Abstract
Abdominal bleeding is a lethal complication in acute pancreatitis (AP) and it is commonly described as a late event. However, spontaneous intra-abdominal bleeding could occur very early but no study focusing on this phenomenon was published yet. In this study, 1137 AP patients were retrospectively screened and 24 subjects suffering early spontaneous bleeding (ESB) were selected. Meanwhile, a 1:1 well-balanced cohort of non-bleeding patients was generated by propensity score match. The clinical characteristics of these patients were compared and a multiple regression analysis was performed to assess the risk factors for ESB. Besides, patients with massive post-intervention bleeding (PIB) were collected for additional comparison. ESB patients suffered significantly worse outcome than the matched cohort evidenced by dramatically higher mortality than the non-bleeding patients and even the PIB group (54.2% versus 20.8%, P = 0.017; 54.2% versus 31.0%, P = 0.049). The regression analysis demonstrated computer tomography severity index (CTSI; OR, 3.34; 95% CI, 1.995–5.59, P < 0.001) and creatinine (OR, 1.008; 95% CI, 1.004–1.012, P < 0.001) were associated with the occurrence of ESB. In conclusion, ESB is a rare but dangerous complication of moderate-to-severe AP and may result in high mortality. CTSI and creatinine are independent risk factors for the development of ESB.
Acute pancreatitis (AP) is a sudden inflammatory disorder of the pancreas, among which, quite a number of cases are self-limited and resolve without severe complications. However, severe acute pancreatitis (SAP) develops in 15–20% patients and is associated with a mortality between 20% to 30%1. Mortality in SAP occurs either early, owing to multiple organ dysfunction syndrome (MODS) or late, due to septic shock and uncontrolled major bleeding.
Intra-abdominal bleeding is a fatal complication of AP, which was considered as the primary cause for >50% death cases of AP2,3. Up till now, it is commonly considered that intra-abdominal bleeding usually develop late in the clinical course2,4,5. According to the study conducted by Balthazar et al.5, bleeding complications were detected from 2 months to 8 years after one or several episodes of pancreatitis. Flati2 and his colleagues summarized massive intra-abdominal bleeding in AP patients and found more than 60% bleeding events occur as a complication of necrosis. It has long been thought that the local inflammation and necrosis in AP would increase the risk of vessel wall injury and finally lead to the bleeding. That is easy to explain why intra-abdominal bleeding usually occur late in the disease course.
However, we noticed that spontaneous intra-abdominal bleeding could occur infrequently at relatively early phase and there was no previous study describing this phenomenon probably due to its limited incidence. Therefore in the present study, we aimed to explore the clinical significance of early spontaneous intra-abdominal bleeding (ESB) in patients with moderate to severe acute pancreatitis using propensity score matched analysis.
Results
Study Population
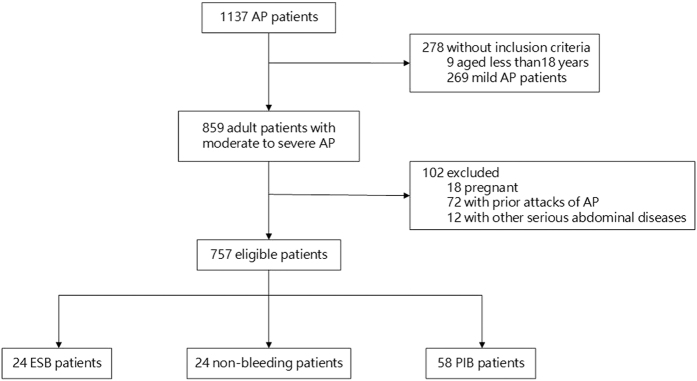
As shown in the Fig. 1, among all 1137 AP patients, 757 eligible patients who met the inclusion and exclusion criteria were screened. Of whom, 24 ESB patients were selected for final analysis. Each ESB patient was exactly and propensity score matched 1:1 with patients without intra-abdominal bleeding (non-bleeding group). In addition, 58 patients who suffered massive post-intervention bleeding (PIB) were enrolled into the PIB group for an additional comparison.
Figure 1. Study flowchart.
Variables between ESB patients and non-bleeding patients
The baseline characteristics including age, gender, body mass index (BMI), time interval from AP onset to admission, etiology and co-morbidities between ESB patients and non-bleeding patients were displayed in Table 1 and no significant difference was found. Table 2 showed the admission status (vital signs, laboratory data, and severity scores, etc.) of ESB patients and non-bleeding patients. Despite the propensity score match we made, significant differences could still be found in the following aspects: blood urea nitrogen (10.7[4.8–20.6] vs. 5.6[2.8–9.5], P = 0.008), creatinine (207.5[58.8–339.5] vs. 58[38–114], P = 0.007) and computer tomography severity index (CTSI; 10[10–10] vs. 8[6–10], P = 0.005).
