Abstract
Lentivirals vectors (LVs) are used for various gene transfer applications, notably for hematopoietic gene therapy, but methods are lacking to precisely evaluate parameters that control the efficiency of transduction in relation with the entry of vectors into target cells. We adapted a fluorescence resonance energy transfer-based human immunodeficiency virus-1 fusion assay to measure the entry of non-replicative recombinant LVs in various cell types, including primary human hematopoietic stem/progenitor cells (HSPCs), and to quantify the level of transduction of the same initially-infected cells. The assay utilizes recombinant LVs containing β-lactamase (BLAM)-Vpr chimeric proteins (BLAM-LVs) and encoding a truncated form of the low affinity nerve growth factor receptor (ΔNGFR). After infection of target cells with BLAM-LVs, the vector entry rapidly leads to BLAM-Vpr release into the cytoplasm which is measured by cleavage of a fluorescent substrate using flow cytometry. Parallel cultures of the same infected cells show transduction efficiency resulting from ΔNGFR expression. This LV-based fusion/transduction assay is a dynamic and versatile tool, revealing for instance the postentry restrictions of LVs known to occur in cells of hematopoietic origin, especially human HSPCs. Furthermore, this BLAM-LV assay allowed us to evaluate the effect of cytokine prestimulation of HSPCs on the entry step of LVs. The assay also shows that transduction enhancers like Vectofusin-1 or Retronectin can partially relieve the postentry block but their effects differ in how they promote LV entry. In conclusion, one such assay should be useful to study hematopoietic postentry restrictions directed against LVs and therefore should allow improvements in various LV-based gene therapy protocols.
INTRODUCTION
Human immunodeficiency virus 1 (HIV-1)-based lentiviral vectors (LVs) are highly efficient tools for gene transfer approaches, especially in the context of ex vivo hematopoietic gene therapies (D’Costa et al., 2009; Naldini, 2011). However, a limiting factor that can be encountered during lentiviral transduction is the low efficacy of the entry or postentry events into target cells. One strategy to optimize the binding and the fusion of LVs with target cells is the capacity to pseudotype LVs with heterologous viral envelope glycoproteins (GPs) or ligand-fused GPs, leading to a cellular entry through different surface receptors (Anliker et al., 2010; Frecha et al., 2008b). Another strategy to improve the entry step is the addition of cofactors during the transduction procedure, such as cationic polymers – For example, polybrene (Cornetta and Anderson, 1989; Toyoshima and Vogt, 1969), DEAE-Dextran (Vogt, 1967) or cationic peptides (e.g. Vectofusin-1 (Fenard et al., 2013b), Retronectin (Pollok and Williams, 1999), protamine sulphate (Cornetta and Anderson, 1989), human Semen Enhancer of Viral Infection (SEVI) (Wurm et al., 2010) or HIV-1 gp120-derived peptides (Yolamanova et al., 2013)). The action mechanism of these cationic additives is mainly described as the capacity to neutralize membrane charges, to improve virus/cell interactions, virus/cell fusion and/or to promote virus aggregation (Davis et al., 2004; Fenard et al., 2013b; Pollok and Williams, 1999; Roan et al., 2009; Yolamanova et al., 2013).
Despite an improvement of the fusion step with the target cell membranes, LVs are sometimes restricted into the cytoplasm during the trafficking from the plasma membrane to the nucleus. These postentry restrictions have been extensively studied in HIV-1-infected T cells, macrophages and monocytes from different species, with the identification of major antiviral factors like SAMHD1, tetherin, TRIM5α, the APOBEC3 family (for reviews, see (Duggal and Emerman, 2012; Harris et al., 2012; Wolf and Goff, 2008)) and some DNA repair pathways (Fenard et al., 2009; Lau et al., 2004; Lloyd et al., 2006; Weil et al., 2013; Yoder et al., 2006). Concerning the human hematopoietic stem/progenitor cells (HSPCs), an attractive target for gene therapy developments, the endogenous proteasome activity and also the cyclin-dependent kinase inhibitor p21Waf1/Cip1/Sdi1 have been shown to exert a strong restriction on the transduction efficiency with LVs or on HIV-1 infection ( Zhang et al., 2005, 2007; Santoni de Sio et al., 2006, 2008).
To evaluate the extent of postentry restriction into HSPCs and to study the effect of transduction enhancers on entry and postentry steps, it is critical to develop an assay that is capable to concurrently study the fusion and the transduction efficiency in the same cell population. Using an HIV-1 fluorescence resonance energy transfer (FRET)-based fusion assay (Cavrois et al., 2002), adapted to study non replicative LVs and recombinant transgene expression, we show that LVs are subjected to a strong postfusion restriction in human hematopoietic cells, either immortalized T lymphocytes or HSPCs expressing the hCD34+ marker. This postfusion restriction can be partially saturated when increasing amounts of LVs are used in the assay and/or when transduction enhancers such as Vectofusin-1 or Retronectin are used in the transduction protocol.
MATERIAL AND METHODS
Reagents
CCF2-AM substrate and the CO2-independent medium were purchased from Life Technologies (St-Aubin, France). Bovine Serum albumin, human γ-globulins, Bafilomycin A1, Paraformaldehyde (PFA) and Probenecid were obtained from Sigma-Aldrich (St-Quentin-Fallavier, France). Retronectin was purchased from Ozyme (St-Quentin-en-Yvelines, France). Vectofusin-1 was synthesized by Genecust (Dudelange, Luxembourg).
Plasmids
The plasmid encoding VAI and VAII (pAdVAntage) and the pCMV4- -lactamase (BlaM)-Vpr were respectively obtained from Promega (Charbonnières-les-Bains, France) or from Addgene, (Cambridge, MA) (plasmid 21950) (Cavrois et al., 2002); The plasmids encoding HIV-1 Rev (pK.Rev), HIV-1 gagpol (pKLgagpol) were described by (Merten et al., 2011). The lentiviral transfer plasmid pRRL-PGK-low affinity nerve growth factor receptor (ΔNGFR) was constructed based on pRRLsin-cPPT-hPGK-GFP-WPRE by substituting the GFP coding sequence, after digestion with BamHI and SalI, with that of ΔNGFR coding sequence obtained by polymerase chain reaction (PCR) from the commercial plasmid pMACS-LNGFR-IRES (Miltenyi Biotec, Paris, France).
Cell line culture
HCT116 cells derived from a human colorectal carcinoma (CCL-247, ATCC) and 293T cells (Merten et al., 2011) were cultured at 37°C and 5% CO2 in Dulbecco’s modified Eagle’s medium(DMEM+Glutamax) supplemented with 10% heat inactivated fetal calf serum (FCS; Life Technologies). Jurkat E6.1 immortalized T cells (TIB-152, ATCC) were maintained in RPMI 1640 (Life Technologies) supplemented with 10% heat-inactivated FCS.
