ABSTRACT
Herpes simplex virus 1 (HSV-1) establishes latency in neural tissues of immunocompetent mice but persists in both peripheral and neural tissues of lymphocyte-deficient mice. Thymidine kinase (TK) is believed to be essential for HSV-1 to persist in neural tissues of immunocompromised mice, because infectious virus of a mutant with defects in both TK and UL24 is detected only in peripheral tissues, but not in neural tissues, of severe combined immunodeficiency mice (T. Valyi-Nagy, R. M. Gesser, B. Raengsakulrach, S. L. Deshmane, B. P. Randazzo, A. J. Dillner, and N. W. Fraser, Virology 199:484–490, 1994, https://doi.org/10.1006/viro.1994.1150). Here we find infiltration of CD4 and CD8 T cells in peripheral and neural tissues of mice infected with a TK-negative mutant. We therefore investigated the significance of viral TK and host T cells for HSV-1 to persist in neural tissues using three genetically engineered mutants with defects in only TK or in both TK and UL24 and two strains of nude mice. Surprisingly, all three mutants establish persistent infection in up to 100% of brain stems and 93% of trigeminal ganglia of adult nude mice at 28 days postinfection, as measured by the recovery of infectious virus. Thus, in mouse neural tissues, host T cells block persistent HSV-1 infection, and viral TK is dispensable for the virus to establish persistent infection. Furthermore, we found 30- to 200-fold more virus in neural tissues than in the eye and detected glycoprotein C, a true late viral antigen, in brainstem neurons of nude mice persistently infected with the TK-negative mutant, suggesting that adult mouse neurons can support the replication of TK-negative HSV-1.
IMPORTANCE Acyclovir is used to treat herpes simplex virus 1 (HSV-1)-infected immunocompromised patients, but treatment is hindered by the emergence of drug-resistant viruses, mostly those with mutations in viral thymidine kinase (TK), which activates acyclovir. TK mutants are detected in brains of immunocompromised patients with persistent infection. However, answers to the questions as to whether TK-negative (TK−) HSV-1 can establish persistent infection in brains of immunocompromised hosts and whether neurons in vivo are permissive for TK− HSV-1 remain elusive. Using three genetically engineered HSV-1 TK− mutants and two strains of nude mice deficient in T cells, we found that all three HSV-1 TK− mutants can efficiently establish persistent infection in the brain stem and trigeminal ganglion and detected glycoprotein C, a true late viral antigen, in brainstem neurons. Our study provides evidence that TK− HSV-1 can persist in neural tissues and replicate in brain neurons of immunocompromised hosts.
KEYWORDS: herpes simplex virus 1, mice, neural tissues, persistent infection, thymidine kinase
INTRODUCTION
Herpes simplex virus 1 (HSV-1) infects more than 80% of adults worldwide (1, 2). During primary infection, HSV-1 replicates in peripheral tissues such as the skin and eye. The virus then spreads to peripheral sensory ganglia, such as trigeminal ganglia (TG), and the central nervous system (CNS) before establishing lifelong latency maintained by the presence of viral genomes in neurons (1). Latent virus can reactivate to cause recurrent infection. Both primary and recurrent infections can induce serious morbidity and even mortality, especially in immunocompromised patients such as those with genetic immunodeficiency, AIDS, transplantation-associated immunosuppression, or cancer chemotherapy (3–5). Acyclovir (ACV) and related nucleoside analogs are commonly used for patient treatment. However, ACV treatment is hindered by the emergence of drug-resistant viruses, which are estimated to occur in ∼6% of treated immunocompromised patients (3, 6). ACV resistance has also been reported in immunocompetent patients with eye infections (7, 8). The majority (>90%) of ACV-resistant clinical isolates contain mutations in the thymidine kinase (tk) gene, which encodes the protein that activates ACV (3, 5).
Genetically engineered, TK-negative (TK−) mutants have been constructed to study the importance of TK in HSV-1 infection. By using TK− mutants, in vitro studies showed that TK is dispensable for viral replication in cells, including sensory neurons cultured from dorsal root ganglia of rat embryos (9, 10). However, in vivo studies found that TK is required for viral replication in neural tissues (TG and brain), but not in peripheral tissues, of immunocompetent adult mice (4, 11–14). Therefore, it is generally believed that adult neurons are postmitotic cells that do not express detectable levels of cellular TK, which presumably supports TK− HSV-1 to replicate in dividing cells (10, 15). This notion is supported by our work showing that the replacement of viral tk with cellular tk enables HSV-1 to replicate in TG (16) and that providing TK activity in trans by coinfection with wild-type virus enables the TK− mutant to replicate in TG (17). In addition, the absence of TK activity inhibits HSV-1 reactivation (11, 12).
