Abstract
Stereocilia are actin-based protrusions on auditory and vestibular sensory cells that are required for hearing and balance. They convert physical force from sound, head movement or gravity into an electrical signal, a process that is called mechanoelectrical transduction. This function depends on the ability of sensory cells to grow stereocilia of defined lengths. These protrusions form a bundle with a highly precise geometry that is required to detect nanoscale movements encountered in the inner ear. Congenital or progressive stereocilia degeneration causes hearing loss. Thus, understanding stereocilia hair bundle structure, development, and maintenance is pivotal to understanding the pathogenesis of deafness. Stereocilia cores are made from a tightly packed array of parallel, crosslinked actin filaments, the length and stability of which are regulated in part by myosin motors, actin crosslinkers and capping proteins. This review aims to describe stereocilia actin regulation in the context of an emerging “tip turnover” model where actin assembles and disassembles at stereocilia tips while the remainder of the core is exceptionally stable.
Keywords: Hearing, Deafness, Stereocilia, Actin
1. Introduction
The specialized auditory and vestibular sensory cells that detect sound, gravity and head position feature a bundle of hair-like projections called stereocilia that couple physical displacement to depolarization of resting membrane potential. This process, known as mechanoelectrical transduction (MET), is essential for hearing; consequently, stereocilia loss or degeneration causes deafness. In mouse models, defective stereocilia homeostasis is one primary cause of progressive and age-related deafness [1–7].
Mammalian auditory hair cells are not replaced if lost, possibly because of their intricate cell shape, position in a complex tissue or requirement to synapse with neurons. Thus, human hair cells must form precisely structured stereocilia bundles and also maintain those bundles for decades. Both events raise questions about the underlying molecular mechanisms, which are relevant to potential therapeutic approaches to treating hearing loss. Here, we first address how the bundle assumes its characteristic shape and cellular position, and next discuss how the stereocilia actin core is regulated to maintain mature stereocilia length.
2. Morphological and molecular structure of stereocilia bundles
2.1. General morphology and organization of bundles
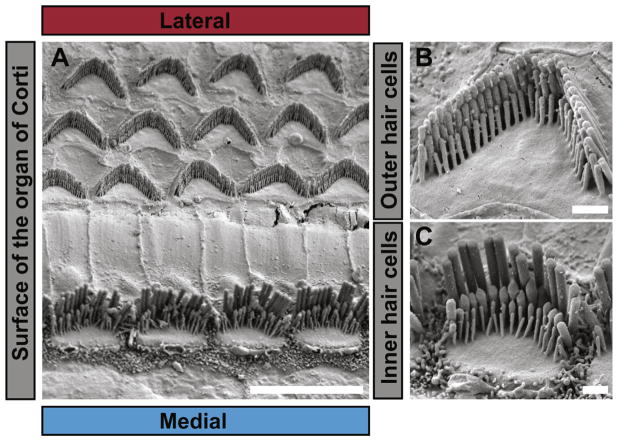
The cochlea, which is the end organ of the auditory system, houses two types of sensory hair cells (Fig. 1). The inner hair cells (IHCs) communicate information to the auditory cortex, while outer hair cells (OHCs) amplify sound waves in the cochlea by rapidly oscillating in length [8,9]. Both cell types are stimulated by stereocilia deflections as small as a few nanometers. To detect these small movements stereocilia are organized into a bundle with three rows of stereocilia that increase in height in a stair-like fashion. From tallest to shortest, the rows are designated as row 1, 2 and 3. Within a row, stereocilia are very nearly the same height. The tips of stereocilia from the shorter rows (2 and 3) are connected to the sides of the adjacent taller stereocilia by tip links (Fig. 2). These thin filaments are formed from heterodimers of the trans-membrane proteins protocadherin-15 (PCDH15) and cadherin-23 (CDH23) [10]. As the taller stereocilia are deflected backwards by force from sound waves, tension is transferred through the tip link, which then mechanically gates an ion channel housed in the tips of row 2 and 3 stereocilia [11]. Sodium, potassium and calcium ions enter through the open channel, depolarize the cell and trigger firing of associated afferent neurons for IHCs or cell contraction for OHCs [12].
Fig. 1.
Inner and outer hair cell stereocilia. A) Scanning electron micrograph of the surface of the organ of Corti from an adult mouse. The tectorial membrane was removed to expose the stereocilia that protrude from the apical surface of the organ of Corti. A single row of inner hair cells (IHC) is located at the medial side while three rows of outer hair cells (OHC) are found towards the lateral side. Bar is 10 μm. B) Higher magnification of an outer hair cell stereocilia bundle showing the three rows of stereocilia within each bundle. The tallest row ordinarily anchors into the overlying tectorial membrane and does not contain functional mechanoelectrical transduction (MET) channels. The shorter two rows have MET channels that are gated by tip links. Bar is 1 μm. C) Higher magnification image of a typical inner hair cell stereocilia bundle, which often have an incomplete 4th row of thin stereocilia. Bar is 1 μm.
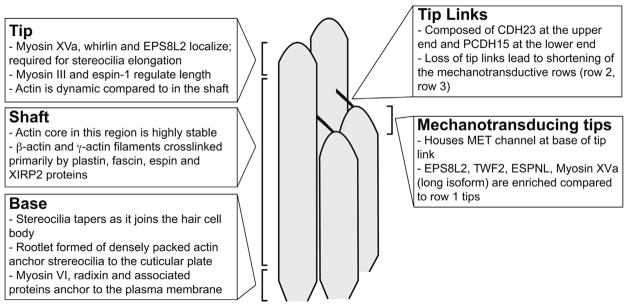
Fig. 2.
Regions of stereocilia are distinguished by protein composition, actin dynamics and function. Stereocilia can be divided into tip, shaft and base segments, each with a unique protein signature. A) The tip region has the highest level of actin turnover which is likely regulated by proteins such as myosin XVa, whirlin, epidermal growth factor receptor pathway substrate 8 (EPS8), myosin III and espin-1. B) Stereocilia shafts comprise the majority of the length. This region is notable for the extreme stability of actin filaments, which are crosslinked into a parallel bundle by espin, plastin and fascin isoforms as well as Xin actin binding repeat containing 2 (XIRP2). C) Stereocilia taper at their base before joining the hair cell body. A rootlet comprised of actin filaments bundled by TRIOBP spans the joint and stabilizes the stereocilia. D) Tip links composed of cadherin 23 (CDH23) and protocadherin 15 (PCDH15) connect the tips of shorter row stereocilia to the sides of adjacent taller stereocilia. E) The tips of mechanotransducing stereocilia (in the shorter rows of the bundle) are specialized. They house mechanoelectrical transduction channels, which are associated with the base of tip links, as well as a different set of actin regulatory proteins such as twinfilin-2 (TWF2), Eps8-like 2 (EPS8L2) and myosin XVa.
