Abstract
Purpose of review:
To identify the different indications for the treatment of neurologic disorders with the potassium channel blockers 4-aminopyridine (4-AP) and 3,4-diaminopyridine (3,4-DAP).
Recent findings:
4-AP is an effective symptomatic treatment for downbeat nystagmus (DBN), episodic ataxia type 2 (EA2) (5–10 mg TID), and impaired gait in multiple sclerosis (MS) (10 mg BID). 3,4-DAP (5 mg/d–20 mg TID) improves symptoms in Lambert-Eaton myasthenic syndrome (LEMS) (randomized placebo-controlled trials for all 4 entities). 4-AP may also be effective in cerebellar gait ataxia of different etiologies (2 case series), upbeat nystagmus, and limb ataxia in MS (single cases). In the recommended dosages, they are well tolerated. The assumed mode of action is a blockade of mainly Kv1.5: in DBN, this increases the excitability of Purkinje cells (PC), and in EA2, restores the precision of resting discharge of PC. In MS, 4-AP improves the conduction of action potentials in demyelinated axons, and in LEMS, 3,4-DAP facilitates the transmission at the neuromuscular endplate by prolonging the action potential duration.
Summary:
There is sufficient evidence that APs are indicated for the symptomatic treatment of DBN, EA2, gait ataxia due to MS and cerebellar disorders, and LEMS with a reasonable risk-benefit profile.
Since the 1950s, it has been known that aminopyridines (AP) inhibit certain axonal K channels, which were named after them (“the A current”).1,2 In particular, they block Kv1.5 voltage-activated potassium channels, thereby prolonging the duration of action potentials in axons because of delayed repolarization. This mode of action is relevant for their efficacy in Lambert-Eaton myasthenic syndrome (LEMS) at the neuromuscular junction leading to an increase of acetylcholine release3 and multiple sclerosis (MS) at demyelinated axons thereby improving action potential conductance.2,4 The common mechanism behind the therapeutic influence of AP in cerebellar disorders lies within their effect on cerebellar Purkinje cells (PCs). In vitro studies demonstrated that 4-AP (in therapeutic concentrations) increases the resting discharge rate and excitability of PCs of the guinea pig cerebellum5 and the precision of pace-making of PCs in the tottering mouse, an animal model of episodic ataxia type 2 (EA2)6; both 4-AP and 3,4-diaminopyridine (3,4-DAP) reduced the frequency of attacks; this is probably because they raise the threshold for their triggering.7 Furthermore, AP normalizes the firing rate and the motor behavior in the ataxin-1 mutant mouse in vivo.8 Remarkably, animals treated early demonstrated better motor function in the longer term, which may be mediated by a neuroprotective effect due to an enhanced electrical activity of PCs.8 Due to an increased activity of PC,5 AP evidently restores the GABAergic inhibitory influence on the deep cerebellar nuclei mediated by the vestibulo-cerebellum. This increased inhibitory influence has been confirmed by neurophysiologic findings in patients with ataxia telangiectasia. Here, the ingestion of 10 mg 4-AP resulted in a reduced time constant of the angular vestibulo-ocular reflex (VOR).9 These various electrophysiologic effects explain why such a simple molecule has so many indications for the treatment of various neurologic disorders with different underlying pathophysiologies such as MS, LEMS, downbeat nystagmus (DBN), upbeat nystagmus (UBN), EA2, and cerebellar gait ataxia. As mentioned above, the drug may also have a neuroprotective effect.8,10 Clinically, both 3,4-DAP and 4-AP are currently used but for different indications: 3,4-DAP in LEMS and 4-AP in central nervous disorders because it better penetrates the blood–brain barrier.11 The clinical efficacy of 4-AP is currently being examined in further randomized controlled trials in EA2 (EAT2TREAT) and cerebellar gait disorders (FACEG). All in all, AP is a fine example of repurposing of a pharmacologic agent. In this overview, the different clinical indications as well as the mode of action of AP are described and an outlook on further indications such as acute vertigo is given.
