Abstract
Administration of second-generation antipsychotic drugs (SGAs) often leads to weight gain and consequent cardio-metabolic side effects. We observed that clozapine but not six other antipsychotic drugs reprogrammed the gene expression pattern of differentiating human adipocytes ex vivo, leading to an elevated expression of the browning marker gene UCP1, more and smaller lipid droplets and more mitochondrial DNA than in the untreated white adipocytes. Laser scanning cytometry showed that up to 40% of the differentiating single primary and Simpson–Golabi–Behmel syndrome (SGBS) adipocytes had the characteristic morphological features of browning cells. Furthermore, clozapine significantly upregulated ELOVL3, CIDEA, CYC1, PGC1A and TBX1 genes but not ZIC1 suggesting induction of the beige-like and not the classical brown phenotype. When we tested whether browning induced by clozapine can be explained by its known pharmacological effect of antagonizing serotonin (5HT) receptors, it was found that browning cells expressed 5HT receptors 2A, 1D, 7 and the upregulation of browning markers was diminished in the presence of exogenous 5HT. Undifferentiated progenitors or completely differentiated beige or white adipocytes did not respond to clozapine administration. The clozapine-induced beige cells displayed increased basal and oligomycin-inhibited (proton leak) oxygen consumption, but these cells showed a lower response to cAMP stimulus as compared with control beige adipocytes indicating that they are less capable to respond to natural thermogenic anti-obesity cues. Our data altogether suggest that novel pharmacological stimulation of these masked beige adipocytes can be a future therapeutic target for the treatment of SGA-induced weight gain.
Introduction
Obesity is one of the major risk factors of metabolic syndrome, coronary heart disease and cancer, which are leading causes of morbidity and mortality today.1, 2 Recent studies showed high incidence of metabolically active brown adipose tissue (BAT), which can dissipate energy directly into heat in healthy adult humans and revealed the strong negative correlation between obesity and the amount of BAT.3, 4, 5 Energy expenditure of BAT is not exclusively mediated by the ab ovo-differentiated classical brown adipocytes.6 Beige cells that can be found interspersed in subcutaneous white adipose tissue (WAT) and generated in a process called ‘browning' also has a significant role.7, 8, 9 Although several studies suggest that a large proportion of the thermogenic fat depots in adult humans is mostly composed of beige cells, there is only limited information about the origin of beige adipocytes and the regulators of beige adipogenesis in humans.10, 11, 12 The available data obtained from mouse models and human samples suggest that the differentiation of beige cells, characterized by the elevated expression of the beige-selective TBX1 gene,10 is mostly induced from a distinct precursor as a result of several stimuli (for example, cold, physical exercise, diet) at least partially regulated by the β-adrenergic signaling pathway.13, 14 Beige adipocyte development can be highly enhanced by several neuro-endocrine or paracrine factors (for example, norepinephrine, irisin, atrial natriuretic peptide, bone morphogenic protein-7), which determine a beige potential or thermogenic competency of each individual.15, 16, 17 When the thermogenic stimulus subsides, ‘masked' beige cells persist that they, however, have a white adipocyte-like morphology in vivo.14, 18 Both ‘mature' and ‘masked' beige adipocytes are able to activate UCP1 expression and their thermogenic capacity in response to a recurring β-adrenergic stimulus.10, 14, 18 Thus, the augmentation of beige potential or the identification and subsequent thermogenic induction of ‘masked' beige adipocytes, for example, by stimulating the β3-adrenergic pathway can be a future therapeutic target against obesity and type 2 diabetes mellitus.19, 20
The prevalence rate of obesity and its comorbidities is at least two times higher in patients suffering from schizophrenia or other severe mental illnesses compared with the general population.21, 22 Moreover, the long-term administration of second-generation antipsychotic drugs (SGAs), especially clozapine, olanzapine, risperidone and quetiapine further increases the incidence of weight gain and metabolic syndrome in patients with severe mental illnesses.23, 24, 25 Different mechanisms were proposed, which might underlie the SGA-induced weight gain.26 These include the increase of appetite related to the activation of hypothalamic AMP kinase via histamine H1 receptors in rodents,27 decrease of insulin sensitivity in rats,28 defects in insulin secretion or clearance in dogs29 or rats,30 altered gut microbiota in rats31 and humans,32 induced low-grade inflammation in the adipose tissue in rats33, 34 and humans35 and direct stimulation of adipogenesis in rodents.36, 37
Previously, we demonstrated that the long-term SGA administration enhanced the expression of several white and general adipogenic marker genes and pro-inflammatory mediators in differentiating human adipocytes.35 Surprisingly, we also observed that the treatment with clozapine but not with other antipsychotic drugs resulted in the elevated expression of the major brown and beige adipocyte marker gene, UCP1. Using gene expression measurements, functional analysis of mitochondrial respiration and the recently developed laser scanning cytometry (LSC)-based technique, which could clearly discriminate between the white and brown adipocytes in cell culture conditions,38 we found that clozapine modified the differentiation program of human adipocyte precursors, presumably via the inhibition of serotonin (5HT) receptor-mediated signaling, leading to the generation of beige adipocytes with masked and not fully responsive thermogenic potential.
Materials and methods
Materials
All the chemicals were from Sigma-Aldrich (Munich, Germany) unless stated otherwise.
Ethics statement
Human adipose-derived mesenchymal stem cells (hADMSCs) were isolated from subcutaneous abdominal adipose tissue of healthy volunteers (body mass index <29.9) aged 20–65 years who underwent a planned surgical treatment (herniotomy). Written informed consent from all the participants was obtained before the surgical procedure. The study protocol was approved by the Ethics Committee of the University of Debrecen, Hungary (No. 3186-2010/DEOEC RKEB/IKEB). All the experiments were carried out in accordance with the approved ethical guidelines and regulations.
Isolation of hADMSCs, cell culture, ex vivo differentiation induction and treatments
The hADMSCs were isolated and cultivated from the stromal-vascular fraction (SVF) of abdominal subcutaneous fat as described previously.35, 38 White35, 38, 39, 40 and positive control beige adipocytes38, 41, 42 were differentiated from the hADMSCs, or from the Simpson–Golabi–Behmel syndrome (SGBS) preadipocyte cell line39, 40 (University Medical Center Ulm, Ulm, Germany) according to the already described protocols. The absence of mycoplasma was checked by polymerase chain reaction (PCR) analysis (PCR Mycoplasma Test Kit I/C, PromoKine, PromoCell France). The antipsychotic drugs were dissolved in dimethyl sulfoxide and differentiating adipocytes were treated every day in the following final concentrations: olanzapine 50 ng ml−1, ziprasidone 50 ng ml−1, quetiapine 50 ng ml−1, aripiprazole 100 ng ml−1, haloperidol 10 ng ml−1 and risperidone 50 ng ml−1.35 Clozapine was administered at 100 ng ml−1 concentration to the hADMSCs and to the fully differentiated white or brown adipocytes for 12 h (short-term treatment) or on the last 2 and 4 days or during the whole white adipogenic differentiation process (long-term treatment). Where indicated, the cells were treated with 5HT at 10 μm concentration during the whole adipocyte differentiation.43 To investigate the response of differentiated adipocytes to thermogenic induction, the cells received a single bolus of dibutyril-cAMP at 500 μm concentration for 4 h.6
RNA and DNA preparation, PCR array and TaqMan real-time PCR
The messenger RNA (mRNA) expressions in response to the treatment with the seven antipsychotic drugs were determined with CAPH09329 Custom Human RT2 Profiler PCR Arrays (SABiosciences, Frederick, MD, USA) as described previously.35 See reference 38 for details about TaqMan Real-time PCR. Mitochondrial DNA quantification by Quantitative PCR was carried out as described previously.38 Quantitative PCR experiments were repeated at least five times with SVFs from independent healthy donors or with SGBS samples from independent passages.
Immunofluorescence staining, image acquisition and quantification of ex vivo brown adipocyte differentiation
The hADMSCs or SGBS cells were plated on Ibidi eight-well microslides and differentiated as previously described. Vital and immunofluorescence stainings were carried out as described previously.38 First- and second-sample scanning was done by iCys Research Imaging Cytometer (iCys, Thorlabs Imaging Systems, Sterling, VA, USA). The images were processed and analyzed (n=3, 2000 cells per donor or SGBS sample) by our high-throughput automatic cell-recognition protocol using the iCys companion software (iNovator Application Development Toolkit, CompuCyte Corporation, Westwood, MA, USA) and CellProfiler (The Broad Institute of MIT, MA, USA). See reference ref. 38 for further details about the analysis.