The cores of stereocilia are composed of parallel actin filaments crosslinked at a frequency of about one crosslinker for every 10 actin subunits. They appear to run the entire length of the stereocilia, extending from the base to within a few nanometers of the membrane at the tip of the stereocilia [13–15]. The actin core is stiff and as a result the structure pivots, lever-like, at the base where the protrusion tapers as it joins the hair cell body. The actin filaments are regulated by several different types of binding proteins, with myosin motors and actin crosslinkers making particularly noteworthy contributions to stereocilia morphogenesis and maintenance.
2.2. The tip, shaft and base are distinct parts of a stereocilium
The tip of the stereocilium is a highly specialized domain (Fig. 2). Several proteins selectively localize to this region and are presumably part of the tip density that is evident in electron micrographs. Some of these, such as myosin XVa (Myo15), whirlin (Whrn), Epidermal growth factor receptor pathway substrate 8 (Eps8), myosin IIIa and IIIb (Myo3a and Myo3b) and espin-1 (splice isoform of Espn) are important for growth of the actin core [16–22]. Others, including CapZ, Eps8-like 2 (Eps8L2) and Twinfilin-2 (Twf2) have actin filament capping activity and likely regulate actin filament elongation [2,23–25]. Interestingly, the protein composition of stereocilia tips differs in the tallest row of the bundle compared to those in shorter rows. In particular, EPS8 is enriched in row 1 while EPS8L2 and TWF2 are largely restricted to rows 2 and 3 along with the long splice isoform of myosin XVa [2,24,26,27]. This localization pattern is likely important for generating or maintaining the staircase morphology of the bundle. In addition, the tips of the shorter row stereocilia are particularly notable as they also house functional mechanosensitive transduction channels [11].
The diameter of the stereocilia narrows near the cell body as only a subset of the actin filaments pass through this region. Within the taper, there is a rootlet structure that anchors stereocilia into the underlying cuticular plate in the cell body [14,28]. The rootlet depends on the actin binding protein TRIOBP to organize actin filaments into an array that is even more tightly packed than in the stereocilia shaft. Stereocilia lacking TRIOBP also lack rootlets and as a result are less rigid and more prone to damage [29]. In addition to having a specialized actin structure, the taper region also features a set of proteins that are required for stereocilia development and maintenance. Myosin VI, radixin (Rdx), chloride intracellular channel 5 (Clic5), and PTPRQ are critical for anchoring the membrane to the cytoskeleton because their loss results in stereocilia fusion, presumably because the membrane lifts from the base to cover multiple stereocilia cores [30–33]. Myosin VI also contributes to stereocilia development by reducing the number of CDH23 links found between stereocilia at early postnatal ages [34]. Additionally, Fam65b forms an oligomeric ring structure at the stereocilia base that is required to restrict taperin localization [35]. Taperin (Tprn), a protein of unknown function that is required for auditory maintenance [32,36], mislocalizes in overly elongated stereocilia that result from loss of Fam65b [35]. Together, these observations highlight that a structured base region regulates stereocilia development.
The stereocilium shaft is defined as the region between the base and the tip (Fig. 2). This region of the core is composed of highly crosslinked actin filaments notable for their uncommon stability. Important features in this region include the upper tip link density (UTLD) where the CDH23 portion of the tip link associates with an electron dense plaque. The UTLD depends on harmonin, a protein that localizes to the UTLD and can directly bind to F-actin [37]. Myosin Ic, myosin VI and myosin VIIa regulate MET adaptation, likely by applying force at the UTLD to adjust tip link tension [34,38–40].
3. Development of stereocilia
At an early developmental stage (~E14 in mouse) the apical surface of hair cells is decorated with two types of protrusions including numerous, small actin-based microvilli and a single microtubule-based primary cilium called the kinocilium. As development proceeds, the kinocilium migrates to the lateral side of the cell and adopts a position at the vertex of the developing stereocilia bundle [41]. Most of the microvilli are disassembled, leaving bare zones on the medial and lateral portions of the cell apex. Other microvilli presumably become stereocilia as the actin core is remodeled by a multitude of proteins including unconventional myosins, actin crosslinkers and actin capping proteins [23,42].
3.1. The kinocilium and bundle development
A central role for kinocilia in forming and orienting the chevron shape of stereocilia bundles is demonstrated by a number of mutations that perturb its structure and/or function [43,44]. For example, axoneme loss due to mutations in the intraflagellar transport (IFT) pathway result in bundles that are frequently misoriented such that the vertex of the bundle is not uniformly pointed along the medial-lateral axis. In addition, bundles are often split, circular or have a less defined V shape. The kinocilium responds to cues from the planar cell polarity pathway as well as cell intrinsic polarity pathways such that it is positioned between cortical domains marked by atypical protein kinase C (aPKC) and G-protein alpha i complex (Gαi) at the medial and lateral sides, respectively, of the cell apex [42,45–47]. Stereocilia bundle formation follows the boundary of these domains, with the vertex of the bundle attached to the kinocilia and the tallest stereocilia forming closest to the lateral side of the cell in a decreasing height gradient to the medial side. Phenotypes similar to mutants lacking kinocilia also arise when non-ciliary proteins are lost that regulate basal body positioning through organization of the microtubule network, including the small GTPases Cdc42 and Rac1 [48–51]. Thus, proper cortical-microtubule interactions are critical for positioning kinocilia in their correct location, and ultimately orienting the newly formed bundles properly along the medial-lateral axis.
In most cell types, primary cilia are solitary organelles thought to coordinate signaling cascades, in pathways such as the Wnt and Hedgehog pathways. In contrast, kinocilia are bound to stereocilia via links formed from the transmembrane proteins CDH23 and PCDH15 [52] (Fig. 3). Disrupting the kinocilia links results in stereocilia bundles that are reminiscent of those in mutants that lack kinocilia altogether. Thus, while kinocilia likely serve as signaling centers, their physical connection to stereocilia indispensably guides bundle shape and positioning [53].
Fig. 3.
