Abstract
Species differences exist in terms of drug oxidation activities, which are mediated mainly by cytochrome P450 (P450) enzymes. To overcome the problem of species extrapolation, transchromosomic mice containing a human P450 3A cluster or chimeric mice transplanted with human hepatocytes have been introduced into the human toxicology research area. In this review, drug metabolism and disposition mediated by humanized livers in chimeric mice are summarized in terms of biliary/urinary excretions of phthalate and bisphenol A and plasma clearances of the human cocktail probe drugs caffeine, warfarin, omeprazole, metoprolol, and midazolam. Simulation of human plasma concentrations of the teratogen thalidomide and its human metabolites is possible with a simplified physiologically-based pharmacokinetic model based on data obtained in chimeric mice, in accordance with reported clinical thalidomide concentrations. In addition, in vivo non-specific hepatic protein binding parameters of metabolically activated 14C-drug candidate and hepatotoxic medicines in humanized liver mice can be analyzed by accelerator mass spectrometry and are useful for predictions in humans.
Graphical Abstract
1. Introduction
The human cytochrome P450 gene superfamily comprises 57 genes and 58 pseudogenes.1 The corresponding P450 (P450 or CYP) enzymes are involved in the oxidative metabolism of a variety of endogenous compounds, medicines, and toxic chemicals. Rodents are often used as animal models in drug development, but it is well known that species differences exist in terms of drug metabolism mediated mainly by rodent and human P450s.2 The relevance and limitations of animal models used in non-clinical safety assessments of investigational products and new medicines needs to be carefully considered. To overcome the species differences, humanized mice have been widely developed by introducing human specific and/or multiple genes for drug metabolizing P450s,3,4 NAD(P)H:quinone oxidoreductase5, and nuclear receptors that control their expression.6,7 Another type of humanized model mice has been also developed by transplanting human hepatocytes in immunodeficient mice.8–10 NOG mice expressing transgenic urokinase-type plasminogen activator in the liver were produced,9 and replacement by human hepatocytes could be estimated by human albumin concentrations in the blood because the humanized mice produces human albumin. Humanized (TK-NOG) mice expressing a herpes simplex virus type 1 thymidine kinase transgene had a human-specific profile of drug metabolism.10
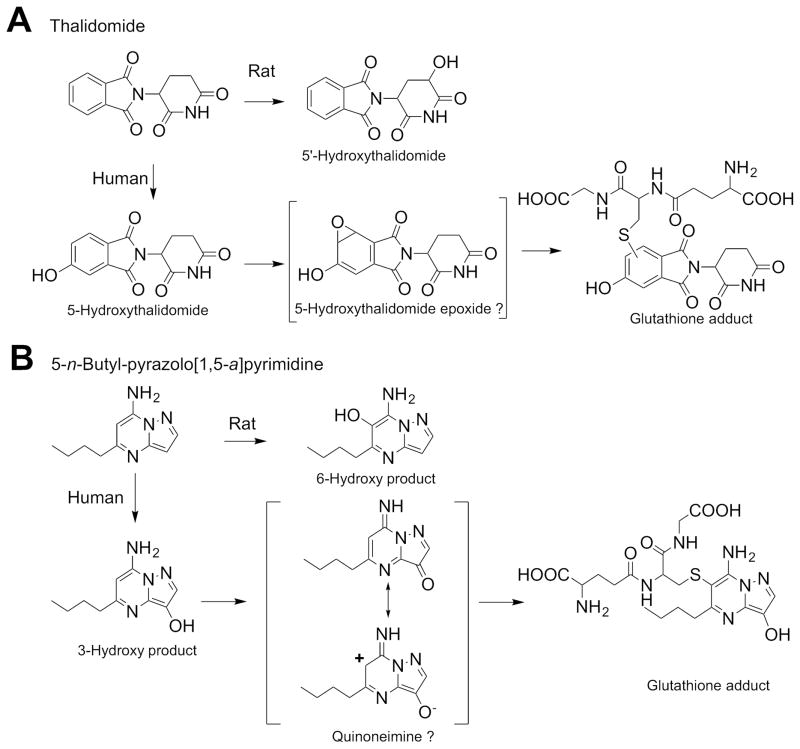
One of the classical species differences is seen in thalidomide, a teratogen in humans or non-human primates11 but not in rodents. Recently, a whole-embryo culture system from transchromosomic mice containing a human cytochrome P450 3A cluster12 (in which the endogenous mouse P450 3a genes were deleted) showed limb abnormalities,13 suggesting that the humanized P450 3A mouse is a useful model for predicting toxicity in humans. Thalidomide is metabolized via two major pathways, 5′-hydroxythalidomide (a major product in rodents) and 5-hydroxythalidomide (human proportionate phenyl ring-based metabolites, Figure 1).14 Furthermore, thalidomide and primary 5-hydroxylated metabolites (including 5,6-dihydroxythalidomide and GSH conjugate(s)) were detected by mass spectrometry (MS) in plasma from chimeric mice with highly “humanized” liver cells harboring cytochrome P450 3A5*1.14 Chimeric mice with humanized liver revealed that the second oxidation step in human proportionate 5-hydroxythalidomide pathway generated a reactive intermediate that can be trapped by GSH to give GSH adducts,14 indicating that this model is useful for predicting toxic metabolites in humans.
Figure 1.
Metabolic pathways of thalidomide (A) and 5-n-butyl-pyrazolo[1,5-a]pyrimidine (B). Proposed formation of the glutathione conjugate (A) is modified from Chowdhury et al.43. Proposed mechanism for a putative quinone imine metabolite and conjugate formation at the 6-position (B) is taken from Kuribayashi et al.50
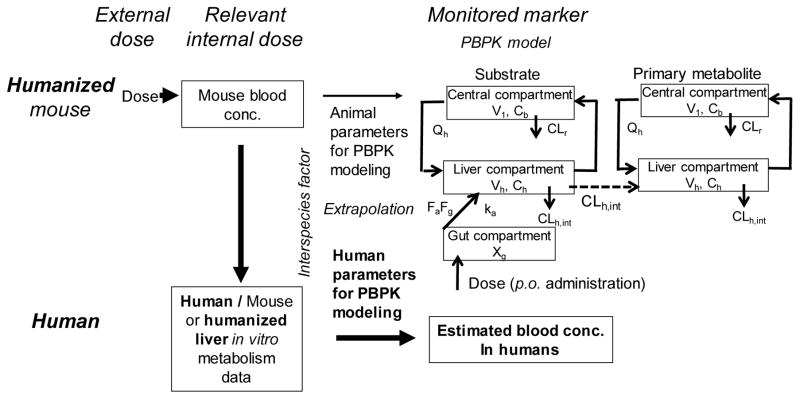
In vitro–in vivo extrapolations of hepatic clearance and disposition have been reported for a wide variety of drugs.15 Simplified physiologically-based pharmacokinetic (PBPK) models consist of a chemical receptor compartment, a metabolizing compartment, and a central compartment (Figure 2).14,16,17 Subsequently, final parameter values (including standard deviation values) for an animal PBPK model can be calculated to give the best fit to measured blood substrate concentration values. Differential equations can be solved to estimate blood concentrations of substrates and/or metabolites after oral administration.14,16,17 These simplified human PBPK models for industrial chemicals with toxicity concerns (e.g., bisphenol A17 and di(2-ethylhexyl)phthalate16 and the pesticides acephate18 and chlorpyrifos18) were recently developed and successfully used to estimate human pharmacokinetic parameters, based on the pharmacokinetics in humanized mice.
