Abstract
MiR-27a rs895819 is a loop-stem structure single nucleotide polymorphism affecting mature miR-27a function. In this study, we performed a comprehensive analysis about the association of rs895819 with gastric cancer risk and prognosis, atrophic gastritis risk, as well as the interactions with environmental factors. A total of 939 gastric cancer patients, 1,067 atrophic gastritis patients and 1,166 healthy controls were screened by direct sequencing and MALDI-TOF-MS. The association of rs895819 with clinical pathological parameters and prognostic survival in 357 gastric cancer patients was also been analyzed. The rs895819 variant genotype increased the risk for atrophic gastritis (1.58-fold) and gastric cancer (1.24-fold). While in stratified analysis, the risk effect was demonstrated more significantly in the female, age >60y, Helicobacter pylori (H. pylori) negative and non-drinker subgroups. Rs895819 and H. pylori showed an interaction effect for atrophic gastritis risk. In the survival analysis, the rs895819 AG heterozygosis was associated with better survival than the AA wild-type in the TNM stage I–II subgroup. In vitro study by overexpressing miR-27a, cells carrying polymorphic-type G allele expressed lower miR-27a than wild-type A allele. In conclusion, miR-27a rs895819 is implicated as a biomarker for gastric cancer and atrophic gastritis risk, and interacts with H. pylori in gastric carcinogenesis.
Single nucleotide polymorphisms (SNPs) are common variations of the hereditary factors thought to be associated with several forms of cancers1,2. MicroRNAs (miRNAs) SNPs are located in the pre-/pri- or mature miRNAs, which could change the binding site with transcriptional factors or Drosha/Dicer enzyme and thus affect the quality and quantity of mature miRNAs3,4. MiR-27a contains two SNPs, rs895819 and rs11671784, with rs895819 being located in the 6 bp downstream of rs11671784 on chromosome 19p13.132. The miR-27a rs895819 polymorphism is an unusual miRNA-SNP due to its location in the coding region of the pre-mir-27a hairpin in the stem-loop, which could be cut by Dicer in the process of pre-miRNA maturation5. Recent studies speculated that the A → G change of rs895819 could shorten the stem-loop structure and affect the processing of miR-27a6,7, suggesting that rs895819 was a functional SNP. The results of previous investigations concerning the association between this polymorphism and cancer risk are controversial. For example, several studies reported the variant allele could decrease the cancer risk7,8,9, while another study found the AG and GG genotypes increased the risk of gastric cancer10, and the others suggested that the association between the variant G allele and cancer risk was statistically insignificant11,12,13. However, for the most important of all, it was reported that direct sequencing or MALDI-TOF Mass-ARRAY using primer extension from one direction (not overlapping with any SNP) is essential for investigations of rs895819 and rs1167178414.
Gastric carcinogenesis is influenced by multiple hereditary and environmental factors, and epidemiological studies have suggested that individual hereditary susceptibility affects the incidence of gastric cancer15. Recently, accumulating evidences have demonstrated environmental factors such as smoking, drinking and Helicobacter pylori (H. pylori) infection, are all critical risk factors for gastric carcinogenesis. In addition, environmental factors and gene polymorphisms may also be involved in gastric carcinogenesis. Helicobacter pylori (H. pylori), as one of the important factors for gastric adenocarcinoma and mucosa-associated lymphoid tissue lymphoma, are able to result in chronic infection16. A synergistic effect of H. pylori infection with gene polymorphisms could contribute to the development of gastric cancer17,18,19. However, it is still unclear whether an interaction effect exists between miR-27a rs895819 SNP and H. pylori for gastric carcinogenesis, of which the exploration will help to the comprehension of the carcinogenic biology.
In this study, sequencing and MALDI-TOF-MS were used as the accepted detection technology to perform a comprehensive analysis for this special SNP, including the association with risk and prognosis, cancerous and precancerous status, and interaction effects with the environmental factor H. pylori.
Results
Patient characteristics
Two-stage study was conducted: in stage 1, the samples came from 724 retrospectively recruited individuals, including 215 gastric cancer patients, 205 atrophic gastritis cases and 304 controls, which attended the First Affiliated Hospital of China Medical University between 2005 and 2010. In stage 2, a total of 2,448 cases were included, consisting of 724 gastric cancer cases, 862 atrophic gastritis cases and 862 matched healthy controls from the Zhuanghe Gastric Diseases Screening Program or from patients who attended the First Affiliated Hospital of China Medical University between 2002 and 2013.
The baseline characteristics of the subjects in the control, atrophic gastritis and gastric cancer groups were listed in Supplementary Table S1. No significant difference was found in terms of age and sex between the case and control groups. In stage 2, the subjects were selected from two different sources, then we analyzed the baseline of the subjects and the distribution frequency of rs895819 and no significant difference was discovered between the two groups (Supplementary Table S2). The characteristics of the subjects in the intestinal-type and diffuse-type gastric cancer subgroups were listed in Supplementary Table S3.
Frequencies of miR-27a rs895819 and rs11671784 determined by direct sequencing
Direct sequencing of miR-27a rs895819 in the 724 samples revealed 392 cases of the AA genotype, 291 cases of the AG genotype and 41 cases of the GG genotype. Among the 724 samples in the control group, only one individual carried the rs11671784 AG genotype, while the other 723 samples all carried GG genotype.
Association of miR-27a rs895819 with gastric disease risk
The miR-27a rs895819 SNP was found to meet with Hardy–Weinberg equilibrium (P > 0.05, Table 1). Rs895819 was associated with an increased risk for atrophic gastritis in stage 2 (GG vs. AA: P = 0.045, OR = 1.58; GG vs. AG + AA: P = 0.045, OR = 1.56). We also found rs895819 was associated with an increased risk of gastric cancer in the analysis of merge data (AG vs. AA: P = 0.027, OR = 1.24; AG + GG vs. AA: P = 0.043, OR = 1.21; Table 1). In addition, the method suggested by Thakkinstian et al.20 was employed to select the optimal genetic models for rs895819 SNP including dominant model, recessive model, codominant model and complete overdominant model. In the merged data, for the atrophic gastritis risk, OR1, OR2 and OR3 were 1.30 (P = 0.174), 1.02 (P = 0.797) and 1.25 (P = 0.248), respectively, which was in accord with the principle of OR1 > OR2 > 1 and OR1 > OR3 > 1, indicating a codominant model effect (Table 1). For the gastric cancer risk, OR1, OR2 and OR3 were 1.03 (P = 0.906), 1.24 (P = 0.027) and 0.81 (P = 0.318), respectively, which was in accord with the principle of OR2 = 1/OR3 ≠ 1 and OR1 = 1, indicating a complete overdominant model effect (Table 1). Thus, for the gastric cancer risk, in the complete overdominant model, AG heterozygote was associated with an increased risk of gastric cancer compared with GG + AA genotypes (P = 0.025, OR = 1.24, Table 1).
