Abstract
Hematopoietic cell transplantation (HCT) is a critical treatment of patients with high-risk hematopoietic malignancies, hematological deficiencies, and other immune diseases. In allogeneic HCT (allo-HCT), donor-derived T cells recognize host tissues as foreign, causing graft-versus-host disease (GVHD) which is a main contributor to morbidity and mortality. The intestine is one of the organs most severely affected by GVHD and research has recently highlighted the importance of bacteria, particularly the gut microbiota, in HCT outcome and in GVHD development. Loss of intestinal bacterial diversity is common during the course of HCT and is associated with GVHD development and treatment with broad-spectrum antibiotics. Loss of intestinal diversity and outgrowth of opportunistic pathogens belonging to the phylum Proteobacteria and Enterococcus genus have also been linked to increased treatment-related mortality including GVHD, infections, and organ failure after allo-HCT. Experimental studies in allo-HCT animal models have shown some promising results for prebiotic and probiotic strategies as prophylaxis or treatment of GVHD. Continuous research will be important to define the relation of cause and effect for these associations between microbiota features and HCT outcomes. Importantly, studies focused on geographic and cultural differences in intestinal microbiota are necessary to define applicability of new strategies targeting the intestinal microbiota.
Introduction
Hematopoietic cell transplantation (HCT) is used as a potentially curative therapy for patients with high-risk hematopoietic malignancies in addition to hematological deficiencies and other immune diseases. In HCT, a fully or partially myeloablative conditioning regimen is followed by infusion of autologous (recipient-derived) or allogeneic (donor-derived) hematopoietic cells. The conditioning regimen depletes malignant or deficient hematopoietic cells as well as healthy hematopoietic cells while the infusion of autologous or allogeneic cells reconstitutes the patient with a new healthy hematopoietic system. In allogeneic HCT (allo-HCT), the donor cells also help to prevent relapse of malignancy by killing residual malignant cells in a process called graft-versus-tumor. Although HCT has increased the survival prospects for many patients, this treatment is not without risks. Intensive pretransplant conditioning regimens cause collateral damage to organs such as the liver and gut and the beneficial alloreactivity that eliminates residual malignant cells also causes graft-versus-host disease (GVHD) in 40% to 50% of allo-HCT patients when alloreactive donor T cells attack healthy tissues.1 GVHD-induced inflammation is commonly seen in the liver, lung, skin, and intestine and is a major contributor to mortality after allo-HCT.2 Moreover, during the interval between hematopoietic depletion and immune reconstitution, the patient’s immune defense suffers an important drawback, leaving patients particularly susceptible to infections by bacteria and from reactivation of latent pathogens such as cytomegalovirus. Research has recently highlighted the importance of bacteria, particularly the gut microbiota, in HCT outcome and in GVHD development. This review addresses current knowledge on the link between gut dysbiosis and intestinal GVHD, and how possible flora interventions may hold promise in the prevention and treatment of GVHD.
The intestinal microbiota
The human body is host to 1013 to 1014 microbial organisms.3 The vast majority of these organisms are bacteria although viruses and fungi are also present at considerable amount and diversity.4-6 The gut is the main site of microbial colonization harboring a complex composition of bacteria referred to as the intestinal microbiota or sometimes as the intestinal microbiome, although the latter more often refers to the total genetic makeup of the microbiota rather than the microorganisms themselves. The intestinal microbiota synthesize essential amino acids and vitamins and process otherwise indigestible components of our diet such as plant polysaccharides.7 A healthy and balanced intestinal microbiota also protects the host from viral and bacterial infections by preventing colonization of pathogens in the gastrointestinal tract.8,9 The importance and impact of the intestinal microbiota on health and disease has been increasingly revealed during the last decades, and imbalances in the intestinal microbiota have been linked to obesity,10 type 2 diabetes,11,12 irritable bowel syndrome,13 and autoimmune diseases such as rheumatoid arthritis.14 New insights have been enabled by the development of molecular analysis methods (ie, 16S ribosomal RNA sequencing15) that overcome the limitations of culture-based techniques in the identification of bacterial strains. The bacterial phyla Firmicutes and Bacteroidetes are the most prevalent commensals in the intestinal microbiota, but Proteobacteria, Fusobacteria, and Actinobacteria are also present.16 Over 1000 different bacterial species have been identified in samples from human intestines, but the composition at the species level varies greatly between individuals and is influenced by diet, lifestyle, and environmental factors17,18 and the number of species present in any single individual may be as low as 160.4 Despite a great variation in abundance of specific bacterial strains, the total set of microbial genes and the presence of genes involved in specific metabolic pathways (the metagenome) are relatively stable between healthy individuals.19
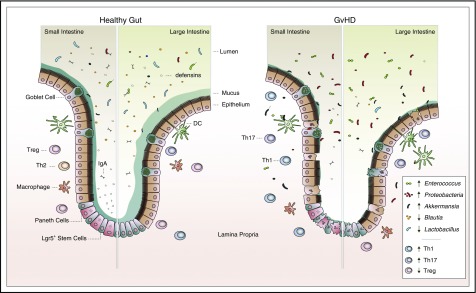
The gut immune system represents the first line of defense to ingested microorganisms and other substances. The immune system has evolved not only to protect against potential pathogens but also to tolerate beneficial commensal bacteria, allowing us to live in symbiosis with our intestinal microbiota. The intestinal immune system keeps the mucosal barrier in check through secretion of antimicrobial peptides and antibodies, and the commensal microbiota reciprocally molds the host gut physiology. Germ-free mice, housed without exposure to any microorganisms, have altered intestinal morphology and slower turnover of intestinal epithelial cells compared with conventionally raised mice.20,21 When exposed to intestinal bacteria, germ-free mice quickly develop innate immune responses, and structural changes in the intestinal epithelial crypts are seen as early as 4 days after exposure to a conventional microbiota, with an increase in tumor necrosis factor-α and interferon-γ (IFN-γ) expression and major histocompatibility complex class I upregulation.20,22 The gut adaptive immune system is also quickly activated after exposure to commensal bacteria, with an increase in the expression of major histocompatibility complex class II molecules and an increase of T cells by day 8.22 Gut exposure to bacteria is also linked to recruitment of T regulatory lymphocytes (Tregs) with the production of interleukin-10 (IL-10) and transforming growth factor-β, as well as Th17 cells which secrete IL-17 and IL-22.23,24 Tregs and Th17 cells are fundamental in order to mount a proper immune response to antigens within the gut but also influence systemic immunity.25,26 Moreover, as cited previously, intestinal bacteria are not only responsible for breaking down complex dietary fibers but are also important for the production of a variety of metabolites with active functions in the gut. The best studied of these are short-chain fatty acids (SCFAs) such as butyrate, propionate, and acetate, which serve as an energy source for intestinal epithelial cells and induce protective regulatory immune responses both locally in the gut and systemically.27-29 In summary, the microbiota have an important role in modulating the immune system during homeostasis and intestinal disease.