Immunoblotting
The polyvinylidene fluoride membranes were probed by polyclonal anti-Ucp1 (U6382) and monoclonal anti-Gapdh (Millipore, Billerica, MA, USA, MAB374) antibodies overnight at 4 °C, followed by incubation with horseradish-peroxidase-conjugated species-corresponding secondary antibodies (Covalab, Villeurbanne, France, lab0252 and lab0273) for 1 h at room temperature. Densitometry was carried out using Image J software (n=3). See reference ref. 38 for a more detailed description.
Oxygen consumption
Oxygen consumption was measured using an XF96 oxymeter (Seahorse Biosciences, North Billerica, MA, USA). The hADMSCs were seeded and differentiated in 96-well XF96 assay plates. After recording the baseline OC, the cells received a single bolus of dibutyril-cAMP at 500 μm concentration modeling adrenergic stimulation. Then, stimulated OC was recorded every 30 min. The final reading took place at 5 h post treatment. To exclude that dibutyril-cAMP enhanced lipolysis might provoke artefactual mitochondrial uncoupling, we repeated the OC measurements including 2% BSA in the respiration buffer.44 The adipocytes were treated with 5 μm etomoxir or with 2 mm β-guanidinopropionic acid to block beta-oxidation and creatine-driven substrate cycle.45 Next, proton leak respiration was determined after adding oligomycin at 2 μm concentration to block ATP synthase activity. As a last step, the cells received a single bolus of Antimycin A at 10 μm concentration for baseline correction. The oxygen consumption rate (OCR) was normalized to protein content and normalized readings were displayed. For statistical analysis (n=4), the fold change of OC levels were calculated comparing the basal, cAMP-stimulated and oligomycin-inhibited (both in unstimulated and stimulated cells) OCRs of each sample to the basal OCR of untreated white adipocytes.38, 42, 46
Statistical analysis
The results are expressed as the mean±s.d. for the number of assays indicated. The sample sizes were chosen to ensure adequate statistical power following the practice of similar studies.35, 38, 40, 42 The normality of distribution of the data was tested by Kolmogorov–Smirnov test. For multiple comparisons of groups, statistical significance was calculated and evaluated by one-way analysis of variance followed by Tukey post hoc test. In comparison of two groups, two-tailed Student's t-test was used. The data were analyzed using Prism 6.01 (GraphPad Software, San Diego, CA, USA).
Results
Long-term clozapine treatment induces a beige-like gene expression pattern in differentiating adipocytes
The hADMSCs were cultivated from the SVF of abdominal subcutaneous fat35, 38 and a previously described differentiation protocol was applied to induce white adipocyte differentiation.35, 39 Using this ex vivo approach, up to 50% of the treated hADMSCs were able to accumulate large lipid droplets, which could be quantified by LSC in a time-dependent manner.40 Primarily, we aimed to investigate the direct effect of seven antipsychotic drugs at doses comparable to their therapeutic plasma concentrations35, 47, 48, 49, 50 on the gene expression of the differentiating adipocytes. After a 14 days long white adipogenic differentiation39 and parallel daily administration of antipsychotics, whole cell lysates were collected, then mRNA expressions were determined by a custom PCR Array.35 We found that clozapine administration resulted in a sixfold upregulation of UCP1, the major brown and beige adipocyte marker gene.51 None of the used other five SGAs (olanzapine, ziprasidone, quetiapine, aripiprazole and risperidone) and a typical antipsychotic (haloperidol) influenced the expression of UCP1 (Supplementary Figure 1).
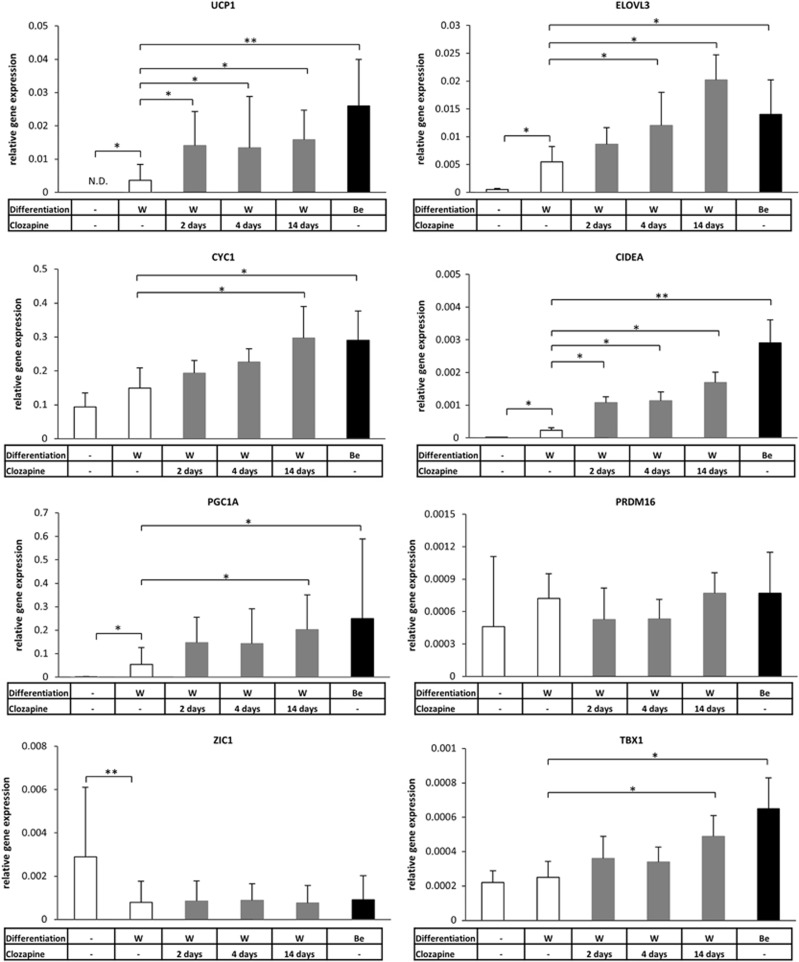
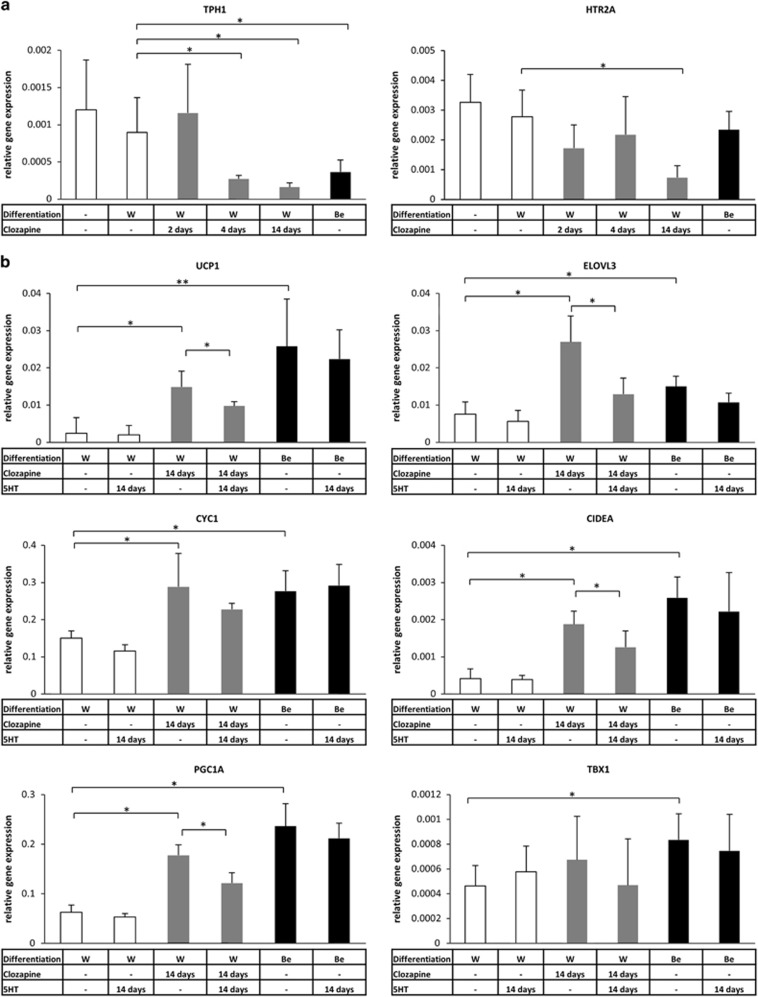
As a next step, we intended to validate the aforementioned effect of clozapine by TaqMan Real-time PCR with reverse transcription. In accordance with our previous results, the expression of UCP1 was undetectable in hADMSCs and showed a basal level in adipocytes differentiated by the white adipogenic protocol for 14 days.38 The differentiating adipocytes were treated with clozapine on the last 2 and 4 days or during the whole white adipogenic differentiation process. We found that UCP1 was expressed fivefold higher as a result of clozapine administration in each case (Figure 1). A previously described brown adipocyte differentiation protocol,41 which turned out to induce a beige program rather than classical brown,38 was applied as positive control and resulted in a high expression level of the UCP1 gene; we are referring to these cells as ‘positive control beige adipocytes'.38, 41 We also aimed to analyze the expression of a core set of brown fat-specific genes (CIDEA,52 PGC1A53 and ELOVL354), an indicator of the mitochondrial enrichment (CYC1),51 a beige (TBX1)10 and a classical brown adipocyte marker (ZIC1)6 in adipocytes differentiated in the presence of clozapine. Significantly elevated expression of CIDEA, CYC1, ELOVL3, PGC1A and the beige indicator TBX1 was found in a concentration-dependent manner compared with the untreated white adipocytes (Figure 1 and Supplementary Figure 2). The long-term clozapine treatment was less effective in the induction of brown and beige-related genes than the above-mentioned browning protocol in most of the cases. The expression of ZIC1 remained at a low level after clozapine administration excluding that clozapine induces classical brown adipocyte differentiation. Furthermore, clozapine treatment moderately