Table 1. The association of miR-27a rs895819 polymorphisms and gastric cancer/atrophic gastritis riska.
| Genotype | NCBI Ref (%) | CON (%) | AG (%) | AG vs CON |
CON (%) | GC (%) | GC vs CON |
||
|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P | OR (95% CI) | P | ||||||
| Stage 1 | n = 304 | n = 205 | n = 304 | n = 215 | |||||
| AA | 21 (48.8) | 171 (56.3) | 110 (53.7) | 1 (Ref) | 171 (56.3) | 111 (51.6) | 1 (Ref) | ||
| AG | 17 (39.5) | 111 (36.5) | 83 (40.5) | 1.15 (0.79–1.68) | 0.463 | 111 (36.5) | 97 (45.1) | 1.32 (0.91–1.91) | 0.141 |
| GG | 5 (11.6) | 22 (7.2) | 12 (5.9) | 0.84 (0.39–1.79) | 0.646 | 22 (7.2) | 7 (3.3) | 0.47 (0.19–1.14) | 0.094 |
| GG + AG VS. AA | 1.10 (0.77–1.58) | 0.559 | 1.17 (0.82–1.67) | 0.386 | |||||
| GG VS. AG + AA | 0.80 (0.38–1.68) | 0.559 | 0.41 (0.17–0.99) | 0.047 | |||||
| PHWEb | 0.590 | 0.498 | |||||||
| Stage 2 | n = 862 | n = 862 | n = 729 | n = 724 | |||||
| AA | 482 (55.9) | 467 (54.2) | 1 (Ref) | 411 (56.4) | 369 (51.0) | 1 (Ref) | |||
| AG | 340 (39.4) | 336 (39.0) | 0.98 (0.80–1.21) | 0.874 | 282 (38.7) | 311 (43.0) | 1.21 (0.97–1.51) | 0.089 | |
| GG | 40 (4.6) | 59 (6.8) | 1.58 (1.01–2.46) | 0.045 | 36 (4.9) | 44 (6.1) | 1.35 (0.84–2.18) | 0.220 | |
| GG + AG VS. AA | 1.04 (0.85–1.27) | 0.684 | 1.23 (0.99–1.52) | 0.061 | |||||
| GG VS. AG + AA | 1.56 (1.01–2.41) | 0.045 | 1.22 (0.76–1.94) | 0.415 | |||||
| Meta-analysis | n = 1166 | n = 1067 | n = 1033 | n = 939 | |||||
| AA | 653 (56.0) | 577 (54.1) | 1 (Ref) | 582 (56.3) | 480 (51.1) | 1 (Ref) | |||
| AG | 451 (38.7) | 419 (39.3) | 1.02 (0.85–1.23) | 0.797 | 393 (38.0) | 408 (43.5) | 1.24 (1.03–1.50) | 0.027 | |
| GG | 62 (5.3) | 71 (6.7) | 1.30 (0.89–1.88) | 0.174 | 58 (5.6) | 51 (5.4) | 1.03 (0.68–1.54) | 0.906 | |
| GG + AG VS. AA | 1.28 (0.89–1.84) | 0.190 | 1.21 (1.01–1.45) | 0.043 | |||||
| GG VS. AG + AA | 1.06 (0.89–1.26) | 0.537 | 0.92 (0.62–1.37) | 0.681 | |||||
| GG vs AA (OR1) | 1.30 (0.89–1.88) | 0.174 | 1.03 (0.68–1.54) | 0.906 | |||||
| AG vs. AA (OR2) | 1.02 (0.85–1.23) | 0.797 | 1.24 (1.03–1.50) | 0.027 | |||||
| GG vs AG (OR3) | 1.25 (0.86–1.83) | 0.248 | 0.81 (0.54–1.22) | 0.318 | |||||
| Codominant modelc | 1.06 (0.89–1.26) | 0.537 | |||||||
| Complete overdominant modeld | 1.24 (1.03–1.49) | 0.025 | |||||||
Note: aUsing Logistic Regession adjusted by sex, age and H. pylori infection; bMeans Hardy–Weinberg Equilibrium in population; cA codominant model was indicated for rs895819 in the comparison of atrophic gastritis vs. control group, and GG was compared with AG and with AA. dA complete overdominant model was implied for rs895819 in the comparison of gastric cancer vs. control group. The original grouping was collapsed and the new group of AG heterozygote was compared with GG and with AA. CON: controls; AG: atrophic gastritis; GC: gastric cancer; NCBI Ref: the reference frequencies of these polymorphisms in Beijing Han, China in NCBI database.
When the gastric cancer group was divided into intestinal-type and diffuse-type according to Lauren classification, no association was found between diffuse-type subgroup and controls, which was also fit for intestinal-type subgroup though the dominant model closely reaching statistical significance (GG vs. AA, P = 0.050, OR = 1.32; Supplementary Table 4).
In the stratified analysis, the heterogeneity test was performed for the host’s characteristics, and the results were shown in Supplementary Table S5. Both the age factor for atrophic gastritis vs. control group and the H. pylori infection factor for gastric cancer vs. control group almost reached statistical significance (Pheterogeneity = 0.095 and 0.078, respectively). Thus, the stratified analysis for all the host’s characteristics was conducted. In the female subgroup, rs895819 was found to be associated with an increased risk of atrophic gastritis and gastric cancer (atrophic gastritis risk: GG vs. AA, P = 0.030, OR = 1.86; gastric cancer risk: AG vs. AA, P = 0.032, OR = 1.44). In the age >60y subgroup, individuals carrying with the AG heterozygosis were associated with an increased gastric cancer risk (AG vs. AA, P = 0.001, OR = 1.74). In the H. pylori-negative subgroup, the GG genotype was associated with an increased risk when compared with the AA wild-type (P = 0.047, OR = 1.62). In the non-drinking subgroup, individuals with the GG variant genotype were associated with increased atrophic gastritis risk (GG vs. AA, P = 0.037, OR = 2.08, Table 2).
Table 2. Association of miR-27a rs895819 polymorphism with the risk of atrophy gastritis and gastric cancer stratified by host characteristics.