Figure 2.
PBPK model for animals, humanized TK-NOG mice, and humans.
There is considerable interest in the importance of drug metabolites as potential determinants of drug safety.19 Guidance notes for Industry Safety Testing of Drug Metabolites (issued in 2008 by the United States Food and Drug Administration) laid out criteria regarding the circumstances under which direct testing of a metabolite in animal toxicology studies is needed to provide a reliable risk assessment of human health. Recent developments in chimeric mice with humanized liver20 and in bioanalytical methodology with LC-MS/MS systems have provided several strategies to generate data that can guide critical decisions related to metabolite quantitation and biomonitoring in plasma.
In this review article, drug metabolism and disposition mediated by humanized livers in chimeric mice are summarized. These models may be useful for evaluating the relationships between biliary and urinary excretion, clearances in plasma, human metabolite formation, and non-specific protein binding of drugs and their metabolites and the potential toxicity in humans. These findings provide examples of the usefulness of transplanted human liver cells in humanized mice to provide accurate preclinical predictions of human drug metabolism and disposition, with the aid of LC-MS/MS systems.
2. Disposition and Clearance of Industrial Chemicals and Human P450 Probe Drugs in Humanized Liver Mice
Species variations in the threshold molecular weight factor for the biliary excretion of orally administered compounds have been recognized in mice (325 ± 50) and humans (500 ± 50);21 urinary excretion is extensive for the compounds of lower molecular weight and tends to decrease with increasing molecular weight because bile and urine are complementary excretory pathways in animals. Although a renal clearance-type drug (cefmetazole) has been mainly excreted in urines of humanized mice urine but not in control mice,22 the hepatic metabolite excretion into urine was not confirmed in humanized mice.
In immunodeficient TK-NOG mice, transplanted human hepatocytes (which express similar human P450 mRNA levels and have catalytic function as transplanted human hepatocytes) were maintained after an initial exposure to a non-toxic dose of ganciclovir to ablate the mouse liver cells.10 The pharmacokinetics of mono(2-ethylhexyl)phthalate (MEHP) (a primary metabolite of di(2-ethylhexyl)phthalate (DEHP))16 and bisphenol A O-glucuronide17 (after oral administration of DEHP (250 mg/kg) and bisphenol A (100 mg/kg)) were determined in order to extrapolate these experimental data from chimeric mice transplanted with human hepatocytes to virtual administration in humans. Biphasic plasma concentration–time curves of MEHP and its glucuronide and high fecal excretion levels of MEHP glucuronide were seen in control mice, although MEHP and its glucuronide were extensively excreted in urine within 24 h in mice when humanized liver mice were used.
For running simple PBPK modeling, physicochemical properties (i.e. plasma unbound fraction and octanol–water partition coefficients) and the pharmacokinetic parameters (e.g., absorption rate constant (ka), volume of the systemic circulation (V1), and hepatic intrinsic clearance (CLh,int)) were estimated and calculated by fitting.16,17 Typical physiological hepatic blood flow rates in mice (0.16 L/h) and humans (97 L/h) were used.23,24 Using known species allometric scaling factors, estimated urine MEHP concentrations in humans based on the pharmacokinetics in mice with humanized liver by a simple PBPK model16 were consistent with the reported concentrations.25 These findings showed that transplanted human hepatocytes could affect the extensive excretion of primary and secondary metabolites of DEHP into urine in chimeric mice, as in the cases of marmosets26 or humans.25 Simplified PBPK models were used with both forward and reverse dosimetry and were able to estimate human plasma and urinary concentrations of MEHP16 and bisphenol A O-glucuronide after ingestion of bisphenol A17. The Fourth National Report on Human Exposure to Environmental Chemicals, Updated Tables, August 2014 (U. S. Centers for Disease Control)27 indicates geometric means and 95th percentile values of urinary MEHP concentrations for men in the USA in 2005–2006 of 3.4 and 50 μg/L, respectively. Urinary total concentrations of bisphenol A in the US population in 2003–2004 were 2.6 and 16 μg/L, respectively, which were the highest values recorded in the period 1999–2010. The MEHP concentrations in urine can be ascribed to exposure of 0.087 μg/kg/day and 1.3 μg/kg/day DEHP and to 0.067 μg/kg/day and 0.41 μg/kg/day bisphenol A, respectively, by reverse dosimetry with the human PBPK model (Table 1), assuming that the reported urinary concentrations had reached steady-state values. These estimated DEHP and bisphenol A exposures are far less than the daily tolerable intake of DEHP (30 μg/kg/day28,29 or 50 μg/kg/day, EU Public Helth, 2008) and daily tolerable intake of bisphenol A (50 μg/kg/day),30 implying little risk of either compound in humans under average conditions.
Table 1.
Reported urinary concentrations of DEHP and bisphenol A and their estimated exposures.
| urinary concentrations for men in USA, μg/L | estimated exposure, μg/kg/day | daily tolerated intake, μg/kg/day | ||
|---|---|---|---|---|
| D(M)EHP (2005–2006) | geometric mean, 95th percentile, | 3.4 50 |
0.087 1.3 |
30 or 50 |
| bisphenol A (2003–2004) | geometric mean, 95th percentile, | 2.6 16 |
0.067 0.41 |
50 |
Details are presented in the text.
