Abstract
Objective
Diabetic sensorimotor peripheral neuropathy (DSP) is the most prevalent complication in diabetes mellitus. Identifying DSP risk is essential for intervening early in the natural history of the disease. Small nerve fibers are affected earliest in the disease progression and evidence of this damage can be identified using in vivo corneal confocal microscopy (IVCCM).
Research design and methods
We applied IVCCM to a cohort of 40 patients with type 1 diabetes to identify their DSP risk profile. We measured standard IVCCM parameters including corneal nerve fiber length (CNFL), and performed nerve conduction studies and quantitative sensory testing.
Results
40 patients (53% female), with a mean age of 48±14, BMI 28.1±5.8, and diabetes duration of 27±18 years were enrolled between March 2014 and June 2015. Mean IVCCM CNFL was 12.0±5.2 mm/mm2 (normal ≥15 mm/mm2). Ten patients (26%) without DSP were identified as being at risk of future DSP with mean CNFL 11.0±2.1 mm/mm2. Six patients (15%) were at low risk of future DSP with mean CNFL 19.0±4.6 mm/mm2, while 23 (59%) had established DSP with mean CNFL 10.5±4.5 mm/mm2.
Conclusions
IVCCM can be used successfully to identify the risk profile for DSP in patients with type 1 diabetes. This methodology may prove useful to classify patients for DSP intervention clinical trials.
Keywords: Neuropathy, Neuropathy Clinical Trials, CCM, Risk Assessment
What is already known about this subject?
In vivo corneal confocal microscopy (IVCCM) is a non-invasive technique that measures small nerve fiber structure. Change in IVCCM corneal nerve fiber length (CNFL) is a biomarker for diabetic sensorimotor polyneuropathy (DSP) progression.
IVCCM measurement of CNFL can be used to diagnose DSP.
What are the new findings?
This paper reports the use of IVCCM measurement to identify the DSP risk profile in patients with type 1 diabetes.
IVCCM measurement of CNFL can stratify DSP risk in patients with type 1 diabetes with a broad spectrum of sensory and motor nerve impairments.
How might these results change the focus of research or clinical practice?
We identified subgroups of patients at high and low future risk of developing DSP and those with established DSP.
This finding will allow for better selection of patients for clinical trials and also for more accurate monitoring of DSP progression.
Introduction
Diabetic sensorimotor polyneuropathy (DSP) is a diffuse and length-dependent injury to peripheral nerves in diabetes mellitus (DM) that impairs function of peripheral sensory and motor nerves.1 DSP is the most common complication of DM affecting at least 50% of the population.2 In the natural history of the disease, small nerve fiber impairment represents the earliest nerve injury in the development of DSP, prior to the development of large fiber injury.3 Symptoms of DSP include pain, numbness, and loss of sensation in feet and legs, and impaired balance and walking. This increases the risk of falls, foot trauma, ulceration, and amputation.4
Our research group and others have recently validated the use of in vivo corneal confocal microscopy (IVCCM) to measure changes in corneal nerve fiber length (CNFL) as a screening tool, diagnosis method, and biomarker of DSP disease progression in type 1 diabetes mellitus (T1DM).5–12 The small, thinly myelinated, and unmyelinated fibers in the cornea reflect the loss of small fibers in other peripheral nerves. Our group and others have reported that impairments in this small nerve fiber measure correlate well with measures of small6 12 and large7 nerve dysfunctions. Recently, we have identified that individuals with IVCCM CNFL below a threshold of 15 mm/mm2 are at risk of developing future DSP11 while normal is >15 mm/mm2.13
In clinical trials of novel therapeutic agents for DSP, it is crucial to select the best study population to show the potential effects of treatment. We aimed to determine whether IVCCM could identify the DSP risk profile in a cohort of patients with T1DM.
Methods
This cross-sectional study characterizes DSP risk in 40 patients with T1DM using IVCCM.
Ethics statement
The study was performed at the Prosserman Family Neuromuscular Clinic at the University Health Network, Toronto, Canada. The protocol and consent procedures were conducted in accordance with Health Canada good clinical practice and were approved by the University Health Network Research Ethics Board. All study patients were at least 18 years of age and provided written informed consent.