GVHD pathophysiology
GVHD is caused by the alloactivation of T cells, which recognize host antigens as foreign, causing an autoimmune-like attack in recipient organs such as lungs, liver, gut, thymus, the hematopoietic system, and possibly even the central nervous system. It is traditionally diagnosed as acute GVHD (aGVHD) and as chronic GVHD, although these 2 disease entities can sometimes overlap.30,31 aGVHD includes intense skin, gastrointestinal and liver damage, whereas chronic GVHD associates to more progressive damage with ulcer-type insult to mucosas and widespread injury to other organs such as lungs.32 The genetic similarity between the donor and the host, measured as the degree of HLA disparity, is linked to GVHD risk,33 although the extent of this may depend on specific loci mismatch and graft source.34 Also, due to standard donor HLA matching, donor minor antigens may play a more determinant role in the current clinical setting.35 Additionally, a GVHD-like syndrome can also be seen in autologous HCT settings though the exact etiological cause for this is still in debate.36 Ferrara and colleagues have summarized GVHD disease progression into 3 phases.32 The first phase consists of injury to the recipient tissues caused by the aggressive conditioning regimens necessary to prevent malignant relapse and graft rejection. Although all organs are affected to varied extents during initial treatment, the hematopoietic and gastrointestinal systems are especially susceptible to conditioning. Total-body irradiation is a common component of the conditioning regimen and is known to induce dose-dependent damage to the gut.37 In mouse models, ionizing radiation causes direct damage to epithelial and endothelial cells and increased p53-mediated epithelial apoptosis.38 Irradiation also induces enteric endothelial damage via induction of plasminogen activator inhibitor-type 1 (PAI-1) production which aggravates enteritis with crypt damage, intestinal inflammation, and fibrosis.39,40 Moreover, these toxic effects may partially be mediated by the gut microbiota: germ-free mice show reduced intestinal damage after radiation41 and broad-spectrum antibiotic treatment or deficiency in the myeloid differentiation primary response gene 88 (MyD88), a crucial adaptor for recognition of microbial molecules, prevents crypt loss and reduces damage to progenitor and stem cells in mouse colon after high doses of radiation.42 The gut microbiota has, however, been shown to protect against dextran sodium sulfate–induced intestinal damage and to reduce damage after lower doses of radiation43; it is possible that the microbiota play different roles in the initiation of damage and the subsequent healing process. Paneth cells and Lgr5+ intestinal stem cells are also targeted after irradiation.44 This is especially important due to the role of Paneth cells in secreting defensins and other microbial products to regulate the microbiota and stem cells in maintaining epithelial enterocyte turnover. Standard chemotherapeutic drugs as cyclophosphamide, busulfan, 5-fluorouracil, and irinotecan (CPT-11) also induce mucositis and chemotherapy-induced diarrhea. Appropriately, organ damage and chemotherapy-induced intestinal inflammation cause an increase in the systemically circulating levels of inflammatory cytokines such as tumor necrosis factor-α, IFN-γ, and IL-6.45
The second phase of GVHD development involves lymphocyte activation by host and later donor antigen-presenting cells, where activated CD4+ T cells acquire T helper effector functions and secrete cytokines, which further accelerates immune activation. GVHD was classically identified as a Th1-type disease due to the high expression of cytokines such as IFN-γ, though subsequent studies also established a role for Th2 and Th17 cells, which produce IL-4 and IL-17, and determine immune targeting for different organs.46,47 As such, a third stage links immune activation to effector cytotoxic functions of mediators as T CD8+ cells, which go on to induce direct damage to target cells and establish typical GVHD injury to organs as the liver, skin, and gut (Figure 1).35,48,49
Figure 1.
Intestinal damage and dysbiosis linked to GVHD. Upon HCT, multiple factors such as conditioning toxicity, antibiotic treatment, and immune activation mediate GVHD. GVHD progression associates with injury to stem cell compartments along with Paneth cells (small intestine) and goblet cells. This leads to increased intestinal permeability, inflammation, and reduction of the mucus layer and antimicrobial products (eg, defensins). Antibiotic treatment and a limited amount of nutrients in the gut also promote gut dysbiosis, furthering gastrointestinal damage and disease. DC, dendritic cell; IgA, immunoglobulin A.