During wild-type HSV-1 infection in immunocompromised mice, such as those deficient in lymphocytes, high virus doses induce death, while inoculation of low virus doses in peripheral tissues enables the virus to establish persistent infection in both peripheral and neural tissues (18–20). A few reports have investigated TK− HSV-1 infection in immunocompromised mice, particularly one study using a TK− mutant (dlsptk) derived from HSV-1 strain KOS to infect severe combined immunodeficiency (SCID) mice deficient in both functional T and B cells (21). In that study, infectious virus was recovered from the eye and skin, but not from the TG and brain, of mice infected for 30 days. Additionally, viral antigens and the mRNAs of viral immediate early, early, and late genes (ICP27, VP5, and gC) were detected in TG but not in the brain. That study therefore concluded that TK− HSV-1 establishes persistent infection in SCID mice with uncontrolled peripheral replication but only marginal nervous system involvement. Therefore, it is generally believed that TK is required for HSV-1 to establish persistent infection in neural tissues of immunocompromised hosts. The detection of ACV-resistant TK mutants in the CNS of immunocompromised patients with persistent infection (22) may be due to their residual TK activity or the TK activity provided in trans by the wild-type virus. Later, it was shown that a deletion in dlsptk that inactivates TK reduces the expression of an adjacent gene (UL24) that also promotes productive ganglionic infection in immunocompetent mice like tk (23, 24). A better understanding of TK− HSV-1 infection in neural tissues of immunocompromised hosts is important in at least two aspects. ACV-resistant HSV-1 infection in immunocompromised patients with neurological diseases has been an increasingly important problem in the clinic (25). The successful use of TK− HSV-1 for experimental therapies of several different intracranial tumors of laboratory animals has been reported (26, 27). Therefore, we reinvestigated this issue using three HSV-1 TK− mutants, including dlsptk, and two backgrounds of nude mice and discovered that all three HSV-1 TK− mutants can efficiently establish persistent infection in the TG and brain stem of nude mice, as measured by the recovery of infectious virus.
RESULTS
T cells infiltrate neural tissues of wild-type mice infected with TK− HSV-1.
We first used the HSV-1 TK− mutant (tkLTRZ1), an insertion mutant constructed from KOS with intact UL24 expression (11), for study. Viral replication in tissues of ICR mice infected with 1 × 107 PFU/eye of tkLTRZ1 or KOS in both eyes was assessed. After infection with KOS, infectious virus was detected in the eye, TG, and brain (Fig. 1). In the eye on days 1, 3, 5, and 7 postinfection (p.i.), average tkLTRZ1 titers were about 3.8 × 105, 4.7 × 103, 1.7 × 103, and 2 PFU/tissue, and average KOS titers were about 1.7 × 106, 1.6 × 104, 2.5 × 103, and 40 PFU/tissue, respectively (Fig. 1A). The replication kinetics of tkLTRZ1 and KOS were similar but with slightly reduced tkLTRZ1 titers from days 1 to 7 p.i. In the TG, average KOS titers were about 0, 3.9 × 104, 2.8 × 104, and 15 PFU/tissue on days 1, 3, 5 and 7, respectively, with a peak on day 3 p.i. (Fig. 1B). In the TG of tkLTRZ1-infected mice, infectious virus was detected only on day 3, but not on days 1, 5, and 7 p.i., in 25% (2/8) of samples and with an average titer of 5 PFU/TG. In the brain stem, average KOS titers were about 11, 1.2 × 104, and 0 PFU/tissue on days 3, 5, and 7, respectively, with a peak on day 5 p.i. (Fig. 1C). Infectious virus was not detected in five brain stems harvested from tkLTRZ1-infected mice on day 5 p.i. The result of infection by TK-defective HSV-1 with impaired growth in neural tissues of immunocompetent mice is consistent with data from previous reports using tkLTRZ1 or TK− mutants derived from KOS or other HSV-1 strains (4, 11–13). Mouse brain stems were examined because high titers of wild-type HSV-1 are detected in this region compared with other CNS regions, such as frontal cortex, cerebellum, and spinal cord, of mice (28) and because TK− HSV-1 is attenuated for neurovirulence, with reduced virus production in the mouse brain stem (14, 29).
FIG 1.
Acute replication of HSV-1 in ICR mice. Viral titers in the eyes (A), trigeminal ganglia (B and D), and brain stems (C and E) of ICR wild-type (WT) and nude mice infected with HSV-1 strain KOS or tkLTRZ1 for the indicated times are shown. (A to C) The data represent means ± SE (error bars) for >3 samples per data point. For panels D and E, each point on the scattergram represents an individual sample, and the horizontal lines represent the mean values for each group.
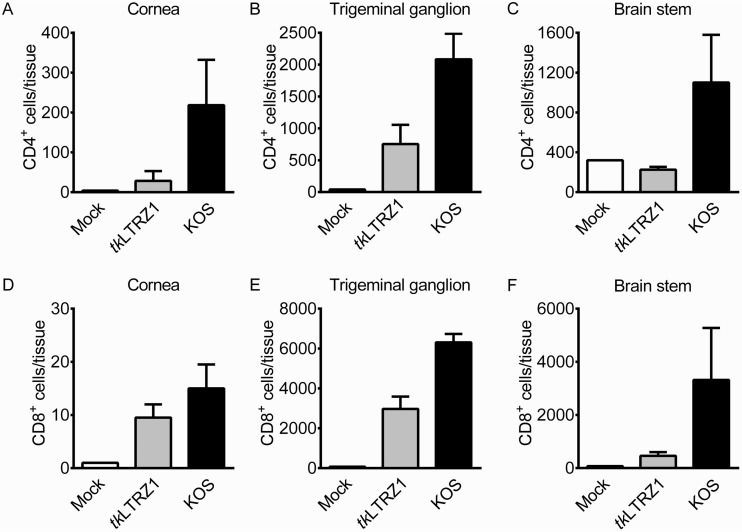
T cells have been shown to infiltrate and inhibit wild-type HSV-1 infection in mouse tissues (30, 31). Very few studies have measured T cells in TK− HSV-1-infected mice, so we quantified T cells in tissues of tkLTRZ1-infected mice. Flow cytometric analysis showed that the numbers of CD4+ T cells in the cornea and TG and the numbers of CD8+ T cells in the cornea, TG, and brain stem of tkLTRZ1-infected mice were elevated compared with those in mock-infected mice on day 7 p.i. (Fig. 2). The levels of CD4+ and CD8+ T cells in the cornea, TG, and brain stem of tkLTRZ1-infected mice were lower than those in KOS-infected mice.