| Variables | Genotype | AG vs CON | OR (95% CI) | P | GC vs CON | OR (95% CI) | P |
|---|---|---|---|---|---|---|---|
| Gendera | n = 1067 vs 1166 | n = 939 vs 1033 | |||||
| Male | AA | 343/377 | 1 (Ref) | 334/377 | 1 (Ref) | ||
| AG | 238/255 | 0.97 (0.76–1.23) | 0.803 | 268/255 | 1.15 (0.92–1.45) | 0.227 | |
| GG | 33/38 | 0.93 (0.56–1.55) | 0.787 | 33/38 | 0.96 (0.58–1.58) | 0.875 | |
| AG + GG VS. AA | 1.00 (0.77–1.22) | 0.772 | 1.13 (0.90–1.41) | 0.282 | |||
| GG VS. AG + AA | 0.95 (0.57–1.56) | 0.829 | 0.89 (0.55–1.46) | 0.648 | |||
| Female | AA | 234/276 | 1 (Ref) | 146/205 | 1 (Ref) | ||
| AG | 181/196 | 1.12 (0.84–1.47) | 0.443 | 140/138 | 1.44 (1.03–2.00) | 0.032 | |
| GG | 38/24 | 1.86 (1.06–3.27) | 0.030 | 18/20 | 1.17 (0.58–2.36) | 0.661 | |
| AG + GG VS. AA | 1.20 (0.92–1.56) | 0.183 | 1.39 (1.01–1.91) | 0.045 | |||
| GG VS. AG + AA | 1.77 (1.03–3.06) | 0.040 | 0.98 (0.50–1.95) | 0.959 | |||
| Agea | n = 1067 vs 1166 | n = 939 vs 1033 | |||||
| ≤60 | AA | 406/477 | 1 (Ref) | 334/406 | 1 (Ref) | ||
| AG | 283/324 | 1.02 (0.82–1.26) | 0.886 | 232/266 | 1.06 (0.84–1.33) | 0.655 | |
| GG | 46/39 | 1.39 (0.87–2.21) | 0.168 | 32/35 | 1.02 (0.61–1.70) | 0.948 | |
| AG + GG VS. AA | 1.06 (0.86–1.30) | 0.609 | 1.05 (0.84–1.32) | 0.667 | |||
| GG VS. AG + AA | 1.37 (0.87–2.16) | 0.173 | 1.01 (0.61–1.66) | 0.983 | |||
| >60 | AA | 171/176 | 1 (Ref) | 146/176 | 1 (Ref) | ||
| AG | 136/127 | 1.07 (0.76–1.49) | 0.712 | 176/127 | 1.74 (1.25–2.42) | 0.001 | |
| GG | 25/23 | 1.14 (0.61–2.14) | 0.682 | 19/23 | 1.10 (0.56–2.14) | 0.785 | |
| AG + GG VS. AA | 1.08 (0.78–1.48) | 0.659 | 1.63 (1.18–2.24) | 0.003 | |||
| GG VS. AG + AA | 1.11 (0.60–2.07) | 0.731 | 0.85 (0.44–1.62) | 0.614 | |||
| H. pylorib | n = 1067 vs 1166 | n = 939 vs 1033 | |||||
| negative | AA | 250/469 | 1 (Ref) | 240/165 | 1 (Ref) | ||
| AG | 179/315 | 1.06 (0.84–1.35) | 0.629 | 204/117 | 1.27 (1.00–1.62) | 0.055 | |
| GG | 36/41 | 1.62 (1.01–2.60) | 0.047 | 27/19 | 1.08 (0.63–1.85) | 0.782 | |
| AG + GG VS. AA | 1.12 (0.89–1.41) | 0.319 | 1.24 (0.98–1.57) | 0.069 | |||
| GG VS. AG + AA | 1.58 (0.99–2.51) | 0.054 | 0.94 (0.55–1.58) | 0.803 | |||
| positive | AA | 327/184 | 1 (Ref) | 240/417 | 1 (Ref) | ||
| AG | 240/136 | 0.98 (0.74–1.30) | 0.899 | 204/276 | 1.19 (0.88–1.61) | 0.262 | |
| GG | 35/21 | 0.92 (0.52–1.64) | 0.785 | 24/39 | 0.98 (0.52–1.82) | 0.936 | |
| AG + GG VS. AA | 0.97 (0.74–1.27) | 0.820 | 1.15 (0.86–1.55) | 0.335 | |||
| GG VS. AG + AA | 0.93 (0.53–1.64) | 0.810 | 0.90 (0.49–1.64) | 0.720 | |||
| Smokingb | n = 548 vs 586 | n = 333 vs 500 | |||||
| Never smoker | AA | 204/224 | 1 (Ref) | 102/180 | 1 (Ref) | ||
| AG | 143/142 | 1.08 (0.78–1.50) | 0.642 | 69/105 | 1.17 (0.78–1.77) | 0.444 | |
| GG | 26/18 | 1.95 (0.99–3.85) | 0.055 | 14/16 | 1.64 (0.74–3.67) | 0.225 | |
| AG + GG VS. AA | 1.17 (0.85–1.60) | 0.330 | 1.23 (0.83–1.82) | 0.295 | |||
| GG VS. AG + AA | 1.87 (0.96–3.64) | 0.066 | 1.53 (0.70–3.34) | 0.288 | |||
| Ever smoker | AA | 94/109 | 1 (Ref) | 74/107 | 1 (Ref) | ||
| AG | 68/82 | 0.95 (0.60–1.50) | 0.828 | 68/81 | 1.13 (0.71–1.82) | 0.606 | |
| GG | 11/11 | 1.21 (0.48–3.09) | 0.685 | 6/11 | 0.60 (0.20–1.84) | 0.376 | |
| AG + GG VS. AA | 0.98 (0.64–1.52) | 0.936 | 1.06 (0.67–1.67) | 0.819 | |||
| GG VS. AG + AA | 1.23 (0.50–3.07) | 0.653 | 0.54 (0.18–1.64) | 0.277 | |||
| Alcohol drinkingb | n = 547 vs 585 | n = 296 vs 499 | |||||
| Nondrinker | AA | 239/256 | 1 (Ref) | 105/211 | 1 (Ref) | ||
| AG | 157/165 | 1.07 (0.78–1.46) | 0.687 | 71/127 | 1.10 (0.74–1.64) | 0.641 | |
| GG | 27/17 | 2.08 (1.04–4.16) | 0.037 | 13/15 | 1.43 (0.62–3.33) | 0.406 | |
| AG + GG VS. AA | 1.15 (0.85–1.55) | 0.358 | 1.13 (0.77–1.66) | 0.521 | |||
| GG VS. AG + AA | 1.99 (1.01–3.90) | 0.047 | 1.35 (0.59–3.08) | 0.484 | |||
| Drinker | AA | 60/76 | 1 (Ref) | 52/75 | 1 (Ref) | ||
| AG | 53/60 | 0.99 (0.58–1.67) | 0.959 | 50/60 | 1.20 (0.70–2.06) | 0.503 | |
| GG | 11/11 | 1.22 (0.49–3.05) | 0.677 | 5/11 | 0.73 (0.23–2.30) | 0.587 | |
| AG + GG VS. AA | 1.03 (0.63–1.70) | 0.902 | 1.14 (0.68–1.93) | 0.620 | |||
| GG VS. AG + AA | 1.33 (0.54–3.27) | 0.542 | 0.71 (0.23–2.21) | 0.555 |
Note: aUsing Logistic Regession adusted by the other two factors of gender, age and H. pylori infection status. bUsing Logistic Regession adusted by gender, age and H. pylori infection status. CON:controls; AG: atrophic gastritis; GC:gastric cancer.
Interaction of rs895819 and H. pylori in the risk of gastric cancer/atrophic gastritis
More significant association was found in the H. pylori subgroup with the risk of gastric cancer and atrophic gastritis compared with that of the total samples. Therefore, we analyzed the interaction effect between rs895819 and H. pylori infection status. The result showed a negative interaction for atrophic gastritis risk (Pinteraction = 0.013, OR = 0.54; Table 3).
Table 3. The interaction of miR-27a rs895819 polymorphism and H. pylori infection statue in the risk of gastric cancer/atrophic gastritisa.
| Genotype | AG vs CON | GC vs CON | |||
|---|---|---|---|---|---|
| H. pylori | H. pylori | ||||
| (−) | (+) | (−) | (+) | ||
| AG + AA | Case/Control | 385/709 | 528/284 | 395/618 | 396/246 |
| OR (95%CI) | 1 (Ref) | 3.44 (2.84–4.16) | 1 (Ref) | 2.53 (2.07–3.10) | |
| GG | Case/Control | 80/116 | 74/57 | 73/114 | 75/55 |
| OR (95%CI) | 1.27 (0.93–1.73) | 2.38 (1.65–3.44) | 1.00 (0.73–1.38) | 2.13 (1.47–3.08) | |
| Pinteraction = 0.013 | Pinteraction = 0.505 | ||||
| OR (95%CI) = 0.54 (0.33–0.88) | OR (95%CI) = 0.84 (0.51–1.39) | ||||
Note: aP for interaction was used Logistic Regression adjusted by gender and age. CON: controls; AG: atrophic gastritis; GC: gastric cancer.
We further analyzed the cumulative effect of these two risk factors for atrophic gastritis and also found a significantly negative effect (Ptrend < 0.001, 95%CI = 0.24–0.32; Table 4).
