Introduction
Combinations of psychoactive treatments are frequently used to treat impulsivity, aggressiveness, or anxiety in patients with resistant schizophrenia.
Antipsychotics, as recommended by the French authorities [Haute Autorité de Santé, 2007], can be co-prescribed with antiepileptic agents in order to reduce impulsivity, to stabilize mood or to treat epilepsy. The prevalence of psychotic disorders in epileptic patients is significantly higher compared with the general population [Clancy et al. 2014].
Clozapine (CLZ; 8-chloro-11-(4-methyl-1-piperazinyl)-5H-dibenzo[b,e][1,4]-diazepine) is an atypical antipsychotic indicated for resistant schizophrenia or in cases of intolerance to the other antipsychotic agents. CLZ has a complex pharmacological profile because of its binding properties. It presents an antagonist action on serotoninergic and dopaminergic systems (5HT2A and D4) and, to a lesser extent, an antagonist activity on histaminergic (H1), cholinergic-like muscarinic (M3 and M1) and adrenergic (α1) systems [Stahl, 2015].
CLZ is recommended as third-line treatment because of the side effects in 25.2% of patients [Mustafa et al. 2015].
The coadministration of treatments may induce the modulation of the pharmacokinetics of CLZ via the enzyme inhibition or induction of cytochrome P450 (CYP450). CLZ is metabolized mainly in N-desmethylclozapine (NCLZ or norclozapine) and CLZ N-oxide by the isoenzymes of CYP450: CYP1A2, CYP3A4 and to a lesser extent by CYP2D6, CYP2C19 and CYP2C9 [Kennedy et al. 2013; Rouleau et al. 2008]. As documented, NCLZ is known to present anticholinergic and hypersalivation effects [de Leon et al. 2012]. Uridine 5′-diphospho-glucuronosyltransferase (UGT) catalyses glucuronidation reactions of NCLZ facilitating its elimination [Rouleau et al. 2008].
A correlation between plasma concentrations of CLZ, NCLZ and the occurrence of side effects have been previously reported [Llorca and Pere, 2004]. This led to an evaluation of the factors of variability in plasma concentration of CLZ and its active metabolites. Plasma levels are also indicative of the quality of compliance. The relative rate of CLZ/NCLZ is also useful to estimate the quality of the observance with CLZ [Couchman et al. 2010].
Oxcarbazepine (OXC; 10, 11-dihydro-10-oxo-5H-dibenzo[b, f]azepine-5-carboxamide) is developed from structural rearrangements of carbamazepine (CBZ) [Schmidt and Elger, 2004] in order to minimize the involvement of isoenzymes of CYP450 in OXC metabolism. This is a choice anti-epileptic treatment, anti-impulsive and mood stabilizer as with CBZ [Stahl, 2015]. OXC is indicated in the treatment of partial seizures with or without secondary generalization. After absorption, an important proportion of OXC undergoes rapid reduction in a pharmacologically active metabolite, monohydroxy derivative (MHD; 10, 11-dihydro-10-hydroxy-5H-dibenzo[f,b]azepine-5-carboxamide or licarbazepine) while a minority of MHD is oxidized in pharmacologically inactive dihydroxy derivative (DHD; carbamazepine-10,11-dihydro-10,11-dihydroxy) [Flesch, 2004]. In comparison with CBZ which is significantly inductive of CYP3A4 (about 46% higher than that of OXC), OXC is known to present a less pronounced dose-dependent effect [Andreasen et al. 2007]. The induction effect is more effective at high doses (900–1200 mg/day). In fact, OXC weakly induced CYP3A4 and UGT in vitro [Patsalos, 2013a,2013b], as documented by a decrease in plasma concentrations of oral contraceptives, felodipine, imatinib and lamotrigine. To our knowledge, there is no report of a putative interaction between CLZ and OXC.
In this paper, we report the case of important variations of CLZ and NCLZ concentrations after OXC introduction. This raises the hypothesis of an interaction between CLZ and OXC in a schizophrenic patient for whom we performed a therapeutic drug monitoring of plasma concentration of CLZ and NCLZ, before, during and after treatment of OXC.
