Abstract
We have developed an unnatural base pair (UBP) and a semi-synthetic organism (SSO) that imports the constituent unnatural nucleoside triphosphates and uses them to replicate DNA containing the UBP. However, propagation of the UBP is at least in part limited by the stability of the unnatural triphosphates, which are degraded by cellular and secreted phosphatases. To circumvent this problem, we now report the synthesis and evaluation of unnatural triphosphates with their β,γ-bridging oxygen replaced with a difluoromethylene moiety, yielding dNaMTPCF2 and dTPT3TPCF2. We find that while dNaMTPCF2 cannot support in vivo replication, likely due to poor polymerase recognition, dTPT3TPCF2 can, and moreover, its increased stability can contribute to increased UBP retention. The data demonstrate the promise of this chemical approach to SSO optimization, and suggest that other modifications should be sought that confer phosphatase resistance without interfering with polymerase recognition.
Graphical Abstract
Introduction
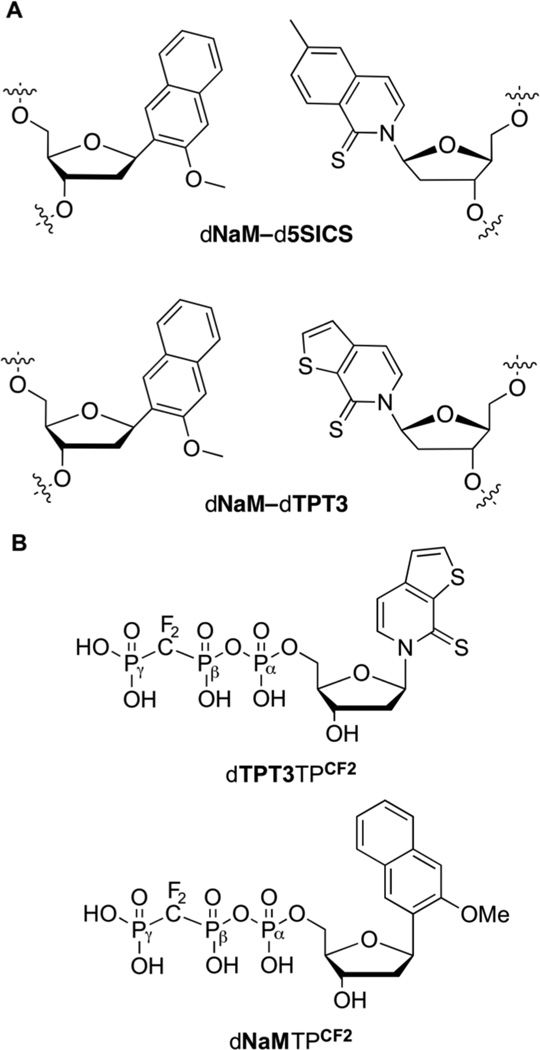
The natural genetic alphabet, conserved throughout nature, is composed of four nucleotide “letters” that pair by complementary hydrogen bonding to provide two base pairs, and these two pairs alone underlie the storage and retrieval of biological information. Significant effort has been directed toward the development of synthetic nucleotides that selectively pair and thus constitute an unnatural base pair (UBP), and three distinct design strategies have yielded UBPs that are well retained in DNA during PCR amplification.1–3 Hirao and Benner have elegantly used these in vitro expanded genetic alphabets to select for unnatural aptamers with improved properties relative to their fully natural counterparts.3–5 While these UBPs were developed by modification of the shape or hydrogen-bonding patterns of natural nucleobases, our approach instead relied on synthetic nucleotides with nucleobases that have little similarity to their natural counterparts and that interact via hydrophobic and packing forces;6 an approach that eventually culminated in the identification of a family of UBPs represented by dNaM-d5SICS7 and dNaM-dTPT38,9 (Fig. 1A). Despite having little to no nucleobase homology with their natural counterparts, these UBPs are efficiently replicated via a unique, mutually induced- fit mechanism.6,10,11
Figure 1.
Unnatural nucleotides. (A) dNaM-d5SICS and dNaM-dTPT3 UBPs. (B) dTPT3TPCF2 and dNaMTPCF2, with α, β, and γ phosphates labeled.
