ABSTRACT
The performance of the Bactec FX blood culture system for detecting Brucella bacteremia within the routine 1-week incubation period was assessed in a prospective study conducted in an area in southern Israel in which Brucella melitensis is endemic. Aerobic vials (BD Bactec Plus Aerobic/F medium) inoculated with blood specimens obtained from adult patients with positive Rose-Bengal screening test results were monitored for 4 consecutive weeks, and blind subcultures of negative vials were performed on solid media on days 7 and 28. During a 16-month period, a total of 31 (35.2%) of 88 cultures, obtained from 19 (38.0%) of 50 patients, were positive for Brucella melitensis. The blood culture instrument identified 30 (96.8%) of 31 positive vials within 7 days of incubation; the single positive vial that was missed by the automated readings was detected only by the blind subculture performed on day 28. It is concluded that the Bactec FX system is able to detect the vast majority of episodes of Brucella bacteremia within the 1-week incubation protocol instituted in most clinical microbiology laboratories and without the need to perform blind subcultures of negative vials, enabling early diagnosis and saving labor and incubation time and space.
KEYWORDS: Brucella melitensis, blood cultures, detection
INTRODUCTION
Although veterinarian control of brucellosis has been successfully achieved in most industrialized countries, the disease still represents a serious health threat to human populations living in the developing world and to travelers to regions in which the disease is endemic (1, 2). In addition, because of the low infecting dose, the high infectivity, and the ability to cause prolonged disability in humans and loss of productivity in animal husbandry, brucellae are potential candidates for biological warfare (3).
The clinical manifestations of human brucellosis are protean and nonspecific, mimicking other infectious and noninfectious conditions, and affected patients require prolonged combination therapy with antibiotics that are not routinely prescribed for other infectious diseases (1). Therefore, laboratory confirmation of the diagnosis is of paramount importance for adequate case management. Although brucellar infections can be ascertained with serological tests and nucleic acid amplification assays, culture detection of circulating Brucella organisms remains a diagnostic cornerstone. Because of the fastidious nature of members of the genus Brucella, prolonged incubation of media and performance of blind subcultures of negative blood culture vials have been traditionally recommended to maximize isolation of these organisms (4, 5). Recent studies, however, have demonstrated that automated blood culture systems based on monitoring of CO2 production have improved sensitivity, compared to manual methods, enabling detection of brucellae in pediatric patients within the customary 1-week incubation protocol followed in most clinical microbiology laboratories (6). Data on the capability of automated technology for the diagnosis of brucellemia in adult patients, however, are scarce. Because of the slow replication time of brucellae (7), coupled with the release of only small quantities of measurable CO2 by members of the genus (8) and the fact that the magnitude of bacteremia in adults is usually lower than that observed in children (9), it is possible that the presence of the bacteria in blood culture media cannot be revealed within the routine incubation period. The implications of false-negative culture results are that diagnosis of the disease may be delayed or overlooked altogether and patients may develop focal complications and/or progress to the difficult-to-treat chronic stages of infection (1).
Brucellosis caused by Brucella melitensis is highly prevalent among the Bedouin population of southern Israel, which maintains herds of unvaccinated camels, sheep, and goats and consumes unpasteurized dairy products and among which the annual incidence in 2012 was as high as 151.9 cases per 100,000 population (10). A prospective study was conducted at a large hospital that serves this area, in which brucellosis is endemic, to assess the performance of the novel Bactec FX system (Becton-Dickinson, Sparks, MD) in detecting Brucella organisms in blood cultures obtained from adult patients.
RESULTS
During the 16-month period between July 2015 and November 2016, 88 blood culture vials, obtained from 50 adult individuals who exhibited positive Rose-Bengal (RB) screening test results, met the inclusion criteria. All patients exhibited standard agglutination test (SAT) titers of >1:160 and thus were considered to have brucellar infections.
Thirty-three patients were male, and the mean ± standard deviation age of the population was 39.2 ± 16.1 years. Patients had been sick for an average ± standard deviation of 5.2 ± 5.8 days (median, 3 days; range, 1 to 30 days). The clinical presentation included fever for 35 patients, arthralgia or arthritis for 9 patients (18.0%), fatigue for 8 patients (16.0%), headache for 6 patients (12%), night sweats or myalgia for 4 patients (8%) each, osteomyelitis for 3 patients (6%), and orchitis for 2 patients (4%). By the time the blood cultures were obtained, 4 patients (2 with positive and 2 with negative culture results) had been already administered potentially effective antibiotic therapy (tetracyclines, aminoglycosides, rifampin, or fluoroquinolones).