Table 1. Baseline characteristics of ESB, Non-bleeding, and PIB Patients.
| Variable | ESB Group (n = 24) | Non-bleeding Group (n = 24) | P Value* | PIB Group (n = 58) | P Value† |
|---|---|---|---|---|---|
| Age, years | 43 (36–47.25) | 45.5 (34.75–49.5) | NS (0.570) | 46 (39–51) | NS (0.155) |
| Gender, M/F | 19/5 | 17/7 | NS (0.505) | 48/10 | NS (0.702) |
| BMI, kg/m2 | 24.8 (22.2–28.5) | 25.5 (21.17–27.75) | NS (0.836) | 25.25 (23.08–27.59) | NS (0.967) |
| Onset to admission, days | 18.5 (5.75–26.5) | 10 (6–41.25) | NS (0.844) | 23 (9.5–40) | NS (0.089) |
| Etiology, no. (%) | NS (0.499) | NS (0.99) | |||
| Biliary | 13 (54.2) | 16 (66.7) | 32 (55.2) | ||
| HTG | 9 (37.5) | 8 (33.3) | 20 (34.5) | ||
| Alcoholic | 1 (4.2) | 0 (0) | 3 (5.2) | ||
| Other | 1 (4.2) | 0 (0) | 3 (5.2) | ||
| Co-morbidities, no. (%) | |||||
| Smoking | 10 (41.7) | 8 (33.3) | NS (0.551) | 29 (50.0) | NS (0.492) |
| Alcohol use | 12 (50) | 9 (37.5) | NS (0.383) | 24 (41.4) | NS (0.474) |
| Hypertension | 7 (29.2) | 11 (45.8) | NS (0.233) | 19 (32.8) | NS (0.750) |
| Diabetes mellitus | 5 (20.8) | 7 (29.2) | NS (0.505) | 9 (15.5) | NS (0.560) |
| Coronary heart disease | 0 (0) | 2 (8.3) | NS (0.149) | 4 (6.9) | NS (0.187) |
| Fatty liver | 3 (12.5) | 5 (20.8) | NS (0.439) | 12 (20.7) | NS (0.383) |
| Chronic respiratory disease | 2 (8.3) | 3 (12.5) | NS (0.637) | 8 (13.8) | NS (0.492) |
| Chronic renal disease | 0 (0) | 1 (4.2) | NS (0.312) | 1 (1.7) | NS (0.517) |
BMI body mass index, HTG hypertriglyceridemia. *ESB Group versus Non-bleeding Group, †ESB Group versus PIB Group. Data are expressed as median (interquartile range) for continuous variables and frequencies (proportions) for categorical variables, as appropriate. Mann–Whitney U-test and Pearson test was used as the circumstances required.
Table 2. Comparison of ESB patients and Non-bleeding patients on admission.
| Variable | ESB Group (n = 24) | Non-bleeding Group (n = 24) | P Value |
|---|---|---|---|
| Temperature, °C | 37.85 (36.53–38.58) | 37.5 (37.08–38.2) | NS (0.702) |
| MAP, mmHg | 94.33 (73.33–108.67) | 93.33 (87.92–103.83) | NS (0.885) |
| Heart rate | 113.5 (100.5–131.5) | 114 (91.25–127.75) | NS (0.680) |
| Respiratory rate | 26 (22.25–34.25) | 24 (18.25–34.5) | NS (0.337) |
| IAP, mmHg | 11.7 (8.7–14.65) | 9.15 (7.45–12.0) | NS (0.066) |
| Hemoglobin, g/L | 81.5 (73.25–99) | 91.5 (78–99.0) | NS (0.312) |
| Platelet count, x109 cells/L | 188 (135.75–299.5) | 203 (121.75–278) | NS (0.975) |
| PT, s | 13.65 (12.8–14.75) | 14.45 (13.03–16.6) | NS (0.208) |
| APTT, s | 36.6 (31.83–42.33) | 36.75 (28.6–43.53) | NS (0.902) |
| INR | 1.2 (1.11–1.4) | 1.23 (1.16–1.29) | NS (0.628) |
| C-reactive protein, mg/L | 125.9 (89.9–182.1) | 117.7 (80.8–183.15) | NS (0.585) |
| Total bilirubin, umol/L | 26.3 (15.6–83.0) | 21.8 (10.78–52.98) | NS (0.364) |
| Unconjugated bilirubin, umol/L | 10.9 (5.0–34.5) | 7.0 (2.93–13.9) | NS (0.197) |
| ALT, U/L | 24.5 (12.25–48.25) | 32.5 (25.25–60.5) | NS (0.173) |
| AST, U/L | 29 (22.5–88.5) | 37 (25.25–67) | NS (0.585) |
| BUN, mmol/L | 10.7 (4.8–20.6) | 5.6 (2.8–9.5) | 0.008 |
| Creatinine, umol/L | 207.5 (58.8–339.5) | 58 (38–114) | 0.007 |
| CTSI | 10 (10–10) | 8 (6–10) | 0.005 |
| APACHE II score | 15.5 (9–22.3) | 15.5 (10.25–22.75) | NS (0.885) |
| SOFA score | 6 (2.25–8.75) | 3 (2–6) | NS (0.076) |
MAP mean arterial pressure, IAP intra-abdominal pressure, PT prothrombin time, APTT activated partial thromboplastin time, INR international normalized ratio, ALT alanine transaminase, AST aspartate transaminase, BUN blood urea nitrogen, CTSI computer tomography severity index, APACHE acute physiology and chronic health evaluation, SOFA sequential organ failure assessment. Data are expressed as median (interquartile range) for continuous variables and frequencies (proportions) for categorical variables, as appropriate. Mann–Whitney U-test and Pearson test was used as the circumstances required.