enhanced the expression of beta-oxidation-related mitochondrial genes (ACOX2, CPT1A, CPT2 and ACADM; Supplementary Figure 3a). In parallel, PRDM1655 (Figure 1), CEBPB56 (transcriptional regulators of brown and beige adipocyte development), CEBPA, FABP4, LEPTIN and PPARG (general adipocyte markers)57 genes expressed at the same level in adipocytes differentiated in the presence of clozapine compared with the untreated ones (Supplementary Figure 4). Undifferentiated progenitors or completely differentiated white or positive control beige adipocytes did not respond to the 12 h-long clozapine treatment (except for the slight upregulation of TBX1 in differentiated positive control beige adipocytes), suggesting that the adipogenic differentiation program is required for the aforementioned effect of clozapine (Supplementary Figure 5). However, the influence of clozapine on the induction of browning genes was complete in the majority of the cases even if applied only during the last 4 or even 2 days of white adipogenic differentiation (Figure 1). In the collected whole cell lysates of adipocytes that were differentiated in the presence of clozapine, a threefold elevated Ucp1 protein level was detected compared with the untreated cells (Figures 2a and b). Thus, direct clozapine administration during ex vivo human white adipocyte differentiation induces a beige-like gene expression pattern and results in the increased amount of Ucp1 protein.
Figure 1.
Relative expression of brown and beige adipogenic markers and key transcriptional regulators of brown adipocyte development in primary human adipocytes as a result of clozapine treatment during ex vivo white or beige adipocyte differentiation. SVF-derived hADMSCs were differentiated for 2 weeks to white (W) or positive control beige (Be) adipocytes. Clozapine (gray bars) was administered at 100 ng ml−1 concentration on the last 2 and 4 days or during the whole white adipogenic differentiation process. The experiment was repeated six times with SVFs from independent healthy donors (Relative gene expression was determined by RT-qPCR, target genes were normalized to GAPDH); *P<0.05, **P<0.01. hADMSC, human adipose-derived mesenchymal stem cell; RT-qPCR, quantitative PCR with reverse transcription; SVF, stromal-vascular fraction.
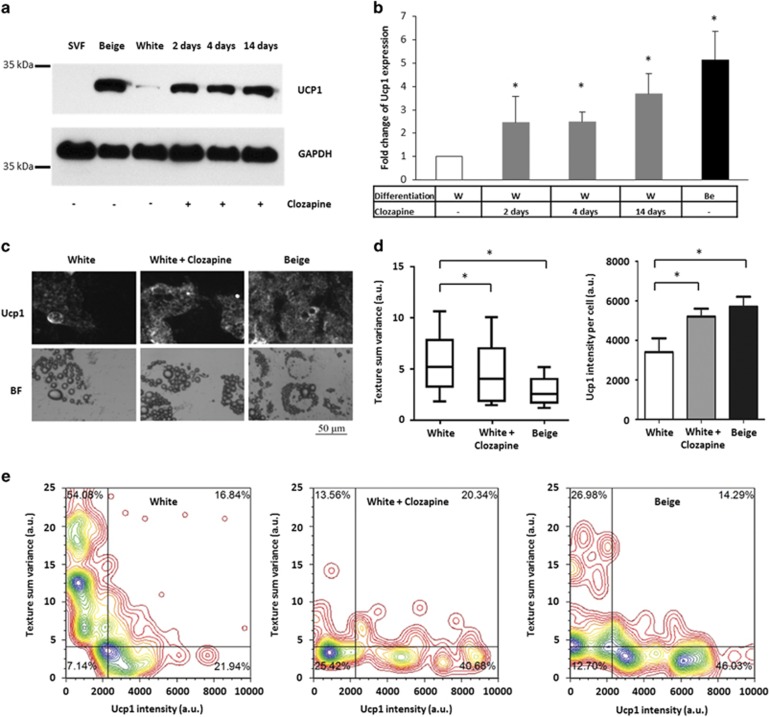
Figure 2.
Laser scanning cytometry-based population scale analysis of brown adipogenic differentiation by texture parameters and Ucp1 protein content of ex vivo-differentiated single primary adipocytes treated with clozapine. SVF-derived hADMSCs were differentiated and treated as in Figure 1. (a) Ucp1 protein expression in one representative adipocyte donor. (b) Protein expression level of Ucp1 in the adipocytes of three different SVF donors quantified by densitometry (as compared with untreated white adipocytes). (c) Distribution of Ucp1 in clozapine-treated human primary adipocytes. The images were collected with an iCys Research Imaging Cytometer. (d) Texture sum variance and Ucp1 protein content of adipocytes per cell. *P<0.05, n=3, 2000 cells each donor. (e) Dot plot figures of one representative donor based on the identification of browning adipocytes containing small lipid droplets and high levels of Ucp1. hADMSC, human adipose-derived mesenchymal stem cell; SVF, stromal-vascular fraction.
More hADMSCs and SGBS preadipocytes differentiate into adipocytes with morphological characteristics of browning as a result of clozapine treatment
It has been reported that a bolus clozapine treatment at the early stage of human adipocyte differentiation resulted in an increased lipid content.58 We have developed a LSC-based approach to analyze the attached adipocyte cultures at consecutive time points.40 This slide-based image cytometry approach combines texture analysis and detection of Ucp1 protein content in single brown adipocytes of mixed cell populations.38 In our experiments, up to 50% of the differentiated hADMSCs progressively accumulated lipid droplets; in this respect, there was no difference between clozapine-treated and untreated adipocytes (Figure 2c). However, instead of the formation of large lipid droplets, we found that the long-term clozapine administration resulted in the occurrence of more and smaller droplets in the differentiated adipocytes, similarly but less extensively than in the case of the positive control beige adipocytes. The quantification of this phenomenon by the texture ‘sum variance'38, 40, 42 is shown in Figure 2d. Significantly elevated Ucp1 protein content was detected simultaneously in single human adipocytes. Representative images are displayed in Figure 2c and the quantification of Ucp1 content in hADMSC-derived adipocytes of three different donors is presented in Figure 2d. In Figure 2e, we plotted Ucp1 content and texture ‘sum variance' for each differentiated adipocyte. Adipocytes with morphological characteristics of browning were recognized as the ones that contained small lipid droplets and high levels of Ucp1 protein (lower right quadrant of density plot images), in contrast to white adipocytes, which accumulated large lipid droplets and expressed low amount of Ucp1 (upper left quadrant of density plot images).38 The biological variance among donors of hADMSCs was summarized in Supplementary Table 1. Using this approach, we demonstrated that depending on individual donors 30–40% of differentiating single human primary adipocytes had the characteristic morphological features of browning cells as a result of clozapine treatment.