Study patients
Individuals of any gender or race aged 18 or above; with previously diagnosed T1DM as defined by the 2008 Canadian Diabetes Association guidelines of any duration; and a Toronto Clinical Neuropathy Score (TCNS) ≥1 were included in the study. The TCNS was used to ensure patients had a sign or symptom of neuropathy and to classify DSP severity (no neuropathy (0–5), mild (6–8), moderate (9–11), and severe (>12) neuropathy).14 To ensure a broad spectrum of DSP, at least seven patients were included in each of the no to moderate neuropathy strata. Patients were also evaluated using established DSP consensus criteria.15 Individuals were excluded if they presented with neuropathy due to non-diabetic causes (eg, familial, alcoholic, nutritional, uremic, etc), current eye infection, corneal damage or severe movement disorder to preclude safe IVCCM examination, or an allergy to proparacaine (the ocular topical anesthetic used for the IVCCM examination).
Study procedures
In vivo corneal confocal microscopy
Patients had bilateral examination of the nerve plexus adjacent to the Bowman's layer of the cornea using the Rostock Cornea Module of the Heidelberg Tomograph III (Heidelberg Engineering, Smithfield RI, USA) to determine CNFL according to our validated procedure.7 9 10 16 Topical anesthetic (Proparacaine hydrochloride 0.5%; Alcan, Mississauga, Canada) and a viscous gel (Tear-gel; Novartis Pharmaceuticals, Dorval, Canada) were applied to the eye to establish a gel bridge between the cornea and the sterile single-use lens cap on the microscope objective lens. After the interface between the corneal epithelium and Bowman's layer was identified, 40 images, taken over a depth of 50 microns in 1.3 micron incremental steps, were obtained using the semiautomated volume scan. This was performed using a 300 μm lens. The most technically sound image from each eye and each magnification lens was analyzed manually, and the parameters were measured using semiautomated analytical software (CCMetrics Image Analysis Software v1.1). CNFL in mm/mm−2 was calculated semiautomatically by having the examiner digitally trace over the nerve fibers and branches of the selected images. The lengths of nerve fibers present in the field were summated to produce the measure in mm standardized to the area of the field in mm2. An automated analysis procedure was also used for comparison (ACCMetrics Image Analysis Software v2.0).5 The number of nerve fibers, branches, and nerve fiber length were divided by the field of view for each lens to provide corneal nerve fiber density (CNFD) (fibers/mm2), corneal nerve branch density (CNBD) (branches/mm2), and CNFL (mm/mm2).7–9
DSP risk profile
DSP risk was classified using IVCCM CNFL and established consensus criteria for DSP diagnosis.15 Patients with no DSP and CNFL <15 mm/mm2 were considered at high risk for future DSP. Patients with no DSP and CNFL >15 mm/mm2 were considered at low risk for future DSP.
Large nerve fiber function
Nerve conduction studies were performed in 39 patients. Ten sensory and motor parameters from the dominant limb peroneal and sural nerves and the non-dominant limb median and ulnar nerves were obtained using the Counterpoint device (Alpine Biomed Corporation; Fountain Valley, USA) according to the standards of the American Association for Neuromuscular and Electrodiagnostic Medicine.15 These include: distal motor and sensory latencies, sensory nerve action potential amplitudes, compound muscle action potential amplitudes, F wave latencies, and motor and sensory nerve conduction velocities.
Vibration perception thresholds testing was performed using the method of limits algorithm17 (Neurothesiometer; Bailey Instruments Limited, UK).
Small nerve fiber function
Three tests of small fibers included: cooling detection thresholds, axon reflex-mediated neurogenic vasodilation in response to cutaneous heating by the laser Doppler imaging flare technique (LDIFLARE) and heart rate variability (HRV). Cooling-detection thresholds testing was done using method of limits algorithms17 (Medoc TSA-II NeuroSensory Analyzer; Ramat-Yishai, Israel). LDIFLARE was measured using a MoorLDI2 Laser Doppler blood perfusion imager (Moor Instruments, Axminster, UK).18 HRV function was measured for 1 min at rest and during deep breathing. Changes in heart rate (adaptation of the diabetes control and complications trial protocol)19 and R-R Variation (on the Counterpoint EMG device) were measured using the beat-to-beat HRV as endorsed by the American Diabetes Association20 and implemented by our research group.21
Clinical Assessment
All patients underwent a comprehensive medical and neurological evaluation, including detailed assessments of symptoms and signs of neuropathy, comorbidities, and biochemical tests including glycated hemoglobin A1c, serum lipids, thyroid stimulating hormone, creatinine, urinary albumin excretion, and serum C reactive protein concentration.