As opposed to in vitro applications, our efforts have focused on the use of the UBPs as the foundation of a semi-synthetic organism (SSO) that stably stores and retrieves increased information. We recently demonstrated that Escherichia coli grown in media supplemented with the unnatural triphosphates dNaMTP and d5SICSTP, and provided with the nucleoside triphosphate transporter PtNTT2 to import them, is able to replicate a plasmid containing the dNaM-d5SICS UBP.12 However, a significant limitation of the nascent SSO is UBP loss with extended or high density growth, which appears to result, at least in part, from the dephosphorylation of the unnatural triphosphates, reducing their concentration below that required for efficient replication.12 While recent efforts have demonstrated that the dNaM-dTPT3 UBP is retained in the SSO better than the originally employed dNaM-d5SICS UBP,13 dTPT3TP is likely also susceptible to the same phospholytic degradation. Efforts to inhibit degradation or identify and delete the genes that encode the phosphatases responsible have so far been unsuccessful (our unpublished results and Ref 12), but suggest that multiple phosphatases may be involved.
While a potential solution to the problem of unnatural triphosphate degradation would be to add them continuously during growth, this would be wasteful and would complicate the use of the SSO. In contrast, chemical stabilization of the triphosphates would be more practical. To explore this chemical approach, we synthesized analogs of the unnatural triphosphates with their β,γ-bridging oxygen atom replaced with a difluoromethylene moiety, yielding dNaMTPCF2 and dTPT3TPCF2 (Fig. 1B). The data reveal that while the β,γ-CF2 modification imparts stability to both triphosphates, it also impacts transporter and polymerase recognition in a nucleobase- dependent manner. The effects on polymerase recognition appear larger and preclude the use of dNaMTPCF2, but not dTPT3TPCF2, suggesting that it or other similarly stabilized unnatural triphosphates should be useful in the effort to expand the genetic alphabet of a living organism.
Experimental Section
General
For synthetic procedures, all reactions were carried out in oven-dried glassware under inert atmosphere, and all solvents were distilled and/or dried over 4 Å molecular sieves. All other chemical reagents were purchased from Aldrich, unless otherwise noted. 1H, 13C, spectra were recorded on Bruker DPX 400 mHz NMR equipped with a QNP probe. 31P and 19F NMR spectra were recorded on Bruker AVIII HD 600 mHz NMR equipped with a CPDCH cryoprobe. Mass spectroscopic data were obtained from the core facilities at The Scripps Research Institute.
All bacterial cultures were grown in liquid 2×YT media (casein peptone 16 g/L, yeast extract 10 g/L, NaCl 5 g/L) supplemented with potassium phosphate (50 mM, pH 7). All experiments involving bacterial growth were conducted in microwell plates (96-well or 48-well, as needed). When noted, antibiotics were used at the following concentrations: chloramphenicol, 5 µg/mL; ampicillin, 50 µg/mL. Cell growth, indicated as OD600, was measured using a Perkin Elmer EnVision 2103 Multilabel Reader with a 590/20 nm filter. Unless otherwise stated, molecular biology reagents were purchased from New England Biolabs (Ipswich, MA) and were used according to the manufacturer’s protocols. As necessary, purification of nucleic acids was accomplished by micro-elution columns (Zymo Research Corp; Irvine, CA). All natural oligonucleotides were purchased from IDT (San Diego, CA), and oligonucleotides containing dNaM or dTPT3 were synthesized by Biosearch Technologies (Petaluma, CA) with purification by reverse phase cartridge and kindly provided by Synthorx (La Jolla, CA).
Degradation of nucleotide triphosphates
An overnight culture (~3 mL) of E. coli BL21(DE3) lacZYA::CmR 13 was diluted 100-fold into fresh media and incubated at 37 °C for 2 h. At this time, cells were diluted back to an OD600 of 0.10 in 500 µL media containing 200 mM nucleotide triphosphate. Cultures were incubated with the triphosphates at 37 °C with shaking and at designated times samples were collected by removing an aliquot (50 µL) and immediately pelleting the cell fraction (9,000 r.c.f. for 5 min, 4 °C). An aliquot (40 µL) of the supernatant (i.e. media fraction) was removed and mixed with acetonitrile (80 µL) and then incubated at room temperature (~23 °C) for 30 min before pelleting (12,000 r.c.f. for 10 min, RT) to remove precipitate. The supernatant was collected, dried (SpeedVac), resuspended in 20 µL 0.1 M triethylammonium bicarbonate (TEAB) pH 7.5, and then analyzed by HPLC (Agilent 1100 series) using a Phenomenex Jupiter LC column (3 mm C18 300 Å, 250 × 4.6 mm) as described previously.12 Percent remaining triphosphate was determined by integrating the area under the curve (AUC) for each species present (i.e. triphosphate, diphosphate, monophosphate), and dividing the AUC of the triphosphate peak by the sum of the three peaks.