Conditioning regimens and initial dysbiosis
Few studies have addressed the impact of chemotherapy and conditioning regimens on the composition of the intestinal microbiota and shown a loss of Clostridium cluster XIVa and Bifidobacteria and increased Enterococcus during high-dose chemotherapy treatment.50-52 One study with focus on HCT patients found increased levels of Proteobacteria including Escherichia species and reduced levels of Firmicutes including Blautia species after chemotherapy.52 However, the causal relationship between the chemotherapy and microbiota is hard to delineate because many of these patients received prophylactic antibiotics concurrently with the chemotherapy.
The intestinal microbiota and GVHD
A normal and healthy intestinal microbiota is diverse and dominated by obligate anaerobic bacteria.16,19 From the time of admission to discharge from the hospital, many HCT patients lose this diversity, and the changes that occur are influenced by antibiotic treatment as well as development of GVHD.53-55 An impact of the microbiota on GVHD development was first proposed in the 1970s when it was demonstrated that mice kept under germ-free conditions developed less gut GVHD.56,57 These results were followed up by clinical studies that initially showed promising results for intestinal decontamination in HCT patients,58,59 though later studies have not reproduced these benefits,60-62 possibly due to varying success of the decontaminating regimens. In addition, a large prospective study targeting anaerobic bacteria in HCT patients showed decreased GVHD development, indicating that selective decontamination may have beneficial effects.63
The loss of intestinal diversity seen in HCT patients is generally associated with loss of Clostridia species known to produce SCFAs from dietary fiber.64 Butyrate is preferred as an energy source of intestinal epithelial cells and a recent article suggests that reduced amounts of butyrate are found in intestinal epithelial cells after allo-HCT in mice, while the addition of these SCFAs reduced gut damage and GVHD.65 Interestingly, this effect was also mimicked by the administration of a mix of bacteria, known to be high producers of butyrate, belonging to the Clostridia class. Although this is so far the only experimental evidence of efficacy of SCFAs in a GVHD model, a clinical study from our group also showed that high intestinal abundance of Blautia, a genus belonging to the Clostridia class, correlates with reduced risk of death from GVHD.66 Antibiotic treatment is probably the main factor that influences the microbiota changes observed during the course of transplant and we recently showed that the choice of antibiotic regimen used to treat neutropenic fever influences the incidence of GVHD. Administration of the broad-spectrum antibiotics imipenem-cilastatin and piperacillin-tazobactam was associated with increased GVHD-related mortality at 5 years.67 This retrospective study did not find an association between metronidazole and reduced GVHD as reported by Beelen et al,63 which could be due to multiple factors including the use of different antibiotic combinations in the retrospective study, as well as geographic and cultural differences in the intestinal flora. Fecal microbiota transplant (FMT) can be used to restore a disrupted intestinal flora and a recent pilot study with the aim to evaluate the safety of FMT in HCT patients with steroid-refractory or steroid-dependent GVHD showed promising results; the treatment was well received and 3 of 4 patients showed resolution of signs and symptoms of GVHD 28 days after FMT.68 However, this very small study did not address long-term efficacy and larger studies with the capacity to control for confounding factors will be necessary to determine the efficacy of FMT as a novel treatment of GVHD. Together, these results indicate that the microbiota present in the gut may not only mirror GVHD host pathology but that the reverse may also be true, where the gut flora may act as an active participant in the recovery or even prevention of GVHD.
A parallel can also be drawn to other intestinal inflammatory diseases such as inflammatory bowel disease (IBD) which has been associated with reduced gut flora diversity.69,70 As in HCT, IBD has been linked to a reduction in Firmicutes71 and a recent study involving 235 IBD patients found a negative correlation of Blautia abundance and IBD and IBD flare.72 These similarities suggest that there may be an underlying link between the intestinal microbiota and intestinal disease that is immune-mediated.
The intestinal injury caused by the conditioning regimen results in increased intestinal permeability that permits translocation of gut bacteria over the intestinal barrier, a condition described as leaky bowel syndrome. As a consequence, immune stimulation from a range of pathogen and danger-associated molecular patterns such as lipopolysaccharide and peptidoglycan reinforce the inflammatory cytokine response and provide the ideal setting for T-cell alloactivation and priming. In line with this, the Toll-like receptors for these molecules have been linked to GVHD development. Clinical and experimental evidence suggests that TLR4 mutations (receptor for lipopolysaccharide) provide a lower risk for acute GVHD, although this receptor may also be important to prevent bacteremia,73,74 providing protection against disease. In addition, host mutations in the intracellular peptidoglycan receptor nucleotide-binding oligomerization domain-containing 2 (NOD2/CARD15) have been associated with acute GVHD and worse survival after HCT.75 Interestingly, this association was reduced in patients with gram-positive decontamination suggesting the involvement of specific gut bacteria in GVHD.75 This was also corroborated by our group in an experimental allo-HCT model where NOD2-deficient mice showed increased GVHD and intestinal inflammation.76
Diversity loss, GVHD, and posttransplant infections
The magnitude of intestinal diversity loss is a risk factor for treatment-related mortality (TRM) including death from GVHD, infections, and organ failure after HCT.77 In line with this, Weber et al showed that the level of urine 3-indoxyl sulfate in the urine, a metabolite derived from indole produced by commensal bacteria, can be used as a measure for the presence of an intact intestinal microbiota and that lower levels of 3-indoxyl sulfate correlated with higher TRM rates and lower overall survival in HCT patients.78 A longitudinal study of pediatric patients undergoing HCT showed that it often takes over 2 months after transplantation to regain intestinal diversity.79 Lost diversity and intestinal domination of opportunistic bacteria have also been shown to precede blood stream infection (BSI) by the same pathogen in HCT patients.55,80 As such, some investigators are exploring alternative antibiotic prophylactic strategies that may better preserve patient flora and maintain gut homeostasis. Rifaximin is a broad-spectrum antibiotic with negligible intestinal absorption81 used clinically to treat irritable bowel syndrome and traveler’s diarrhea.82 The use of rifaximin in HCT patients has been found to preserve intestinal diversity and associates with lower TRM rates compared with patients treated with a combination of ciprofloxacin and metronidazole in a retrospective study.83
Despite standard prophylaxis with antibiotics after HCT, many days of neutropenia leaves patients vulnerable to bacterial infections including BSI.84 The incidence of BSI increases with acute GVHD,85 and treatment of acute GVHD with corticosteroids such as methylprednisolone further suppresses the immune system and is associated with a higher risk of infection after transplant.84 Corticosteroid-refractory GVHD patients also present high risk for mortality and infection-related death without an existing consensus on the effectiveness of second-line treatment.86,87 Studies also show that early BSI may increase the risk of subsequent GVHD grade 2 or higher.88 Common sources of opportunistic infectious bacteria are the oral cavity and the gut, hence the importance of clarifying the role of the intestinal microbiota on posttransplant infection.