Human CD34+ cell culture
Umbilical cord blood (UCB) samples were obtained in accordance with international ethics principles and French national law under declaration number DC-201-1655 to the French Ministry of Research and Higher Studies. UCB samples were collected from the Centre Hospitalier Sud-Francilien in Evry after uncomplicated births. hCD34+ cells were obtained by immunomagnetic selection (Miltenyi Biotec) from the mononuclear cell fraction of UCB. hCD34+ cells were preactivated as described previously (Charrier et al., 2011). Briefly, hCD34+ cells were incubated overnight with X-vivo 20 medium (Lonza, Levallois-Perret, France) supplemented with 50 U/ml penicillin, 50 μg/ml streptomycin and 2mM L-glutamine (Life Technologies), SCF (25 ng/ml), Flt-3 ligand (50 ng/ml), TPO (25 ng/ml) and IL-3 (10 ng/ml) (Miltenyi Biotec).
Viral vector production and vector titering
HIV-1 derived LVs were generated by transient calcium phosphate transfection of 293T cells with a 6-plasmid vector system as described in Supplementary Table S1. The viral supernatants were collected 48 hr post-transfection, filtered (0.45 μm), aliquoted, and stored at −80°C before use. An ultracentrifugation step is added before freezing for experiments requiring high titers of LVs. Infectious titers, expressed as the number of infectious genomes per milliliter (ig/ml), were determined by quantitative PCR on genomic DNA of serially infected HCT116 cells as described previously (Merten et al., 2011). Physical particle titers were determined by measuring HIV-1 p24 capsid contents using a commercial enzyme-linked immunosorbent assay kit (Perkin Elmer, Courtaboeuf, France).
Fusion and transduction assay
HCT116 cells (4 × 105 plated cells/well), Jurkat T cells (5 × 105 cells/well) or preactivated hCD34+ cells (7 × 104 cells/well) were incubated with VSV-G-BLAM-LVs for 2.5 hr at 37°C and 5% CO2. For Retronectin conditions, LV particles were pre-adsorbed on plate-bound Retronectin (7μg/cm2) for 2 hr and hCD34+ cells were subsequently added to the well for 2.5 hr at 37°C and 5% CO2. Next, for each condition, the cell sample was split in two: an aliquot of cells was transferred into a new well and further cultured for 4–7 days, while the remaining cells were processed for the fusion assay (Cavrois et al., 2004). Briefly, cells were washed with 200 μl of CO2-independent medium and incubated with 100 μl of CCF2/AM loading solution for 1 hr at room temperature in the dark. After the loading step, cells were washed and incubated with 200 μl of CO2-independent medium supplemented with 10% FCS and 2.5 mM probenecid, for 16 hr at room temperature in the dark. Cells were then washed with PBS1x and fixed with 1.2% PFA. The change in emission fluorescence of CCF2 was monitored by flow cytometry using an LSRII and data were analyzed with DiVa software (BD Biosciences, Le Pont de Claix-Cedex, France). To evaluate the level of transduction, cells maintained in culture for 4–7 days were washed and the nonspecific binding on hCD34+ cells was prevented by pre-incubating the cells with human γ-globulins. Next, cells were labeled for 30 min at 4°C with Allophycocyanin-conjugated antihuman LNGFR (Miltenyi Biotech). Cells were then washed and analyzed by flow cytometry.
RESULTS
Production of LVs containing BLAM-Vpr and encoding ΔNGFR
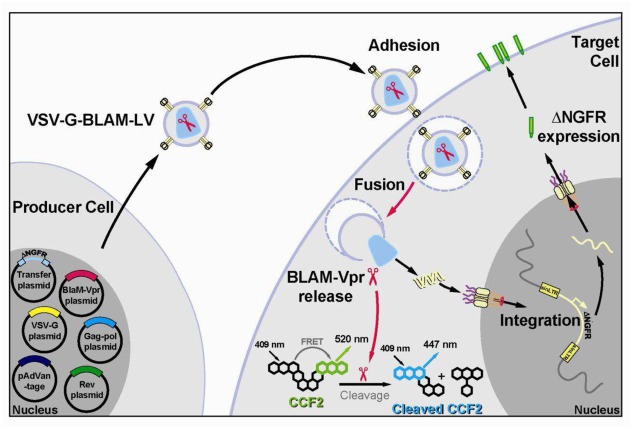
To better understand the parameters that control the efficiency of transduction in relation with the entry of gene transfer vectors into target cells, we developed a new method based on a FRET-based fusion assay (BLAM fusion assay) previously developed to study the fusion of HIV-1 virions with target cells. Such assay could also be used to monitor the level of infection by following the expression of de novo intracellular HIV-1 gag expression after few days (Cavrois et al., 2002). However, with non-replicative LVs (i.e. the proviral DNA is devoided of gag-pol-env gene sequences), it is not possible to follow intracellular HIV-1 gag de novo production as described in the original method. Hence, BLAM-LVs (LVs carrying the fusion protein BLAM-Vpr) were designed to encode a truncated form of the low-affinity nerve growth factor receptor (ΔNGFR) (Bonini et al., 2003; Rudoll et al., 1996) under the control of the ubiquitously expressed PGK promoter. This transmembrane protein is a safe and easily detectable marker at the surface of any transduced cells, including primary cells (e.g. HSPCs). High titers of recombinant BLAM-LVs pseudotyped with the VSV-G envelope GP (VSV-G-BLAM-LVs) were generated by transient calcium phosphate transfection of 293T cells with a six-plasmid vector system (Fig. 1 and Supplementary Table S1). Four typical plasmids are used to produce third generation LVs: the plasmids encoding the HIV-1 rev protein, the HIV-1 gagpol polyprotein, the VSV-G envelope GP and the transfer vector plasmid encoding ΔNGFR. For BLAM-LV production, two other plasmids have to be included (Supplementary Table S1). The plasmid pCMV4-BLAM-Vpr, encoding the BLAM enzyme fused to the HIV-1 Vpr protein, specifically incorporated into lentiviral particles through its interaction with the p6 region of HIV-1 p55gag (Kondo et al., 1995; Paxton et al., 1993) and the pAdVAntage plasmid, encoding adenoviral VAI/VAII RNAs, acting as specific inhibitors of the Protein Kinase R (Supplementary Table S1). This last plasmid improves the level of expression of the BLAM-Vpr chimeric construct (Cavrois et al., 2002) and is also used to improve the efficiency of LV production (Pernod et al., 2004). The use of these NGFR-encoding BLAM-LVs allows us to monitor, in the same cell population, the efficiency of lentiviral fusion and the level of effective transduction of target cells (Fig. 1).
Fig. 1. Schematic representation of the BLAM-LV system.
Representation of the VSV-G-BLAM-LV production using a six-plasmid vector system (left) and the target cell transduction with VSV-G-BLAM-LVs (right) allowing the concurrent study of the fusion efficiency (CCF2 cleavage) and the transduction efficiency ( NGFR expression at the cell surface). NGFR, low-affinity nerve growth factor receptor; BLAM, -lactamase; LV, lentiviral vector.