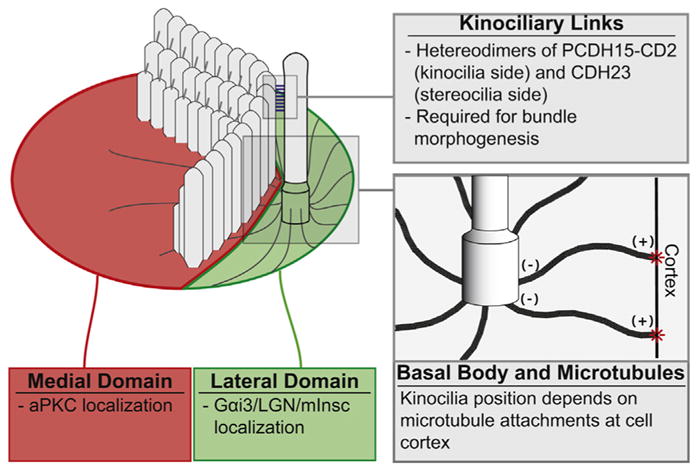
The kinocilia and stereocilia bundle morphogenesis. The bundle is positioned at the interface between two apical domains, which in turn depend on core planar cell polarity pathway and kinocilia position. In the medial domain, atypical protein kinase C (aPKC) localizes to the apical surface. G-protein alpha i (Gαi) complex localizes to the apical surface in the lateral domain. Based on this signaling, the basal body is positioned by microtubules associated with the cell cortex. Kinocilia are attached to the bundle by links composed of cadherin 23 (CDH23) and protocadherin 15 (PCDH15-CD2). When these links are disrupted by knockout of PCDH15-CD2, bundle development is abnormal and resembles that resulting from loss of kinocilia.
3.2. Actin binding proteins required for stereocilia morphogenesis
Stereocilia growth requires the addition of new filaments to increase width and actin polymerization to increase filament length. Both types of growth require unconventional myosins, many of which move along actin filaments towards the stereocilia tips, and their cargo proteins. Myosin XVa binds the adaptor protein whirlin and localizes actin binding protein EPS8 to stereocilia tips [16,18,20,54]. Loss of any component of this tripartite complex markedly reduces stereocilia length and causes a notably diminished height difference between the rows of stereocilia. EPS8, which is lost from tips in myosin XVa deficient hair cells, may stimulate growth of the stereocilia actin core through its actin crosslinking and/or capping activities [18,20].
While myosin XVa is necessary to elongate stereocilia, increasing stereocilia width depends on myosin IIIa and its paralog myosin IIIb as mice lacking both proteins have longer, thinner stereocilia [55]. Myosin IIIa and myosin IIIb also localize to stereocilia tips along with their binding partner, espin-1, which is the longest splice isoform produced from the transcriptionally regulated Espn gene. Stereocilia are thin and unstable in hair cells lacking all espin isoforms while selective knockout of the espin-1 isoform also results in longer and thinner stereocilia, at least on extrastriolar utricular hair cells [6,17]. All espin isoforms have an actin bundling domain that is necessary to form stable stereocilia [56]. Espin-1 and espin-2B, which is not highly expressed in cochlea, possess an additional actin-binding domain called xAB that allows them to form larger bundles of actin [57,58]. When espin-1 was expressed in cultured cells, this activity was sufficient to convert the treadmilling actin filaments normally observed in microvilli into a stable, non-treadmilling array. The espin-1 xAB is autoinhibited unless bound to either myosin IIIa or IIIb [58–60]. In addition to activating espin-1, a single myosin IIIa crosslinks two molecules of espin-1 together promoting the formation of even larger actin bundles [61]. Thus myosin III regulates espin-1 bundling activity to increase the diameter of stereocilia as they develop.
Crosslinking and bundling may promote stereocilia growth by making stiff actin arrays that do not buckle under membrane tension, a physical property that is necessary for extending cellular protrusions in general. Additionally, crosslinking may stabilize newly polymerized actin against depolymerization such that filament growth ratchets forward.
4. Stereocilia actin dynamics: the tip turnover model
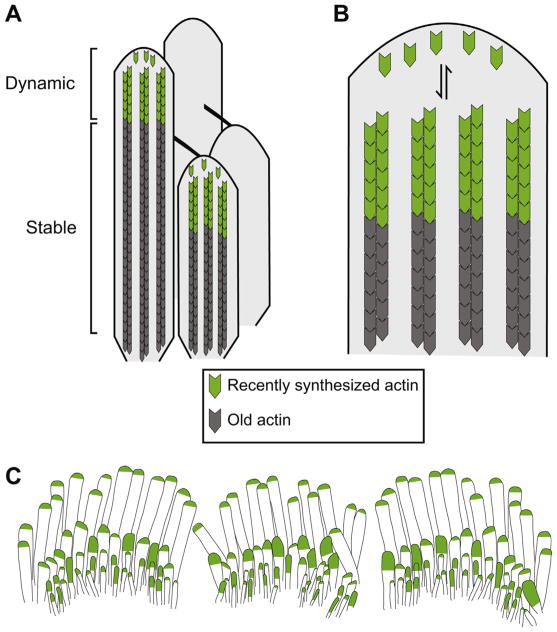
Stereocilia lengths are dictated by the assembly and disassembly of their actin-based cores. Actins are a family of very similar proteins encoded by different genes. Hair cells express two isoforms, β-actin and γ-actin (encoded by Actb and Actg1, respectively), which are 99% identical in amino acid sequence [62,63]. All actins can reversibly polymerize into polarized filaments (filamentous or F-actin) with barbed and pointed ends, which were named for their appearance when decorated with myosin S1 fragments [64]. The barbed ends of the actin filaments are oriented towards the stereocilia tip, while the pointed ends are directed towards the base [65]. Although actin can associate and dissociate from either end of the filament, the barbed end has a higher affinity for actin monomers than the pointed end. As a result, at steady-state in solution, actin subunits dissociate from the pointed end while there is concurrent polymerization at the barbed end. An actin monomer that adds to the barbed end will therefore eventually dissociate from the pointed end, a process referred to as treadmilling. Importantly, actin dynamics are regulated by several different types of actin binding proteins including filament nucleation factors that stimulate the production of new filaments, proteins that modulate assembly and disassembly at filament ends as well as crosslinking proteins that link adjacent filaments and bundling proteins that join several filaments together [14,65]. Corresponding to actin treadmilling observed in solution, early work in hair cell explants transfected with GFP-β-actin suggested that the stereocilia actin core was rapidly renewed by a similar mechanism [66,67]. However, recent reports from multiple groups in mouse systems have instead provided compelling support for a tip turnover model [68–70] (Fig. 4). In this model, actin monomers are in dynamic equilibrium with F-actin barbed ends at stereocilia tips while F-actin in the remainder of the stereocilia is static.
Fig. 4.
