Abstract
Multiple sclerosis (MS)-induced neuropathic pain deteriorates quality of life in patients but is often refractory to treatment. In experimental autoimmune encephalomyelitis (EAE), a rodent model of MS, animals develop neuropathy and inflammation-induced tissue acidosis, which suggests the involvement of acid-sensing ion channels (ASICs). Also, peripheral neuropathy is reported in MS patients. However, the involvement of the peripheral nervous system (PNS) in MS neuropathic pain remains elusive. This study investigated the contribution of ASICs and peripheral neuropathy in MS-induced neuropathic pain. Elicited pain levels were as high in Asic1a−/−, Asic2−/− and Asic3−/− mice as wild-type mice even though only Asic1a−/− mice showed reduced EAE disease severity, which indicates that pain in EAE was independent of disease severity. We thus adopted an EAE model without pertussis toxin (EAEnp) to restrain activated immunity in the periphery and evaluate the PNS contribution to pain. Both EAE and EAEnp mice showed similar pain behaviors and peripheral neuropathy in nerve fibers and DRG neurons. Moreover, pregabalin significantly reduced neuropathic pain in both EAE and EAEnp mice. Our findings highlight the essential role of the PNS in neuropathic pain in EAE and pave the way for future development of analgesics without side effects in the CNS.
Multiple sclerosis (MS) is a demyelinating autoimmune disease causing motor deficit due to neuronal damage-induced inflammation and demyelination in the central nervous system (CNS)1,2. It is the most frequent debilitating disease in young adults. Among MS-associated symptoms, chronic neuropathic pain severely reduces quality of life. Pain is reported by approximately 50% of MS patients and can be reported by 90%3,4,5. Furthermore, MS-induced chronic pain is significantly associated with MS-related disability and depression4. Therefore, mitigating chronic neuropathic pain in MS patients can improve their quality of life. However, the underling mechanisms of MS-induced chronic pain remain unclear.
Acid-sensing ion channels (ASICs), which are activated in tissue acidosis conditions, participate in pain sensation6. Inflammation in MS is often accompanied by acidosis in the spinal cord, which may lead to activation of ASICs. Lars Fugger and colleagues found ASIC1a involved in disease development in experimental autoimmune encephalomyelitis (EAE)7, a well-established animal model of MS. However, the role of ASICs in MS-induced chronic pain is still unknown.
Pain-like behaviors have been explored in EAE, and they occur before the onset of motor deficit and autoimmune infiltration in the CNS8,9. In addition, these pain-like behaviors in EAE mice, similar to clinical administration, can be ameliorated by anticonvulsant and antidepressant drugs8,9,10,11,12,13. Hence, the EAE model is suitable for exploring the underlying molecular and pathophysiological mechanisms as well as developing more effective analgesic candidates in MS-associated chronic neuropathic pain.
Comprehensive investigation of the cellular and pathological mechanism of chronic pain development has elucidated the importance of peripheral sensory-neuron activity. For instance, studies of fibromyalgia-associated pain in humans and animal models indicate that peripheral administration of lidocaine, blocking local neuronal activity, can relieve mechanical hyperalgesia and allodynia14,15,16,17,18,19,20. Also, damaged peripheral sensory neurons are involved in neuropathic pain associated with diabetes, chemotherapy, post-herpetic neuroglia, radiculopathy, etc.21,22. However, whether peripheral sensory neurons contribute to MS-induced neuropathic pain is largely unknown.
Accumulating evidence has shown that MS-induced pathological and functional damage in the peripheral nervous system (PNS) may be involved in the development of neuropathic pain. First, biopsy specimens of sural nerves from MS patients showed higher frequency of internodes with at least 50% reduced myelin thickness23. Second, in humans and rodents, many myelin proteins, including myelin basic protein (MBP), myelin proteolipid protein, myelin oligodendrocyte glycoprotein (MOG) and galactocerebroside, thought to be exclusively expressed in the CNS, are expressed in the PNS, so autoimmunity may attack the PNS24,25,26,27,28. Third, a case report of an MS patient demonstrated PNS symptoms, including areflexia and specific nerve conductive features of inflammatory demyelinating polyneuropathy (CIDP), dominating the clinical features, without relapsing MS for at least 6 years29. This temporal and clinical association between MS and CIDP suggests that MS-induced neuronal damage in both the CNS and PNS might share the same insults from T-cell activation to target same myelin proteins. This evidence indicates that peripheral sensory-nerve injury may contribute to chronic pain development in MS.
Here we aimed to probe the neurobiological basis of EAE-induced chronic neuropathic pain. We first evaluated the role of ASICs in pain development and whether the PNS damage contributes to clinical and pathological deficits in EAE mice with or without pertussis toxin (PTX). Next, we examined the neuropathic injury of dorsal root ganglia (DRG) in EAE mice by staining cyclic AMP-dependent transcription factor-3 (ATF-3), a well-known neuropathic pain marker, then verified the identity of injured DRG neurons. Last, we aimed to determine whether the anticonvulsant drug pregabalin has an analgesic effect in EAE mice with or without PTX injection.
Results
All ASIC-subtype knockout mice elicit similar levels of EAE-induced pain to wild-type mice regardless of their different contributions to clinical severity
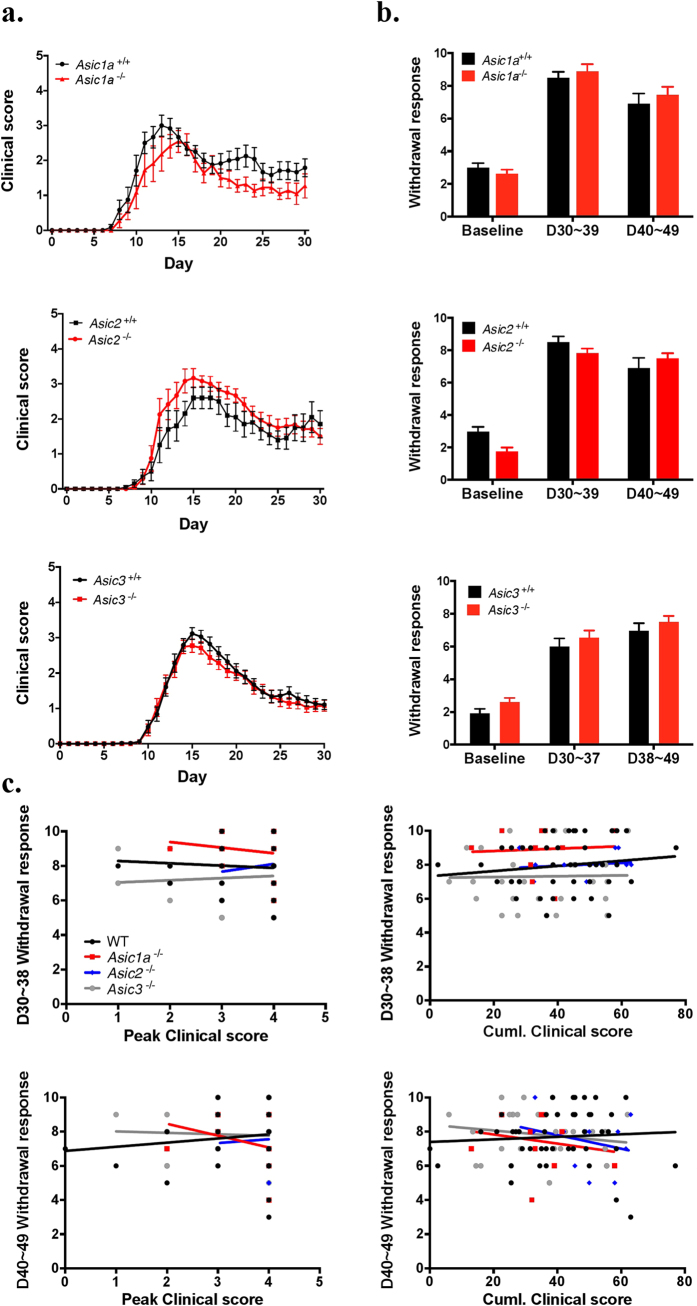
A previous study showed that Asic1a knockout ameliorated EAE-induced clinical deficits in mice, but its effect on pain was not explored7. Since EAE induced acidosis (pH 6.5~6.6) in the spinal cord, we tested whether specific ASIC subtype would affect the EAE-induced neuropathic pain. Here we conducted a comprehensive exam of EAE-induced neuronal disorders and pain in mice lacking Asic1a, Asic2, and Asic3. On analysis of clinical severity in EAE mice, Asic1a−/− mice showed significantly milder disease progression than Asic1a+/+ mice (genotype: F(1,21) = 5.4, P = 0.03). Asic2−/− and Asic3−/− mice showed no difference in disease progression as compared with wild-type littermates, although Asic2−/− mice showed a trend to increased disease severity (F(1,20) = 1.778, P = 0.19) (Fig. 1a). Previous studies demonstrated that ASIC2 expression could modulate the expression of ASIC1a30,31, so we performed western blot analysis to investigate the membrane proteins of DRG and found that loss of ASIC2 did not alter the expression of ASIC1a in DRG (See Supplementary Fig. S1 and Methods).
Figure 1. ASIC-knockout EAE mice showed pain comparable to the wild type despite different contribution to severity.
