Graphical abstract
Keywords: Hydrogels, Membranes, Wound dressings, Skin substituent, Biomedical applications
Abstract
This review presents the past and current efforts with a brief description on the featured properties of hydrogel membranes fabricated from biopolymers and synthetic ones for wound dressing applications. Many endeavors have been exerted during past ten years for developing new artificial polymeric membranes, which fulfill the demanded conditions for the treatment of skin wounds. This review mainly focuses on representing specifications of ideal polymeric wound dressing membranes, such as crosslinked hydrogels compatible with wound dressing purposes. But as the hydrogels with single component have low mechanical strength, recent trends have offered composite or hybrid hydrogel membranes to achieve the typical wound dressing requirements.
Introduction
Hydrogels are vastly hydrophilic macromolecular networks, which are produced by chemical or physical crosslinking of soluble polymers. Due to peculiar properties of hydrogels such as, high-sensitive to physiological environments, hydrophilic nature, soft tissue-like water content and adequate flexibility, make them excellent candidates for biomedical applications. Hydrogels can swell and de-swell water in a reversible direction, showing specific environmental stimuli-responsive e.g. temperature, pH, and ionic strength. Thus, such smart physiological response of hydrogels toward changes of physiological variable suggests their use in several biomedical applications [1]. Hydrogels have been introduced for the first time as crosslinked 2-hydroxyethyl methacrylate (HEMA) hydrogels by Wichterle and Lim have been applied in numerous biomedical applications e.g. drug carrier, absorbable sutures, osteoporosis, and as neoplasm owing to their hydrophilic characters [2]. At that time, Lim and Sun [3] have offered calcium-alginate microcapsules for cell engineering and then, Yannas 's group have fabricated synthetic hydrogels composed of natural polymer e.g. collagen to obtain novel dressing materials, showing optimal conditions for healing burns and wound dressing [4]. Subsequently, polymeric hydrogels have much attentions and interests of biomaterials scientists for many years. After revolution of hydrogel appearance which facilitated the production of wound dressings from the passive material to active and functionalized ones (Fig. 1). Earlier to 1960s, polymeric dressings have been considered as just passive materials that have the minimum role in the healing process. Winter [5] has introduced the first generation of polymeric dressings and provided the optimal environments for wound repair. In the mid 1970s, polymeric membranes have been investigated in the development of synthetic dressing materials. In 1978, several chitin-based polymeric materials have been used as the earliest wounds covers, owing to their appealing biological activities, showing that polymers are excellent candidates for developing dressing materials [6]. The research and development trend of hydrogels as polymeric transitory dressing membranes are becoming the main commercial target. Thus, some commercial hydrogels have appeared in the markets under brand names e.g. “Geliperm®, Curasol® and Tegagel®” hydrogel dressing materials. These hydrogels were synthesized by chemical crosslinking of acrylamide and methylene-bis-acrylamide including polysaccharides [7], [8].
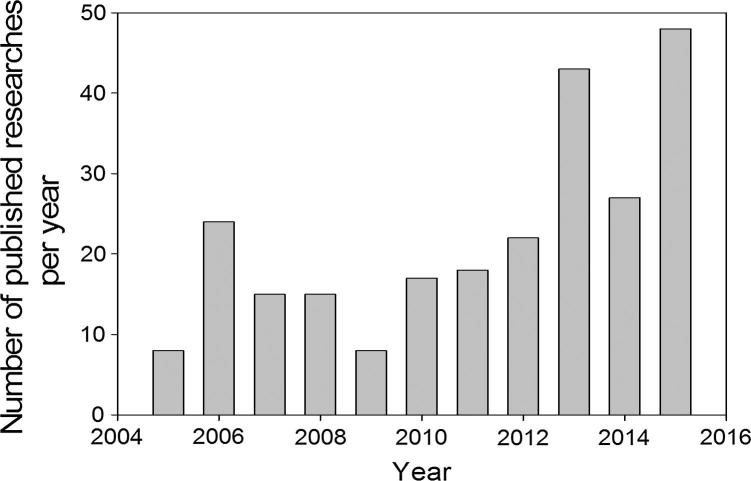
Fig. 1.
Developing of number of published researches in the last ten years, regarding the use of hydrogel membranes as wound dressings.
Sources: PubMed, and Scopus; updated results were taken in Feb. 15, 2016.
According to the first attempts, biomaterial scientists suggested that hydrogels as dressing membranes meet all requirements for wound healing and proper candidate for burn patients in the shortest time possible because of the following justifications: (1) hydrogels control the lost fluids and liquid from the body, (2) maintain the wettability and moist in the wound zone, and (3) they possess tissue-like structure and compatibility. Most polymeric dressing materials e.g. hydrogels, foams, films, hydrocolloids, and alginates have their own aforementioned advantages. However, hydrogels are reported the best choice compared to other dressing forms due to they have the needed requirements for the ideal wound dressings (Table 1). The only disadvantage of hydrogels is their poor mechanical stability at swollen state. This drawback has been addressed by using “composite or hybrid hydrogel membranes” system consisting of more than one polymer in the dressing composition. Hydrogels are usual stabilized by means of chemical or physical interactions among the polymer chains which are known as crosslinking method. After crosslinking hydrogels have ability to retain great absorbed water in their mesh structure. The in situ forming hydrogels via physical crosslinking or radiation-crosslinking, hydrogels are preferred over the chemical crosslinking particularly in biomedical applications. Because of the physical method avoids the presence of crosslinking agents, organic solvents, and chemical reagents overcomes toxicity issue, as compared to the second crosslinking method [9]. Thus, physical crosslinking methods e.g. hydrogen bonding, Van der Waals bonds, or freeze-thawing (F-T) consecutive cycles are the safest crosslinking method for hydrogel formation which are mainly used for wound dressings and in-situ crosslinking cases. Fig. 1 represents a comparison of the annual number of scientific publications distribution around the world based on “polymeric wound dressing materials” which was done using SciFinder Scholar search system in the beginning of 2016. The data analysis in Fig. 1 shows the obvious annual publications number growth with the term “polymeric wound dressings” during last five years, referring to the importance and big attentions in this medical approach. Thus, the annual costs of caring for chronic wounds in USA reached to 25 billion USD. In addition the wound management markets are estimated to raise a value of 4.4 billion USD in 2019 compared to the value of 3.1 billion USD in 2012. Further, statistics reported that between 6 and 15 billion USD was estimated as annual cost for healing of chronic wounds in USA [1]. Accordingly, many significant efforts have been done to mitigate the cost management of wound care approaches [1]. Strangely enough, although polymeric dressing’s progress has shown potential promising and has existed apparently in literatures for several decades, its understanding is still very limited and their publication numbers is a relatively exclusive and scatter. On the light of last contributions, polyvinyl alcohol (PVA)-hybrid hydrogel membranes were considered the most frequently polymeric membrane candidate for wound dressings and covers. This review aims to recollect the most polymers which previously blended with PVA for using as polymeric wound dressings, knowing that these modern dressings are suggested to initiate the healing efficiency and rate of wound instead of just to cover it. Thus, the review focuses on showing the remarkable polymers e.g. bio, synthetic, or composite polymers which were reported in literature as PVA-based hydrogel dressings.
Table 1.
Polymeric wound dressings: Advantages and disadvantages.
| Type of polymeric dressings | Properties | Advantages | Disadvantages |
|---|---|---|---|
| Films | Polyurethanes thin, flexible, transparent and self adhesion films Suitable for: uncontaminated, laser, and superficial wounds |
Good gases-permeability, impermeable to bacteria and fluids, easy wound monitoring through film transparency, and less maceration and painless | Difficult handled, adherence to wound bed, non-absorbent allowing wound exudates accumulation, easy bacterial invasion and infection and impermeable for proteins and drugs |
| Foams | Polyurethane, PEG, and silicone bilaminate layers Suitable for: burns, chronic wounds, cavities wound-shape, and deep ulcers |
High absorbent, keep moist environment, non-leakage against bacterial invasion, very easy used, and costless | Very adherent, forming opaque layer which complicates wound monitoring, semi-permeable for gases, non-applicable for dried wounds, and poor stability |
| Hydrogels | Natural and synthetic polymers, high absorption capacity, and used as soothing and cooling agent for cutaneous wounds. Suitable for: most wounds and burns types |
High exudates capacity, non-adherent, easily removed from wound, accelerate the healing, pain and inflammatory reduction, costless, easily developed and handled | Semi-transparent, semi-permeable to gases and water vapor, poor bacterial barrier, and sometimes poor mechanical stability |
| Alginates | Alginate polymer in woven -fibers form. Ion exchange occurs with Ca-alginate and Na-blood serum Suitable for: surgical wounds and full burns |
High absorbent, non adherent, high mechanical stability, stable for long time, easily removed by saline solution, and good bacterial barrier | Very cost, unpleasant odor, difficult handled and unavailable due to their scarcity presence |
| Hydrocolloids | Two phase’s systems from starch immobilized with iodine, dextran-PEG, or plastic woven polymers Suitable for: chronic ulcers and burns |
High absorbent materials, easily removed by saline or sterilized water, non-adherent, high density, water-proof materials, and no pain dressings | Have varied antimicrobial activity based structure, little cytotoxicity, volumetrically unstable, high leakage exudates, dextran hydrocolloid delays healing, impermeable to gases, unpleasant odor and color changes |
The concept of hydrogels as wound dressings
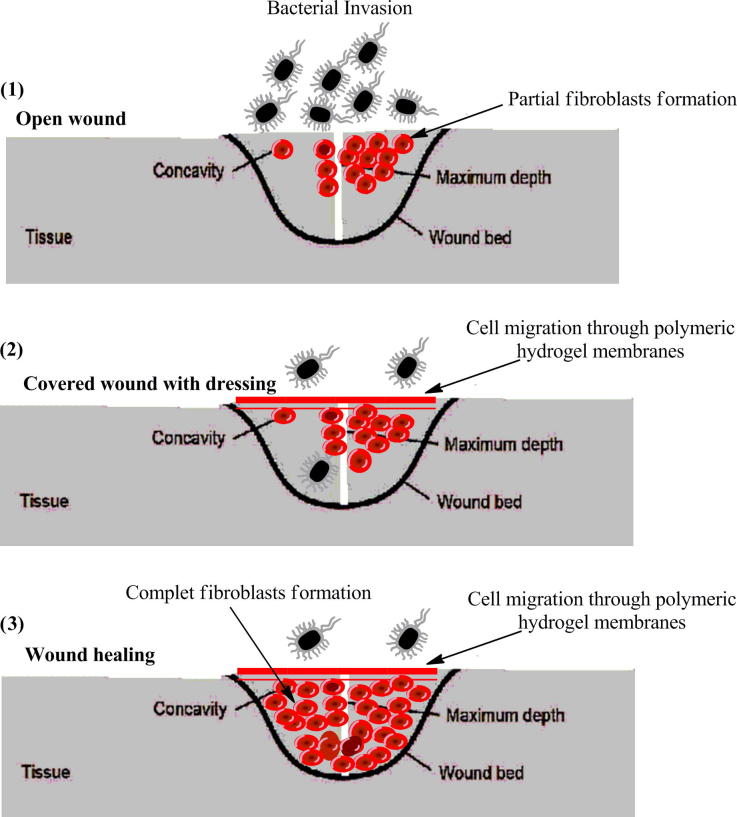
Intrinsic properties of membranes as wound dressings to endorse the skin healing and to protect the skin defect zone from infection, have been progressively investigated and applied in the clinical sectors since early eighties [1]. The mechanism of hydrogels as wound dressings can be ascribed as follows. Hydrogels can absorb and retain the wound exudates, which promote fibroblast proliferation and keratinocyte migration. The last two processes are very necessary for complete epithelialization and healing of the wound [5], [10]. In addition the tight mesh size of hydrogels structure protects the wound from infection and prevents microorganism and bacteria to reach the wound area. However, hydrogels structure allows transporting bioactive molecules e.g. antibiotics, and pharmaceuticals to wound centre. Such molecules can be entrapped into hydrogel networks during gelling process, while these molecules can be exchanged with absorbing the wound exudates during the sustainable release process after contacting hydrogels with the wound surface. The significant tissue-like water content of hydrogels provides the needed flexibility and elasticity to adapt wounds located in different body sites.