Nutrition and GVHD
Patient nutritional status is also of great concern after bone marrow transplantation. As described, conditioning regimens profoundly compromise gastrointestinal physiology. This frequently leads patients to require supplemental feeding, which is most often administered parenterally and consequently reduces the amount of nutrients that reach the gut epithelium. Therefore, it is likely that some of the changes in the microbiota observed during HCT and GVHD may be due to insufficient nutrients in the gut to maintain a balanced flora. Indeed, our study showing an association between Blautia abundance and GVHD also showed a correlation of longer parenteral feeding regimens in patients with low Blautia.66 In mouse models, imipenem-cilastatin treatment, which was linked to worse GVHD and increased gut damage, was also associated with an increase in intestinal Akkermansia, a mucolytic bacteria belonging to the Verrucomicrobia phylum.67 Interestingly, increased abundance of Akkermansia is also associated with restricted calorie intake and bariatric surgery.89,90 This suggests that enteral feeding, as opposed to parenteral feeding, may exert a beneficial effect on intestinal flora after HCT and perhaps accelerate patient recovery. In line with this, enteral nutrition has been associated with reduced aGVHD and lower infection-related mortality, in addition to shorter neutrophil engraftment time.91
The gut microbiota have been found to play an important role in a wide range of diseases including patients with hematological malignancies undergoing HCT, and increasing attention has been given to the potential of probiotics and prebiotics to prevent and treat intestinal dysbiosis. Probiotics are nutritional supplements containing a defined amount of viable microorganisms that upon administration confer a benefit to the host,92 whereas prebiotics consist of nondigestible food ingredients (eg, undigested fibers) that support the growth of beneficial bacteria. Only a few studies have addressed the feasibility of prebiotic or probiotic treatment in HCT, and there will be major safety issues involved in administering viable bacteria to severely immune-compromised HCT patients.93 Two experimental studies in mice have shown that oral administration of Lactobacillus results in improved survival and reduced aGVHD and intestinal inflammation following HCT.53,94 Assessment of the safety of Lactobacillus administration to human HCT patients has shown promising results95 but the impact on GVHD development has yet to be reported. Although the use of these treatments seem promising, future studies should clarify both the safety and efficiency for these therapies in the clinical setting. Another important aspect to the efficacy of probiotic treatment is the capacity of ingested microorganisms to survive the low pH of the stomach and bile acids released in the small intestine. Many strains of Lactobacillus, including the ones found in common diary products, are sensitive to low pH and could not be reisolated in fecal samples after administration to humans,96 making the causative effect of probiotic treatment more challenging to interpret.
Concluding remarks
Many studies support the hypothesis that the composition of the intestinal microbiota is associated with differences in GVHD development and other outcomes after HCT (Table 1). Challenges that remain to be addressed include the relationship of cause and effect between specific microbiota features and clinical outcomes. Future research regarding antibiotics and GVHD should include prospective clinical studies that better control for confounding factors in determining causality. Another major challenge is the effect of varying diets, lifestyles, ethnic, and environmental factors on the intestinal microbiota17,18 which is a critical issue in associative studies of microbiota features and clinical outcomes because most studies so far were performed at a single center. Additional studies from other centers and ideally multicenter studies will be key to validate the associations between features of the microbiota and GVHD. Moreover, better assessment of microbial function through whole-genome sequencing will likely increase mechanistic insight and add more granularity to microbial analyses as has been the case in IBD patients where studies have shown that 12% of metabolic pathways were altered compared with 2% of species genera when IBD patients were compared with healthy controls.97 Therefore, metagenomic data from such experiments could give rise to more accurate mechanistic hypotheses.
Table 1.
Intestinal bacteria associations in allo-HCT
| Bacteria | Effect on GVHD/HCT | Comment |
|---|---|---|
| Clostridium cluster XIVa/Blautia | Positive | Reduced during chemotherapy treatment50,51 |
| Low relative abundance associated with increased GVHD mortality in HCT patients66 | ||
| Low abundance associated with IBD72 | ||
| Ameliorates experimental GVHD in mice65 | ||
| Lactobacillus | Positive | Improves survival and reduces experimental aGVHD in mice53,94 |
| Reported as a safe supplement to HCT patients95 | ||
| Enterococcus | Negative | Increased during chemotherapy treatment50-52 |
| Intestinal dominance associated with blood stream infection after HCT55,80 |
Acknowledgments
The authors sincerely thank Robert Jenq for constructive comments on the manuscript and Kori Anahi Porosnicu Rodriguez for assistance with literature review.
