Abstract
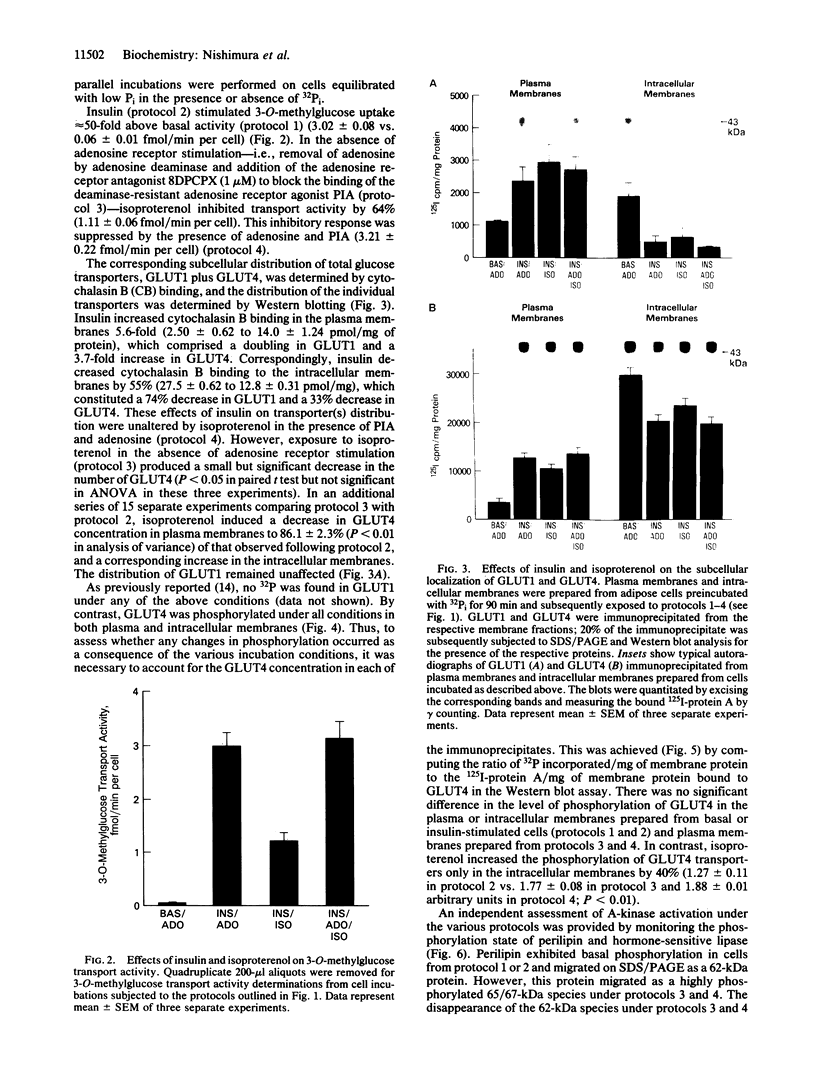
The acute effects of insulin, adenosine, and isoproterenol on the activity, subcellular distribution, and phosphorylation state of the GLUT4 glucose transporter isoform were investigated in rat adipocytes under conditions carefully controlled to monitor changes in cAMP-dependent protein kinase (A-kinase) activity. In contrast to GLUT1, which has not been shown to be phosphorylated even when cells are exposed to any of the above agents, GLUT4 was partially phosphorylated (0.1-0.2 mol/mol) when the activity of the A-kinase was suppressed, and remained unchanged in response to insulin. Isoproterenol elicited a 64% inhibition of insulin-stimulated glucose transport activity in the absence, but not the presence, of adenosine receptor agonists. However, in either the presence or the absence of agonists, A-kinase was activated as assessed by examining the phosphorylation of the major adipocyte A-kinase substrate, perilipin. Similarly, under either condition, phosphorylation of GLUT4 was enhanced 1.4-fold in the intracellular membranes, but no significant change was observed in the plasma membrane. In the absence of adenosine receptor agonists, isoproterenol exerted a small (14%) but significant inhibition of the insulin-induced translocation of GLUT4 but had no effect on the translocation of GLUT1. Thus, changes in the phosphorylation state and/or subcellular distribution of GLUT4 cannot account for the inhibition of insulin-stimulated glucose activity induced by isoproterenol.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beavo J. A., Bechtel P. J., Krebs E. G. Preparation of homogeneous cyclic AMP-dependent protein kinase(s) and its subunits from rabbit skeletal muscle. Methods Enzymol. 1974;38:299–308. doi: 10.1016/0076-6879(74)38046-9. [DOI] [PubMed] [Google Scholar]
- Bell G. I., Kayano T., Buse J. B., Burant C. F., Takeda J., Lin D., Fukumoto H., Seino S. Molecular biology of mammalian glucose transporters. Diabetes Care. 1990 Mar;13(3):198–208. doi: 10.2337/diacare.13.3.198. [DOI] [PubMed] [Google Scholar]
- Birnbaumer L. G proteins in signal transduction. Annu Rev Pharmacol Toxicol. 1990;30:675–705. doi: 10.1146/annurev.pa.30.040190.003331. [DOI] [PubMed] [Google Scholar]
- Cushman S. W., Wardzala L. J. Potential mechanism of insulin action on glucose transport in the isolated rat adipose cell. Apparent translocation of intracellular transport systems to the plasma membrane. J Biol Chem. 1980 May 25;255(10):4758–4762. [PubMed] [Google Scholar]
- Cushman S. W., Wardzala L. J. Potential mechanism of insulin action on glucose transport in the isolated rat adipose cell. Apparent translocation of intracellular transport systems to the plasma membrane. J Biol Chem. 1980 May 25;255(10):4758–4762. [PubMed] [Google Scholar]
- Egan J. J., Chang M. K., Londos C. A tandem chromatographic column method for assaying cAMP-dependent protein kinase and protein kinase C with synthetic peptide substrates. Anal Biochem. 1988 Dec;175(2):552–561. doi: 10.1016/0003-2697(88)90583-0. [DOI] [PubMed] [Google Scholar]
