SA, Duffy MM, Sutton CE, Weidhofer GA, Miller MN, Nath KA, Mills KH, Ceredig R, Griffin MD: Interleukin-1 accounts for intrarenal Th17 cell activation during ureteral obstruction. Kidney Int 81: 379–390, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dekker FJ, van den Bosch T, Martin NI: Small molecule inhibitors of histone acetyltransferases and deacetylases are potential drugs for inflammatory diseases. Drug Discov Today 19: 654–660, 2014 [DOI] [PubMed] [Google Scholar]
- 50.Gupta J, Kumar S, Li J, Krishna Murthy Karuturi R, Tikoo K: Histone H3 lysine 4 monomethylation (H3K4me1) and H3 lysine 9 monomethylation (H3K9me1): Distribution and their association in regulating gene expression under hyperglycaemic/hyperinsulinemic conditions in 3T3 cells. Biochimie 94: 2656–2664, 2012 [DOI] [PubMed] [Google Scholar]
- 51.Brown JD, Lin CY, Duan Q, Griffin G, Federation AJ, Paranal RM, Bair S, Newton G, Lichtman AH, Kung AL, Yang T, Wang H, Luscinskas FW, Croce KJ, Bradner JE, Plutzky J: NF-κB directs dynamic super enhancer formation in inflammation and atherogenesis. Mol Cell 56: 219–231, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Amir-Zilberstein L, Ainbinder E, Toube L, Yamaguchi Y, Handa H, Dikstein R: Differential regulation of NF-kappaB by elongation factors is determined by core promoter type. Mol Cell Biol 27: 5246–5259, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Barboric M, Nissen RM, Kanazawa S, Jabrane-Ferrat N, Peterlin BM: NF-kappaB binds P-TEFb to stimulate transcriptional elongation by RNA polymerase II. Mol Cell 8: 327–337, 2001 [DOI] [PubMed] [Google Scholar]
- 54.Berger SL: Gene activation by histone and factor acetyltransferases. Curr Opin Cell Biol 11: 336–341, 1999 [DOI] [PubMed] [Google Scholar]
- 55.Bettelli E, Korn T, Oukka M, Kuchroo VK: Induction and effector functions of T(H)17 cells. Nature 453: 1051–1057, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Isailovic N, Daigo K, Mantovani A, Selmi C: Interleukin-17 and innate immunity in infections and chronic inflammation. J Autoimmun 60: 1–11, 2015 [DOI] [PubMed] [Google Scholar]
- 57.Mease PJ, Genovese MC, Greenwald MW, Ritchlin CT, Beaulieu AD, Deodhar A, Newmark R, Feng J, Erondu N, Nirula A: Brodalumab, an anti-IL17RA monoclonal antibody, in psoriatic arthritis. N Engl J Med 370: 2295–2306, 2014 [DOI] [PubMed] [Google Scholar]
- 58.Park TY, Park SD, Cho JY, Moon JS, Kim NY, Park K, Seong RH, Lee SW, Morio T, Bothwell AL, Lee SK: RORγt-specific transcriptional interactomic inhibition suppresses autoimmunity associated with TH17 cells. Proc Natl Acad Sci U S A 111: 18673–18678, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mele DA, Salmeron A, Ghosh S, Huang HR, Bryant BM, Lora JM: BET bromodomain inhibition suppresses TH17-mediated pathology. J Exp Med 210: 2181–2190, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bäckdahl L, Bushell A, Beck S: Inflammatory signalling as mediator of epigenetic modulation in tissue-specific chronic inflammation. Int J Biochem Cell Biol 41: 176–184, 2009 [DOI] [PubMed] [Google Scholar]
- 61.Du P, Kibbe WA, Lin SM: Lumi: A pipeline for processing Illumina microarray. Bioinformatics 24: 1547–1548, 2008 [DOI] [PubMed] [Google Scholar]
- 62.Smyth GK: Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3: 3, 2004 [DOI] [PubMed]
- 63.Huang W, Sherman BT, Lempicki RA: Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4: 44–57, 2009 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.