Concurrent study of the lentiviral fusion and transduction of hematopoietic and non-hematopoietic cell lines
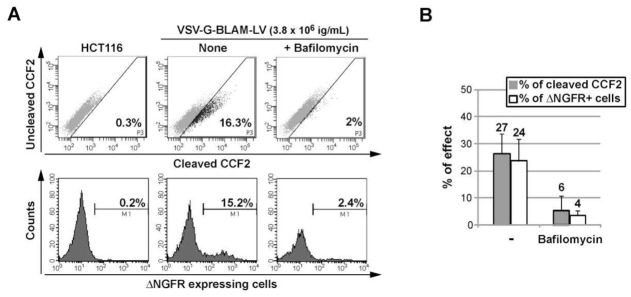
First, the BLAM-LV assay has been performed on adherent HCT116 cells (Fig. 2A), a non-hematopoietic cell line, highly permissive to VSV-G lentiviral peudotypes. As shown in Fig. 2, the use of only 3.8 × 106 ig/ml of VSV-G-BLAM-LVs can lead to comparable efficiencies of fusion (27%) and transduction (24%) in an infection protocol of only two hours and in the absence of any culture additives. To evaluate the specificity and the sensitivity of this assay, an inhibitor of the vacuolar proton ATPase (Bafilomycin A1) was added. As expected, the fusogenic activity of the pH-dependent VSV-G envelope GP is strongly inhibited in presence of Bafilomycin (Fig. 2A and B).
Fig. 2. Concurrent study of VSV-G-BLAM-LV fusion/transduction with a non-hematopoietic cell line.
(A) Analysis of VSV-G-BLAM-LV fusion in the HCT116 human colorectal cancer cell line (upper panels). Analysis of the cell transduction (day 5) with VSV-G-BLAM-LVs after NGFR expression at the surface of HCT116 cells (lower panels). (B) Histograms are representing the average fusion (gray) and transduction (white) efficiency obtained for HCT116 cells ± SD (n=3). SD, standard deviations.
Next, VSV-G-BLAM-LVs were used to transduce Jurkat cells, a highly permissive immortalized human T cell line. As shown in Fig. 3A, the use of 7.5 × 106 ig/ml of VSV-G-BLAM-LVs can lead to a strong lentiviral fusion with Jurkat cells (above 77%). However, in the same cell suspension, the transduction efficiency is reaching only 29%. Interestingly, the ratio between the fusion and the transduction efficiency (F/T ratio) is decreasing when increasing amounts of VSV-G-BLAM-LVs are used in the assay (Fig. 3B). This discrepancy between fusion and transduction is not an artefact since the transduction efficiencies obtained with Jurkat cells at 7.5 × 106 ig/ml (27%) are comparable to the one obtained with HCT116 cells (around 24%) in which we observed a comparable level of fusion and transduction (Fig. 3B).
Fig. 3. Concurrent study of VSV-G-BLAM-LV fusion/transduction with an immortalized human T cell line.
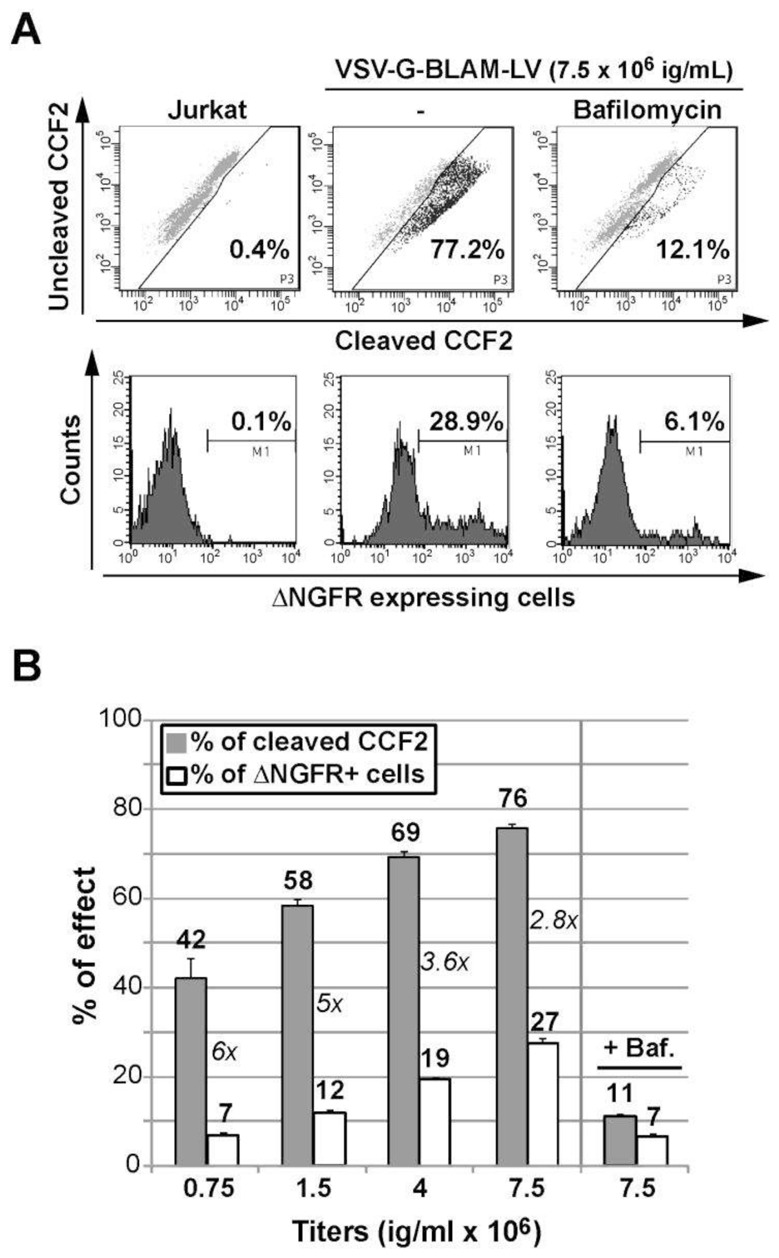
(A) Analysis of the fusion and the transduction of Jurkat T cells with VSV-G-BLAM-LV. The fusion-positive cell gate was established based on untransduced living cells loaded with the CCF2 substrate (upper left panel). In the presence of VSV-G-BLAM-LV, a control condition including the VSV-G fusion inhibitor Bafilomycin (150 nM) was used. The cell transduction efficiency with VSV-G-BLAM-LVs was monitored at day 5 by following NGFR expression at the cell surface (Lower panels). (B) Histograms are representing the fusion (gray) and the transduction (white) efficiency obtained for Jurkat T cells incubated with various infectious titers (ig/ml) of VSV-G-BLAM-LVs. A control condition including Bafilomycin (Baf.) was added for the highest dose of vector. Data are represented as the average values ± SD (n=3). The numbers in italic represent the ratio between fusion and transduction efficiency (F/T ratio) for each vector dose.