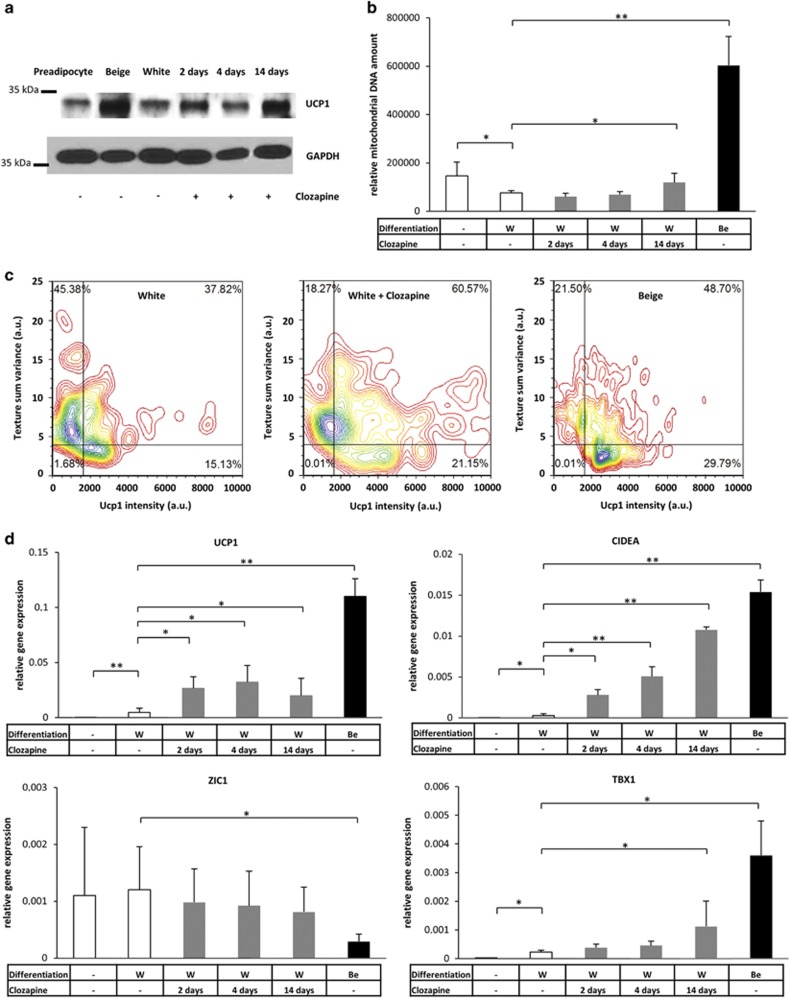
To exclude the possible impact of heterogeneity and the lower differentiation capacity of the primary cell model, we also investigated the aforementioned effect of clozapine in differentiating SGBS preadipocytes. SGBS cells, which are derived from a patient with SGBS, represent a well-accepted model of human white adipocyte differentiation.39, 40, 59, 60 We found elevated Ucp1 protein expression (Figure 3a) and moderately increased mitochondrial DNA amount (Figure 3b) in the cell lysates of SGBS adipocytes that were differentiated in the presence of clozapine compared with the untreated white fat cells. Morphological features of browning (Figure 3c) and a beige-like gene expression pattern (Figure 3d) could be also detected in clozapine-treated SGBS adipocytes. Thus, the data from both primary cells and an adipogenic cell line corroborate that clozapine can shift the differentiation program towards browning.
Figure 3.
Induction of browning in SGBS human preadipocyte cell line as a result of clozapine treatment during white adipocyte differentiation. Preadipocytes were differentiated for 2 weeks to white (W) or positive control beige (Be) adipocytes. Clozapine (gray bars) was administered at 100 ng ml−1 concentration on the last 2 and 4 days or during the whole white adipogenic differentiation process. (a) Ucp1 protein expression in one representative SGBS sample. (b) Relative mitochondrial DNA amount of SGBS adipocytes determined by qPCR in six different samples. (c) Laser scanning cytometry-based population scale analysis of browning in one representative SGBS sample by texture parameters and Ucp1 protein content of adipocytes. (d) Relative expression of browning marker genes in SGBS adipocytes. The experiment was repeated six times with samples from independent passages (relative gene expression was determined by RT-qPCR, target genes were normalized to GAPDH); *P<0.05, **P<0.01. RT-qPCR, quantitative PCR with reverse transcription; SGBS, Simpson–Golabi–Behmel syndrome.
Clozapine-treated white adipocytes have increased thermogenic competency but low sensitivity to thermogenic induction ex vivo
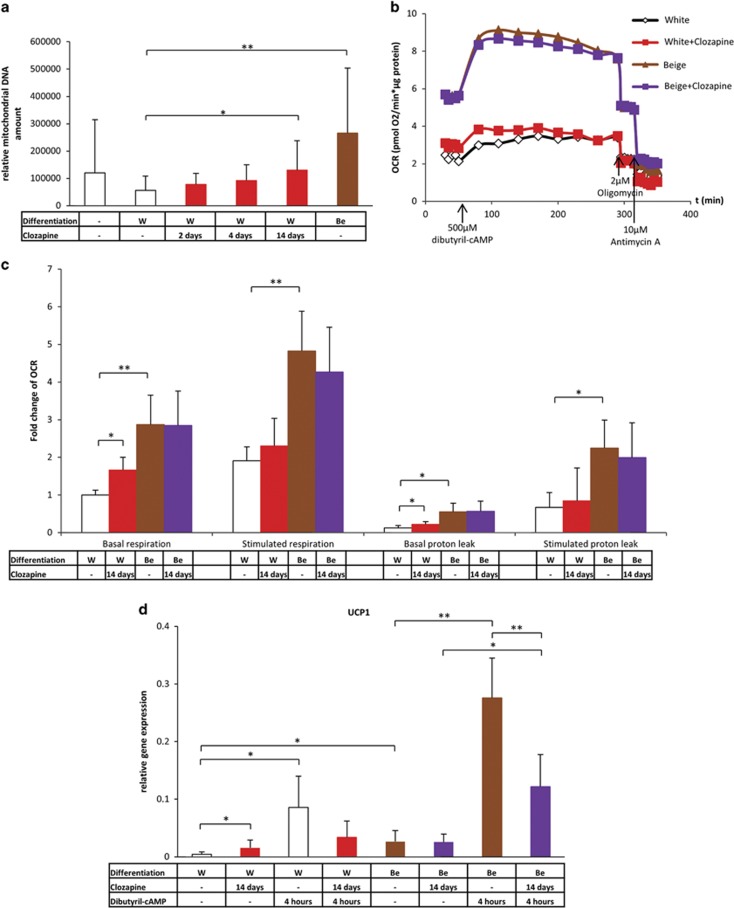
As a next step, we intended to investigate the functional capacity of human primary adipocytes differentiated in the presence of clozapine. Clozapine-treated primary adipocytes had a moderately increased mitochondrial DNA content, to a lesser extent as positive control beige adipocytes (Figure 4a). Figure 4b demonstrates the OC of SVF-derived adipocytes of one representative donor measured by an XF96 oxymeter. The basal mitochondrial respiration of clozapine-treated white adipocytes was moderately elevated as compared with the untreated white cells. In accordance with our previous results,38 ex vivo-differentiated positive control beige adipocytes had higher basal OCR than the white adipocytes; the presence or absence of clozapine during the differentiation did not affect the basal OCR of positive control beige adipocytes. After the cells received a single bolus dose of cell permeable dibutyril-cAMP mimicking adrenergic stimulation, we found that adipocytes that were differentiated in the presence of clozapine were less capable than the untreated cells to induce their respiration. At the maximal level of induced respiration, the difference in OCR demonstrated at the basal level between clozapine-treated and untreated white adipocytes was abolished. A similar trend was observed when cells were treated with oligomycin that blocks the activity of ATP synthase and provides information on proton leak respiration (Figure 4c). Considering the fact that dibutyril-cAMP enhanced lipolysis might increase the release of fatty acids by adipocytes, which can stimulate respiration by artefactual mitochondrial uncoupling,44 we carried out OC measurements including 2% BSA in the respiration buffer. Although we observed weaker induction of OCR in response to dibutyril-cAMP in each differentiated sample, the aforementioned effects of clozapine on the metabolic parameters of adipocytes could be detected in this experimental condition as well (Supplementary Figure 6).
Figure 4.