Statistical Analysis
All statistical analysis was performed using SPSS v22 (IBM, Armonk, New York, USA). Descriptive data are shown as means ±SD for normally distributed data, or means ±IQR for non-parametric data. Comparison of IVCCM and small and large fiber tests were performed using independent samples t-test for normally distributed or Mann-Whitney rank sum test for non-parametric data with patients separated into no-DSP (DSP (−)) or diagnosed-DSP (DSP(+)) according to England et al.15 We used qualitative analysis of a 2×2 contingency table to explore the frequency of DSP with IVCCM CNFL </> 15 mm/mm2 and a one-way analysis of variance to compare the difference in CNFL between groups and subgroup analysis of clinical variables and CNFL. Statistical significance for all tests was set at p≤0.05.
Results
Between March 2014 and June 2015, 46 individuals with T1DM were evaluated of whom 40 were eligible and enrolled in the study. Baseline demographic data for the study patients are shown in table 1. One participant declined to participate in nerve conduction studies (NCS), therefore DSP criteria and NCS data are shown as n=39, while IVCCM measures were available on all 40 participants. TCNS classification of the patients shows a broad spectrum of DSP in those recruited: 16 (40%) had no neuropathy (TCNS 1–5), 14 (35%) had mild neuropathy (TCNS 6–8), and 10 (25%) had moderate-neuropathy (TCNS 9–11). DSP criteria showed that 16 (41%) had no neuropathy while 23 (59%) had neuropathy.
Table 1.
Baseline demographic data on the 40 type 1 diabetes study participants
| Clinical characteristics | |
| Age (y) | 48±14 |
| Female, n (%) | 21 (53) |
| Diabetes duration (y) | 27±14 |
| BMI | 28.1±5.8 |
| SBP/DBP (mm Hg) | 126/70±13/9 |
| Smoking, n (%) | 7 (18) |
| Alcohol use (%) | 32 (80) |
| Family history of diabetes (%) | 22 (55) |
| Family history of neuropathy (%) | 5 (13) |
| Toronto Clinical Neuropathy Score | |
| No neuropathy (1–5) (%) | 16 (40) |
| Mild neuropathy (6–8) (%) | 14 (35) |
| Moderate neuropathy (9–11) (%) | 10 (25) |
| DSP (England criteria)(n=39) | |
| Absent neuropathy (%) | 16 (41) |
| Diagnosed neuropathy (%) | 23 (59) |
| IVCCM CNFL | |
| 300 μm lens manual (mm/mm2) | 12.0±5.2 |
| 300 μm lens automated (mm/mm2) | 8.3±2.3 |
| No DSP, low risk—CNFL >15 mm/mm2 (n) |
19.0±4.6 (6) |
| No DSP, high risk—CNFL <15 mm/mm2, (n) |
11.0±2.1(10) |
| Established DSP—CNFL <15 mm/mm2, (n) |
10.5±4.5 (23) |
| Biochemical characteristics | |
| A1C (%) | 7.6±1.0 |
| Total cholesterol (mmol/L) | 4.23±0.95 |
| LDL cholesterol (mmol/L) | 2.20±0.70 |
| HDL cholesterol (mmol/L | 1.61±0.44 |
| Non-HDL cholesterol (mmol/L) | 2.61±0.91 |
| Triglycerides (mmol/L) | 1.25±2.20 |
| TSH (mIU/L) | 2.28±1.41 |
| Creatinine (μmol/L) | 72±17 |
| eGFR (mL/min/1.73 m2) | 87±16 |
| CRP (mg/L) | 3.8±7.5 |
BMI, body mass index; CRP, C reactive protein; CNFL, corneal nerve fiber length; DBP, diastolic blood pressure; DSP, diabetic sensorimotor polyneuropathy; eGFR, estimated glomerular filtration rate; HDL, high-density lipoproteins; LDL, low-density lipoproteins; SBP, systolic blood press; TSH, thyroid stimulating hormone.