Table 4. Cumulative effect of the interacting risk factors of miR-27a rs895819-H. pylori infection on the atrophic gastritis risk.
| No. of interacting risk factors | Total population |
||
|---|---|---|---|
| Cases/controls | P a | OR (95% CI) | |
| 0 | 329/592 | 1 (ref) | |
| 1 | 502/259 | <0.001 | 3.53 (2.89–4.33) |
| 2 | 31/11 | <0.001 | 5.06 (2.51–10.21) |
| P trend <0.001, 95%CI = 0.24–0.32 | |||
Note: aAdjusted by sex and age.
Rs895819 genotype and clinical pathological characteristics
To investigate the association of genotype with phenotype, we analyzed the association between rs895819 and the clinical pathological characteristics of gastric cancer patients. No significant association was found between age and sex, macroscopic type, Lauren grade, TNM stage, depth of invasion, lymphatic metastasis and rs895819 genotypes (Supplementary Table 6).
Association of miR-27a rs895819 with cancer survival prognosis
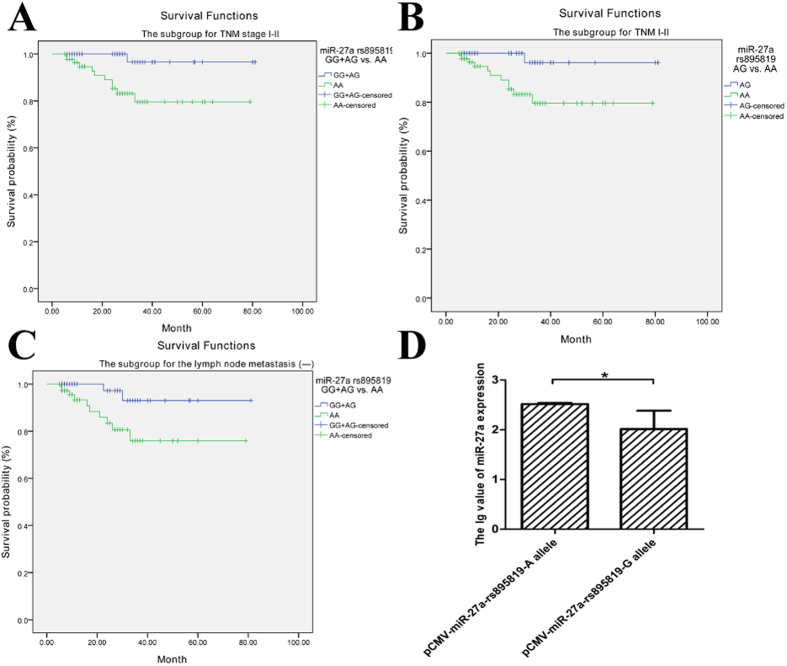
To analyze the relationship between rs895819 and survival time, univariate and multivariate Cox proportional hazard analysis were performed adjusted by adding all SNP variables to the clinicopathological parameters with P < 0.05 (TNM stage, lymphatic metastasis and depth of invasion; Supplementary Table 7). No significant difference was discovered between rs895819 and survival, while the result was totally contrary in the subgroups. In the TNM stage I–II subgroup, the overall survival in the subjects carrying with rs895819 AG heterozygosis was better than the AA wild-type (univariate: P = 0.036, HR = 0.11; multivariate: P = 0.041, HR = 0.12, Fig. 1-A,B). In the lymphatic metastasis subgroup, individuals carrying with rs895819 AG + GG genotype had better survival compared with AA wild-type (univariate: P = 0.040, HR = 0.20, Table 5, Fig. 1-C).
Figure 1. The effect of miR-27a rs895819 A/G SNP on the mature miR-27a expression in vivo and vitro.
(A) Kaplan–Meier survival curve analysis with the different genotypes of miR-27a rs895819 (GG + AG vs. AA) for the subgroup for TNM I-II stage. (B) Kaplan–Meier survival curve analysis with the different genotypes of miR-27a rs895819 (AG vs. AA) for the subgroup for TNM I-II stage. (C) Kaplan–Meier survival curve analysis with the different genotypes of miR-27a rs895819 (GG + AG vs. AA) for the subgroup for lymph node metastasis (−). (D) The cell mature miR-27a expression transinfected by different miR-27a rs895819 plasmid. *P = 0.05.
Table 5. Univariate and multivariate cox proportional hazard analysis for miR-27a rs895819 polymorphism.
| Genotype | All GC | Death | MSTa (M) | Univariate |
Multivariatec |
||
|---|---|---|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | ||||
| n = 357 | n = 89 | ||||||
| AA | 184 | 48 | 58.4b | 1 (Ref) | 1 (Ref) | ||
| AG | 148 | 36 | 58.8b | 0.89 (0.58–1.37) | 0.597 | 0.84 (0.54–1.29) | 0.415 |
| GG | 25 | 5 | 45.4b | 0.75 (0.30–1.88) | 0.535 | 0.62 (0.25–1.57) | 0.316 |
| AG + GG VS. AA | 0.87 (0.57–1.32) | 0.510 | 0.80 (0.53–1.21) | 0.294 | |||
| GG VS. AG + AA | 0.80 (0.32–1.97) | 0.625 | 0.68 (0.27–1.67) | 0.398 | |||
| Subgroup analysis for TNM stage I–II | |||||||
| I–II | n = 171 | n = 12 | |||||
| AA | 91 | 11 | 67.0b | 1 (Ref) | 1 (Ref) | ||
| AG | 68 | 1 | 79.0b | 0.11 (0.01–0.87) | 0.036 | 0.12 (0.02–0.91) | 0.041 |
| GG | 12 | 0 | NA | NA | NA | NA | NA |
| AG + GG VS. AA | 0.10 (0.01–0.77) | 0.027 | 0.10 (0.01–0.80) | 0.030 | |||
| GG VS. AG + AA | NA | NA | NA | NA | |||
| Subgroup analysis for Lymphatic metastasis (−) | |||||||
| Negative | n = 141 | n = 12 | |||||
| AA | 74 | 10 | 64.7b | 1 (Ref) | 1 (Ref) | ||
| AG | 58 | 2 | 76.9b | 0.22 (0.05–1.02) | 0.054 | 0.25 (0.06–1.15) | 0.076 |
| GG | 9 | 0 | NA | NA | NA | NA | NA |
| AG + GG VS. AA | 0.20 (0.05–0.93) | 0.040 | 0.2 (0.05–1.03) | 0.054 | |||
| GG VS. AG + AA | NA | NA | NA | NA | |||
Note: HR, hazard ratio; CI, confidence interval; aMST, median survival time (months). bwhen MST could not be calculated, mean survival time was provided. cMultivariate survival analysis was carried out by adding all the SNP variables to the clinicopathological parameters with P < 0.05 including TNM stage, lymphatic metastasis and depth of invasion. NA, not available.
The effect of rs895819 on miR-27a expression
We first analyzed the differential expression of miR-27a between cancer patients and control groups, as well as between tumor tissues and paired non-cancer tissues. miR-27a expression in cancer patients was significantly higher than that of the control groups in serum expression level (P < 0.001, Table 6). And the same tendency was revealed in tissue expression level rather than reaching the statistical significance (P = 0.063).