The tip turnover model. A) The actin core in the shaft of stereocilia (grey) is exceptionally stable. Actin incorporation in this region is not detected over a months long timecourse. In contrast, actin incorporation is much more dynamic in the tips of all stereocilia (green). B) Enlarged view of the tip showing actin monomers in dynamic equilibrium with F-actin barbed ends at stereocilia tips. Dynamic equilibrium is posited because mature stereocilia length is constant, suggesting that polymerization must be balanced by an equal amount of disassembly. The equilibrium is likely shifted to net shrinkage or elongation by factors such as regulation of actin polymerization by myosin XVa-whirlin-EPS8, myosin III/espin-1 and actin depolymerizing factor (ADF) as well as the stability of the actin core. C) Mechanotransducing stereocilia incorporate more actin at their tips. A uniform level of actin is incorporated at tips of the tallest stereocilia, which lack functional mechanoelectrical transduction (MET) channels. In contrast, the level of actin incorporation varies amongst mechanotransducing stereocilia, suggesting that tip link mediated membrane tension and ion concentration from MET channel activity influence actin turnover.
If actin filaments in stereocilia shafts are stable, the length of mature stereocilia remain constant and actin continues to incorporate at the tips, then actin subunits presumably must dissociate from the filament barbed ends in equal measure. Although barbed end actin disassembly runs counter to actin’s behavior in solution, this behavior was previously observed in Drosophila bristles. Like stereocilia, Drosophila bristles contain numerous (~500/bristle) unipolar actin filaments, which are packed together by a combination of several different actin crosslinkers [71]. The actin core eventually disassembles by depolymerizing filaments from the barbed ends of actin filaments demonstrating that large, parallel bundles of highly crosslinked actin can be primarily regulated from the barbed end [72].
4.1. Evidence supporting tip turnover model
The initial discovery suggesting stereocilia cores are highly stable was based on the measurement of total protein turnover by multi-imaging mass spectrometry (MIMS) [70]. This technique can detect the ratio of 14N and 15N isotopes with nanometer level spatial resolution. Frogs and mice were fed 15N labeled food, and MIMS was used to determine the change in the ratio of 15N to 14N along the length of stereocilia at timepoints ranging from a few days to a few months. Even at the longest timepoint, approximately half of the protein content in the stereocilia shaft did not incorporate 15N label, indicating that proteins in this region were highly stable. In contrast, protein in the cell body and at stereocilia tips more rapidly incorporated 15N label [70]. Although MIMS measures total protein incorporation, several different approaches demonstrate that the F-actin is the stable fraction of protein in stereocilia.
To more directly assess actin dynamics in stereocilia in vivo, either the β-actin or γ-actin isoform were deleted in adolescent mice via conditional cre-mediated recombination [70]. β-actin and γ-actin, which are 99% identical in amino acid sequence, colocalize along the entire length of inner hair cell stereocilia [62]. Following knockout, actin isoform specific antibody staining demonstrated that the knocked out isoform was lost from IHC stereocilia tips within a week but was retained in the remainder of the stereocilia shaft for at least several months [70]. Similar results were also observed in utricular hair cells (unpublished observation, BJP).
Expression of GFP-β-actin has also been used to monitor actin incorporation and dynamics, with the resulting data further supporting the tip turnover model. In vivo, GFP-β-actin was conditionally induced in adolescent mice [69]. Since GFP-β-actin was not expressed prior to induction, the newly expressed GFP-β-actin should incorporate rapidly into dynamic actin structures but not into stable structures. Consistent with the tip turnover model, GFP-β-actin was incorporated at stereocilia tips indicating this region was dynamic. In contrast, GFP-β-actin was largely excluded from the rest of the stereocilia core even after months of expression, again demonstrating the stable nature of these structures [69].
Temporally resolved analysis of individual cells also provides critical evidence supporting the tip turnover model because it avoids potential confusion that might arise from studying only populations of cells fixed at a single timepoint. Several independent efforts have been made along these lines using fluorescence recovery after photobleaching (FRAP) and live cell imaging to measure GFP-β-actin dynamics. In cultured explants from mice expressing transgenic GFP-β-actin in hair cells, fluorescence only recovered at the tips of IHC stereocilia following photobleaching [69]. In cultured utricles from the same transgenic line, photobleached lines across bundles neither recovered fluorescence over several days of further culture nor moved relative to their initial position, again consistent with a highly stable actin core [70]. Interestingly, in a similar experiment in zebrafish, fluorescent actin recovered much more rapidly, though still without apparent tip to base treadmilling [73]. One intriguing possibility for this difference is that the stereocilia actin core is less stable in species that can readily regenerate hair cells.
Finally, actin incorporation in mammalian stereocilia has perhaps been most accurately quantified by timelapse microscopy of GFP-β-actin transfected mouse utricular explants [68]. Tracking GFP-β-actin incorporation in individual cells over time again established that actin at stereocilia tips is relatively dynamic and that actin does not generally incorporate into the actin core of existing stereocilia. Importantly, because these observations were time resolved, stereocilia growth or genesis could be distinguished from treadmilling of the stereocilia core. Growing stereocilia incorporated GFP-β-actin into the newly formed portion of the stereocilia, but not into the existing regions of the stereocilia core. Thus, growing stereocilia add actin to their tips so that the actin core elongated while mature stereocilia only exchanged actin at their tips [68].
4.2. Stabilization of actin in stereocilia by filament crosslinking and capping
The stability of the stereocilia actin core is exceptional, especially considering the actin filament arrays that form other cellular structures such as microvilli, filopodia, dendritic spines and stress fibers are turned over in seconds to minutes. The unique complement and high concentration of actin crosslinkers and capping proteins expressed in hair cells likely stabilize the stereocilia actin core.
Multiple actin crosslinking proteins including plastin-1 (Pls1), fascin-2 (Fscn2), Xin actin binding repeat containing 2 (Xirp2), espin-3A and espin-1 (splice isoforms of Espn) are found at high levels in stereocilia [7,57,74,75]. By binding adjacent filaments together and grouping crosslinked filaments into larger bundles, these proteins likely reduce the rate of actin depolymerization in stereocilia. Demonstrating this idea, fascin-crosslinked actin filaments depolymerize at a slower rate in biochemical assays because bonds to both the actin filament and crosslinker must break for the actin subunit to dissociate [76]. The high density of crosslinks in stereocilia, estimated to be one crosslinker every 10 actin subunits [13], should contribute to stabilizing the core against disassembly, and in fact seem to promote elongation. For example, overexpression of espin isoforms in culture models and fascin-2b in zebrafish hair cells increases the length of microvilli and stereocilia, respectively [73,77]. Correspondingly, loss or mutation of plastin-1, fascin-2, EPS8L2 and XIRP2 causes stereocilia degeneration, often particularly so at the tips of stereocilia where actin monomers can dissociate from filament ends [2,7,18,74,75,78].