The disease course and prognosis of ESB patients were largely different from the non-bleeding patients. As shown in the Table 3, ESB patients had a higher risk of developing organ failures including shock, acute kidney injury (AKI), acute liver injury (ALI) and multiple organ dysfunction syndrome (MODS) than non-bleeding patients. Moreover, ESB patients suffered more invasive treatment measures including continuous renal replacement therapy (CRRT), percutaneous catheter drainage (PCD), negative pressure irrigation (NPI) and digital subtraction angiography (DSA). In addition, the proportion of severe acute pancreatitis in ESB patients was significantly higher than non-bleeding patients (22, 91.7% vs. 11, 45.8%, P = 0.001), so were the mortality (13, 54.2% vs. 5, 20.8%, P = 0.017), intensive care unit (ICU) duration and cost.
Table 3. Clinical course and outcome between ESB patients and Non-bleeding patients.
| Variable | ESB Group (n = 24) | Non-bleeding Group (n = 24) | P Value |
|---|---|---|---|
| IPN, no. (%) | 19 (79.2) | 15 (62.5) | NS (0.204) |
| Digestive tract fistula, no. (%) | 10 (41.7) | 6 (25.0) | NS (0.221) |
| SVT, no. (%) | 5 (20.8) | 5 (20.8) | NS (1) |
| ACS, no. (%) | 7 (29.2) | 0 (0) | 0.004 |
| Organ failure | |||
| Shock, no. (%) | 16 (66.7) | 8 (33.3) | 0.021 |
| ARDS, no. (%) | 19 (79.2) | 15 (62.5) | NS (0.204) |
| AKI, no. (%) | 17 (70.8) | 10 (41.6) | 0.042 |
| ALI, no. (%) | 13 (54.2) | 6 (25.0) | 0.039 |
| MODS, no. (%) | 18 (75.0) | 8 (33.3) | 0.004 |
| Management | |||
| CRRT, no. (%) | 16 (66.7) | 8 (33.3) | 0.021 |
| PCD, no. (%) | 20 (83.3) | 13 (54.2) | 0.029 |
| NPI, no. (%) | 18 (75.0) | 8 (33.3) | 0.004 |
| ED, no. (%) | 11 (45.8) | 8 (33.3) | NS (0.376) |
| DSA, no. (%) | 17 (70.8) | 5 (20.8) | 0.001 |
| Laparotomy, no. (%) | 12 (50.0) | 6 (25.0) | NS (0.074) |
| Revised Atlanta classification | 0.001 | ||
| Mild, no. (%) | 0 (0) | 0 (0) | |
| Moderate severe, no. (%) | 2 (8.3) | 13 (54.2) | |
| Severe, no. (%) | 22 (91.7) | 11 (45.8) | |
| Mortality, no. (%) | 13 (54.2) | 5 (20.8) | 0.017 |
| Hospital duration, days | 35 (19.5–83.75) | 13.5 (8–64.5) | NS (0.060) |
| ICU duration, days | 24.5 (12.75–46.25) | 9 (4.25–33.75) | 0.042 |
| Cost, thousand CNY | 293 (183–459) | 120 (49–449) | 0.026 |
IPN infected pancreatic necrosis, SVT splanchnic venous thrombosis, ACS abdominal compartment syndrome, ARDS acute respiratory distress syndrome, AKI acute kidney injury, ALI acute liver injury, MODS multiple organ dysfunction syndrome, CRRT continuous renal replacement therapy, PCD percutaneous catheter drainage, NPI negative pressure irrigation, ED endoscopic necrosectomy, DSA digital subtraction angiography, ICU intensive care unit. Data are expressed as median (interquartile range) for continuous variables and frequencies (proportions) for categorical variables, as appropriate. Mann–Whitney U-test and Pearson test was used as the circumstances required.