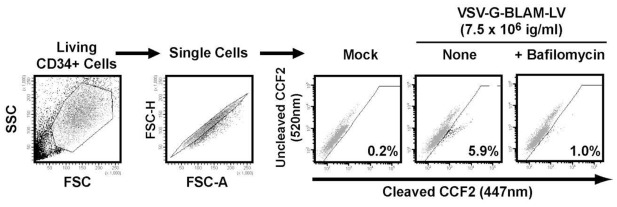
Numerous LV-based hematopoietic gene therapies target hCD34+ HSPCs. The fusion efficiency of VSV-G lentiviral pseudotypes with HSPCs has never been studied before. For that, hCD34+ HSPCs isolated by positive immunomagnetic selection from cord blood samples were used (Fig. 4). hCD34+ HSPCs were transduced with low doses of VSV-G-BLAM-LVs for only 2 hr which results in low levels of detectable vector-infected cells (6%) (Fig. 4). As expected, this percentage was downregulated in presence of Bafilomycin (1%). This result highlights the strong difference in lentiviral fusion potency between hCD34+ cells and Jurkat T cells since the percentage of fusion is one log higher in Jurkat T cells (77%) using the same infectious titer of VSV-G-BLAM-LVs (Fig. 2). Therefore, the BLAM-LV assay is a powerful tool to highlight postfusion restrictions into target cells and shows that these restrictions are cell-type dependent and sensitive to the amount of LVs used in the assay.
Fig. 4. Gating strategy for analysis of hCD34+ cells transduced with VSV-G-BLAM-LVs.
Preactivated hCD34+ cells were left untransduced (Mock) or were transduced with VSV-G-BLAM-LVs for 2 hr at 37°C in the absence (None) or presence of a fusion inhibitor (Bafilomycin A1, 150nM). Subsequently, the viral fusion efficiency was estimated by measuring the level of cleaved CCF2 substrate.
Effect of hCD34+ HSPC prestimulation with cytokines on lentiviral fusion/transduction efficiencies
Current transduction protocols of hCD34+ cells with LVs include a cytokine prestimulation step to increase infection of HSPCs. The BLAM-LV assay was used to investigate whether or not the cytokine prestimulation step had an effect on the entry of lentiviral pseudotypes. hCD34+ cells were cultured in absence (Mock) or presence of cytokines and transduced in parallel during 2.5 hr with VSV-G-BLAM-LVs. Figure 5A shows that the treatment of hCD34+ cells with cytokines significantly improved the fusion efficiency of VSV-G-BLAM-LVs, on average by 1.5 fold (27 to 39% of cleaved CCF2+ cells). In addition, cytokines also enhanced the transduction efficiency (Fig. 5B) to the same extent as fusion (16 to 23% of NGFR+ cells). This result suggests that the effect of cytokines on HSPCs during lentiviral transduction is mainly exerted at the level of entry.
Fig. 5. Effect of hCD34+ cells prestimulation on the efficiency of lentiviral fusion and transduction.
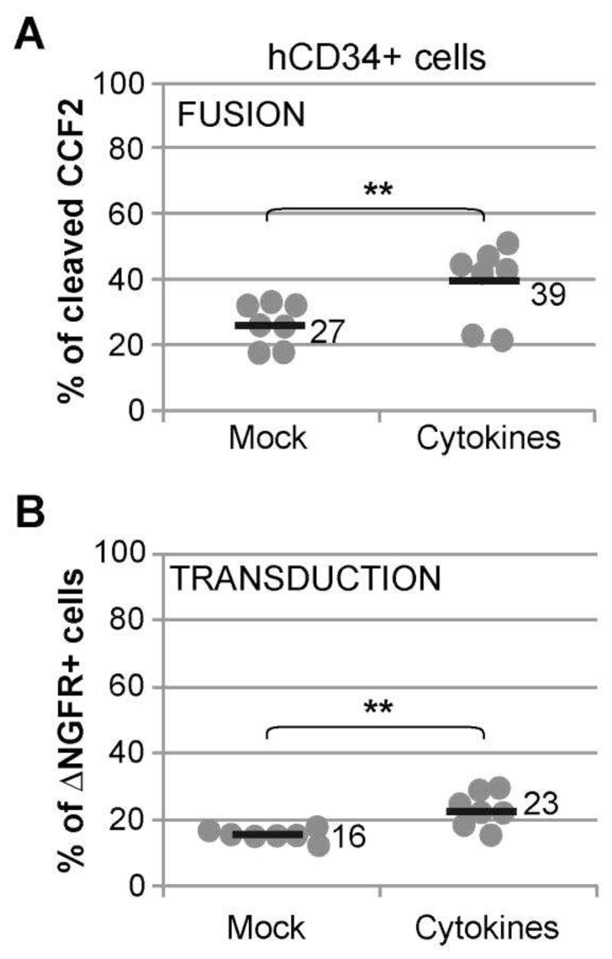
hCD34+ cells cultured overnight in absence (Mock) or presence of a cocktail of cytokines (see the Material and Methods section), were incubated 2.5 hr with VSV-G-BLAM-LVs (108 ig/ml), precoated for 2 hr on plate-bound Retronectin (7 μg/cm2). The viral fusion and transduction efficiency were estimated by measuring the level of cleaved CCF2 substrate (A) and the level of NGFR expression (B), respectively. Data are obtained from five different cord blood donors in simplicate or duplicate. Bars indicate the mean value of the distributions (** p<0.01, bilateral paired Student’s t-test).
Influence of transduction enhancers on postfusion restrictions observed for VSV-G lentiviral pseudotypes in human CD34+ HSPCs
Most of the gene therapy protocols describing HSPC transduction are currently using a fragment of the human Fibronectin protein, called Retronectin, as a transduction enhancer, but the effects of Retronectin on the fusion of lentiviral particles and target cell membranes have never been evaluated. Furthermore, we have recently described the Vectofusin-1 peptide as a strong enhancer of lentiviral adhesion and fusion (Fenard et al., 2013b). Vectofusin-1 acts in a dose-dependent manner and in solution in the presence of LVs and cells, thus providing a practical system to study the dynamics of LV entry and transduction. To test the influence of Retronectin and Vectofusin-1 on fusion and transduction, various concentrations of VSV-G-BLAM-LVs were used to transduce hCD34+ HSPCs in absence or presence of the culture additives (Fig. 6A and B). The average F/T ratios calculated for each condition were plotted on a 2D contour chart (Fig. 6C). The lines in the contour chart delineate different F/T ratios ranging from 1–5 fold to 15–20 fold. The use of increasing concentrations of LV with hCD34+ cells decreases the F/T ratio, suggesting a partial saturation process of postentry restrictions (Fig. 6C). Such effects of increasing concentrations of LVs are also found on Jurkat cells, as observed in Fig. 2. In presence of Retronectin, the level of lentiviral fusion is not statistically different from the control conditions in the absence of additive (Fig. 6A), whereas a slight effect on transduction is observed (Fig. 6B). Concerning the Vectofusin-1 peptide, the F/T ratio evolves in a more pronounced manner when this additive is added in the hCD34+ cell cultures and depending on the concentration of LV added (Fig. 6C), showing that Vectofusin-1-mediated fusion (Fig. 6A) is able to partially saturate the postentry restrictions without the need to increase LV input. This Vectofusin-1 effect on the F/T ratio is observed at the optimal concentration of 12μg/ml but is also effective at concentrations as low as 6μg/ml (Supplementary Fig. S1). As shown in Fig. 6, the Retronectin effect does not seem to be optimal when low doses of VSV-G-BLAM-LVs are used. Hence, a high infectious titer of vector (108 ig/ml), typically used for transduction of hCD34+ cells in the presence of Retronectin, has been incubated with plate-bound Retronectin and hCD34+ HSPCs during 2 hr (Fig. 7). The fusion assay performed in these conditions showed a slight effect of Retronectin on the fusion process, from 37 to 46% (Fig. 7A) while at the same time, the level of transduction was increased by 2-fold from 12 to 24% (Fig. 7B), which constitutes a greater effect. In these conditions, the presence of Retronectin decreased the F/T ratio from 3 to 2-fold.
