Abstract
Purpose
To evaluate the effects of valproic acid (VPA), a histone deacetylase inhibitor (HDACI), on the expression of hypoxia-inducible factor-1 alpha (HIF-1α) and vascular endothelial growth factor (VEGF) in human retinal Müller cells under hypoxic conditions.
Methods
Chemical hypoxia was induced in human retinal Müller cells (MIO-M1) by treatment with increasing concentrations of cobalt(II) chloride (CoCl2). Müller cells were also treated with a set concentration of CoCl2, along with various concentrations of VPA. The expression of HIF-1α and VEGF in the treated Müller cells was determined by enzyme-linked immunosorbent assay.
Results
Exposure of human retinal Müller cells to increasing concentrations of CoCl2 produced a dose-dependent increase in HIF-1α expression. The addition of increasing concentrations of VPA lead to a dose-dependent decrease in expression of HIF-1α and VEGF in Müller cells exposed to a set concentration of CoCl2.
Conclusions
HDACI VPA downregulated the expressions of HIF-1α and VEGF in human retinal Müller cells under hypoxic conditions. Using HDACI to target HIF-1α expression in Müller cells could be a new therapeutic strategy for the treatment of retinal vascular diseases.
Keywords: Histone deacetylase inhibitors, Hypoxia-inducible factor 1 alpha, Retinal Muller cells, Valproic acid, Vascular endothelial growth factor
Diseases such as age-related macular degeneration, diabetic retinopathy, and branch retinal vein occlusion are serious conditions that can decrease visual acuity and potentially lead to blindness [1,2]. An important factor in these diseases is neovascularization, which can be induced by hypoxia, ischemia, and inflammatory reactions. Vascular endothelial growth factor (VEGF), in particular, is thought to be a critical promoter of ophthalmic neovascularization [3,4,5]. VEGF is essential for neovascularization in the retina and choroid. During hypoxia, the expression of VEGF is increased to improve vascularization and vascular permeability [6,7]. Intravitreal injection of anti-VEGF has emerged as a promising treatment for neovascularization-associated ophthalmic disorders, with the drugs bevacizumab (Avastin) and ranibizumab (Lucentis) widely used in clinical settings.
In the retina, VEGF is expressed in the Müller cells, pigment epithelium, endothelium, astrocytes, and ganglion cells [8,9,10,11,12]. Müller cells are distributed throughout the retinal layer, perform a variety of functions, and serve as important mediators of neovascularization [13,14].
To adapt to hypoxic conditions, human cells and organs express a number of genes that affect neovascularization, metabolic processes, cell proliferation, and cell survival. The primary mediator that controls the expression of these genes is hypoxia-inducible factor-1 alpha (HIF-1α), which regulates the production of VEGF [15,16]. HIF-1α is composed of a β and an α subunit, the latter of which reacts to oxygen. In the presence of normal oxygen concentrations, HIF-1α is degraded; however, in a hypoxic state, HIF-1α is not degraded and functions to alter the expression of more than 100 genes. HIF-1α is also involved in the regulation of vascular tone, cell proliferation, apoptosis, and other metabolic pathways [17]. Therapeutic agents that target HIF-1α directly could provide widespread control of VEGF-induced neovascularization, an important factor in retinal hypoxic diseases, and potentially serve as alternative therapeutics to drugs targeting only individual processes in hypoxia-associated disease.
Therapeutics that target HIF-1α have been studied thoroughly in the field of cancer treatment and many agents that target HIF-1α directly have been developed [18,19,20,21,22,23]. Among these agents are histone deacetylase inhibitors (HDACIs), which inhibit the histone deacetylase enzyme. This allows the induction of gene transcription by hyperacetylation and reduces the activity of HIF-1α [24]. One of the HDAC inhibitors is valproic acid (VPA), which has been widely used for the treatment of epilepsy [25]. Several studies have shown that VPA also inhibits tumor angiogenesis through suppression of angiogenic factors such as VEGF [26,27]. While HDACIs have been widely used in cancer treatment, these agents have yet to be applied to the treatment of ophthalmic disease [28,29,30].
In this study, the effects of the HDACI, VPA, on the expression of HIF-1α and VEGF in human retinal Müller cells during hypoxia was evaluated.