Risk factors predicting ESB
A multivariable regression model including 16 indices (age, gender, etiology of AP, acute physiology and chronic health evaluation II score, etc.) was performed to evaluate risk factors of ESB. Univariate logistic regression analysis (Table 4) revealed significant correlations between ESB and prothrombin time (PT; P = 0.027), creatinine (P = 0.011) and CTSI (P < 0.001). Taking the three significant variables together into the multiple logistic regression model, CTSI (OR, 3.34; 95% CI, 1.995–5.59, P < 0.001) and creatinine (OR, 1.008; 95% CI, 1.004–1.012, P < 0.001) were proved to be independent risk factors for ESB (shown in Table 5).
Table 4. Univariate logistic regression analysis of ESB.
| Variable | Odds Ratio | 95% CI |
P Value | |
|---|---|---|---|---|
| Lower | Upper | |||
| Age | 0.955 | 0.896 | 1.018 | 0.155 |
| Gender | 1.105 | 0.141 | 8.679 | 0.925 |
| Etiology | 1.136 | 0.399 | 3.234 | 0.812 |
| Smoking | 0.978 | 0.162 | 5.909 | 0.981 |
| Alcohol use | 0.169 | 0.023 | 1.236 | 0.080 |
| Platelet count | 1.002 | 0.995 | 1.009 | 0.611 |
| PT | 0.444 | 0.216 | 0.913 | 0.027 |
| APTT | 0.997 | 0.954 | 1.042 | 0.889 |
| INR | 1497.253 | 0.385 | 5822920.583 | 0.083 |
| C-reactive protein | 0.991 | 0.979 | 1.004 | 0.173 |
| Total bilirubin | 0.996 | 0.982 | 1.010 | 0.585 |
| Unconjugated bilirubin | 1.050 | 0.982 | 1.123 | 0.152 |
| BUN | 0.962 | 0.772 | 1.198 | 0.730 |
| Creatinine | 1.018 | 1.004 | 1.032 | 0.011 |
| CTSI | 5.764 | 2.378 | 13.971 | <0.001 |
| APACHE II score | 0.906 | 0.793 | 1.036 | 0.148 |
PT prothrombin time, APTT activated partial thromboplastin time, INR international normalized ratio, BUN blood urea nitrogen, CTSI computer tomography severity index, APACHE acute physiology and chronic health evaluation.
Table 5. Independent prognostic factors in a multivariate logistic regression analysis of ESB.
| Variable | Odds Ratio | 95% CI |
P Value | |
|---|---|---|---|---|
| Lower | Upper | |||
| CTSI | 3.340 | 1.995 | 5.590 | <0.001 |
| Creatinine | 1.008 | 1.004 | 1.012 | <0.001 |
CTSI computer tomography severity index.
Variables between ESB patients and post-intervention patients
As shown in the Table 1, there was no significant difference regarding the baseline characteristics between ESB and PIB groups. Besides, Table 6 showed the admission status and prognosis of the two groups, among which, significant difference could be detected in the following aspects: creatinine, acute physiology and chronic health evaluation (APACHE) II score, time interval from AP onset to bleeding, infected pancreatic necrosis (IPN), acute liver injury (ALI) and mortality, indicting a more severe clinical course of ESB patients even compared with PIB patients. Unexpectedly, the occurrence of ESB was associated with an even lower IPN rate. Furthermore, as Fig. 2 showed, the time interval from disease onset to death of ESB patients was significantly shorter than PIB patients (46[30.5–72] days versus 73.5[57.75–112.75] days, P = 0.010). Additionally, there were 17 patients(26 times) in the ESB group and 50 patients(70 times) in the PIB group received digital subtraction angiography (DSA) examination, respectively, among which, positive findings were found in 15 (14 artery and 1 vein) cases and 35 (33 artery and 2 vein) cases. Transcatheter arterial embolization (TAE) was successfully performed for 13 times in 9 of the 24 ESB patients and 27 times in 22 of the 58 PIB patients.
Table 6. Admission status, clinical course and outcome between ESB patients and PIB patients.