FIG 2.
Presence of T cells in tissues of ICR mice infected with HSV-1. The indicated tissues of mice that were mock infected or infected with HSV-1 strain KOS or tkLTRZ1 for 7 days were harvested to quantify cells positive for CD4 (A to C) or CD8 (D to F) by flow cytometry. The data represent means + SE (error bars) for >3 samples per group, with samples being derived from individual mice.
TK− HSV-1 can efficiently establish persistent infection in neural tissues of nude mice.
The presence of both CD4+ and CD8+ T cells in tissues of infected wild-type ICR mice prompted us to investigate the influence of T cells on TK− HSV-1 infection using ICR nude mice deficient in T cells. We monitored viral replication in nude mice inoculated with 1 × 107 PFU/eye of the virus in both eyes. In the eye on days 1, 3, and 7 p.i., average KOS titers were about 4.5 × 105, 1.9 × 104, and 1.2 × 104 PFU/tissue, and average tkLTRZ1 titers were about 1.8 × 105, 140, and 0 PFU/tissue, respectively (Fig. 1A). In the TG of KOS-infected mice, average viral titers were about 384, 2.1 × 105, and 3.0 × 104 PFU/tissue on days 1, 3, and 7 p.i., respectively (Fig. 1B). In tkLTRZ1-infected mice, infectious virus was detected in 29% (4/14) and 25% (4/16) of TG on days 3 and 7 p.i., respectively, with average titers of 58 and 8 PFU/tissue (Fig. 1B and D). In the brain stem of KOS-infected mice, average viral titers were about 0, 3.7 × 103, and 1.4 × 104 PFU/tissue on days 1, 3, and 7 p.i., respectively (Fig. 1C). Intriguingly, in tkLTRZ1-infected mice, infectious virus was detected in 25% (1/4) and 100% (3/3) of brain stems on days 3 and 7 p.i., respectively, with average titers of 4 and 12 PFU/tissue (Fig. 1C and E). Results for tkLTRZ1 obtained from ICR wild-type and nude mice showed the following. In the eye, the replication kinetics of tkLTRZ1 in nude and wild-type mice were similar. Notably, tkLTRZ1 titers in nude mice were decreased by about 2- and 34-fold on days 1 and 3 p.i., respectively, compared with those of wild-type mice (Fig. 1A). Although T-cell deficiency fails to increase tkLTRZ1 titers in the eye, it increases tkLTRZ1 titers in mouse neural tissues.
In immunocompetent mice infected with wild-type or TK− HSV-1, infectious virus is cleared by about 10 days p.i. However, in SCID mice infected with dlsptk for 30 days, while infectious virus was detected in the eye, but not in the TG and brain, viral mRNA and antigens associated with productive infection were detected in TG (21). In our study, all tkLTRZ1-infected nude mice survived. We harvested mouse tissues on day 28 p.i. to determine viral titers. Low levels of virus ranging from 1 to 7 PFU/eye were detected in 15% (4/26) of eyes, with an average titer of about 1 PFU/eye (Fig. 3A and Table 1). Notably, infectious virus was detected in 46% (12/26) of TG and 85% (11/13) of brain stems, with average titers of 31 and 233 PFU/tissue, respectively. The viral titers and percentages of persistent infection detected in neural tissues were significantly higher than those in the eye (P < 0.05). Moreover, the viral titers and percentages of persistent infection detected in the brain stem were significantly higher than those in the TG (P < 0.05).
FIG 3.

Persistent infection by tkLTRZ1 in ICR nude mice. (A) Viral titers in the eyes, trigeminal ganglia, and brain stems of mice infected with HSV-1 strain tkLTRZ1 for 28 days are shown. Each point on the scattergram represents an individual sample, and the horizontal lines represent the mean values for each group. BD, below the detection limit. **, P < 0.01; ***, P < 0.001. (B and C) Brain stems of mock-infected or infected mice were harvested at 28 days postinfection and stained with antibodies against HSV-1 (B) or with antibodies against HSV-1 glycoprotein C (gC) or the neuron-specific marker NeuN or with Hoechst dye for DNA in nucleus (C). Data are representative of results from at least 3 samples per group from two independent experiments.
TABLE 1.
Persistent TK− HSV-1 infection in tissues of nude mice
| Tissue examined | % infection (PFU/tissue)a |
|||
|---|---|---|---|---|
| ICR mice infected with TK− strain: |
BALB/c mice infected with TK− strain 294dlTKA | |||
| tkLTRZ1 | dlsptk | 294dlTKA | ||
| Eye | 15 (1) | 45 (323) | 72 (1,726) | 93 (5,068) |
| TG | 46 (31) | 50 (32) | 66 (44) | 93 (174) |
| Brain stem | 85 (233) | 100 (157) | 100 (358) | 71 (5) |
The eyes, TG, and brain stems of ICR or BALB/c nude mice infected with tkLTRZ1, dlsptk, or 294dlTKA were harvested 28 days after infection to assay for the virus. Results are the percentages of tissue with persistent infection, and the PFU of virus per tissue are shown in parentheses. The P value was <0.05 for percentages of persistent infection between tissues of ICR nude mice: the eye versus the TG of tkLTRZ1-infected mice, the TG versus the brain stem of tkLTRZ1-infected mice, the eye versus the brain stem of tkLTRZ1-infected mice, the eye versus the brain stem of dlsptk-infected mice, and the TG versus the brain stem of dlsptk-infected mice.