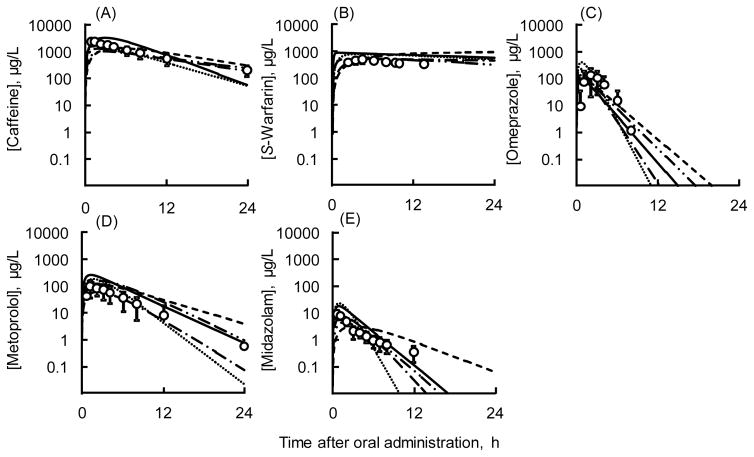
This simple system was also successful in estimating human plasma concentrations of various P450 probes based on non-human primate, dog, and minipig plasma data31–33. This simple system was also successful in estimating human plasma concentrations of various P450 probes in humans extrapolated from corresponding plasma data in humanized liver mice and marmosets,32 dogs31, minipigs31, and monkeys.33 Pharmacokinetic parameters for humanized liver mice and other animals (marmosets, monkeys, dogs, and minipigs) determined in in vivo experiments with P450 cocktail probes using LC-MS/MS methods described elsewhere.31–33 are described (Table 2). Briefly, observed plasma concentrations of caffeine, warfarin, omeprazole, metoprolol, and midazolam in chimeric TK-NOG mice with humanized liver were scaled to human oral monitoring equivalents using known species allometric scaling factors. Human plasma concentration profiles of the five P450 probes estimated by simplified human PBPK models (based on the observed pharmacokinetics in mice with humanized liver) were consistent with previously published pharmacokinetic data in Caucasians (Figure 3). Similarly, using the same approach, the previously reported pharmacokinetics of the five P450 probes in marmosets, monkeys, dogs, and minipigs (Supporting Table S1) were also scaled to reported equivalents in humans using in vitro metabolic clearance data.31,33 The results in Figure 3 (created with the parameters in Supporting Table S1) suggest that mice with humanized liver and/or marmosets, monkeys, dogs, and minipigs can be used as suitable pharmacokinetic models for humans during research with many new drugs, especially when used in combination with simple PBPK models with LC-MS/MS analytical systems for drug monitoring. Human hepatic clearance values of omeprazole, metoprolol, and midazolam in the simple PBPK models based on humanized mice and average parameters for humans were 52.2 and 40.9 (± 9.9) L/h, 46.5 and 55.9 (±10.6) L/h, and 31.7 and 41.5 (± 10.4) L/h, respectively (Table 2). The expected AUClast values on virtual administrations with average parameters for five human models of omeprazole (407 ± 216 ng h−1 mL−1), metoprolol (939 ± 347 ng h−1 mL−1), and midazolam (37.5 ± 17.4 ng h−1 mL−1) were consistent with 30 reported human averages (± SD values) for omeprazole (368 ± 250 ng h−1 mL−1), metoprolol (449 ± 303 ng h−1 mL−1), and midazolam (27.3 ± 8.0 ng h−1 mL−1). Caffeine and S-warfarin, having intermediate and low hepatic extraction ratio drugs, respectively, also showed good consistency between estimated and reported AUC values in the present system (Table 2).
Table 2.
Mean parameters for simplified human PBPK models for caffeine, warfarin, omeprazole, metoprolol, and midazolam calculated from parameters in humanized mice, marmosets, cynomolgus monkey, dog, and minipig models.
| probe drug | PBPK model | absorbed × intestinal availability, FaFg | absorption rate constant, ka (1/h) | distribution volume, V1 (L) | hepatic intrinsic clearance, CLh,int, in vivo (L/h) | hepatic clearance, CLh (L/h) | AUClast, ng h−1 mL−1 |
|---|---|---|---|---|---|---|---|
| caffeine | human, from humanized mice | 1 | 0.50 | 18.6 | 4.91 | 4.35 | 26200 |
| human, average (± SD) | 1 | 0.97 (± 0.57) | 58.1 (± 29.4) | 7.25 (± 1.71) | 4.83 (± 2.52) | 16700 (± 6000) | |
| literature | 21,300 (± 9,530) | ||||||
| S-warfarin | human, from humanized mice | 1 | 7.51 | 5.90 | 2.54 | 0.259 | 17000 |
| human, average (± SD) | 1 | 2.74 (± 3.03) | 9.7 (± 4.4) | 2.90 (± 2.41) | 0.25 (± 0.26) | 13900 (± 3380) | |
| literature | 16,000 (± 4,340) | ||||||
| omeprazole | human, from humanized mice | 1 | 2.84 | 77.2 | 2020 | 52.2 | 229 |
| human, average (± SD) | 1 | 2.59 (± 1.84) | 37.2 (± 26.9) | 1340 (± 513) | 40.9 (± 9.9) | 407 (± 216) | |
| literature | 368 (± 250) | ||||||
| metoprolol | human, from humanized mice | 1 | 1.65 | 174 | 105 | 46.5 | 1300 |
| human, average (± SD) | 1 | 0.91 (± 0.48) | 200 (± 66) | 178 (± 110) | 55.9 (± 10.6) | 939 (± 347) | |
| literature | 449 (± 303) | ||||||
| midazolam | Human, from humanized mice | 1 | 2.37 | 62 | 475 | 31.7 | 53.5 |
| Human, average (± SD) | 1 | 1.00 (± 0.82) | 71 (± 33) | 827 (± 352) | 41.5 (± 10.4) | 37.5 (± 17.4) | |
| Literature | 23.7 (± 8.0) |
Figure 3.