| Variable | ESB Group (n = 24) | PIB Group (n = 58) | P Value |
|---|---|---|---|
| Hemoglobin, g/L | 81.5 (73.25–99) | 91 (79.75–101.5) | NS (0.142) |
| Platelet count, ×109 cells/L | 188 (135.75–299.5) | 186 (106.75–284) | NS (0.795) |
| PT, s | 13.65 (12.8–14.75) | 13.45 (12.15–14.8) | NS (0.71) |
| APTT, s | 36.6 (31.83–42.33) | 33.5 (27.95–40.35) | NS (0.154) |
| INR | 1.2 (1.11–1.4) | 1.17 (1.055–1.2725) | NS (0.177) |
| C-reactive protein, mg/L | 125.9 (89.9–182.1) | 131.45 (96.675–191) | NS (0.744) |
| Total bilirubin, umol/L | 26.3 (15.6–83) | 24.15 (13.5–55.25) | NS (0.488) |
| Unconjugated bilirubin, umol/L | 10.9 (5.0–34.5) | 6.55 (3.55–13.075) | NS (0.06) |
| ALT, U/L | 24.5 (12.25–48.25) | 32 (21–56) | NS (0.243) |
| AST, U/L | 29 (22.5–88.5) | 34.5 (24–54.25) | NS (0.919) |
| BUN, mmol/L | 10.7 (4.8–20.6) | 9.1 (5.35–14.725) | NS (0.415) |
| Creatinine, umol/L | 207.5 (58.8–339.5) | 81.5 (49.5–188.5) | 0.037 |
| APACHE II score | 15.5 (9–22.3) | 11.5 (8.75–15) | 0.032 |
| Onset to bleeding, days | 17.5 (10–20) | 51 (26.75–70.75) | <0.001 |
| IPN, no. (%) | 19 (79.2) | 56 (96.6) | 0.010 |
| Digestive tract fistula, no. (%) | 10 (41.7) | 28 (48.3) | NS (0.585) |
| SVT, no. (%) | 5 (20.8) | 13 (22.4) | NS (0.875) |
| ACS, no. (%) | 7 (29.2) | 10 (17.2) | NS (0.225) |
| Organ failure | |||
| Shock, no. (%) | 16 (66.7) | 37 (63.8) | NS (0.804) |
| ARDS, no. (%) | 19 (79.2) | 39 (67.2) | NS (0.280) |
| AKI, no. (%) | 17 (70.8) | 34 (58.6) | NS (0.299) |
| ALI, no. (%) | 13 (54.2) | 15 (25.9) | 0.014 |
| MODS, no. (%) | 18 (75.0) | 37 (63.8) | NS (0.326) |
| Revised Atlanta classification | NS (0.230) | ||
| Mild, no. (%) | 0 (0) | 0 (0) | |
| Moderate severe, no. (%) | 2 (8.3) | 11 (19.0) | |
| Severe, no. (%) | 22 (91.7) | 47 (81.0) | |
| Mortality, no. (%) | 13 (54.2) | 18 (31.0) | 0.049 |
| Hospital duration, days | 35 (19.5–83.75) | 52 (27–78) | NS (0.343) |
| ICU duration, days | 24.5 (12.75–46.25) | 35 (18.25–53.25) | NS (0.375) |
| Cost, thousand CNY | 293 (183–459) | 416 (256–553) | NS (0.433) |
PT prothrombin time, APTT activated partial thromboplastin time, INR international normalized ratio, ALT alanine transaminase, AST aspartate transaminase, BUN blood urea nitrogen, APACHE acute physiology and chronic health evaluation, IPN infected pancreatic necrosis, SVT splanchnic venous thrombosis, ACS abdominal compartment syndrome, ARDS acute respiratory distress syndrome, AKI acute kidney injury, ALI acute liver injury, MODS multiple organ dysfunction syndrome, ICU intensive care unit. Data are expressed as median (interquartile range) for continuous variables and frequencies (proportions) for categorical variables, as appropriate. Mann–Whitney U-test and Pearson test was used as the circumstances required.
Figure 2. Distribution of survival time between ESB group and PIB group (Mann–Whitney U-test).
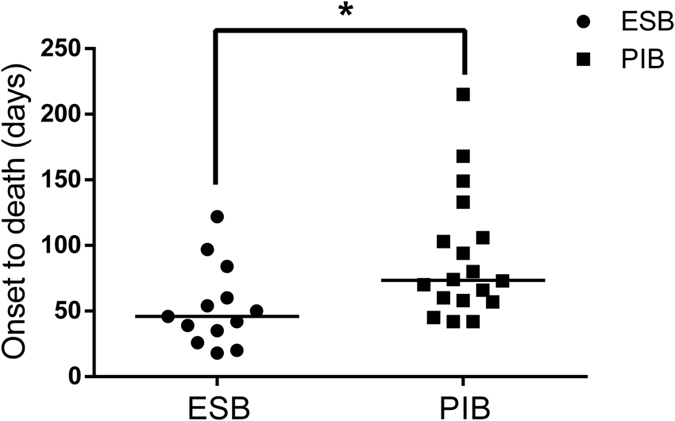
The lines in the figure refer to the median survival time of patients (ESB group: 46[30.5–72] days vs PIB group: 73.5[57.75–112.75] days, P = 0.010).