Table 6. The effect of miR-27a rs895819 polymorphism to its mature miR-27a expression.
| Serum expression |
Tissue expression in situ |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cancer |
Controls |
Cancer Tissue |
Noncancer Tissues |
|||||||
| n | Mean ± SD | n | Mean ± SD | n | Mean ± SD | n | Mean ± SD | |||
| Total | 87 | 2.12 ± 1.14 | 83 |
1.46 ± 0.73 | 31 | 8.71 ± 0.37 | 32 | 8.55 ± 0.31 | ||
| Pa | <0.001 | 0.063 | ||||||||
| Genotype | ||||||||||
| AA | 38 | 2.25 ± 1.13 | 44 | 1.57 ± 0.78 | 14 | 8.84 ± 0.40 | 16 | 8.53 ± 0.32 | ||
| AG | 47 | 2.01 ± 1.14 | 34 | 1.33 ± 0.71 | 16 | 8.64 ± 0.30 | 16 | 8.56 ± 0.30 | ||
| GG | 2 | 2.02 ± 1.90 | 5 | 1.35 ± 0.18 | 1 | 8.18 | 0 | NA | ||
| Pb | 0.630 | 0.340 | 0.103 | 0.744 | ||||||
Note: aMeans the dependent sample-t test was used to calculate the difference betweenthe expressions of tumor and controls; bMeans the variance analysis was used to calculate the difference between the expressions of three genotypes.
To preliminarily explore the effect of rs895819 on mature miR-27a expression, we analyzed the mature miRNA expression based on different SNP genotypes both in tissue and in serum or in cancerous and non-cancerous groups. No significant effect of different rs895819 genotypes on mature miR-27a expression in tissues or in serum was observed (Table 6).
In the vitro level, AGS cell line was selected by the screening of miR-27a expression. And the AGS cells were transinfected into two plasmids, pCMV-miR-27a-rs895819-A and pCMV-miR-27a-rs895819-G. After 24 hours, the miR-27a expression level of variant G allele was significantly lower compared with the ancient A allele (P = 0.05, Fig. 1-D).
Discussion
We comprehensively and systematically conducted the risk and survival study for the special miR-27a rs895819 SNP, and further performed the expression study in tissue and in serum or in vivo as well as in vitro. We found that the rs895819 SNP was associated with an increased risk of atrophic gastritis and gastric cancer. An interaction between rs895819 and H. pylori for atrophic gastritis risk was been found. Individuals carrying with the rs895819 AG heterozygosis had better survival in the TNM stage I–II subgroup. And cells carrying with polymorphic-type G allele expressed lower miR-27a level than wild-type A allele.
In this study, we found that rs895819 AG genotype was associated with an increased risk of gastric cancer. It has been reported that rs895819 increased the gastric cancer risk in Chinese population, and could affect transcription proceeding from pri-miRNA to pre-miRNA resulting in the changes in the expression of mature miRNA2. It suggested that rs895819 was a functional SNP, and our result was consistent with the previously published study2. Only one study has shown negative association of this SNP with atrophic gastritis risk21. It has been suggested by Lauren that gastric cancer, especially intestinal-type gastric cancer, develops from atrophic gastritis, which is known to be an important precancerous disease22. Our analysis of a second set of samples by MALDI-TOF-MS revealed that the rs895819 SNP was associated with a 1.56-fold increase in the risk of atrophic gastritis, suggesting that this variant is involved in the progression of gastric carcinogenesis, and furthermore, implicating this SNP could be a biomarker of the risk of precancerous atrophic gastritis.
Environmental factors consist of the host’s natural and social environment. The natural environment contains geographical and occupational factors as well as bacterial or viral infection. H. pylori infection is an important environmental factor that influences the risk of gastric cancer. In this study, when stratified by the H. pylori infection status, the OR value of the H. pylori-negative subgroup was found to be higher than that of the overall population (1.62 vs. 1.58, respectively). This suggests that the role of rs895819 SNP will be more apparent when the H. pylori factor is removed from the analysis. Further analysis based on this finding showed that rs895819 and H. pylori infection status could exert a negative interaction effect. Surprisingly, linear regression analysis showed that accumulation of the risk factors including the rs895819 risk genotype and H. pylori infection was associated with a protective effect against carcinogenesis. This accumulation effect revealed by linear regression analysis was consistent with the interaction analysis, which suggested that the association of this rs895819 SNP with H. pylori infection was based on antagonism. Both rs895819 SNP and H. pylori infection are risk factors for carcinogenesis, as H. pylori is a tumour accelerator and the rs895819 SNP is a polymorphism associated with increased gastric cancer risk. However, it can be speculated that rs895819 SNP weakens the carcinogenic role of H. pylori in individuals carrying with both two risk factors. Similar situations were observed in the associations of H. pylori infection and microsatellite instability with gastric cancer prognosis. A recent meta-analysis of 2,454 gastric cancer patients suggested a protective role for infection in prognosis, and also that H. pylori-induced inflammation might modulate antitumor immunity23. Microsatellite instability is a hallmark of the DNA-mismatch repair deficiency, which is one of the pathways of gastric carcinogenesis although microsatellite alterations are related to better post-operative survival24,25. Therefore, it is plausible that the rs895819 SNP has a negative interaction with H. pylori infection in gastric carcinogenesis. In our research, we also found that miR-27a rs895819 GG genotype was associated with an increased risk of atrophic gastritis in stage 2. Some scholars have also reported that miRNA expression changes have been already detectable in early stages of gastric carcinogenesis including H. pylori induced atrophic gastritis26. But whether the variations in miRNAs, especially the SNP in miR-27a, were associated with H. pylori-related atrophic gastritis was still not clear. Several investigators were concerned with the association between rs895819 and atrophic gastritis risk, but no significant association21,27,28 was shown. Thus, large-scales and multi-central studies are needed in the near future.
We also found that in the TNM stage I–II or non-lymphatic metastasis subgroups, the patients carrying with miR-27a rs895819 AG + GG genotypes had better prognosis. It has been reported that miR-27a rs895819 G is associated with better survival29, which is consistent with the results of our study. In the risk study, this rs895819 variant genotype showed a risk function, although in the prognosis study, the same variant genotype showed a protective function. These conflicting results are consistent with the observation that the rs895819 SNP weakens the carcinogenic role of H. pylori. Several studies have shown that miRNA polymorphism increases cancer risk, but exerts protective function in cancer survival. For example, in one study, miR-146a polymorphism was found to have risk function in the non-smoking subgroup of gastric cancer patients, but played a protective role in the intestinal-type subgroup of gastric cancer patients30. In another study, miR-196a2 polymorphism exerted a protective function in head and neck cancer, but was associated with a negative effect on survival in gastric cancer patients31. These contradictory findings may result from the different types of cancer, or the association with the effect of miRNA polymorphism on miRNA expression in carcinogenesis.
MiRNA polymorphism could affects miRNA transcription and mature miRNA production32. To further explore the possible mechanism of the association between rs895819 and gastric cancer risk as well as prognosis we analyzed mature miR-27a expression in accordance with the polymorphic stratification. We did not found significant statistical effects of the SNP on the mature miR-27a expression both in the serum and in tissue or in cancer and non-cancerous groups. But in the vivo experiment, significantly lower mature miR-27a expression level were detected in AGS cells containing pCMV-miR-27a-rs895819-G allele than that of wild-A allele suggesting the rs895819 may effect mature miR-27a expression to a certain extent, which could partly interpret the mechanism involved in the protective function in cancer survival but controversial the increased gastric cancer susceptibility. This phenomenon is very puzzling and interesting. Other scholars also reported controversial results for the miR-27a function, for example, Zhou L et al. found that miR-27a could promote cancer cell proliferation33; while Wang X et al. reported that miR-27a acted as a tumor suppressor by suppressing oncogene MAP2K4 in vivo experiment34. Accordingly, some thought it was an “oncogene”33,35 but some believed it could induce the apoptosis36. Thus, it is still unclear whether the mature miR-27a acts as an “oncogene” or “tumor suppressor” during carcinogenesis. Maybe miR-27a play a role as double-edged sword, in that way, it is also still unclear that when miR-27a acts as an “oncogene” or “tumor suppressor”. The real function of miR-27a still needs to be investigated in the future.