Results of simplified human PBPK models for caffeine (A), S-warfarin (B), omeprazole (C), metoprolol (D), and midazolam (E) after virtual single oral doses. Solid (
 ) and broken (
) and broken (
 ), (
), (
 ), (
), (
 ), and (
), and (
 ) lines show simplified human PBPK models based on humanized TK-NOG mouse, marmoset, cynomolgus monkey, dog, and minipig PBPK models, respectively. Circles (with SD bars) show reported mean human plasma concentrations after single oral administration of a combination of five probe drugs to 30 Caucasian subjects (2.1 mg/kg for midazolam, 10 mg for S-warfarin, 20 mg for omeprazole, and 100 mg for caffeine and metoprolol).57
) lines show simplified human PBPK models based on humanized TK-NOG mouse, marmoset, cynomolgus monkey, dog, and minipig PBPK models, respectively. Circles (with SD bars) show reported mean human plasma concentrations after single oral administration of a combination of five probe drugs to 30 Caucasian subjects (2.1 mg/kg for midazolam, 10 mg for S-warfarin, 20 mg for omeprazole, and 100 mg for caffeine and metoprolol).57
3. Metabolic Activation of Thalidomide in Humanized Liver Mice
The metabolism of thalidomide is important for both teratogenicity and anti-cancer efficacy. Thalidomide is metabolized via P450-mediated oxidation.34 Various P450s oxidize thalidomide to 5-hydroxy-, 5′-hydroxy-, and dihydroxythalidomide products (Figure 1A), with a major one being P450 2C19.35,36 Recently we reported that human P450 3A4 and 3A5 also oxidize thalidomide to the 5-hydroxy and dihydroxy metabolites.37–39 The second oxidation step in the P450 3A4 pathway generates a reactive intermediate, possibly an arene oxide (as initially suggested by Gordon et al.40 that can be trapped by GSH to give GSH adducts, as confirmed in humanized mouse models using LC-MS/MS methods.38,39 The secondary oxidation of 5-hydoxythalidomide was faster than the primary thalidomide 5-hydoxylation mediated by recombinant human P450 3A4/5.37 Thalidomide and its human metabolite 5-hydroxythalidomide were oxidized by auto-induced human P450 3A enzymes41 to reactive intermediates (with substrate cooperativity42), with reactive sites on the aromatic ring, i.e. epoxides and o-quinones that were trapped as glutathione conjugates.37,39
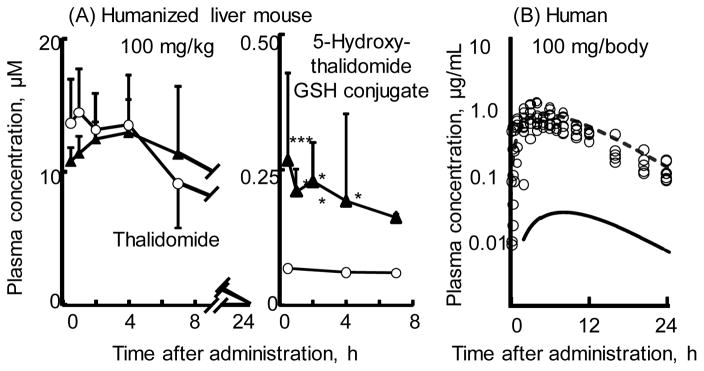
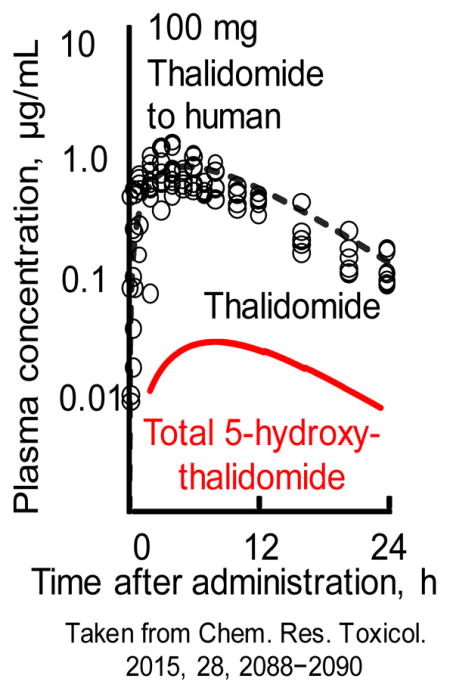
The primary metabolite 5-hydroxythalidomide was found to be extensively oxidized by human P450 enzymes to a dihydroxy metabolite.43 The dihydroxy metabolite is further oxidized to a quinone intermediate that can by trapped with GSH to give a dihydroxythalidomide-GSH conjugate. The observation that quinones are known to undergo redox cycling to generate reactive oxygen species may be consistent a proposed reactive oxygen species hypothesis for toxicity.44 Based on the in vivo experiments in humanized liver mice45 (Figure 4A), results following administration of a low dose of 100 mg thalidomide to human subjects could be reasonably estimated by the current simplified human PBPK model (Figure 4A).14
Figure 4.
Plasma concentrations of thalidomide and 5-hydroxythalidomide-GSH conjugate (A) measured in control TK-NOG mice (open circles) and chimeric TK-NOG mice with humanized liver cells (solid triangles) and thalidomide (open circles, reported by Eriksson et al.58; broken lines) and the sum of 5-hydroxythalidomide metabolites containing 5-hydroxythalidomide-GSH conjugate and 5,6-dihyrdoxythalidomide (solid lines) estimated in humans in silico after oral administration of a single dose of thalidomide (100 mg/kg for mice and 100 mg for humans). Results are expressed as mean values (± SD) obtained with four mice (**p < 0.01, and *p < 0.05, two-way ANOVA with Bonferroni post tests). Results are reproduced from Nishiyama et al.14
4. Metabolic Activation of Hepatoxicant and Non-Specific Binding in Humanized Liver Mice
Drug-induced liver injury is one of the most frequent single causes of safety-related withdrawals of drugs from the market.46 The National Institutes of Health LiverTox database (http://livertox.nih.gov/) is a free online source of textual documents on liver injury caused by prescription and nonprescription drugs, collected from various databases.47 Recently, systematic research into metabolic drug activation has become more comprehensive and more complex.48 Drug-induced toxicity may be caused by active intermediates, formed especially by human cytochrome P450 enzymes. In a clinical study of 5-n-butyl-7-(3,4,5-trimethoxybenzoylamino)pyrazolo[1,5-a]pyrimidine (OT-7100, an amide moiety-bearing pyrazolopyrimidine derivative with potential analgesic effects),49 limited elevations in the serum levels of aspartate or alanine aminotransferase were occasionally observed in humans; these elevations were not predicted from regulatory animal or in vitro studies.50 As an example of species differences in metabolic drug activation, human liver P450 1A2 differed from rat P450 1A2 in bioactivation of the primary metabolite of 5-n-butyl-7-(3,4,5-trimethoxybenzoylamino)pyrazolo[1,5-a]pyrimidine (an amide moiety-bearing pyrazolopyrimidine derivative with potential analgesic effects).50 A primary metabolite was oxidized by human P450 1A2 to form a proximate metabolite, which was conjugated with a peptide to form an adduct (Figure 1B).51 In rats, the same P450 1A2 enzyme formed some proximate metabolites but predominantly mediated the formation of another metabolite that exhibited no toxicity.52 These metabolic differences highlight some of the perils of relying solely on animal testing of drug candidates for metabolite toxicity.