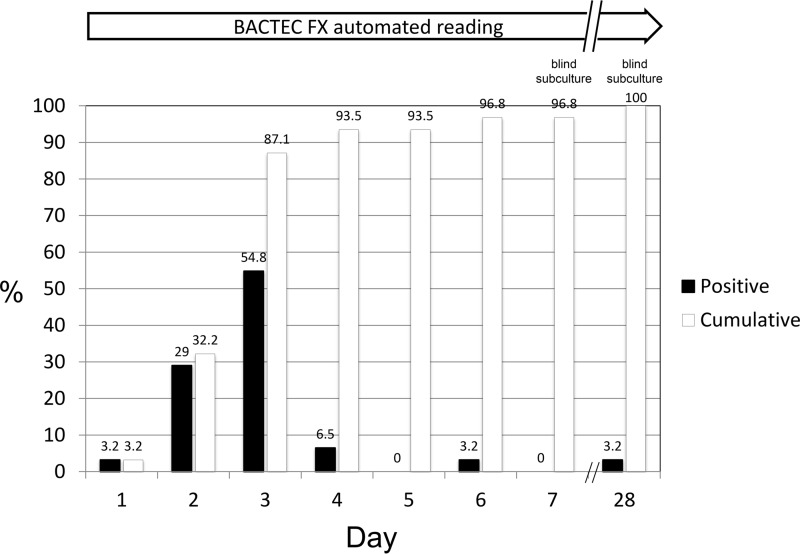
Overall, 31 cultures (35.2%) drawn from 19 patients (38.0%) grew Brucella organisms (all later identified as B. melitensis). The automated readings by the Bactec FX instrument detected 30 (96.8%) of 31 positive vials within the routine 7-day incubation period; the remaining vial was detected only by the blind subculture of the broth performed on day 28. Figure 1 shows the positivity rates and cumulative percentages of positive vials detected by the automated instrument and by blind subcultures during the 28-day period.
FIG 1.
Time to detection of Brucella melitensis by the Bactec FX blood culture system.
The blood volume inoculated into the 31 positive vials was 3.9 ± 2.5 ml (range, 0.7 to 14.3 ml; median, 2.9 ml), and that inoculated into the negative vials was 4.2 ± 2.6 ml (range, 1.2 to 13.2 ml; median, 3.3 ml; P = 0.688). For the 30 positive vials detected by the instrument, the time to positivity showed a weak and nonsignificant inverse correlation with the blood volume inoculated into the vial (r = −0.298; P = 0.110).
Culture-positive vials and those that remained negative by the Bactec FX instrument after a 28-day incubation period differed significantly in terms of SAT titers. Culture-positive patients exhibited an average ± standard deviation SAT titer of 3,802.1 ± 4,859.7, compared to 790.6 ± 1,887.7 for culture-negative patients (P < 0.001). No significant differences between culture-positive and culture-negative patients in terms of patient age or gender, duration of symptoms, clinical presentation, or previous antibiotic therapy were observed (data not shown).
DISCUSSION
Recovery of brucellae from normally sterile body fluids or tissues is considered irrefutable evidence of the disease (1). Isolation of the organisms permits identification to the species level and typing of the isolates for epidemiological studies, enables determination of antibiotic susceptibility when indicated, and makes it possible to diagnosis brucellosis in the early stages of infection, when serological test results may still be negative or demonstrate borderline antibody titers (1, 11). An additional advantage of the culture approach is that, in contrast to serological and nucleic acid amplification assays, it allows diagnosis among patients for whom the infection is not suspected (6).
Because of the slow generation time of brucellae, compared with other human pathogens, incubation of inoculated media for up to 30 days and periodic performance of blind subcultures of seemingly negative blood culture vials have been traditionally recommended (4, 6). In addition, members of the genus are characterized by low levels of production of CO2, which is the metabolic parameter detected by modern blood culture systems (6, 8). Therefore, limiting the monitoring of blood culture vials to the 1-week period instituted in most clinical microbiology laboratories cannot be routinely recommended unless it is firmly demonstrated that, with adoption of this approach, no significant numbers of positive cultures are missed. The time to detection is thus a crucial issue that needs to be specifically addressed in well-designed prospective studies. Adequate assessment of the ability of any blood culture system to detect brucellae requires keeping vials for long periods and performing blind subcultures of negative vials to ensure that the presence of the organism is not missed by the curtailed incubation protocol.
Experience with the isolation of Brucella spp. by the modern automated blood culture instruments of the Bactec series has been accumulating at a slow pace (6, 8). Although the disease is still prevalent in many developing countries, the use of modern bacteriological techniques is restricted in resource-poor regions because of the high cost; in the more affluent Western world, where the use of modern automated blood culture technology is widespread, brucellosis has been successfully controlled or eradicated altogether (1).
The present investigation is the first to evaluate the ability of the novel Bactec FX blood culture system to detect circulating Brucella organisms in adult patients. The study results demonstrate that the automated instrument, coupled with BD Bactec Plus Aerobic/F medium, enables identification of the vast majority of B. melitensis-positive blood cultures with the customary 1-week incubation protocol followed by clinical microbiology laboratories. These results are especially notable because of the low concentrations of circulating Brucella organisms (6) and the fact that the blood specimens inoculated into the vials were generally small (12). It may be speculated that, if larger blood samples (8 to 10 ml) had been drawn, as recommended by the manufacturer, then additional positive cultures could have been detected.