Materials and Methods
Growth of cell lines
Human retinal Müller cells (MIO-M1) were grown in a humidified incubator at 37℃, 5% CO2, with DMEM + GlutaMAX-I (Gibco BRL, Grand Island, NY, USA) growth media containing 10% FBS (Gibco BRL), 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were provided with fresh media every 2 to 3 days.
Induction of hypoxia
Human retinal Müller cells were seeded at 5 × 103 cells per well in 96-well cell culture plates. For enzyme-linked immunosorbent assay, the cells were seeded at 1.0 × 105 cells per dish in 100 mm culture dishes. After 24 hours, the growth media was replaced with serum-free media and the cells were starved for 16 hours. Chemical hypoxia was induced by incubating cells for 24 hours in serum-free media containing various concentrations of cobalt(II) chloride (CoCl2).
Cell viability assay
To investigate the optimal concentration of CoCl2 for inducing chemical hypoxia, human retinal Müller cells were exposed to 0, 100, 200, 300, 400, 500, 600, 700, 800, 900, or 1,000 µM CoCl2. The cells were then washed twice with phosphate buffered saline and the cell counting kit-8 (CCK-8; Dojindo Molecular Technologies, Rockville, MD, USA) was used to measure cell viability. CCK-8 measures the amount of live cells by measuring formazan that is produced by dehydrogenase in cells. Ten microliters of CCK-8 and 90 µL of serum-free media were mixed in each well. After 1 hour, optical density was measured by a microplate reader at 450 nm wavelength.
VPA treatment
VPA 1 g/mL (Sigma-Aldrich, Silver Spring, MO, USA) was diluted serially in an aqueous solution of 0.1 N sodium hydroxide (NaOH) to 10 mg/mL and added to wells containing hypoxic Müller cells that were treated with 400 µM CoCl2. The wells were exposed to serum-free media for 22 hours with adequate concentrations of VPA (10, 25, 50, 75, 100 µg/mL). The control group (0 µg/mL) was treated with the same a mount of 0.1 N NaOH as the experimental groups.
Detection of VEGF and HIF-1α
VEGF and HIF-1α proteins were detected using the Quantikine enzyme-linked immunosorbent assay and Surveyor IC kits (R&D Systems, Minneapolis, MN, USA), respectively, according to the manufacturer's instructions.
Statistical analyses
The mean ± standard deviation values were reported for the results of three independent experiments and all statistics were calculated using the IBM SPSS Statistics ver. 19.0 (IBM Co., Armonk, NY, USA). The Wilcoxon signed-rank test was used to compare differences between the experimental and control groups, with a p-value <0.05 considered as statistically significant.
Results
Induction of chemical hypoxia and expression of HIF-1α in human retinal Müller cells
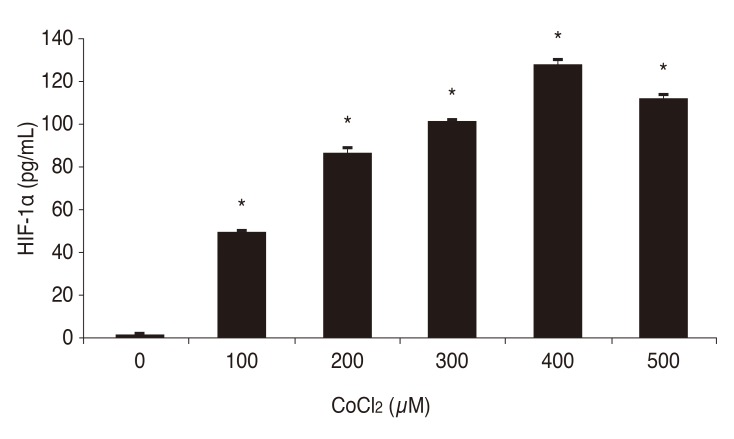
Chemical hypoxia was induced by treating human retinal Müller cells with 0, 100, 200, 300, 400, and 500 µM of CoCl2. A dose-dependent increase in the expression of HIF-1α was observed with increasing concentrations of CoCl2, with maximal HIF-1α expression at 400 µM CoCl2 (Fig. 1).
Fig. 1. Effects of cobalt(II) chloride (CoCl2) on the expression of hypoxia-inducible factor-1 alpha (HIF-1α) in human retinal Müller cells. *p-value < 0.05.