(a) EAE was induced in Asic1a−/− (N = 11), Asic2−/− (N = 13), Asic3−/− (N = 33) and littermate control Asic1a+/+ (N = 16), Asic2+/+ (N = 10), Asic3+/+ (N = 34) mice. Disease severity was monitored by daily clinical scoring for 30 days. (b) Mechanical sensitivity was assessed by von Frey test before EAE induction as baseline, 30 to 39 days post-induction and 40 to 49 days post-induction. (c) Regression line of peak score and cumulative score verses pain levels at 30 to 39 days and 40 to 49 days post-induction of EAE in wild-type, Asic1a−/−, Asic2−/− and Asic3−/− mice. Statistics were analyzed by two-way ANOVA. Genotype effect: Asic1a−/−: F(1,21) = 5.421, P = 0.03; Asic2−/−: F(1,20) = 1.778, P = 0.1974; Asic3−/−: F(1,65) = 0.4818, P = 0.4901; Time effect: Asic1a−/−: F(30,630) = 30.54, P < 0.001; Asic2−/−: F(30,600) = 42.30, P < 0.001; Asic3−/−: F(30,1905) = 120, P < 0.001. Interaction: no significance.
We next probed the effect of Asic knockout on EAE-evoked neuropathic pain. Since it has been reported that mechanical and thermal hyperalgesia is present in pre-onset of EAE mice9,11, we examined the mechanical and thermal hyperalgesia in different stages after EAE induction, including pre-onset (days 6 to 7), early onset (days 11 to 12), and recovery (days 30–37, 38–49). In both pre-onset and early-onset phases, mechanical and thermal sensitivity did not differ among Asic knockout groups as compared with the wild type (Supplementary Fig. S2). We found a significant interaction effect in mechanical hyperalgesia, and post-hoc analysis showed only Asic1a−/− mice with lower mechanical responses to von Frey stimuli on days 6–7 than Asic3−/− mice. On days 6–7, only Asic3−/− mice showed significant mechanical hyperalgesia as compared with their pre-immune phase; the other groups of mice showed significant mechanical hyperalgesia on days 11–12. The onset of EAE-induced neuropathic pain differed greatly among genotypes and may be complicated by differential disease onset among mice. In the recovery phases, EAE induced mechanical hyperalgesia but not thermal hyperalgesia (Supplementary Fig. S3). All Asic1a−/−, Asic2−/−, and Asic3−/− mice showed similar mechanical sensitivity as wild-type mice after EAE (Fig. 1b). The clinical deficits were not associated with levels of mechanical hyperalgesia in the recovery phase in EAE mice. Therefore, EAE disease severity may not be associated with pain.
To further demonstrate whether these two aspects are correlated with each other in EAE, we further analyzed the correlation between EAE-induced mechanical hyperalgesia and two parameters of clinical deficit: peak score and cumulative score (see Methods) and found no correlation with each other (Fig. 1c, Table 1). Therefore, EAE disease severity is independent of pain levels, which suggests EAE-induced motor deficits (clinical scores) and neuropathy pain develop by 2 independent mechanisms.
Table 1. Spearman correlation analysis of pain levels verses peak scores or cumulative scores at 30–39 and 40–49 days post-induction in wild-type, Asic1a −/−, Asic2 −/− and Asic3 −/− EAE mice.
| D30-39 |
D40-49 |
|||||||
|---|---|---|---|---|---|---|---|---|
| Wildtype | Asic1a−/− | Asic2−/− | Asic3−/− | Wildtype | Asic1a−/− | Asic2−/− | Asic3−/− | |
| Max. score | ||||||||
| Spearman r | −0.08778 | −0.02700 | 0.2722 | 0.1248 | 0.1077 | −0.3021 | 0.08944 | −0.06561 |
| 95% CI | −0.3826 to 0.2233 | −0.6294 to 0.5957 | −0.3743 to 0.7406 | −0.2870 to 0.4977 | −0.1970 to 0.3934 | −0.7720 to 0.3813 | −0.5248 to 0.6424 | −0.4313 to 0.3186 |
| p value | 0.5710 | 0.6190 | 0.6636 | 0.5436 | 0.4764 | 0.1498 | 0.3727 | 0.7353 |
| Significance | ns | ns | ns | ns | ns | ns | ns | ns |
| Cumulative score | ||||||||
| Spearman r | 0.1566 | 0.1541 | 0.1539 | 0.003135 | 0.1892 | −0.1125 | −0.1986 | −0.2094 |
| 95% CI | −0.1559 to 0.4407 | −0.5066 to 0.7008 | −0.4758 to 0.6793 | −0.3949 to 0.4002 | −0.1157 to 0.4615 | −0.6785 to 0.5374 | −0.7033 to 0.4393 | −0.5430 to 0.1811 |
| p value | 0.3099 | 0.6529 | 0.6494 | 0.9879 | 0.2080 | 0.6680 | 0.4043 | 0.2755 |
| Significance | ns | ns | ns | ns | ns | ns | ns | ns |
EAEnp mice show increased mechanical hypersensitivity with almost normal mobility
Although most of the MS studies of EAE models focused on the CNS, whether the CNS or PNS contributes to EAE-induced neuropathic pain remains unclear. To investigate the mechanisms underlying EAE-induced neuropathic pain, we used an approach to separate the CNS and PNS effect. We used a modified EAE immunization model without PTX (named EAEnp), because PTX in EAE facilitates activated leukocyte infiltration into the CNS by increasing the permeability of the blood–brain barrier. In the EAEnp model, MOG-activated T cells are restrained in the periphery, so we could specifically explore the PNS effect.
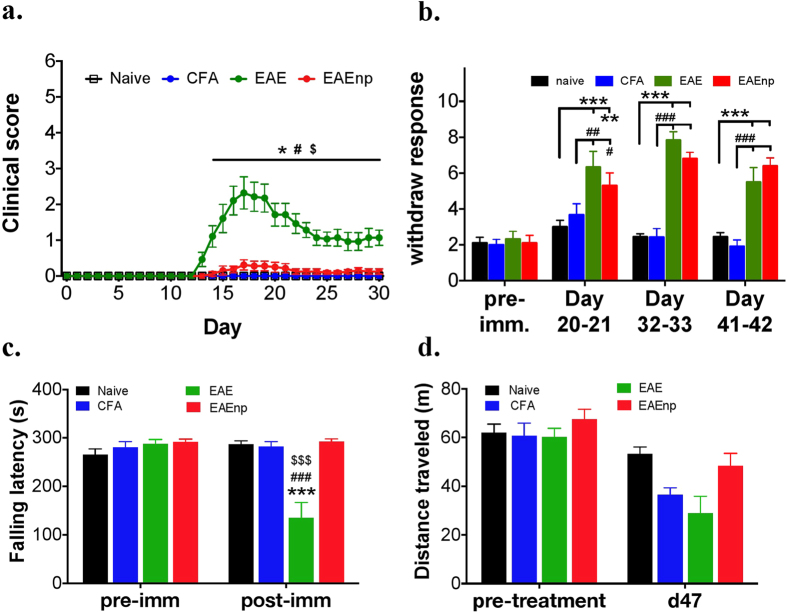
We assessed the disease course in 4 experimental groups: naïve, CFA, EAE and EAEnp mice (Fig. 2a). EAE mice showed clinical deficits as previously reported, whereas EAEnp mice showed very mild clinical deficits. Mice injected with CFA showed no disease onset like naïve mice. During chronic pain assessment, EAE mice elicited hyperalgesia as was reported by previous studies, whereas EAEnp mice exhibited similar mechanical hypersensitivity as EAE mice at 20, 32–33 and 41–42 days after induction (Fig. 2b). Furthermore, EAE and EAEnp mice elicited similar mechanical hypersensitivity in the early-onset phase, whereas only EAE mice (but not EAEnp mice) showed decreased thermal response in the early-onset phase (Supplementary Fig. S4). In open-field and rota-rod tests, only EAE mice showed decreased locomotor activity in the rota-rod test, whereas mice in both CFA and EAEnp groups showed locomotor activity comparable to naïve mice (Fig. 2c,d). Thus, EAEnp mice showed elevated mechanical sensitivity without motor deficit, which suggests that the CNS damage may not be the direct cause of the neuropathic pain in EAE models.
Figure 2. EAEnp mice developed pain without clinical deficits.
(a) Clinical scores of mice in naive (N = 7), CFA (N = 7), EAE (N = 14), EAEnp (N = 16) groups at different days after immunization. Statistics were analyzed by two-way ANOVA (Interaction: F(90,1200) = 10.08; Time: F(30,1200) = 10.64; Treatment: F(3,40) = 20.44). Post-Bonferroni analysis presented in the figure, *p < 0.05 vs. naive group; #p < 0.05 vs. CFA group; $p < 0.01 vs. EAEnp group. (b) von Frey test conducted at selected time points showing hyperalgesia in both EAE and EAEnp mice. Naive: N = 9, CFA: N = 12, EAE: N = 6, EAEnp: N = 10. Statistics were analyzed by two-way ANOVA (Interaction: F(9,99) = 7.769, p < 0.001; Treatment: F(3,33) = 30.03, p < 0.001; Time: F(3,99) = 29.18, p < 0.001). Post-Tukey analysis presented in the figure, **p < 0.01, ***p < 0.001 vs. naive group; ##p < 0.01, ###p < 0.001 vs. CFA group. (c) Rota-rod test shows significantly shorter falling latency in EAE mice than other groups at 47 days post-immunization. Naive: N = 12, CFA: N = 11, EAE: N = 9, EAEnp: N = 11. Statistics were analyzed by two-way ANOVA (Interaction: F(3,39) = 26.3; Time: F(1,39) = 18.1; Treatment: F(3,39) = 11.5). Post-Bonferroni analysis presented in the figure, ***p < 0.001 vs. naive group; ###p < 0.001 vs. CFA group; $$$p < 0.001 vs. EAEnp group. (d) Open field test shows no difference in traveling distance between treatment groups.