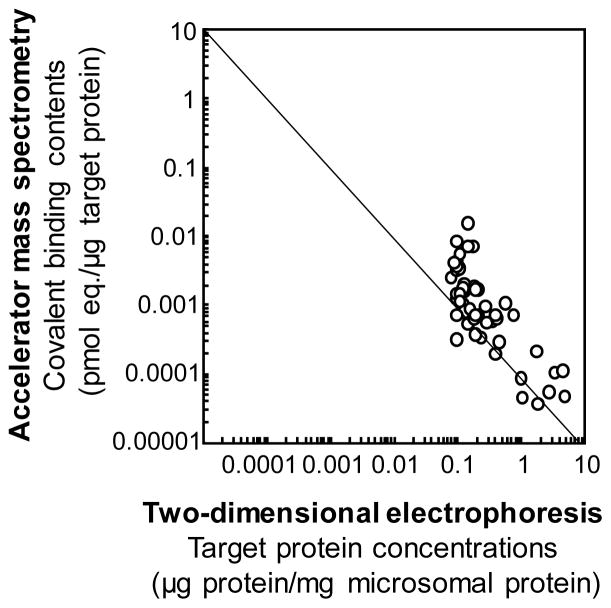
Electrophoretic zone analysis coupled with accelerator MS methods revealed that bioactivated radiolabeled diazepam (rarely hepatotoxic) and 5-n-butyl-pyrazolo[1,5-a]pyrimidine (Figure 5, limited hepatotoxicity) bound nonspecifically in vivo to a variety of microsomal and/or cytosolic proteins present in liver from chimeric mice with humanized liver.53 A line is drawn though convenient axis intersections to indicate an inverse relationship (Figure 5). In contrast, radiolabeled troglitazone and flutamide (both known to be hepatotoxic in humans) showed relatively little covalent binding at concentrations needed for target protein binding.53 These two idiosyncratic hepatotoxic drugs were activated to reactive metabolites and apparently bound to different target proteins. Thus, testing whether protein binding data of new drug candidates are unbalanced with respect to deviation from an inverse relationship like the case in Figure 5 or the presence of data points in the high covalent binding/high protein concentration zone can be an important concept in evaluating hepatotoxic potential.53 To understand the roles of human P450 enzymes in drug metabolism, safety assessment of drug metabolites in engineered mouse models is proposed for more extensive use.
Figure 5.
Covalent binding profiles of liver microsomal protein fractions separated by two-dimensional electrophoresis. Loaded liver protein samples (100 μg) were subjected to isoelectric focusing (pI 3–10) and were then separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (10–225 kDa). In vivo liver protein bindings with metabolically activated 14C-substrates in humanized liver mice were analyzed by accelerator mass spectrometry. The results for 5-n-butyl-pyrazolo[1,5-a]pyrimidine, a new drug candidate OT-7100 metabolite, are taken from Yamazaki et al.51
5. Conclusions and future perspective
Current research collectively suggests that studies of drug metabolism using transplanted human liver cells in humanized mice may be useful in evaluating the drug clearance and disposition, human metabolite formation, and non-specific protein binding tendency of drugs and their metabolites and potential toxicity in humans. Recently, transplantation of three-dimensional-cultured hepatoma-derived cell line HepaRG cells has been reported to yield hepatocyte-like colonies in in vivo mouse bodies, like primary human hepatocytes, suggesting a possible human cell source for steady generation of humanized liver TK-NOG mice.54,55 Humanized mice reconstituted with human immune systems are also essential to study human immune reactions in vivo and are expected to be useful for studying human allergies. A novel transgenic NOG strain bearing human interleukin-3 and granulocyte macrophage colony-stimulating factor genes has been developed.56 Combinations of transplanted human hepatocytes and immune system may become available in the future to study human-type drug metabolite identification and resulting human immune reactions in the combined model of humanized mice. These humanized model mice provide accurate preclinical predictions of human drug metabolism and disposition, coupled with MS methods.
Supplementary Material
Acknowledgments
The authors thank Prof. Norio Shibata and Drs. Koichiro Adachi, Takamori Miyaguchi, Sayako Nishiyama, Satomi Shida, Mirai Kawano, Shotaro Uehara, and Norie Murayama for their experimental support.
Funding Sources. This work was supported in part by the Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research 26460206 (H.Y.), the MEXT (Ministry of Education, Science, Sports and Culture of Japan)-Supported Program for the Strategic Research Foundation at Private Universities, 2013–2018. and United States Public Health Service grant R01 GM118122 (F.P.G.).