Actin dynamics at stereocilia tips are likely also regulated by proteins that bind and cap the barbed ends of filaments, reducing both polymerization and depolymerization of subunits. Although several proteins with capping activity have been localized to stereocilia tips, relatively little is known about their effect on stereocilia development and maintenance. Members include capping protein, TWF2 and gelsolin, which also has severing activity. Moreover, EPS8 and EPS8L2 have capping activity in addition to their actin crosslinking function. TWF2, gelsolin and EPS8L2 are enriched at the tips of stereocilia in the shorter rows of the bundle compared to the tallest row, in contrast EPS8 appears to primarily localize to the tallest row [2,20,24,79]. These data suggest that limiting dynamics at actin filament barbed ends is important for making stereocilia of different lengths. In keeping with this idea, loss of gelsolin results in long, thin and disorganized stereocilia, though this phenotype is limited to OHCs in the apex [80]. Proteins that sever actin filaments, thereby generating new, uncapped barbed ends, may antagonize barbed-end capping. In other cellular systems, this results in a burst of actin polymerization. Correspondingly, loss of the severing protein actin depolymerizing factor (ADF, also known as destrin) caused stereocilia shortening, consistent with increased capping protein occupancy preventing stereocilia growth [69].
4.3. Maintenance of mechanotransducing stereocilia
The stereocilia in the shorter rows of the bundle, in contrast to those in the tallest row, feature functional mechanosensitive channels at their tips [11]. Mechanotransducing stereocilia tips are attached to the base of a tip link, which transmits tensile force that gates the associated MET channel [12]. The stability of mature tip links relies on Ca2+ dependent interactions between the amino termini of CDH23 homodimers at the upper end and PCDH15 homodimers at the lower end [10,81,82]. CDH23 trans-membrane domains are anchored into the side of stereocilia in the taller row at the upper tip link density (UTLD) where it attaches to the actin cytoskeleton of stereocilia by interacting with myosin VIIa/sans/harmonin (a tripartite complex that localizes to the UTLD) through its carboxyl terminus [37,83].
Consistent with their fragile appearance, tip link loss and regeneration seems likely to be involved in the long-term preservation of auditory function [81]. In mouse models, relatively mild hypomorphic expression of CDH23 causes progressive, high-frequency hearing loss that resembles presbycusis [3,84]. Reduced expression may decrease the efficiency of tip link regeneration, a process that occurs over a period of several hours [3,85,86]. If tip links are lost and cannot properly reform then the shorter mechanotransducing rows of the bundle shorten and are eventually lost while row 1 heights remain unaffected [87,88].
Tip links may be required to maintain mechanotransducing stereocilia for several reasons. One possibility is that actin regulatory proteins directly bind to the cytoplasmic domain of PCDH15 and are unable to localize when tips links are lost. Another model is that tip links pull on the membrane at stereocilia tips, reducing the membrane tension force on the stereocilia core that would otherwise favor actin disassembly. These mechanisms may also apply to length regulation of enterocyte brush border microvilli, which maintain uniform height registration with their neighbors, a property that depends on tip links resembling those from stereocilia in structure and function [89]. Membrane tension transmitted though links may ordinarily limit elongation or shortening, which becomes irregular when the links are ablated. In contrast to microvilli, stereocilia tip links gate mechanotransduction channels, so ion flux may also regulate stereocilia actin dynamics and length. Morphometric analysis of stereocilia from mice with TMIE and TMC1/2 mutations that affect channel activity but not tip link integrity may separate the effects of tension and channel activity on stereocilia length regulation [90].
Regardless of the mechanism of stereocilia shortening following tip link loss, subsequent regrowth would require actin polymerization at tips. Monitoring GFP-β-actin incorporation in stereocilia revealed that row 1, whose lengths are unaffected by tip link loss, incorporated a uniform amount of GFP-β-actin across all members of the row and at a lower level than in mechanotransducing stereocilia. In contrast, individual mechanotransducing stereocilia incorporated varied levels of GFP-β-actin, consistent with stochastic tip link loss followed by stereocilia shortening and regrowth [69] (Fig. 4C). The morphology of the mechanotransducing stereocilia is also particularly sensitive to perturbation of the actin cytoskeleton as loss or mutation of several cytoskeletal proteins, including the long isoform of myosin XVa, fascin-2, β-actin, Esp8L2, actin interacting protein 1 (AIP1) and ADF result in shortening of these stereocilia while not impacting the length of the non-mechanotransducing row [2,4,5,69,27].
5. Conclusion
Stereocilia are highly specialized protrusions that feature a core of extremely stable actin filaments. The reason for such remarkable stability is unknown, but it seems likely to be important for regulation of length and longevity, two defining characteristics of stereocilia. Beyond understanding how such remarkable stability of the actin core is achieved, the apparent lack of regular actin turnover raises questions about how damaged actin filaments or subunits are repaired. One possibility is that, in a slow and unsynchronized fashion, individual actin filaments either turn over by localized treadmilling or depolymerize completely before being replaced by a new filament. These processes could lead to a gradual renewal of the actin core over time. While stability is the most notable feature of the stereocilia actin core, the dynamic region at stereocilia tips is also intriguing because actin subunit addition and loss must be regulated to maintain stereocilia length. Actin binding proteins with activities ranging from filament capping, severing and nucleation are obvious candidates to regulate tip turnover, though their actual effect on stereocilia actin dynamics and length regulation remains to be determined.
A clearer understanding of stereocilia actin regulation may prove to be useful for devising new therapeutic approaches for treating or preventing forms of progressive hearing loss that result from stereocilia degeneration. For example, perhaps a drug could stimulate endogenous repair pathways that regrow shortened stereocilia by promoting actin assembly at stereocilia tips. Targeting existing hair cells is appealing because it avoids the challenge faced by stem cell therapies of inserting a new cell into an existing and highly patterned tissue. However, manipulating existing cells faces its own challenges, including that presumably only a subset of progressive hearing loss cases result from defects in stereocilia length maintenance. This concern may be reduced if knowledge of stereocilia repair mechanisms can be combined with medical genomics to identify individuals who would most likely benefit from stereocilia restoration.
Acknowledgments
We thank Kevin J. Sonnemann for critically reading this manuscript. This work was supported by the Indiana University—Purdue School of Science and NIH grant R03DC12354 to BJP.
Footnotes
Conflict of interest
The authors have no financial or non-financial competing interests to declare.