L4 spinal cord of EAEnp mice at disease peak shows no immune cells infiltrating and demyelination with very mild microglia activated in gray matter
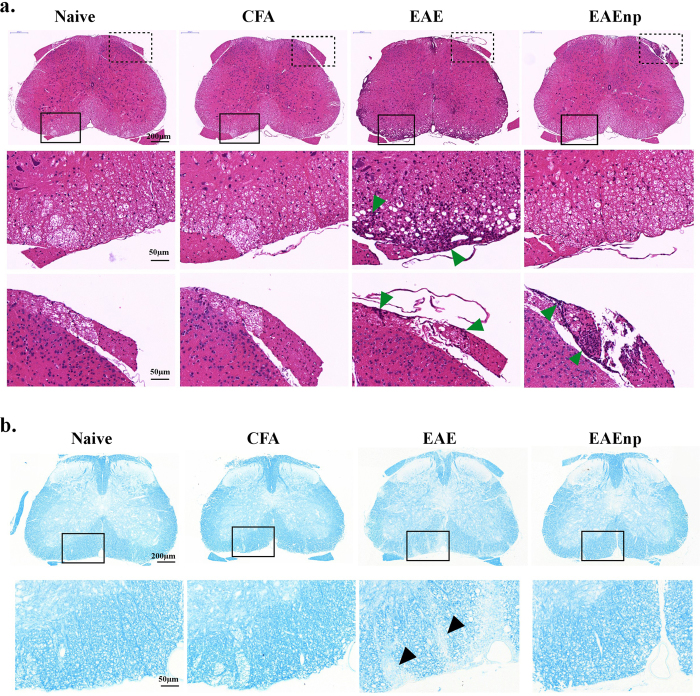
We next examined the pathological aspect of the CNS in the 4 groups of mice, focusing on infiltration of immune cells in the L4 spinal cord. On H&E staining, the L4 spinal cord of naïve and CFA mice showed little or no immune cell infiltration, whereas the EAE mouse spinal cord showed massive immune cell infiltration especially in the ventral horn (Fig. 3a). The spinal cords of EAEnp mice showed little immune cell infiltration similar to naïve or CFA mice. EAEnp mice showed accumulation of immune cells in the dorsal root, the sensory input from PNS, as did EAE mice (Fig. 3a).
Figure 3. Immune-cell infiltration and demyelination occurred in EAE but not EAEnp mice.
Lumbar spinal cord (L4) of each treatment was collected at the peak of disease (d15) and underwent staining. (a) H&E staining of L4 spinal cord. Magnified images of square areas in solid, the ventral horn, and dashed line, the dorsal root, are shown in the second and third rows, respectively.  , sites of immune-cell infiltration. (b) Luxol fast blue staining of L4 spinal cord. Magnified images of dashed line square are shown in the second row. ▴, sites of demyelination. Full-sized images and magnified images are in the same scale, respectively.
, sites of immune-cell infiltration. (b) Luxol fast blue staining of L4 spinal cord. Magnified images of dashed line square are shown in the second row. ▴, sites of demyelination. Full-sized images and magnified images are in the same scale, respectively.
Demyelination is another feature characteristic of EAE model. Here, Luxol fast blue staining in spinal cords of naïve and CFA mice demonstrated negligible demyelination, with EAE mice showing obvious demyelination (Fig. 3b). Notably, EAEnp mice showed no signs of demyelination.
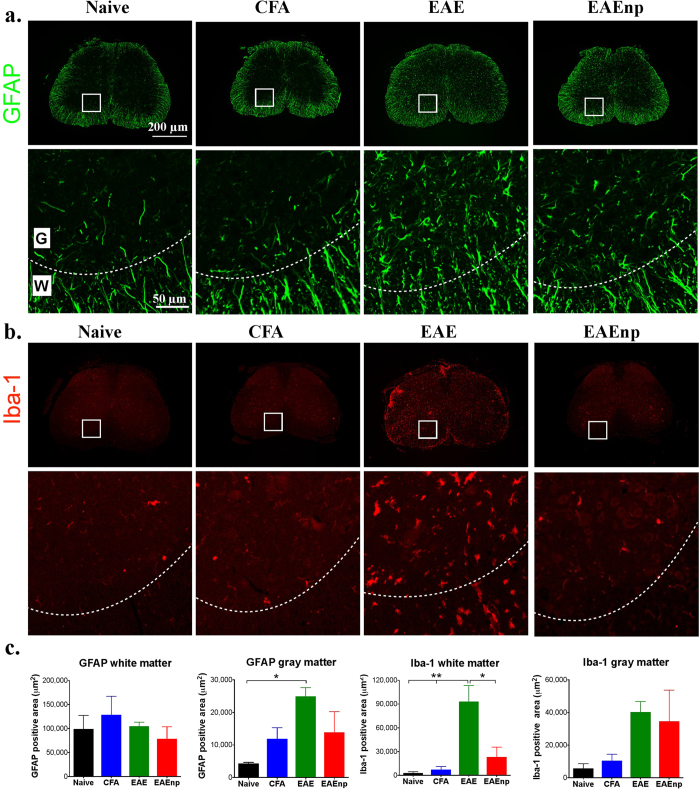
We further characterized the infiltrated cells by immunohistochemistry. Staining of the astrocyte marker GFAP in the L4 spinal cord revealed increased astrocyte number in EAE mice in gray matter as compared with other groups (Fig. 4a). Although astrocyte number was slightly increased in EAEnp gray matter, the result was not significantly different from other groups. Microglia was monitored by Iba-1 staining, and elevated microglia number was apparent in the L4 spinal cord in EAE mice (Fig. 4b). Quantitative analysis indicated significantly greater GFAP expression in L4 spinal-cord gray matter in EAE than naïve mice (Fig. 4c). Iba-1 expression was significantly elevated in EAE mouse L4 spinal-cord white matter as compared with other groups. Therefore, the spinal cord, representing the CNS, remained mostly undisturbed in EAEnp mice.
Figure 4. Astrocytes and microglia activation in the spinal cord observed in EAE mice.
Lumbar spinal cord (L4) for each treatment was collected at the peak of disease (d15) and underwent immunofluorescence staining. Magnified images of squared area are shown in the second row of each staining. Dashed curves indicate the border between white matter and gray matter. (a) GFAP staining for astrocytes in each treatment. (b) Iba-1 staining of microglia in each treatment. (c) Quantification of GFAP and Iba-1 staining in white matter and gray matter. Statistics were analyzed by one-way ANOVA (GFAP white matter: F(3,10) = 0.6455, p = 0.6033; GFAP gray matter: F(3,10) = 3.998, p = 0.0414, Naive vs. EAE: p = 0.0427; Iba-1 white matter: F(3,10) = 9.327, p = 0.0030. Naive vs. EAE: p = 0.0055, CFA vs. EAE: p = 0.0075, EAE vs. EAEnp: p = 0.0167; Iba-1 gray matter: F(3,10) = 2.023, p = 0.1747). *p < 0.05, **p < 0.01. Full-sized images and magnified images are in the same scale, respectively. W: white matter; G: gray matter.