Abbreviations
- MS
mass spectrometry
- NOG mice
non-obese diabetes-severe combined immunodeficiency-interleukin-2 receptor gamma chain-deficient mice
Footnotes
Description of the Supporting Information material. Table S1 (Parameters for simplified PBPK models for caffeine, warfarin, omeprazole, metoprolol, and midazolam.). This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Nelson DR, Zeldin DC, Hoffman SMG, Maltais LJ, Wain HM, Nebert DW. Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants. Pharmacogenetics. 2004;14:1–18. doi: 10.1097/00008571-200401000-00001. [DOI] [PubMed] [Google Scholar]
- 2.Guengerich FP. Cytochrome P450 and chemical toxicology. Chem Res Toxicol. 2008;21:70–83. doi: 10.1021/tx700079z. [DOI] [PubMed] [Google Scholar]
- 3.Gonzalez FJ, Fang ZZ, Ma X. Transgenic mice and metabolomics for study of hepatic xenobiotic metabolism and toxicity. Expert Opin Drug Metab Toxicol. 2015;11:869–881. doi: 10.1517/17425255.2015.1032245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Scheer N, Kapelyukh Y, Rode A, Oswald S, Busch D, McLaughlin LA, Lin D, Henderson CJ, Wolf CR. Defining human pathways of drug metabolism in vivo through the development of a multiple humanized mouse model. Drug Metab Dispos. 2015;43:1679–1690. doi: 10.1124/dmd.115.065656. [DOI] [PubMed] [Google Scholar]
- 5.Levova K, Moserova M, Nebert DW, Phillips DH, Frei E, Schmeiser HH, Arlt VM, Stiborova M. NAD(P)H:quinone oxidoreductase expression in Cyp1a-knockout and CYP1A-humanized mouse lines and its effect on bioactivation of the carcinogen aristolochic acid I. Toxicol Appl Pharmacol. 2012;265:360–367. doi: 10.1016/j.taap.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 6.Moriguchi T, Motohashi H, Hosoya T, Nakajima O, Takahashi S, Ohsako S, Aoki Y, Nishimura N, Tohyama C, Fujii-Kuriyama Y, Yamamoto M. Distinct response to dioxin in an arylhydrocarbon receptor (AHR)-humanized mouse. Proc Natl Acad Sci U S A. 2003;100:5652–5657. doi: 10.1073/pnas.1037886100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ma X, Shah Y, Cheung C, Guo GL, Feigenbaum L, Krausz KW, Idle JR, Gonzalez FJ. The PREgnane X receptor gene-humanized mouse: a model for investigating drug-drug interactions mediated by cytochromes P450 3A. Drug Metab Dispos. 2007;3:194–200. doi: 10.1124/dmd.106.012831. [DOI] [PubMed] [Google Scholar]
- 8.Tateno C, Yoshizane Y, Saito N, Kataoka M, Utoh R, Yamasaki C, Tachibana A, Soeno Y, Asahina K, Hino H, Asahara T, Yokoi T, Furukawa T, Yoshizato K. Near completely humanized liver in mice shows human-type metabolic responses to drugs. Am J Pathol. 2004;165:901–912. doi: 10.1016/S0002-9440(10)63352-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Suemizu H, Hasegawa M, Kawai K, Taniguchi K, Monnai M, Wakui M, Suematsu M, Ito M, Peltz G, Nakamura M. Establishment of a humanized model of liver using NOD/Shi-scid IL2Rgnull mice. Biochem Biophys Res Commun. 2008;377:248–252. doi: 10.1016/j.bbrc.2008.09.124. [DOI] [PubMed] [Google Scholar]
- 10.Hasegawa M, Kawai K, Mitsui T, Taniguchi K, Monnai M, Wakui M, Ito M, Suematsu M, Peltz G, Nakamura M, Suemizu H. The reconstituted ‘humanized liver’ in TK-NOG mice is mature and functional. Biochem Biophys Res Commun. 2011;405:405–410. doi: 10.1016/j.bbrc.2011.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Poswillo DE, Hamilton WJ, Sopher D. The marmoset as an animal model for teratological research. Nature. 1972;239:460–462. doi: 10.1038/239460a0. [DOI] [PubMed] [Google Scholar]
- 12.Kazuki Y, Kobayashi K, Aueviriyavit S, Oshima T, Kuroiwa Y, Tsukazaki Y, Senda N, Kawakami H, Ohtsuki S, Abe S, Takiguchi M, Hoshiya H, Kajitani N, Takehara S, Kubo K, Terasaki T, Chiba K, Tomizuka K, Oshimura M. Trans-chromosomic mice containing a human CYP3A cluster for prediction of xenobiotic metabolism in humans. Hum Mol Genet. 2013;22:578–592. doi: 10.1093/hmg/dds468. [DOI] [PubMed] [Google Scholar]
- 13.Kazuki Y, Akita M, Kobayashi K, Osaki M, Satoh D, Abe S, Takehara S, Kazuki K, Yamazaki H, Kamataki T, Oshimura M. Thalidomide-induced limb abnormalities in a humanized CYP3A mouse model. Sci Rep. 2016;6:21419. doi: 10.1038/srep21419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nishiyama S, Suemizu H, Shibata N, Guengerich FP, Yamazaki H. Simulation of human plasma concentrations of thalidomide and primary 5-hydroxylated metabolites explored with pharmacokinetic data in humanized TK-NOG mice. Chem Res Toxicol. 2015;28:2088–2090. doi: 10.1021/acs.chemrestox.5b00381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Poulin P, Haddad S. Hepatocyte composition-based model as a mechanistic tool for predicting the cell suspension: aqueous phase partition coefficient of drugs in in vitro metabolic studies. J Pharm Sci. 2013;102(8):2806–2818. doi: 10.1002/jps.23602. [DOI] [PubMed] [Google Scholar]
- 16.Adachi K, Suemizu H, Murayama N, Shimizu M, Yamazaki H. Human biofluid concentrations of mono(2-ethylhexyl)phthalate extrapolated from pharmacokinetics in chimeric mice with humanized liver administered with di(2-ethylhexyl)phthalate and physiologically based pharmacokinetic modeling. Environ Toxicol Pharmacol. 2015;39:1067–1073. doi: 10.1016/j.etap.2015.02.011. [DOI] [PubMed] [Google Scholar]
- 17.Miyaguchi T, Suemizu H, Shimizu M, Shida S, Nishiyama S, Takano R, Murayama N, Yamazaki H. Human urine and plasma concentrations of bisphenol A extrapolated from pharmacokinetics established in in vivo experiments with chimeric mice with humanized liver and semi-physiological pharmacokinetic modeling. Regul Toxicol Pharmacol. 2015;72:71–76. doi: 10.1016/j.yrtph.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 18.Suemizu H, Sota S, Kuronuma M, Shimizu M, Yamazaki H. Pharmacokinetics and effects on serum cholinesterase activities of organophosphorus pesticides acephate and chlorpyrifos in chimeric mice transplanted with human hepatocytes. Regul Toxicol Pharmacol. 2014;70:468–473. doi: 10.1016/j.yrtph.2014.08.010. [DOI] [PubMed] [Google Scholar]