Treatment of different types of nystagmus with 4-AP
Downbeat nystagmus
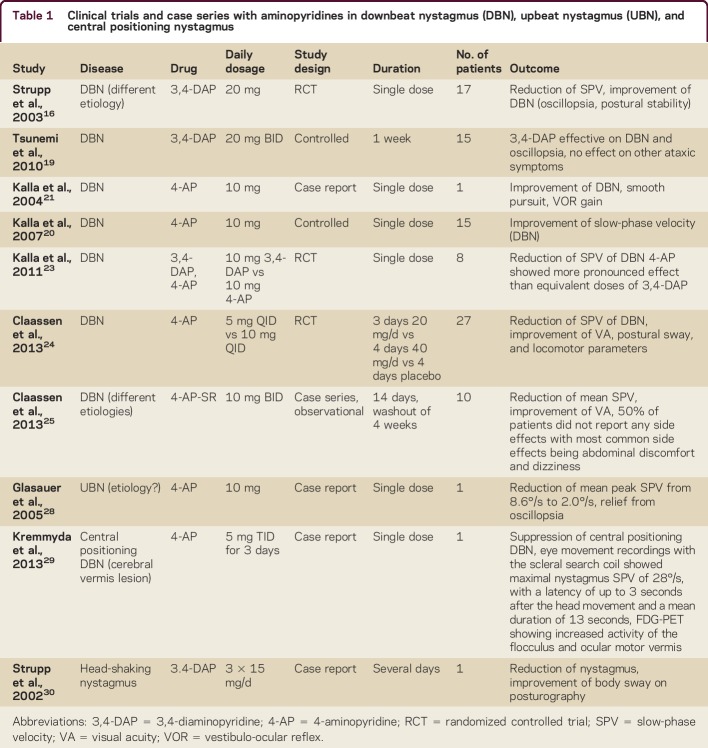
DBN is a frequent form of acquired persisting fixation nystagmus,12 mostly due to cerebellar degeneration leading to a floccular hypofunction.13,14 Patients with DBN predominantly have postural imbalance and cerebellar gait ataxia, as well as oscillopsia, which leads to reduced visual acuity (VA).15 In 2003, the first randomized controlled trial (RCT) showed a significant effect of a single dosage of 20 mg 3,4-DAP on peak slow-phase velocity (SPV) 30 minutes after ingestion.16 The drug was well tolerated.16 These findings were confirmed by others.17–19 In spinocerebellar ataxia type 6, 3,4-DAP also reduced mean peak SPV, but other ataxic symptoms and postural control remained unaffected after treatment over 1 week (20 mg BID).19 Other studies showed that APs worked best in patients with cerebellar atrophy without having considerable effects in patients with structural brainstem lesions, most likely because the target structure is no longer intact.20 4-AP restores the function of the vertical and horizontal neural integrator, which also explains its effects on gaze-evoked nystagmus.20,21 Equal doses of 4-AP were superior to 3,4-DAP because 4-AP crosses the blood–brain barrier more easily22 and should, therefore, be preferred.23 In 2013, an RCT proved the effect of 4-AP (5 mg QID) in DBN (reduction of mean SPV of DBN by about 50%, improvement of VA).24 At higher dosages (4-AP 10 mg QID), a reduction of postural sway and an improvement of motor performance assessed by the timed Get Up and Go Test was demonstrated, although the patients did not notice any subjective improvement; this was discussed as probably being caused by the short half-life of 4-AP.24 This shortage may be overcome by the sustained release form of 4-AP (4-AP-SR; Fampyra, Biogen Idec, Mississauga, Canada), which is also efficient in observational case series in a dosage of 10–20 mg/d,25 but further RCTs are needed. The published RCTs and observational studies as well as case series on APs are summarized in table 1.
Table 1.
Clinical trials and case series with aminopyridines in downbeat nystagmus (DBN), upbeat nystagmus (UBN), and central positioning nystagmus
Based on the current state-of-the-art RCTs, the use of 4-AP (5 mg 2–4 times daily) or 4-AP-SR (10–20 mg/d) is generally recommended for the treatment of DBN.25–27
UBN, central positioning, and head-shaking nystagmus
UBN—a central fixation nystagmus beating upwards in primary position—is rarer than DBN. Oscillopsia is often irritating, but the symptoms of UBN usually persist only for months.15 4-AP has been shown to be effective in UBN28 and central positional nystagmus,29 while 3,4-DAP suppressed head-shaking nystagmus by an improvement of nerve conduction or an increase of cerebellar inhibitory input30 (table 1).
Episodic ataxia type 2
EA2 is the most frequent subtype of episodic ataxia and belongs to the growing number of ion channel disorders. CACNA1A gene mutations, encoding the α-subunit of the P/Q-type calcium channel, are found in about 60% of patients.31–33 The leading symptoms are recurrent attacks of ataxic symptoms, lasting for hours to days, often provoked by physical or emotional stress or alcohol. More than 90% of patients present with persisting central cerebellar ocular disturbances in between the attacks, such as gaze-holding deficits, saccadic smooth pursuit, impaired suppression of the VOR, and especially DBN.31 These clinical signs help to differentiate patients with EA2 from patients with migraine, who only present with minor ocular motor dysfunction.32,34 In the past, the treatment of choice was the carbonic anhydrase inhibitor acetazolamide in a dosage of 250–1,000 mg/d.35 Acetazolamide had been shown to suppress the number and severity of attacks in EA2, and effects on interictal oculomotor and postural function have also been documented.36 However, the effect has only been described in case series; there are so far no RCTs on its efficacy.26,27 Furthermore, its adverse effects (e.g., kidney stones, nephrocalcinosis, hyperhidrosis, paresthesia, muscle stiffening with easy fatigability, and gastrointestinal disturbances, dose-related) considerably limit its use in clinical practice.