Peripheral neuropathy after EAE or EAEnp induction
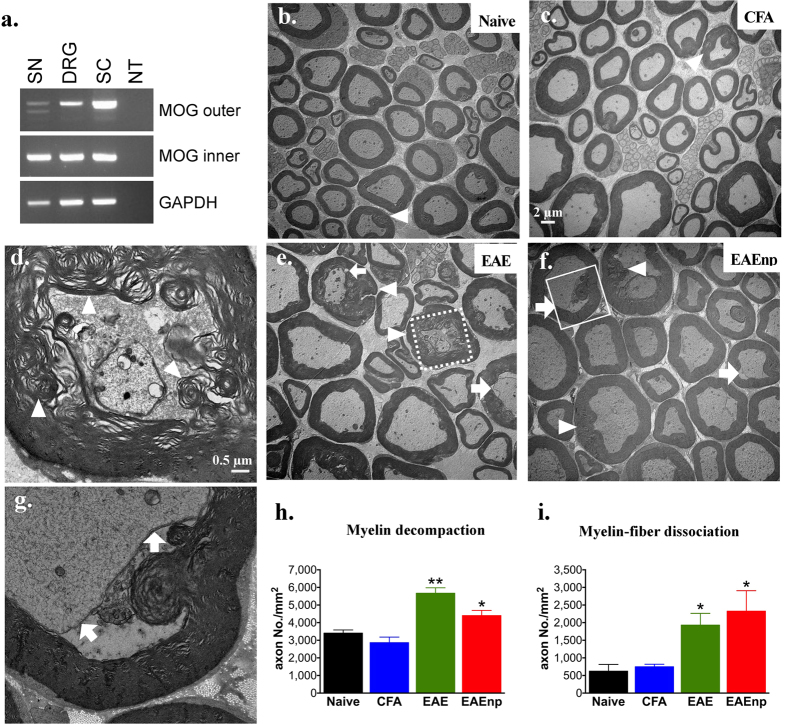
We next examined the possible contribution of the PNS to the EAE-induced neuropathic pain. MOG35-55 peptide, belonging to the CNS, has been widely used for EAE induction in mice; however, its expression in the PNS has not been confirmed. Here we showed MOG transcripts (493–667 bp) in mouse DRG and sciatic nerve (a representative peripheral nerve) by nested RT-PCR (Fig. 5a). To determine whether the expression of MOG in DRG could cause demyelination of peripheral nerves during EAE, we used electron microscopy to analyze the sciatic nerves from naïve, CFA, EAE and EAEnp mice. The myelin sheath of naïve and CFA mice was characterized by a regular and well-organized myelin sheath (Fig. 5b,c). However, the sciatic nerves of EAE and EAEnp mice showed a relatively decompacted myelin sheath, characterized by a dismantled myelin sheath (Fig. 5d,e,f). Also, many axons of EAE and EAEnp mice showed myelin-fiber dissociation, with the membrane of neurons dissociated from the myelin sheath (Fig. 5g). The frequency of decompacted myelin sheath or myelin-fiber dissociation was higher in axons of EAE and EAEnp than CFA and naïve mice (Fig. 5h,i). These results illustrate MOG expression in the PNS and myelin disruption in the PNS after EAE or EAEnp induction and suggest that the neuropathic pain induced in EAE results from peripheral neuropathy derived from demyelination.
Figure 5. Myelin disruption shown in peripheral nerves in EAE and EAEnp mice.
(a) Myelin oligodendrocyte glycoprotein (MOG) transcripts were detected by nested RT-PCR in sciatic nerve (SN), dorsal root ganglion (DRG), and spinal cord (SC) of naive wild-type mice. NT: negative control. (b,c,e,f) Sciatic nerves of each treatment were dissected at peak of disease (d15) for electron-microscopy image. (d) A representative image of an axon showing decompacted myelin in EAE, square area of dashed line in (e). (g) A representative image of an axon showing myelin-fiber dissociation in EAEnp, square area of solid line in (f). (h) Quantification of decompacted myelin. Statistics were analyzed by unpaired t test (EAE: p = 0.0022, EAEnp: p = 0.0437). (i) Quantification of myelin–fiber dissociation. Statistics were analyzed by unpaired t test (EAE: p = 0.0272, EAEnp: p = 0.0497). ⇧, axons with myelin–fiber dissociation. Δ, axons with decompacted myelin. *p < 0.05 vs. naive group. Naive N = 3, CFA N = 3, EAE N = 4, EAEnp N = 3. Scale in (c,e,f) is the same as (b). (d,g) are in the same magnification.
Injured DRG neurons are identified in EAE and EAEnp mice and partially characterized as myelinated or non-peptidergic neurons
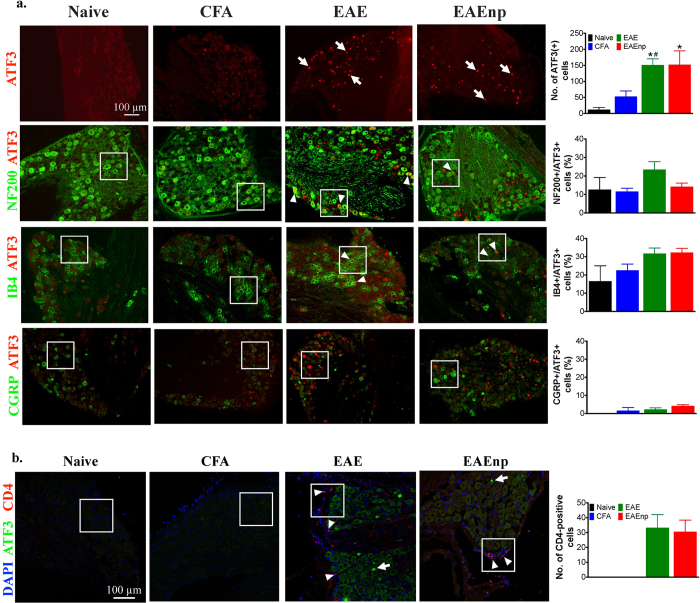
To better understand whether the PNS is involved in EAE-induced neuropathic pain, we examined the expression of activating transcription factor (ATF3), a commonly used neuron injury marker and related to neuropathic pain20,32, in the fourth lumbar DRG (L4 DRG) of naïve, CFA, EAE and EAEnp mice. ATF3-positive neurons were observed in L4 DRG neurons from both EAE and EAEnp mice and were absent in naïve and CFA mice (Fig. 6, Supplementary Fig. S5). Next, we further characterized the ATF3-positive DRG neurons (Fig. 6a; See Supplementary Fig. S6 for large magnification). DRG neurons can be approximately characterized as large-diameter, myelinated neurons that express NF-200 and small-diameter, non-myelinated neurons, which mainly encode nociceptive signals and can be divided into isolectin B4 (IB4)-positive non-peptidergic and IB4-negative peptidergic neurons33. The peptidergic population expresses neuropeptides such as substance P and calcitonin gene-related peptide (CGRP). About 20% of ATF3-positive DRG neurons were NF-200–positive and about 30% of ATF3-positive DRG neurons were IB4–positive in EAE mice. In EAEnp mice, NF-200 and IB4–positive cells accounted for 10% and 30% of ATF3-positive DRG neurons respectively. However, less than 5% of the ATF3-positive neurons were CGRP-positive in EAE and EAEnp mouse L4 DRG. CD4-positive T lymphocytes were present in L4 DRG of EAE and EAEnp mice but not DRG of naïve and CFA mice (Fig. 6b). The cell type of CD4-positive T lymphocytes was mainly Th1 cells, which express interferon γ but not interleukin 17 (Supplementary Fig S7 and Methods). Our results elucidate the EAE-induced neuronal injury, which might be associated with CD4-positive cell infiltration, in DRG neurons, containing myelinated neurons and non-peptidergic neurons.
Figure 6. Characterization of ATF3-positive neurons and infiltrating T-helper cells in EAE and EAEnp mice.
DRG at the 4th lumbar segment was isolated from mice of each treatment at disease peak (d15). (a) DRG sections were stained with ATF3 to assess nerve injury. Characterization of injured neurons was assessed by co-staining ATF3 with different neuron markers. NF-200, neurofilament-200 (a marker of myelinated neurons); IB4, isolectin-B4 (a marker of non-peptidergic neurons); CGRP, calcitonin gene-related peptide (a marker of peptidergic neurons). ⇑, ATF3-positive neurons. Δ, co-localization of ATF3 with each neuron marker. Quantitative analysis of ATF-positive neurons and their subtypes in different treatment groups are shown in the right column. Statistics were analyzed by one-way ANOVA, *p < 0.05 vs. naïve group. #p < 0.05 vs. CFA group. All images are in the same scale as indicated in ATF3 staining of Naïve group. b. DRG sections were stained with ATF3 and CD4 to assess the anatomical distribution of injured neurons and infiltrating T-helper cells. ⇑, ATF3-positive neurons. Δ, CD4-positive cells. Quantitative analysis of CD4-positive cells is indicated in the right column. Magnified images of square area are shown in Supplementary Fig. S6.
EAE- and EAEnp-induced mechanical hyperalgesia was ameliorated by pregabalin
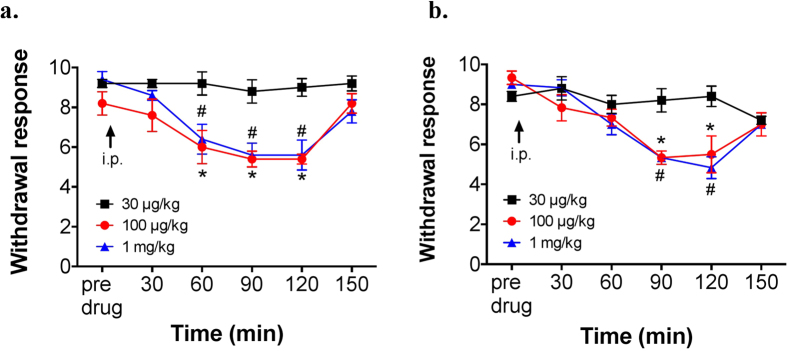
Gabapentinoids (e.g., gabapentin, pregabalin) have been recommended and found effective in ameliorating pain associated with MS in humans and rodents34,35. Hence, we tested whether pregabalin has an analgesic effect in EAE and EAEnp mice. Pregabalin showed similar dose-dependent analgesic effects in both EAE and EAEnp mice, with effective concentrations of 100 μg/kg and 1 mg/kg (i.p.) (Fig. 7). Hence, both EAE and EAEnp models may induce neuropathic pain via a shared pathway, the peripheral neuropathy.
Figure 7. Analgesic effects of pregabalin in EAE and EAEnp mice.