Skin and wound structure
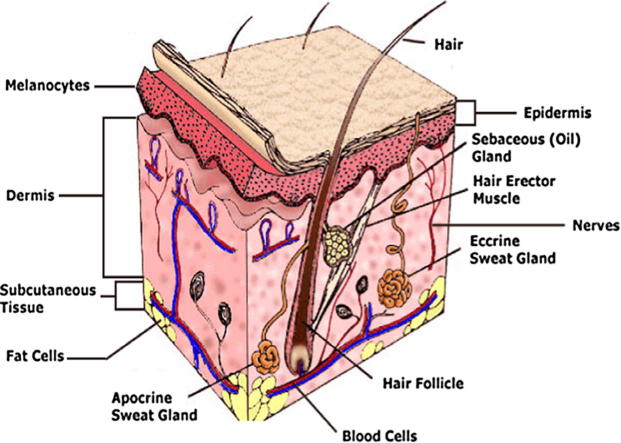
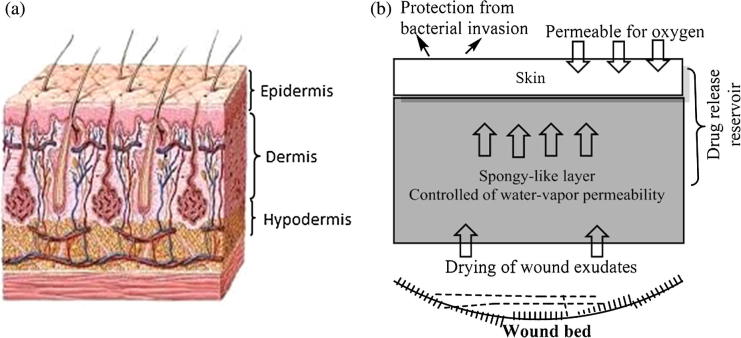
Skin is the principle exterior defense system which protects the inner body systems from microorganism’s attack, contamination, infection, and the effect of external environment. It was found that, the skin of body adult can cover area of 2 m2 and its weight is more than 10 wt.% of total weight of adult body. Skin plays a vital role to regulate the temperature of body and transmitting the external environment information, like pains and heat. Skin composed form three multi-histological layers, the epidermis, the dermis, and the hypodermis or subcutaneous layer (Fig. 2). The epidermis upper layers or the stratum corneum layers have been revealed for building the main barrier to percutaneous penetration of any external invasion. Dermis layer exists exactly adjacent to epidermis layer and consist of connective tissues matrix which provide the elasticity and deformation resistance for skin. Also, dermis layer contain the blood vessels which give the layers with nutrients and oxygen. The hypodermis layer are subcutaneous fat tissue followed the epidermis and dermis layers which provide the thermal isolation and mechanical protection to the body [11]. Fig. 2 shows the diagnostic structure details of the human normal skin and the work mechanism of wound dressing materials at the wound bed. A wound is described as a break or defect in the skin, which formed due to physicochemical or thermal damage. The wound is divided into acute and chronic wounds. Acute wounds are injured skin that needs a healing over periods 8–12 weeks, such as burns, and chemical injuries which result in attrition contact between skin and hard surface. Chronic wounds need a long healing time reaches to months, and leaves serious scars. There are some reasons which delay the healing of chronic wounds such as, diabetes, wound strong dryness and infections [12]. Whereas, the healing of wound passes during four stages, homeostasis, inflammation, granulation tissue formation and remodeling which timely interfered. These healing phases affected on some specific and individual factors e.g. nutrition, patient age, diseases, size, depth and causation of wound [13] (Fig. 2).
Fig. 2.
Schematic representation of normal skin structure (a) and design of an ideal wound dressing membrane (b).
Wound dressings
Thousands of patients suffered annually from different kinds of epidermal or skin damage or burns by hot water, flames, accidents, and boiling oil. These accidents normally accompany with disabilities on treatment and high cost treatment or even sometimes death. As the World Health Organization, more than 30,000 deaths per year occur, owing to scalds and burns forms [14]. Notably, both two patients’ cases of adults and overage people are suffering over challenges that dermis regeneration cannot occur spontaneously again. Since auto-skin repair has somewhat accessibility and accompanied by further scarring. This traditional approach for substantial loss of dermis cannot meet the requirements, and polymeric dressing materials became inevitable for skin tissue repair or healing with time.
History of wound dressings
Until the mid 1962, the researches of wound dressing and healing were somewhat neglected. It was supposed previously that the wound heal is faster and more efficient, if it is kept dried and maintained uncovered. This speculation was assumed before establishing the ideal requirements for wound healing materials. The pioneering work of Winter [5] designed the first generation of wound film or “dressings”, where he revealed that the epithelial repair of wounded pig skin was at least twice compared to the air-exposed wounds. Since this date, the studies and researches of wound dressing development were further heightened; suggesting ideal wound dressings should keep a wetted environment with high biocompatibility and prohibit the bacterial infection for accelerating the tissue regeneration. In eighties, the wound dressings were classified according to their wettability degree into dried and wet dressings. The idea of the dried wound dressings was regarded the proper for wound healing. Until the mid of 1970s, the woven cotton gauze or non-woven blends of rayon with other fibers like polyester or cotton, were known as the most frequently dressings in the market [15]. The traditional or dried dressings maintained the wound area dry, reduced the wound size from view, allowed to absorb all wound exudates and fluids causing a leakage and provided further contamination. Thus, this behavior was classified as a hostile for bacterial proliferation and also for viability of the mammalian cells and tissue repair. According last investigations, Winter [5] found that in case dried dressings, a scab can covers the whole wound area which decreases the epithelialization rate and inhibit the dressing rate. Therefore the gauze-cotton dressings (dried dressings) which were earlier frequently utilized as wound dressings became now not so useful because their own drawbacks, as follows [16]: (i) Inability to protect the wound from microbial invasion, (ii) cause strong pains for patients during the removal time, due to their big adhere on wound surface, (iii) accumulated wound exudates at wound surface owing to their low absorption rate, which facilitate the microbial attack, (iv) low gases permeability, (v) not suitable for chronic wounds, and (vi) keep the wound dried which decreases the epithelialization rate and cell proliferation.
Requirements of ideal wound dressings
It was demonstrated that the wound healing with wet dressings is faster than that with dried dressings. This fact is ascribed to the healing and the renewed skin without formation of eschars or inflammation; can be only taken place in a wet environment [5]. Thus, the wetted or moisten dressings were taken in account to be the suitable dressings candidate for skin repair and wound dressing. Also, they were considered with high water content and inherent permeability. An ideal wound dressing materials should fulfill the following characteristics (Fig. 2): (a) keep a local moist environment around wound, (b) good gases transmission, (c) remove excess exudates, but do not reach to saturation on wound outer surface, (d) protect the wound from micro-organisms, infections, or contaminations, (e) stop the wound desiccation, (f) reduce the wound surface necrosis, (g) stimulate the growth factor, (h) possess mechanical protection, (i) are easy and comfortable to remove and change, non allergic, non-toxic and biocompatible, biodegradable and elastic, (j) aid to reduce the pain from the wound and costly/commercially acceptable, and (h) easily sterilized [17]. Toward this end, wound healing takes place efficiently and faster in wet environment, as provided by hydrogel than dried environment. Interestingly, hydrogels can control the scab formation and allow for cell proliferation and epthilialization process. For emphasizing that hydrogels are eventually the best choice for wound dressings. It was found that the water and fluids losses from wounded skin are almost 20 times greater than the normal skin. Thus, the water permeability of used dressings should control the extensive dehydration as well as building up of exudates and scabs without affecting on the epithelialization or cell proliferation processes, as provided by hydrogels. Researchers reported that the regular lost-water from normal skin is ∼250 gm−2/day at 35 °C, this amount of water loss extensively increase to 5000 gm−2/day based on the nature of wound. This exhibits that, the used dressing should be thin-film or membrane which possesses a high water-vapor permeability, high-liquid capacity, bacterial growth, and repair recovery occurs [18]. As such, wound dressings should have a satisfied and balanced wound wall adherent but easy to remove after treatment, simultaneously. A schematic representation of the ability of polymeric dressings was designed as a physical barrier to protect the wound from microbial invasion, infection and supporting fibroblasts migration accelerating the healing rate, is showing in Fig. 3.
Fig. 3.
Schematic representation of the role of hydrogel membrane materials for enhancing and accelerating the wound healing phases.
Classification of wound dressings
Dressings materials which are used for wounds or burns are known as “artificial skin”, should possess properties of usual skin to accelerate the recovery within wounded or destroyed skin areas. They are used for wounds and burns were classified into traditional, biological, and artificial dressings [19].
Traditional dressings
The most famous example of this category is gauze or gauze-woven cotton composite dressings, which were offered since the mid 1970s [15]. These materials were characterized with their low cost, easy use and fabrication. However, the popular disadvantages of these materials restricted their use, can be described. The gauze and gauze-cotton composite possesses much absorbent capacity of wound exudates which causes a fast dehydration and promotes the bacterial growth and contamination. In addition, at the end of treatment the cover removal is a somewhat difficult that causes bleeding or damage to the renewed epithelial flora. Thus, many attentions have been exerted to address these disadvantages by grafting the gauze-cotton composite with non-adhesive inner surface were fabricated to relieve the pain or minimize the damage to the renewed skin when removing the dressings. Advanced cotton gauze composite have been recently developed to meet all requirements of typical dressings. Cotton gauze was coated with chitosan-Ag-ZnO nanocomposites [20]. Obtained results illustrated that, treating of cotton gauze membrane with chitosan-Ag-ZnO increased swelling capacity and improved antibacterial activity versus Escherichia coli and Staphylococcus aureus.
Biological dressings
The biological dressings as called “auto-grafting” are regarded the most suitable materials for complete healing of deep, chronic wounds and burns. This method is depending upon the donation of normal and fresh skin from foreigner bodies e.g. human, animals, or cadavers. Such materials of this method are from collagen-type structure including elastin and lipids. The basic drawback of these materials is sometimes inadequate for donating skin part for deep or large wound, which results in the search to new tissue donor [21].