Viability of human retinal Müller cells during chemical hypoxia
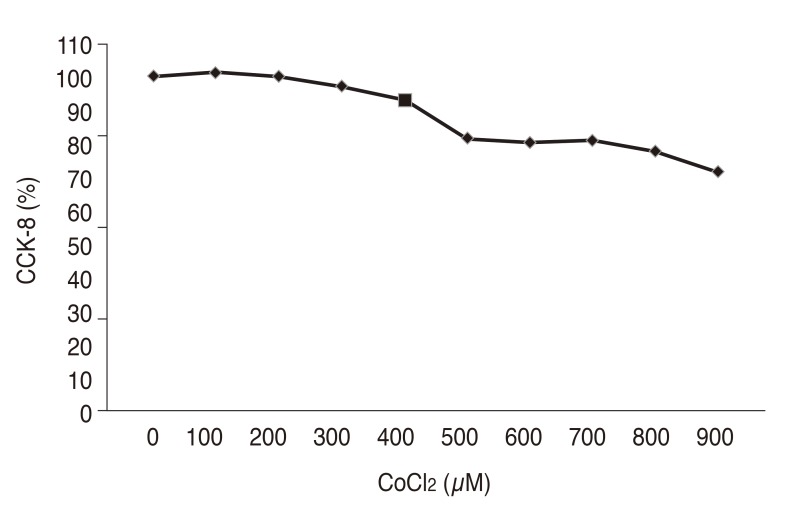
Müller cells were treated with 0, 100, 200, 300, 400, 500, 600, 700, 800, 900, and 1,000 µM CoCl2 and cell viability was assessed using the CCK-8 assay in order to determine the optimal concentration of CoCl2 to induce chemical hypoxia without reducing cell viability. Cell viability decreased gradually with increasing concentrations of CoCl2 (Fig. 2). Cell viability decreased even more at 500 µM, but the highest HIF-1α expression was observed with 400 µM of CoCl2. Therefore, the optimal concentration of CoCl2 to induce chemical hypoxia was selected to be 400 µM, based on the results for induction of HIF-1α expression as described above.
Fig. 2. Effects of cobalt(II) chloride (CoCl2) on the survival of human retinal Müller cells. CCK-8 = cell counting kit-8.
Effect of VPA treatment on the expression of HIF-1α and VEGF by human retinal Müller cells during hypoxia
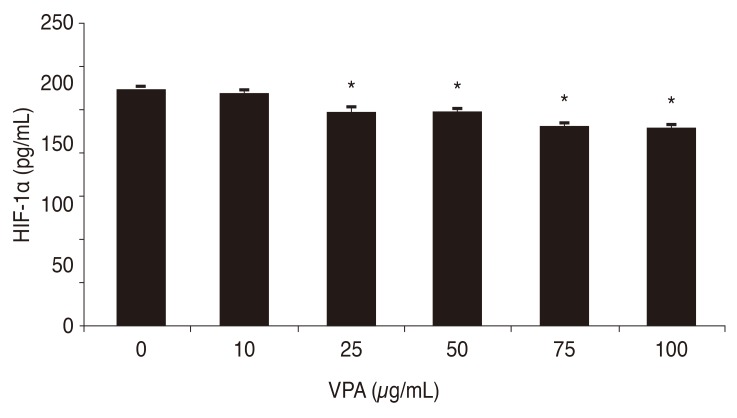
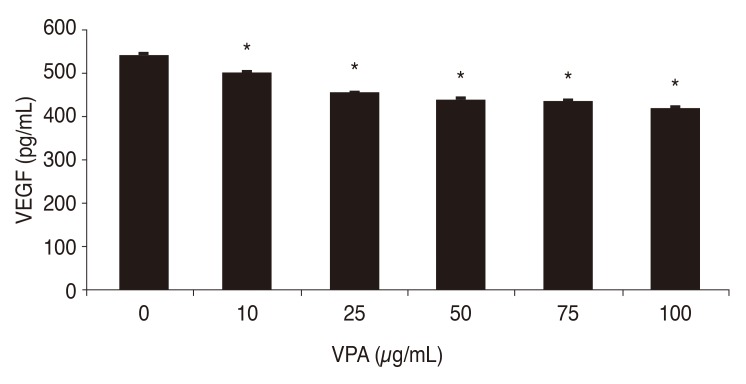
After exposing human retinal Müller cells to 400 µM CoCl2 to induce hypoxia, the cells were treated with 0, 10, 25, 50, 75, and 100 µg/mL of VPA. Expression of HIF-1α decreased significantly with increasing concentrations of VPA (p < 0.05), in a dose-dependent manner (Fig. 3). Similar results were found for the expression of VEGF (Fig. 4).