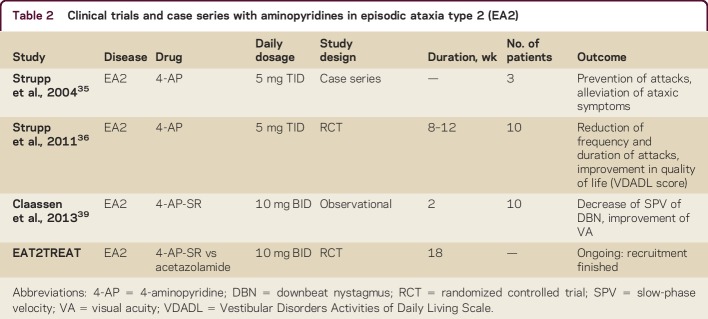
Nowadays the treatment of choice is 4-AP, starting with a first observational study in 2004 on 3 patients35 and confirmed in an RCT on patients with EA2 and familial ataxias in 2011.36 The published data on APs are summarized in table 2. The recommended dosage is 5–10 mg TID.
Table 2.
Clinical trials and case series with aminopyridines in episodic ataxia type 2 (EA2)
In the tottering mouse, the animal model of EA2,6,7 the evident effect was in increasing the precision of pace-making of PCs, while other animal studies suggested an extrafloccular effect37 contrary to the mathematical model of increased excitability in EA2.2,38 In ataxin-1-mutant mice, AP was proven to normalize the firing rate as well as the motor behavior.8 Therefore, a neuroprotective effect due to changes in electrical activity of PCs was discussed in mice treated earlier with AP.8 As a consequence, an earlier treatment in neurodegenerative ataxia may be positive for the long-term course and clinical outcome.8 This may also be true for other neurodegenerative cerebellar diseases such as EA2 or DBN as well.
Gait ataxia in MS
Walking impairment is a clinical hallmark of MS and has a fundamental effect on everyday functioning and quality of life. Besides the treatment of fatigue symptoms with AP, the drug gained momentum after several controlled trials demonstrated that 4-AP-SR improved the walking capability in patients with MS. In 2 phase II clinical trials with 36 and 206 patients, respectively, with definite MS, post hoc analysis revealed a response rate of ∼37% during the treatment with 10–40 mg 4-AP-SR BID compared to 9% during placebo. The mean percent walking speed improvements in a 25-foot walk test were ∼25%.40
The following phase III trials used the dosage of 10 mg 4-AP-SR BID and enrolled 301 and 239 patients, respectively, with definite MS. The responder rates for treatment were ∼35% (with ∼25% walking speed improvement) for treatment compared to ∼8% for placebo.e1 Responders had reduced self-assessed ambulation-related disability and improved lower extremity muscle functioning during treatment. Extension studies of the 2 phase III trials evaluated the open-label, long-term usage of 10 mg 4-AP-SR BID.e2 The studies illustrated typical walking speed decline when 4-AP-SR was discontinued and prompt improvements when the drug was restarted. Data from both extension studies showed that improvements were maintained during long-term usage. The effect of 4-AP-SR on multiple postural domains besides walking speed, e.g., coordination of stepping, dynamic balance, and sit-to-stand transitions, had been evaluated as well. The open-label exploratory study showed improvements in all these domains after 4 weeks of treatment with 4-AP-SR.e3 A more recent observational study suggested the suitability of pretreatment motor evoked potentials in order to predict the therapeutic response to 4-AP-SR. All patients with normal central motor conduction time prior to treatment were identified as nonresponders in this study.e4 Moreover, there is evidence of a functional improvement of everyday mobility induced by the gait improvement. Patients with an increased walking pace under treatment with 4-AP-SR also showed increased physical activity in accelerometer-based long-term measurements.e4 The putative mode of action in MS is the restoration of nerve conduction via blockade of the potassium channels that become exposed during demyelination.2,4 In MS, a beneficial effect on upper limb tremor also was reported in one patient.e5
Gait ataxias of other etiologies
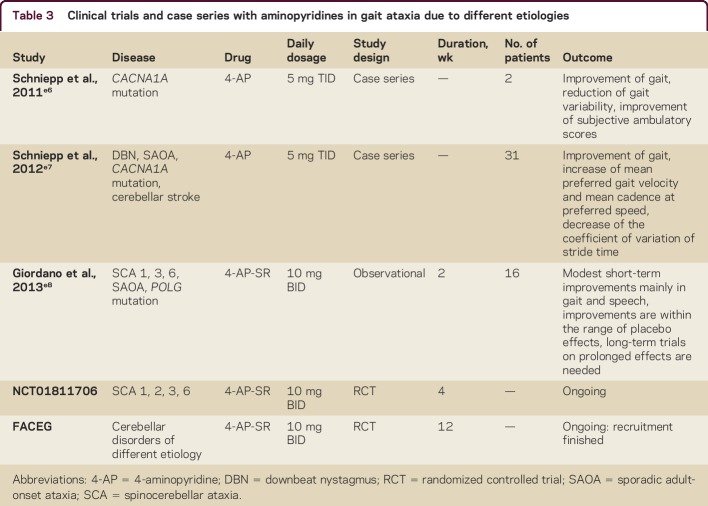
Based on the positive effects of AP on cerebellar oculomotor functions, one can hypothesize about a possible effect of AP on cerebellar locomotion control (table 3). In a case description of 2 patients with progressive cerebellar ataxia (CA) due to mutations of the CACNA1A gene, 4-AP improved the precision of stepping (the coefficient of variation of stride time); the effect was speed-dependent, with the highest magnitude during fast walking.e6 This finding was further extended by a retrospective case series with 31 patients with cerebellar gait ataxia due to different etiologies (multisystem degeneration with CA, sporadic adult-onset ataxia, cerebellar stroke, CACNA1A mutations, DBN syndrome; no patient with MS included).e7 On an individual basis, 25 patients showed an improvement of gait performance. Treatment with 