Fig. 6. Influence of the infectious titers and the transduction enhancers on the F/T ratio in hCD34+ cells.
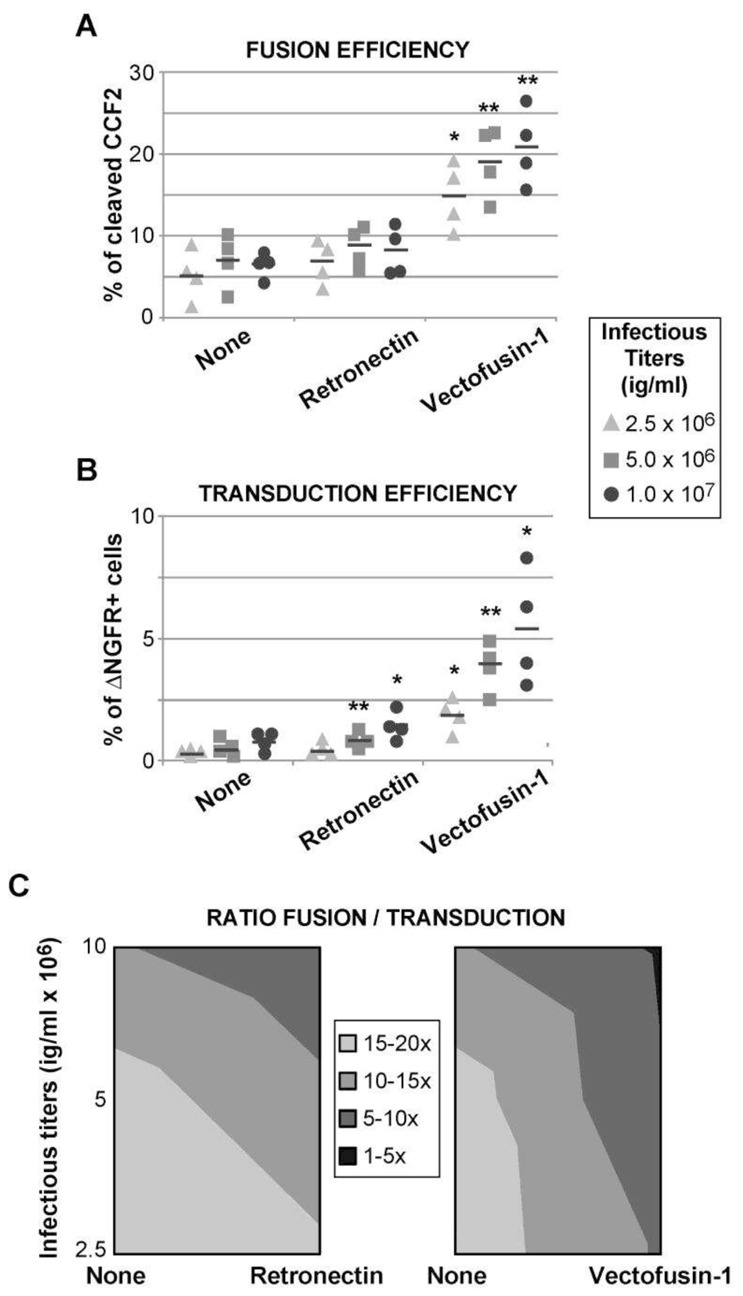
Fusion (A) and transduction (B) efficiencies obtained for hCD34+ HSPCs isolated from four independent cord blood donors in presence of three vector doses of VSV-G-BLAM-LVs and in the absence (none) or presence of Retronectin (7μg/cm2) or Vectofusin-1 (12 μg/ml). Bars indicate the mean value of the distributions. (C) The average F/T ratios obtained for each condition from (A) and (B)have been plotted on a 2D contour chart (2D view of the surface chart from above obtained with the Microsoft Excel software). The lines in the contour chart delineate different F/T ratios ranging from 1–5 to 15–20 fold (** p<0.01, * p<0.05, bilateral paired Student’s t-test).
Fig. 7. Influence of plate-bound Retronectin on the lentiviral fusion/transduction ratio using high titers of VSV-G-BLAM-LVs.