Functional analysis of ex vivo-differentiated primary adipocytes treated with clozapine. SVF-derived hADMSCs were differentiated for 2 weeks to white (W) or positive control beige (Be) adipocytes. Clozapine (red bars) was administered at 100 ng ml−1 concentration on the last 2 and 4 days or during the whole adipogenic differentiation process. (a) Relative mitochondrial DNA amount of human adipocytes determined by quantitative PCR in five different donors. (b) Oxygen consumption of SVF-derived adipocytes of one representative donor measured with an XF96 oxymeter. After recording the baseline oxygen consumption, the cells received a single bolus dose of dibutyril-cAMP. Then, stimulated oxygen consumption was recorded at every 30 min. Proton leak respiration was determined after adding oligomycin to block ATP synthase activity. (c) Basal, cAMP-stimulated and oligomycin-inhibited oxygen consumption levels (as compared with the basal OCR of white adipocytes) in four different SVF-derived adipocyte donors. (d) Effect of short-term cAMP treatment on the expression of UCP1 gene in primary adipocytes. The experiment was repeated five times with SVFs from independent healthy donors (relative gene expression was determined by RT-qPCR, target gene was normalized to GAPDH); *P<0.05, **P<0.01. hADMSC, human adipose-derived mesenchymal stem cell; OCR, oxygen consumption rate; RT-qPCR, quantitative PCR with reverse transcription; SVF, stromal-vascular fraction.
Recent findings revealed a substrate cycle of creatine metabolism, which enhances energy expenditure in the mitochondria of beige adipocytes. Mitochondrial creatine kinase 1 or 2 catalyzes the conversion of creatine and utilizes ATP to create phosphocreatine (PCr) and ADP, before Phospho1 liberates the high-energy phosphate group from PCr.45 When adipocytes differentiated in the presence of clozapine were treated with etomoxir (O-carnitine palmitoyltransferase-1 inhibitor) or β-guanidinopropionic acid (creatine analog which reduces creatine levels in the cells), the basal and cAMP-stimulated OCRs were decreased at a higher extent then in the case of white adipocytes. These data demonstrate that the clozapine-induced browning cells (similar to positive control beige adipocytes) utilize more fatty acids by beta-oxidation than white adipocytes and further increase their energy expenditure by activating the recently identified futile cycle of creatine metabolism (Supplementary Figures 3b and 7).
In parallel, we analyzed the changes in the gene expression of the previously shown brown and beige adipocyte markers as a result of a 4 h-long dibutyril-cAMP treatment that serves as an ex vivo model of thermogenic induction.6 In line with the previously described results, white adipocytes expressed UCP1 gene at a basal level. White adipocytes differentiated in the presence of clozapine had a significantly elevated UCP1 expression compared with the untreated cells. Positive control beige adipocytes expressed UCP1 mRNA at a higher level; in this case the clozapine treatment did not affect UCP1 expression. A similar trend was observed in the case of PGC1A. The UCP1 and PGC1A expression showed a robust (ten- and fivefold, respectively) upregulation in response to cAMP treatment of white and positive control beige adipocytes differentiated in the absence of clozapine. This effect of thermogenic induction was less manifested in clozapine-generated beige adipocytes, with regard to UCP1. The responsiveness of positive control beige adipocytes differentiated in the presence of clozapine was also reduced (Figure 4d and Supplementary Figure 8). The expression of CYC1, CIDEA, ELOVL3, PRDM16, TBX1 and ZIC1 did not change significantly as a result of thermogenic induction in either clozapine-treated or untreated adipocytes (data not shown). Of note, beta-oxidation and creatine-driven substrate cycle was enhanced by cAMP in a similar extent in clozapine-induced as in positive control beige adipocytes (Supplementary Figures 3b and 7). Although the demonstrated gene expression, morphological and functional data suggest that clozapine treatment induces the thermogenic competency of differentiating white adipocytes, these ‘masked' beige cells are less capable to respond to β-adrenergic induction of thermogenesis.
Clozapine induces browning via inhibiting 5HT-receptor-mediated signaling
Next, we intended to learn the molecular mechanism, which can explain the browning effect of clozapine. Although initially introduced as a dopamine receptor antagonist in the clinical practice, clozapine has a very diverse molecular interaction profile. It binds to different 5HT, muscarinic and histamine receptors and antagonizes adrenergic alpha 1 and 2 or various dopamine receptors.61, 62, 63 Fourteen different 5HT-receptor types have been already described64 and most of them are expressed by white65, 66 and brown adipocytes67, 68 in rodents. In human adipocytes, the expression of HTR1D, HTR2A, HTR3A and HTR7 were reported in different studies.12, 69
Primarily, we investigated the expression of the above-mentioned 5HT-receptor genes. In our experiments, we found that HTR2A was expressed at the highest level in hADMSCs and in differentiated adipocytes. Interestingly, long-term clozapine administration resulted in the downregulation of HTR2A gene (Figure 5a). In addition, the HTR1D and HTR7 genes were expressed at a lower extent. In contrast to HTR2A, their expression was not altered in response to clozapine treatment (Supplementary Figure 9). We found a similar expression pattern in SGBS adipocytes, except for the undetectable expression level of HTR7 (data not shown). The rate-limiting step of 5HT synthesis in peripheral tissues is catalyzed by Tryptophan Hydroxylase 1 (TPH1).70 The expression of TPH1 was detectable in hADMSCs (Figure 5a) and in SGBS preadipocytes (data not shown), and did not change as a result of white adipocyte differentiation, suggesting that these cells are capable of autonomously generating and releasing 5HT during adipogenesis. However, we found decreased TPH1 expression in clozapine-induced and in positive control beige adipocytes (Figure 5a). As a next step, exogenous 5HT was administered daily to the differentiating primary adipocytes. We found that the upregulation of the majority of the browning markers was significantly attenuated when clozapine-induced beige adipocytes were differentiated in the presence of 5HT (Figure 5b). The expression of white and general adipogenic markers was not changed as a result of 5HT administration (Supplementary Figure 10). The SGBS adipocytes differentiated in the presence of clozapine similarly responded to the 5HT treatment (data not shown). Of note, the 5HT administration alone did not modify the gene expression pattern of differentiating white and positive control beige adipocytes. Our results suggest that disturbance of 5HT production or 5HT-mediated signaling might, at least partially, explain the browning effect of clozapine.
Figure 5.
Effect of 5HT on the induction of browning in ex vivo-differentiated primary human adipocytes treated with clozapine. SVF was differentiated for 2 weeks to white (W) or positive control beige (Be) adipocytes. 100 ng ml−1 clozapine (gray bars) and/or 10 μm 5HT was administered on the last 2 and 4 days or during the whole adipogenic differentiation process. (a) Relative expression of Tryptophan Hydroxylase 1 and HTR2A receptor in adipocytes in response to clozapine administration. (b) Relative expression of browning marker genes as a result of clozapine and 5HT treatment. The experiment was repeated five times with SVFs from independent healthy donors (Relative gene expression was determined by RT-qPCR, target genes were normalized to GAPDH); *P<0.05, **P<0.01. 5HT, serotonin; RT-qPCR, quantitative PCR with reverse transcription; SVF, stromal-vascular fraction.
Discussion
The mechanisms underlying SGA-induced metabolic dysfunctions remain barely understood.26 Only a few studies investigated the direct effect of SGAs on differentiating human adipocytes.47, 58 To fill this hiatus and to follow up our previous study where we looked at selected adipogenic, cell cycle-related and pro-inflammatory genes in SGA-treated human white adipocytes,35 in the present study we found that clozapine administration unexpectedly resulted in the upregulation of UCP1 (Supplementary Figure 1). This prompted us to examine how the propensity of hADMSCs and SGBS preadipocytes to differentiate into heat-generating brown or beige cells is influenced by clozapine. The administration of the drug at 20-fold higher concentration than the upper limit of therapeutic reference range resulted in a significant inhibition of the differentiation of a murine brown preadipocyte cell line.71 However, toxic effects of the treatment for the cultured cells cannot be excluded at this high clozapine concentration.47 On the other hand, clozapine administration at the concentrations of therapeutic reference range had a slight positive effect on the expression of the selected BAT marker genes in an in vitro mouse model.71 In our experiments, clozapine treatment resulted in significantly elevated expression of several brown marker genes, including the beige indicator TBX1, in differentiating human adipocytes.