5 mg 4-AP enhanced the preferred walking speed and decreased the coefficient of variation of stride time (both measures are linked to better dynamic stability during walking and are associated with a higher risk of falls). The effect of 4-AP on gait was independent of the severity of ataxia as assessed in the Scale for Assessment and Rating of Ataxia but was associated with a high magnitude of temporal gait variability prior to treatments. Thus, temporal gait variability might serve as a prognostic factor for the response to 4-AP in CA.e7 A randomized, placebo-controlled monocentric study with Fampyra (FACEG) is currently being performed to further evaluate the effect of 4-AP-SR on gait stability, walking performance, and falls in patients with different forms of ataxia. As mentioned above, 4-AP improves the precision of the intrinsic pacemaker functions of PCs.6 A higher precision of this function might result in a higher regularity of stepping in humans, but this is speculative.
Table 3.
Clinical trials and case series with aminopyridines in gait ataxia due to different etiologies
Lambert-Eaton myasthenic syndrome
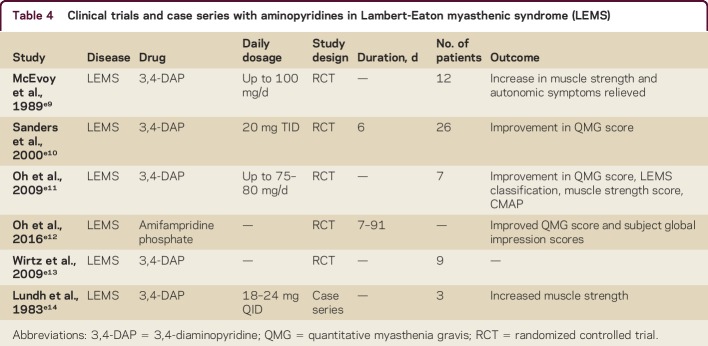
3,4-DAP is an effective drug in LEMS as well as congenital myasthenic syndrome (CMS) (table 4).e10–e13,e15 LEMS was first described in 1956 and is a rare neuromuscular autoimmune disorder. A total of 50%–60% of the cases are paraneoplastic and associated with a tumor, which is almost always small-cell lung carcinoma (SCLC)e16; the remaining are nontumor LEMS. Because of the high incidence of SCLC and manifestation of LEMS before detection of SCLC, tumor screening is crucial and causal treatment has to be performed accordingly. Clinical features of LEMS are proximal muscle weakness, autonomic symptoms, and reduced tendon reflexes. Clinical symptoms are caused by autoantibodies to P/Q-type voltage-gated calcium channels (VGCC) on the presynaptic nerve terminal.e17,e18 VGCC autoantibodies can be found in about 85%–90% of patients with LEMS (in 60%–70% of them, SCLC is detected).e19 However, in these patients, a SCLC is much less likely to be diagnosed.e20 The presence of VGCC antibodies in the neuromuscular junction leads to a reduced Ca2+ influx during depolarization and therefore a decreased acetylcholine (ACh) vesicle release from the presynaptic membrane.e16 In 1983, the drug 3,4-DAP was introduced as an innovative symptomatic treatment for LEMS.e14 3,4-DAP blocks potassium channels and prolongs the duration of depolarization, subsequently leading to increased ACh release.3 Since then, 4 studies have confirmed its efficacy and safety.e9–e11,e13,e21 Two were based on clinical endpoints (quantitative muscle strength, quantitative myasthenia gravis [QMG] score, LEMS classification, muscle strength scoree10,e11) and all 4 proved the efficacy on electrophysiologic measurements (including repetitive nerve stimulation test, single-fiber EMG, and amplitudes of compound muscle action potentials). Side effects consisted of paresthesias, gastrointestinal symptoms, and an epileptic seizure in one patient.e9–e11,e13 3,4-DAP should be the drug of first choice in LEMS, starting with 4 × 10 mg/d (up to 5 × 20 mg/d). In 2010, the 3,4-DAP phosphate salt amifampridine (Firdapse, 10 mg tablets, BioMarin, San Rafael, CA) was given market authorization for LEMS by the European Medicines Agency under European drug legislation. The approval was given based on previous trials on the base form of 3,4-DAP. In the United States, the latter has been available through compounding pharmacies and from Jacobus Pharmaceutical Co., Inc. (Princeton, NJ), under a compassionate use Investigational New Drug program. Recently, a phase 3 clinical trial showed that amifampridine (Firdapse) is efficacious in the symptomatic treatment of LEMS (QMG score). Furthermore, a second study on 3,4-DAP in LEMS has been completed but has not yet been published (for further information, see reference e12 at http://cp.neurology.org/lookup/doi/10.1212/CPJ.0000000000000321). There is also one study about expanded access in amifampridine in LEMS, CMS, and DBN (NCT02189720) and one about amifampridine in pediatric CMS (NCT02562066). Recently, numerous physicians brought to public attention their concern about the potential cost increase of 3,4-DAP after submission for US FDA approval under the Orphan Drug Act by Jacobus Pharmaceutical Co., Inc. and Catalyst Pharmaceuticals, Inc. (Coral Gables, FL).e12
Table 4.