In 2012, Yang reported that rs895819 polymorphism is associated with an unusual structure making primer extension possible from only one direction (not overlapping with any SNP). The use of direct sequencing or MALDI-TOF Mass-ARRAY is essential for miR-27a rs895819 and rs11671784 genotyping because qPCR and PCR-RFLP are associated with unacceptable risk of false-positive genotyping14. Later in 2013, other researchers noted that only the direct sequencing or MALDI-TOF Mass-ARRAY was acceptable for genotyping this rs895819 polymorphism37. Subsequently, however, the special structure of rs895819 was rarely considered and Taqman or PCR-RFLP technologies were still used, rendering the association of this polymorphism and disease risk being inconsistent. To make the unusual structure of this rs895819 polymorphism being compatible, we used direct sequencing and Mass-ARRAY technology to increase the credibility of our study. Furthermore, we found only one case of the rs11671784 AG genotype among 724 samples was analyzed by direct sequencing. This distribution frequency is in accordance with the NCBI databases (http://www.ncbi.nlm.nih.gov/projects/SNP/snp_ref.cgi?rs=11671784). Several studies2,38 investigated this rs11671784 polymorphism using PCR-RFLP or Taqman technology and the distribution frequency of the variant allele was higher than 50% both in the control and cancerous groups, which increases the risk of false genotyping.
There are some limitations of our study. First, the small sample size limited the subgroup analysis of rare genotypes. Second, complete information for other environmental factors is required to analyze polymorphism interaction effects.
In summary, we found that the rs895819 variant genotype was associated with an increased risk of atrophic gastritis and gastric cancer. In the stratified analysis, these effects were more significant in the female, age >60, H. pylori-negative and non-drinkers subgroups. The negative interaction between rs895819 and H. pylori in atrophic gastritis risk should be noted. For the survival analysis, individuals carrying with the rs895819 AG heterozygosis had better survival compared with individuals with the AA wild-type genotype in the TNM stage I–II subgroup. And it was confirmed by the vitro study that cells carrying polymorphic-type G allele expressed lower miR-27a than wild-type A allele. In conclusion, miR-27a rs895819 is implicated as a biomarker for gastric cancer and atrophic gastritis risk, and interacts with H. pylori in gastric carcinogenesis.
Methods
Study design
This study was approved by the Ethics Committee of the First Affiliated Hospital of China Medical University and the methods were carried out in accordance with the relevant guidelines and regulations. All participants were non-consanguineous ethnic Han Chinese. This study was divided into three parts focusing on risk, prognosis and miRNA expression. For the evaluation of risk, the study design comprised two stages using two different detection methods (direct sequencing and Sequenom Mass-ARRAY analysis) to investigate a total of 939 gastric cancer patients, 1,067 atrophic gastritis patients and 1,166 controls. To explore the association between miR-27a rs895819 with the risk of gastric cancer and atrophic gastritis, the first stage of the study involved direct sequencing of the samples from 724 retrospectively recruited individuals, consisting of 215 gastric cancer patients, 205 atrophic gastritis cases and 304 controls who attended the First Affiliated Hospital of China Medical University between 2005 and 2010. In the second stage of the study, the Sequenom Mass-ARRAY platform was used to investigate a total of 2,448 cases, consisting of 724 gastric cancer cases, 862 atrophic gastritis cases and 862 matched healthy controls from the Zhuanghe Gastric Diseases Screening Program39 or from patients who attended the First Affiliated Hospital of China Medical University between 2002 and 2013. All the subjects in this study were endoscopically and histologically confirmed. The classification of gastric cancer was based on Lauren’s classification22, which was divided into intestinal-type and diffuse-type for subgroup analysis. The classification and grading of gastritis were based on the Updated Sydney System40,41. Subjects who were endoscopically and histologically confirmed with normal mucosa or only minimal gastritis without other systemic disease and stomach diseases served as controls. Written informed consent was obtained from all participants. Medical histories (including details of age, sex, smoking, and alcohol consumption) were obtained by questionnaire and the records were computerized.
To further investigate the correlation of risk-associated polymorphisms with clinicopathologic parameters and survival in gastric cancer patients, we analyzed the data of 357 gastric cancer cases, which were all suffered surgical resection and the information of death or survival was available. The tumour histological grade was evaluated according to the World Health Organization criteria and tumours were staged using the 7th edition of the TNM Staging System of the International Union Against Cancer (UICC)/American Joint Committee on Cancer (AJCC) (2010) based on post-operative pathologic examinations. Patients (i) with distant metastasis found preoperatively, (ii) who underwent preoperative radiotherapy or chemotherapy, or (iii) with incomplete pathological data entries were excluded from the survival analysis. The information on clinicopathologic parameters were collected at baseline and the complete pathological data were obtained including macroscopic type, histologic grade, depth of invasion, number of LNs retrieved, number of metastatic LNs, and number of tumor deposits retrieved. The following data were obtained for all patients: date of death (if applicable), cause of death (if applicable), and date of follow-up. The primary endpoint was cause-specific survival duration from the date of gastric cancer diagnosis to the date of death. The median follow-up time was 23.0 months.
For the evaluation of a correlation between rs895819 and expression of the corresponding mature miRNA in serum, 170 cases consisting of 87 gastric cancer patients and 83 healthy controls were examined. In addition, for the assessment of correlation between rs895819 and miR-27a expression in gastric tissue, 32 non-cancerous specimens and 31 gastric cancerous specimens were obtained from 35 patients who underwent gastrectomy at the First Affiliated Hospital of China Medical University between 2009 and 2013.
Genotyping of subjects
Genomic DNA was extracted as described previously42, with some modifications. The genotyping in stage 1 was performed by direct sequencing carried out by Huada Gene Company (Shenzhen, China) to identify rs895819 and rs11671784 genotypes. The fragments for sequencing were amplified by PCR using the primers F: 5′-AACTTAGCCACTGTGAACACG-3′ and R: 5′-AGTTGCTGTAGCCTCCTTGTC-3′ with an annealing temperature of 59 °C. The genotyping in stage 2 assay was performed using the Sequenom Mass-ARRAY platform (Sequenom, San Diego, CA, USA) by Biomiao (Beijing, China) as described previously43. 5% samples in the same groups were random chosen to be detected in both the direct sequencing and MALDI-TOF Mass-ARRAY methods to test the consistency of these two methods, with a concordance rate of 100%. Another 5% samples were random chosen to be detected in the same method, and the consistency also reached 100%.
Serum H. pylori-IgG titer determination
Serum H. pylori-IgG titers were determined by enzyme linked immunosorbent assay (ELISA, Helicobacter pylori-IgG kit; Biohit, Helsinki, Finland) according to a previously described method44. H. pylori-IgG titers >35 was judged to be positive.