Mechanical sensitivity to a 0.02-g von Frey filament was evaluated in mice at 41 to 50 days post-immunization. (a) Dose-dependent effect of pregabalin in EAE mice (30 μg/kg N = 5, 100 μg/kg N = 5, 1 mg/kg N = 5, i.p.). Statistics were analyzed by two-way ANOVA (EAE: Interaction: F(10,60) = 3.264; Time: F(5,60) = 14.77; Drug dose: F(2,12) = 12.35). (b) Dose-dependent effect of pregabalin in EAEnp mice (30 μg/kg N = 5, 100 μg/kg N = 6, 1 mg/kg N = 6, i.p.). Statistics were analyzed by two-way ANOVA (EAEnp:Interaction: F(10,70) = 4.121; Time: F(5,70) = 17.53; Drug dose: F(2,14) = 5.176). Post-Bonferroni analysis presented in the figure, *P < 0.05, 30 μg/kg vs. 100 μg/kg; #P < 0.05, 30 μg/kg vs. 1 mg/kg.
Discussion
In this study, we showed that ASIC1a is involved in EAE-induced motor deficits, but the channel was not associated with the development of neuropathic pain. Using a modified EAE model without PTX, EAEnp, we demonstrated that EAEnp mice elicit neuropathic pain without immune cells infiltrating in CNS, which suggests the contribution of the PNS in EAE-induced neuropathic pain. The neuropathic pain in EAE mice may be partly attributable to myelinated peripheral nerve injury as well as IB4-positive non-peptidergic nociceptors. Together, the current work highlights a specific type of peripheral neuropathy contributing to the development of MS-associated chronic pain and thus provides a new direction to treat the intractable pain.
ASIC1a is known to play an important role for sensing acidosis during ischemic stroke, seizure, fear, panic, and MS, as well as synaptic transmission7,36,37,38,39,40. A previous study also showed that blockade of ASIC1a activity would result in activation of the endogenous enkephalin pathway in the CNS and thus produce analgesia for neuropathic pain41. We were surprised to find normal development of EAE-induced chronic neuropathic pain in Asic1a−/− mice, although they showed milder EAE clinical scoring than wild-type mice. This result excludes a role for ASIC1a in modulating EAE-induced pain and also raises a question of the CNS contribution to the pain development. Although the MS-associated pain is complex and we cannot totally exclude the contribution of the CNS, probing a possible role for the PNS in EAE-induced neuropathic pain is of interest.
Despite numerous studies of MS pain, evidence regarding the PNS contribution is scarce because of the limited diagnostic methods. Pollock and colleagues reported that many MS patients have abnormal sensory nerves, with internodes showing at least 50% reduction in myelin thickness23. In 2011, Gartzen et al. used electrophysiological diagnosis to investigate the PNS effect on MS pain and found that about 30% of MS patients showed lower conduction velocity in peripheral myelinated nerve fibers, despite no abnormalities in pain-related evoked potentials42. Ayromlou et al. further analyzed nerve electrophysiological properties of motor and sensory fibers in MS patients and found subtle sensory fiber abnormality43. Although diagnostic methods are insufficient to clearly and entirely investigate the peripheral sensory fiber function, these studies indicate that peripheral sensory neurons might be injured in MS patients and contribute to sensory illness.
However, previous studies using EAE models to investigate the neuropathic pain mechanism in MS revealed the involvement of both neurological and immunological factors in the PNS, including satellite glia-cell activation44 and elevated levels of tumor necrosis factor α45 and brain-derived neurotrophic factor46, in DRG of EAE animals. Moreover, our EAEnp mouse model is supported by evidence of mild symptoms accompanied by hyperalgesia in the rat EAE model, which also does not contain PTX13,47. To our knowledge, we are the first to use the EAEnp mouse model to investigate pain mechanisms in EAE and show that it is feasible in behavioral or pathological experiments.
We found immune cells infiltrating the dorsal roots of both EAE and EAEnp mice (Fig. 3a), which suggests that activated immune cells might infiltrate the spinal cord through dorsal roots. This observation might explain why approximately 25% of EAEnp mice showed mild disease signs such as tail weakness, although immune cell infiltration to the spinal cord was rarely found in EAEnp mice. Nevertheless, 75% of our EAEnp mice showed mechanical hypersensitivity without any clinical deficit and motor function interference. Hence, the EAEnp model is suitable and accurate for assessing neuropathic pain responses in EAE mice regardless of motor impairment during disease.
Spinal cord immune-cell infiltration and demyelination is characteristic of the EAE model but is absent in EAEnp mice. Thus, peripheral nerve insult by MOG-triggered autoimmunity may play an important role in EAE-induced neuropathic pain. Although MOG is predominantly expressed in the CNS, evidence of MOG mRNA expression in the PNS has been shown in primates and rats48 and in mice in our study. In the EAEnp mice, MOG-specific T cells might be trapped in the PNS and related to ATF3 activation in DRG neurons. A recent study also showed that PTX injection alone could induce ATF3 expression in DRG neurons in mice, but the ATF3 expression was much lower in PTX-only group than in EAE mice49. Comparing a previous study49 with our results of EAEnp mice, peripheral nerve injury in EAE might only partially result from PTX injection. Of note, previous studies used a rat EAE model, which did not include PTX injection, to study pain13,47. In addition to rats, human studies have also illustrated peripheral nerve injury in MS patients23. Together with data from the EAEnp model, we demonstrate that PNS is important for EAE neuropathic pain.
DRG is a highly heterogeneous tissue, composed of different types of sensory neuron populations. Each population may refer to a specific sensory pathway. Thus, characterization of the injured neurons in EAE may further reveal the possible factors involved in the neuropathic pain development. Here we identified that EAE-induced injured neurons were mainly NF-200–positive myelinated neurons (~20%) and IB4–positive non-peptidergic neurons (~30%), whereas CGRP-positive peptidergic neurons were almost never injured. NF-200–positive neurons are myelinated A fibers, including A-δ fibers that transmit acute pain and innervate in the lamina I, II outer and V parts of the spinal cord dorsal horn33,50. Previous studies showed that A-fiber demyelination would induce chronic neuropathic pain. Demyelination-induced loss of conduction in A fibers would result in plastic changes of C-polymodal nociceptors, which would generate spontaneous activity and hypersensitivity to innocuous stimuli51. IB4-positive neurons encode unmyelinated non-peptidergic C fibers that innervate into the lamina II inner layer and transmit neuropathic pain due to neuron injury33,52,53,54. In many neuropathic pain models, increased ATF3 expression is often coupled with upregulation of P2X3 in IB4-positive neurons55,56. However, CGRP-positive neurons are unmyelinated peptidgeric C fibers that innervate into the lamina I and II outer layers of the spinal cord dorsal horn and are involved in inflammatory pain33,53,54,57. Although NF-200−, IB4−, and CGRP-positive neurons represent almost >95% of DRG neurons54,58,59,60, more than 40% of ATF3-positive DRG neurons are not in these populations. Further studies should focus on the identity of these ATF-positive DRG neurons, to determine a full picture of EAE-induced peripheral neuropathy. Candidate markers for the non-NF-200, non-IB4, and non-CGRP DRG neuron populations may include galanin, neuropeptide Y, nitric oxide synthase, and lysophosphatidic acid receptor33,61,62.
Pharmacological studies of MS neuropathic pain treatment have revealed a limited effect of current analgesics such as morphine63. Anti-epileptic drugs such as gabapentin and pregabalin are some of the common drugs prescribed for MS pain in clinical practice35. Clinical research has indicated the effects of gabapentin and pregabalin in relieving pain in MS patients. Thus, we assessed the analgesic effect of pregabalin in EAE and EAEnp mice and found that it could alleviate pain in both models at a relatively high dose. Pregabalin is known to relieve neuropathic pain by reducing calcium influx and neurotransmitter release in the spinal cord by inhibiting the α2δ-1 subunit of T-type calcium channel trafficking to sensory nerve terminals64,65. Together with our results for EAE and EAEnp mice, pregabalin eliciting anti-nociception through the periphery can further support our hypothesis that PNS is involved in EAE neuropathic pain. Thus, we demonstrate the predicted validity of using the EAEnp model to explore the EAE-induced neuropathic pain and highlight the involvement of peripheral neuropathy in the development of MS-related pain.
Methods
Mice
Asic1a knockout (Asic1a−/−) mice were a kind gift from Dr. Chen-Chang Lien of National Yang-Ming University66. Asic2 knockout (Asic2−/−) mice were purchased from the Jackson Lab. Asic3 knockout (Asic3−/−) mice were generated as previously described67. All mutant mouse lines were congenic after backcrossing to C57BL6/J for at least 10 generations. Wild type and knockout mice were littermates from heterozygote breeding. All experiments involved 8- to 12-week-old female mice housed in a group (3–5) at a temperature-controlled environment with a 12-hr light/dark cycle from 8:00 to 20:00. All procedures followed the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No. 85-12, revised 1996) and approved by the Institutional Animal Care and Utilization Committee of Academia Sinica. We aimed to minimize the use of animals used and their suffering without compromising the quality of the experiments.