References
- 1.Belyantseva IA, et al. Gamma-actin is required for cytoskeletal maintenance but not development. Proc Natl Acad Sci U S A. 2009;106(24):9703–9708. doi: 10.1073/pnas.0900221106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Furness DN, et al. Progressive hearing loss and gradual deterioration of sensory hair bundles in the ears of mice lacking the actin-binding protein Eps8L2. Proc Natl Acad Sci U S A. 2013;110(34):13898–13903. doi: 10.1073/pnas.1304644110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Noben-Trauth K, Zheng QY, Johnson KR. Association of cadherin 23 with polygenic inheritance and genetic modification of sensorineural hearing loss. Nat Genet. 2003;35(1):21–23. doi: 10.1038/ng1226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Perrin BJ, Sonnemann KJ, Ervasti JM. β-actin and γ-actin are each dispensable for auditory hair cell development but required for stereocilia maintenance. PLoS Genet. 2010;6(10):e1001158. doi: 10.1371/journal.pgen.1001158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Perrin BJ, et al. beta-Actin and fascin-2 cooperate to maintain stereocilia length. J Neurosci. 2013;33(19):8114–8121. doi: 10.1523/JNEUROSCI.0238-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sekerková G, Richter CP, Bartles JR. Roles of the espin actin-bundling proteins in the morphogenesis and stabilization of hair cell stereocilia revealed in CBA/CaJ congenic jerker mice. PLoS Genet. 2011;7(3):e1002032. doi: 10.1371/journal.pgen.1002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Taylor R, et al. Absence of plastin 1 causes abnormal maintenance of hair cell stereocilia and a moderate form of hearing loss in mice. Hum Mol Genet. 2014;24(1):37–49. doi: 10.1093/hmg/ddu417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ashmore J. Cochlear outer hair cell motility. Physiol Rev. 2008;88(1):173–210. doi: 10.1152/physrev.00044.2006. [DOI] [PubMed] [Google Scholar]
- 9.Brownell WE, et al. Evoked mechanical responses of isolated cochlear outer hair cells. Science. 1985;227(4683):194–196. doi: 10.1126/science.3966153. [DOI] [PubMed] [Google Scholar]
- 10.Kazmierczak P, et al. Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature. 2007;449(7158):87–91. doi: 10.1038/nature06091. [DOI] [PubMed] [Google Scholar]
- 11.Beurg M, et al. Localization of inner hair cell mechanotransducer channels using high-speed calcium imaging. Nat Neurosci. 2009;12(5):553–558. doi: 10.1038/nn.2295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fettiplace R, Hackney CM. The sensory and motor roles of auditory hair cells. Nat Rev Neurosci. 2006;7(1):19–29. doi: 10.1038/nrn1828. [DOI] [PubMed] [Google Scholar]
- 13.Shin JB, et al. Molecular architecture of the chick vestibular hair bundle. Nat Neurosci. 2013;16(3):365–374. doi: 10.1038/nn.3312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tilney LG, Derosier DJ, Mulroy MJ. The organization of actin filaments in the stereocilia of cochlear hair cells. J Cell Biol. 1980;86(1):244–259. doi: 10.1083/jcb.86.1.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tilney LG, et al. Actin filaments, stereocilia, and hair cells of the bird cochlea. II. Packing of actin filaments in the stereocilia and in the cuticular plate and what happens to the organization when the stereocilia are bent. J Cell Biol. 1983;96(3):822–834. doi: 10.1083/jcb.96.3.822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Belyantseva IA, et al. Myosin-XVa is required for tip localization of whirlin and differential elongation of hair-cell stereocilia. Nat Cell Biol. 2005;7(2):148–156. doi: 10.1038/ncb1219. [DOI] [PubMed] [Google Scholar]
- 17.Ebrahim S, et al. Stereocilia-staircase spacing is influenced by myosin III motors and their cargos espin-1 and espin-like. Nat Commun. 2016;7:10833. doi: 10.1038/ncomms10833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Manor U, et al. Regulation of stereocilia length by myosin XVa and whirlin depends on the actin-regulatory protein Eps8. Curr Biol. 2011;21(2):167–172. doi: 10.1016/j.cub.2010.12.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Salles FT, et al. Myosin IIIa boosts elongation of stereocilia by transporting espin 1 to the plus ends of actin filaments. Nat Cell Biol. 2009;11(4):443–450. doi: 10.1038/ncb1851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zampini V, et al. Eps8 regulates hair bundle length and functional maturation of mammalian auditory hair cells. PLoS Biol. 2011;9(4):e1001048. doi: 10.1371/journal.pbio.1001048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Merritt RC, et al. Myosin IIIB uses an actin-binding motif in its espin-1 cargo to reach the tips of actin protrusions. Curr Biol. 2012;22(4):320–325. doi: 10.1016/j.cub.2011.12.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Delprat B, et al. Myosin XVa and whirlin, two deafness gene products required for hair bundle growth, are located at the stereocilia tips and interact directly. Hum Mol Genet. 2005;14(3):401–410. doi: 10.1093/hmg/ddi036. [DOI] [PubMed] [Google Scholar]
- 23.Avenarius MR, et al. Correlation of actin crosslinker and capper expression levels with stereocilia growth phases. Mol Cell Proteomics. 2014;13(2):606–620. doi: 10.1074/mcp.M113.033704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Peng AW, et al. Twinfilin 2 regulates actin filament lengths in cochlear stereocilia. J Neurosci. 2009;29(48):15083–15088. doi: 10.1523/JNEUROSCI.2782-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schafer DA, Jennings PB, Cooper JA. Dynamics of capping protein and actin assembly in vitro: uncapping barbed ends by polyphosphoinositides. J Cell Biol. 1996;135(1):169–179. doi: 10.1083/jcb.135.1.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rzadzinska AK, et al. MyosinVIIa interacts with Twinfilin-2 at the tips of mechanosensory stereocilia in the inner ear. PLoS One. 2009;4(9):e7097. doi: 10.1371/journal.pone.0007097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fang Q, et al. The 133-kDa N-terminal domain enables myosin 15 to maintain mechanotransducing stereocilia and is essential for hearing. elife. 2015;4:e08627. doi: 10.7554/eLife.08627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Furness DN, et al. The dimensions and composition of stereociliary rootlets in mammalian cochlear hair cells: comparison between high- and low-frequency cells and evidence for a connection to the lateral membrane. J Neurosci. 2008;28(25):6342–6353. doi: 10.1523/JNEUROSCI.1154-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kitajiri S, et al. Actin-Bundling Protein TRIOBP Forms Resilient Rootlets of Hair Cell Stereocilia Essential for Hearing. Cell. 2010;141(5):1–22. doi: 10.1016/j.cell.2010.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Goodyear RJ, et al. Hair bundle defects and loss of function in the vestibular end organs of mice lacking the receptor-like inositol lipid phosphatase PTPRQ. J Neurosci. 