- 19.Gao H, Jacobs A, White RE, Booth BP, Obach RS. Meeting report: Metabolites in safety testing (MIST) Symposium-Safety assessment of human metabolites: What’s REALLY necessary to ascertain exposure coverage in safety tests? AAPS J. 2013;15:970–973. doi: 10.1208/s12248-013-9502-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kitamura S, Sugihara K. Current status of prediction of drug disposition and toxicity in humans using chimeric mice with humanized liver. Xenobiotica. 2014;44:123–134. doi: 10.3109/00498254.2013.868062. [DOI] [PubMed] [Google Scholar]
- 21.Hirom PC, Millburn P, Smith RL, Williams RT. Species variations in the threshold molecular-weight factor for the biliary excretion of organic anions. Biochem J. 1972;129:1071–1077. doi: 10.1042/bj1291071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Okumura H, Katoh M, Sawada T, Nakajima M, Soeno Y, Yabuuchi H, Ikeda T, Tateno C, Yoshizato K, Yokoi T. Humanization of excretory pathway in chimeric mice with humanized liver. Toxicol Sci. 2007;97:533–538. doi: 10.1093/toxsci/kfm041. [DOI] [PubMed] [Google Scholar]
- 23.Kato M, Shitara Y, Sato H, Yoshisue K, Hirano M, Ikeda T, Sugiyama Y. The quantitative prediction of CYP-mediated drug interaction by physiologically based pharmacokinetic modeling. Pharm Res. 2008;25:1891–1901. doi: 10.1007/s11095-008-9607-2. [DOI] [PubMed] [Google Scholar]
- 24.Gargas ML, Andersen ME, Teo SKO, Batra R, Fennell TR, Kedderis GL. A physiologically based dosimetry description of acrylonitrile and cyanoethylene oxide in the rat. Toxicol Appl Pharmacol. 1995;134:185–194. doi: 10.1006/taap.1995.1183. [DOI] [PubMed] [Google Scholar]
- 25.Kurata Y, Shimamura N, Katoh M. Metabolite profiling and identification in human urine after single oral administration of DEHP. J Toxicol Sci. 2012;37:401–414. doi: 10.2131/jts.37.401. [DOI] [PubMed] [Google Scholar]
- 26.Kurata Y, Makinodan F, Shimamura N, Katoh M. Metabolism of di (2-ethylhexyl) phthalate (DEHP): comparative study in juvenile and fetal marmosets and rats. J Toxicol Sci. 2012;37:33–49. doi: 10.2131/jts.37.33. [DOI] [PubMed] [Google Scholar]
- 27.Silva MJ, Barr DB, Reidy JA, Malek NA, Hodge CC, Caudill SP, Brock JW, Needham LL, Calafat AM. Urinary levels of seven phthalate metabolites in the U.S. population from the National Health and Nutrition Examination Survey (NHANES) 1999–2000. Environ Health Persp. 2004;112:331–338. doi: 10.1289/ehp.6723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Koch HM, Preuss R, Angerer J. Di(2-ethylhexyl)phthalate (DEHP): human metabolism and internal exposure--an update and latest results. Int J Androl. 2006;29:155–165. doi: 10.1111/j.1365-2605.2005.00607.x. [DOI] [PubMed] [Google Scholar]
- 29.Rusyn I, Corton JC. Mechanistic considerations for human relevance of cancer hazard of di(2-ethylhexyl) phthalate. Mutat Res. 2012;750:141–158. doi: 10.1016/j.mrrev.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Krishnan K, Gagne M, Nong A, Aylward LL, Hays SM. Biomonitoring equivalents for bisphenol A (BPA) Regul Toxicol Pharmacol. 2010;58:18–24. doi: 10.1016/j.yrtph.2010.06.005. [DOI] [PubMed] [Google Scholar]
- 31.Shida S, Yamazaki H. Human plasma concentrations of five cytochrome P450 probes extrapolated from pharmacokinetics in dogs and minipigs using physiologically based pharmacokinetic modeling. Xenobiotica. 2016;46:759–764. doi: 10.3109/00498254.2015.1118650. [DOI] [PubMed] [Google Scholar]
- 32.Utoh M, Suemizu H, Mitsui M, Kawao M, Toda A, Uehara S, Uno Y, Shimizu M, Sasaki E, Yamazaki H. Human plasma concentrations of cytochrome P450 probe cocktails extrapolated from pharmacokinetics in mice transplanted with human hepatocytes and from pharmacokinetics in common marmosets using physiologically based pharmacokinetic modeling. Xenobiotica. doi: 10.3109/00498254.2016.1147102. in press. [DOI] [PubMed] [Google Scholar]
- 33.Shida S, Utoh M, Murayama N, Shimizu M, Uno Y, Yamazaki H. Human plasma concentrations of cytochrome P450 probes extrapolated from pharmacokinetics in cynomolgus monkeys using physiologically based pharmacokinetic modeling. Xenobiotica. 2015;45:881–886. doi: 10.3109/00498254.2015.1028511. [DOI] [PubMed] [Google Scholar]
- 34.Nakamura K, Matsuzawa N, Ohmori S, Ando Y, Yamazaki H, Matsunaga T. Clinical evidence of the pharmacokinetics change in thalidomide therapy. Drug Metab Pharmacokinet. 2013;28:38–43. doi: 10.2133/dmpk.dmpk-12-rv-089. [DOI] [PubMed] [Google Scholar]
- 35.Ando Y, Fuse E, Figg WD. Thalidomide metabolism by the CYP2C subfamily. Clin Cancer Res. 2002;8:1964–1973. [PubMed] [Google Scholar]
- 36.Lu J, Helsby N, Palmer BD, Tingle M, Baguley BC, Kestell P, Ching LM. Metabolism of thalidomide in liver microsomes of mice, rabbits, and humans. J Pharmacol Exp Ther. 2004;310:571–577. doi: 10.1124/jpet.104.067793. [DOI] [PubMed] [Google Scholar]