- Egan J. J., Greenberg A. S., Chang M. K., Londos C. Control of endogenous phosphorylation of the major cAMP-dependent protein kinase substrate in adipocytes by insulin and beta-adrenergic stimulation. J Biol Chem. 1990 Nov 5;265(31):18769–18775. [PubMed] [Google Scholar]
- Green A. Glucagon inhibition of insulin-stimulated 2-deoxyglucose uptake by rat adipocytes in the presence of adenosine deaminase. Biochem J. 1983 Apr 15;212(1):189–195. doi: 10.1042/bj2120189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenberg A. S., Egan J. J., Wek S. A., Garty N. B., Blanchette-Mackie E. J., Londos C. Perilipin, a major hormonally regulated adipocyte-specific phosphoprotein associated with the periphery of lipid storage droplets. J Biol Chem. 1991 Jun 15;266(17):11341–11346. [PubMed] [Google Scholar]
- Haspel H. C., Birnbaum M. J., Wilk E. W., Rosen O. M. Biosynthetic precursors and in vitro translation products of the glucose transporter of human hepatocarcinoma cells, human fibroblasts, and murine preadipocytes. J Biol Chem. 1985 Jun 25;260(12):7219–7225. [PubMed] [Google Scholar]
- Holman G. D., Kozka I. J., Clark A. E., Flower C. J., Saltis J., Habberfield A. D., Simpson I. A., Cushman S. W. Cell surface labeling of glucose transporter isoform GLUT4 by bis-mannose photolabel. Correlation with stimulation of glucose transport in rat adipose cells by insulin and phorbol ester. J Biol Chem. 1990 Oct 25;265(30):18172–18179. [PubMed] [Google Scholar]
- Hopkirk T. J., Denton R. M. Studies on the specific activity of [gamma-32P]ATP in adipose and other tissue preparations incubated with medium containing [32P]phosphate. Biochim Biophys Acta. 1986 Feb 21;885(2):195–205. doi: 10.1016/0167-4889(86)90089-3. [DOI] [PubMed] [Google Scholar]
- James D. E., Brown R., Navarro J., Pilch P. F. Insulin-regulatable tissues express a unique insulin-sensitive glucose transport protein. Nature. 1988 May 12;333(6169):183–185. doi: 10.1038/333183a0. [DOI] [PubMed] [Google Scholar]
- James D. E., Hiken J., Lawrence J. C., Jr Isoproterenol stimulates phosphorylation of the insulin-regulatable glucose transporter in rat adipocytes. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8368–8372. doi: 10.1073/pnas.86.21.8368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joost H. G., Weber T. M., Cushman S. W., Simpson I. A. Activity and phosphorylation state of glucose transporters in plasma membranes from insulin-, isoproterenol-, and phorbol ester-treated rat adipose cells. J Biol Chem. 1987 Aug 15;262(23):11261–11267. [PubMed] [Google Scholar]
- Kahn B. B., Flier J. S. Regulation of glucose-transporter gene expression in vitro and in vivo. Diabetes Care. 1990 Jun;13(6):548–564. doi: 10.2337/diacare.13.6.548. [DOI] [PubMed] [Google Scholar]
- Karnieli E., Zarnowski M. J., Hissin P. J., Simpson I. A., Salans L. B., Cushman S. W. Insulin-stimulated translocation of glucose transport systems in the isolated rat adipose cell. Time course, reversal, insulin concentration dependency, and relationship to glucose transport activity. J Biol Chem. 1981 May 25;256(10):4772–4777. [PubMed] [Google Scholar]
- Kashiwagi A., Huecksteadt T. P., Foley J. E. The regulation of glucose transport by cAMP stimulators via three different mechanisms in rat and human adipocytes. J Biol Chem. 1983 Nov 25;258(22):13685–13692. [PubMed] [Google Scholar]