We failed to detect infectious virus in the lungs, livers, and sera of five ICR nude mice with tkLTRZ1 present in their brain stems on day 28 p.i., suggesting the failure of TK− HSV-1 to establish systemic infection or to spread by viremia in nude mice. We performed staining to detect HSV-1 antigens and cells expressing viral antigens in five brainstem samples from nude mice infected with tkLTRZ1 for 28 days. Immunohistochemical staining detected viral antigens and damage in three samples, with two shown in Fig. 3B. Immunofluorescence double staining using antibodies against mouse NeuN (a neuron-specific protein in the nucleus) or HSV-1 glycoprotein C, a true late antigen, detected some neurons in the brain stem expressing glycoprotein C (Fig. 3C).
We also tested infection of ICR nude mice with lower doses of tkLTRZ1. In mice infected with a 10-fold-lower dose (1 × 106 PFU/eye in both eyes), infectious virus was detected in 38% (3/8) of TG and 75% (3/4) of brain stems, with average titers of 160 and 127 PFU/tissue, respectively, but not in the eye (n = 8) on day 28 p.i. In mice infected with 1 × 103 PFU/eye in both eyes, infectious virus was not detected in eyes, TG, and brain stems of five mice on day 28 p.i. These results reveal that the frequencies of persistent infection established by tkLTRZ1 in mouse tissues are positively correlated with the inoculum dose.
Because tkLTRZ1 efficiently establishes persistent infection in neural tissues of nude mice, we also tested dlsptk. Surprisingly, infectious virus was detected in 45% (11/24) of eyes, 50% (12/24) of TG, and 100% (12/12) of brain stems, with average titers of 323, 32, and 157 PFU/tissue, respectively, in ICR nude mice inoculated with 6 × 106 to 1 × 107 PFU/eye of the virus in both eyes for 28 days (Table 1 and Fig. 4). Similarly to tkLTRZ1, dlsptk established persistent infection in the brain stem, with percentages being significantly higher than those detected in the TG and eye (P < 0.05) (Table 1) and with viral titers being significantly higher than those detected in TG (P < 0.001) (Fig. 4).
FIG 4.
Persistent infection by dlsptk in ICR nude mice. Viral titers in the eyes, trigeminal ganglia, and brain stems of mice infected with HSV-1 strain dlsptk for 28 days are shown. Each point on the scattergram represents an individual sample, and the horizontal lines represent the mean values for each group. BD, below the detection limit. ***, P < 0.001.
Our additional studies found that the mortality rates of ICR nude mice infected with 1 × 104 or 1 × 103 PFU/eye of KOS in both eyes were 100% (5/5) and 20% (1/5), respectively. We harvested tissues from four surviving mice infected with 1 × 103 PFU/eye of KOS to measure viral titers on day 28 p.i. Infectious virus was detected in 50% (4/8), 50% (4/8), and 25% (1/4) of eyes, TG, and brain stems, respectively, with average titers of <1 PFU/tissue.
We next assessed whether a TK− mutant derived from another HSV-1 strain can also establish persistent infection in neural tissues of nude mice using 294dlTKA, which is genetically engineered from strain 294.1, has a similar deletion, and also displays impaired replication in the TG of immunocompetent mice like dlsptk (13). In ICR nude mice inoculated with 1 × 107 PFU/eye of 294dlTKA in both eyes for 28 days, infectious virus was detected in 72% (13/18) of eyes, 66% (12/18) of TG, and 100% (9/9) of brain stems, with average titers of 1,726, 44, and 358 PFU/tissue, respectively (Table 1 and Fig. 5A). Viral titers detected in the brain stem were significantly higher than those detected in the TG (P < 0.01). Results with tkLTRZ1, dlsptk, and 294dlTKA show that ICR nude mice support persistent TK− HSV-1 infection, with high percentages in the brain stem (85 to 100%) compared to the TG (46 to 66%) and eye (15 to 72%).
FIG 5.
Persistent infection by 294dlTKA in ICR or BALB/c nude mice. Viral titers in the eyes, trigeminal ganglia, and brain stems of ICR nude mice (A) or BALB/c nude mice (B) infected with HSV-1 strain 294dlTKA for 28 days are shown. Each point on the scattergram represents an individual sample, and the horizontal lines represent the mean values for each group. BD, below the detection limit. **, P < 0.01; ***, P < 0.001.
ICR mice are an outbred strain, so we also examined nude mice generated in the inbred strain BALB/c for supporting persistent infection by TK− HSV-1 in neural tissues. In BALB/c nude mice inoculated with 1 × 107 PFU/eye of 294dlTKA in both eyes for 28 days, infectious virus was detected in 93% (13/14) of eyes, 93% (13/14) of TG, and 71% (5/7) of brain stems, with average titers of 5,068, 174, and 5 PFU/tissue, respectively (Table 1 and Fig. 5B). Although infectious virus was detected five brain stems, the tissue viral titers were low, with 34 PFU in one specimen and 1 PFU/sample in the other four specimens. Viral titers detected in the eye were significantly higher than those detected in TG (P < 0.01), and viral titers detected in TG were significantly higher than those detected in the brain stem (P < 0.01). Our results for three TK mutants and two mouse strains collectively show that the persistent infection established by TK− HSV-1 in neural tissues of nude mice is not specific to a particular TK− mutant or background of mice.