We complemented the measurements of gene expression changes from total cell lysates with the recently described LSC approach that helped us to specifically identify browning cells in mixed adipocyte populations.38 When the adipocytes were differentiated in the presence of clozapine for two weeks, the rate of browning cells increased by approximately 1.5-fold. Clozapine did not, however, alter the gene expression pattern of fully differentiated white or positive control beige adipocytes. This indicates that clozapine, most probably, induces the commitment of mesenchymal adipocyte progenitor cells to beige adipocytes at some point during the differentiation. The ratio of energy-dissipating beige and energy-storing white adipocytes (which fundamentally defines the thermogenic competency of each individual) is determined, at least partially, during early differentiation of mesenchymal progenitors into adipocyte subtypes.14, 16, 17 The upregulation of the marker genes of browning was nearly the same when the administration of clozapine took place only on the last 2 or 4 days of the white adipogenic differentiation suggesting that clozapine is able to induce this developmental shift from white to beige even at the later stage of the ex vivo differentiation process.
Recently, two groups reported independently that peripheral 5HT reduced the beige potential and the sensitivity of brown and beige adipocytes to thermogenic induction in a cell autonomous manner in mice.43, 70 Increased levels of peripheral 5HT72 and polymorphisms in the TPH1 gene73 are associated with obesity. We found that TPH1, the enzyme which catalyzes the rate-limiting step of 5HT production outside the central nervous system, and 5HT-receptor genes are expressed in human primary and SGBS adipocytes and by undifferentiated precursors. Furthermore, exogenous 5HT partially abrogated the upregulation of browning marker genes that were induced by clozapine administration. In our experiments, out of the 5HT receptors, HTR2A was expressed at the highest level. HTR2A initiates Gq signaling, which was recently reported to abolish browning in mice and in human adipocytes. In parallel, the expression of Gq protein correlated inversely with UCP1 in WAT in vivo.74 As clozapine is able to antagonize several 5HT receptors, with the highest affinity to HTR2A;62 the disturbance of 5HT-receptor-mediated Gq signaling might, at least partially, explain the unexpected phenomenon described in the present study assuming that 5HT or similarly acting compounds are generated and released from cells during white adipocyte differentiation.
Previously, it was found that clozapine enhances the accumulation of lipids in differentiating human adipocytes which could stand in line with its documented effect to promote obesity.58 However, we failed to detect more or larger lipid droplets in the clozapine-treated adipocytes by LSC. These cells had smaller lipid droplets in multilocular arrangement, more mitochondrial DNA and expressed more Ucp1 protein than the untreated white adipocytes. On the other hand, it should be emphasized that beige fat cells differentiated from precursors (which process is enhanced by clozapine administration) do not activate the effective thermogenic program unless they are stimulated with certain inducers, for example β-adrenergic agonists.7, 10, 75 The question may arise why in vivo administration of clozapine leads to fat gain and obesity. A similar discrepancy has been observed with thiazolidinediones that act through the nuclear receptor PPARG.76, 77 Clinical trials implicated that rosiglitazone increases the risk of obesity, congestive heart failure and myocardial infarction in patients who received the drug for type 2 diabetes mellitus,78, 79, 80 while it was capable of increasing the thermogenic competency of WAT in mice7 and inducing browning of human adipocytes ex vivo.41 Our data suggest that adipocytes differentiated in the presence of clozapine are less sensitive to cAMP stimulation. In line with this, altered cAMP signaling has been reported in the brain as a result of SGA treatment in rodents81, 82 and in humans.83 The observed incomplete thermogenic response of clozapine-generated beige cells to β-adrenergic agonist can be one of the reasons why these ‘masked' beige adipocytes function ineffectively also in vivo. Results from in vivo studies focusing on the effect of SGAs on the sympathetic nervous system have been conflicting.84, 85, 86, 87 The β3-adrenergic pathway which has a key role in the activation of thermogenesis in brown and beige adipocytes20, 88, 89 has never, to our knowledge, been investigated in this regard. Further investigations will help to identify the ‘masked' beige cells differentiated in response to clozapine treatment in vivo and to find ways to induce their thermogenic function independently from the sympathetic nervous system.
Acknowledgments
This research was supported by the European Union and the State of Hungary, co-financed by the European Social Fund in the framework of TÁMOP-4.2.4.A/2-11/1-2012-0001 ‘National Excellence Program', which provided personal support to EK and AKS, TÁMOP-4.2.2.A-11/1/KONV-2012-0023, TÁMOP-4.2.2.A-11/1/KONV-2012-0025, GINOP-2.3.2-15-2016-00006 grant, the European Union Framework Programme 7 TRANSCOM IAPP 251506, OTKA K108308, NK105046 and the Hungarian Academy of Sciences. We thank Dr András Mádi and Dr Máté Demény for their consultation and for reviewing the manuscript. We acknowledge Jennifer Nagy and Szilvia Szalóki for excellent technical assistance.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
Supplementary Material
References
- Haslam DW, James WP. Obesity. Lancet 2005; 366: 1197–1209. [DOI] [PubMed] [Google Scholar]
- Park J, Euhus DM, Scherer PE. Paracrine and endocrine effects of adipose tissue on cancer development and progression. Endocr Rev 2011; 32: 550–570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med 2009; 360: 1509–1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T, Nio-Kobayashi J et al. High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 2009; 58: 1526–1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoneshiro T, Aita S, Matsushita M, Kayahara T, Kameya T, Kawai Y et al. Recruited brown adipose tissue as an antiobesity agent in humans. J Clin Invest 2013; 123: 3404–3408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cypess AM, White AP, Vernochet C, Schulz TJ, Xue R, Sass CA et al. Anatomical localization, gene expression profiling and functional characterization of adult human neck brown fat. Nat Med 2013; 19: 635–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrovic N, Walden TB, Shabalina IG, Timmons JA, Cannon B, Nedergaard J. Chronic peroxisome proliferator-activated receptor gamma (PPARgamma) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J Biol Chem 2010; 285: 7153–7164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J et al. Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 2011; 121: 96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harms M, Seale P. Brown and beige fat: development, function and therapeutic potential. Nat Med 2013; 19: 1252–1263. [DOI] [PubMed] [Google Scholar]
- Wu J, Boström P, Sparks LM, Ye L, Choi JH, Giang AH et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012; 150: 366–376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee P, Werner CD, Kebebew E, Celi FS. Functional thermogenic beige adipogenesis is inducible in human neck fat. Int J Obes (Lond) 2014; 38: 170–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinoda K, Luijten IH, Hasegawa Y, Hong H, Sonne SB, Kim M et al. Genetic and functional characterization of clonally derived adult human brown adipocytes. Nat Med 2015; 21: 389–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee YH, Petkova AP, Mottillo EP, Granneman JG. In vivo identification of bipotential adipocyte progenitors recruited by β3-adrenoceptor activation and high-fat feeding. Cell Metab 2012; 15: 480–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang QA, Tao C, Gupta RK, Scherer PE. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat Med 2013; 19: 1338–1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins S. A heart-adipose tissue connection in the regulation of energy metabolism. Nat Rev Endocrinol 2014; 10: 157–163. [DOI] [PubMed] [Google Scholar]