- 37.Chowdhury G, Murayama N, Okada Y, Uno Y, Shimizu M, Shibata N, Guengerich FP, Yamazaki H. Human liver microsomal cytochrome P450 3A enzymes involved in thalidomide 5-hydroxylation and formation of a glutathione conjugate. Chem Res Toxicol. 2010;23:1018–1024. doi: 10.1021/tx900367p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yamazaki H, Suemizu H, Igaya S, Shimizu M, Shibata M, Nakamura M, Chowdhury G, Guengerich FP. In vivo formation of a glutathione conjugate derived from thalidomide in humanized uPA-NOG mice. Chem Res Toxicol. 2011;24:287–289. doi: 10.1021/tx200005g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yamazaki H, Suemizu H, Shimizu M, Igaya S, Shibata N, Nakamura N, Chowdhury G, Guengerich FP. In vivo formation of dihydroxylated and glutathione conjugate metabolites derived from thalidomide and 5-hydroxythalidomide in humanized TK-NOG mice. Chem Res Toxicol. 2012;25:274–276. doi: 10.1021/tx300009j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gordon GB, Spielberg SP, Blake DA, Balasubramanian V. Thalidomide teratogenesis: evidence for a toxic arene oxide metabolite. Proc Natl Acad Sci U S A. 1981;78:2545–2548. doi: 10.1073/pnas.78.4.2545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Murayama N, van Beuningen R, Suemizu H, Guguen-Guillouzo C, Shibata N, Yajima K, Utoh M, Shimizu M, Chesne C, Nakamura M, Guengerich FP, Houtman R, Yamazaki H. Thalidomide increases human hepatic cytochrome P450 3A enzymes by direct activation of pregnane X receptor. Chem Res Toxicol. 2014;27:304–308. doi: 10.1021/tx4004374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yamazaki H, Suemizu H, Murayama N, Utoh M, Shibata N, Nakamura M, Guengerich FP. In vivo drug interactions of the teratogen thalidomide with midazolam: Heterotropic cooperativity of human cytochrome P450 in humanized TK-NOG mice. Chem Res Toxicol. 2013;26:486–489. doi: 10.1021/tx400008g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chowdhury G, Shibata N, Yamazaki H, Guengerich FP. Human cytochrome P450 oxidation of 5-hydroxythalidomide and pomalidomide, an amino analog of thalidomide. Chem Res Toxicol. 2014;27:147–156. doi: 10.1021/tx4004215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Parman T, Wiley MJ, Wells PG. Free radical-mediated oxidative DNA damage in the mechanism of thalidomide teratogenicity. Nat Med. 1999;5:582–585. doi: 10.1038/8466. [DOI] [PubMed] [Google Scholar]
- 45.Nishiyama S, Suemizu H, Shibata N, Guengerich FP, Yamazaki H. Simulation of human plasma concentrations of thalidomide and primary 5-hydroxylated metabolites explored with pharmacokinetic data in humanized TK-NOG mice. Chem Res Toxicol. 2015;28:2088–2090. doi: 10.1021/acs.chemrestox.5b00381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kaplowitz N. Idiosyncratic drug hepatotoxicity. Nat Rev Drug Discov. 2005;4:489–499. doi: 10.1038/nrd1750. [DOI] [PubMed] [Google Scholar]
- 47.Hoofnagle JH, Serrano J, Knoben JE, Navarro VJ. LiverTox: a website on drug-induced liver injury. Hepatology. 2013;57:873–874. doi: 10.1002/hep.26175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yamazaki H. Drug-induced liver toxicity studies: research into human metabolites clarifies their role in drug development. Drug Metab Pharmacokinet. 2014;29:111. doi: 10.2133/dmpk.dmpk-14-pf-902. [DOI] [PubMed] [Google Scholar]
- 49.Kuribayashi S, Ueda N, Naito S, Yamazaki H, Kamataki T. Species differences in hydrolase activities toward OT-7100 responsible for different bioavailability in rats, dogs, monkeys and humans. Xenobiotica. 2006;36:301–314. doi: 10.1080/00498250600571798. [DOI] [PubMed] [Google Scholar]
- 50.Kuribayashi S, Goto K, Naito S, Kamataki T, Yamazaki H. Human cytochrome P450 1A2 involvement in the formation of reactive metabolites from a species-specific hepatotoxic pyrazolopyrimidine derivative, 5-n-butyl-7-(3,4,5-trimethoxybenzoylamino)pyrazolo[1,5-a]pyrimidine. Chem Res Toxicol. 2009;22:323–331. doi: 10.1021/tx8003592. [DOI] [PubMed] [Google Scholar]
- 51.Yamazaki H, Kuribayashi S, Inoue T, Tateno C, Nishikura Y, Oofusa K, Harada D, Naito S, Horie T, Ohta S. Approach for in vivo protein binding of 5-n-butyl-pyrazolo[1,5-a]pyrimidine bioactivated in chimeric mice with humanized liver by two-dimensional electrophoresis with accelerator mass spectrometry. Chem Res Toxicol. 2010;23:152–158. doi: 10.1021/tx900323a. [DOI] [PubMed] [Google Scholar]
- 52.Kuribayashi S, Uno Y, Naito S, Yamazaki H. Different metabolites of human hepatotoxic pyrazolopyrimidine derivative 5-n-butyl-pyrazolo[1,5-a]pyrimidine produced by human, rat, and monkey cytochrome P450 1A2 and liver microsomes. Basic Clin Pharmacol Toxicol. 2012;110:405–408. doi: 10.1111/j.1742-7843.2011.00823.x. [DOI] [PubMed] [Google Scholar]
- 53.Yamazaki H, Kuribayashi S, Inoue T, Honda T, Tateno C, Oofusa K, Ninomiya S, Ikeda T, Izumi T, Horie T. Zone analysis by two-dimensional electrophoresis with accelerator mass spectrometry of in vivo protein bindings of idiosyncratic hepatotoxicants troglitazone and flutamide bioactivated in chimeric mice with humanized liver. Toxicol Res. 2015;4:106–111. [Google Scholar]
- 54.Higuchi Y, Kawai K, Kanai T, Yamazaki H, Chesne C, Guguen-Guillouzo C, Suemizu H. Functional polymer-dependent 3D culture accelerates the differentiation of HepaRG cells into mature hepatocytes. Hepatol Res. doi: 10.1111/hepr.12644. in press. [DOI] [PubMed] [Google Scholar]
- 55.Higuchi Y, Kawai K, Yamazaki H, Nakamura M, Bree F, Guillouzo C, Suemizu H. The human hepatic cell line HepaRG cells, possible cell source for steady generation of humanized liver TK-NOG mice. Xenobiotica. 2014;44:146–153. doi: 10.3109/00498254.2013.836257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ito R, Takahashi T, Katano I, Kawai K, Kamisako T, Ogura T, Ida-Tanaka M, Suemizu H, Nunomura S, Ra C, Mori A, Aiso S, Ito M. Establishment of a human allergy model using human IL-3/GM-CSF-transgenic NOG mice. J Immunol. 2013;191:2890–2899. doi: 10.4049/jimmunol.1203543. [DOI] [PubMed] [Google Scholar]
- 57.Turpault S, Brian W, Van HR, Santoni A, Poitiers F, Donazzolo Y, Boulenc X. Pharmacokinetic assessment of a five-probe cocktail for CYPs 1A2, 2C9, 2C19, 2D6 and 3A. Br J Clin Pharmacol. 2009;68:928–935. doi: 10.1111/j.1365-2125.2009.03548.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Eriksson T, Bjorkman S, Roth B, Bjork H, Hoglund P. Hydroxylated metabolites of thalidomide: formation in-vitro and in-vivo in man. J Pharm Pharmacol. 1998;50:1409–1416. doi: 10.1111/j.2042-7158.1998.tb03368.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.