DISCUSSION
It is generally believed that TK is required for HSV-1 to persist in neural tissues of immunocompromised mice, because Valyi-Nagy et al. found infectious virus in the eye, but not in the TG and brain stem, of SCID mice 30 days after infection with dlsptk (21). Few studies question this assumption because it is consistent with the finding that TK is needed for HSV-1 to replicate in neural tissues of immunocompetent mice (4, 11–13). Here we discover that all three HSV-1 TK− mutants tested (at an inoculum dose of ≥1 × 106 PFU/eye) can efficiently establish persistent infection in neural tissues of adult nude mice. Thus, in mouse neural tissues, viral TK is dispensable for HSV-1 to establish persistent infection, and host T cells prevent TK− mutants from establishing persistent infection. Additionally, our tkLTRZ1 results showing more virus in neural tissues than in the eye and the presence of the viral true late antigen glycoprotein C in brainstem neurons during persistent infection suggest that neurons of adult nude mice are permissive for TK− HSV-1.
Valyi-Nagy et al. used 2 × 106 PFU/eye of dlsptk to infect SCID mice derived from a C.B-17 [BALB/c.C57BL/Ka-Igh-1b/ICR(N17F13)] background without an indication of whether one or both eyes were inoculated (21). The dlsptk dose used in our study may be 3- to 10-fold higher than that used by Valyi-Nagy et al. As our tkLTRZ1 results reveal that the frequencies of persistent infection detected in all three mouse tissues and the viral titers detected in the eye and brain stem are correlated with the amount of input virus, the low viral dose used by Valyi-Nagy et al. may explain the failure to detect persistent infection in mouse neural tissues. Although we observed that both ICR and BALB/c nude mice support persistent infection by 294dlTKA in neural tissues, we cannot exclude the possibility that C.B-17 SCID mice may not support persistent infection by dlsptk in neural tissues. Notably, the results for 294dlTKA obtained from ICR and BALB/c nude mice show that these two mouse strains vary in their capacities to support persistent infection in the brain stem, with a significantly high viral titer being detected in ICR nude mice by 71-fold (P < 0.001). In addition to nude mice, wild-type ICR and BALB/c mice also differ in their abilities to support wild-type HSV-1 (KOS) replication in the brain stem, with a high viral titer being found in ICR mice (32). The genetic basis for the variation in the abilities of mouse strains to support HSV-1 replication in the brain stem needs further investigation.
In addition to persistent infection, we investigated the effect of T cells on the acute replication of TK− HSV-1 in mouse tissues due to the paucity of studies on this issue. T-cell deficiency fails to increase TK− HSV-1 (tkLTRZ1) titers in the eye during acute infection. Although acute tkLTRZ1 replication in neural tissues of nude mice was still severely impaired, T-cell deficiency increased the viral titer in TG by about 11-fold on day 3 p.i., enabled the virus to persist in some TG on day 5 p.i., and allowed the virus spread to (or viral replication in) the brain. Accordingly, T-cell deficiency promotes the acute replication of TK− HSV-1 in neural tissues but not in the peripheral tissue.
We also compared KOS results obtained from ICR wild-type and nude mice during acute infection. In the eye, T-cell deficiency reduced the average viral titer on day 1 p.i. by about 4-fold but increased average viral titers on days 3 and 7 p.i. by about 1.2- and 304-fold, respectively. Accordingly, T-cell deficiency failed to increase the peak viral titer on day 1 p.i. but prevented virus clearance from days 3 to 7 p.i. In TG, T-cell deficiency increased average viral titers on days 1, 3, and 7 p.i. by about 384-, 5-, and 2,024-fold, respectively. In the brain stem, T-cell deficiency increased average viral titers on days 3 and 7 p.i. by about 336- and 14,170-fold, respectively. Based on these results, T-cell deficiency promotes the acute replication of wild-type HSV-1 in both peripheral and neural tissues. Collectively, we show the significance of T cells in the clearance of HSV-1 from mouse tissues.
T cells can target neurons to block the reactivation of wild-type HSV-1 from latency in mouse TG by several different mechanisms, such as secreting interferon gamma or noncytotoxic lytic granules (31, 33, 34). The exact mechanism that renders TK− HSV-1 susceptible to T-cell scrutiny in mouse neural tissues remains to be elucidated. In the brain stem of ICR nude mice infected with tkLTRZ1, we found less virus on days 3 and 7 p.i. than on day 28 p.i., by 58- and 19-fold, respectively. During acute infection, HSV-1 can induce innate antiviral effectors, such as interferons (35). However, the induction of type I interferon is very transient, with a peak serum level at 6 h p.i., which then declines substantially at 24 h p.i., before abundant virus can be detected in tissues of wild-type mice (35). In nude mice, interferon production is also transient and declines to a basal level comparable to that in wild-type mice 24 h after infection with Newcastle disease virus (36). The transient induction of host innate responses may explain the reduced viral titer detected during acute infection compared to that detected during persistent infection. Our previous in vitro study (37) showing that tkLTRZ1 is susceptible to treatments with type I interferons (interferon alpha or beta) and especially the combination of type I (interferon beta) and type II (interferon gamma) interferons supports this possibility.