Clinical trials and case series with aminopyridines in Lambert-Eaton myasthenic syndrome (LEMS)
Side effects, contraindications, and precautions of 3,4-DAP and 4-AP
Regarding 3,4-DAP (Firdapse), precautions must be taken if the patient has a history of asthma, convulsions, or kidney or liver problems. Drugs that may prolong the QT interval must not be combined with 3,4-DAP. Side effects commonly include tingling and numbness around the mouth and extremities, reduced sense of touch or sensation, nausea, dizziness, increased sweating, stomachache, and cold extremities. Contraindications are hypersensitivity to the active substance, uncontrolled asthma, congenital QT syndromes, and concomitant use with various drugs that prolong QTc (for further information, see reference e22). Precautions for 4-AP (Fampyra) are similar. Treatment with this drug increases seizure risk and renal impairment has to be checked. It should not be administered to patients with a creatinine clearance <80 mL/min. Furthermore, caution is necessary when it is administered in combination with drugs that are OCT2 substrates such as carvedilol, propranolol, and metformin, as well as in patients with a history of rhythm disorders. The most common reported side effect is urinary tract infection (for further information, see reference e23).
Other possible indications
Acute vertigo/acute unilateral vestibulopathy
There are currently few animal studies on the effects of pharmacologic agents on central compensation (overview in reference e24). An in vivo study in a rat model of acute unilateral vestibulopathy showed improvement of postural symptoms with 4-AP but on the other hand impairment of the course of vestibular compensation, underlining the hypothesis that symptomatic treatment in acute unilateral vestibulopathy should only be given for a short time period and only if neccessary.e25 These findings are the basis for an observational clinical study on the symptomatic effects of 4-AP in acute vertigo.e25
Lower urinary tract symptoms
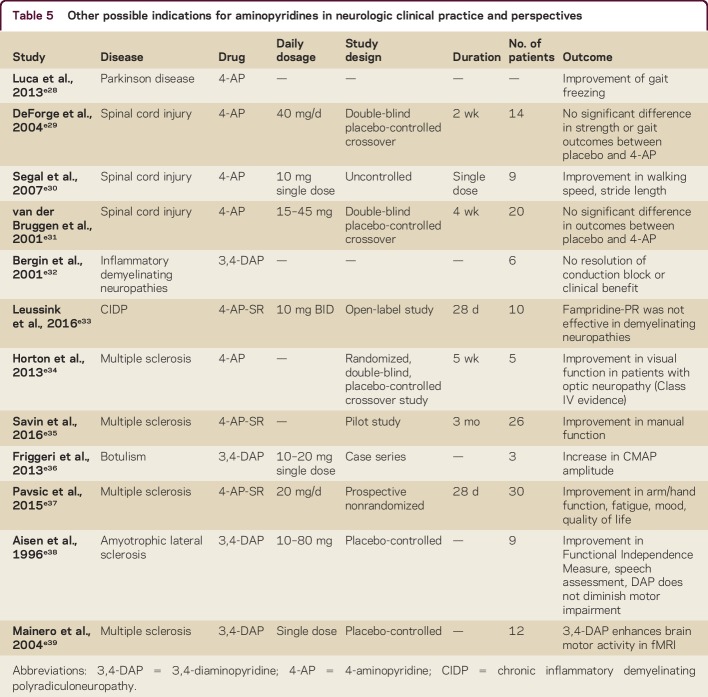
The efficacy of 4-AP-SR has been demonstrated in a single case of a patient with DBN and lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia.e26 The reported subjective improvement of LUTS during the 4-AP treatment was monitored using uroflowmetry.e26 In this case, it is assumed that 4-AP can increase the excitability of both the sympathetic and the parasympathetic branches of the autonomic nervous systems and an increased activity of these nerves during voiding can also improve bladder function.e26,e27 For further information and indications, see table 5.
Table 5.