Artificial dressings
Artificial dressings are fabricated from synthetic materials such as, non-biological materials and polymers which are not found in skin ingredients [22]. Synthetic dressing’s composition should be a harmless, mechanically stable, biodegradable, and presents a proper environment for the tissue repair. Recently, there are huge demands for polymeric membrane materials to be applied for wound dressings. The polymeric wound dressings have been used recently in different forms such as films, foams, hydrogels, alginates, and hydrocolloids (Table 1). Table 1 shows the classification of artificial polymeric wound dressings according to the shape including their positives and negatives. While, Table 2 describes the possible different polymeric dressing materials in the world markets and their commercial names, the optimal use and the utilization percentage in the sector of wounds and burns care [19].
Table 2.
List of currently available polymeric wound dressing materials in the world markets.
| Type of dressings | Brand names® | Description | Use for | Global utilization rate (%) | |
|---|---|---|---|---|---|
| Artificial Dressing Materials | Polymeric Films |
|
Films were synthesized from polyurethane or any other polymeric materials |
|
8 |
| Polymeric Foams |
|
Foams dressing were synthesized from hydrophilic foams and hydrophobic backing or semi-permeable with non absorbent membranes, e.g. polyoxyethylene glycol surrounded with polyurethane or silicone/polyester |
|
5 | |
| Polymeric Hydrogels | Cultinova Gel, Biolex, TegaGel, Carrasyn, NuGel, 2nd Skin Flexderm, Exu Dry Dressing, CarraSorb, and GRX wound Gel | Hydrogels were synthesized from crosslinked hydrophilic polymers, e.g. polyvinyl alcohol, polyvinyl pyrrolidone, or polyethylene oxide |
|
43 | |
| Polymeric Alginates |
|
Alginate hydrogels were synthesized from chemical crosslinking of sodium alginate algae with Ca, Mg, or Zn salt solutions |
|
20 | |
| Polymeric Hydrocolloides | Iodosorb (Cadexomer), Debrisan (Dextranomer), and Sorbex, Duoderm (polymer blend) | Hydrocolloides were synthesized by immobilization of iodine onto water-soluble modified starch based on cadexomer-iodine beads; the gel was formed by iodine exchange between polymeric material and wound exudates. The second form is Dextranomer, where crosslinked dextran is grafted with polyethylene glycol |
|
24 | |
Hydrogel dressings had large share for wound dressing applications due to their advantages excelled on own disadvantages (Fig. 3). At present, PVA is one of the most frequently and the oldest synthetic polymer have been employed as, wound dressings, wound management, drug delivery systems [23], artificial organs [24], and contact lenses [25]. However, PVA hydrogel has inadequate elasticity, stiff membrane, and very incomplete hydrophilic characteristics which restrict its use alone as wound dressing polymeric membranes. Among the various hydrogels described in literatures, hydrogels prepared using PVA blended with some natural polysaccharides (Fig. 4) and some other synthetic ones (Fig. 5) are attractive and the most widespread route of membranes synthesis, because of the abundance of such polymers, easily for chemical derivatization or modification, and usually good biocompatible [26]. Moreover, Kamoun et al. [14] have reported that the properties of hydrogels based PVA-composite polymers have outperformed their counterparts of other polymers. Natural and synthetic polymers have been used individually or combined with each other for preparing of dressings as showed in Fig. 4, Fig. 5. Sood et al., [1] have reported that polymeric wound dressings based on hydrogels facilitated the healing of pressure ulcers patient’s by promoting more rapid epithelialization. Hence, the number healed wounds with hydrogels as dressings were 85%, compared to 50% with those which were healed by traditional gauze dressings [1], [19].
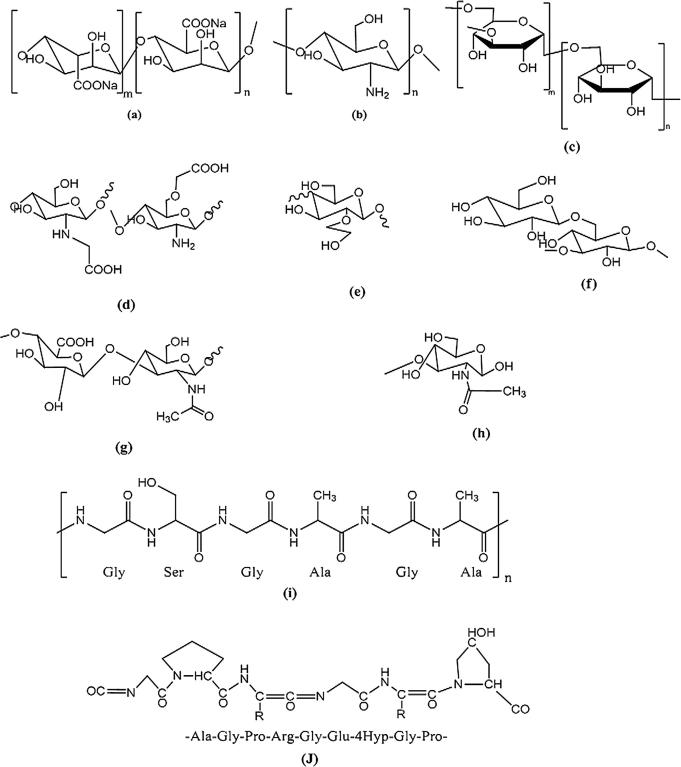
Fig. 4.
Chemical structures of natural polymers and their derivatives which were employed formerly as hydrogels membranes for wound dressings or skin substitutes, (a. sodium alginate, b. chitosan, c. dextran, d. N-O-carboxymethyl chitosan, e. hydroxyethyl starch, f. glucan, g. hyaluronic acid, h. poly-N-acetylglucosamine, i. silk, j. gelatin).
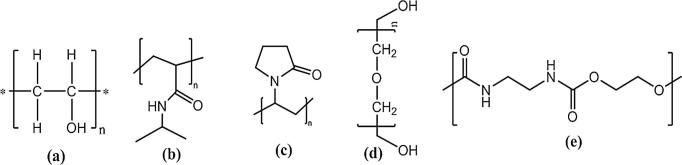
Fig. 5.
Chemical structures of synthetic polymers which were employed formerly as hydrogel membranes for wound dressings or skin substitutes, (a. polyvinyl alcohol, b. poly (N-isopropylacrylamide), c. poly (N-vinylpyrrolidone), d. polyethylene glycol, e. polyurethane).
Natural polymers
Collagen
Collagen type-I is of the first, the oldest, and the most frequently dressings till date, it is a biocompatible abundant protein existing in connective tissues of body. The collagen was first used in human body by Knapp et al. [27]. Collagen was used the first time as injectable and building materials under the dermis in wounds in 1980. Different collagen forms e.g. suspensions, foams, wound dressings materials, suture, sponges and gels have been employed in literatures for dermal injections, while collagen has not been used itself yet, as wound dressing membranes. Collagen-rich pig skin was used previously as wound dressings [28]. Recently, biodegradable collagen type-I membranes were used for rapid epithelialization in surgical defects of oral mucosa [29], [30], for burns/chronic wounds [31], and for superficial partial thickness burns in children [32]. Collagen was not combined with PVA in a wide scope as wound dressings, this might owing to collagen is used as a skin regenerative templates. Collagen plays major role in haemostasis in order to promote the wound healing and supports fibroblasts growth, attachment, differentiation, and migration of kerationcytes directly. However, collagen was combined with other polymers e.g. hyaluronic acid as dressing membranes, skin regenerative templates or skin substitute matrix [28]. Nanofibrous composite membranes composed of collagen-chitosan-PEO was synthesized by electrospinning as dressing membranes [33]. The common disadvantage of collagen membranes usage is, they have a big permeable to bacteria and microorganisms.
Chitin derivatives and chitosan
Chitin is a copolymer polysaccharide composed of N-acetyl-D-glucosamine and N-glucosamine units are distributed in block or random form during biopolymer chains. This biopolymer is called as chitin or chitosan depending on the ratio between N-acetyl-glucosamine or N-glucosamine is greater than 50% in the biopolymer chain, respectively (Fig. 4). Chitosan is soluble in a dilute acidic solution of pH ∼ 2, as a result of it possesses a rigid crystalline structure [34]. Chitin and chitosan are regarded the second common biopolymer used in the past after collagen as wound dressings, due to their antimicrobial activity, resistance against environmental conditions, adhesive nature, antifungal, and excellent oxygen permeability. Thus, chitin and chitosan were employed as excellent membranes for wound and burn dressings. It was noticed that chitosan enhances the granulation of wound which candidates it as accelerator agent for treating open and deep wounds. Due to its haemostatic effect and it accelerates fibroblasts layer formation which increases the healing rate. Chitosan as wound dressing was fabricated in different forms, such as films, hydrogels, fibers, powders, and nanoparticles [35]. The popular disadvantage of chitosan use as dressings is its high cost and difficult handled.
Chitosan was applied in two main directions as wound dressings, first as coated-textile materials, and second in a combination with PVA composite membranes using freeze-thawing (F-T) cycles method. Chitosan was first used as coating martial-based textiles for wound dressings [8]. Cotton textile was layered with chitosan and polyethylene glycol (PEG) afterwards freeze-dried, where chitosan-PEG formed a very porous thin dressing film. They found that an addition of chitosan controlled the film surface morphology, while PEG was used as pores forming agent. Yu et al. [36] have designed a composite hydrogel membrane from polyvinyl alcohol-poly(N-vinyl pyrrolidone)-chitosan containing antibiotic. The prepared membrane showed a quick antibiotic release at the beginning then becomes slower with time, which recommended for the healing management usage, while this membrane showed a good mechanical stability and elongation percentage. Samah et al. [37] have prepared chitosan-coated gauze dressings, they reported a considered microbial resistance against E. coli and Lactobacillus growth, while thermal and physicochemical properties were evaluated. Chitosan-alginate membranes for wound dressing and healing using poly electrolyte complex crosslinking concept, were prepared by Wang et al. [38]. Obtained membranes showed a quick healing rate for incision wounds in rat-model and high exudates capacity, compared to commercial gauzes. PVA-chitosan blend hydrogel membranes as wound dressings were designed for the first time by Cascone et al. [39], using repeated (F-T) cycles. The results revealed that an addition high content of chitosan to membranes prevents the crystalline phase formation of PVA, which affects slightly on the thermal stability and forms porous and filamentous morphological structure. New crosslinking method of PVA-chitosan hydrogel membranes was offered by Yang and co-workers [40], [41]. Hydrogel membranes were formed by freeze-thawing cycles followed by γ-irradiation, or the reverse. They reported that hydrogels crosslinked with irradiation then F-T cycles showed high swelling capacity, high thermal/mechanical stability, and low water evaporation compared to those crosslinked by F-T cycles then irradiation or F-T cycles alone. In addition hydrogels crosslinked by F-T, then irradiation have a good resistance against Escherichia coli (E. coli) and with high chitosan contents in membranes. Also, results of Yang et al. [41] showed a rapid healing for full-thickness wound after addition glycerol into-PVA-chitosan membranes, which exhibited non-toxicity toward L929 mouse fibroblast cells in MTT-assay test.