Fig. 3. Effects of increasing concentrations of valproic acid (VPA) on hypoxia-inducible factor-1 alpha (HIF-1α) expression by human retinal Müller cells during hypoxia. *p-value < 0.05.
Fig. 4. Effects of increasing concentrations of valproic acid (VPA) on vascular endothelial growth factor (VEGF) expression by human retinal Müller cells during hypoxia. *p-value < 0.05.
Discussion
Neovascularization in the retina leads to vision loss and blindness in age-related macular degeneration, diabetic retinopathy, branch retinal vein occlusion, and other ophthalmic disorders. The primary cause of this neovascularization is the physiological response to hypoxia. HIF-1α controls many of the reactions to hypoxia, as well as the expression of other genes, including placental growth factor, stromal-derived factor 1, angiopoietin 2, platelet-derived growth factor B, erythropoietin, and VEGF [31,32,33].
In ophthalmic diseases associated with neovascularization, improvements in vision have been demonstrated using therapeutics that target VEGF. However, these improvements appeared in only half of the patients and the effects were not long-lasting, requiring repeated, expensive treatments [34,35].
As a result, a number of studies have attempted to design t herapeutics targeting mediators other than V EGF. HIF-1α, in particular, has been one of the more promising targets. As noted, many potential mechanisms are available to suppress HIF-1α. Studies of the pro-apoptotic, anti-angiogenic effects of HDACIs as anti-cancer therapeutics have demonstrated that HDACI-mediated suppression of HIF-1α has a significant anti-angiogenic effect [36,37,38]. The HDACI VPA has been used widely in the treatment of epilepsy and bipolar disorder and this compound has been found to have neuroprotective or neuroregenerative effects on retinal ganglion cells in these patients [39,40,41].
This study determined that VPA suppresses HIF-1α expression by human retinal Müller cells during hypoxia (Fig. 3), suggesting that VPA could possibly be used to target HIF-1α in humans. However, further studies are needed to determine the precise mechanism by which HDACIs suppress HIF-1α [42,43,44].
This study found that VPA suppresses VEGF expression in human retinal Müller cells during hypoxia (Fig. 4), likely due to suppression of HIF-1α, which normally activates transcription of the VEGF gene [45].
The HIFs are heterodimeric nuclear proteins consisting of α and β subunits. There are three types of oxygen-reactive α subunits: HIF-1α, HIF-2α, and HIF-3α. HIF-3α had not been studied extensively and it is currently unknown whether HIF-1α or HIF-2α has a greater affect in the retina [46,47]. This study analyzed HIF-1α, but further studies are needed to determine whether HIF-1α or HIF-2α is more important in human retinal Müller cells.
One of the limitations of this study is that targeting HIF-1α is more non-specific than treatments targeting V EGF, because it controls the upper regulatory pathway. Treatments for specific targets would be better pharmacologically, but current widely used treatments that directly target VEGF have limitations, as mentioned above. Thus, controlling various hypoxia-induced pathways could be advantageous. This study identified some of the regulatory interactions between HIF-1α and VEGF, but further studies on other factors that are controlled by hypoxia are needed. This was an in vitro study, so further in vivo studies should also be performed before making conclusions about the actual effects of VPA on human retinal Müller cells.
In this study, CoCl2 was used to simulate the hypoxic state. One drawback of the cobalt model is that it represents only the chronic activation state of HIF-1, although it is widely established and thought to be an appropriate model for hypoxic states. With this concern in mind, experiments using CoCl2 may not actually reflect a hypoxic state with respect to the upper regulatory level of HIF-1.