RNA Extraction and real-time PCR analysis of miRNA expression in vivo
MiRNA was extracted from serum and tissue samples as described previously45 with some modifications. The One Step Prime Script miRNA cDNA (Perfect Real-Time) Kit (TAKARA Biotechnology Co., Ltd, Dalian, China) was used to generate cDNA and the miRcute miRNA qPCR detection kit (SYBR) (TIANGEN Biotech Co., Ltd, Beijing, China) was used for real-time PCR analysis with the primer 5′-CGCGTTCACCGTGGCTAAGTTCC-3′. The methods used have been described previously46,47.
Transient transfection and Real-time PCR reaction for miRNA expression in vitro
The commercial expression plasmid pCMV-miR-27a-rs895819-A was purchased from Genechem Company (Genechem Biotech Co., Ltd, Shanghai, China). And the polymorphic plasmid was conducted for site-specific mutagenesis from A to G (pCMV-miR-27a-rs895819-G) by Genechem Company and confirmed by sequencing. Then, the lowest miR-27a expression cell line, AGS, was selected for transfection (More details see Supplementary Methods and Supplementary Figure 1). After 24 hours, the total RNA of cells was extracted and Real-time PCR was used to detect miR-27a expression after reverse transcription in order to compare the mature miR-27a produced by pCMV-miR-27a-rs895819-G vs. pCMV-miR-27a-rs895819-A.
Statistical analysis
To determine the genetic models for rs895819 SNP, we employed the method suggested by Thakkinstian et al.20. The strength of association between genotype and disease risk was assessed by odds ratio (OR) and 95% confidence interval (CI) measures, adjusted by sex, age and status of H. pylori infection. Genetic effects for three pairwise comparisons were calculated to determine the optimal genetic model for each polymorphism, including OR1 for GG versus AA, OR2 for AG versus AA, and OR3 for GG versus AG. If OR1 = OR3 ≠ 1 and OR2 = 1, a recessive model is suggested and GG was compared with the group of AG plus AA. If OR1 = OR2 ≠ 1 and OR3 = 1, a dominant model is indicated and the group of GG plus AG was compared with AA. If OR2 = 1/OR3 ≠ 1 and OR1 = 1, a complete overdominant model is suggested and AG was compared with the group of GG and AA. If OR1 > OR2 > 1 and OR1 > OR3 > 1, or OR1 < OR2 < 1 and OR1 < OR3 < 1, a codominant model is indicated and GG was compared with AG and with AA. Hardy–Weinberg equilibrium (HWE) among the controls was analyzed using the χ2 test. Continuous variables were represented as mean ± standard deviation (SD) and compared by analysis of variance (ANOVA), while the discrete variables were represented as frequencies and percentages and compared by the χ2 test. Multivariate logistic regression with adjustment for age, sex and H. pylori infection was used to assess the association between miR-27a polymorphism and gastric disease risk. The heterogeneity test for the gender, age and H. pylori infection status was performed by STATA software, version 11.0 (STATA Corp., College Station, TX, USA). Data for smoking and alcohol consumption was missing in nearly a third of cases and was not suitable for being adjustment factors. Therefore, these characteristics were used only as stratified factors for analysis of the association between the miR-27a polymorphism and disease risks48. Univariate and multivariate survival analyses were carried out using the log-rank test and the Cox proportional hazards model. The survival curves were mapped using the Kaplan–Meier method. Multivariate survival analysis was carried out by adding details of the SNP to all clinicopathological parameters with P < 0.05. In addition, the lg value of the copies of miRNA was used for a normal distribution, and the effect of miR-27a polymorphism on expression level was tested by ANOVA. The lg value of the miR-27a copies expressed by the AGS cells carrying with different miR-27a alleles were analyzed by nonparametric test because of the abnormal distribution. Statistical analysis was performed using SPSS version 18.0 software (SPSS, Chicago, IL, USA) and P-values < 0.05 was considered to be of statistical significance.
Additional Information
How to cite this article: Xu, Q. et al. MiR-27a rs895819 is involved in increased atrophic gastritis risk, improved gastric cancer prognosis and negative interaction with Helicobacter pylori. Sci. Rep. 7, 41307; doi: 10.1038/srep41307 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
This work is supported by grants from the National Science and technology support project (2015BAI13B07), the National Key Basic Research Program of China (973 Program ref no. 2010CB529304), the National Natural Science Foundation of China (Ref No. 31200968).
Footnotes
The authors declare no competing financial interests.
Author Contributions Study design: Y.Y. Perform experiments: Q.X. and T.J.C. Data interpretation: C.Y.H., and L.P.S. Drafting manuscript: Q.X. and Y.Y. Revising manuscript content: Q.X., J.W.L. and Y.Y. Approving final version of manuscript: Q.X., T.J.C., C.Y.H., L.P.S., J.W.L., Y.Y.
References
- Shastry B. S. SNPs: impact on gene function and phenotype. Methods in molecular biology (Clifton, NJ, 578, 3–22 (2009). [DOI] [PubMed] [Google Scholar]
- Song B., Yan G., Hao H. & Yang B. rs11671784 G/A and rs895819 A/G polymorphisms inversely affect gastric cancer susceptibility and miR-27a expression in a Chinese population. Medical science monitor: international medical journal of experimental and clinical research 20, 2318–2326 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pivarcsi A., Stahle M. & Sonkoly E. Genetic polymorphisms altering microRNA activity in psoriasis–a key to solve the puzzle of missing heritability? Experimental dermatology 23, 620–624 (2014). [DOI] [PubMed] [Google Scholar]
- Ha M. & Kim V. N. Regulation of microRNA biogenesis. Nature reviews Molecular cell biology 15, 509–524 (2014). [DOI] [PubMed] [Google Scholar]
- Xu Q., He C. Y., Liu J. W. & Yuan Y. Pre-miR-27a rs895819A/G polymorphisms in cancer: a meta-analysis. PLoS One 8, e65208 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng Y., Yi R. & Cullen B. R. Recognition and cleavage of primary microRNA precursors by the nuclear processing enzyme Drosha. The EMBO journal 24, 138–148 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R. et al. A genetic variant in the pre-miR-27a oncogene is associated with a reduced familial breast cancer risk. Breast cancer research and treatment 121, 693–702 (2010). [DOI] [PubMed] [Google Scholar]
- Kontorovich T. et al. Single nucleotide polymorphisms in miRNA binding sites and miRNA genes as breast/ovarian cancer risk modifiers in Jewish high-risk women. International journal of cancer 127, 589–597 (2010). [DOI] [PubMed] [Google Scholar]
- Shi D. et al. A genetic variant in pre-miR-27a is associated with a reduced renal cell cancer risk in a Chinese population. PLoS One 7, e46566 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Q. et al. Hsa-mir-27a genetic variant contributes to gastric cancer susceptibility through affecting miR-27a and target gene expression. Cancer science 101, 2241–2247 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M. et al. Associations of miRNA polymorphisms and female physiological characteristics with breast cancer risk in Chinese population. European journal of cancer care 21, 274–280 (2012). [DOI] [PubMed] [Google Scholar]
- Hezova R. et al. Evaluation of SNPs in miR-196-a2, miR-27a and miR-146a as risk factors of colorectal cancer. World journal of gastroenterology: WJG 18, 2827–2831 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catucci I. et al. The SNP rs895819 in miR-27a is not associated with familial breast cancer risk in Italians. Breast cancer research and treatment 133, 805–807 (2012). [DOI] [PubMed] [Google Scholar]