2012;32(8):2762–2772. doi: 10.1523/JNEUROSCI.3635-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pataky F, Pironkova R, Hudspeth AJ. Radixin is a constituent of stereocilia in hair cells. Proc Natl Acad Sci U S A. 2004;101(8):2601–2606. doi: 10.1073/pnas.0308620100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Salles FT, et al. CLIC5 stabilizes membrane-actin filament linkages at the base of hair cell stereocilia in a molecular complex with radixin, taperin, and myosin VI. Cytoskeleton (Hoboken) 2014;71(1):61–78. doi: 10.1002/cm.21159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhao H, et al. Large membrane domains in hair bundles specify spatially constricted radixin activation. J Neurosci. 2012;32(13):4600–4609. doi: 10.1523/JNEUROSCI.6184-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Marcotti W, et al. The acquisition of mechano-electrical transducer current adaptation in auditory hair cells requires myosin VI. J Physiol. 2016;594(13):3667–3681. doi: 10.1113/JP272220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhao B, Wu Z, Muller U. Murine Fam65b forms ring-like structures at the base of stereocilia critical for mechanosensory hair cell function. eLife. 2016:e14222.5. doi: 10.7554/eLife.14222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rehman AU, et al. Targeted capture and next-Generation sequencing identifies C9orf75, encoding taperin, as the mutated gene in nonsyndromic deafness DFNB79. Am J Hum Genet. 2010 doi: 10.1016/j.ajhg.2010.01.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Grillet N, et al. Harmonin mutations cause mechanotransduction defects in cochlear hair cells. Neuron. 2009;62(3):375–387. doi: 10.1016/j.neuron.2009.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gillespie PG, Cyr JL. Myosin-1c, the hair cell’s adaptation motor. Annu Rev Physiol. 2004;66:521–545. doi: 10.1146/annurev.physiol.66.032102.112842. [DOI] [PubMed] [Google Scholar]
- 39.Stauffer EA, et al. Fast adaptation in vestibular hair cells requires myosin-1c activity. Neuron. 2005;47(4):541–553. doi: 10.1016/j.neuron.2005.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kros CJ, et al. Reduced climbing and increased slipping adaptation in cochlear hair cells of mice with Myo7a mutations. Nat Neurosci. 2002;5(1):41–47. doi: 10.1038/nn784. [DOI] [PubMed] [Google Scholar]
- 41.Tilney LG, Tilney MS, DeRosier DJ. Actin filaments, stereocilia, and hair cells: how cells count and measure. Annu Rev Cell Biol. 1992;8:257–274. doi: 10.1146/annurev.cb.08.110192.001353. [DOI] [PubMed] [Google Scholar]
- 42.Tarchini B, Jolicoeur C, Cayouette M. A molecular blueprint at the apical surface establishes planar asymmetry in cochlear hair cells. Dev Cell. 2013;27(1):88–102. doi: 10.1016/j.devcel.2013.09.011. [DOI] [PubMed] [Google Scholar]
- 43.Jones C, et al. Ciliary proteins link basal body polarization to planar cell polarity regulation. Nat Genet. 2008;40(1):69–77. doi: 10.1038/ng.2007.54. [DOI] [PubMed] [Google Scholar]
- 44.May-Simera HL, et al. Ciliary proteins Bbs8 and Ift20 promote planar cell polarity in the cochlea. Development. 2015;142(3):555–566. doi: 10.1242/dev.113696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bhonker Y, et al. The GPSM2/LGN GoLoco motifs are essential for hearing. Mamm Genome. 2016;27(1–2):29–46. doi: 10.1007/s00335-015-9614-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ezan J, et al. Primary cilium migration depends on G-protein signalling control of subapical cytoskeleton. Nat Cell Biol. 2013;15(9):1107–1115. doi: 10.1038/ncb2819. [DOI] [PubMed] [Google Scholar]
- 47.Ezan J, Montcouquiol M. Revisiting planar cell polarity in the inner ear. Semin Cell Dev Biol. 2013;24(5):499–506. doi: 10.1016/j.semcdb.2013.03.012. [DOI] [PubMed] [Google Scholar]
- 48.Grimsley-Myers CM, et al. The small GTPase Rac1 regulates auditory hair cell morphogenesis. J Neurosci. 2009;29(50):15859–15869. doi: 10.1523/JNEUROSCI.3998-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kirjavainen A, et al. The Rho GTPase Cdc42 regulates hair cell planar polarity and cellular patterning in the developing cochlea. Biol Open. 2015;4(4):516–526. doi: 10.1242/bio.20149753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sipe CW, et al. Lis1 mediates planar polarity of auditory hair cells through regulation of microtubule organization. Development. 2013;140(8):1785–1795. doi: 10.1242/dev.089763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ueyama T, et al. Maintenance of stereocilia and apical junctional complexes by Cdc42 in cochlear hair cells. J Cell Sci. 2014;127(Pt 9):2040–2052. doi: 10.1242/jcs.143602. [DOI] [PubMed] [Google Scholar]
- 52.Goodyear RJ, et al. Asymmetric distribution of cadherin 23 and protocadherin 15 in the kinocilial links of avian sensory hair cells. J Comp Neurol. 2010;518(21):4288–4297. doi: 10.1002/cne.22456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Webb SW, et al. Regulation of PCDH15 function in mechanosensory hair cells by alternative splicing of the cytoplasmic domain. Development. 2011;138(8):1607–1617. doi: 10.1242/dev.060061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Delprat B, et al. Deafness and cochlear fibrocyte alterations in mice deficient for the inner ear protein otospiralin. Mol Cell Biol. 2005;25(2):847–853. doi: 10.1128/MCB.25.2.847-853.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lelli A, et al. Class III myosins shape the auditory hair bundles by limiting microvilli and stereocilia growth. J Cell Biol. 2016;212(2):231–244. doi: 10.1083/jcb.201509017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bartles JR, et al. Small espin: a third actin-bundling protein and potential forked protein ortholog in brush border microvilli. J Cell Biol. 1998;143(1):107–119. doi: 10.1083/jcb.143.1.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sekerkova G, et al. Differential expression of espin isoforms during epithelial morphogenesis: stereociliogenesis and postnatal maturation in the developing inner ear. Dev Biol. 2006;291(1):83–95. doi: 10.1016/j.ydbio.2005.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zheng L, Beeler DM, Bartles JR. Characterization and regulation of an additional actin filament-binding site in large isoforms of the stereocilia actin-bundling protein espin. J Cell Sci. 2014;127(Pt 6):1306–1317. doi: 10.1242/jcs.143255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ebrahim S, et al. Stereocilia-staircase spacing is influenced by myosin III motors and their cargos espin-1 and espin-like. Nat Commun. 2016;7:10833. doi: 10.1038/ncomms10833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu H, et al. Myosin III-mediated cross-linking and stimulation of actin bundling activity of Espin. elife. 2016;5:e12856. doi: 10.7554/eLife.12856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liu H, et al. Myosin III-mediated cross-linking and stimulation of actin bundling activity of Espin. Elife. 2016;5 doi: 10.7554/eLife.12856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Perrin BJ, Sonnemann KJ, Ervasti JM. beta-actin and gamma-actin are each dispensable for auditory hair cell development but required for Stereocilia maintenance. PLoS Genet. 2010;6(10):e1001158. doi: 10.1371/journal.pgen.1001158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Slepecky NB, Savage JE. Expression of actin isoforms in the guinea pig organ of Corti: muscle isoforms are not detected. Hear Res. 1994;73(1):16–26. doi: 10.1016/0378-5955(94)90278-x. [DOI] [PubMed] [Google Scholar]