El-Salmawi [42] suggested γ-radiation induced for crosslinking PVA-chitosan dressing membranes. Results revealed that mechanical properties and microbe penetration test increased with increasing PVA content or irradiation doses, due to high crosslinked structure formed. While swelling capacity and antimicrobial activity increased with chitosan contents in membranes. New grafting method was used by Don et al. [43] for synthesis of chitosan-g-PVA-PVA hydrogel membranes. Results showed that blood compatibility of chitosan-g-PVA-PVA hydrogel membranes was improved by addition of pure PVA or low moieties of PVA-chitosan, while pure chitosan showed poor blood compatibility. Sung et al. [44] suggested minocycline-loaded PVA-chitosan hydrogel films as wound dressings using F-T cycles. Results showed that an addition of chitosan increased swelling capacity, water vapor transmission, and pores areas. On the contrary, it decreased mechanical and thermal stability of membranes. Similarly, an addition of minocycline into membranes accelerated the healing rat of dorsum wound, as compared with PVA-chitosan membranes without minocycline. Recently, Zhang et al. [45] have proposed new dressing membranes from carboxyl-modified PVA-chitosan hydrogel containing gentamicin sulfate, using solution-casting method and chemical crosslinking based on formation of amide linkage. Their bioevaluation tests showed that, gentamicin-loaded membranes possess sustainable release profile, and restrain the bacterial proliferation and protect the wound area from infection.
Chitosan derivatives have ability to soluble in acidic, medium and basic conditions, were found better than chitosan itself, due to the antibacterial activity of chitosan was noticed only in acidic physiological conations and inadequate homogeneity between pure chitosan and PVA. N-O-carboxymethyl chitosan (CM-chitosan) [46], carboxyethyl chitosan (CE-chitosan) [47], and quaternary chitosan (Q-chitosan) [48], have given much attentions due to their excellent antibacterial and antifungal activities (Fig. 4). Zhao et al. [49] have prepared PVA-CM-chitosan membranes as wound dressings using electron beam irradiation. PVA-CM-chitosan membranes showed a biological resistance against E.coli even with low CM-chitosan concentrations. Moreover, grafting interaction among membrane compositions was observed by electron beam irradiation, and full homogeneity between them was noticed too. They have also prepared PVA-CE-chitosan nanofibers mats using electrospinning for skin regeneration and healing. Ignatova et al. [50] have used electrospinning technique to prepare PVA-Q-chitosan nano-fibrous mats, which exhibited a significant antibacterial activity and antifungal growth, due to presence of quaternary ammonium groups. A commercial N-carboxybutyl chitosan-agarous as topical membranes-type wound dressing were synthesized using a supercritical solvent impregnation, by Dias et al. [51]. Results showed that membranes have a high water holding capacity and good vapor transmission rate which gave them in typical range of commercial topical wound dressings.
Alginic acid and sodium alginate
Alginic acid is a natural linear polysaccharide extracted from brown marine algae (seaweed) such as Laminaria and Ascophyllum species. Alginic acid was first patented by Stanford in 1880, and was first discussed in literature in 1883. Alginic acid consists of a linear copolymer of α-L-guluronic acid and β-D-mannuronic acid (Fig. 4), where the ratio between them adjusts the elasticity of obtained crosslinked alginic gels [52]. Alginic acid and its salts were first used as wound dressings and treatment in the form of gels and sponges by Thomas [53]. Thomas reported that alginic acid dressings improved the healing rate and cellular activity properties of wound such as, haemostatic, adhesion and cells proliferation. Kim et al. [54] have prepared PVA-alginate hydrogel membranes containing nitrofurazone, ampicillin, and clindamycin, respectively as wound dressings using F-T cycles method. They found that, the addition of alginate in membranes increased swelling ability, thermal stability and in vitro protein adsorption resulting in reduction of blood compatibility. However, it decreased the mechanical stability of membranes. In addition, alginate content has a prominent impact on the release behavior of ampicillin or clindamycin from dressing membranes. Nam et al. [55] have prepared PVA-alginate hydrogel membranes using the 60Co γ-ray irradiation techniques. Results revealed that alginate increases, swelling capacity of membranes increases too. Moreover, the foreign reaction due to implantation of PVA-alginate membranes in wound rats was a quit limit and exhibited small sizes with non-scars healed wound. This behavior is ascribed to alginate membranes have good haemostatic, adhesion, absorptive properties for wound healing. Tarun et al. [56] have developed a new concept to prepare PVA-alginate composite webs for wound healing, using electrospinning technique. They found PVA-alginate composite fibers with high calcium alginate content exhibited high water vapor transmission character which provides the moist environment resulting in wound healing acceleration. Interestingly, Thomas [53] has classified the crosslinked PVA-alginate membranes into three categories. The most important category is based on contacting with an exuding wound, and an ion-exchange reaction occurs between Ca2+ ions in membranes and Na+ ions in blood serum or wound fluids [14].
Starch and starch derivatives
Starch is a famous plentiful-source and cost-effective polysaccharides. Starch contains about 30% amylose, a linear (1–4) glucan, and 70% amylopectin [57]. Starch has a poor hydrophilicity which limits its use alone in various applications. This disadvantage can limit pure starch usage in wound dressings, because starch does not provide the demanded moist environment for wound. Chemically modified starch through graft copolymerization of vinyl monomers onto starch has comprehensively accomplished as follows. PVA-starch composite membranes were developed by Zhai et al. [57], using γ-rays and electron beam irradiation for biomedical applications. They demonstrated that amylose content in starch and irradiation dose have affected sharply on grafting of starch onto PVA hydrogels.
Hydroxyethyl starch (HES) is one of the most popular and applicable starch derivatives (Fig. 4). HES is prepared by reaction occurring with amylopectin and ethylene oxide, it was previously used as blood-plasma expander, cryo-preservative and as drug delivery polymers [58]. They have introduced PVA-HES hydrogel membranes for wound dressing, using F-T cycles. They suggested that high HES content increased thermal stability, protein adsorption, released ampicillin, and swelling ability of PVA-HES membranes. This fact was ascribed to high hydrophilicity and long HES chains.
Dextran
Dextran is a bacterial polysaccharide, consists of α-1,6- linked D-glucopyranose residues with a low contents of α-1,2-, α-1,3- and α-1,4-linked side chains (Fig. 4) [59]. Dextran is extracted from Leuconostoc mesenteroides contains about 5% of α-1,3-glycopyranosidic linkages. Dextran was formerly utilized for drug delivery and blood plasma polymer expander. Cascone et al. [39] developed PVA-dextran hydrogel membranes using F-T crosslinking cycles, showing more ordered and uniform physically crosslinked PVA membranes. This study explored that PVA-dextran membranes possessed more ordered membranes structure, good swelling capacity, and adequate release profile due to excellent miscibility degree between PVA and dextran, as compared to PVA-chitosan membranes. Fathi et al. [60] have synthesized PVA-dextran-gentamicin hydrogel membranes and PVA-dextran xerogel membranes for wound dressings using F-T crosslinking cycles. They reported that, an addition of dextran influenced sharply on membrane properties. As such, it increased in vitro protein adsorption, water vapor transmission, and swelling capacity of membranes. Notably, the released gentamicin from PVA-dextran membranes reduced appreciably the wound size and spots in rat dorsum, as compared to membranes free-gentamicin. They have described intensively the morphology and thermal stability of PVA-dextran hydrogel membranes crosslinked by F-T cycles for wound dressings. This study proved that the dextran content in membranes has no clear impact on thermal stability; however it has a significant impact on surface morphology and crystallinity distribution areas in membranes. The main disadvantages of dextran usage in wound dressing is very expensive and non available in most markets.
Glucan
Glucan is a water-soluble biodegradable polymer derived from fermentation of incubated plants [61]. It consists of β-(1–3) and β-(1–6) linked-D-glucose residues (Fig. 4). Lee et al. [61] designed a grafted skin substitute materials composed of grafted gelatin-glucan films. Results demonstrated that glucan films have a significant antibacterial, antiviral effects, and anti-inflammatory properties. PVA-glucan membranes for wound dressing have been prepared by Huang and Yang [62], using a physical blending followed by drying at 110 °C. It was found that the wound area was remarkably healed in a short time by 50%, due to the free-release of glucan from membranes based on its anti-inflammatory property. Free-release of glucan from membranes is ascribed to no covalent bond between PVA and glucan membranes. Thus, glucan is considered an important healing agent additive and accelerator for healing rate in deep and superficial wound dressings.
Gelatin
Gelatin is the most abundant protein is obtained by an incomplete denaturalization of collagen extracted from the boiling of some materials such as, bones (27%), connective tissues or organs (28%), and skin of certain animals (44%, usually cows and pigs) (Fig. 4). Due to, the gelatin has a nature origin, therefore it owns biological features and was formerly used for biomedical applications. Gelatin was formerly employed to prepare strong hydrogel membranes and transparent film with glucan as described above [61]. Also, gelatin was blended physically with PVA to prepare hydrogel membranes containing-glutaminase for wound dressing using enzymatic crosslinking, followed by F-T crosslinking cycles [63]. Results showed that, the gelatin content in membranes has a strong impact on fibroblast culture and proliferation behavior of wound due to its nature.
Poly-N-acetyl glucosamine
Poly-N-acetyl glucosamine is biodegradable marine polymer extracted from microalgae (Fig. 4). It was early used as supporting materials nano-fibers for wound healing. It was characterized as an effective haemostatic agent, and as excellent antibacterial materials. Therefore, it has used to accelerate the wound healing and burns. Hence, granulation tissue exhibited quicker rate of proliferation and vascularization after 1-h treatment than the 24-h or untreated wound of mouse [64]. This polymer is somewhat rare in literatures due to its high cost-extraction and scarcity sources. However, it’s appealing biological properties were detected as supporting materials for wound dressings.
Hyaluronic acid or hyaluronan
Hyaluronic acid is a naturally occurring biodegradable-biopolymer, consists of d-glucouronic acid and D-N-acetylglucosamine (Fig. 4). It was derived from bond and connective tissues of mammals and synovial fluids [65]. It has been extensively used in cartilage and tissue repair since its discovery in 1934. Park et al. [66] reported that hyaluronan can interact with most tissue components such as proteins and growth factors which facilitates the healing rate of most wound types and tissue repair. Price et al. [67] have suggested hyaluronic acid in sheets to heal angiogenesis and to convert injured areas of chronic wound into acute wound. Miller et al. [68] have used hyaluronic ester merogel for nasal wound dressing in sinusitis; they found that hyalyronic acid content in nasal dressing protected the injured area from bacterial invasion and most infections. Recently, Fahmy et al. [69] have proposed PVA-hyaluronan hydrogel membranes containing ampicillin for wound dressing, using F-T crosslinking cycles. The bioevaluation results evidenced that prepared PVA-hyaluronan membranes free-ampicillin showed biological resistance against Candida albicans, owing to HA presence.
Bacterial cellulose (BC)
Bacterial cellulose is biosynthetic cellulose of plant-origin, derived from strain Gramm-negative bacterium Acetobacter xylinum using glucose units as substrate. BC film possesses appealing properties such as high purity, good tensile strength for never-dried film, high exudates capacity, biocompatible/biodegradable polymer, and has a unique nano-fibril morphology network structure [70], [71]. BC membranes were recently used as excellent topical wound dressings for burns. Curiously, BC membranes showed a very bad inherent antibacterial activity property, thus their lack ability to prevent bacterial infection in wounded area, mainly restrict their use as dressings.