Case report
We report the case of a 29-year-old White male inpatient with schizophrenia associated with antisocial personality and hospitalization for 5 years after serious aggressive behavior. The patient was hospitalized in an institution that does not require ethics approval for reporting individual cases nor the patient’s written consent. This latter point is acceptable due to the fact that the patient’s personal information was completely restricted to maintain the confidentiality of his identity in this report. The patient reported having a personal history of addiction to cannabis, weaned for 9 years. He was treated with CLZ 900 mg/day for 5 years at the same dosage and also treated with paliperidone at 100 mg/28 days (over 1 year), macrogol at 2 sachets/day, phloroglucinol at 1 tablet as needed, uvesterol at 1 bulb/day, and Uvedose (vitamin D3-based cholecalciferol) at 1 bulb/3 months. Each of these treatments were prescribed for more than 1 year. During hospitalization, the patient presented with a seizure characterized by lower limb clonus followed by falls, deviations of gaze and consciousness disorder. OXC at a dosage of 750 mg/day (period B) has been introduced after the diagnosis of partial epilepsy with unknown etiology. Partial improvement in neurologic symptoms has been observed (persistence of only slight clonus) associated with an increase of dissociative symptoms such as listening attitudes and thoughts disorders. These symptoms were consistent with those usually observed in this patient during psychotic exacerbation.
Weekly and biweekly blood samples were drawn by a nurse in the psychiatric unit in which the patient was hospitalized. They were collected in two tubes without gel or preservative agent at 12 h post-dose (in the morning at 8 am for a treatment taken at 8 pm). CLZ doses were administrated in the presence of the nurse who verified the adherence each time.
The sample was performed by high-performance liquid chromatography after liquid/liquid extraction and detection in ultraviolet light [Rosland et al. 2007].
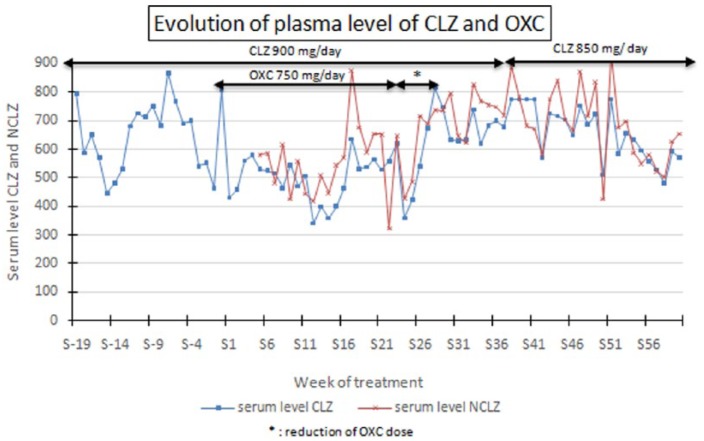
Figure 1 illustrates the evolution of concentrations of CLZ and NCLZ over a period starting 20 weeks before the introduction of OXC (period A) and extending to 5 months following the withdraw of antiepileptic therapy (period C and D). Before the OXC introduction (period A) the plasma concentrations of CLZ were at the concentrations usually recommended, however, closer to the upper limit with a mean (± standard deviation) concentration of 650 μg/l (± 122 μg/l) over a period of 20 weeks prior to the introduction of OXC (Table 1).
Figure 1.
Progress of drug doses and plasma levels of CLZ and NCLZ before OXC introduction, when receiving simultaneous treatment with CLZ and OXC, after OXC withdrawal.
CLZ, clozapine; NCLZ, N-desmethylclozapine; OXC, oxcarbazepine.
Table 1.
Dose of CLZ and OXC and average plasma concentrations of CLZ and NCLZ.
| Treatment prescribed | CLZ dose (mg/day) | OXC dose (mg/day) | Average plasma concentration (µg/l) (5th percentile–95th percentile) |
||
|---|---|---|---|---|---|
| CLZ | Norclozapine | ||||
| Period A | CLZ alone | 900 | 0 | 650.05 (410.57; 889.52) | – |
| Period B | CLZ + OXC | 900 | 750 | 492.08 (333.45; 650.70) | 547.9 (303.37; 787.48) |
| Period C | CLZ alone | 900 | 0 | 667.5 (584.07; 754.20) | 726.71 (636.69; 866.73) |
| Period D | CLZ alone | 850 | 0 | 643.87 (476.40; 811.33) | 685.34 (518.30; 852.39) |
CLZ, clozapine; NCLZ, N-desmethylclozapine; OXC, oxcarbazepine.
The plasma concentrations of CLZ, calculated during the period of association with OXC at 750 mg/day (period B) using a Student’s t- test with a significance level of p < 0.05 were significantly lower (p < 0.001) with a mean CLZ concentration of 492 μg/l (± 80 μg/l).