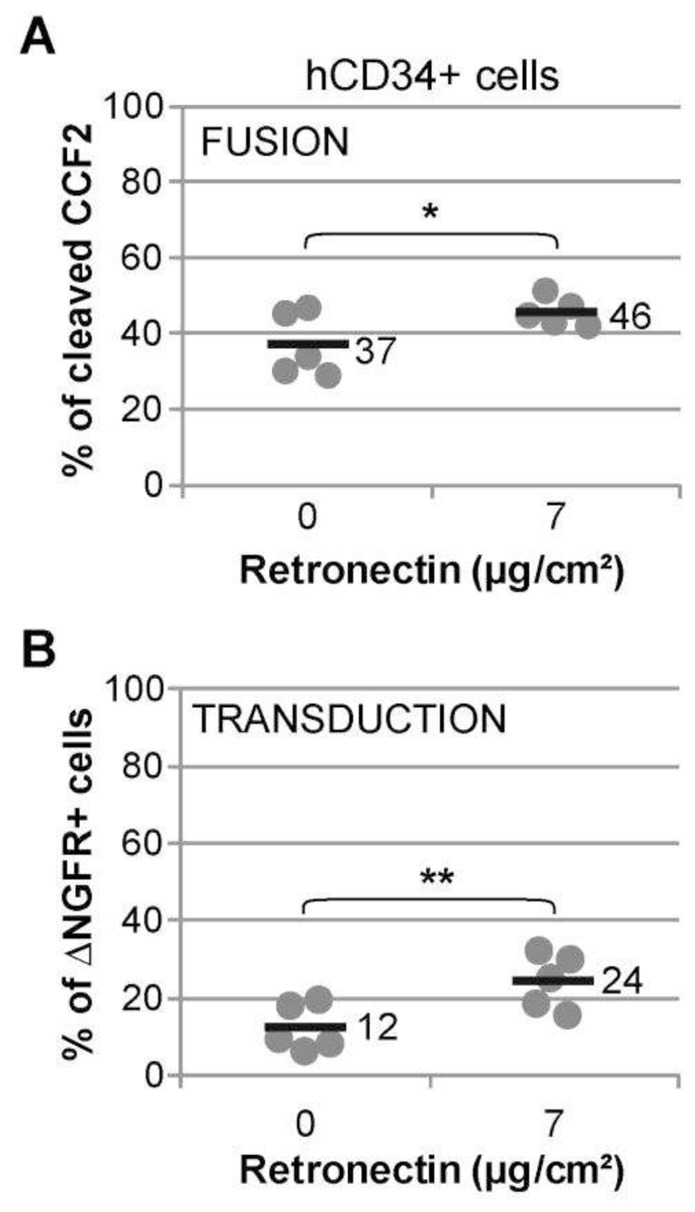
Study of the fusion (A) and transduction (B) efficiencies of hCD34+ cells with high titers of VSV-G-BLAM-LVs (108 ig/ml) in absence or presence of plate-bound Retronectin (7 μg/cm2) for 2 hr. Data are obtained from five different cord blood donors. Bars indicate the mean value of the distributions (* p<0.05, ** p<0.01, bilateral paired Student’s t-test).
The concurrent study of the lentiviral fusion and transduction step therefore provides insights on the effects of culture additives such as Vectofusin-1 or Retronectin. Although both improve transduction, they utilize different mechanisms of action, acting mainly on the fusion and consequently on the postfusion steps for Vectofusin-1, while Retronectin seems to act mainly on the postfusion steps.
DISCUSSION
In this report, an HIV-1 viral fusion assay was adapted to study non-replicative LVs (BLAM-LVs) pseudotyped with the VSV-G envelope GP. The BLAM-LV assay allows the concurrent monitoring of the fusion efficiency between cellular and viral membranes and the determination of the effective transduction level in the same cell population.
The BLAM-LV assay shows that at low vector doses, the fusion of the vector with immortalized T cells or with hCD34+ HSPCs is a lot more efficient than the effective transduction level achieved, revealing postentry restrictions in these cells. Using cord blood hCD34+ HSPCs that were either unstimulated or stimulated with mild concentrations of cytokines, the BLAM-LV assay shows that the positive effect of the cytokines on transduction is mainly exerted at the level of entry, probably through plasma membrane reorganization events. Indeed, it has been shown previously that early acting cytokines induce the polarization of plasma membranes and the redistribution of lipid raft markers on HSPCs (Giebel et al., 2004; Yamazaki et al., 2007). We also describe two distinct culture additives that enhance LV transduction through distinct mechanisms of action. For instance, the Vectofusin-1 peptide, derived from the LAH4-L1 transfection agent (Mason et al., 2006; Lam et al., 2012; Fenard et al., 2013a; Kichler et al., 2013), is a new culture additive that potently enhances the fusion and transduction of HSPCs with LVs (Fenard et al., 2013b). Using this additive, postentry restrictions observed in hCD34+ HSPCs were partially overcome, much more efficiently than the strategy consisting to use increasing concentrations of vector only. When the effect of the broadly used transduction enhancer Retronectin was studied, the latter was shown to act mainly on postentry restrictions rather than the fusion step per se, potentially a consequence of the stimulation of VLA-4 and VLA-5 cell surface integrins by the Retronectin sub-domains CS-1 and cell binding domain respectively (Yokota et al., 1998; Horiuchi et al., 2009).
Most of the lentiviral transduction protocols developed for clinical applications are using high concentrations of VSV-G-LVs, with sometimes two rounds of infection to reach the highest level of engraftment of genetically modified target cells in the patient. Despite encouraging results using this approach, it could be of high interest to be able to decrease the quantity of clinical-grade LVs to increase the number of patients that could be treated per clinical batch of vector and to reduce the potential toxicity associated with the manipulation of cells (especially with highly fusogenic VSV-G lentiviral pseudotypes). Studies aiming to improve the postentry steps are therefore of high interest, either through a better understanding of the molecular mechanisms responsible for this restriction or through the developments of compounds than can boost the entry step, or saturate or abrogate the postentry restrictions.
It could also be interesting to study various lentiviral pseudotypes with the BLAM-LV assay. Retargeting of LV entry to cell types of interest using heterologous viral GPs or ligand-fused GPs (Frecha et al., 2008b; Anliker et al., 2010) could be a way to avoid postentry restrictions. For instance, envelope GPs harboring a strong hematopoietic-tropism, such as the modified gibbon ape leukemia virus (GALVTR) GP (Christodoulopoulos and Cannon 2001), the modified feline endogenous virus (RD114TR) GP (Sandrin et al., 2002) or the Edmonston measles virus hemagglutinin/fusion (H/F) GP (Frecha et al., 2008a), may behave differently at the level of entry since they are using different target receptors. Of note, the use of H/F-LVs has been shown to efficiently transduce quiescent T and B lymphocytes, a particular feature described as the consequence of the specific interaction of H/F-LVs to both CD46 and SLAM receptors (Frecha et al., 2011).
In conclusion, this BLAM-LV fusion assay is a useful tool (1) to evaluate the degree of postentry restriction for various LV pseudotypes into attractive target cells (e.g. HSPCs, lymphocytes); (2) to determine which cells are targeted during the entry process in comparison with cells that are really transduced in the same bulk population; (3) to better characterize culture additives that are able to improve the adhesion and fusion steps of LVs; and (4) to better define and develop compounds that can attenuate or abrogate the postentry restrictions. All these applications of the BLAM-LV assay will help to improve our knowledge of the lentiviral transduction and lead to the development of more effective transduction protocols for LV-based gene therapies.
Supplementary Material
Supplementary Fig. S1. Influence of the infectious titers and Vectofusin-1 (6μg/ml) on the fusion/transduction (F/T) ratio in hCD34+ cells. Fusion (A) and transduction efficiencies (B) obtained for hCD34+ HSPCs isolated from three independent cord blood donors in presence of three vector doses of VSV-G-BLAM-LVs and in absence or presence of 6μg/ml of Vectofusin-1. Bars indicate the mean value of the distributions. (C) The average F/T ratio obtained for each condition from (A) and (B) have been plotted on a 2D contour chart as in Fig. 6. The lines in the contour chart delineate different F/T ratios ranging from 1–5 to 40–50 fold (** p<0.01, * p<0.05, bilateral paired Student’s t-test).