Other possible indications for aminopyridines in neurologic clinical practice and perspectives
Recommendations
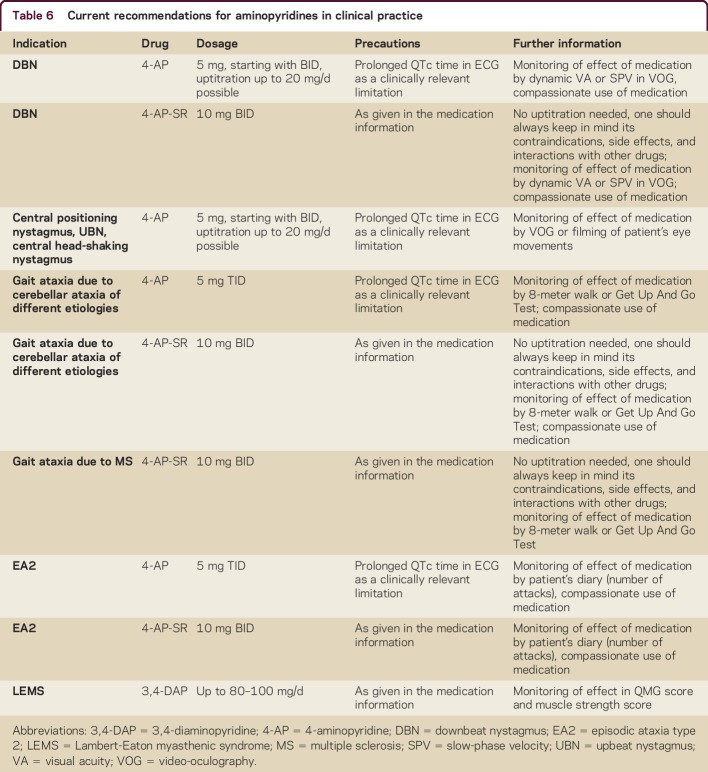
Based on the given data, recommendations for treatment in neurologic diseases are given in table 6.
Table 6.
Current recommendations for aminopyridines in clinical practice
Perspectives and ongoing trials
Further controlled trials are needed, as the results of experimental and clinical studies on 4-AP support the importance of this substance for the treatment of vestibular, cerebellar, and ocular motor disorders, and for back-translational research to further elucidate its mode of action in animal models.
EA2TREAT
There is currently an ongoing placebo-controlled trial of 4-AP-SR and acetazolamide compared with placebo (University of Munich).
FACEG
There are currently 2 ongoing placebo-controlled trials of 4-AP-SR for cerebellar gait disorders (University of Florida [NCT01811706] and University of Munich).
Use of 4-AP in acute vertigo
Based on the pronounced effects of 4-AP in an animal model of acute unilateral vestibulopathy, a clinical trial in humans with this disease seems to be justified to evaluate its symptomatic effects. However, duration of treatment should be limited to 3 days to avoid a delay of central compensation.
Neuroprotective effect
Due to the convincing findings in animal models, long-term studies in humans with neurodegenerative cerebellar disorders are justified.
Mode of action
Since there is debate about the mode of action of AP in cerebellar disorders, further basic research is necessary in different animal models of cerebellar disorders.
AUTHOR CONTRIBUTIONS
M. Strupp and K. Feil: drafting and revising the manuscript for intellectual content, including medical writing for content. J. Teufel, A. Zwergal, R. Schniepp, and K. Khodakhah: revising the manuscript for intellectual content, including medical writing for content (review, critique, revising).
STUDY FUNDING
Part of the presented work was supported by the Federal Ministry for Education and Science (BMBF, no. 80121000-49) and the German Hertie Foundation. In addition, parts were supported by a grant from the BMBF to the IFB (grant code 01 EO 0901).
DISCLOSURES
M. Strupp serves on scientific advisory boards for Abbott, Actelion, Sensorion, Heel, IntraBio, and Pierre-Fabre; has received speaker honoraria from Abbott, GlaxoSmithKline, Merck Serono, HENNIG ARZNEIMITTEL GmbH & Co. KG, Pierre Fabre Laboratories, UCB, TEVA, Heel, Biogen Idec, MSD, and Eisai Inc.; serves as Joint-Chief Editor of the Journal of Neurology, Editor-in-Chief of Frontiers in Neuro-otology, and Section Editor of F1000; and receives publishing royalties for Leitsymptom Schwindel (Springer, 2012) and Vertigo and Dizziness: Common Complaints (Springer, 2013). J. Teufel reports no disclosures. A. Zwergal has received speaker honoraria from Schwabe Pharma GmbH, Actelion Pharmaceuticals GmbH, Bayer Vital GmbH, and the German Federal Ministry of Education and Health (BMBF) in connection with the foundation of the German Center for Vertigo and Balance Disorders (DSGZ). R. Schniepp reports no disclosures. K. Khodakhah serves on a scientific advisory board for Ataxion and receives research support from Ataxion and NIH. K. Feil reports no disclosures. Full disclosure form information provided by the authors is available with the full text of this article athttp://cp.neurology.org/lookup/doi/10.1212/CPJ.0000000000000321.