- 64.Pollard TD. Actin and actin-binding proteins. Cold Spring Harb Perspect Biol. 2016;8(8):a018226. doi: 10.1101/cshperspect.a018226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Flock A, Cheung HC. Actin filaments in sensory hairs of inner ear receptor cells. J Cell Biol. 1977;75(2 Pt 1):339–343. doi: 10.1083/jcb.75.2.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rzadzinska AK, et al. An actin molecular treadmill and myosins maintain stereocilia functional architecture and self-renewal. J Cell Biol. 2004;164(6):887–897. doi: 10.1083/jcb.200310055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schneider ME, et al. Rapid renewal of auditory hair bundles. Nature. 2002;418(6900):837–838. doi: 10.1038/418837a. [DOI] [PubMed] [Google Scholar]
- 68.Drummond MC, et al. Live-cell imaging of actin dynamics reveals mechanisms of stereocilia length regulation in the inner ear. Nat Commun. 2015;6:6873. doi: 10.1038/ncomms7873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Narayanan P, et al. Length regulation of mechanosensitive stereocilia depends on very slow actin dynamics and filament-severing proteins. Nat Commun. 2015;6:6855. doi: 10.1038/ncomms7855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhang DS, et al. Multi-isotope imaging mass spectrometry reveals slow protein turnover in hair-cell stereocilia. Nature. 2012;481(7382):520–524. doi: 10.1038/nature10745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tilney LG, et al. F-actin bundles in Drosophila bristles are assembled from modules composed of short filaments. J Cell Biol. 1996;135(5):1291–1308. doi: 10.1083/jcb.135.5.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Guild GM, et al. Actin filament turnover removes bundles from Drosophila bristle cells. J Cell Sci. 2002;115(Pt 3):641–653. doi: 10.1242/jcs.115.3.641. [DOI] [PubMed] [Google Scholar]
- 73.Hwang P, et al. The stereociliary paracrystal is a dynamic cytoskeletal scaffold In vivo. Cell Rep. 2015;13(7):1287–1294. doi: 10.1016/j.celrep.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Scheffer DI, et al. XIRP2, an actin-binding protein essential for inner ear hair-cell stereocilia. Cell Rep. 2015;10(11):1811–1818. doi: 10.1016/j.celrep.2015.02.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shin JB, et al. The R109H variant of fascin-2, a developmentally regulated actin crosslinker in hair-cell stereocilia, underlies early-onset hearing loss of DBA/2J mice. J Neurosci. 2010;30(29):9683–9694. doi: 10.1523/JNEUROSCI.1541-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Schmoller KM, Semmrich C, Bausch AR. Slow down of actin depolymerization by cross-linking molecules. J Struct Biol. 2011;173(2):350–357. doi: 10.1016/j.jsb.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 77.Loomis PA, et al. Espin cross-links cause the elongation of microvillus-type parallel actin bundles in vivo. J Cell Biol. 2003;163(5):1045–1055. doi: 10.1083/jcb.200309093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Francis SP, et al. A short splice form of Xin-actin binding repeat containing 2 (XIRP2) lacking the Xin repeats is required for maintenance of stereocilia morphology and hearing function. J Neurosci. 2015;35(5):1999–2014. doi: 10.1523/JNEUROSCI.3449-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Olt J, et al. The actin-binding proteins eps8 and gelsolin have complementary roles in regulating the growth and stability of mechanosensory hair bundles of mammalian cochlear outer hair cells. PLoS One. 2014;9(1):e87331. doi: 10.1371/journal.pone.0087331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mburu P, et al. Gelsolin plays a role in the actin polymerization complex of hair cell stereocilia. PLoS One. 2010;5(7):e11627. doi: 10.1371/journal.pone.0011627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Alagramam KN, et al. Mutations in protocadherin 15 and cadherin 23 affect tip links and mechanotransduction in mammalian sensory hair cells. PLoS One. 2011;6(4):e19183. doi: 10.1371/journal.pone.0019183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Siemens J, et al. Cadherin 23 is a component of the tip link in hair-cell stereocilia. Nature. 2004;428(6986):950–955. doi: 10.1038/nature02483. [DOI] [PubMed] [Google Scholar]
- 83.Grati M, Kachar B. Myosin VIIa and sans localization at stereocilia upper tip-link density implicates these Usher syndrome proteins in mechanotransduction. Proc Natl Acad Sci U S A. 2011;108(28):11476–11481. doi: 10.1073/pnas.1104161108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Johnson KR, et al. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear Res. 1997;114(1–2):83–92. doi: 10.1016/s0378-5955(97)00155-x. [DOI] [PubMed] [Google Scholar]
- 85.Indzhykulian AA, et al. Molecular remodeling of tip links underlies mechanosensory regeneration in auditory hair cells. PLoS Biol. 2013;11(6):e1001583. doi: 10.1371/journal.pbio.1001583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Miyasaka Y, et al. Compound heterozygosity of the functionally null Cdh23(v-ngt) and hypomorphic Cdh23(ahl) alleles leads to early-onset progressive hearing loss in mice. Exp Anim. 2013;62(4):333–346. doi: 10.1538/expanim.62.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Caberlotto E, et al. Coupling of the mechanotransduction machinery and F-actin polymerization in the cochlear hair bundles. Bioarchitecture. 2011;1(4):169–174. doi: 10.4161/bioa.1.4.17532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Caberlotto E, et al. Usher type 1G protein sans is a critical component of the tip-link complex, a structure controlling actin polymerization in stereocilia. Proc Natl Acad Sci U S A. 2011;108(14):5825–5830. doi: 10.1073/pnas.1017114108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Crawley SW, Mooseker MS, Tyska MJ. Shaping the intestinal brush border. J Cell Biol. 2014;207(4):441–451. doi: 10.1083/jcb.201407015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zhao B, et al. TMIE is an essential component of the mechanotransduction machinery of cochlear hair cells. Neuron. 2014;84(5):954–967. doi: 10.1016/j.neuron.2014.10.041. [DOI] [PMC free article] [PubMed] [Google Scholar]