Kinetic assay
Steady-state kinetic assays were performed as described previously14 with oligonucleotide primers and templates described in Table S1. The reaction products were resolved by denaturing PAGE, and the resulting gel was imaged on a Typhoon 9210 flatbed laser scanner (GE Healthcare). ImageStudioLite software was used to quantify band density of imaged gels. Kinetic parameters (KM and Vmax) were determined by plotting the observed rate of incorporation against triphosphate concentration and fitting the data to the Michaelis-Menten equation. The reported values are the average and standard deviation of at least three independent determinations.
Inhibition-based uptake assay
An overnight culture (~3 mL) of the E. coli SSO YZ313 was diluted 100-fold in media supplemented with chloramphenicol (~8 mL) and incubated at 37 °C to an OD600 ~ 0.4–0.6 (~1.5 h). To an aliquot of cells (92.5 µL) was added a solution of an unnatural triphosphate (500 µM), ATP (50 µM), and [α-32P]-ATP (4 µCi/mL, Perkin Elmer), to a final volume of 100 µL. Each sample was run in parallel with an aliquot of cells receiving no unnatural triphosphate as an ATP-only control. Treated cells were incubated at 37 °C for 10 min, collected through a 96-well 0.65 mm glass fiber filter plate (MultiScreen, EMD Millipore) under vacuum, and then washed with ddH2O (2 × 100 µL). Filters were then exposed overnight to a storage phosphor screen, which was then imaged as described above. Inhibition of ATP uptake was determined by densitometric analysis of the resulting image, normalizing radioactive density to the ATP-only control and expressing the value as percent inhibition of ATP uptake. Reported values are the average and standard deviation of at least three independent determinations.
Analysis of in vivo replication. Plasmids containing the UBP were prepared and used to transform the SSO as described previously. 12,13 After recovery, cells were diluted into media containing different combination and concentrations of dNaMTP and dTPT3 or their β,γ-CF2 modified counterparts. Plasmids were recovered at defined times and analyzed for retention of the UBP as described previously12,13 and in Supporting Information.
Results and Discussion
To explore the chemical stabilization of the unnatural triphosphates, we synthesized dNaMTPCF2 and dTPT3TPCF2. Although the β,γ-CF2 modification may impact polymerase binding, its electronegativity should favor nucleophilic attack at the a phosphate,15–18 while eliminating phospholytic cleavage between the β and γ phosphates, likely the major route of degradation. Briefly, the free nucleosides dNaM and dTPT3 were synthesized as reported previously,14,19 and then, using the method reported by McKenna et al., they were converted to dNaMTPCF2 and dTPT3TPCF2.20