In summary, VPA was found to decrease the expression of HIF-1α and VEGF in human retinal Müller cells during hypoxia. Using VPA or other HDACIs to target HIF-1α in retinal Müller cells could be a potential therapeutic strategy for the treatment of retinal vascular diseases.
Acknowledgements
Human retinal Müller cells (Moorfields/Institute of Ophthalmology-Müller 1, MIO-M1) were provided generously by professor G. Astrid Limb (Institute of Ophthalmology, University College London, London, UK).
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Congdon N, O'Colmain B, Klaver CC, et al. Causes a nd prevalence of visual impairment among adults in the United States. Arch Ophthalmol. 2004;122:477–485. doi: 10.1001/archopht.122.4.477. [DOI] [PubMed] [Google Scholar]
- 2.Congdon NG, Friedman DS, Lietman T. Important causes of visual impairment in the world today. JAMA. 2003;290:2057–2060. doi: 10.1001/jama.290.15.2057. [DOI] [PubMed] [Google Scholar]
- 3.Stalmans I, Ng YS, Rohan R, et al. Arteriolar and venular patterning in retinas of mice selectively expressing VEGF isoforms. J Clin Invest. 2002;109:327–336. doi: 10.1172/JCI14362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haigh JJ, Morelli PI, Gerhardt H, et al. Cortical and retinal defects caused by dosage-dependent reductions in VEGF-A paracrine signaling. Dev Biol. 2003;262:225–241. doi: 10.1016/s0012-1606(03)00356-7. [DOI] [PubMed] [Google Scholar]
- 5.Marneros AG, Fan J, Yokoyama Y, et al. Vascular endothelial growth factor expression in the retinal pigment epithelium is essential for choriocapillaris development and visual function. Am J Pathol. 2005;167:1451–1459. doi: 10.1016/S0002-9440(10)61231-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aiello LP, Avery RL, Arrigg PG, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331:1480–1487. doi: 10.1056/NEJM199412013312203. [DOI] [PubMed] [Google Scholar]
- 7.Adamis AP, Miller JW, Bernal MT, et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol. 1994;118:445–450. doi: 10.1016/s0002-9394(14)75794-0. [DOI] [PubMed] [Google Scholar]
- 8.Pierce EA, Avery RL, Foley ED, et al. Vascular endothelial growth factor/vascular permeability factor expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci U S A. 1995;92:905–909. doi: 10.1073/pnas.92.3.905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Miller JW, Adamis AP, Aiello LP. Vascular endothelial growth factor in ocular neovascularization and proliferative diabetic retinopathy. Diabetes Metab Rev. 1997;13:37–50. doi: 10.1002/(sici)1099-0895(199703)13:1<37::aid-dmr174>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 10.Aiello LP, Northrup JM, Keyt BA, et al. Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol. 1995;113:1538–1544. doi: 10.1001/archopht.1995.01100120068012. [DOI] [PubMed] [Google Scholar]
- 11.Stone J, Itin A, Alon T, et al. Development of retinal vasculature is mediated by hypoxia-induced vascular endothelial growth factor (VEGF) expression by neuroglia. J Neurosci. 1995;15(7 Pt 1):4738–4747. doi: 10.1523/JNEUROSCI.15-07-04738.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stone J, Chan-Ling T, Pe'er J, et al. Roles of vascular endothelial growth factor and astrocyte degeneration in the genesis of retinopathy of prematurity. Invest Ophthalmol Vis Sci. 1996;37:290–299. [PubMed] [Google Scholar]
- 13.Bai Y, Ma JX, Guo J, et al. Muller cell-derived VEGF is a significant contributor to retinal neovascularization. J Pathol. 2009;219:446–454. doi: 10.1002/path.2611. [DOI] [PubMed] [Google Scholar]