This work was supported by National Institutes of Health (NIH), National Cancer Institute award numbers P01-CA023766 (M.R.M.v.d.B.) and Project 4 of P01-CA023766 (M.R.M.v.d.B.); NIH, National Heart, Lung, and Blood Institute award numbers R01-HL069929 (M.R.M.v.d.B.) and 1R01HL123340-01A1 (M.R.M.v.d.B.); NIH, National Institute of Allergy and Infectious Diseases award numbers R01-AI101406 (M.R.M.v.d.B.) and R01–AI100288 (M.R.M.v.d.B.). Support was also received from The Lymphoma Foundation, The Susan and Peter Solomon Divisional Genomics Program, The Memorial Sloan Kettering Cancer Center Cycle for Survival, and P30 CA008748 Memorial Sloan Kettering Cancer Center Support Grant/Core Grant, the European Union's Seventh Programme for research, technological development and demonstration under grant agreement No [602587], the Swedish Research Council 2016-00149 (A.S.), the Swedish Society for Medical Research P14-0090 (A.S.), the Swedish Society of Medicine SLS-499181 (A.S.), and São Paulo Research Foundation 2015/14370-0 (M.B.d.S.).
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Authorship
Contribution: A.S. and M.B.d.S. performed literature review and prepared the manuscript and figure; and M.R.M.v.d.B. prepared the manuscript.
Conflict-of-interest disclosure: The Memorial Sloan Kettering Cancer Center has filed patent applications related to this work (PCT/US2015/062734 entitled “Intestinal microbiota and GVHD”; inventors include M.R.M.v.d.B.). M.R.M.v.d.B. received research support from Seres Therapeutics. The remaining authors declare no competing financial interests.
Correspondence: Marcel R. M. van den Brink, Medicine and Immunology, Memorial Sloan Kettering Cancer Center, 1275 York Ave, Box 111, New York, NY 10065; e-mail: vandenbm@mskcc.org.
References
- 1.Jagasia M, Arora M, Flowers ME, et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood. 2012;119(1):296-307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pasquini MC, Zhu X. Current uses and outcomes of hematopoietic stem cell transplantation: CIBMTR summary slides. Available at: http://www.cibmtr.org. Accessed 18 September 2016.
- 3.Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, Gordon JI. The human microbiome project. Nature. 2007;449(7164):804-810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Qin J, Li R, Raes J, et al. ; MetaHIT Consortium. A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464(7285):59-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ott SJ, Kühbacher T, Musfeldt M, et al. Fungi and inflammatory bowel diseases: alterations of composition and diversity. Scand J Gastroenterol. 2008;43(7):831-841. [DOI] [PubMed] [Google Scholar]
- 6.Reyes A, Haynes M, Hanson N, et al. Viruses in the faecal microbiota of monozygotic twins and their mothers. Nature. 2010;466(7304):334-338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bäckhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI. Host-bacterial mutualism in the human intestine. Science. 2005;307(5717):1915-1920. [DOI] [PubMed] [Google Scholar]
- 8.Kamada N, Chen GY, Inohara N, Núñez G. Control of pathogens and pathobionts by the gut microbiota. Nat Immunol. 2013;14(7):685-690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vollaard EJ, Clasener HA. Colonization resistance. Antimicrob Agents Chemother. 1994;38(3):409-414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027-1031. [DOI] [PubMed] [Google Scholar]
- 11.Karlsson FH, Tremaroli V, Nookaew I, et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013;498(7452):99-103. [DOI] [PubMed] [Google Scholar]
- 12.Qin J, Li Y, Cai Z, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55-60. [DOI] [PubMed] [Google Scholar]
- 13.Distrutti E, Monaldi L, Ricci P, Fiorucci S. Gut microbiota role in irritable bowel syndrome: new therapeutic strategies. World J Gastroenterol. 2016;22(7):2219-2241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Scher JU, Sczesnak A, Longman RS, et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. eLife. 2013;2:e01202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zoetendal EG, Akkermans AD, De Vos WM. Temperature gradient gel electrophoresis analysis of 16S rRNA from human fecal samples reveals stable and host-specific communities of active bacteria. Appl Environ Microbiol. 1998;64(10):3854-3859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Eckburg PB, Bik EM, Bernstein CN, et al. Diversity of the human intestinal microbial flora. Science. 2005;308(5728):1635-1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.David LA, Maurice CF, Carmody RN, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505(7484):559-563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Obregon-Tito AJ, Tito RY, Metcalf J, et al. Subsistence strategies in traditional societies distinguish gut microbiomes. Nat Commun. 2015;6:6505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486(7402):207-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu C, Sartor RB, Huang K, Tonkonogy SL. Transient activation of mucosal effector immune responses by resident intestinal bacteria in normal hosts is regulated by interleukin-10 signalling. Immunology. 2016;148(3):304-314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith K, McCoy KD, Macpherson AJ. Use of axenic animals in studying the adaptation of mammals to their commensal intestinal microbiota. Semin Immunol. 2007;19(2):59-69. [DOI] [PubMed] [Google Scholar]
- 22.El Aidy S, van Baarlen P, Derrien M, et al. Temporal and spatial interplay of microbiota and intestinal mucosa drive establishment of immune homeostasis in conventionalized mice. Mucosal Immunol. 2012;5(5):567-579. [DOI] [PubMed] [Google Scholar]