Keratin and silk
Keratin is naturally occurring insoluble protein; it associates as intermediate filaments to create the bulk cytoplasmic layers of epithelia and epidermal structures (Fig. 4). Keratin was classified into soft and hard keratins based on the sulfur content in its structure and also in terms of the number and the sequence of amino acids components. Due to its poor solubility, keratin was utilized as wound dressing in forms of foams and gels, for absorbing the wound exudates [72]. Strangely enough, both soft/hard keratin protein was not blended with PVA as wound dressing membranes, it might due to it’s a very hard solubility compared to PVA. However, it was utilized as wound healing agent, due to its supporting for fibroblasts formation and easy absorbing heavy metal ions and volatile organic compounds for skin regeneration. Silk fibers are mixture of protein amorphous biopolymer; it is naturally derived from silkworm and belongs to the type of secretion animal-fibers. Silk consists from two simplest amino acids mixture of glycine and alanine. It possesses remarkable biological properties such as, high mechanical strength, excellent elasticity, and biocompatible/biodegradable. Limited and very rare studies have been discussed the silk-wound dressing film or membrane. Padol et al. [73] revealed that silk-protein film as a novel wound healing agent is very efficient with epidermal growth factor in case acute wound. Roh et al. [74] have tested silk protein alginate sponge for dressing of full thickness skin defect on rat-model.
Synthetic polymers
Polyurethane
Polyurethane is copolymers from urethanes groups produced by a conjugation between diol and diisocyanate groups via polymerization reaction. Nontoxic and non biodegradable polyurethane elastomers were rarely used for wound dressings that provided earlier epithelial cover to burned/injured patients as an exterior film [75].
Poly (methyl methacrylate)
Methyl methacrylate is a synthetic non-biodegradable polymer (Fig. 5), which is used as anti-both heat and UV. Thus, it has been utilized as supporting dressing materials in surgical plastic, oral cavity or dental tissue, and little of injuries treatment [76]. This polymer has not been reported as a direct wound dressing membranes yet.
Proplast or alloplastics
Proplast or alloplastic are high biocompatible synthetic polymers such as, low density polyethylene, polydimethylsiloxane, polyethyleneterphthalate, or Teflon (polytetrafluoro ethylene) [77], which were first used as surgeons plastic and alternative to auto-logous tissues [78]. These polymers are characterized as; they are inert materials, easily sterilized, insoluble in polar or non-polar solvents, and non-carcinogenetic materials which make them the best synthetic dressings particularly for injured areas. Alloplastic copolymers from polyether-polyester- hydroxyapatite composite tympanic membranes were fabricated and implanted to accelerate the healing of epidermis and epithelium layers [79].
Poly(N-vinylpyrrolidone) (PVP)
PVP is a water-soluble and biocompatible/biodegradable synthetic polymer (Fig. 5) [7]. It has been utilized previously as hydrogel membranes for skin substitutes, due to its low toxicity, good water vapor transmission, and impermeable for bacteria. Crosslinked PVP hydrogel membrane with radiation polymerization exhibited impermeable behavior toward bacterial invasion, and very elasticity. However, cell attachments test showed that PVP membranes are very suitable only for healthy skin or wounded skin in tropical environment [80]. Also, they proved that an addition of PEG enhanced the porosity of PVP hydrogel membranes toward good water vapor transmitting and hindered the bacterial barrier. PVA-PVP hydrogel membranes crosslinked by 6Co γ irradiation were synthesized by Razzak et al. [7]. They found that PVA addition improved the barrier of membranes against microbes and E. coli. Also, PVA-PVP-sterculia gum hydrogel membranes crosslinked by irradiation, have been synthesized for wound dressing [81]. They demonstrated that swelling exudates capacity of membranes increased with increasing PVP and sterculia gum contents in membranes, where the gum was incorporated into membranes as antibacterial agent. Park et al. [82] developed a new route for synthesis of PVA-PVP-glycerin-chitosan hydrogel membranes covered with polyurethane for wound dressing, crosslinked with γ-irradiation or F-T cycles and then irradiation. Results were summarized that membranes made by F-T cycles followed by irradiation, showed higher crosslinked membrane structure, compared to that made by irradiation process only. Moreover, water-vapor transmission of membranes which have been covered by polyurethane was lower than those of un-covered. PVA-PVP-kappa-carrageenen-silk powder hydrogel membranes for wound dressing were prepared using electron beam and γ-irradiation crosslinking methods [83]. They found that addition of PVP and silk powder enhanced significantly mechanical properties of membranes, while carrageenan addition reduced the water vapor transmission. Interestingly, PVA-PVP-carrageenen-silk hydrogel membranes do not shown any toxicity, which recommended their use for fast wound and burn healing. Uslu et al. [84] have prepared PVA-PVP-PEG nanofibers mats for wound dressing, using electrospinning method. It was worthwhile mentioned that, they have added hydroxypropyl methylcellulose (HPMC) into nanofibers due to its high retention to water uptake, and Aloe-vera as healing agent. It was found that addition of Aloe-vera affected sharply on morphology of nanofibers resulting in porous structure and good vapor and oxygen-permeable. Furthermore, its excellent therapeutic effect against microbial growth was investigated; accordingly it has been used as an excellent healing accelerator agent.
Polyethylene glycol (PEG)
PEG is a water-soluble synthetic viscous amphiphilic polymer (Fig. 5). Due to its featured biological properties such as non toxicity, biocompatibility/biodegradability, transparent, and cost-efficient, this makes it a fit candidate for several medical applications. PVA-PEG-CaCl2 hydrogel membranes for wound dressing have been crosslinked by γ-irradiation [85]. CaCl2 solution was used as gelling and plasticizer to improve synergistic effect of dressing membranes, while the thermal stability increased after adding the PEG. This type of membranes introduced good biological characteristics such as, no inhibition for cells proliferation was investigated, while PEG accelerated wound healing size and rate.
Poly(N-isopropylacrylamide) (PNIPAm)
N-isopropylacrylamide is a water-soluble monomer; its polymer is a thermal-reversible polymer with a low critical solution temperature in water at 32 °C (i.e. this polymer can swell and de-swell at 32 °C below and above LCST, respectively), (Fig. 5). Thus, PNIPAm had much attentions to be used under physiological conditions particularly its own other biological properties such as, nontoxic, biocompatible/biodegradable and physiological responding. PVA-PNIPAm-levothyroxine (T4) nanofibrous mats, were prepared using electro-spinning technique for advanced application of wound dressing such as, reducing deposits of adipose tissue on skin [86]. They concluded that the sustained release of T4 from mats was enhanced with increasing PNIPAm content. Furthermore, PVA-PNIPAm blend nanofibrous mats preserved on T4, compared to PNIPAm containing T4.
Nanoparticles composite-polymers
The main target behind the incorporation of nanoparticles into membranes is to enhance the mechanical and thermal stability with swelling capacity and water vapor transmission of resultant membranes specially under stress area [12]. Inorganic nanoparticles which have been used previously for biomedical application such as, clays (e.g. montmorillonite (MMT), laponite RD, bentonite), metal oxides (e.g. Zno, ZrO2, TiO2, carbon based materials (graphene G, graphene oxide (GO)) and other nanoparticles (bioactive glass (BG), AgNO3 nanoparticles, hydroxyapatite (HAP)). They do not possess biodegradability property, thus their usage in wound dressing field have been used only for superficial and thin wounds.
Clay nanocomposite-membranes
PVA-MMT nanocomposite membranes have been prepared using F-T crosslinking cycles for wound dressing [17]. Their results showed that clay-composite membranes afforded feasible and high ability to swell, keep moist membranes and controlled water-vapor transmission with high elasticity under high stress area. Thence, they meet ideal wound dressing requirements for a good healed wound. Recently, advanced bio-functionalized MMT clay with chitosan sulfate to form novel chitosan-SMMT membranes for wound dressing, using genipin as a crosslinker, have been evaluated by Moghadas et al. [87]. These membranes were recommended to the low and moderate exudates wounds, due to their controlled water-vapor transmission rate and good resistance against E. coli growth. Likewise, PVA-bentonite, PVA-Ag and PVA-clove extract membranes for wound dressing were synthesized by F-T cycles method [88]. They suggested that swelling capacity, water-vapor transmission rate, mechanical stability, and antibacterial activity against E. coli have improved entirely after addition of bentonite filler and silver nanoparticles into membranes. However, PVA-clove extract exhibited non-homogeneous membrane.
Metal oxides composite-membranes
ZnO nanoparticles are regarded the famous metal oxide which has been incorporated in membranes for wound dressing applications. While, other metal oxide have been used as nano-fillers for bone regeneration and cartilage repair. ZnO nanoparticles have been incorporated formerly into PVA-chitosan membranes and PVA-alginate nanofibers mats using physical blending between membrane compositions and electrospinning technique, respectively. Shalumon et al. and Vicentini et al. [89], [90] have noticed that PVA-chitosan-ZnO membranes possessed an anti-bacterial activity against S. aureus growth, due to presence of ZnO nanoparticles, while glycerin or tween-80 was added to membranes as plasticizer. Shalumon et al. [89] reported that PVA-alginate-ZnO nanofibers mats exhibited a feasible antibacterial activity against E. coli and S. aureus growth due to incorporating low contents of ZnO, while this behavior has not been investigated in case incorporation high contents of ZnO nanoparticles in mats. Notably, addition of ZnO nanoparticles with either membranes or nanofibers mats enhanced mechanical and thermal properties of obtained fragile hydrogels free-ZnO. Chitosan-ZnO nanocomposite hydrogels were prepared as wound bandages [91]. Chitosan-ZnO nanocomposite bandages showed enhanced swelling, oxygen-vapor transmission, blood clotting rate, antibacterial activity, faster re-epithelialization, and collagen deposition, due to the ZnO incorporation.
Recently, ZnO nanoparticles were incorporated into collagen-dextran composite-hydrogel membranes for wound dressing using glutaraldehyde crosslinker, while the spongious composites were obtained by using F-T crosslinking cycles [92]. Obtained results illustrated that, incorporation 50% of ZnO into collagen-dextran hydrogels, reduced significantly the water-uptake and the biodegradation or enzymatic degradation of collagen and dextran can be easily adjusted up to 50% of ZnO. However, dextran content in hydrogels does not affect on the rheology properties of composite membranes. Also, alginate-ZnO nanocomposite-hydrogel bandages or dressings for infected wounds, have been developed using freeze-drying crosslinking method [93]. The addition of ZnO nanoparticles decreased water uptake degree, controlled degradation rate, and faster blood-clotting compared to alginate hydrogel free-ZnO. Interestingly, alginate-ZnO nanocomposite hydrogels exhibited no cytotoxicity with low ZnO concentration, and slight cell viability reduction with high ZnO content.