- Kuroda M., Honnor R. C., Cushman S. W., Londos C., Simpson I. A. Regulation of insulin-stimulated glucose transport in the isolated rat adipocyte. cAMP-independent effects of lipolytic and antilipolytic agents. J Biol Chem. 1987 Jan 5;262(1):245–253. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lawrence J. C., Jr, Hiken J. F., James D. E. Phosphorylation of the glucose transporter in rat adipocytes. Identification of the intracellular domain at the carboxyl terminus as a target for phosphorylation in intact-cells and in vitro. J Biol Chem. 1990 Feb 5;265(4):2324–2332. [PubMed] [Google Scholar]
- Lawrence J. C., Jr, Hiken J. F., James D. E. Stimulation of glucose transport and glucose transporter phosphorylation by okadaic acid in rat adipocytes. J Biol Chem. 1990 Nov 15;265(32):19768–19776. [PubMed] [Google Scholar]
- Lawrence J. C., Jr, James C. Activation of glycogen synthase by insulin in rat adipocytes. Evidence of hormonal stimulation of multisite dephosphorylation by glucose transport-dependent and -independent pathways. J Biol Chem. 1984 Jun 25;259(12):7975–7982. [PubMed] [Google Scholar]
- Londos C., Cooper D. M., Rodbell M. Receptor-mediated stimulation and inhibition of adenylate cyclases: the fat cell as a model system. Adv Cyclic Nucleotide Res. 1981;14:163–171. [PubMed] [Google Scholar]
- Shibata Y., Flanagan J. E., Smith M. M., Robinson F. W., Kono T. Sedimentation characteristics of vesicles associated with insulin-sensitive intracellular glucose transporter from rat adipocytes. Biochim Biophys Acta. 1987 Aug 7;902(1):154–158. doi: 10.1016/0005-2736(87)90147-7. [DOI] [PubMed] [Google Scholar]
- Simpson I. A., Cushman S. W. Hormonal regulation of mammalian glucose transport. Annu Rev Biochem. 1986;55:1059–1089. doi: 10.1146/annurev.bi.55.070186.005211. [DOI] [PubMed] [Google Scholar]
- Smith P. K., Krohn R. I., Hermanson G. T., Mallia A. K., Gartner F. H., Provenzano M. D., Fujimoto E. K., Goeke N. M., Olson B. J., Klenk D. C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985 Oct;150(1):76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- Smith U., Kuroda M., Simpson I. A. Counter-regulation of insulin-stimulated glucose transport by catecholamines in the isolated rat adipose cell. J Biol Chem. 1984 Jul 25;259(14):8758–8763. [PubMed] [Google Scholar]
- Suzuki K., Kono T. Evidence that insulin causes translocation of glucose transport activity to the plasma membrane from an intracellular storage site. Proc Natl Acad Sci U S A. 1980 May;77(5):2542–2545. doi: 10.1073/pnas.77.5.2542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor W. M., Mak M. L., Halperin M. L. Effect of 3':5'-cyclic AMP on glucose transport in rat adipocytes. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4359–4363. doi: 10.1073/pnas.73.12.4359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zorzano A., Wilkinson W., Kotliar N., Thoidis G., Wadzinkski B. E., Ruoho A. E., Pilch P. F. Insulin-regulated glucose uptake in rat adipocytes is mediated by two transporter isoforms present in at least two vesicle populations. J Biol Chem. 1989 Jul 25;264(21):12358–12363. [PubMed] [Google Scholar]