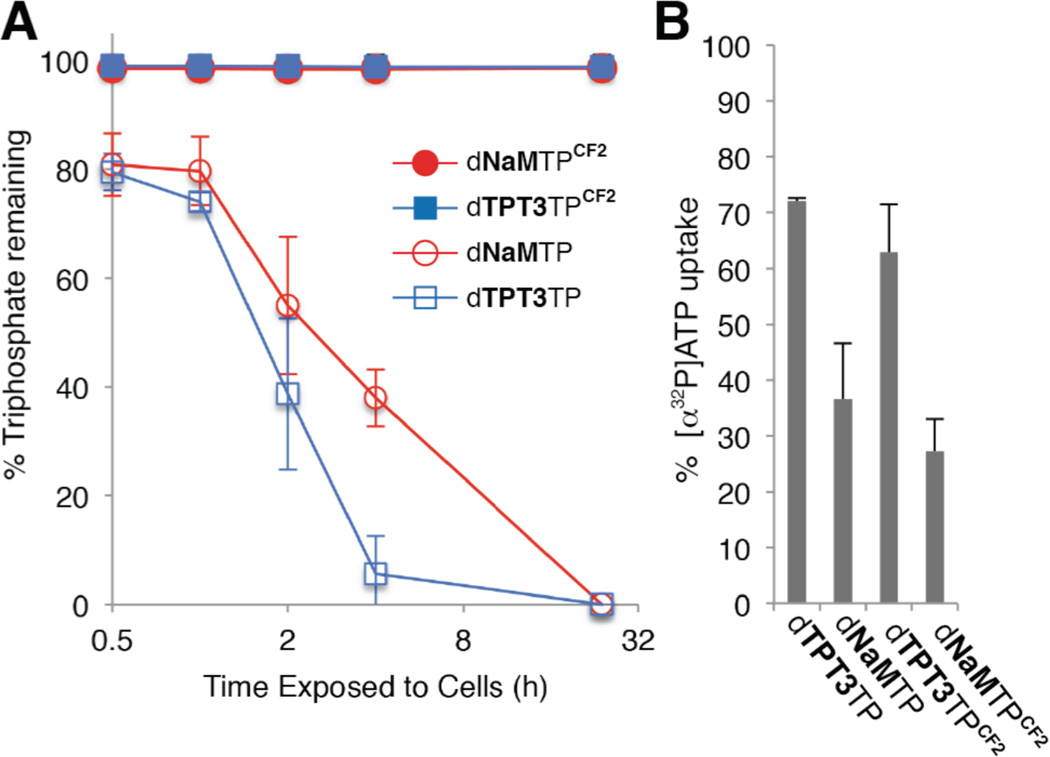
With these analogs in hand, we first tested their extracellular stability in cultures of actively growing bacteria. E. coli BL21(DE3) was grown to early-log phase (OD600 ~0.1), at which time 200 µM of a modified or unmodified unnatural triphosphate was added. Following incubation at 37 °C, cells were removed by centrifugation, soluble proteins in the recovered growth medium were precipitated, and the amount of intact triphosphate was determined by HPLC (Fig. 2A). Without modification, no triphosphate was detected after 24 h, consistent with rapid degradation, presumably by secreted phosphatases. However, no degradation of either dNaMTPCF2 or dTPT3TPCF2 was apparent, confirming that the β,γ-CF2 moiety significantly stabilizes the triphosphates under conditions employed to grow the SSO.
Figure 2.
Extracellular stability and transporter recognition of modified and unmodified unnatural triphosphates. (A) β,γ-CF2 modification stabilizes the unnatural triphosphates to degradation by secreted phosphatases. (B) The β,γ-CF2 modified unnatural triphosphates are still recognized by the SSO’s PtNTT2 transporter as detected by the inhibition of [α-32P]ATP uptake (see text and Supporting Information for details).
To determine whether β,γ-CF2 modification is compatible with polymerase-mediated replication, we next performed steady-state kinetic assays with the Klenow fragment of E. coli DNA polymerase I (Supporting Information), which served as a model polymerase during UBP development. Even under forcing conditions, we were unable to detect any insertion of dNaMTPCF2 opposite dTPT3. In contrast, we found that the polymerase does insert dTPT3TPCF2 opposite dNaM; however, while it does so with a reasonable KM of 15.7 ± 7.5 nM, the kcat of 1.4 ± 0.5 min−1 is somewhat less than that observed for dTPT3TP (our unpublished results). This data suggests that dNaMTPCF2 might not be compatible with replication within the SSO, and that while replication may occur with dTPT3TPCF2, the β,γ-CF2 moiety may lower its efficiency.
For use with the SSO, the stabilized unnatural triphosphates must first be recognized as a substrate by PtNTT2 and imported into the cell. To begin to examine recognition by PtNTT2, we characterized the ability of each modified and unmodified analog to decrease the import of [α-32P]ATP due to competitive inhibition, as previously described.21 The addition of 500 mM of either dNaMTPCF2 or dTPT3TPCF2 inhibited the uptake of [α-32P]ATP (Fig. 2B and Supporting Information). Although the level of inhibition was somewhat less than with dNaMTP and dTPT3TP, the data suggests that CF2 modification does not fully ablate recognition by the SSO’s transporter.
