Abbreviations
- ICG
indocyanine green
- NTCP
sodium‐taurocholate cotransporting polypeptide
- OATP
organic anion transporting polypeptide
- P1‐P4
patients 1‐4
The indocyanine green (ICG) retention test is widely used for preoperative evaluation of liver function. Individuals who have markedly poor ICG clearance without severe liver diseases are defined as patients with a “constitutional ICG excretory defect.”1 However, the underlying molecular mechanisms remain unknown. We hypothesized that the ICG clearance defect involved impaired function of hepatic uptake transporters such as organic anion transporting polypeptide 1B1 (OATP1B1, gene symbol SLCO1B1), OATP1B3 (SLCO1B3), and sodium‐taurocholate cotransporting polypeptide (NTCP, SLC10A1). Homozygous inactivation of both SLCO1B1 and SLCO1B3 causes Rotor syndrome, for which three disease‐causing haplotypes linked to SLCO1B1 and SLCO1B3 are documented.2 We also reported a Japanese population‐specific haplotype: a homozygous c.1738C>T (p.R580X) nonsense mutation in SLCO1B1, and a homozygous insertion of a 6.5‐kbp long interspersed element (LINE‐1, L1) retrotransposon in intron 5 of SLOC1B3 that induced exon skipping and a premature stop codon that generated truncated proteins.3 A recent case report showed that homozygous c.755G>A (p.R252H) missense mutation in SLC10A1 led to the functional loss of NTCP resulting in hypercholanemia.4 Here, by genotyping these transporters, we report cases of an ICG clearance defect due to a deficiency of OATP1B3.
Cases
Four patients (P1‐P4) underwent an ICG retention test—popular in Japan—for assessing liver function. All four patients exhibited extremely high ICG retention at 15 minutes, ranging from 70.8% to 90.7% (Table 1A). ICG elimination rate constants for P3 and P4 were markedly low. Background liver diseases included primary biliary cholangitis (Ludwig stage I), fatty liver, and hepatocellular carcinoma. P4 was taking metformin, telmisartan, rosuvastatin, and alogliptin, while the other patients were not taking any drugs. Except for slight elevations in serum alanine aminotransferase and alkaline phosphatase levels, laboratory data were normal. Liver biopsy denied the presence of liver cirrhosis or cholestasis in all patients.
Table 1.
Patient Characteristics and Genotyping of SLCO1B1, SLCO1B3, and SLC10A1
| Subject |
Age (years) |
Gender | Liver Disease |
Platelets (×104/μL) |
Albumin (g/dL) |
ALT (IU/L) |
ALP (IU/L) |
T. Bil (mg/dL) |
D. Bil (mg/dL) |
Total bile acid (μmol/L) |
PT‐INR |
ICG‐ R15 (%) |
ICG‐k |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A. Patient Characteristics a | |||||||||||||
| P1 | 44 | F |
PBC (stage I) |
12.6 | 4.3 | 63 | 417 | 0.7 | 0.2 | 3.3 | 0.88 | 90.7 | NA |
| P2 | 58 | F |
PBC (stage I) |
23.6 | 4.1 | 32 | 443 | 0.8 | 0.3 | 2.5 | 0.95 | 79.3 | NA |
| P3 | 62 | M | Fatty liver | 23.4 | 4.4 | 38 | 313 | 0.8 | 0.3 | 1.0 | 0.97 | 86 | 0.024 |
| P4 | 53 | F | HCC | 27.1 | 4.6 | 27 | 201 | 1.4 | 0.1 | 5.2 | 1.26 | 70.8 | 0.023 |
| Subject | SLCO1B1 | SLCO1B3 | SLC10A1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| c.388A>G | c.571T>C | c.757C>T | c.1738C>T |
chr12: 21239315T>C |
chr12: 21239628T>G |
chr12: 21239652A>G |
c.1747+1 | L1 | c.225G>A | c.263T>C | c.755G>A | |
| p.N130D | p.L191L | p.R253X | p.R580X | 3′UTR | 3′UTR | 3′UTR | G>A | insertion | p.T75T | p.I88T | p.R252H | |
| rs2306283 | rs4149057 | rs183501729 | rs71581941 | rs4149087 | rs4149088 | rs4646285 | rs148467625 | rs147226818 | ||||
| B. Genotyping of SLCO1B1, SLCO1B3, and SLC10A1 b | ||||||||||||
| P1 | A/G | T/C | C/C | C/C | T/C | T/T | A/A | G/G | L1/L1 | G/A | T/C | G/G |
| P2 | G/G | T/T | C/C | C/C | T/T | T/G | A/G | G/G | L1/L1 | G/G | T/T | G/G |
| P3 | G/G | T/T | C/C | C/C | T/T | T/G | A/G | G/G | L1/L1 | G/G | T/T | G/G |
| P4 | G/G | T/T | C/C | C/C | T/T | T/G | A/G | G/G | L1/L1 | G/G | T/T | G/G |
Normal reference values of laboratory data: platelets, 13.0‐36.9 × 104/μL; albumin, 3.8‐5.2 g/dL; ALT, 5‐40 IU/L; ALP, 115‐359 IU/L; total bilirubin, 0.3‐1.2 mg/dL; direct bilirubin, <0.4 mg/dL; total bile acid, <10 μmol/L; PT‐INR, 0.85‐1.15; ICG‐R15, <10%; ICG‐k, 0.179‐0.199.
c.757C>T and c.1738C>T in SLCO1B1 and c.1747+1G>A and L1 insertion in SLCO1B3 are variants associated with Rotor syndrome, and 755G>A in SLC10A1 is a variant associated with hypercholanemia (shown in bold). The following variants have not been reported to be pathogenic: c.388A>G, c.571T>C, chr12:21239315T>C, chr12:21239628T>G and chr12:21239652A>G in SLCO1B1, and c.225G>A and c.263T>C in SLC10A1.