In the clinic, ACV-resistant HSV-1 deficient in TK poses a significant problem in immunocompromised patients with persistent infection. Our present mouse study finds that TK− HSV-1 can persist and cause damage in the brain of nude mice. In humans, T cells may also play a significant role in the clearance of HSV-1 from the brain, and TK− HSV-1 may be capable of establishing persistent infection in the brain of immunocompromised patients to cause damage. Previous reports on the failure of TK− HSV-1 to replicate and persist in neural tissues of immunocompetent and immunocompromised mice have promoted the use of TK− mutants as a vector for gene therapy for neurons (26, 27, 38). TK-negative mutants are not currently used for these approaches, in part because they are neurovirulent (27, 29). In addition to drug resistance and neurovirulence, here we uncover another pathogenic property of TK− HSV-1, the capacity to establish persistent infection in neural tissues of immunocompromised mice. Moreover, the present study establishes a model with persistent ACV-resistant virus infection in mouse neural tissues, which provides a system for testing anti-HSV-1 therapies.
MATERIALS AND METHODS
Cells, viruses, and mice.
Vero cells were maintained and propagated according to instructions provided by the American Type Culture Collection. HSV-1 strain KOS, KOS-derived TK− mutants (tkLTRZ1 and dlsptk) (12, 38), and the HSV-1 strain 294.1-derived TK deletion mutant 294dlTKA (13) were propagated and titrated on Vero cell monolayers. The TK activities of dlsptk and tkLTRZ1 are below the level of detection, as measured by a plaque autoradiography assay (12, 13). Using a plaque radiography assay, we failed to detect the TK activity of 294dlTKA, which has a 400-bp deletion in tk similar to that of dlsptk. ICR mice were purchased from The Jackson Laboratory (Bar Harbor, ME), and ICR nude (Narl:ICR-Foxn1nu) mice deficient in T cells because of athymia due to mutations in the transcription factor gene Foxn1 (39) were purchased from Charles River Laboratories International, Inc. ICR and ICR nude mice were maintained under specific-pathogen-free conditions in the laboratory animal center of our college. BALB/c nude (BALB/cAnN.Cg-Foxn1nu/CrlNarl) mice purchased from Charles River Laboratories International, Inc. (Japan), were maintained in the National Laboratory Animal Center of Taiwan.
Infection of mice.
All protocols for mouse experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of National Cheng Kung University (IACUC approval no. 101149). Six- to eight-week-old mice were anesthetized and inoculated with the virus in both eyes following scarification of the cornea with a needle 20 times. Mouse corneas remained clear after infection.
Measurement of viral titers.
Mouse eyeballs, TG, and brain stems were harvested and frozen at −80°C. Tissues were thawed, homogenized, and frozen again. Tissue homogenates were thawed, sonicated, centrifuged, and titrated for infectious virus by a plaque assay on Vero cell monolayers overlaid with medium containing methylcellulose. For plaque assays, tissue samples harvested during acute infection were incubated for 3 days, and tissue samples harvested 28 days after infection were incubated for 4 days.
Flow cytometry.
Mouse eyes, TG, and brain stems were processed and assayed as described in our previous reports (40, 41), using antibodies against mouse CD4 (clone GK1.5; eBioscience) or CD8a (clone 53-6.7; eBioscience).
Immunohistochemical and immunofluorescence staining.
Mouse brain stems were processed and stained as described in our previous reports (40, 42), using Hoechst dye for DNA and antibodies against HSV-1 (Dako), HSV-1 glycoprotein C (clone 3G9; Abcam), or mouse NeuN (clone A60; Millipore).
Statistical analyses.
Data are expressed as means ± standard errors (SE). For statistical comparison, tissue viral titers were analyzed by a Mann-Whitney U test, and the percentages of tissue with persistent infection were analyzed by Fisher's exact test.
ACKNOWLEDGMENTS
We thank Donald Coen for providing viruses used in this study and helpful suggestions, Robert Anderson for critical readings of the manuscript, and Robert Lausch for helpful suggestions.
This work was supported by a grant from the Ministry of Science and Technology in Taiwan (MOST 105-2320-B-006-024-MY3) and by funding from the Infectious Disease and Signal Research Center of National Cheng Kung University.