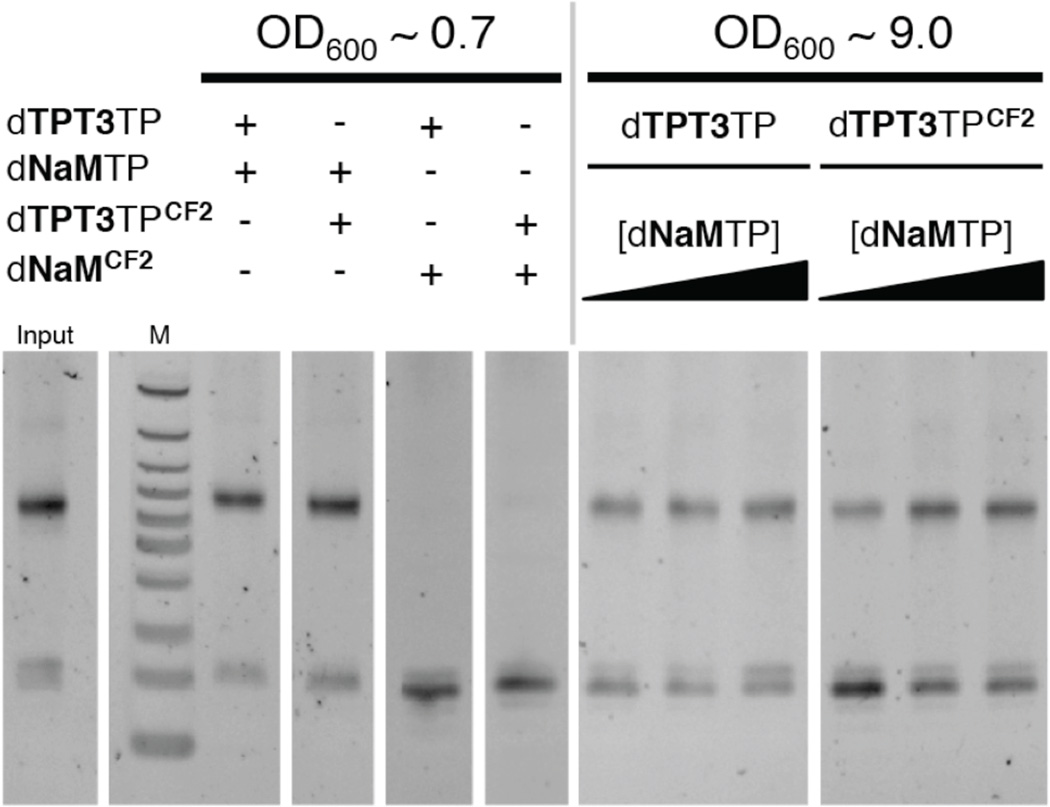
We next proceeded to examine whether dNaMTPCF2 and/or dTPT3TPCF2 could support replication of the dNaM-dTPT3 UBP within the SSO. A pUC19 plasmid containing a single dNaM-dTPT3 UBP embedded within the TK1 sequence,12 was constructed via Golden Gate assembly, and used to transform the SSO. Cells were grown to an OD600 of ~0.7 in media supplemented with 3.0 µM dTPT3TP and 250 µM dNaMTP, or with one of the unnatural triphosphates replaced with its CF2-modified analog at the same concentration, and plasmids were recovered and analyzed for UBP retention (Fig. 3 and Supporting Information). Under these relatively permissive conditions, as expected, providing dNaMTP and dTPT3TP in the media resulted in high UBP retention (99.1 ± 2.1%). While retention was undetectable when dNaMTPCF2 and dTPT3TP were provided, it was significant when dNaMTP and dTPT3TPCF2 were provided (86.0 ±11.4%). When combined with the data described above, this suggests that both of the modified unnatural triphosphates are imported, but dNaMTPCF2 is not recognized during replication, and while dTPT3TPCF2 is recognized, it is recognized with somewhat reduced fidelity than its unmodified counterpart.
Figure 3.