- 14.Wang J, Xu X, Elliott MH, et al. Muller cell-derived VEGF is essential for diabetes-induced retinal inflammation and vascular leakage. Diabetes. 2010;59:2297–2305. doi: 10.2337/db09-1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kaelin WG., Jr The von Hippel-Lindau tumor suppressor protein and clear cell renal carcinoma. Clin Cancer Res. 2007;13:680s–684s. doi: 10.1158/1078-0432.CCR-06-1865. [DOI] [PubMed] [Google Scholar]
- 16.Liao D, Johnson RS. Hypoxia: a key regulator of angiogenesis in cancer. Cancer Metastasis Rev. 2007;26:281–290. doi: 10.1007/s10555-007-9066-y. [DOI] [PubMed] [Google Scholar]
- 17.Ke Q, Costa M. Hypoxia-inducible f actor-1 (HIF-1) Mol Pharmacol. 2006;70:1469–1480. doi: 10.1124/mol.106.027029. [DOI] [PubMed] [Google Scholar]
- 18.Pili R, Donehower RC. Is HIF-1 alpha a valid therapeutic target? J Natl Cancer Inst. 2003;95:498–499. doi: 10.1093/jnci/95.7.498. [DOI] [PubMed] [Google Scholar]
- 19.Welsh SJ, Powis G. Hypoxia inducible factor as a cancer drug target. Curr Cancer Drug Targets. 2003;3:391–405. doi: 10.2174/1568009033481732. [DOI] [PubMed] [Google Scholar]
- 20.Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–732. doi: 10.1038/nrc1187. [DOI] [PubMed] [Google Scholar]
- 21.Giaccia A, Siim BG, Johnson RS. HIF-1 as a target for drug development. Nat Rev Drug Discov. 2003;2:803–811. doi: 10.1038/nrd1199. [DOI] [PubMed] [Google Scholar]
- 22.Powis G, Kirkpatrick L. Hypoxia inducible factor-1alpha as a cancer drug target. Mol Cancer Ther. 2004;3:647–654. [PubMed] [Google Scholar]
- 23.Brown JM, Wilson WR. Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer. 2004;4:437–447. doi: 10.1038/nrc1367. [DOI] [PubMed] [Google Scholar]
- 24.Poon E, Harris AL, Ashcroft M. Targeting the hypoxia-inducible factor (HIF) pathway in cancer. Expert Rev Mol Med. 2009;11:e26. doi: 10.1017/S1462399409001173. [DOI] [PubMed] [Google Scholar]
- 25.Gottlicher M, Minucci S, Zhu P, et al. Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J. 2001;20:6969–6978. doi: 10.1093/emboj/20.24.6969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang ZH, Hao CL, Liu P, et al. Valproic acid inhibits tumor angiogenesis in mice transplanted with Kasumi1 leukemia cells. Mol Med Rep. 2014;9:443–449. doi: 10.3892/mmr.2013.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shan Z, Feng-Nian R, Jie G, Ting Z. Effects of valproic acid on proliferation, apoptosis, angiogenesis and metastasis of ovarian cancer in vitro and in vivo. Asian Pac J Cancer Prev. 2012;13:3977–3982. doi: 10.7314/apjcp.2012.13.8.3977. [DOI] [PubMed] [Google Scholar]
- 28.Marks PA, Richon VM, Breslow R, Rifkind RA. Histone deacetylase inhibitors as new cancer drugs. Curr Opin Oncol. 2001;13:477–483. doi: 10.1097/00001622-200111000-00010. [DOI] [PubMed] [Google Scholar]
- 29.Drummond DC, Noble CO, Kirpotin DB, et al. Clinical development of histone deacetylase inhibitors as anticancer agents. Annu Rev Pharmacol Toxicol. 2005;45:495–528. doi: 10.1146/annurev.pharmtox.45.120403.095825. [DOI] [PubMed] [Google Scholar]
- 30.Johnstone RW, Licht JD. Histone deacetylase inhibitors in cancer therapy: is transcription the primary target? Cancer Cell. 2003;4:13–18. doi: 10.1016/s1535-6108(03)00165-x. [DOI] [PubMed] [Google Scholar]
- 31.DeNiro M, Al-Halafi A, Al-Mohanna FH, et al. Pleiotropic effects of YC-1 selectively inhibit pathological retinal neovascularization and promote physiological revascularization in a mouse model of oxygen-induced retinopathy. Mol Pharmacol. 2010;77:348–367. doi: 10.1124/mol.109.061366. [DOI] [PubMed] [Google Scholar]