Carbon-based materials composite-membranes
A novel composite hydrogel membrane for wound dressing composed of acrylic acid-N, N-methylenebisacrylamide-graphene-Ag hydrogel membranes, were prepared by Fan et al. [94], using ammonium persulphate as a thermal initiator. Graphene is created from graphene oxide (GO) with a large-scale and low cost. Graphene composite hydrogel has a porous structure which can absorb large amounts of water, and excellent biocompatibility which makes it a conductive for cellular adhesion and growth. Resultant membranes showed antibacterial activity particularly against Gramm-negative bacterial, due to the presence of graphene. Additionally, presence of Ag nanoparticles accelerated the healing rate of artificial wound in rat-model. Although, the reported antimicrobial activity of GO compounds, but the introduction of GO in PVA membranes for wound dressings is still a very rare. This fact might be attributed to the limited exfoliation degree of GO in the PVA matrix. Recently, Polymer nanocomposites films (PVA/GO/starch/Ag films) are successfully prepared by one pot synthesis in a biological autoclave where PVA acts as matrix and starch as green reducing agent for biomedical application [95]. Results reveled that the incorporation of GO nanoparticles into PVA/starch/Ag films, enhanced significantly the antimicrobial activity of films versus Gramm positive and negative bacteria.
Other nanoparticles composite-membranes
AgNO3 nanoparticles have been incorporated into PVA-cellulose acetate-gelatin membranes and PVA-chitosan nanofibers for wound dressing, using γ-irradiation crosslinking and electrospinning technique, respectively. Wu et al. [71] indicated that the antimicrobial activity of PVA-cellulose acetate-gelatin-AgNO3 membranes have improved by increasing Ag nanoparticles contents. Unexpectedly, the latter membranes showed lower inhibition for bacterial-adhesion than membranes free-Ag. Li et al. [96] found a notable antibacterial activity for PVA-chitosan-AgNO3 nanofibers against E.coli and S. aureus bacterial growth, due to presences of Ag nanoparticles in membrane composition which facilitated the wound healing. Recently, silver sulfadiazine (SSD) particles were suspended carefully with bacterial cellulose (BC) for preparing wound dressing composite-membranes [70]. In vitro antibacterial tests of prepared BS-SSD membranes exhibited excellent resistance activity against Staphylococcus aureus, Pseudomonas aeruginosa and E. coli. Thus, BS-SSD membranes have dressed wound and burns. Bioactive glass (BG) nanoparticles were blended previously with chitosan for preparing temporary guided tissue regeneration membranes for periodontal wounds, using solution-casting crosslinking method [97]. They decided that the incorporation of BG nanoparticles reduced the mechanical stability of chitosan membranes, and conversely increased membranes bioactivity against microbe growth.
Conclusions
In this review, the most update progress of the use of polymeric hydrogel membranes as wound dressings were recollected and summarized. The wet-retentive dressings have been chosen previously based on the type of wound-shape. Conversely, polymeric hydrogel dressings were found currently a convenient for any wound and burn types regardless the wound-shape. The traditional gauze cotton-dressings can re-injure the wound upon removal time. Thus, the occlusive polymeric dressings were preferred over the semi-occlusive and traditional dressings. The first category of polymeric dressings is based on natural polymers, which are among the core topics intensively discussed in literatures. Chitosan, glucan, alginates, and hyaluronan are more efficient as a wound-healing accelerator than synthetic polymers. Notably, wounds covered with biopolymers, e.g. chitosan-based dressings showed fast healing rate and scarless healing, which are similar the normal skin. The second category of dressings is based on the combination between biopolymers and synthetic ones using favorable physical crosslinking method which is a convenient for healing process. PVA-biopolymers composites membranes exhibited better biological and antimicrobial activities than those composite with synthetic polymers, particularly PVA-chitosan and PVA-alginate membranes. Moreover, PVA-biopolymers composite membranes containing healing agents (e.g. Aloe vera, PEG, sterculia/Arabic gums) or antibiotics (e.g. sod. ampicillin or gentamicin) suggested being typical dressings for acute and chronic wounds. The third category of dressings is based on PVA-nanoparticles-composite membranes were exploited to achieve the features of polymer and nanofillers for improving the performance of dressings of faster healing rate, pain relieving role, and easier removal. Finally, it have decided that natural polymers based dressings have outperformed the synthetic polymers, while additives were incorporated to accelerate the healing rate and improve the mechanical potential, which were lately found advanced therapeutic impact as wound dressings.
Conflicts of Interest
The authors have declared no conflict of interest.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Acknowledgements
This work was supported and financially funded by Science and Technology Development Fund (STDF-Egypt), program of Young Research (project ID No. 5571).
Biographies

Elbadawy Kamoun was born and educated in Kafr El-Sheikh, Egypt. He received his B.Sc. and M.Sc. in Applied Chemistry from Faculty of Science Al-Azhar University-Cairo in 2001 and 2003, respectively. He also received his Ph.D. (Dr. rer. nat.) in macromolecular chemistry from Braunschweig University of Technology (TU-BS), Germany in Feb., 2011. Dr. Kamoun has obtained four post-doctoral fellowships funded by DAAD and Higher education Ministry of Egypt to (TU-BS)-Germany and Fudan University in Shanghai, respectively in 2012, 2013, 2014 and 2016. Dr. Kamoun is currently as an associate professor since April 2016 in SRTA-City, Alexandria, Egypt. Dr. Kamoun has research interest mainly focused on antimicrobial polymers, biomedical applications of polymers, and polymeric membranes for fuel cells and industrial applications and polymers for electronic packaging.

El-Refaie Kenawy is the head of Chemistry department and he is distinguished Professor of polymer chemistry at University of Tanta, Egypt. He is internationally recognized in the field of polymer chemistry. He is a graduate of Tanta University, Egypt. He did his PhD. work according to channel Scheme at Strathclyde University, UK. He worked as postdoctoral fellow and visiting professor at many international universities as Pisa University, Gent University, Virginia Commonwealth University, Tokyo Institute of Technology, and Tanta University. Professor Kenawy is a member of editorial board of many international journals. He participated actively in many international conferences. In 2004, he was Abdul Hameed Shoman Award for Young Arab Scientists in Chemistry 2004. Recent award for Professor Kenawy in 2012 is the most cited award from Miser El-Kher Foundation, Egypt. In 2014, he was awarded Tanta University Prize, and in 2016 he was awarded Tanta University award for excellence in academic performance. He has more than 120 publications and reviews in international journals. He is the vice president of National committee of Pure and Applied Chemistry in Egypt.

Xin (Terry) Chen received his B.S. degree in 1990, M.S. degree in 1993 and Ph.D. degree in 1996 from Fudan University, China. From 1996 to 2000, he was a lecturer in the Department of Macromolecular Science, Fudan University. In 1999, he was a visiting scientist in Brookhaven National Laboratory, USA. From 2000 to 2001, he was a research staff in the Department of Zoology, the University of Oxford, UK. From 2001 to 2007, he was an associate professor, and since 2008, he has been a full professor in the Department of Macromolecular Science, Fudan University. He has published more than 170 peer-reviewed papers. His current research interests include natural polymers, biomedical materials, and synchrotron radiation techniques.
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Sood A., Granick M.S., Tomaselli N.L. Wound dressings and comparative effectiveness data. Adv Wound Care. 2014;3(8):511–529. doi: 10.1089/wound.2012.0401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gibs I., Janik H. Review: synthetic polymer hydrogels for biomedical applications. Chem Chem Tech. 2010;4(4):297–304. [Google Scholar]
- 3.Lim F., Sun A.M. Microencapsulated islets as bioartificial endocrine pancreas. Science. 1980;210:908–910. doi: 10.1126/science.6776628. [DOI] [PubMed] [Google Scholar]
- 4.Yannas, Gordon PL, Huang C, Silver FH, Burke, JF. Crosslinked Collagen-Mucopolysaccharide Composite Materials. U.S. Pat. 4,280,954 1981.
- 5.Winter G.D. Formation of the scab and the rate of epithelization of superficial wounds in the skin of the young domestic pig. Nature. 1962;193:293–294. doi: 10.1038/193293a0. [DOI] [PubMed] [Google Scholar]
- 6.Balassa LL, Prudden JF. Chitosan, a wound healing accelerator. Proc. 1st Int. Congr. Chitin/Chitosan (R.A.A. Muzarelli and Pariser E.R., eds.) 1978, Cambridge, MA.