Streptavidin gel shifts characterizing extent of UBP retention in vivo. The recovered plasmids are PCR amplified with a dNaMTP analog that is biotin tagged and thus the percentage of the slower migrating (upper) band, relative to the sum of the slower and faster migrating bands represents the percent UBP retention. Note that the faster migrating (unshifted) band sometimes appears as a doublet, but this is observed with the input plasmid, as well, and thus is not related to in vivo replication. The percentage shifted is normalized to that measured for the input plasmid (shown on left). A molecular weight marker is included in the lane labeled “M.” See Supporting Information for details.
To explore whether the increased stability of dTPT3TPCF2 can result in increased intracellular concentrations that result in more efficient UBP replication, despite its inherently reduced fidelity, we examined replication of the dNaM-dTPT3 UBP within the SSO under less permissive conditions, growing cells to deep stationary phase (OD600 ~ 9) in the presence of 0.75 µM dTPT3TP or dTPT3TPCF2, and 250, 1000, or 2000 µM dNaMTP (Fig. 3). For dTPT3TP, the level of UBP retention remained relatively constant across the different dNaMTP concentrations, at 72 ± 9%, 83 ± 4%, and 81 ± 9%, respectively. However, with dTPT3TPCF2, UBP retentions were 51 ± 11%, 80 ± 9%, and 85 ± 14%, respectively, with increasing dNaMTP concentrations. It is unclear if the ~85% retention, which was also observed under the permissive conditions, is significant, perhaps representing the maximal fidelity possible with polymerase saturation. Regardless, it is clear that under these more challenging growth conditions, compensating for dNaMTP degradation by increasing its concentration can increase UBP retention, but this obviously requires that sufficient levels of its cognate triphosphate also be maintained, which appears to be accomplished by CF2-modification.
The goal of synthetic biology is to impart living organisms with new and unnatural traits and even to create artificial life,22 and at its heart is the power to draw upon both synthetic chemistry and more biology-focused approaches. While early SSO development in our laboratory focused on the chemical optimization of the unnatural nucleotides, more recent efforts have focused on the optimization of the cell itself for import and retention of the UBP. The data reveal that, as expected, the β,γ-CF2 modification of either unnatural triphosphate imparts resistance to degradation, but that it also impacts transporter and polymerase recognition in a manner that depends on the nucleobase. The reduced polymerase recognition of dNaMTPCF2 precludes its use in the SSO, but the stability and at least reasonable uptake and polymerase recognition of dTPT3TPCF2 demonstrate the promise of this chemical approach to SSO optimization. Further modifications should be sought with the goal of stabilizing both unnatural triphosphates and in a manner that does not interfere with uptake or replication.
Supplementary Material
Acknowledgments
Funding Sources
This work was supported by the National Institutes of Health (Grant Nos. GM060005 and GM118178 to F.E.R.) and National Science Foundation Graduate Research Fellowships (Grant No. NSF/DGE-1346837 to A.W.F.)
Footnotes
ASSOCIATED CONTENT
Supporting Information
The Supporting Information is available free of charge on the ACS Publications website at DOI: xx.xxxx/xx.
Supporting methods, synthesis and characterization of dTPT3TPCF2 and dNaMTPCF2 (PDF)
No competing financial interests have been declared..