Supplementary Material
ACKNOWLEDGMENT
The authors thank Katie Ogston for copyediting the manuscript.
Footnotes
Supplemental data at http://cp.neurology.org/lookup/doi/10.1212/CPJ.0000000000000321

REFERENCES
- 1.Hille B. Ionic Channels of Excitable Membranes, 3rd ed Sunderlang: Sinauer; 2001. [Google Scholar]
- 2.Sherratt RM, Bostock H, Sears TA. Effects of 4-aminopyridine on normal and demyelinated mammalian nerve fibres. Nature 1980;283:570–572. [DOI] [PubMed] [Google Scholar]
- 3.Molgo J, Lundh H, Thesleff S. Potency of 3,4-diaminopyridine and 4-aminopyridine on mammalian neuromuscular transmission and the effect of pH changes. Eur J Pharmacol 1980;61:25–34. [DOI] [PubMed] [Google Scholar]
- 4.Bostock H, Sears TA, Sherratt RM. The effects of 4-aminopyridine and tetraethylammonium ions on normal and demyelinated mammalian nerve fibres. J Physiol 1981;313:301–315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Etzion Y, Grossman Y. Highly 4-aminopyridine sensitive delayed rectifier current modulates the excitability of guinea pig cerebellar Purkinje cells. Exp Brain Res 2001;139:419–425. [DOI] [PubMed] [Google Scholar]
- 6.Alvina K, Khodakhah K. The therapeutic mode of action of 4-aminopyridine in cerebellar ataxia. J Neurosci 2010;30:7258–7268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weisz CJ, Raike RS, Soria-Jasso LE, Hess EJ. Potassium channel blockers inhibit the triggers of attacks in the calcium channel mouse mutant tottering. J Neurosci 2005;25:4141–4145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hourez R, Servais L, Orduz D, et al. . Aminopyridines correct early dysfunction and delay neurodegeneration in a mouse model of spinocerebellar ataxia type 1. J Neurosci 2011;31:11795–11807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shaikh AG, Marti S, Tarnutzer AA, et al. . Effects of 4-aminopyridine on nystagmus and vestibulo-ocular reflex in ataxia-telangiectasia. J Neurol 2013;260:2728–2735. [DOI] [PubMed] [Google Scholar]
- 10.Abbasi S, Abbasi A, Sarbaz Y. Introducing treatment strategy for cerebellar ataxia in mutant med mice: combination of acetazolamide and 4-aminopyridine. Comput Methods Programs Biomed 2014;113:697–704. [DOI] [PubMed] [Google Scholar]
- 11.Hayes KC, Katz MA, Devane JG, et al. . Pharmacokinetics of an immediate-release oral formulation of Fampridine (4-aminopyridine) in normal subjects and patients with spinal cord injury. J Clin Pharmacol 2003;43:379–385. [DOI] [PubMed] [Google Scholar]
- 12.Wagner JN, Glaser M, Brandt T, Strupp M. Downbeat nystagmus: aetiology and comorbidity in 117 patients. J Neurol Neurosurg Psychiatry 2008;79:672–677. [DOI] [PubMed] [Google Scholar]
- 13.Kalla R, Deutschlander A, Hufner K, et al. . Detection of floccular hypometabolism in downbeat nystagmus by fMRI. Neurology 2006;66:281–283. [DOI] [PubMed] [Google Scholar]
- 14.Zee DS, Yamazaki A, Butler PH, Gucer G. Effects of ablation of flocculus and paraflocculus of eye movements in primate. J Neurophysiol 1981;46:878–899. [DOI] [PubMed] [Google Scholar]
- 15.Leigh RJ, Zee DS, editors. The Neurology of Eye Movements, 5th ed Oxford: Oxford UP; 2015. [Google Scholar]
- 16.Strupp M, Schuler O, Krafczyk S, et al. . Treatment of downbeat nystagmus with 3,4-diaminopyridine: a placebo-controlled study. Neurology 2003;61:165–170. [DOI] [PubMed] [Google Scholar]
- 17.Helmchen C, Gottschalk S, Sander T, Trillenberg P, Rambold H, Sprenger A. Beneficial effects of 3,4-diaminopyridine on positioning downbeat nystagmus in a circumscribed uvulo-nodular lesion. J Neurol 2007;254:1126–1128. [DOI] [PubMed] [Google Scholar]
- 18.Sprenger A, Zils E, Rambold H, Sander T, Helmchen C. Effect of 3,4-diaminopyridine on the postural control in patients with downbeat nystagmus. Ann NY Acad Sci 2005;1039:395–403. [DOI] [PubMed] [Google Scholar]
- 19.Tsunemi T, Ishikawa K, Tsukui K, Sumi T, Kitamura K, Mizusawa H. The effect of 3,4-diaminopyridine on the patients with hereditary pure cerebellar ataxia. J Neurol Sci 2010;292:81–84. [DOI] [PubMed] [Google Scholar]