CNFL using manual tracing showed a group mean of 12.0±5.2 mm/mm2 and automated tracing showed a group mean of 8.3±3.1 mm/mm2. CNFL measured by manual tracing were significantly longer than automated tracing by 41±3% (p<0.01). CNBD and CNFD were also higher when measured by manual tracing (p≤0.01). Comparison of the CNFL for those with established DSP and no DSP shows a significant difference between groups (p=0.04) (figure 1A).
Figure 1.
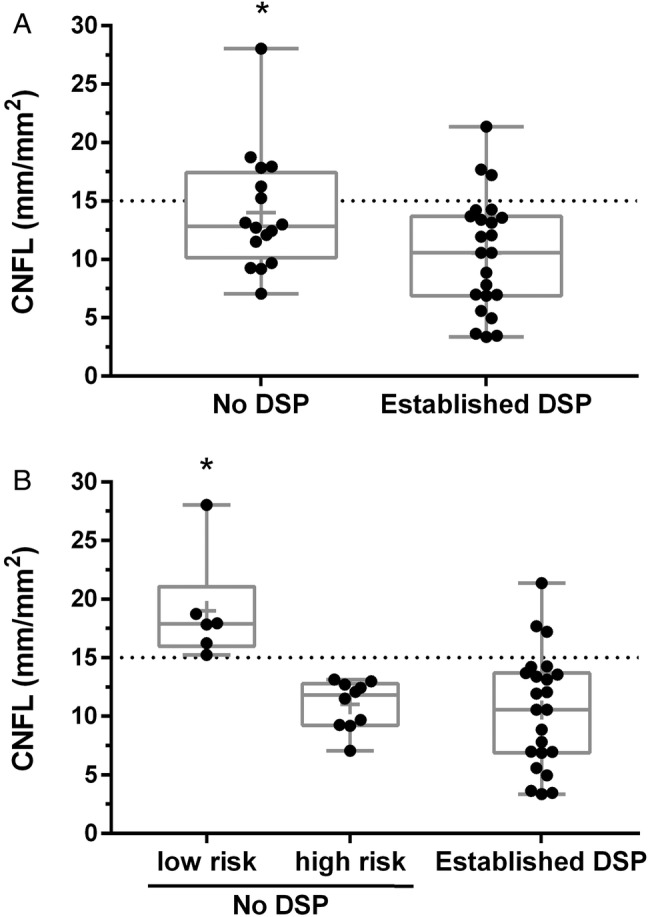
Box- and whisker plots demonstrating the distribution of 300 μm manual trace corneal nerve fiber length (CNFL) in 39 type 1 diabetic participants. The dashed line represents CNFL 15 mm/mm2. Panel A shows CNFL in those with and without diagnosed DSP. * Indicates significantly higher than Established DSP cases. Panel B shows participants divided into three groups based on DSP status and future risk: No DSP, low risk (CNFL >15 mm/mm2), No DSP, high risk (CNFL <14.9 mm/mm2), Established DSP. + Bonferroni post hoc analysis indicates significantly higher CNFL than other groups. CNFL, corneal nerve fiber length; DSP, diabetic sensorimotor polyneuropathy.
We examined the proportion of patients at high and low risk for future DSP using the 15 mm/mm2 CNFL threshold (table 2). There are 10 patients at high risk for future DSP and six that are at low future risk. A distribution of CNFL for patients grouped by DSP risk is shown in figure 1B. NCS and sensory nerve data showed no difference between high-risk and low-risk subgroups (p>0.05). Those without DSP had lower TCNS symptom scores and diabetes duration compared with those with established DSP (p≤0.04). NCS and sensory nerve function was better in patients with no DSP compared with those with established DSP (p<0.001) (table 3).
Table 2.