EAE induction
Mice were subcutaneously injected in the hind limb with emulsified equal volume of MOG35-55 peptide (100 μg/mouse. MDBio, Inc., Taipei) in PBS and complete Freund’s adjuvant (CFA, 400 μg of heat-killed Mycobacterium tuberculosis H37RA/mouse, Sigma-Aldrich Co., St. Louis, MO), then intraperitoneally injected with pertussis toxin (PTX, 200 ng/mouse, List Biological Laboratories, Campbell, CA) on the day of immunization and 2 days later. CFA control mice received emulsion without MOG35-55 and PTX. As for EAEnp mice, they received the same MOG-CFA emulsion without PTX.
EAE scoring criteria, analysis parameters and disease phases
The clinical scoring index following a previous report68 was assigned daily for 30 days and at the day of behavioral and pathological examination: 0: no clinical signs, 1: paralyzed tail, 2: ataxia of lower body, 3: paraparesis of one or both hind limbs, 4: paraplegia of one or both hind limbs, 5: paraplegia of one or both hind limbs with incomplete paralyzed forelimb, 6: death. Two parameters were used for correlation analysis: peak score, the maximum score within 30 days after induction; accumulated score, summation of all the score in 30 days. For further behavioral experiments, we defined three phases of the EAE disease: pre-onset (day 6 to 7), early onset (day 11 to 12) and recovery (after day 30).
Mechanical response test
A 0.02-g von Frey filament was applied to each hind paw for 5 times and total withdrawal response was recorded. A positive mechanical response was defined as paw withdrawal, licking or flipping in response to von Frey stimulation. Mechanical response assessment was conducted within 3 days before induction and on selected time points with exclusion of EAE mice with score >3.
Rota-rod test
The rota-rod test was conducted before EAE induction and on 48 days post-immunization. A rota-rod machine with automatic timers and falling sensors (MK-660D, Muromachi KiKai, Tokyo) were used. In the training session, a constant speed of 4 rpm with 60-sec cut-off time for 3 trials was used. In the test session, the rod accelerated from 4 to 40 rpm with 300-sec cut-off time was used for 3 trials. The longest falling latency was selected to represent the motor function of each mouse.
Open field test
A transparent square chamber made of plexiglass with area 48 × 48 cm and height 30 cm was used. After 1 hr of habituation in the home cage, mice were allowed to explore the whole test chamber for 20 min and were videotaped for analysis. Top scan software was utilized to analyze the video (Clever System, Reston, VA). The total distance each mouse traveled was analyzed.
RT-PCR
Mouse spinal cord, sciatic nerve and DRG were freshly collected and stored at −80 °C for further RNA extraction. RNA extraction was performed by using TRIzol reagent (Thermo Fisher Scientific, Waltham, MA). cDNA was synthesized by using non-specific primer Oligo(dT). MOG transcripts were examined by using the following primers: MOG-outer-F: 5′-TGGAAGATCCCTTCTATTGGG-3′, MOG-outer-R: 5′-CTG CCAGTCTTCGGTGCAGCCA-3′, MOG-inner-F: 5′-ATCCTCCTGCAGGTCTC TGT-3′, MOG-inner-R: 5′-CAACCAGGGGTCCAAGAACA-3′, GAPDH-F: 5′-GG AGCCAAAAGGGTCATCATCTC-3′, GAPDH-R: 5′-GAGGGGCCATCCACAGT CTTCT-3′. The MOG outer primers will amplify the MOG coding sequence from 431–709 bp (279 bp) and MOG inner primers will amplify 493–667 bp (175 bp).
Morphology of axons by transmission electron microscopy
Freshly prepared sciatic nerve was isolated from mice anesthetized with 13% urethane and prefixed in 4% paraformaldehyde with 2.5% glutaraldehyde in 0.1 M sodium Cacodylate buffer (pH 7.2~7.4) overnight. Tissues were immersed in 1% OsO4 in s-collidine buffer for post-fixation. Dehydration was performed with ethanol and acetone. Resin infiltration followed increased concentration by 100% acetone: Spurr’s resin (3:1, 1:1, 1:3), each for 2~4 hr and the third step overnight. Tissues were immersed in pure Spurr’s resin for 2 hr or overnight before pure resin for 2~4 hr. After embedding in a mode and polymerization under 60 °C, ultrathin sections 70~90-nm thick were cut by using an ultramicrotome (Leica EM UC7), then mounted on a copper grid. Images were obtained by using FEI Tecnai G2 F20 S-TWIN (FEI, Hillsboro, Oregon). The amount of axon with myelin decompaction or axon-myelin dissociation was counted in 6 randomly selected images in each group.
Immunohistochemistry
Mice were anesthetized with an intraperitoneal injection of 1.3 mg/kg urethane (Sigma-Aldrich, St Louis, MO, USA) followed by 4% paraformaldehyde fixation (PFA, Merck, Germany). We used 12-μm–thick DRG cryosections and 5-μm–thick spinal cord paraffin-embedded sections for staining. The following primary antibodies were incubated at 4 °C overnight: rabbit anti-ATF3 (1:1000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rat anti-mouse CD4 (1:100, BD Biosciences, CA, USA), mouse anti-NF200 (1:500, Sigma-Aldrich, St Louis, MO, USA), goat anti-CGRP (1:1000, BioRad, Hercules, CA, USA), rabbit-anti mouse Iba-1 (1:500, Wako Pure Chemical Industry, Osaka, Japan), Alexa Flour 488-conjugated rat anti-mouse GFAP (1:100). Tissue was incubated with secondary antibodies for 1 hr at room temperature: Alexa Fluor 488 donkey anti-goat, goat anti-mouse and goat anti-rabbit; Alexa Fluor 594 donkey anti-rabbit (1:200, Invitrogen technologies, Carlsbad, CA, USA); Alexa Fluor 568 goat anti-rabbit (1:1000); and Alexa Fluor 555 goat anti-rat (1:500, both Thermo Fisher Scientific, Waltham, MA). As for IB4 staining, slides were then immersed in IB4-FITC (Sigma-Aldrich, Inc., St. Louis, MO, USA) containing 1 μM of Mg2+, Mn2+ and Ca2+ for 1.5 hr. Images of DRG sections were acquired under a Ziess Axiovert 200 M fluorescence microscope and processed with ImageJ 64. The maximal background signal was defined as 5 times of the SEM above mean intensity, which is calculated from 9 randomly selected 20 μm × 20 μm square areas from no primary antibody stained images. Images of spinal cord sections were observed by confocal microscopy (LSM 700 stage, Carl Zeiss, Oberkochenm, Germany) and acquired with Zen software (Carl Zeiss, Oberkochen, Germany). Immunofluorescence quantification was analyzed with Integrated Intensity quantification in Metamorph software. To include all possible signals, threshold was set at 70 for Iba-1 staining and 60 for GFAP staining. To further exclude non-specific particles, area limit filter was set at 20 pixel for Iba-1 staining and 50 pixel for GFAP staining. Sections of 3 mice in total were counted in each group.
H&E and Luxol fast blue (LFB) staining
Spinal cord sections were stained for H&E and LFB as previously described68. Briefly, for H&E staining, sections were immersed with Meyer’s hematoxylin followed by 0.5% Eosin Y disodium in 70% alcohol. For LFB staining, sections were stained with 0.1% LFB solution, containing 1 g Solvent blue 38 (Sigma-Aldrich, Inc., St. Louis, MO, USA), 1000 ml 95% alcohol and 5 ml 10% acetic acid, at 60 °C overnight. Photos were obtained by using Pannoramic 250 Flash II (3DHistech Ltd., Budapest, Hungary) and analyzed by panoramic viewer (3DHistech Ltd., Budapest, Hungary).
Analgesic drug application
The analgesic effects of pregabalin were investigated in the chronic phase of EAE. Following pre-drug assessment, the von Frey test was conducted at 30, 60, 90, 120 and 150 min after drug application. For drug application, pregabalin (Toronto Research Chemicals, Toronto, Canada) was dissolved in normal saline to a stock concentration of 78.5 mM and further diluted with normal saline to a final concentration. Each mouse received pregabalin at doses of 30 or 100 μg/kg or 1 mg/kg intraperitoneally.
Statistical analysis
Data are presented as mean ± SEM and analyzed by use of Prism 6. Statistical analysis of clinical severity, rota-rod, open-field, mechanical response and drug response test were conducted by repeat measured two-way ANOVA with Bonferroni difference post-hoc testing or Tukey test. One-way ANOVA was used to analyze the activation level of glial cells in the spinal cord and immunofluorescence results. Unpaired t test was used to analyze the quantification of axon with axon-myelin association and myelin decompaction. P < 0.05 was considered statistically significant.
Additional Information
How to cite this article: Wang, I-C. et al. Peripheral sensory neuron injury contributes to neuropathic pain in experimental autoimmune encephalomyelitis. Sci. Rep. 7, 42304; doi: 10.1038/srep42304 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
We would like to thank Dr. Shing-Hong Lin for consultation in statistics analyses. We thank the Imaging Core Facility of the Institute of Cellular and Organismic Biology, Academia Sinica, especially Yao-Kuan Huang, for technical support in electron microscopy; the Core Facility and Pathology core, IBMS, Academia Sinica, for helpful instruction on confocal microscopy and spinal cord staining respectively. We appreciate for the kindly assistance from Dr. Yen, Jeffery J.Y., Dr. Peng-Peng Ip and Ya-Lin Lin in immune cell IHC and quantitative PCR. This work was supported by grants from Academia Sinica in Taiwan.