- 23.Round JL, Lee SM, Li J, et al. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science. 2011;332(6032):974-977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ivanov II, Atarashi K, Manel N, et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139(3):485-498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee YK, Menezes JS, Umesaki Y, Mazmanian SK. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc Natl Acad Sci USA. 2011;108(suppl 1):4615-4622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Teng F, Klinger CN, Felix KM, et al. Gut microbiota drive autoimmune arthritis by promoting differentiation and migration of Peyer’s patch T follicular helper cells. Immunity. 2016;44(4):875-888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Donohoe DR, Garge N, Zhang X, et al. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011;13(5):517-526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smith PM, Howitt MR, Panikov N, et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science. 2013;341(6145):569-573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.den Besten G, Bleeker A, Gerding A, et al. Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes. 2015;64(7):2398-2408. [DOI] [PubMed] [Google Scholar]
- 30.Martin PJ, Schoch G, Fisher L, et al. A retrospective analysis of therapy for acute graft-versus-host disease: initial treatment. Blood. 1990;76(8):1464-1472. [PubMed] [Google Scholar]
- 31.Sullivan KM, Agura E, Anasetti C, et al. Chronic graft-versus-host disease and other late complications of bone marrow transplantation. Semin Hematol. 1991;28(3):250-259. [PubMed] [Google Scholar]
- 32.Ferrara JL, Levine JE, Reddy P, Holler E. Graft-versus-host disease. Lancet. 2009;373(9674):1550-1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morishima Y, Sasazuki T, Inoko H, et al. The clinical significance of human leukocyte antigen (HLA) allele compatibility in patients receiving a marrow transplant from serologically HLA-A, HLA-B, and HLA-DR matched unrelated donors. Blood. 2002;99(11):4200-4206. [DOI] [PubMed] [Google Scholar]
- 34.Fernandez-Viña MA, Wang T, Lee SJ, et al. Identification of a permissible HLA mismatch in hematopoietic stem cell transplantation. Blood. 2014;123(8):1270-1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dickinson AM, Wang XN, Sviland L, et al. In situ dissection of the graft-versus-host activities of cytotoxic T cells specific for minor histocompatibility antigens. Nat Med. 2002;8(4):410-414. [DOI] [PubMed] [Google Scholar]
- 36.Otegbeye F, Gale RP, Lazarus HM. Autologous GVHD? Bone Marrow Transplant. 2014;49(11):1349-1351. [DOI] [PubMed] [Google Scholar]
- 37.Leibowitz BJ, Wei L, Zhang L, et al. Ionizing irradiation induces acute haematopoietic syndrome and gastrointestinal syndrome independently in mice. Nat Commun. 2014;5:3494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang X, Wei L, Cramer JM, et al. Pharmacologically blocking p53-dependent apoptosis protects intestinal stem cells and mice from radiation. Sci Rep. 2015;5:8566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rannou E, François A, Toullec A, et al. In vivo evidence for an endothelium-dependent mechanism in radiation-induced normal tissue injury. Sci Rep. 2015;5:15738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Abderrahmani R, François A, Buard V, et al. PAI-1-dependent endothelial cell death determines severity of radiation-induced intestinal injury. PLoS One. 2012;7(4):e35740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jervis HR, McLaughlin MM, Johnson MC. Effect of neutron-gamma radiation on the morphology of the mucosa of the small intestine of germfree and conventional mice. Radiat Res. 1971;45(3):613-628. [PubMed] [Google Scholar]
- 42.Lai XY, Egan LJ. Suppression of radiation-induced DNA double-strand break repair by MyD88 is accompanied by apoptosis and crypt loss in mouse colon. Oncogenesis. 2013;2:e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rakoff-Nahoum S, Paglino J, Eslami-Varzaneh F, Edberg S, Medzhitov R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell. 2004;118(2):229-241. [DOI] [PubMed] [Google Scholar]
- 44.Metcalfe C, Kljavin NM, Ybarra R, de Sauvage FJ. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell. 2014;14(2):149-159. [DOI] [PubMed] [Google Scholar]
- 45.Hechinger AK, Smith BA, Flynn R, et al. Therapeutic activity of multiple common γ-chain cytokine inhibition in acute and chronic GVHD. Blood. 2015;125(3):570-580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nikolic B, Lee S, Bronson RT, Grusby MJ, Sykes M. Th1 and Th2 mediate acute graft-versus-host disease, each with distinct end-organ targets. J Clin Invest. 2000;105(9):1289-1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Carlson MJ, West ML, Coghill JM, Panoskaltsis-Mortari A, Blazar BR, Serody JS. In vitro-differentiated TH17 cells mediate lethal acute graft-versus-host disease with severe cutaneous and pulmonary pathologic manifestations. Blood. 2009;113(6):1365-1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Serody JS, Burkett SE, Panoskaltsis-Mortari A, et al. T-lymphocyte production of macrophage inflammatory protein-1alpha is critical to the recruitment of CD8(+) T cells to the liver, lung, and spleen during graft-versus-host disease. Blood. 2000;96(9):2973-2980. [PubMed] [Google Scholar]