A 2×2 contingency table of in vivo corneal confocal microscopy (IVCCM) CNFL DSP threshold versus clinical DSP diagnosis
| England criteria |
||
|---|---|---|
| DSP (−) | DSP (+) | |
| 300 μm CNFLmanual >15 mm/mm2 | 6 | 3 |
| 300 μm CNFLmanual <15 mm/mm2 | 10 | 20 |
CNFL, corneal nerve fiber length; DSP, diabetic sensorimotor polyneuropathy.
Table 3.
Quantitative sensory testing, heart rate, and nerve conduction study data
| Group mean N=36 |
DSP(−) | DSP(+) | p Value | |
|---|---|---|---|---|
| Quantitative sensory testing | ||||
| VPT hand (volts) | 4.5±2.7 | 2.9±1.3 | 5.3±2.9 | 0.01 |
| VPT toe (volts) | 17.3±12.4 | 7.0±3.9 | 16.8±15.6 | <0.001 |
| Cooling detection threshold (°C) | 23.8±7.4 | 28.8±3.1 | 23.0±6.6 | 0.001 |
| LDIFLARE area (cm2) | 1.9±0.7 | 2.0±1.4 | 1.8±0.5 | 0.2 |
| Heart rate | ||||
| Resting heart rate (bpm) | 62±7 | 60±10 | 62±12 | 0.4 |
| Resting heart rate, deep breathing (bpm) | 62±8 | 60±7 | 61±9 | 0.4 |
| HRV normal breathing (%) | 15±10 | 24±20 | 11±9 | 0.03 |
| HRV deep breathing (%) | 24±16 | 28±34 | 18±18 | 0.03 |
| Nerve conduction study | ||||
| Sural nerve amplitude (μv) | 2.7±9.1 | 13.0±9.8 | 1.9±2.3 | <0.001 |
| Sural nerve conduction velocity (m/s) | 43.8±11.0 | 48.3±7.6 | 39.6±11.3 | <0.001 |
| Peroneal nerve amplitude, ankle (mV) | 3.2±4.6 | 5.8±2.9 | 1.6±3.0 | <0.001 |
| Peroneal nerve conduction velocity, fibular head (m/s) | 38.8±6.6 | 45.0±3.6 | 36.2±5.8 | <0.001 |
| Peroneal nerve F-wave (ms) | 55.5±18.3 | 48.6±5.1 | 57.8±19.2 | <0.001 |
HRV, heart rate variability, LDIFLARE, laser Doppler imaging flare; VPT, vibration perception threshold.
Conclusions
This paper reports the use of IVCCM to identify DSP risk in a cohort of patients with type 1 diabetes. The 40 patients enrolled in this study present with a range of diabetes duration (2 to 55 years) and a broad spectrum of DSP severity similar to previous investigations of DSP in T1DM(6; 18; 21). In this IVCCM analysis, we have identified a subgroup of patients without diagnosed DSP but predicted to be at high future risk of DSP. This finding provides the opportunity to intervene in the earliest stages of this disease.
IVCCM CNFL provides a novel measure of changes in small nerve fiber structure that precede large fiber electrophysiological changes.5–11 Previously, we proposed a diagnostic threshold of ≤14.0 mm/mm2 using a 300 μm lens and manual tracing to rule in DSP from a cross-sectional analysis.7 Findings from our recently published 4-year T1DM cohort study has supported the idea that IVCCM CNFL can serve as a predictive biomarker for the future risk of DSP.11 A threshold of <15 mm/mm2 predicted onset of DSP irrespective of the results of quantitative sensory testing and NCS.
In this analysis, 59% of patients have diagnosed DSP according to TCNS and current consensus criteria.15 Of the 41% of patients without DSP, 62% of these patients are at risk of developing future DSP base on IVCCM CNFL <15 mm/mm2 (table 2). This stratification of the T1DM population provides an opportunity to select patients early in the natural history of DSP and might allow IVCCM to detect early changes in small fiber structure in response to therapeutic interventions. Previous therapeutic trials in DSP have generally failed22–24 possibly because trial patients had advanced nerve damage that may not respond to therapy. IVCCM promises to identify a DSP population earlier in the natural history of the disease, which might be more amenable to intervention.