REFERENCES
- 1.Malyshev DA, Dhami K, Quach HT, Lavergne T, Ordoukhanian P, Torkamani A, Romesberg FE. Proc. Natl. Acad. Sci. USA. 2012;109:12005–12010. doi: 10.1073/pnas.1205176109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kimoto M, Hirao I. In: Chemical Biology of Nucleic Acids: Fundamentals and Clinical Applications. Erdmann AV, Markiewicz TW, Barciszewski J, editors. Berlin, Heidelberg: Springer Berlin Heidelberg; 2014. pp. 131–148. [Google Scholar]
- 3.Zhang L, Yang Z, Sefah K, Bradley KM, Hoshika S, Kim MJ, Kim HJ, Zhu G, Jimenez E, Cansiz S, Teng IT, Champanhac C, McLendon C, Liu C, Zhang W, Gerloff DL, Huang Z, Tan W, Benner SA. J. Am. Chem. Soc. 2015;137:6734–6737. doi: 10.1021/jacs.5b02251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kimoto M, Yamashige R, Matsunaga K, Yokoyama S, Hirao I. Nat. Biotechnol. 2013;31:453–457. doi: 10.1038/nbt.2556. [DOI] [PubMed] [Google Scholar]
- 5.Sefah K, Yang Z, Bradley KM, Hoshika S, Jimenez E, Zhang L, Zhu G, Shanker S, Yu F, Turek D, Tan W, Benner SA. Proc. Natl. Acad. Sci. USA. 2014;111:1449–1454. doi: 10.1073/pnas.1311778111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Malyshev DA, Romesberg FE. Angew. Chem. Int. Ed. 2015;54:11930–11944. doi: 10.1002/anie.201502890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Leconte AM, Hwang GT, Matsuda S, Capek P, Hari Y, Romesberg FE. J. Am. Chem. Soc. 2008;130:2336–2343. doi: 10.1021/ja078223d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dhami K, Malyshev DA, Ordoukhanian P, Kubelka T, Hocek M, Romesberg FE. Nucleic Acids Res. 2014;42:10235–10244. doi: 10.1093/nar/gku715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lavergne T, Malyshev DA, Romesberg FE. Chem. Eur. J. 2012;18:1231–1239. doi: 10.1002/chem.201102066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Betz K, Malyshev D, Lavergne T, Welte W, Diederichs K, Romesberg FE, Marx A. J. Am. Chem. Soc. 2013;135:18637–18643. doi: 10.1021/ja409609j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Betz K, Malyshev DA, Lavergne T, Welte W, Diederichs K, Dwyer TJ, Ordoukhanian P, Romesberg FE, Marx A. Nat. Chem. Biol. 2012;8:612–614. doi: 10.1038/nchembio.966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Malyshev DA, Dhami K, Lavergne T, Chen T, Dai N, Foster JM, Correa IR, Jr, Romesberg FE. Nature. 2014;509:385– 388. doi: 10.1038/nature13314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang Y, Lamb B, Feldman AW, Zhou AX, Lavergne T, Li L, Romesberg FE. Proc. Natl. Acad. Sci. USA. 2017 doi: 10.1073/pnas.1616443114. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Seo YJ, Hwang GT, Ordoukhanian P, Romesberg FE. J. Am. Chem. Soc. 2009;131:3246–3252. doi: 10.1021/ja807853m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sucato CA, Upton TG, Kashemirov BA, Batra VK, Martinek V, Xiang Y, Beard WA, Pedersen LC, Wilson SH, McKenna CE, Florian J, Warshel A, Goodman MF. Biochemistry. 2007;46:461–471. doi: 10.1021/bi061517b. [DOI] [PubMed] [Google Scholar]
- 16.Sucato CA, Upton TG, Kashemirov BA, Osuna J, Oertell K, Beard WA, Wilson SH, Florian J, Warshel A, McKenna CE, Goodman MF. Biochemistry. 2008;47:870–879. doi: 10.1021/bi7014162. [DOI] [PubMed] [Google Scholar]
- 17.Batra VK, Pedersen LC, Beard WA, Wilson SH, Kashemirov BA, Upton TG, Goodman MF, McKenna CE. J. Am. Chem. Soc. 2010;132:7617–7625. doi: 10.1021/ja909370k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Oertell K, Chamberlain BT, Wu Y, Ferri E, Kashemirov BA, Beard WA, Wilson SH, McKenna CE, Goodman MF. Biochemistry. 2014;53:1842–1848. doi: 10.1021/bi500101z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li L, Degardin M, Lavergne T, Malyshev DA, Dhami K, Ordoukhanian P, Romesberg FE. J. Am. Chem. Soc. 2014;136:826–829. doi: 10.1021/ja408814g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McKenna CE, Kashemirov BA, Upton TG, Batra VK, Goodman MF, Pedersen LC, Beard WA, Wilson SH. J. Am. Chem. Soc. 2007;129:15412–15413. doi: 10.1021/ja072127v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ast M, Gruber A, Schmitz-Esser S, Neuhaus HE, Kroth PG, Horn M, Haferkamp I. Proc. Natl. Acad. Sci. USA. 2009;106:3621– 3626. doi: 10.1073/pnas.0808862106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Leduc S. The Mechanisms of Life. New York: Rebman Company; 1911. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.