- Kajimura S, Spiegelman BM, Seale P. Brown and beige fat: physiological roles beyond heat generation. Cell Metab 2015; 22: 546–559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynes MD, Tseng YH. The thermogenic circuit: regulators of thermogenic competency and differentiation. Genes Dis 2015; 2: 164–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenwald M, Perdikari A, Rülicke T, Wolfrum C. Bi-directional interconversion of brite and white adipocytes. Nat Cell Biol 2013; 15: 659–667. [DOI] [PubMed] [Google Scholar]
- Whittle AJ, López M, Vidal-Puig A. Using brown adipose tissue to treat obesity—the central issue. Trends Mol Med 2011; 17: 405–411. [DOI] [PubMed] [Google Scholar]
- Cypess AM, Weiner LS, Roberts-Toler C, Franquet Elía E, Kessler SH, Kahn PA et al. Activation of human brown adipose tissue by a β3-adrenergic receptor agonist. Cell Metab 2015; 21: 33–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennekens CH, Hennekens AR, Hollar D, Casey DE. Schizophrenia and increased risks of cardiovascular disease. Am Heart J 2005; 150: 1115–1121. [DOI] [PubMed] [Google Scholar]
- De Hert M, Schreurs V, Vancampfort D, Van Winkel R. Metabolic syndrome in people with schizophrenia: a review. World Psychiatry 2009; 8: 15–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey DE, Haupt DW, Newcomer JW, Henderson DC, Sernyak MJ, Davidson M et al. Antipsychotic-induced weight gain and metabolic abnormalities: implications for increased mortality in patients with schizophrenia. J Clin Psychiatry 2004; 65(Suppl 7): 4–18. [PubMed] [Google Scholar]
- Newcomer JW. Second-generation (atypical) antipsychotics and metabolic effects: a comprehensive literature review. CNS Drugs 2005; 19(Suppl 1): 1–93. [DOI] [PubMed] [Google Scholar]
- De Hert M, Schreurs V, Sweers K, Van Eyck D, Hanssens L, Sinko S et al. Typical and atypical antipsychotics differentially affect long-term incidence rates of the metabolic syndrome in first-episode patients with schizophrenia: a retrospective chart review. Schizophr Res 2008; 101: 295–303. [DOI] [PubMed] [Google Scholar]
- Gohlke JM, Dhurandhar EJ, Correll CU, Morrato EH, Newcomer JW, Remington G et al. Recent advances in understanding and mitigating adipogenic and metabolic effects of antipsychotic drugs. Front Psychiatry 2012; 3: 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SF, Huang AS, Snowman AM, Teuscher C, Snyder SH. From the cover: antipsychotic drug-induced weight gain mediated by histamine H1 receptor-linked activation of hypothalamic AMP-kinase. Proc Natl Acad Sci USA 2007; 104: 3456–3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houseknecht KL, Robertson AS, Zavadoski W, Gibbs EM, Johnson DE, Rollema H. Acute effects of atypical antipsychotics on whole-body insulin resistance in rats: implications for adverse metabolic effects. Neuropsychopharmacology 2007; 32: 289–297. [DOI] [PubMed] [Google Scholar]
- Ader M, Kim SP, Catalano KJ, Ionut V, Hucking K, Richey JM et al. Metabolic dysregulation with atypical antipsychotics occurs in the absence of underlying disease: a placebo-controlled study of olanzapine and risperidone in dogs. Diabetes 2005; 54: 862–871. [DOI] [PubMed] [Google Scholar]
- Chintoh AF, Mann SW, Lam L, Giacca A, Fletcher P, Nobrega J et al. Insulin resistance and secretion in vivo: effects of different antipsychotics in an animal model. Schizophr Res 2009; 108: 127–133. [DOI] [PubMed] [Google Scholar]
- Davey KJ, Cotter PD, O'Sullivan O, Crispie F, Dinan TG, Cryan JF et al. Antipsychotics and the gut microbiome: olanzapine-induced metabolic dysfunction is attenuated by antibiotic administration in the rat. Transl Psychiatry 2013; 3: e309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bahr SM, Tyler BC, Wooldridge N, Butcher BD, Burns TL, Teesch LM et al. Use of the second-generation antipsychotic, risperidone, and secondary weight gain are associated with an altered gut microbiota in children. Transl Psychiatry 2015; 5: e652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victoriano M, de Beaurepaire R, Naour N, Guerre-Millo M, Quignard-Boulangé A, Huneau JF et al. Olanzapine-induced accumulation of adipose tissue is associated with an inflammatory state. Brain Res 2010; 1350: 167–175. [DOI] [PubMed] [Google Scholar]
- Zhang Q, He M, Deng C, Wang H, Huang XF. Effects of olanzapine on the elevation of macrophage infiltration and pro-inflammatory cytokine expression in female rats. J Psychopharmacol 2014; 28: 1161–1169. [DOI] [PubMed] [Google Scholar]
- Sárvári AK, Veréb Z, Uray IP, Fésüs L, Balajthy Z. Atypical antipsychotics induce both proinflammatory and adipogenic gene expression in human adipocytes in vitro. Biochem Biophys Res Commun 2014; 450: 1383–1389. [DOI] [PubMed] [Google Scholar]
- Vestri HS, Maianu L, Moellering DR, Garvey WT. Atypical antipsychotic drugs directly impair insulin action in adipocytes: effects on glucose transport, lipogenesis, and antilipolysis. Neuropsychopharmacology 2007; 32: 765–772. [DOI] [PubMed] [Google Scholar]
- Yang LH, Chen TM, Yu ST, Chen YH. Olanzapine induces SREBP-1-related adipogenesis in 3T3-L1 cells. Pharmacol Res 2007; 56: 202–208. [DOI] [PubMed] [Google Scholar]
- Kristóf E, Doan-Xuan QM, Bai P, Bacso Z, Fésüs L. Laser-scanning cytometry can quantify human adipocyte browning and proves effectiveness of irisin. Sci Rep 2015; 5: 12540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer-Posovszky P, Newell FS, Wabitsch M, Tornqvist HE. Human SGBS cells - a unique tool for studies of human fat cell biology. Obes Facts 2008; 1: 184–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doan-Xuan QM, Sarvari AK, Fischer-Posovszky P, Wabitsch M, Balajthy Z, Fesus L et al. High content analysis of differentiation and cell death in human adipocytes. Cytometry A 2013; 83: 933–943. [DOI] [PubMed] [Google Scholar]
- Elabd C, Chiellini C, Carmona M, Galitzky J, Cochet O, Petersen R et al. Human multipotent adipose-derived stem cells differentiate into functional brown adipocytes. Stem Cells 2009; 27: 2753–2760. [DOI] [PubMed] [Google Scholar]
- Abdul-Rahman O, Kristóf E, Doan-Xuan QM, Vida A, Nagy L, Horváth A et al. AMP-activated kinase (AMPK) activation by AICAR in human white adipocytes derived from pericardial white adipose tissue stem cells induces a partial beige-like phenotype. PLoS One 2016; 11: e0157644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh CM, Namkung J, Go Y, Shong KE, Kim K, Kim H et al. Regulation of systemic energy homeostasis by serotonin in adipose tissues. Nat Commun 2015; 6: 6794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Fromme T, Schweizer S, Schöttl T, Klingenspor M. Taking control over intracellular fatty acid levels is essential for the analysis of thermogenic function in cultured primary brown and brite/beige adipocytes. EMBO Rep 2014; 15: 1069–1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazak L, Chouchani ET, Jedrychowski MP, Erickson BK, Shinoda K, Cohen P et al. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 2015; 163: 643–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tews D, Schwar V, Scheithauer M, Weber T, Fromme T, Klingenspor M et al. Comparative gene array analysis of progenitor cells from human paired deep neck and subcutaneous adipose tissue. Mol Cell Endocrinol 2014; 395: 41–50. [DOI] [PubMed] [Google Scholar]
- Sertié AL, Suzuki AM, Sertié RA, Andreotti S, Lima FB, Passos-Bueno MR et al. Effects of antipsychotics with different weight gain liabilities on human in vitro models of adipose tissue differentiation and metabolism. Prog Neuropsychopharmacol Biol Psychiatry 2011; 35: 1884–1890. [DOI] [PubMed] [Google Scholar]
- Coryell W, Miller DD, Perry PJ. Haloperidol plasma levels and dose optimization. Am J Psychiatry 1998; 155: 48–53. [DOI] [PubMed] [Google Scholar]
- Broich K, Heinrich S, Marneros A. Acute clozapine overdose: plasma concentration and outcome. Pharmacopsychiatry 1998; 31: 149–151. [DOI] [PubMed] [Google Scholar]
- Bergemann N, Frick A, Parzer P, Kopitz J. Olanzapine plasma concentration, average daily dose, and interaction with co-medication in schizophrenic patients. Pharmacopsychiatry 2004; 37: 63–68. [DOI] [PubMed] [Google Scholar]
- Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev 2004; 84: 277–359. [DOI] [PubMed] [Google Scholar]