- 7.Razzak M.T., Erizal Zainuddin, Dewi S.P., Lely H., Taty E. The characterization of dressing component materials and radiation formation of PVA-PVP hydrogel. Radiat Phys Chem. 1999;55:153–165. [Google Scholar]
- 8.Gupta B., Agarwal R., Alam M.S. Textile-based smart wound dressings. Ind J Fibre Textile Res. 2010;35:174–187. [Google Scholar]
- 9.Agostino A.D., Gatta A.L., Rosa M., Schiraldi C. Semi-interpenetrated hydrogels composed of PVA and hyaluronan or chondroitin sulphate: chemico-physical and biological characterization. Biotech Biomater. 2012;2(4):1–6. [Google Scholar]
- 10.Bullock A.J., Pickavance P., Haddow D.B., Rimmer S., MacNeil S. Development of calcium-chelating hydrogel for treatment of superficial burns and scalds. Regen Med. 2010;5:55–64. doi: 10.2217/rme.09.67. [DOI] [PubMed] [Google Scholar]
- 11.Schaefer H., Redelmeier T.E. Skin barrier: principles of percutaneous absorption. Karger, Basel. 1996;19:1–6. [Google Scholar]
- 12.Boateng J.S., Matthews K.H., Stevens H.N.E., Eccleston G.M. Wound healing dressings and drug delivery systems: a review. J Pharm Sci. 2008;97(8):2892–2923. doi: 10.1002/jps.21210. [DOI] [PubMed] [Google Scholar]
- 13.Purna S.K., Babu M. Collagen based dressing- a review. Burns. 2000;26(1):54–62. doi: 10.1016/s0305-4179(99)00103-5. [DOI] [PubMed] [Google Scholar]
- 14.Kamoun E.A., Chen X., Mohy Eldin M.S., Kenawy E.S. Crosslinked poly(vinyl alcohol) hydrogels for wound dressing applications: a review of remarkably blended polymers. Arab J Chem. 2015;8(1):1–14. [Google Scholar]
- 15.Edwards J.V., Yager D.R., Cohen I.K., Diegelmann R.F., Montante S., Bertoniere N. Modified cotton gauze dressings that selectively absorb neutrophil elastase activity in solution. Wound Repair Regen. 2001;9(1):50–58. doi: 10.1046/j.1524-475x.2001.00050.x. [DOI] [PubMed] [Google Scholar]
- 16.Waring M.J., Parsons D. Physico-chemical characterisation of carboxymethylated spun cellulose fibres. Biomater. 2001;22(9):903–912. doi: 10.1016/s0142-9612(00)00254-4. [DOI] [PubMed] [Google Scholar]
- 17.Kokabi M., Sirousazar M., Hassan Z.M. PVA–clay nanocomposite hydrogels for wound dressing. Europ Polym J. 2007;43:773–781. [Google Scholar]
- 18.Fansler R.F., Taheri P., Cullinane C., Sabates B., Flint L.M. Polypropylene mesh closure of the complicated abdominal wound. Am J Surg. 1995;170:15–18. doi: 10.1016/s0002-9610(99)80244-x. [DOI] [PubMed] [Google Scholar]
- 19.Stashak T.S., Farstvedt E., Othic A. Update on wound dressings: Indications and best use. Clin Tech Equine Pract. 2004;3:148–163. [Google Scholar]
- 20.Abbasipour M., Mirjalili M., Khajavi R., Majidi M.M. Coated cotton gauze with Ag/ZnO/chitosan nanocomposite as a modern wound dressing. J Eng Fibers Fabrics. 2014;9(1):124–130. [Google Scholar]
- 21.Kearney J.N. Clinical evaluation of skin substitutes. Burns. 2001;27:545–551. doi: 10.1016/s0305-4179(01)00020-1. [DOI] [PubMed] [Google Scholar]
- 22.Van der Veen V.C., Van der Wal M., van Leeuwev M.C.E., Magda M.W.U. Biological background of dermal substitutes. Burns. 2010;36(3):305–321. doi: 10.1016/j.burns.2009.07.012. [DOI] [PubMed] [Google Scholar]
- 23.Li J.K., Wang N., Wu X.S. Poly(vinyl alcohol) nanoparticles prepared by freezing–thawing process for protein/peptide drug delivery. J Control Rel. 1998;56:117–126. doi: 10.1016/s0168-3659(98)00089-3. [DOI] [PubMed] [Google Scholar]
- 24.Chen D.H., Leu J.C., Huang T.C. Transport and hydrolysis of urea in a reactor–separator combining an anion exchange membrane and immobilized urease. J Chem Technol Biotechnol. 1994;61:351–357. doi: 10.1002/jctb.280610411. [DOI] [PubMed] [Google Scholar]
- 25.Hyon S.H., Cha W.I., Ikada Y., Kita M., Ogura Y., Honda Y. Poly(vinyl alcohol) hydrogels as soft contact lens material. J Biomater Sci Polym Ed. 1994;5:397–406. doi: 10.1163/156856294x00103. [DOI] [PubMed] [Google Scholar]
- 26.Kamoun E.A., Kenawy E.S., Tamer T.M., El-Meligy M.A., Mohy Eldina M.S. Poly (vinyl alcohol)-alginate physically crosslinked hydrogel membranes for wound dressing applications: characterization and bio-evaluation. Arab J Chem. 2015;8(1):38–47. [Google Scholar]
- 27.Knapp T.R., Kaplan E.N., Daniels J.R. Injectable collagen for soft tissue augmentation. Plast Reconstr Surg. 1977;60:398–405. [PubMed] [Google Scholar]
- 28.Park S.N., Kim J.K., Suh H. Evaluation of antibiotic-loaded collagen-hyaluronic acid matrix as a skin substitute. Biomaterials. 2004;25:3689–3698. doi: 10.1016/j.biomaterials.2003.10.072. [DOI] [PubMed] [Google Scholar]
- 29.Rastogi S., Modi M., Sathian B. The efficacy of collagen membrane as a biodegradable wound dressing material for surgical defects of oral mucosa: a prospective study. J Oral Maxillofac Surg. 2009;67(8):1600–1606. doi: 10.1016/j.joms.2008.12.020. [DOI] [PubMed] [Google Scholar]
- 30.Almazrooa S.A., Noonan V., Woo S.B. Resorbable collagen membranes: histopathologic features. Oral Surg Oral Med Oral Pathol Oral Rad. 2014;118(2):236–240. doi: 10.1016/j.oooo.2014.04.006. [DOI] [PubMed] [Google Scholar]
- 31.Singh O., Gupta S.S., Soni M., Moses S., Shukla S., Mathur R.K. Collagen dressing versus conventional dressings in burn and chronic wounds: a retrospective study. J Cutan Aesthet Surg. 2011;4(1):12–16. doi: 10.4103/0974-2077.79180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ramakrishnan K.M., Babu M., Jayaraman M.V., Shankar J. Advantages of collagen based biological dressings in the management of superficial and superficial partial thickness burns in children. Ann Burns Fire Disasters. 2013;26(2):98–104. [PMC free article] [PubMed] [Google Scholar]
- 33.Chen J.P., Chang G.Y., Chen J.K. Electrospun collagen/chitosan nanofibrous membrane as wound dressing. Colloids Surf A: Physicochem Eng Aspects. 2008;313–314:183–188. [Google Scholar]
- 34.Krajewska B. Application of chitin and chitosan based materials for enzyme immobilizations: a review. Enzyme Microbial Tech. 2004;35:126–139. [Google Scholar]
- 35.Mi F., Wu Y., Shyu S., Schoung J., Huang Y., Tsai Y. Control of wound infections using a bilayer chitosan wound dressing with sustaniable antibiotic delivery. J Biomed Mater Res. 2002;59:438–449. doi: 10.1002/jbm.1260. [DOI] [PubMed] [Google Scholar]
- 36.Yu H., Xu X., Chen X., Hao J., Jing X. Medicated wound dressings based on poly(vinyl alcohol)/poly(N-vinyl pyrrolidone)/chitosan hydrogels. J Appl Polym Sci. 2006;101:2453–2463. [Google Scholar]
- 37.Samah NA, Zakaria MA, Yarmo S, Ayub MK. Preparation of chitosan coated gauze and its morphological and physical properties. The NSF Worshop 2001, Kuala Lumpur, Malaysia.
- 38.Wang L., Khor E., Wee A., Lim L.Y. Chitosan-alginate PEC membrane as a wound dressing: assessment of incisional wound healing. J Biomed Mater Res. 2002;63(5):610–618. doi: 10.1002/jbm.10382. [DOI] [PubMed] [Google Scholar]
- 39.Cascone M.G., Maltinti S., Barbani N. Effect of chitosan and dextran on the properties of poly(vinyl alcohol) hydrogels. J Mater Sci Mater Med. 1999;10:431–435. doi: 10.1023/a:1008983215833. [DOI] [PubMed] [Google Scholar]
- 40.Yang X., Liu Q., Chen X., Yu F., Zhu Z. Investigation of PVA/ws-chitosan hydrogels prepared by combined gama-irradiation and freeze-thawing. Carbohyd Polym. 2008;73:401–408. [Google Scholar]
- 41.Yang X., Yang K., Wu S., Chen X., Yu F., Li J. Cytotoxicity and wound healing properties of PVA/ws Chitosan/glycerol hydrogels made by irradiation followed by freeze-thawing. Radiat Phys Chem. 2010;79:606–611. [Google Scholar]
- 42.El-Salmawi K.M. Gamma radiation-induced crosslinked PVA/Chitosan blends for wound dressing. J Macromol Sci Part A Pure Appl Chem. 2007;44:541–545. [Google Scholar]
- 43.Don T.M., King C.F., Chiu W.Y., Peng C.A. Preparation and characterization of chitosan-g-poly(vinyl alcohol)/poly(vinyl alcohol) blends used for the evaluation of blood-contacting compatibility. Carbohyd Polym. 2006;63:331–339. [Google Scholar]
- 44.Sung J.H., Hwang M.R., Kim J.O., Lee J.H., Kim Y.I., Sun J.H. Gel characterization and in vivo evaluation of minocycline-loaded wound dressing with enhanced wound healing using polyvinyl alcohol and chitosan. Int J Pharm. 2010;392:232–240. doi: 10.1016/j.ijpharm.2010.03.024. [DOI] [PubMed] [Google Scholar]
- 45.Zhang D., Zhou W., Wei B., Wang X., Tang R., Nie J. Carboxy-modified poly(vinyl alcohol)-crosslinked chitosan hydrogel films for potential wound dressing. Carbohyd Polym. 2015;125:189–199. doi: 10.1016/j.carbpol.2015.02.034. [DOI] [PubMed] [Google Scholar]
- 46.Liu X.F., Guan Y.L., Yang Z.D., Li Z., Yao K.D. Antibacterial action of chitosan and carboxymethylated chitosan. J Appl Polym Sci. 2001;79(7):1324–1335. [Google Scholar]
- 47.Zhou Y., Yang D., Chen X., Xu Q., Lu F., Nie J. Electrospun water-soluble carboxyethyl chitosan/poly(vinyl alcohol) nanofibrous membrane as potential wound dressing for skin regeneration. Biomacromolecules. 2008;9:349–354. doi: 10.1021/bm7009015. [DOI] [PubMed] [Google Scholar]
- 48.Tashiro T. Antibacterial and bacterium adsorbing macromolecules Macromol. Mater Eng. 2001;286:63–87. [Google Scholar]
- 49.Zhao L., Mitomo H., Zhai M., Yoshii F., Nagasawa N., Kume T. Synthesis of antibacterial PVA/CM-chitosan blend hydrogels with electron beam irradiation. Carbohyd Polym. 2003;53:439–446. [Google Scholar]
- 50.Ignatova M., Manolova N., Rashkov I. Novel antibacterial fibers of quaternized chitosan and poly(vinyl pyrrolidone) prepared by electrospinning. Europ Polym J. 2007;43:1112–1122. [Google Scholar]
- 51.Dias A.M., Braga M.E., Seabra I.J., Ferreira P., Gil M.H., de Sousa H.C. Development of natural-based wound dressings impregnated with bioactive compounds and using supercritical carbon dioxide. Int J Pharm. 2011;408(1–2):9–19. doi: 10.1016/j.ijpharm.2011.01.063. [DOI] [PubMed] [Google Scholar]
- 52.Lee K.Y., Mooney D.J. Alginate: properties and biomedical applications. Prog Polym Sci. 2012;37:106–126. doi: 10.1016/j.progpolymsci.2011.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Thomas S. Alginate dressings in surgery and wound management- Part 3. J Wound Care. 2000;9(4):163–166. doi: 10.12968/jowc.2000.9.4.25973. [DOI] [PubMed] [Google Scholar]