- 49.Kim YM, Sachs T, Asavaroengchai W, Bronson R, Sykes M. Graft-versus-host disease can be separated from graft-versus-lymphoma effects by control of lymphocyte trafficking with FTY720. J Clin Invest. 2003;111(5):659-669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van Vliet MJ, Tissing WJ, Dun CA, et al. Chemotherapy treatment in pediatric patients with acute myeloid leukemia receiving antimicrobial prophylaxis leads to a relative increase of colonization with potentially pathogenic bacteria in the gut. Clin Infect Dis. 2009;49(2):262-270. [DOI] [PubMed] [Google Scholar]
- 51.Zwielehner J, Lassl C, Hippe B, et al. Changes in human fecal microbiota due to chemotherapy analyzed by TaqMan-PCR, 454 sequencing and PCR-DGGE fingerprinting. PLoS One. 2011;6(12):e28654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Montassier E, Batard E, Massart S, et al. 16S rRNA gene pyrosequencing reveals shift in patient faecal microbiota during high-dose chemotherapy as conditioning regimen for bone marrow transplantation. Microb Ecol. 2014;67(3):690-699. [DOI] [PubMed] [Google Scholar]
- 53.Jenq RR, Ubeda C, Taur Y, et al. Regulation of intestinal inflammation by microbiota following allogeneic bone marrow transplantation. J Exp Med. 2012;209(5):903-911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Holler E, Butzhammer P, Schmid K, et al. Metagenomic analysis of the stool microbiome in patients receiving allogeneic stem cell transplantation: loss of diversity is associated with use of systemic antibiotics and more pronounced in gastrointestinal graft-versus-host disease. Biol Blood Marrow Transplant. 2014;20(5):640-645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ubeda C, Taur Y, Jenq RR, et al. Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J Clin Invest. 2010;120(12):4332-4341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.van Bekkum DW, Roodenburg J, Heidt PJ, van der Waaij D. Mitigation of secondary disease of allogeneic mouse radiation chimeras by modification of the intestinal microflora. J Natl Cancer Inst. 1974;52(2):401-404. [DOI] [PubMed] [Google Scholar]
- 57.Jones JM, Wilson R, Bealmear PM. Mortality and gross pathology of secondary disease in germfree mouse radiation chimeras. Radiat Res. 1971;45(3):577-588. [PubMed] [Google Scholar]
- 58.Storb R, Prentice RL, Buckner CD, et al. Graft-versus-host disease and survival in patients with aplastic anemia treated by marrow grafts from HLA-identical siblings. Beneficial effect of a protective environment. N Engl J Med. 1983;308(6):302-307. [DOI] [PubMed] [Google Scholar]
- 59.Vossen JM, Heidt PJ, van den Berg H, Gerritsen EJ, Hermans J, Dooren LJ. Prevention of infection and graft-versus-host disease by suppression of intestinal microflora in children treated with allogeneic bone marrow transplantation. Eur J Clin Microbiol Infect Dis. 1990;9(1):14-23. [DOI] [PubMed] [Google Scholar]
- 60.Passweg JR, Rowlings PA, Atkinson KA, et al. Influence of protective isolation on outcome of allogeneic bone marrow transplantation for leukemia. Bone Marrow Transplant. 1998;21(12):1231-1238. [DOI] [PubMed] [Google Scholar]
- 61.Petersen FB, Buckner CD, Clift RA, et al. Infectious complications in patients undergoing marrow transplantation: a prospective randomized study of the additional effect of decontamination and laminar air flow isolation among patients receiving prophylactic systemic antibiotics. Scand J Infect Dis. 1987;19(5):559-567. [DOI] [PubMed] [Google Scholar]
- 62.Russell JA, Chaudhry A, Booth K, et al. Early outcomes after allogeneic stem cell transplantation for leukemia and myelodysplasia without protective isolation: a 10-year experience. Biol Blood Marrow Transplant. 2000;6(2):109-114. [DOI] [PubMed] [Google Scholar]
- 63.Beelen DW, Elmaagacli A, Müller KD, Hirche H, Schaefer UW. Influence of intestinal bacterial decontamination using metronidazole and ciprofloxacin or ciprofloxacin alone on the development of acute graft-versus-host disease after marrow transplantation in patients with hematologic malignancies: final results and long-term follow-up of an open-label prospective randomized trial. Blood. 1999;93(10):3267-3275. [PubMed] [Google Scholar]
- 64.Ganapathy V, Thangaraju M, Prasad PD, Martin PM, Singh N. Transporters and receptors for short-chain fatty acids as the molecular link between colonic bacteria and the host. Curr Opin Pharmacol. 2013;13(6):869-874. [DOI] [PubMed] [Google Scholar]
- 65.Mathewson ND, Jenq R, Mathew AV, et al. Gut microbiome-derived metabolites modulate intestinal epithelial cell damage and mitigate graft-versus-host disease. Nat Immunol. 2016;17(5):505-513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jenq RR, Taur Y, Devlin SM, et al. Intestinal blautia is associated with reduced death from graft-versus-host disease. Biol Blood Marrow Transplant. 2015;21(8):1373-1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shono Y, Docampo MD, Peled JU, et al. Increased GVHD-related mortality with broad-spectrum antibiotic use after allogeneic hematopoietic stem cell transplantation in human patients and mice. Sci Transl Med. 2016;8(339):339ra71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kakihana K, Fujioka Y, Suda W, et al. Fecal microbiota transplantation for patients with steroid-resistant/dependent acute graft-versus-host disease of the gut [published online ahead of print 26 July 2016]. Blood. doi:10.1182/blood-2016-05-717652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ott SJ, Musfeldt M, Wenderoth DF, et al. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut. 2004;53(5):685-693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Willing BP, Dicksved J, Halfvarson J, et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology. 2010;139(6):1844-1854. [DOI] [PubMed] [Google Scholar]