At the same time the average concentration (± standard deviation) of NCLZ was 547 μg/l (± 120 μg/l) (Table 1). Clonazepam, for which no interaction with the CLZ or OXC was reported [Wisher, 2011] was introduced with a dose of 30 drops/d at the 18th week (S18) to reinforce the antiepileptic effect of OXC, then gradually decreased over 6 months.
During the period of OXC dose reduction and after its withdrawal (period C), an increase in CLZ and NCLZ concentrations has been found leading to new levels significantly different (p < 0.001) in comparison with values obtained from the period when the two treatments were co-administered: respectively to 669 μg/l (± 43 μg/l) for CLZ and 726 μg/l (± 71 μg/l) for NCLZ (Figure 1). Blood levels could reach 738 μg/l for CLZ and 827 μg/l for NCLZ in the 33th week (S33).
The decrease of dosage of CLZ, from 900 mg/day to 850 mg/day (period D) leads logically to a decrease in mean concentrations: 657 μg/l (± 95 μg/l) for CLZ and 685 μg/l (± 132 μg/l) for NCLZ and moreover a decrease of neurological symptoms was observed.
At the beginning of the observation only CLZ concentrations are mentioned, those of NCLZ were not measured by the laboratory during this period. The calculation of the ratio CLZ/NCLZ was always higher than 0.5 which suggested a good adherence.
Discussion
CLZ is considered to be a risk factor for epilepsy, because it reduces the seizure threshold. It is also sometimes necessary to prescribe CLZ for inpatients with risk factors or a history of seizure. Despite such risk, some patients may require prescription CLZ at effective doses, thus requiring the co-prescription of an antiepileptic treatment. This case suggests for the first time an interaction between OXC and CLZ. There were significant differences in mean plasma concentrations of CLZ and NCLZ between the three periods (A, B and C). These difference are characterized by a decrease in concentrations of CLZ and NCLZ when OXC was prescribed concomitantly. The hypothesis, as suggested in the literature [Bang and Goa, 2004], is an induction of OXC on CYP450 isoenzymes and UGT [Patsalos, 2013a, 2013b] but this induction is known to be more evident at high doses (900–1200 mg/day). Another study [Muscatello et al. 2005] confirms the necessity of a high dose of OXC and shows that coadministration of oxcarbazepine at 900–1200 mg/day with risperidone or olanzapine, causes minimal and no significant changes in steady-state plasma concentrations of risperidone (and its metabolite) and olanzapine. In this case, the patient received a dose of 750 mg/day OXC. To date, a significant induction at lower dosages of OXC has not been observed. But in this case OXC, via its modest action on CYP3A4, may have increased the catabolism of CLZ resulting in decreased plasma concentrations of the parent compound. OXC may have also induced UGT, favoring the elimination of NCLZ, as evidenced by the decrease of NCLZ concentrations. Both these effects on CLZ and NCLZ lead to a ratio of CLZ/NCLZ that is constant over time. This induction phenomenon is highlighted about 2 weeks after the therapeutic change. The return to the previous state after stopping OXC treatment also took a few weeks.
A limitation is the on-time occurrences of important differences in concentration of CLZ and NCLZ even if statistically significant differences have been noticed between co-prescription period and withdrawal OXC period. However, neither the inter-assay reproducibility of the analytical method, nor the rigorous taking of treatments (at a fixed time and under supervision limiting noncompliance) seemed to induce such variability. Another limitation of this clinical case could be the existence of a bias to other pharmacological or toxic interactions. However, this patient was constantly hospitalized, was nonsmoking and the other treatments were perfectly stable over a long period before the introduction of OXC. In this patient there were, a priori, no other factors already described in the literature that may have affected the pharmacokinetics of CLZ but the influence of an inducing factor cannot be excluded [Rouleau et al. 2008].
Conclusion
In the absence of historical data, it is important that other studies affirm or refute the hypothesis of an interaction between the OXC and CLZ raised by our clinical observation. The possibility of the existence of an inducing effect of OXC on CLZ led us to recommend the monitoring of CLZ concentrations when associated with OXC in clinical setting, because of the risk of a reduced effectiveness of the antipsychotic treatment.
Acknowledgments
The authors are grateful to Pr Parienti JJ for advice and Mrs Cadix for translation assistance.
Footnotes
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Contributor Information
Hazel Yousra, Department of Psychiatry, Esquirol Center, University Hospital of Caen, Caen, France.
Lebain Pierrick, Department of Psychiatry, Esquirol Center, University Hospital of Caen, 14000 Caen, France.