Supplementary Table S1. Six-plasmid vector system for VSV-G-BLAM-LV production
Acknowledgments
This work was supported by the Association Française contre les Myopathies (AFM) and the Fondation pour la Recherche Médicale (FRM). We thank Genethon collaborators: in particular, the Flow Cytometry Core for technical assistance, Armelle Viornery for cord blood processing, and Nathalie Holic for critical reading of the article. We are very grateful to the staff of the Maternité de l’hopital Louise Michel, Evry, France, and the mothers for providing us with UCB samples. We also thank Warner C. Greene for the pCMV4-BlaM-Vpr plasmid and Veronique Parietti for constructing the pRRL-PGK-ΔNGFR.
Footnotes
AUTHOR DISCLOSURE STATEMENT
The authors declare no competing financial interests.
References
- Anliker B, Abel T, Kneissl S, et al. Specific gene transfer to neurons, endothelial cells and hematopoietic progenitors with lentiviral vectors. Nat Methods. 2010;7:929–35. doi: 10.1038/nmeth.1514. [DOI] [PubMed] [Google Scholar]
- Bonini C, Grez M, Traversari C, et al. Safety of retroviral gene marking with a truncated NGF receptor. Nat Med. 2003;9:367–9. doi: 10.1038/nm0403-367. [DOI] [PubMed] [Google Scholar]
- Cavrois M, De Noronha C, Greene WC. A sensitive and specific enzyme-based assay detecting HIV-1 virion fusion in primary T lymphocytes. Nat Biotechnol. 2002;20:1151–4. doi: 10.1038/nbt745. [DOI] [PubMed] [Google Scholar]
- Cavrois M, Neidleman J, Bigos M, Greene WC. Fluorescence resonance energy transfer-based HIV-1 virion fusion assay. Methods Mol Biol. 2004;263:333–44. doi: 10.1385/1-59259-773-4:333. [DOI] [PubMed] [Google Scholar]
- Charrier S, Ferrand M, Zerbato M, et al. Quantification of lentiviral vector copy numbers in individual hematopoietic colony-forming cells shows vector dose-dependent effects on the frequency and level of transduction. Gene Ther. 2011;18:479–87. doi: 10.1038/gt.2010.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christodoulopoulos I, Cannon PM. Sequences in the cytoplasmic tail of the gibbon ape leukemia virus envelope protein that prevent its incorporation into lentivirus vectors. J Virol. 2001;75:4129–38. doi: 10.1128/JVI.75.9.4129-4138.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornetta K, Anderson WF. Protamine sulfate as an effective alternative to polybrene in retroviral-mediated gene-transfer: implications for human gene therapy. J Virol Methods. 1989;23:187–94. doi: 10.1016/0166-0934(89)90132-8. [DOI] [PubMed] [Google Scholar]
- D’Costa J, Mansfield SG, Humeau LM. Lentiviral vectors in clinical trials: Current status. Curr Opin Mol Ther. 2009;11:554–64. [PubMed] [Google Scholar]
- Davis HE, Rosinski M, Morgan JR, Yarmush ML. Charged polymers modulate retrovirus transduction via membrane charge neutralization and virus aggregation. Biophys J. 2004;86:1234–42. doi: 10.1016/S0006-3495(04)74197-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duggal NK, Emerman M. Evolutionary conflicts between viruses and restriction factors shape immunity. Nat Rev Immunol. 2012;12:687–95. doi: 10.1038/nri3295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenard D, Genries S, Scherman D, et al. Infectivity enhancement of different HIV-1-based lentiviral pseudotypes in presence of the cationic amphipathic peptide LAH4-L1. J Virol Methods. 2013a;189:375–8. doi: 10.1016/j.jviromet.2013.02.005. [DOI] [PubMed] [Google Scholar]
- Fenard D, Houzet L, Bernard E, et al. Uracil DNA Glycosylase 2 negatively regulates HIV-1 LTR transcription. Nucleic Acids Res. 2009;37:6008–18. doi: 10.1093/nar/gkp673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenard D, Ingrao D, Seye A, et al. Vectofusin-1, a new viral entry enhancer, strongly promotes lentiviral transduction of human hematopoietic stem cells. Mol Ther Nucleic Acids. 2013b doi: 10.1038/mtna.2013.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frecha C, Costa C, Negre D, et al. Stable transduction of quiescent T cells without induction of cycle progression by a novel lentiviral vector pseudotyped with measles virus glycoproteins. Blood. 2008a;112:4843–52. doi: 10.1182/blood-2008-05-155945. [DOI] [PubMed] [Google Scholar]
- Frecha C, Levy C, Costa C, et al. Measles virus glycoprotein-pseudotyped lentiviral vector-mediated gene transfer into quiescent lymphocytes requires binding to both SLAM and CD46 entry receptors. J Virol. 2011;85:5975–85. doi: 10.1128/JVI.00324-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frecha C, Szecsi J, Cosset FL, Verhoeyen E. Strategies for targeting lentiviral vectors. Curr Gene Ther. 2008b;8:449–60. doi: 10.2174/156652308786848003. [DOI] [PubMed] [Google Scholar]
- Giebel B, Corbeil D, Beckmann J, et al. Segregation of lipid raft markers including CD133 in polarized human hematopoietic stem and progenitor cells. Blood. 2004;104:2332–8. doi: 10.1182/blood-2004-02-0511. [DOI] [PubMed] [Google Scholar]
- Harris RS, Hultquist JF, Evans DT. The restriction factors of human immunodeficiency virus. J Biol Chem. 2012;287:40875–83. doi: 10.1074/jbc.R112.416925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horiuchi Y, Onodera M, Miyagawa Y, et al. Kinetics and effect of integrin expression on human CD34(+) cells during murine leukemia virus-derived retroviral transduction with recombinant fibronectin for stem cell gene therapy. Hum Gene Ther. 2009;20:777–83. doi: 10.1089/hum.2008.159. [DOI] [PubMed] [Google Scholar]
- Kichler A, Mason AJ, Marquette A, Bechinger B. Histidine-rich cationic amphipathic peptides for plasmid DNA and siRNA delivery. Methods Mol Biol. 2013;948:85–103. doi: 10.1007/978-1-62703-140-0_7. [DOI] [PubMed] [Google Scholar]
- Kondo E, Mammano F, Cohen EA, Gottlinger HG. The p6gag domain of human immunodeficiency virus type 1 is sufficient for the incorporation of Vpr into heterologous viral particles. J Virol. 1995;69:2759–64. doi: 10.1128/jvi.69.5.2759-2764.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam JK, Liang W, Lan Y, et al. Effective endogenous gene silencing mediated by pH responsive peptides proceeds via multiple pathways. J Control Release. 2012;158:293–303. doi: 10.1016/j.jconrel.2011.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau A, Kanaar R, Jackson SP, O’Connor MJ. Suppression of retroviral infection by the RAD52 DNA repair protein. EMBO J. 2004;23:3421–9. doi: 10.1038/sj.emboj.7600348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lloyd AG, Tateishi S, Bieniasz PD, et al. Effect of DNA repair protein Rad18 on viral infection. PLoS Pathog. 2006;2:e40. doi: 10.1371/journal.ppat.0020040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason AJ, Martinez A, Glaubitz C, et al. The antibiotic and DNA-transfecting peptide LAH4 selectively associates with, and disorders, anionic lipids in mixed membranes. Faseb J. 2006;20:320–2. doi: 10.1096/fj.05-4293fje. [DOI] [PubMed] [Google Scholar]