- 71.Frank DN, St Amand AL, Feldman RA, Boedeker EC, Harpaz N, Pace NR. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci USA. 2007;104(34):13780-13785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sokol H, Leducq V, Aschard H, et al. Fungal microbiota dysbiosis in IBD [published online ahead of print 3 February 2016]. Gut. doi:10.1136/gutjnl-2015-310746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lorenz E, Schwartz DA, Martin PJ, et al. Association of TLR4 mutations and the risk for acute GVHD after HLA-matched-sibling hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2001;7(7):384-387. [DOI] [PubMed] [Google Scholar]
- 74.Zhao Y, Liu Q, Yang L, et al. TLR4 inactivation protects from graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Cell Mol Immunol. 2013;10(2):165-175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Holler E, Rogler G, Brenmoehl J, et al. Prognostic significance of NOD2/CARD15 variants in HLA-identical sibling hematopoietic stem cell transplantation: effect on long-term outcome is confirmed in 2 independent cohorts and may be modulated by the type of gastrointestinal decontamination. Blood. 2006;107(10):4189-4193. [DOI] [PubMed] [Google Scholar]
- 76.Penack O, Smith OM, Cunningham-Bussel A, et al. NOD2 regulates hematopoietic cell function during graft-versus-host disease. J Exp Med. 2009;206(10):2101-2110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Taur Y, Jenq RR, Perales MA, et al. The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood. 2014;124(7):1174-1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Weber D, Oefner PJ, Hiergeist A, et al. Low urinary indoxyl sulfate levels early after transplantation reflect a disrupted microbiome and are associated with poor outcome. Blood. 2015;126(14):1723-1728. [DOI] [PubMed] [Google Scholar]
- 79.Biagi E, Zama D, Nastasi C, et al. Gut microbiota trajectory in pediatric patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2015;50(7):992-998. [DOI] [PubMed] [Google Scholar]
- 80.Taur Y, Xavier JB, Lipuma L, et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin Infect Dis. 2012;55(7):905-914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Scarpignato C, Pelosini I. Rifaximin, a poorly absorbed antibiotic: pharmacology and clinical potential. Chemotherapy. 2005;51(suppl 1):36-66. [DOI] [PubMed] [Google Scholar]
- 82.Koo HL, DuPont HL. Rifaximin: a unique gastrointestinal-selective antibiotic for enteric diseases. Curr Opin Gastroenterol. 2010;26(1):17-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Weber D, Oefner PJ, Dettmer K, et al. Rifaximin preserves intestinal microbiota balance in patients undergoing allogeneic stem cell transplantation. Bone Marrow Transplant. 2016;51(8):1087-1092. [DOI] [PubMed] [Google Scholar]
- 84.Sayer HG, Longton G, Bowden R, Pepe M, Storb R. Increased risk of infection in marrow transplant patients receiving methylprednisolone for graft-versus-host disease prevention. Blood. 1994;84(4):1328-1332. [PubMed] [Google Scholar]
- 85.Hong J, Moon SM, Ahn HK, et al. Comparison of characteristics of bacterial bloodstream infection between adult patients with allogeneic and autologous hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2013;19(6):994-999. [DOI] [PubMed] [Google Scholar]
- 86.Martin PJ, Rizzo JD, Wingard JR, et al. First- and second-line systemic treatment of acute graft-versus-host disease: recommendations of the American Society of Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2012;18(8):1150-1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Deeg HJ. How I treat refractory acute GVHD. Blood. 2007;109(10):4119-4126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Poutsiaka DD, Price LL, Ucuzian A, Chan GW, Miller KB, Snydman DR. Blood stream infection after hematopoietic stem cell transplantation is associated with increased mortality. Bone Marrow Transplant. 2007;40(1):63-70. [DOI] [PubMed] [Google Scholar]
- 89.Dao MC, Everard A, Aron-Wisnewsky J, et al. ; MICRO-Obes Consortium. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. 2016;65(3):426-436. [DOI] [PubMed] [Google Scholar]
- 90.Liou AP, Paziuk M, Luevano JM Jr, Machineni S, Turnbaugh PJ, Kaplan LM. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5(178):178ra41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Seguy D, Duhamel A, Rejeb MB, et al. Better outcome of patients undergoing enteral tube feeding after myeloablative conditioning for allogeneic stem cell transplantation. Transplantation. 2012;94(3):287-294. [DOI] [PubMed] [Google Scholar]
- 92.de Vrese M, Schrezenmeir J. Probiotics, prebiotics, and synbiotics. Adv Biochem Eng Biotechnol. 2008;111:1-66. [DOI] [PubMed] [Google Scholar]
- 93.Stadlbauer V. Immunosuppression and probiotics: are they effective and safe? Benef Microbes. 2015;6(6):823-828. [DOI] [PubMed] [Google Scholar]
- 94.Gerbitz A, Schultz M, Wilke A, et al. Probiotic effects on experimental graft-versus-host disease: let them eat yogurt. Blood. 2004;103(11):4365-4367. [DOI] [PubMed] [Google Scholar]
- 95.Ladas EJ, Bhatia M, Chen L, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-266. [DOI] [PubMed] [Google Scholar]
- 96.Jacobsen CN, Rosenfeldt Nielsen V, Hayford AE, et al. Screening of probiotic activities of forty-seven strains of Lactobacillus spp. by in vitro techniques and evaluation of the colonization ability of five selected strains in humans. Appl Environ Microbiol. 1999;65(11):4949-4956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Morgan XC, Tickle TL, Sokol H, et al. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 2012;13(9):R79. [DOI] [PMC free article] [PubMed] [Google Scholar]