REFERENCES
- 1.Roizman B, Knipe DM, Whitley RJ. 2013. Herpes simplex viruses, p 1823–1897. In Knipe DM, Howley PM, Cohen JI, Griffin DE, Lamb RA, Martin MA, Racaniello VR, Roizman B (ed), Fields virology, 6th ed, vol 2 Lippincott Williams & Wilkins, Philadelphia, PA. [Google Scholar]
- 2.Smith JS, Robinson NJ. 2002. Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review. J Infect Dis 186:S3–S28. doi: 10.1086/343739. [DOI] [PubMed] [Google Scholar]
- 3.Coen DM, Schaffer PA. 2003. Antiherpesvirus drugs: a promising spectrum of new drugs and drug targets. Nat Rev Drug Discov 2:278–288. doi: 10.1038/nrd1065. [DOI] [PubMed] [Google Scholar]
- 4.Horsburgh BC, Chen SH, Hu A, Mulamba GB, Burns WH, Coen DM. 1998. Recurrent acyclovir-resistant herpes simplex in an immunocompromised patient: can strain differences compensate for loss of thymidine kinase in pathogenesis? J Infect Dis 178:618–625. doi: 10.1086/515375. [DOI] [PubMed] [Google Scholar]
- 5.Stranska R, van Loon AM, Polman M, Beersma MF, Bredius RG, Lankester AC, Meijer E, Schuurman R. 2004. Genotypic and phenotypic characterization of acyclovir-resistant herpes simplex viruses isolated from haematopoietic stem cell transplant recipients. Antivir Ther 9:565–575. [PubMed] [Google Scholar]
- 6.Christophers J, Clayton J, Craske J, Ward R, Collins P, Trowbridge M, Darby G. 1998. Survey of resistance of herpes simplex virus to acyclovir in northwest England. Antimicrob Agents Chemother 42:868–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bacon TH, Levin MJ, Leary JJ, Sarisky RT, Sutton D. 2003. Herpes simplex virus resistance to acyclovir and penciclovir after two decades of antiviral therapy. Clin Microbiol Rev 16:114–128. doi: 10.1128/CMR.16.1.114-128.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Duan R, de Vries RD, Osterhaus AD, Remeijer L, Verjans GM. 2008. Acyclovir-resistant corneal HSV-1 isolates from patients with herpetic keratitis. J Infect Dis 198:659–663. doi: 10.1086/590668. [DOI] [PubMed] [Google Scholar]
- 9.Jamieson AT, Gentry GA, Subak-Sharpe JH. 1974. Induction of both thymidine and deoxycytidine kinase activity by herpes viruses. J Gen Virol 24:465–480. doi: 10.1099/0022-1317-24-3-465. [DOI] [PubMed] [Google Scholar]
- 10.Wilcox CL, Crnic LS, Pizer LI. 1992. Replication, latent infection, and reactivation in neuronal culture with a herpes simplex virus thymidine kinase-negative mutant. Virology 187:348–352. doi: 10.1016/0042-6822(92)90326-K. [DOI] [PubMed] [Google Scholar]
- 11.Chen SH, Pearson A, Coen DM, Chen SH. 2004. Failure of thymidine kinase-negative herpes simplex virus to reactivate from latency following efficient establishment. J Virol 78:520–523. doi: 10.1128/JVI.78.1.520-523.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coen DM, Kosz-Vnenchak M, Jacobson JG, Leib DA, Bogard CL, Schaffer PA, Tyler KL, Knipe DM. 1989. Thymidine kinase-negative herpes simplex virus mutants establish latency in mouse trigeminal ganglia but do not reactivate. Proc Natl Acad Sci U S A 86:4736–4740. doi: 10.1073/pnas.86.12.4736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Griffiths A, Chen SH, Horsburgh BC, Coen DM. 2003. Translational compensation of a frameshift mutation affecting herpes simplex virus thymidine kinase is sufficient to permit reactivation from latency. J Virol 77:4703–4709. doi: 10.1128/JVI.77.8.4703-4709.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Efstathiou S, Kemp S, Darby G, Minson AC. 1989. The role of herpes simplex virus type 1 thymidine kinase in pathogenesis. J Gen Virol 70:869–879. doi: 10.1099/0022-1317-70-4-869. [DOI] [PubMed] [Google Scholar]
- 15.Yamagami S, Mori K, Kawakita Y. 1972. Changes of thymidine kinase in the developing rat brain. J Neurochem 19:369–376. doi: 10.1111/j.1471-4159.1972.tb01346.x. [DOI] [PubMed] [Google Scholar]
- 16.Chen SH, Cook WJ, Grove KL, Coen DM. 1998. Human thymidine kinase can functionally replace herpes simplex virus type 1 thymidine kinase for viral replication in mouse sensory ganglia and reactivation from latency upon explant. J Virol 72:6710–6715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen SH, Lin YW, Griffiths A, Huang WY, Chen SH. 2006. Competition and complementation between thymidine kinase-negative and wild-type herpes simplex virus during co-infection of mouse trigeminal ganglia. J Gen Virol 87:3495–3502. doi: 10.1099/vir.0.82223-0. [DOI] [PubMed] [Google Scholar]
- 18.Ghiasi H, Cai S, Perng GC, Nesburn AB, Wechsler SL. 2000. Both CD4+ and CD8+ T cells are involved in protection against HSV-1 induced corneal scarring. Br J Ophthalmol 84:408–412. doi: 10.1136/bjo.84.4.408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Metcalf JF, Hamilton DS, Reichert RW. 1979. Herpetic keratitis in athymic (nude) mice. Infect Immun 26:1164–1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Minagawa H, Sakuma S, Mohri S, Mori R, Watanabe T. 1988. Herpes simplex virus type 1 infection in mice with severe combined immunodeficiency (SCID). Arch Virol 103:73–82. doi: 10.1007/BF01319810. [DOI] [PubMed] [Google Scholar]
- 21.Valyi-Nagy T, Gesser RM, Raengsakulrach B, Deshmane SL, Randazzo BP, Dillner AJ, Fraser NW. 1994. A thymidine kinase-negative HSV-1 strain establishes a persistent infection in SCID mice that features uncontrolled peripheral replication but only marginal nervous system involvement. Virology 199:484–490. doi: 10.1006/viro.1994.1150. [DOI] [PubMed] [Google Scholar]