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; D. Bil, direct bilirubin; HCC, hepatocellular carcinoma; ICG‐R15, indocyanine green retention rate at 15 minutes; ICG‐k, indocyanine green elimination rate constant; NA, not available; PBC, primary biliary cholangitis; PT‐INR, international normalized ratio of prothrombin time; T. Bil, total bilirubin.
Sequencing of all SLCO1B1 and SLC10A1 exons after obtaining written informed consent did not reveal any pathogenic variants, whereas all patients were homozygous for the L1 insertion in intron 5 of SLOC1B3 (Table 1B). We confirmed the lack of hepatic OATP1B3 expression by immunohistochemistry in three patients (Fig. 1).
Figure 1.
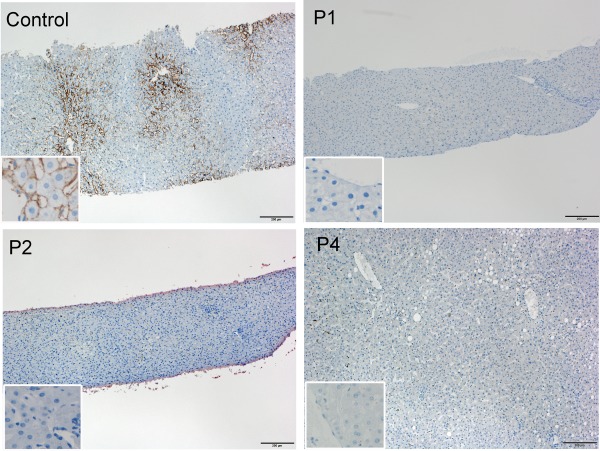
Hepatic OATP1B3 protein expression in patients P1, P2, and P4 and a control. Positive staining for OATP1B3 is observed along the basolateral membrane of hepatocytes in centrilobular areas in control liver tissue (antibody HPA004943; Sigma‐Aldrich, St. Louis, MO). In contrast, OATP1B3 expression is not detected in any of the patients. Scale bar, 200 μm. Inset, high magnification.
Discussion
OATP1B3 and NTCP can transport ICG in vitro.5 An unrevealed deficiency of NTCP in addition to the absence of OATP1B3 might have contributed to the lack of ICG uptake in our patients. However, this seems unlikely because NTCP deficiency–associated hypercholanemia4 was not observed (Table 1A). ICG is reported to accumulate in some hepatocellular carcinoma nodules after intravenous injection. Hepatocellular carcinoma tissues with ICG accumulation had stronger OATP1B3 expression than those without ICG accumulation, suggesting the involvement of this transporter in ICG uptake.
We conclude that OATP1B3 is the major hepatic transporter for ICG uptake and that its deficiency profoundly impairs ICG clearance. A solitary defect in OATP1B3 does not appear to cause conjugated hyperbilirubinemia as reported.2 The allele frequency of the L1 insertion in SLCO1B3 was 0.054 in a Japanese population,3 suggesting that homozygosity occurs in approximately 1 in 400 individuals. Because OATP1B3 is involved in the hepatic uptake of various drugs such as statins, bosentan, olmesartan, erythromycin, and docetaxel, the risk of drug‐induced toxicity is likely increased in these individuals.
Patients with impaired function of a specific transporter, such as those reported here, can provide important insights into understanding the complicated hepatic transport systems.
Acknowledgment
We thank Reiko Orii, Satsuki Ieda, Tadayuki Sato, Haruko Ogawa, Masayuki Tanaka, Hideki Hayashi, Hideyuki Matsuzawa, and Noboru Kawabe for technical assistance.
Potential conflict of interest: Nothing to report.
REFERENCES
- 1. Namihisa T, Nambu M, Kobayashi N, Kuroda H. Nine cases with marked retention of indocyanine green test and normal sulfobromophthalein test without abnormal liver histology: constitutional indocyanine green excretory defect. Hepatogastroenterology 1981;28:6‐12. [PubMed] [Google Scholar]
- 2. van de Steeg E, Stranecky V, Hartmannova H, Noskova L, Hrebicek M, Wagenaar E, et al. Complete OATP1B1 and OATP1B3 deficiency causes human Rotor syndrome by interrupting conjugated bilirubin reuptake into the liver. J Clin Invest 2012;122:519‐528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kagawa T, Oka A, Kobayashi Y, Hiasa Y, Kitamura T, Sakugawa H, et al. Recessive inheritance of population‐specific intronic LINE‐1 insertion causes a Rotor syndrome phenotype. Hum Mutat 2015;36:327‐332. [DOI] [PubMed] [Google Scholar]
- 4. Vaz FM, Paulusma CC, Huidekoper H, de Ru M, Lim C, Koster J, et al. Sodium taurocholate cotransporting polypeptide (SLC10A1) deficiency: conjugated hypercholanemia without a clear clinical phenotype. Hepatology 2015;61:260‐267. [DOI] [PubMed] [Google Scholar]
- 5. de Graaf W, Hausler S, Heger M, van Ginhoven TM, van Cappellen G, Bennink RJ, et al. Transporters involved in the hepatic uptake of (99m)Tc‐mebrofenin and indocyanine green. J Hepatol 2011;54:738‐745. [DOI] [PubMed] [Google Scholar]