Discussion
In this study, early spontaneous intra-abdominal bleeding developed in 24 of all 757 moderate to severe AP patients (3.2%). The mortality of ESB patients was up to 54%, which was extremely high considering the mortality of AP patients in our department was only 6~8% during the study period. With almost the same baseline characteristics and admission status, ESB patients suffered much more severe disease compared with not only non-bleeding patients but also PIB patients, suggesting ESB is a rare but lethal complication of AP.
The multivariable regression model demonstrated that a high CTSI (more pancreatic necrosis) and a high creatinine level (worse renal function) may lead to the development of ESB. As far as we know, the erosion and rupture of blood vessel and pseudoaneurysm which resulted from severe pancreatic necrosis, abscesses, and pseudocysts, local inflammation and aggressive surgical intervention were the leading causes of intra-abdominal bleeding in AP patients2,4,6,7,8. According to the previous study, local infected fluid collections (necrosis, pseudocyst, and abscess) were the most common risk factors for hemorrhage in AP which account for more than 60% cases2. In this regard, it is easy to explain why intra-abdominal bleeding is more likely to happen in patients with more pancreatic necrosis, namely, a high CTSI. Besides, bleeding event was also considered as a major complication of renal failure, especially in the pre-dialysis era9,10. Although, the underling mechanism of hemorrhage in patients with kidney injury is still unclear, the need for anticoagulation like heparin during the renal replacement therapy and effects related to uraemic thrombopathy may be two primary causes11,12,13,14. Our previous work also showed AKI was an independent risk factor for intra-abdominal bleeding in AP patients15. However, the cause-and-effect relationship between renal failure and bleeding event remains unclear, intra-abdominal bleeding may also severely influence the renal function due to the accompanying volume deficiency, intra-abdominal hypertension, and secondary infection, etc.
As mentioned before, the development of intra-abdominal bleeding in AP patients was considered as a progressive process caused by persistent accumulation of vessel injuries. Thus, intra-abdominal bleeding tends to occur late in the course of AP, which has already been described in a series of previous studies2,4,5. However, in this study, the median time interval from AP onset to bleeding event of ESB patients was only 17.5 (10–20) days, dramatically shorter than PIB patients (51[26.75–70.75] days). Besides, the statistical results indicted ESB patients suffered significantly worse prognosis than PIB patients, except for a lower IPN rate which may be attributed to the shorter interval from disease onset to death in some ESB patients. The exact cause of ESB remains unknown, but activated pancreatic enzymes might play a role in the development of ESB. It is known that extravasation of exocrine enzyme-rich fluids within areas of parenchymal necrosis may further aggravates the necrotizing process and increases the risk of damaging the walls of vessels16. Considering the more pancreatic necrosis ESB patients suffered, they should had a high local enzyme level, which may contribute substantially to the occurrence of ESB. Taking together, the underlying mechanism of ESB may be different from PIB and further studies are required.
Certain limitations of this study need to be addressed. Due to the small number of study patients and limited incidence of ESB, the statistical power of the analyses could be relatively weak and some of the findings may be not significant in a larger sample. Moreover, as a retrospective study, selection bias can hardly be avoided especially between the ESB group and PIB group which we could not conduct a propensity score match because of limited sample size.
Conclusion
Early spontaneous bleeding is a rare but potentially lethal complication of AP which occurs in approximately 3% of moderate to severe AP patients, and is associated with a high mortality of 54%. CTSI and creatinine are independent risk factors for the development of ESB in AP patients.
Methods
This retrospective propensity matched cohort study was conducted at the Department of General Surgery in Jinling Hospital between January 2013 and December 2015. Jinling Hospital is a teaching hospital affiliated to the Nanjing University, and serves patients from various regions all over China. As a retrospective study, no ethics approval is required in our institute. To retrieve data from the electric database, we ask for an approval from the Acute Pancreatitis Database Management Committee. All the analyses were performed in compliance with the committee’s regulation. Informed consent regarding data storage and publication was obtained from each patients who were recorded in the database during their hospitalization.