Limitations of this study are: (1) the IVCCM CNFL threshold of <15 mm/mm2 for determining the risk of future onset of DSP in T1DM,11 needs additional confirmation in future investigations with T1DM cohorts; and (2) this study population excludes patients with advanced DSP (TCNS >12) and therefore limits the generalizability of our findings. We recruited participants with a broad spectrum of neuropathy to limit self-selection of patients with severe DSP, as shown by our results. Image selection bias is minimal as a single research technician masked as to the clinical assessment s selected the IVCCM images. We have limited information bias through the simultaneous use of gold-standard NCS along with the novel small fiber measure of CNFL.
In conclusion, the patients with T1DM in this cohort have a broad spectrum of DSP based on TCNS and DSP diagnostic criteria. IVCCM CNFL can identify a subgroup of patients at high risk of DSP that may prove to be an optimal group to enroll in future therapeutic clinical trials.
Footnotes
Contributors: EJHL, BAP, RPB, TMSW, and VB designed the study. EJHL and LEL performed the statistical analysis. All authors contributed to the discussion, reviewed, and edited the manuscript. EJHL and VB are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding: This work was supported by the Canadian Diabetes Association (#OG-2-13-4093-VB), a Banting and Best Diabetes Centre Studentship (EJHL), an Ontario Graduate Scholarship (EJHL), a James F. Crothers Family Fellowship in Peripheral Nerve Damage (EJHL), and a Peterborough K. M. Hunter Graduate Studentship (EJHL).
Competing interests: TMSW is a part owner, president, and medical director of Glycemic Index Laboratories, Toronto, Canada and has authored several popular diet books on the glycemic index for which he has received royalties from Phillipa Sandall Publishing Services and CABI Publishers. He has received consultant fees, honoraria, travel funding, or served on the scientific advisory board for McCain Foods, Temasek Polytechnic, Singapore, Glycemic Index Symbol program, CreaNutrition AG, and the National Sports and Conditioning Association. His wife is an employee and part owner of Glycemic Index Laboratories.
Patient consent: Obtained.
Ethics approval: University Health Network Research Ethics Board.
Provenance and peer review: Not commissioned; internally peer reviewed.
Data sharing statement: All data from this study was published.
References
- 1.Tesfaye S, Chaturvedi N, Eaton SE et al. . Vascular risk factors and diabetic neuropathy. N Engl J Med 2005;352:341–50. 10.1056/NEJMoa032782 [DOI] [PubMed] [Google Scholar]
- 2.Vinik AI, Park TS, Stansberry KB et al. . Diabetic neuropathies. Diabetologia 2000;43:957–73. 10.1007/s001250051477 [DOI] [PubMed] [Google Scholar]
- 3.Breiner A, Lovblom LE, Perkins BA et al. . Does the prevailing hypothesis that small-fiber dysfunction precedes large-fiber dysfunction apply to type 1 diabetic patients? Diabetes Care 2014;37:1418–24. 10.2337/dc13-2005 [DOI] [PubMed] [Google Scholar]
- 4.Bril V. Treatment for diabetic neuropathy. J Peripher Nerv Syst 2012;17(Suppl 2):22–7. 10.1111/j.1529-8027.2012.00391.x [DOI] [PubMed] [Google Scholar]
- 5.Petropolous IN, Alam U, Fadavi H et al. . Rapid automated diagnosis of diabetic peripheral neuropathy with in vivo corneal confocal microscopy. Invest Ophthalmol Vis Sci 2014;55:2071–8. 10.1167/iovs.13-13787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sivaskandarajah GA, Halpern EM, Lovblom LE et al. . Structure-function relationship between corneal nerves and conventional small-fiber tests in type 1 diabetes. Diabetes Care 2013;36:2748–55. 10.2337/dc12-2075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahmed A, Bril V, Orsazag A et al. . Detection of diabetic sensorimotor polyneuropathy by corneal confocal microscopy in type 1 diabetes: a concurrent validity study. Diabetes Care 2012;35:821–8. 10.2337/dc11-1396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hertz P, Bril V, Orszag A et al. . Reproducibility of in vivo corneal confocal microscopy as a novel screening test for early diabetic sensorimotor polyneuropathy. Diabet Med 2011;28: 1253–60. 10.1111/j.1464-5491.2011.03299.x [DOI] [PubMed] [Google Scholar]