- Yang R. & Burwinkel B. A bias in genotyping the miR-27a rs895819 and rs11671784 variants. Breast cancer research and treatment 134, 899–901 (2012). [DOI] [PubMed] [Google Scholar]
- de Martel C., Forman D. & Plummer M. Gastric cancer: epidemiology and risk factors. Gastroenterology clinics of North America 42, 219–240 (2013). [DOI] [PubMed] [Google Scholar]
- Meng W. et al. Role of Helicobacter pylori in gastric cancer: advances and controversies. Discovery medicine 20, 285–293 (2015). [PubMed] [Google Scholar]
- Hong J. B., Zuo W., Wang A. J. & Lu N. H. Helicobacter pylori Infection Synergistic with IL-1beta Gene Polymorphisms Potentially Contributes to the Carcinogenesis of Gastric Cancer. International journal of medical sciences 13, 298–303 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang R. G., Duan G. C., Fan Q. T. & Chen S. Y. Role of Helicobacter pylori infection in pathogenesis of gastric carcinoma. World journal of gastrointestinal pathophysiology 7, 97–107 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figura N., Marano L., Moretti E. & Ponzetto A. Helicobacter pylori infection and gastric carcinoma: Not all the strains and patients are alike. World journal of gastrointestinal oncology 8, 40–54 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thakkinstian A. et al. A method for meta-analysis of molecular association studies. Statistics in medicine 24, 1291–1306 (2005). [DOI] [PubMed] [Google Scholar]
- Song M. Y. et al. Genetic polymorphisms of miR-146a and miR-27a, H. pylori infection, and risk of gastric lesions in a Chinese population. PLoS One 8, e61250 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lauren P. The Two Histological Main Types of Gastric Carcinoma: Diffuse and So-Called Intestinal-Type Carcinoma. an Attempt at a Histo-Clinical Classification. Acta pathologica et microbiologica Scandinavica 64, 31–49 (1965). [DOI] [PubMed] [Google Scholar]
- Wang F. et al. Protective role of Helicobacter pylori infection in prognosis of gastric cancer: evidence from 2,454 patients with gastric cancer. PLoS One 8, e62440 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu M. S. et al. Alterations of BAT-26 identify a subset of gastric cancer with distinct clinicopathologic features and better postoperative prognosis. Hepato-gastroenterology 49, 285–289 (2002). [PubMed] [Google Scholar]
- Lee H. S. et al. Distinct clinical features and outcomes of gastric cancers with microsatellite instability. Modern pathology: an official journal of the United States and Canadian Academy of Pathology, Inc. 15, 632–640 (2002). [DOI] [PubMed] [Google Scholar]
- Link A., Kupcinskas J., Wex T. & Malfertheiner P. Macro-role of microRNA in gastric cancer. Digestive diseases (Basel, Switzerland) 30, 255–267 (2012). [DOI] [PubMed] [Google Scholar]
- Arisawa T. et al. A polymorphism of microRNA 27a genome region is associated with the development of gastric mucosal atrophy in Japanese male subjects. Digestive diseases and sciences 52, 1691–1697 (2007). [DOI] [PubMed] [Google Scholar]
- Kupcinskas J. et al. Gene polymorphisms of micrornas in Helicobacter pylori-induced high risk atrophic gastritis and gastric cancer. PLoS One 9, e87467 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amstutz U. et al. Polymorphisms in MIR27A Associated with Early-Onset Toxicity in Fluoropyrimidine-Based Chemotherapy. Clin Cancer Res. 21, 2038–2044 (2015). [DOI] [PubMed] [Google Scholar]
- Ahn D. H. et al. Association of the miR-146aC > G, miR-149T > C, miR-196a2T > C, and miR-499A > G polymorphisms with gastric cancer risk and survival in the Korean population. Molecular carcinogenesis (2012). [DOI] [PubMed] [Google Scholar]
- Christensen B. C. et al. Mature microRNA sequence polymorphism in MIR196A2 is associated with risk and prognosis of head and neck cancer. Clin Cancer Res. 16, 3713–3720 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calin G. A. et al. A MicroRNA signature associated with prognosis and progression in chronic lymphocytic leukemia. The New England journal of medicine 353, 1793–1801 (2005). [DOI] [PubMed] [Google Scholar]
- Liu T. et al. MicroRNA-27a functions as an oncogene in gastric adenocarcinoma by targeting prohibitin. Cancer letters 273, 233–242 (2009). [DOI] [PubMed] [Google Scholar]
- Wan X. et al. Androgen-induced miR-27A acted as a tumor suppressor by targeting MAP2K4 and mediated prostate cancer progression. The international journal of biochemistry & cell biology 79, 249–260 (2016). [DOI] [PubMed] [Google Scholar]
- Zhou L. et al. MiR-27a-3p functions as an oncogene in gastric cancer by targeting BTG2. Oncotarget (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie M. et al. miR-23a and miR-27a promote human granulosa cell apoptosis by targeting SMAD5. Biology of reproduction 93, 98 (2015). [DOI] [PubMed] [Google Scholar]
- Zhang N. et al. A genetic variant in pre-miR-27a is associated with a reduced breast cancer risk in younger Chinese population. Gene 529, 125–130 (2013). [DOI] [PubMed] [Google Scholar]
- Yang Q. et al. Genetic variations in miR-27a gene decrease mature miR-27a level and reduce gastric cancer susceptibility. Oncogene 33, 193–202 (2014). [DOI] [PubMed] [Google Scholar]
- Tu H. et al. Temporal changes in serum biomarkers and risk for progression of gastric precancerous lesions: a longitudinal study. International journal of cancer 136, 425–434 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixon M. F., Genta R. M., Yardley J. H. & Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. The American journal of surgical pathology 20, 1161–1181 (1996). [DOI] [PubMed] [Google Scholar]
- Stolte M. & Meining A. The updated Sydney system: classification and grading of gastritis as the basis of diagnosis and treatment. Canadian journal of gastroenterology = Journal canadien de gastroenterologie 15, 591–598 (2001). [DOI] [PubMed] [Google Scholar]
- Xu Q. et al. Risk of gastric cancer is associated with the MUC1 568 A/G polymorphism. International journal of oncology 35, 1313–1320 (2009). [DOI] [PubMed] [Google Scholar]
- Xu Q. et al. SNP-SNP interactions of three new pri-miRNAs with the target gene PGC and multidimensional analysis of H. pylori in the gastric cancer/atrophic gastritis risk in a Chinese population. Oncotarget 7, 23700–23714 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong Y. H., Sun L. P., Jin S. G. & Yuan Y. Comparative study of serology and histology based detection of Helicobacter pylori infections: a large population-based study of 7,241 subjects from China. Eur J Clin Microbiol Infect Dis. 29, 907–911 (2010). [DOI] [PubMed] [Google Scholar]
- Liu R. et al. A five-microRNA signature identified from genome-wide serum microRNA expression profiling serves as a fingerprint for gastric cancer diagnosis. Eur J Cancer 47, 784–791 (2011). [DOI] [PubMed] [Google Scholar]
- Xu Q. et al. Expression of serum miR-20a-5p, let-7a, and miR-320a and their correlations with pepsinogen in atrophic gastritis and gastric cancer: a case-control study. BMC clinical pathology 13, 11 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Q. et al. A New Polymorphism Biomarker rs629367 Associated with Increased Risk and Poor Survival of Gastric Cancer in Chinese by Up-Regulated miRNA-let-7a Expression. PLoS One 9, e95249 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Q. et al. Promoter polymorphisms in trefoil factor 2 and trefoil factor 3 genes and susceptibility to gastric cancer and atrophic gastritis among Chinese population. Gene 529, 104–112 (2013). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.