Definitions
Different diagnostic approaches of spontaneous bleeding were described previously3,17,18. In our study, early spontaneous bleeding (ESB) was defined when the bleeding could be detected on contrast-enhanced computed tomography (CECT) within 30 days from initial presentation without prior minimally-invasive or operative intervention. Considering the bleeding risk resulted from surgical interventions2,7,8,19,20, we had excluded patients with prior intervention to reduce the bias. CT scans for patients were reviewed separately by two radiologists with rich experience in abdominal imaging. Surgical interventions including percutaneous catheter drainage (PCD), negative pressure irrigation (NPI), endoscopic necrosectomy (EN), and laparotomy were considered when patients were diagnosed with infected pancreatic necrosis, digestive tract fistula, persistent abdominal compartment syndrome (ACS), and/or uncontrolled hemorrhage and so on. Besides, patients who suffered massive intra-abdominal bleeding after any abovementioned surgical intervention were collected as the post intervention bleeding (PIB) group for an additional comparison. The definition of massive bleeding was a decrease of hemoglobin concentration >2 g/dL and/or significant hemodynamic deterioration caused by intra-abdominal bleeding2,15. Organ failures including shock, acute respiratory distress syndrome (ARDS), acute kidney injury (AKI) and acute liver injury (ALI) were assessed. Shock was defined as systolic blood pressure <90 mmHg or need for inotropic agent, respiratory failure was defined as PaO2/FiO2 ≤ 300 mmHg, renal failure was defined as serum creatinine ≥176umol/L (2.0 mg/dL) and the criteria for hepatic failure was defined as a score of ≥2 using the Marshall scoring system21. The diagnoses of local and systemic complications were made based on the revised Atlanta classification of acute pancreatitis22. According to the guidelines of World Society of Abdominal Compartment Syndrome23, abdominal compartment syndrome (ACS) was defined as a sustained intra-abdominal pressure more than 20 mmHg accompanied with new-onset organ dysfunction/failure. CTSI was evaluated according to the contrast-enhanced computed tomography using the Balthazar’s CT score24.
Patients
We screened all consecutive adult patients (age ≥ 18 years) who were diagnosed with moderate to severe AP in our center between January 2013 and December 2015. Patients with mild AP were not included because they rarely suffered intra-abdominal bleeding thus may bring in additional selection bias. Diagnostic criteria for moderate to severe AP were defined according to the revised Atlanta criteria22. Exclusion criteria included pregnancy, prior attacks of acute pancreatitis, and combining with other serious abdominal diseases. Finally, 757 eligible patients were enrolled for further analysis.
Early spontaneous bleeding could be defined in 24 patients (ESB group). Every case was 1:1 propensity score matched with another patient without intra-abdominal bleeding (non-bleeding group). Matching was based on the admission APACHE II score, demographics (age, gender, and body mass index), time interval from AP onset to admission, and etiology (biliary, hypertriglyceridemia, alcoholic, and other). The propensity score match could effectively decrease bias and improve control for confounding variables25. To compare with patients who suffered “classic” AP-related bleeding and avoid potential overlap, 58 patients who suffered massive intra-abdominal bleeding after surgical intervention (PIB group) was additionally studied to compare with the ESB group in terms of clinical prognosis. All patients received the same standard26,27 treatment including fluid therapy, pain control, nutritional support, antibiotics and so on. In patients with massive intra-abdominal bleeding, resuscitation was applied at the first time, digital subtraction angiography (DSA) and subsequent transcatheter arterial embolization (TAE) were attempted consecutively if possible. Laparotomy was only performed in patients who cannot be controlled with non-operative measures. DSA was also applied in some non-bleeding patients.
Data Collection
The data of patients was obtained from our AP database which contains information of more than two thousand AP patients. Collected variables included demographics, medical history, etiology, co-morbidities, vital signs, laboratory tests (blood routine examination, blood biochemistry, coagulation routine, etc.), imaging data, systemic and local complications, special treatments, and outcomes (mortality, hospital and intensive care unit durations, cost, etc.). The Acute Physiology and Chronic Health Evaluation (APACHE) II score and Sequential Organ Failure Assessment (SOFA) score were also acquired from the database.
Statistical Analysis
All analyses were performed using SPSS 22.0 for windows (IBM Analytics, Armonk, NY). Data are expressed as median (interquartile range) for continuous variables and frequencies (proportions) for categorical variables. Mann–Whitney U-test and Pearson test was used as the circumstances required. To identify the risk factors for ESB, several series of univariate logistics regression analyses using 16 indices were performed. Variables that showed statistical significance were tested in further multiple logistic regression analyses. Odds ratio was expressed with 95% confidence interval (CI). All statistical tests were two-tailed, and the statistical significance was considered as P < 0.05.
Additional Information
How to cite this article: Chen, Y. et al. Early Spontaneous Abdominal Bleeding is associated with Poor Outcome in Moderate to Severe Acute Pancreatitis Patients: A Propensity Matched Study. Sci. Rep. 7, 42607; doi: 10.1038/srep42607 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Footnotes
The authors declare no competing financial interests.
Author Contributions Study concept and design: Y.C., J.Z. & L.K.; Statistical analysis: Y.C., J.Z. & G.L.; Acquisition of data; analysis and interpretation of data: Y.C. & J.D.; Drafting of the manuscript: Y.C. & Y.P.; Critical revision of the manuscript for important intellectual content: L.K. & W.L.; Administrative, technical, or material support: L.K. & Z.T.; Study supervision: L.K., W.L. & J.L. All of the authors read and approve the manuscript.