- 32.Adams JM, Difazio LT, Rolandelli RH, et al. HIF-1: a key mediator in hypoxia. Acta Physiol Hung. 2009;96:19–28. doi: 10.1556/APhysiol.96.2009.1.2. [DOI] [PubMed] [Google Scholar]
- 33.Aiello LP, Pierce EA, Foley ED, et al. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci U S A. 1995;92:10457–10461. doi: 10.1073/pnas.92.23.10457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rey S, Semenza GL. Hypoxia-inducible factor-1-dependent mechanisms of vascularization and vascular remodelling. Cardiovasc Res. 2010;86:236–242. doi: 10.1093/cvr/cvq045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;355:1432–1444. doi: 10.1056/NEJMoa062655. [DOI] [PubMed] [Google Scholar]
- 36.Yu F, White SB, Zhao Q, Lee FS. HIF-1alpha binding to VHL is regulated by stimulus-sensitive proline hydroxylation. Proc Natl Acad Sci U S A. 2001;98:9630–9635. doi: 10.1073/pnas.181341498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mie Lee Y, Kim SH, Kim HS, et al. Inhibition of hypoxia-induced angiogenesis by FK228, a specific histone deacetylase inhibitor, via suppression of HIF-1alpha activity. Biochem Biophys Res Commun. 2003;300:241–246. doi: 10.1016/s0006-291x(02)02787-0. [DOI] [PubMed] [Google Scholar]
- 38.Williams RJ. Trichostatin A, an inhibitor of histone deacetylase, inhibits hypoxia-induced angiogenesis. Expert Opin Investig Drugs. 2001;10:1571–1573. doi: 10.1517/13543784.10.8.1571. [DOI] [PubMed] [Google Scholar]
- 39.Biermann J, Grieshaber P, Goebel U, et al. Valproic acid-mediated neuroprotection and regeneration in injured retinal ganglion cells. Invest Ophthalmol Vis Sci. 2010;51:526–534. doi: 10.1167/iovs.09-3903. [DOI] [PubMed] [Google Scholar]
- 40.Zhang Z, Qin X, Zhao X, et al. Valproic acid regulates antioxidant enzymes and prevents ischemia/reperfusion injury in the rat retina. Curr Eye Res. 2012;37:429–437. doi: 10.3109/02713683.2011.653616. [DOI] [PubMed] [Google Scholar]
- 41.Zhang Z, Qin X, Tong N, et al. Valproic acid-mediated neuroprotection in retinal ischemia injury via histone deacetylase inhibition and transcriptional activation. Exp Eye Res. 2012;94:98–108. doi: 10.1016/j.exer.2011.11.013. [DOI] [PubMed] [Google Scholar]
- 42.Jeong JW, Bae MK, Ahn MY, et al. Regulation and destabilization of HIF-1alpha by ARD1-mediated acetylation. Cell. 2002;111:709–720. doi: 10.1016/s0092-8674(02)01085-1. [DOI] [PubMed] [Google Scholar]
- 43.Kim MS, Kwon HJ, Lee YM, et al. Histone deacetylases induce angiogenesis by negative regulation of tumor suppressor genes. Nat Med. 2001;7:437–443. doi: 10.1038/86507. [DOI] [PubMed] [Google Scholar]
- 44.Kong X, Lin Z, Liang D, et al. Histone deacetylase inhibitors induce VHL and ubiquitin-independent proteasomal degradation of hypoxia-inducible factor 1alpha. Mol Cell Biol. 2006;26:2019–2028. doi: 10.1128/MCB.26.6.2019-2028.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Qian DZ, Kachhap SK, Collis SJ, et al. Class II histone deacetylases are associated with VHL-independent regulation of hypoxia-inducible factor 1 alpha. Cancer Res. 2006;66:8814–8821. doi: 10.1158/0008-5472.CAN-05-4598. [DOI] [PubMed] [Google Scholar]
- 46.Lin M, Chen Y, Jin J, et al. Ischaemia-induced retinal neovascularisation and diabetic retinopathy in mice with conditional knockout of hypoxia-inducible factor-1 in retinal Muller cells. Diabetologia. 2011;54:1554–1566. doi: 10.1007/s00125-011-2081-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mowat FM, Luhmann UF, Smith AJ, et al. HIF-1alpha and HIF-2alpha are differentially activated in distinct cell populations in retinal ischaemia. PLoS One. 2010;5:e11103. doi: 10.1371/journal.pone.0011103. [DOI] [PMC free article] [PubMed] [Google Scholar]