- 9.Hume DA, Lovblom LE, Ahmed A et al. . Higher magnification lenses versus conventional lenses for evaluation of diabetic neuropathy by corneal in vivo confocal microscopy. Diabetes Res Clin Pract 2012;97:e37–40. 10.1016/j.diabres.2012.04.010 [DOI] [PubMed] [Google Scholar]
- 10.Halpern E, Lovblowm L, Orlov S et al. . The existence of rapid small fiber decline in type 1 diabetes: a potential proxy for neuropathy progression using corneal in vivo confocal microscopy. In 22nd Annual Meeting of the Diabetic Neuropathy Study Group Dresden, Germany. [Google Scholar]
- 11.Lovblom LE, Halpern EM, Wu T et al. . In vivo corneal confocal microscopy and prediction of future-incident neuropathy in type 1 diabetes: a preliminary longitudinal analysis. Can J Diabetes 2015;39:390–7. 10.1016/j.jcjd.2015.02.006 [DOI] [PubMed] [Google Scholar]
- 12.Tavakoli M, Quattrini C, Abbott C et al. . Corneal confocal microscopy: a novel noninvasive test to diagnose and stratify the severity of human diabetic neuropathy. Diabetes Care 2010;33:1792–7. 10.2337/dc10-0253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pritchard N, Edwards K, Russell AW et al. . Corneal confocal microscopy predicts 4-year incident peripheral neuropathy in type 1 diabetes. Diabetes Care 2015;38:671–5. 10.2337/dc14-2114 [DOI] [PubMed] [Google Scholar]
- 14.Bril V, Perkins BA. Validation of the Toronto clinical scoring system for diabetic polyneuropathy. Diabetes Care 2002;25:2048–52. [DOI] [PubMed] [Google Scholar]
- 15.England JD, Gronseth GS, Franklin G et al. . Distal symmetric polyneuropathy: a definition for clinical research: report of the American Academy of Neurology, the American Association of Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. Neurology 2005;64:199–207. 10.1212/01.WNL.0000149522.32823.EA [DOI] [PubMed] [Google Scholar]
- 16.Wu T, Ahmed A, Bril V et al. . Variables associated with corneal confocal microscopy parameters in healthy volunteers: implications for diabetic neuropathy screening. Diabetes Med 2012;29:297–303. [DOI] [PubMed] [Google Scholar]
- 17.Yarnitsky D. Quantitative sensory testing. Muscle Nerve 1997;20:198–204. [DOI] [PubMed] [Google Scholar]
- 18.Nabavi Nouri M, Ahmed A, Bril V et al. . Diabetic neuropathy and axon reflex-mediated neurogenic vasodilatation in type 1 diabetes. PLoS ONE 2012;7:e34807 10.1371/journal.pone.0034807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Factors in development of diabetic neuropathy. Baseline analysis of neuropathy in feasibility phase of diabetes control and complications trial (DCCT). The DCCT research group. Diabetes 1988;37: 476–81. [PubMed] [Google Scholar]
- 20.Boulton AJ, Vinik AI, Arezzo JC et al. . American Diabetes Association.Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care 2005;28: 956–62. [DOI] [PubMed] [Google Scholar]
- 21.Orlov S, Bril V, Orszag A et al. . Heart rate variability and sensorimotor polyneuropathy in type 1 diabetes. Diabetes Care 2012;35:809–16. 10.2337/dc11-1652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Apfel SC, Schwartz S, Adornato BT et al. . Efficacy and safety of recombinant human nerve growth factor in patients with diabetic polyneuropathy: a randomized controlled trial. JAMA 2000;284:2215–21. [DOI] [PubMed] [Google Scholar]
- 23.Greene DA, Arezzo JC, Brown MB. Effect of aldose reductase inhibition on nerve conduction and morphometry in diabetic neuropathy: Zenarestat Study Group. Neurology 1999;53:580–91. [DOI] [PubMed] [Google Scholar]
- 24.Wahren J, Foyt H, Daniels M et al. . Long-acting C-peptide and neuropathy in type 1 diabetes: a 12-month clinical trial. Diabetes Care 2016;39:596–602. 10.2337/dc15-2068 [DOI] [PubMed] [Google Scholar]