Footnotes
The authors declare no competing financial interests.
Author Contributions C.C.C. and C.H.L. conceived and designed the research study, analyzed the data, and wrote the manuscript. I.C.W., C.H.L. designed and conducted the experiments, and analyzed the data. C.Y.C. and F.L. designed the experiments. All authors have read and approved the final manuscript.
References
- Kipp M., van der Valk P. & Amor S. Pathology of multiple sclerosis. CNS & neurological disorders drug targets 11, 506–517 (2012). [DOI] [PubMed] [Google Scholar]
- Pouly S. & Antel J. P. Multiple Sclerosis and Central Nervous System Demyelination. Journal of Autoimmunity 13, 297–306, doi: http://dx.doi.org/10.1006/jaut.1999.0321 (1999). [DOI] [PubMed] [Google Scholar]
- Compston A. & Coles A. Multiple sclerosis. Lancet 359, 1221–1231, doi: 10.1016/S0140-6736(02)08220-X (2002). [DOI] [PubMed] [Google Scholar]
- Toosy A., Ciccarelli O. & Thompson A. Symptomatic treatment and management of multiple sclerosis. Handb Clin Neurol 122, 513–562, doi: 10.1016/B978-0-444-52001-2.00023-6 (2014). [DOI] [PubMed] [Google Scholar]
- Beiske A. G., Pedersen E. D., Czujko B. & Myhr K. M. Pain and sensory complaints in multiple sclerosis. Eur J Neurol 11, 479–482, doi: 10.1111/j.1468-1331.2004.00815.x (2004). [DOI] [PubMed] [Google Scholar]
- Sun W. H. & Chen C. C. Roles of Proton-Sensing Receptors in the Transition from Acute to Chronic Pain. Journal of dental research 95, 135–142, doi: 10.1177/0022034515618382 (2016). [DOI] [PubMed] [Google Scholar]
- Friese M. A. et al. Acid-sensing ion channel-1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system. Nature medicine 13, 1483–1489, doi: 10.1038/nm1668 (2007). [DOI] [PubMed] [Google Scholar]
- Rodrigues D. H., Sachs D. & Teixeira A. L. Mechanical hypernociception in experimental autoimmune encephalomyelitis. Arq Neuropsiquiatr 67, 78–81 (2009). [DOI] [PubMed] [Google Scholar]
- Olechowski C. J., Truong J. J. & Kerr B. J. Neuropathic pain behaviours in a chronic-relapsing model of experimental autoimmune encephalomyelitis (EAE). Pain 141, 156–164, doi: 10.1016/j.pain.2008.11.002 (2009). [DOI] [PubMed] [Google Scholar]
- Aicher S. A., Silverman M. B., Winkler C. W. & Bebo B. F. Jr. Hyperalgesia in an animal model of multiple sclerosis. Pain 110, 560–570, doi: 10.1016/j.pain.2004.03.025 (2004). [DOI] [PubMed] [Google Scholar]
- Lu J. et al. Pain in experimental autoimmune encephalitis: a comparative study between different mouse models. J Neuroinflammation 9, 233, doi: 10.1186/1742-2094-9-233 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan N. & Smith M. T. Multiple sclerosis-induced neuropathic pain: pharmacological management and pathophysiological insights from rodent EAE models. Inflammopharmacology 22, 1–22, doi: 10.1007/s10787-013-0195-3 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thibault K., Calvino B. & Pezet S. Characterisation of sensory abnormalities observed in an animal model of multiple sclerosis: a behavioural and pharmacological study. Eur J Pain 15, 231 e231–216, doi: 10.1016/j.ejpain.2010.07.010 (2011). [DOI] [PubMed] [Google Scholar]
- Staud R., Nagel S., Robinson M. E. & Price D. D. Enhanced central pain processing of fibromyalgia patients is maintained by muscle afferent input: a randomized, double-blind, placebo-controlled study. Pain 145, 96–104, doi: 10.1016/j.pain.2009.05.020 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staud R., Robinson M. E., Weyl E. E. & Price D. D. Pain variability in fibromyalgia is related to activity and rest: role of peripheral tissue impulse input. J Pain 11, 1376–1383, doi: 10.1016/j.jpain.2010.03.011 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staud R., Weyl E. E., Bartley E., Price D. D. & Robinson M. E. Analgesic and anti-hyperalgesic effects of muscle injections with lidocaine or saline in patients with fibromyalgia syndrome. Eur J Pain 18, 803–812, doi: 10.1002/j.1532-2149.2013.00422.x (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasu T., Taguchi T. & Mizumura K. Persistent deep mechanical hyperalgesia induced by repeated cold stress in rats. Eur J Pain 14, 236–244, doi: 10.1016/j.ejpain.2009.05.009 (2010). [DOI] [PubMed] [Google Scholar]
- Lin C. C. et al. An antinociceptive role for substance P in acid-induced chronic muscle pain. Proc Natl Acad Sci USA 109, E76–83, doi: 10.1073/pnas.1108903108 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sluka K. A. et al. Chronic hyperalgesia induced by repeated acid injections in muscle is abolished by the loss of ASIC3, but not ASIC1. Pain 106, 229–239 (2003). [DOI] [PubMed] [Google Scholar]
- Chen W. N. et al. Roles of ASIC3, TRPV1, and NaV1.8 in the transition from acute to chronic pain in a mouse model of fibromyalgia. Mol Pain 10, 40, doi: 10.1186/1744-8069-10-40 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenstein M. Neuropathy: A name for their pain. Nature 535, S10–S11, doi: 10.1038/535S10a (2016). [DOI] [PubMed] [Google Scholar]
- Lin J. H., Chiang Y. H. & Chen C. C. Research strategies for pain in lumbar radiculopathy focusing on acid-sensing ion channels and their toxins. Current topics in medicinal chemistry 15, 617–630 (2015). [DOI] [PubMed] [Google Scholar]
- Pollock M., Calder C. & Allpress S. Peripheral nerve abnormality in multiple sclerosis. Ann Neurol 2, 41–48, doi: 10.1002/ana.410020107 (1977). [DOI] [PubMed] [Google Scholar]
- Ben-Nun A. et al. The autoimmune reactivity to myelin oligodendrocyte glycoprotein (MOG) in multiple sclerosis is potentially pathogenic: effect of copolymer 1 on MOG-induced disease. J Neurol 243, S14–22 (1996). [DOI] [PubMed] [Google Scholar]
- Itoyama Y., Webster H. D., Richardson E. P. Jr. & Trapp B. D. Schwann cell remyelination of demyelinated axons in spinal cord multiple sclerosis lesions. Ann Neurol 14, 339–346, doi: 10.1002/ana.410140313 (1983). [DOI] [PubMed] [Google Scholar]
- Puckett C. et al. Myelin-specific proteolipid protein is expressed in myelinating Schwann cells but is not incorporated into myelin sheaths. J Neurosci Res 18, 511–518, doi: 10.1002/jnr.490180402 (1987). [DOI] [PubMed] [Google Scholar]
- Griffiths I. R. et al. Expression of myelin protein genes in Schwann cells. J Neurocytol 18, 345–352 (1989). [DOI] [PubMed] [Google Scholar]
- Pagany M. et al. Myelin oligodendrocyte glycoprotein is expressed in the peripheral nervous system of rodents and primates. Neurosci Lett 350, 165–168 (2003). [DOI] [PubMed] [Google Scholar]
- Falcone M. et al. Spreading of autoimmunity from central to peripheral myelin: two cases of clinical association between multiple sclerosis and chronic inflammatory demyelinating polyneuropathy. Neurol Sci 27, 58–62, doi: 10.1007/s10072-006-0565-3 (2006). [DOI] [PubMed] [Google Scholar]
- Harding A. M., Kusama N., Hattori T., Gautam M. & Benson C. J. ASIC2 subunits facilitate expression at the cell surface and confer regulation by PSD-95. PloS one 9, e93797, doi: 10.1371/journal.pone.0093797 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zha X. et al. ASIC2 Subunits Target Acid-Sensing Ion Channels to the Synapse via an Association with PSD-95. J Neurosci 29, 8438–8446, doi: 10.1523/jneurosci.1284-09.2009 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsujino H. et al. Activating transcription factor 3 (ATF3) induction by axotomy in sensory and motoneurons: A novel neuronal marker of nerve injury. Molecular and cellular neurosciences 15, 170–182, doi: 10.1006/mcne.1999.0814 (2000). [DOI] [PubMed] [Google Scholar]
- Ruscheweyh R., Forsthuber L., Schoffnegger D. & Sandkuhler J. Modification of classical neurochemical markers in identified primary afferent neurons with Abeta-, Adelta-, and C-fibers after chronic constriction injury in mice. The Journal of comparative neurology 502, 325–336, doi: 10.1002/cne.21311 (2007). [DOI] [PubMed] [Google Scholar]
- Turcotte D. et al. Nabilone as an adjunctive to gabapentin for multiple sclerosis-induced neuropathic pain: a randomized controlled trial. Pain medicine (Malden, Mass.) 16, 149–159, doi: 10.1111/pme.12569 (2015). [DOI] [PubMed] [Google Scholar]