- Hallberg M, Morganstein DL, Kiskinis E, Shah K, Kralli A, Dilworth SM et al. A functional interaction between RIP140 and PGC-1alpha regulates the expression of the lipid droplet protein CIDEA. Mol Cell Biol 2008; 28: 6785–6795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 1998; 92: 829–839. [DOI] [PubMed] [Google Scholar]
- Tvrdik P, Asadi A, Kozak LP, Nedergaard J, Cannon B, Jacobsson A. Cig30, a mouse member of a novel membrane protein gene family, is involved in the recruitment of brown adipose tissue. J Biol Chem 1997; 272: 31738–31746. [DOI] [PubMed] [Google Scholar]
- Seale P, Bjork B, Yang W, Kajimura S, Chin S, Kuang S et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature 2008; 454: 961–967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajimura S, Seale P, Kubota K, Lunsford E, Frangioni JV, Gygi SP et al. Initiation of myoblast to brown fat switch by a PRDM16-C/EBP-beta transcriptional complex. Nature 2009; 460: 1154–1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowe CE, O'Rahilly S, Rochford JJ. Adipogenesis at a glance. J Cell Sci 2011; 124: 2681–2686. [DOI] [PubMed] [Google Scholar]
- Hemmrich K, Gummersbach C, Pallua N, Luckhaus C, Fehsel K. Clozapine enhances differentiation of adipocyte progenitor cells. Mol Psychiatry 2006; 11: 980–981. [DOI] [PubMed] [Google Scholar]
- Claussnitzer M, Dankel SN, Kim KH, Quon G, Meuleman W, Haugen C et al. FTO obesity variant circuitry and adipocyte browning in humans. N Engl J Med 2015; 373: 895–907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wabitsch M, Brenner RE, Melzner I, Braun M, Möller P, Heinze E et al. Characterization of a human preadipocyte cell strain with high capacity for adipose differentiation. Int J Obes Relat Metab Disord 2001; 25: 8–15. [DOI] [PubMed] [Google Scholar]
- Naheed M, Green B. Focus on clozapine. Curr Med Res Opin 2001; 17: 223–229. [DOI] [PubMed] [Google Scholar]
- Correll CU. From receptor pharmacology to improved outcomes: individualising the selection, dosing, and switching of antipsychotics. Eur Psychiatry 2010; 25(Suppl 2): S12–S21. [DOI] [PubMed] [Google Scholar]
- Taly A. Novel approaches to drug design for the treatment of schizophrenia. Expert Opin Drug Discov 2013; 8: 1285–1296. [DOI] [PubMed] [Google Scholar]
- Nichols DE, Nichols CD. Serotonin receptors. Chem Rev 2008; 108: 1614–1641. [DOI] [PubMed] [Google Scholar]
- Kinoshita M, Ono K, Horie T, Nagao K, Nishi H, Kuwabara Y et al. Regulation of adipocyte differentiation by activation of serotonin (5-HT) receptors 5-HT2AR and 5-HT2CR and involvement of microRNA-448-mediated repression of KLF5. Mol Endocrinol 2010; 24: 1978–1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stunes AK, Reseland JE, Hauso O, Kidd M, Tømmerås K, Waldum HL et al. Adipocytes express a functional system for serotonin synthesis, reuptake and receptor activation. Diabetes Obes Metab 2011; 13: 551–558. [DOI] [PubMed] [Google Scholar]
- Uldry M, Yang W, St-Pierre J, Lin J, Seale P, Spiegelman BM. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab 2006; 3: 333–341. [DOI] [PubMed] [Google Scholar]
- Timmons JA, Wennmalm K, Larsson O, Walden TB, Lassmann T, Petrovic N et al. Myogenic gene expression signature establishes that brown and white adipocytes originate from distinct cell lineages. Proc Natl Acad Sci USA 2007; 104: 4401–4406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tchkonia T, Lenburg M, Thomou T, Giorgadze N, Frampton G, Pirtskhalava T et al. Identification of depot-specific human fat cell progenitors through distinct expression profiles and developmental gene patterns. Am J Physiol Endocrinol Metab 2007; 292: E298–E307. [DOI] [PubMed] [Google Scholar]
- Crane JD, Palanivel R, Mottillo EP, Bujak AL, Wang H, Ford RJ et al. Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat Med 2015; 21: 166–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh JE, Cho YM, Kwak SN, Kim JH, Lee KW, Jung H et al. Inhibition of mouse brown adipocyte differentiation by second-generation antipsychotics. Exp Mol Med 2012; 44: 545–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim HJ, Kim JH, Noh S, Hur HJ, Sung MJ, Hwang JT et al. Metabolomic analysis of livers and serum from high-fat diet induced obese mice. J Proteome Res 2011; 10: 722–731. [DOI] [PubMed] [Google Scholar]
- Kwak SH, Park BL, Kim H, German MS, Go MJ, Jung HS et al. Association of variations in TPH1 and HTR2B with gestational weight gain and measures of obesity. Obesity (Silver Spring) 2012; 20: 233–238. [DOI] [PubMed] [Google Scholar]
- Klepac K, Kilić A, Gnad T, Brown LM, Herrmann B, Wilderman A et al. The Gq signalling pathway inhibits brown and beige adipose tissue. Nat Commun 2016; 7: 10895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaus S, Ely M, Encke D, Heldmaier G. Functional assessment of white and brown adipocyte development and energy metabolism in cell culture. Dissociation of terminal differentiation and thermogenesis in brown adipocytes. J Cell Sci 1995; 108: 3171–3180. [DOI] [PubMed] [Google Scholar]
- Ahmadian M, Suh JM, Hah N, Liddle C, Atkins AR, Downes M et al. PPARγ signaling and metabolism: the good, the bad and the future. Nat Med 2013; 19: 557–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen ED, Spiegelman BM. What we talk about when we talk about fat. Cell 2014; 156: 20–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kung J, Henry RR. Thiazolidinedione safety. Expert Opin Drug Saf 2012; 11: 565–579. [DOI] [PubMed] [Google Scholar]
- Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 2007; 356: 2457–2471. [DOI] [PubMed] [Google Scholar]
- Graham DJ, Ouellet-Hellstrom R, MaCurdy TE, Ali F, Sholley C, Worrall C et al. Risk of acute myocardial infarction, stroke, heart failure, and death in elderly Medicare patients treated with rosiglitazone or pioglitazone. JAMA 2010; 304: 411–418. [DOI] [PubMed] [Google Scholar]
- Dwivedi Y, Rizavi HS, Pandey GN. Differential effects of haloperidol and clozapine on [(3)H]cAMP binding, protein kinase A (PKA) activity, and mRNA and protein expression of selective regulatory and catalytic subunit isoforms of PKA in rat brain. J Pharmacol Exp Ther 2002; 301: 197–209. [DOI] [PubMed] [Google Scholar]
- Turalba AV, Leite-Morris KA, Kaplan GB. Antipsychotics regulate cyclic AMP-dependent protein kinase and phosphorylated cyclic AMP response element-binding protein in striatal and cortical brain regions in mice. Neurosci Lett 2004; 357: 53–57. [DOI] [PubMed] [Google Scholar]
- Marazziti D, Baroni S, Palego L, Betti L, Giannaccini G, Castagna M et al. Clozapine effects on adenylyl cyclase activity and serotonin type 1 A receptors in human brain post-mortem. J Psychopharmacol 2014; 28: 320–328. [DOI] [PubMed] [Google Scholar]
- Elman I, Goldstein DS, Green AI, Eisenhofer G, Folio CJ, Holmes CS et al. Effects of risperidone on the peripheral noradrenegic system in patients with schizophrenia: a comparison with clozapine and placebo. Neuropsychopharmacology 2002; 27: 293–300. [DOI] [PubMed] [Google Scholar]
- Monda M, Viggiano A, Viggiano A, Fuccio F, De Luca V. Clozapine blocks sympathetic and thermogenic reactions induced by orexin A in rat. Physiol Res 2004; 53: 507–513. [PubMed] [Google Scholar]
- Savoy YE, Ashton MA, Miller MW, Nedza FM, Spracklin DK, Hawthorn MH et al. Differential effects of various typical and atypical antipsychotics on plasma glucose and insulin levels in the mouse: evidence for the involvement of sympathetic regulation. Schizophr Bull 2010; 36: 410–418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blessing WW, Seaman B, Pedersen NP, Ootsuka Y. Clozapine reverses hyperthermia and sympathetically mediated cutaneous vasoconstriction induced by 3,4-methylenedioxymethamphetamine (ecstasy) in rabbits and rats. J Neurosci 2003; 23: 6385–6391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inokuma K, Okamatsu-Ogura Y, Omachi A, Matsushita Y, Kimura K, Yamashita H et al. Indispensable role of mitochondrial UCP1 for antiobesity effect of beta3-adrenergic stimulation. Am J Physiol Endocrinol Metab 2006; 290: E1014–E1021. [DOI] [PubMed] [Google Scholar]
- Mund RA, Frishman WH. Brown adipose tissue thermogenesis: β3-adrenoreceptors as a potential target for the treatment of obesity in humans. Cardiol Rev 2013; 21: 265–269. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.