References
- Banks P. A. & Freeman M. L. Practice Parameters Committee of the American College of, G. Practice guidelines in acute pancreatitis. The American journal of gastroenterology 101, 2379–2400 (2006). [DOI] [PubMed] [Google Scholar]
- Flati G., Andren-Sandberg A., La Pinta M., Porowska B. & Carboni M. Potentially fatal bleeding in acute pancreatitis: pathophysiology, prevention, and treatment. Pancreas 26, 8–14 (2003). [DOI] [PubMed] [Google Scholar]
- Phillip V., Rasch S., Gaa J., Schmid R. M. & Algul H. Spontaneous bleeding in pancreatitis treated by transcatheter arterial coil embolization: a retrospective study. PLoS One 8, e72903 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson E., Ansari D. & Andersson R. Major haemorrhagic complications of acute pancreatitis. British Journal of Surgery 97, 1379–1384 (2010). [DOI] [PubMed] [Google Scholar]
- Balthazar E. J. & Fisher L. A. Hemorrhagic complications of pancreatitis: radiologic evaluation with emphasis on CT imaging. Pancreatology 1, 306–313 (2001). [DOI] [PubMed] [Google Scholar]
- Tsiotos G. G., Munoz Juarez M. M. & Sarr M. G. Intraabdominal hemorrhage complicating surgical management of necrotizing pancreatitis. Pancreas 12, 126–130 (1996). [DOI] [PubMed] [Google Scholar]
- Bello B. & Matthews J. B. Minimally invasive treatment of pancreatic necrosis. World journal of gastroenterology 18, 6829–6835 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pupelis G. et al. Focused open necrosectomy in necrotizing pancreatitis. HPB (Oxford) 15, 535–540 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remuzzi G. Bleeding in renal failure. Lancet 1, 1205–1208 (1988). [DOI] [PubMed] [Google Scholar]
- Sagripanti A. & Barsotti G. Bleeding and thrombosis in chronic uremia. Nephron 75, 125–139 (1997). [DOI] [PubMed] [Google Scholar]
- Ostermann M., Dickie H., Tovey L. & Treacher D. Heparin algorithm for anticoagulation during continuous renal replacement therapy. Critical care 14, 419 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remuzzi G. et al. Parathyroid hormone inhibits human platelet function. Lancet 2, 1321–1323 (1981). [DOI] [PubMed] [Google Scholar]
- Vlachoyannis J. & Schoeppe W. Adenylate cyclase activity and cAMP content of human platelets in uraemia. European journal of clinical investigation 12, 379–381 (1982). [DOI] [PubMed] [Google Scholar]
- Livio M., Benigni A., Vigano G., Mecca G. & Remuzzi G. Moderate doses of aspirin and risk of bleeding in renal failure. Lancet 1, 414–416 (1986). [DOI] [PubMed] [Google Scholar]
- Shen X. et al. Risk Factors and Outcome for Massive Intra-Abdominal Bleeding Among Patients With Infected Necrotizing Pancreatitis. Medicine (Baltimore) 94, e1172 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley E. L. 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, Ga, September 11 through 13, 1992. Archives of surgery 128, 586–590 (1993). [DOI] [PubMed] [Google Scholar]
- Balachandra S. & Siriwardena A. K. Systematic appraisal of the management of the major vascular complications of pancreatitis. Am J Surg 190, 489–495 (2005). [DOI] [PubMed] [Google Scholar]
- Yoon W., Jeong Y. Y. & Kim J. K. Acute gastrointestinal bleeding: contrast-enhanced MDCT. Abdominal imaging 31, 1–8 (2006). [DOI] [PubMed] [Google Scholar]
- Rumstadt B., Schwab M., Korth P., Samman M. & Trede M. Hemorrhage after pancreatoduodenectomy. Ann Surg 227, 236–241 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita Y. et al. Risk factors for and management of delayed intraperitoneal hemorrhage after pancreatic and biliary surgery. Am J Surg 193, 454–459 (2007). [DOI] [PubMed] [Google Scholar]
- Marshall J. C. et al. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Critical care medicine 23, 1638–1652 (1995). [DOI] [PubMed] [Google Scholar]
- Banks P. A. et al. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut 62, 102–111 (2013). [DOI] [PubMed] [Google Scholar]
- Kirkpatrick A. W. et al. Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 39, 1190–1206 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balthazar E. J., Robinson D. L., Megibow A. J. & Ranson J. H. Acute pancreatitis: value of CT in establishing prognosis. Radiology 174, 331–336 (1990). [DOI] [PubMed] [Google Scholar]
- Schneeweiss S. et al. High-dimensional propensity score adjustment in studies of treatment effects using health care claims data. Epidemiology 20, 512–522 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenner S., Baillie J. & DeWitt J., Vege S. S. American College of, G. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol 108, 1400–1415, 1416 (2013). [DOI] [PubMed] [Google Scholar]
- IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology 13, e1–e15 (2013). [DOI] [PubMed] [Google Scholar]