- 20.Kalla R, Glasauer S, Buttner U, Brandt T, Strupp M. 4-aminopyridine restores vertical and horizontal neural integrator function in downbeat nystagmus. Brain 2007;130:2441–2451. [DOI] [PubMed] [Google Scholar]
- 21.Kalla R, Glasauer S, Schautzer F, et al. . 4-aminopyridine improves downbeat nystagmus, smooth pursuit, and VOR gain. Neurology 2004;62:1228–1229. [DOI] [PubMed] [Google Scholar]
- 22.Judge SI, Bever CT Jr. Potassium channel blockers in multiple sclerosis: neuronal Kv channels and effects of symptomatic treatment. Pharmacol Ther 2006;111:224–259. [DOI] [PubMed] [Google Scholar]
- 23.Kalla R, Spiegel R, Claassen J, et al. . Comparison of 10-mg doses of 4-aminopyridine and 3,4-diaminopyridine for the treatment of downbeat nystagmus. J Neuroophthalmol 2011;31:320–325. [DOI] [PubMed] [Google Scholar]
- 24.Claassen J, Spiegel R, Kalla R, et al. . A randomised double-blind, cross-over trial of 4-aminopyridine for downbeat nystagmus: effects on slowphase eye velocity, postural stability, locomotion and symptoms. J Neurol Neurosurg Psychiatry 2013;84:1392–1399. [DOI] [PubMed] [Google Scholar]
- 25.Claassen J, Feil K, Bardins S, et al. . Dalfampridine in patients with downbeat nystagmus: an observational study. J Neurol 2013;260:1992–1996. [DOI] [PubMed] [Google Scholar]
- 26.Ilg W, Bastian AJ, Boesch S, et al. . Consensus paper: management of degenerative cerebellar disorders. Cerebellum 2014;13:248–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Feil K, Bremova T, Muth C, Schniepp R, Teufel J, Strupp M. Update on the pharmacotherapy of cerebellar ataxia and nystagmus. Cerebellum 2016;15:38–42. [DOI] [PubMed] [Google Scholar]
- 28.Glasauer S, Kalla R, Buttner U, Strupp M, Brandt T. 4-aminopyridine restores visual ocular motor function in upbeat nystagmus. J Neurol Neurosurg Psychiatry 2005;76:451–453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kremmyda O, Zwergal A, la Fougere C, Brandt T, Jahn K, Strupp M. 4-Aminopyridine suppresses positional nystagmus caused by cerebellar vermis lesion. J Neurol 2013;260:321–323. [DOI] [PubMed] [Google Scholar]
- 30.Strupp MQV, Eggert T, Brandt T. Potassium channel blocker 3,4-diaminopyridine improves severe head-shaking nystagmus. J Vestib Res 2002;11:263. [Google Scholar]
- 31.Jen JC. Hereditary episodic ataxias. Ann NY Acad Sci 2008;1142:250–253. [DOI] [PubMed] [Google Scholar]
- 32.Jen JC, Baloh RW. Familial episodic ataxia: a model for migrainous vertigo. Ann NY Acad Sci 2009;1164:252–256. [DOI] [PubMed] [Google Scholar]
- 33.Ophoff RA, Terwindt GM, Vergouwe MN, et al. . Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell 1996;87:543–552. [DOI] [PubMed] [Google Scholar]
- 34.Strupp M, Zwergal A, Brandt T. Episodic ataxia type 2. Neurotherapeutics 2007;4:267–273. [DOI] [PubMed] [Google Scholar]
- 35.Strupp M, Kalla R, Dichgans M, Freilinger T, Glasauer S, Brandt T. Treatment of episodic ataxia type 2 with the potassium channel blocker 4-aminopyridine. Neurology 2004;62:1623–1625. [DOI] [PubMed] [Google Scholar]
- 36.Strupp M, Kalla R, Claassen J, et al. . A randomized trial of 4-aminopyridine in EA2 and related familial episodic ataxias. Neurology 2011;77:269–275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Stahl JS, Thumser ZC. 4-aminopyridine does not enhance flocculus function in tottering, a mouse model of vestibulocerebellar dysfunction and ataxia. PLoS One 2013;8:e57895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Glasauer S, Rossert C, Strupp M. The role of regularity and synchrony of cerebellar Purkinje cells for pathological nystagmus. Ann NY Acad Sci 2011;1233:162–167. [DOI] [PubMed] [Google Scholar]
- 39.Claassen J, Teufel J, Kalla R, Spiegel R, Strupp M. Effects of dalfampridine on attacks in patients with episodic ataxia type 2: an observational study. J Neurol 2013;260:668–669. [DOI] [PubMed] [Google Scholar]
- 40.Goodman AD, Cohen JA, Cross A, et al. . Fampridine-SR in multiple sclerosis: a randomized, double-blind, placebo-controlled, dose-ranging study. Mult Scler 2007;13:357–368. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.