- 22.Safrin S. 1992. Treatment of acyclovir-resistant herpes simplex virus infections in patients with AIDS. J Acquir Immune Defic Syndr 5(Suppl 1):S29–S32. [PubMed] [Google Scholar]
- 23.Pearson A, Coen DM. 2002. Identification, localization, and regulation of expression of the UL24 protein of herpes simplex virus type 1. J Virol 76:10821–10828. doi: 10.1128/JVI.76.21.10821-10828.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jacobson JG, Chen SH, Cook WJ, Kramer MF, Coen DM. 1998. Importance of the herpes simplex virus UL24 gene for productive ganglionic infection in mice. Virology 242:161–169. doi: 10.1006/viro.1997.9012. [DOI] [PubMed] [Google Scholar]
- 25.Mitterreiter JG, Titulaer MJ, van Nierop GP, van Kampen JJ, Aron GI, Osterhaus AD, Verjans GM, Ouwendijk WJ. 2016. Prevalence of intrathecal acyclovir resistant virus in herpes simplex encephalitis patients. PLoS One 11:e0155531. doi: 10.1371/journal.pone.0155531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martuza RL, Malick A, Markert JM, Ruffner KL, Coen DM. 1991. Experimental therapy of human glioma by means of a genetically engineered virus mutant. Science 252:854–856. doi: 10.1126/science.1851332. [DOI] [PubMed] [Google Scholar]
- 27.Markert JM, Malick A, Coen DM, Martuza RL. 1993. Reduction and elimination of encephalitis in an experimental glioma therapy model with attenuated herpes simplex mutants that retain susceptibility to acyclovir. Neurosurgery 32:597–603. doi: 10.1227/00006123-199304000-00016. [DOI] [PubMed] [Google Scholar]
- 28.Chen SH, Yao HW, Huang WY, Hsu KS, Lei HY, Shiau AL, Chen SH. 2006. Efficient reactivation of latent herpes simplex virus from mouse central nervous system tissues. J Virol 80:12387–12392. doi: 10.1128/JVI.01232-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pelosi E, Mulamba GB, Coen DM. 1998. Penciclovir and pathogenesis phenotypes of drug-resistant herpes simplex virus mutants. Antiviral Res 37:17–28. doi: 10.1016/S0166-3542(97)00054-5. [DOI] [PubMed] [Google Scholar]
- 30.Khanna KM, Bonneau RH, Kinchington PR, Hendricks RL. 2003. Herpes simplex virus-specific memory CD8+ T cells are selectively activated and retained in latently infected sensory ganglia. Immunity 18:593–603. doi: 10.1016/S1074-7613(03)00112-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Knickelbein JE, Khanna KM, Yee MB, Baty CJ, Kinchington PR, Hendricks RL. 2008. Noncytotoxic lytic granule-mediated CD8+ T cell inhibition of HSV-1 reactivation from neuronal latency. Science 322:268–271. doi: 10.1126/science.1164164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yao HW, Ling P, Chen SH, Tung YY, Chen SH. 2012. Factors affecting herpes simplex virus reactivation from the explanted mouse brain. Virology 433:116–123. doi: 10.1016/j.virol.2012.07.018. [DOI] [PubMed] [Google Scholar]
- 33.Decman V, Kinchington PR, Harvey SA, Hendricks RL. 2005. Gamma interferon can block herpes simplex virus type 1 reactivation from latency, even in the presence of late gene expression. J Virol 79:10339–10347. doi: 10.1128/JVI.79.16.10339-10347.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu T, Khanna KM, Chen X, Fink DJ, Hendricks RL. 2000. CD8(+) T cells can block herpes simplex virus type 1 (HSV-1) reactivation from latency in sensory neurons. J Exp Med 191:1459–1466. doi: 10.1084/jem.191.9.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Su YH, Oakes JE, Lausch RN. 1990. Ocular avirulence of a herpes simplex virus type 1 strain is associated with heightened sensitivity to alpha/beta interferon. J Virol 64:2187–2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pantelouris EM, Pringle CR. 1976. Interferon production in athymic nude mice. J Gen Virol 32:149–152. doi: 10.1099/0022-1317-32-1-149. [DOI] [PubMed] [Google Scholar]
- 37.Huang WY, Su YH, Yao HW, Ling P, Tung YY, Chen SH, Wang X, Chen SH. 2010. Beta interferon plus gamma interferon efficiently reduces acyclovir-resistant herpes simplex virus infection in mice in a T-cell-independent manner. J Gen Virol 91:591–598. doi: 10.1099/vir.0.016964-0. [DOI] [PubMed] [Google Scholar]
- 38.Davar G, Kramer MF, Garber D, Roca AL, Andersen JK, Bebrin W, Coen DM, Kosz-Vnenchak M, Knipe DM, Breakefield XO, Isascon O. 1994. Comparative efficacy of expression of genes delivered to mouse sensory neurons with herpes virus vectors. J Comp Neurol 339:3–11. doi: 10.1002/cne.903390103. [DOI] [PubMed] [Google Scholar]
- 39.Segre JA, Nemhauser JL, Taylor BA, Nadeau JH, Lander ES. 1995. Positional cloning of the nude locus: genetic, physical, and transcription maps of the region and mutations in the mouse and rat. Genomics 28:549–559. doi: 10.1006/geno.1995.1187. [DOI] [PubMed] [Google Scholar]
- 40.Shen FH, Wang SW, Yeh TM, Tung YY, Hsu SM, Chen SH. 2013. Absence of CXCL10 aggravates herpes stromal keratitis with reduced primary neutrophil influx in mice. J Virol 87:8502–8510. doi: 10.1128/JVI.01198-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shen FH, Shen TJ, Chang TM, Su IJ, Chen SH. 2014. Early dexamethasone treatment exacerbates enterovirus 71 infection in mice. Virology 464–465:218–227. doi: 10.1016/j.virol.2014.07.021. [DOI] [PubMed] [Google Scholar]
- 42.Yao HW, Lin PH, Shen FH, Perng GC, Tung YY, Hsu SM, Chen SH. 2014. Tranylcypromine reduces herpes simplex virus 1 infection in mice. Antimicrob Agents Chemother 58:2807–2815. doi: 10.1128/AAC.02617-13. [DOI] [PMC free article] [PubMed] [Google Scholar]