- 54.Kim J.O., Choi J.Y., Park J.K., Kim J.H., Jin S.G., Chang S.W. Development of clindamycin-loaded wound dressing with polyvinyl alcohol and sodium alginate. Biolog Pharm Bull. 2008;31:2277–2288. doi: 10.1248/bpb.31.2277. [DOI] [PubMed] [Google Scholar]
- 55.Nam S.Y., Nho Y.C., Hong S.H., Chae G.T., Jang H.S., Suh T.S. Evaluation of poly(vinyl alcohol) alginate hydrogels cross-lined by γ-ray irradiation technique. Macromol Res. 2004;12(2):219–224. [Google Scholar]
- 56.Tarun K., Gobi N. Calcium alginate/PVA blended nano fiber matrix for wound dressing. Ind J Fibr Tex Res. 2012;37:127–132. [Google Scholar]
- 57.Zhai M., Yoshii F., Kume T., Hashim K. Syntheses of PVA/starch grafted hydrogels by irradiation. Carbohyd Polym. 2002;50:295–303. [Google Scholar]
- 58.Kamoun E.A., Menzel H. HES-HEMA nanocomposite polymer hydrogel: swelling behavior and characterization. J Polym Res. 2012;19:9815–9865. [Google Scholar]
- 59.Sidebotham R.L. Dextrans. Adv Carbohyd Chem Biochem. 1974;30:371–444. doi: 10.1016/s0065-2318(08)60268-1. [DOI] [PubMed] [Google Scholar]
- 60.Fathi E., Atyabi N., Imani M., Alinejad Z. Physically crosslinked polyvinyl alcohol–dextran blend xerogels: morphology and thermal behavior. Carbohyd Polym. 2011;84:145–152. [Google Scholar]
- 61.Lee S.B., Jeon H.W., Lee Y.M., Lee Y.W., Song K.W., Park M.H. Bio-artificial skin composed of gelatin and (1–3), (1–6)-β-Glucan. Biomater. 2003;24:2503–2511. doi: 10.1016/s0142-9612(03)00003-6. [DOI] [PubMed] [Google Scholar]
- 62.Huang M.H., Yang M.C. Evaluation of glucan/poly(vinyl alcohol) blend wound dressing using rat models. Int J Pharm. 2008;346:38–46. doi: 10.1016/j.ijpharm.2007.06.021. [DOI] [PubMed] [Google Scholar]
- 63.Hago E.E., Li X. Interpenetration polymer network hydrogels based on gelatin and PVA by biocompatible approaches: synthesis and characterization. Adv Mater Sci Eng. 2013:1–8. [Google Scholar]
- 64.Pietramaggiori G., Yang H.J., Scherer S.S., Kaipainen A., Chan R.K., Alperovich M. Effects of poly-N-acetyl glucosamine (pGlcNAc) patch on wound healing in db/db mouse. J Trauma. 2008;64(3):803–808. doi: 10.1097/01.ta.0000244382.13937.a8. [DOI] [PubMed] [Google Scholar]
- 65.Kirker K.R., Luo Y., Nielson J.H., Shelby J., Prestwich G.D. Glycosaminoglycan hydrogel films as bio-interactive dressings for wound healing. Biomaterials. 2002;23:3661–3671. doi: 10.1016/s0142-9612(02)00100-x. [DOI] [PubMed] [Google Scholar]
- 66.Park S.N., Lee H.J., Lee K.H., Suh H. Biological characterization of EDC-crosslinked collagen- hyaluronic acid matrix in dermal tissue restoration. Biomaterials. 2003;24:1631–1641. doi: 10.1016/s0142-9612(02)00550-1. [DOI] [PubMed] [Google Scholar]
- 67.Price R.D., Myers S., Leigh I.M., Navsaria H.A. The role of hyaluronic acid in wound healing: assessment of clinical evidence. Am J Clin Dermatol. 2005;6(6):393–402. doi: 10.2165/00128071-200506060-00006. [DOI] [PubMed] [Google Scholar]
- 68.Miller R.S., Steward D.L., Tami T.A., Sillars M.J., Seiden A.M., Shete M. The clinical effects of hyaluronic acid ester nasal dressing (Merogel) on intranasal wound healing after functional endoscopic sinus surgery. Otolaryngol-Heat Neck Surg. 2003;128:862–869. doi: 10.1016/S0194-59980300460-1. [DOI] [PubMed] [Google Scholar]
- 69.Fahmy A., Kamoun E.A., El-Eisawy R., El-Fakharany E.M., Taha T.H., El-Damhougy B.K. Poly(vinyl alcohol)-hyaluronic acid membranes for wound dressing applications: synthesis and in vitro bio-evaluations. J Braz Chem Soc. 2015;26(7):1357–1366. [Google Scholar]
- 70.Wen X., Zheng Y., Wu J., Yue L., Wang C., Luan J. In vitro and in vivo investigation of bacterial cellulose dressing containing uniform silver sulfadiazine nanoparticles for burn wound healing. Prog Nat Sci: Mater Int. 2015;25(3):197–203. [Google Scholar]
- 71.Wu J., Zheng Y., Song W., Luan J., Wen X., Wu Z. In situ synthesis of silver-nanoparticles/bacterial cellulose composites for slow-released antimicrobial wound dressing. Carbohyd Polym. 2014;15(102):762–771. doi: 10.1016/j.carbpol.2013.10.093. [DOI] [PubMed] [Google Scholar]
- 72.Kalin M., Kuru S., Kismet K., Barsal A.M., Akgun Y.A., Astarci H.M. The effectiveness of pocine dermal collagen (Permacol) on wound healing in the rat model. Ind J Surg. 2013;1:1–5. doi: 10.1007/s12262-013-0854-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Padol A.R., Jayakumar K., Shridhar N., Swamy H.N., Swamy M.N., Mohan K. Safety evaluation of skin protein film (A novel wound healing agent) in terms of acute dermal toxicity, acute dermal irritation and skin sensitization. Toxicol Int. 2011;18(1):17–21. doi: 10.4103/0971-6580.75847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Roh D.H., Kang S.Y., Kim J.Y., Kwon Y.B., Young-Kweon H., Lee K.G. Wound healing effect of skin fibroin/alginate-blended sponge in full thickness skin defect of rat. J Mater Sci Mater Med. 2006;17(6):547–552. doi: 10.1007/s10856-006-8938-y. [DOI] [PubMed] [Google Scholar]
- 75.Wright K.A., Nadire K.B., Busto P., Tubo R., McPherson J.M., Wentworth B.M. Alternative delivery of keratinocytes using a polyurethane membrane and the implications for its use in the treatment of full-thickness burn injury. Burns. 1998;24(1):7–17. doi: 10.1016/s0305-4179(97)00075-2. [DOI] [PubMed] [Google Scholar]
- 76.Nakabayashi N. Dental biomaterials and the healing of dental tissues. Biomaterials. 2003;24:2437–2439. doi: 10.1016/s0142-9612(03)00112-1. [DOI] [PubMed] [Google Scholar]
- 77.Lee J.J., Worthington P. Reconstruction of the temporomandibular joint using calvarial bone after a failed Teflon-Proplast implant. J Oral Maxillofac Surg. 1999;57(4):457–461. doi: 10.1016/s0278-2391(99)90290-2. [DOI] [PubMed] [Google Scholar]
- 78.Senyuva C., Yücel A., Erdamar S., Cetinkale O., Seradjmir M., Ozdemir C. The fate of alloplastic materials placed under a burn scar: an experimental study. Burns. 1997;23(6):484–489. doi: 10.1016/s0305-4179(97)00043-0. [DOI] [PubMed] [Google Scholar]
- 79.Grote J.J., Bakker D., Hesseling S.C., van Blitterswijk C.A. New alloplastic tympanic membrane material. Am J Otol. 1991;12(5):329–335. [PubMed] [Google Scholar]
- 80.Himly N., Darwis D., Hardiningsih L. Poly(n-vinylpyrrolidone) hydrogels: 2.Hydrogel composites as wound dressing for tropical environment. Radiat Phys Chem. 1993;42(4-6):911–914. [Google Scholar]
- 81.Singh B., Pal L. Radiation crosslinking polymerization of sterculia polysaccharide-PVA-PVP for making hydrogel wound dressings. Int J Biolog Macromol. 2011;48:501–510. doi: 10.1016/j.ijbiomac.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 82.Park K.R., Chang N.Y. Synthesis of PVA/PVP hydrogels having two-layer by radiation and their physical properties. Rad Phys Chem. 2003;67:361–365. [Google Scholar]
- 83.Wu M., Bao B., Yoshii F., Makuuchi K. Irradiation of crosslinked, poly(vinyl alcohol) blended hydrogel for wound dressing. J Rad Nuclear Chem. 2001;250(2):391–395. [Google Scholar]
- 84.Uslu I., Keskin S., Gul A., Karabulut T.C., Aksu M.L. Preparation and properties of electrospun poly(vinyl alcohol) blended hybrid polymer with Aloe veraand HPMC as wound dressing. Hacettepe J Biol Chem. 2010;38(1):19–25. [Google Scholar]
- 85.Dutta J. Synthesis and characterization of γ -irradiated PVA/PEG/CaCl2 hydrogel for wound dressing. Am J Chem. 2012;2(2):6–11. [Google Scholar]
- 86.Azarbayjani A.F., Venugopal J.R., Ramakrishna S., Lim P.F.C., Chan Y.W., Chan S.Y. Smart polymeric nanofibers for topical delivery of levothyroxine. J Pharm Pharmaceut Sci. 2010;13(3):400–410. doi: 10.18433/j3ts3g. [DOI] [PubMed] [Google Scholar]
- 87.Moghadas B., Dashtimoghadam E., Mirzadeh H., Seidi F., Hasani-Sadrabadi M.M. Novel chitosan-based nanobiohybrid membranes for wound dressing applications. RSC Adv. 2016;6:7701–7711. [Google Scholar]
- 88.Gonzalez J.S., Maiolo A.S., Ponce A.G., Alvarez V.A. Composites based on poly (vinyl alcohol) hydrogels for wound dressing. XVIII The Argentine Congress of Bioengineering and Clinical Engineering Conference VII, SABI. 2011;2011:1–4. [Google Scholar]
- 89.Shalumon K.T., Anulekha K.H., Nair S.V., Chennazhi K.P., Jayakumar R. Sodium alginate/poly(vinyl alcohol) nano ZnO composite nanofibers for antibacterial wound dressing. Int J Biolog Macromol. 2011;49:247–254. doi: 10.1016/j.ijbiomac.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 90.Vicentini DS, Jr AS, Laranjeira MCM. Chitosan/poly (vinyl alcohol) films containing ZnO nanoparticles and plasticizers. Mater Sci Eng C 2010; 30: 503-508.
- 91.Sudheesh Kumar P.T., Lakshmanan V.K., Anilkumar T.V., Ramya C., Reshmi P., Unnikrishnan A.G. Flexible and microporous chitosan hydrogel/nano ZnO composite bandages for wound dressing. in vitro and in vivo evaluation. ACS Appl Mater Interf. 2012;4(5):2618–2629. doi: 10.1021/am300292v. [DOI] [PubMed] [Google Scholar]
- 92.Păunica-Panea G., Ficai A., Marin M.M., Marin S., Albu M.G., Constantin V.D. New collagen-dextran-zinc oxide composites for wound dressing. J Nanomater. 2016;2016:1–7. [Google Scholar]
- 93.Mohandas A., Kumar P.T.S., Raja B., Lakshmanan V.K., Jayakumar R. Exploration of alginate hydrogel/nano zinc oxide composite bandages for infected wounds. Int J Nanomed. 2015;10:53–66. doi: 10.2147/IJN.S79981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Fan Z., Liu B., Wang j., Zhang S., Lin Q., Gong P. A novel wound dressing based on Ag/graphene polymer hydrogel: effectively kill bacteria and accelerate wound healing. Adv Funct Mater. 2014;24:3933–3943. [Google Scholar]
- 95.Usman A., Hussain Z., Riaz A., Khan A.N. Enhanced mechanical, thermal and antimicrobial properties of poly(vinyl alcohol)/graphene oxide/starch/silver nanocomposites. Carbohyd Polym. 2016;153:592–599. doi: 10.1016/j.carbpol.2016.08.026. [DOI] [PubMed] [Google Scholar]
- 96.Li C., Fu R., Yu C., Li Z., Guan H., Hu D. Silver nanoparticle/chitosan oligosaccharide/poly(vinyl alcohol) nanofibers as wound dressing: a preclinical study. Int J Nanomed. 2013;8:4131–4145. doi: 10.2147/IJN.S51679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mota J., Yu N., Caridade S.G., Luz G.M., Gomes M.E., Reis R.L. Chitosan/bioactive glass nanoparticle composite membranes for periodontal regeneration. Acta Biomater. 2012;8:4173–4180. doi: 10.1016/j.actbio.2012.06.040. [DOI] [PubMed] [Google Scholar]