- O’Connor A. B., Schwid S. R., Herrmann D. N., Markman J. D. & Dworkin R. H. Pain associated with multiple sclerosis: systematic review and proposed classification. Pain 137, 96–111, doi: 10.1016/j.pain.2007.08.024 (2008). [DOI] [PubMed] [Google Scholar]
- Ziemann A. E. et al. Seizure termination by acidosis depends on ASIC1a. Nature neuroscience 11, 816–822, doi: 10.1038/nn.2132 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziemann A. E. et al. The amygdala is a chemosensor that detects carbon dioxide and acidosis to elicit fear behavior. Cell 139, 1012–1021, doi: 10.1016/j.cell.2009.10.029 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du J., Reznikov L. R. & Welsh M. J. Expression and activity of acid-sensing ion channels in the mouse anterior pituitary. PloS one 9, e115310, doi: 10.1371/journal.pone.0115310 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Z. G. et al. Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 118, 687–698, doi: 10.1016/j.cell.2004.08.026 (2004). [DOI] [PubMed] [Google Scholar]
- Wemmie J. A. Neurobiology of panic and pH chemosensation in the brain. Dialogues in clinical neuroscience 13, 475–483 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzuca M. et al. A tarantula peptide against pain via ASIC1a channels and opioid mechanisms. Nature neuroscience 10, 943–945, doi: 10.1038/nn1940 (2007). [DOI] [PubMed] [Google Scholar]
- Gartzen K., Katzarava Z., Diener H. C. & Putzki N. Peripheral nervous system involvement in multiple sclerosis. Eur J Neurol 18, 789–791, doi: 10.1111/j.1468-1331.2010.03149.x (2011). [DOI] [PubMed] [Google Scholar]
- Ayromlou H. et al. Electrodiagnostic evaluation of peripheral nervous system changes in patients with multiple sclerosis. The Malaysian journal of medical sciences: MJMS 20, 32–38 (2013). [PMC free article] [PubMed] [Google Scholar]
- Warwick R. A., Ledgerwood C. J., Brenner T. & Hanani M. Satellite glial cells in dorsal root ganglia are activated in experimental autoimmune encephalomyelitis. Neurosci Lett 569, 59–62, doi: 10.1016/j.neulet.2014.03.033 (2014). [DOI] [PubMed] [Google Scholar]
- Begum F., Zhu W., Cortes C., MacNeil B. & Namaka M. Elevation of tumor necrosis factor alpha in dorsal root ganglia and spinal cord is associated with neuroimmune modulation of pain in an animal model of multiple sclerosis. J Neuroimmune Pharmacol 8, 677–690, doi: 10.1007/s11481-013-9449-5 (2013). [DOI] [PubMed] [Google Scholar]
- Wenjun Zhu a E. E. F. a., Farhana Begum b, Parvez Vora a, Kelvin Au a, Yuewen Gong a, Brian MacNeil c, Prakash Pillai a, b, Mike Namaka a, b. The role of dorsal root ganglia activation and brain-derived neurotrophic factor in multiple sclerosis. J. Cell. Mol. Med. Vol. 16, pp. 1856–1865 (2012). [DOI] [PMC free article] [PubMed]
- Perera C. J. et al. Effects of active immunisation with myelin basic protein and myelin-derived altered peptide ligand on pain hypersensitivity and neuroinflammation. Journal of neuroimmunology 286, 59–70, doi: 10.1016/j.jneuroim.2015.07.004 (2015). [DOI] [PubMed] [Google Scholar]
- Paganya Maria, Jagodicb Maja & Schubarta Anna Danielle Pham-Dinhc, Corinne Bachelinc, Anne Baron van Evercoorenc, François Lachapellec, Tomas Olssonb, Christopher Liningtona, d. Myelin oligodendrocyte glycoprotein is expressed in the peripheral nervous system of rodents and primates. Neuroscience letter, doi: 10.1016/S0304-3940(03)00899-1 (2003). [DOI] [PubMed] [Google Scholar]
- Frezel N., Sohet F., Daneman R., Basbaum A. I. & Braz J. M. Peripheral and central neuronal ATF3 precedes CD4+ T-cell infiltration in EAE. Experimental neurology 283, 224–234, doi: 10.1016/j.expneurol.2016.06.019 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basbaum A. I., Bautista D. M., Scherrer G. & Julius D. Cellular and Molecular Mechanisms of Pain. Cell 139, 267–284, doi: 10.1016/j.cell.2009.09.028 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y. L., Xie Z. L., Wu Y. W., Duan W. R. & Xie Y. K. Early demyelination of primary A-fibers induces a rapid-onset of neuropathic pain in rat. Neuroscience 200, 186–198, doi: 10.1016/j.neuroscience.2011.10.037 (2012). [DOI] [PubMed] [Google Scholar]
- Abrahamsen B. et al. The cell and molecular basis of mechanical, cold, and inflammatory pain. Science (New York, N.Y.) 321, 702–705, doi: 10.1126/science.1156916 (2008). [DOI] [PubMed] [Google Scholar]
- Snider W. D. & McMahon S. B. Tackling Pain at the Source: New Ideas about Nociceptors. Neuron 20, 629–632, doi: 10.1016/S0896-6273(00)81003-X (1998). [DOI] [PubMed] [Google Scholar]
- Shields S. D. et al. Nav1.8 expression is not restricted to nociceptors in mouse peripheral nervous system. Pain 153, 2017–2030, doi: 10.1016/j.pain.2012.04.022 (2012). [DOI] [PubMed] [Google Scholar]
- Burnstock G. Purinergic Mechanisms and Pain. Advances in pharmacology (San Diego, Calif.) 75, 91–137, doi: 10.1016/bs.apha.2015.09.001 (2016). [DOI] [PubMed] [Google Scholar]
- Hsieh Y. L., Chiang H., Lue J. H. & Hsieh S. T. P2X3-mediated peripheral sensitization of neuropathic pain in resiniferatoxin-induced neuropathy. Exp Neurol 235, 316–325, doi: 10.1016/j.expneurol.2012.02.013 (2012). [DOI] [PubMed] [Google Scholar]
- McCoy E. S., Taylor-Blake B. & Zylka M. J. CGRPalpha-expressing sensory neurons respond to stimuli that evoke sensations of pain and itch. PloS one 7, e36355, doi: 10.1371/journal.pone.0036355 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quick K. et al. TRPC3 and TRPC6 are essential for normal mechanotransduction in subsets of sensory neurons and cochlear hair cells. Open biology 2, 120068, doi: 10.1098/rsob.120068 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kestell G. R., Anderson R. L., Clarke J. N., Haberberger R. V. & Gibbins I. L. Primary afferent neurons containing calcitonin gene-related peptide but not substance P in forepaw skin, dorsal root ganglia, and spinal cord of mice. The Journal of comparative neurology 523, 2555–2569, doi: 10.1002/cne.23804 (2015). [DOI] [PubMed] [Google Scholar]
- Bennett D. L. H. et al. A Distinct Subgroup of Small DRG Cells Express GDNF Receptor Components and GDNF Is Protective for These Neurons after Nerve Injury. The Journal of Neuroscience 18, 3059–3072 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shortland P. J. et al. ATF3 expression in L4 dorsal root ganglion neurons after L5 spinal nerve transection. The European journal of neuroscience 23, 365–373, doi: 10.1111/j.1460-9568.2005.04568.x (2006). [DOI] [PubMed] [Google Scholar]
- Ueda H. Peripheral mechanisms of neuropathic pain - involvement of lysophosphatidic acid receptor-mediated demyelination. Molecular pain 4, 11, doi: 10.1186/1744-8069-4-11 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalman S., Osterberg A., Sorensen J., Boivie J. & Bertler A. Morphine responsiveness in a group of well-defined multiple sclerosis patients: a study with i.v. morphine. European journal of pain (London, England) 6, 69–80, doi: 10.1053/eujp.2001.0307 (2002). [DOI] [PubMed] [Google Scholar]
- Bauer C. S. et al. The anti-allodynic α2δ ligand pregabalin inhibits the trafficking of the calcium channel α2δ-1 subunit to presynaptic terminals in vivo. Biochemical Society Transactions 38, 525–528 (2010). [DOI] [PubMed] [Google Scholar]
- Field M. J. et al. Identification of the α(2)-δ-1 subunit of voltage-dependent calcium channels as a molecular target for pain mediating the analgesic actions of pregabalin. Proc Natl Acad Sci USA 103, 17537–17542, doi: 10.1073/pnas.0409066103 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu P.-Y. et al. Acid-Sensing Ion Channel-1a Is Not Required for Normal Hippocampal LTP and Spatial Memory. The Journal of Neuroscience 33, 1828–1832, doi: 10.1523/jneurosci.4132-12.2013 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C.-C. et al. A role for ASIC3 in the modulation of high-intensity pain stimuli. Proceedings of the National Academy of Sciences 99, 8992–8997, doi: 10.1073/pnas.122245999 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung C.-Y. & Liao F. CXCR3 signaling in glial cells ameliorates experimental autoimmune encephalomyelitis by restraining the generation of a pro-Th17 cytokine milieu and reducing CNS-infiltrating Th17 cells. Journal of Neuroinflammation 13, 1–17, doi: 10.1186/s12974-016-0536-4 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.