- Merten OW, Charrier S, Laroudie N, et al. Large-scale manufacture and characterization of a lentiviral vector produced for clinical ex vivo gene therapy application. Hum Gene Ther. 2011;22:343–56. doi: 10.1089/hum.2010.060. [DOI] [PubMed] [Google Scholar]
- Naldini L. Ex vivo gene transfer and correction for cell-based therapies. Nat Rev Genet. 2011;12:301–15. doi: 10.1038/nrg2985. [DOI] [PubMed] [Google Scholar]
- Paxton W, Connor RI, Landau NR. Incorporation of Vpr into human immunodeficiency virus type 1 virions: requirement for the p6 region of gag and mutational analysis. J Virol. 1993;67:7229–37. doi: 10.1128/jvi.67.12.7229-7237.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pernod G, Fish R, Liu JW, Kruithof EK. Increasing lentiviral vector titer using inhibitors of protein kinase R. Biotechniques. 2004;36:576–8. 580. doi: 10.2144/04364BM02. [DOI] [PubMed] [Google Scholar]
- Pollok KE, Williams DA. Facilitation of retrovirus-mediated gene transfer into hematopoietic stem and progenitor cells and peripheral blood T-lymphocytes utilizing recombinant fibronectin fragments. Curr Opin Mol Ther. 1999;1:595–604. [PubMed] [Google Scholar]
- Roan NR, Munch J, Arhel N, et al. The cationic properties of SEVI underlie its ability to enhance human immunodeficiency virus infection. J Virol. 2009;83:73–80. doi: 10.1128/JVI.01366-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudoll T, Phillips K, Lee SW, et al. High-efficiency retroviral vector mediated gene transfer into human peripheral blood CD4+ T lymphocytes. Gene Ther. 1996;3:695–705. [PubMed] [Google Scholar]
- Sandrin V, Boson B, Salmon P, et al. Lentiviral vectors pseudotyped with a modified RD114 envelope glycoprotein show increased stability in sera and augmented transduction of primary lymphocytes and CD34+ cells derived from human and nonhuman primates. Blood. 2002;100:823–32. doi: 10.1182/blood-2001-11-0042. [DOI] [PubMed] [Google Scholar]
- Santoni de Sio FR, Cascio P, Zingale A, et al. Proteasome activity restricts lentiviral gene transfer into hematopoietic stem cells and is down-regulated by cytokines that enhance transduction. Blood. 2006;107:4257–65. doi: 10.1182/blood-2005-10-4047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santoni de Sio FR, Gritti A, Cascio P, et al. Lentiviral vector gene transfer is limited by the proteasome at postentry steps in various types of stem cells. Stem Cells. 2008;26:2142–52. doi: 10.1634/stemcells.2007-0705. [DOI] [PubMed] [Google Scholar]
- Toyoshima K, Vogt PK. Enhancement and inhibition of avian sarcoma viruses by polycations and polyanions. Virology. 1969;38:414–26. doi: 10.1016/0042-6822(69)90154-8. [DOI] [PubMed] [Google Scholar]
- Vogt PK. DEAE-dextran: enhancement of cellular transformation induced by avian sarcoma viruses. Virology. 1967;33:175–7. doi: 10.1016/0042-6822(67)90109-2. [DOI] [PubMed] [Google Scholar]
- Weil AF, Ghosh D, Zhou Y, et al. Uracil DNA glycosylase initiates degradation of HIV-1 cDNA containing misincorporated dUTP and prevents viral integration. Proc Natl Acad Sci USA. 2013;110:E448–57. doi: 10.1073/pnas.1219702110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf D, Goff SP. Host restriction factors blocking retroviral replication. Annu Rev Genet. 2008;42:143–63. doi: 10.1146/annurev.genet.42.110807.091704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wurm M, Schambach A, Lindemann D, et al. The influence of semen-derived enhancer of virus infection on the efficiency of retroviral gene transfer. J Gene Med. 2010;12:137–46. doi: 10.1002/jgm.1429. [DOI] [PubMed] [Google Scholar]
- Yamazaki S, Iwama A, Morita Y, et al. Cytokine signaling, lipid raft clustering, and HSC hibernation. Ann NY Acad Sci. 2007;1106:54–63. doi: 10.1196/annals.1392.017. [DOI] [PubMed] [Google Scholar]
- Yoder K, Sarasin A, Kraemer K, et al. The DNA repair genes XPB and XPD defend cells from retroviral infection. Proc Natl Acad Sci USA. 2006;103:4622–7. doi: 10.1073/pnas.0509828103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokota T, Oritani K, Mitsui H, et al. Growth-supporting activities of fibronectin on hematopoietic stem/progenitor cells in vitro and in vivo: structural requirement for fibronectin activities of CS1 and cell-binding domains. Blood. 1998;91:3263–72. [PubMed] [Google Scholar]
- Yolamanova M, Meier C, Shaytan AK, et al. Peptide nanofibrils boost retroviral gene transfer and provide a rapid means for concentrating viruses. Nat Nanotechnol. 2013;8:130–6. doi: 10.1038/nnano.2012.248. [DOI] [PubMed] [Google Scholar]
- Zhang J, Attar E, Cohen K, et al. Silencing p21(Waf1/Cip1/Sdi1) expression increases gene transduction efficiency in primitive human hematopoietic cells. Gene Ther. 2005;12:1444–52. doi: 10.1038/sj.gt.3302544. [DOI] [PubMed] [Google Scholar]
- Zhang J, Scadden DT, Crumpacker CS. Primitive hematopoietic cells resist HIV-1 infection via p21. J Clin Invest. 2007;117:473–81. doi: 10.1172/JCI28971. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Fig. S1. Influence of the infectious titers and Vectofusin-1 (6μg/ml) on the fusion/transduction (F/T) ratio in hCD34+ cells. Fusion (A) and transduction efficiencies (B) obtained for hCD34+ HSPCs isolated from three independent cord blood donors in presence of three vector doses of VSV-G-BLAM-LVs and in absence or presence of 6μg/ml of Vectofusin-1. Bars indicate the mean value of the distributions. (C) The average F/T ratio obtained for each condition from (A) and (B) have been plotted on a 2D contour chart as in Fig. 6. The lines in the contour chart delineate different F/T ratios ranging from 1–5 to 40–50 fold (** p<0.01, * p<0.05, bilateral paired Student’s t-test).
Supplementary Table S1. Six-plasmid vector system for VSV-G-BLAM-LV production