Lecardeur Laurent, Department of Psychiatry, Esquirol Center, University Hospital of Caen, Caen, France.
Debruyne Danièle, Department of Pharmacology and Toxicology, University Hospital of Caen, Caen, France.
References
- Andreasen A., Brøsen K., Damkier P. (2007) A comparative pharmacokinetic study in healthy volunteers of the effect of carbamazepine and oxcarbazepine on cyp3a4. Epilepsia 48: 490–496. [DOI] [PubMed] [Google Scholar]
- Bang L., Goa K. (2004) Spotlight on oxcarbazepine in epilepsy. CNS Drugs 18: 57–61. [DOI] [PubMed] [Google Scholar]
- Clancy M., Clarke M., Connor D., Cannon M., Cotter D. (2014) The prevalence of psychosis in epilepsy; a systematic review and meta-analysis. BMC Psychiatry 14: 75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couchman L., Morgan P., Spencer E., Flanagan R. (2010) Plasma clozapine, norclozapine, and the clozapine:norclozapine ratio in relation to prescribed dose and other factors: data from a therapeutic drug monitoring service, 1993–2007. Ther Drug Moni 32: 438–447. [DOI] [PubMed] [Google Scholar]
- de Leon J., Santoro V., D’Arrigo C., Spina E. (2012) Interactions between antiepileptics and second-generation antipsychotics. Expert Opin Drug Metab Toxicol 8: 311–334. [DOI] [PubMed] [Google Scholar]
- Flesch G. (2004) Overview of the clinical pharmacokinetics of oxcarbazepine. Clin Drug Investig 24: 185–203. [DOI] [PubMed] [Google Scholar]
- Haute Autorite de Sante (2007) Guide, Affection Longue Duréee, Schizophrenies. Available at: www.has-sante.fr
- Kennedy W., Jann M., Kutscher E. (2013) Clinically significant drug interactions with atypical antipsychotics. CNS Drugs 27: 1021–1048. [DOI] [PubMed] [Google Scholar]
- Llorca P., Pere J. (2004) Leponex®, 10 ans après–Une revue clinique. L’Encéphale 30: 474–491. [DOI] [PubMed] [Google Scholar]
- Muscatello R., Pacetti M., Cacciola M., La Torre D., Zoccali R., D’Arrigo C., et al. (2005) Plasma concentrations of risperidone and olanzapine during coadministration with oxcarbazepine. Epilepsia 46: 771–774. [DOI] [PubMed] [Google Scholar]
- Mustafa F., Burke J., Abukmeil S., Scanlon J., Cox M. (2015) ‘schizophrenia past clozapine’: reasons for clozapine discontinuation, mortality, and alternative antipsychotic prescribing. Pharmacopsychiatry 48: 11–14. [DOI] [PubMed] [Google Scholar]
- Patsalos P. (2013a) Drug interactions with the newer antiepileptic drugs (AEDs)–Part 1: pharmacokinetic and pharmacodynamic interactions between AEDs. Clin Pharmacokinet 52: 927–966. [DOI] [PubMed] [Google Scholar]
- Patsalos P. (2013b) Drug interactions with the newer antiepileptic drugs (AEDs)–Part 2: pharmacokinetic and pharmacodynamic interactions between AEDs and drugs used to treat non-epilepsy disorders. Clin Pharmacokinet 52: 1045–1061. [DOI] [PubMed] [Google Scholar]
- Rosland M., Szeto P., Procyshyn R., Barr A., Wasan K. (2007) Determination of clozapine and its metabolite, norclozapine in various biological matrices using high-performance liquid chromatography. Drug Dev Ind Pharm 33: 1158–1166. [DOI] [PubMed] [Google Scholar]
- Rouleau B., Vincent P., Martel J. (2008) Le monitorage thérapeutique de la clozapine: une nécessité clinique. Pharmactuel: 87–93. [Google Scholar]
- Schmidt D., Elger C. (2004) what is the evidence that oxcarbazepine and carbamazepine are distinctly different antiepileptic drugs? Epilepsy Behav 5: 627–635. [DOI] [PubMed] [Google Scholar]
- Stahl S. (2015) Psychopharmacologie Essentielle, 4° éd. Librairie Lavoisier. [Google Scholar]
- Wisher D. (2011) Stockley’s drug interactions. 9th ed. and Stockley’s drug interactions 2010 pocket companion. JMLA 99: 174–175. [Google Scholar]