The sensitivity of the Bactec FX system allows detection of organisms without the need to perform subcultures of vials exhibiting negative automated readings at the end of the 7-day period. These favorable results imply that use of the system results in early detection of brucellar infections and makes diagnosis possible even in cases in which the disease is not suspected. Human brucellosis is a “great imitator,” and individuals with the infection may present with a variety of symptoms suggesting other medical conditions; frequently, the unexpected recovery of the organism in a blood culture is the first and only evidence of the true nature of the disease. From the laboratory perspective, use of the automated Bactec FX system limits the exposure of technicians to dangerous solid media growing contagious brucellae and translates to savings of incubation space, time, and labor.
MATERIALS AND METHODS
Study site and sample testing.
The Soroka University Medical Center is the only hospital that provides inpatient services in southern Israel and the only site where blood cultures are obtained. Blood specimens for culture and Brucella serological testing are drawn at the hospital emergency departments and inpatient wards at the physicians' discretion. Blood cultures are processed with the Bactec FX system according the manufacturer's recommendations. For the serological diagnosis of brucellosis, sera are initially screened with the Rose-Bengal (RB) assay and positive samples are further tested with the SAT. SAT titers of 1:160 are considered diagnostic for the disease (11).
Bacteriological methods. (i) Blood culture protocol.
Even in southern Israel, where the disease is highly endemic, the prevalence of brucellae in blood cultures is rather low, i.e., 514 (2.5%) of 20,620 positive blood cultures detected between 2002 and 2009 (13). To eliminate the need to investigate a large population of patients at low risk for the disease, the present study was designed to restrict investigation to blood cultures obtained from individuals who were natural candidates for Brucella bacteremia. A daily list of all adult patients from whom positive RB test results were obtained was compiled. The inoculated aerobic blood culture vials (BD Bactec Plus Aerobic/F medium) drawn from those individuals were located by searching the Bactec FX EpiCenter operating system. The blood culture vials thus identified were monitored by the automated instrument for 28 consecutive days or until the vials were detected as positive, in which case the time to detection was recorded. To detect Brucella-containing vials missed by the automated readings, blind subcultures of negative vials were performed on days 7 and 28, on Trypticase soy agar medium with 5% added sheep hemoglobin (blood agar) and on chocolate agar plates. Vials in which Brucella organisms were detected by the blind subculture performed on day 7 were kept in the Bactec FX cabinet and monitored until day 28, to determine the time to positivity with the automated system beyond the routine 1-week incubation protocol. All blood culture vials from which Brucella organisms were detected either by the Bactec FX system or by blind subcultures were weighed, to assess the influence of the inoculated blood volume on the time to detection of the organisms.
Because brucellae are strictly aerobic bacteria, anaerobic vials (Bactec Plus Anaerobic/F medium) were not included in the study. The RB assay was chosen instead of the more specific SAT because it has been demonstrated that, with a SAT cutoff value as low as 1:160, a substantial fraction of patients with brucellemia are routinely missed (11).
(ii) Identification of Brucella organisms.
The culture broth from positive vials was subjected to Gram staining, subcultured on blood agar and chocolate agar plates, and incubated at 35°C in a 5% CO2-enriched atmosphere. For safety reasons, blood culture vials flagged as positive by the Bactec FX instrument and seeded solid media were handled in a class II safety cabinet. Organisms were identified as brucellae on the bases of typical morphological and staining findings (small Gram-negative coccobacilli), positive oxidase, catalase, and urease test results, no fermentation of sugars, and lack of motility and hemolysis, with confirmation by positive agglutination reactions with specific antiserum. Identification of the isolates to the species level was performed at the Kimron Veterinary Institute (Beth-Dagan, Israel). Susceptibility of recovered brucellae to phages Tb and Iz identified the isolates as B. melitensis (14).
Data analysis.
The number of blood culture vials from which brucellae were detected by the Bactec FX instrument within the first week of incubation, of the total number of vials from which the organisms were detected within 28 days by the automated system and/or by blind subcultures, was determined. Blood culture vials from which both brucellae and contaminants were isolated were omitted from the data analysis because the presence of accompanying organisms would have spuriously increased the CO2 contents of the vials, decreasing the time to positivity.
The blood volumes inoculated into culture-positive and culture-negative vials were determined by weight and compared, to assess the association between the sensitivity of the blood cultures and the volume of the specimens. In addition, Pearson's correlation analysis was performed to assess the linear association between the inoculated blood volume and the time to detection for the vials read as positive by the automated instrument.
The patients' medical records were also reviewed, and relevant demographic and clinical data were extracted. The statistical significance of the comparisons of numerical data was assessed with the Student t test and the Mann-Whitney U test, as appropriate. Proportions were compared with the chi-square test. P values of <0.05 were considered significant for all comparisons. The local ethics committee approved the performance of the study.
REFERENCES
- 1.Pappas G, Akritidis N, Bosilkovski M, Tsianos E. 2005. Brucellosis. N Engl J Med 352:2325–2336. doi: 10.1056/NEJMra050570. [DOI] [PubMed] [Google Scholar]
- 2.Young EJ. 1995. An overview of human brucellosis. Clin Infect Dis 21:283–290. doi: 10.1093/clinids/21.2.283. [DOI] [PubMed] [Google Scholar]
- 3.Doganay GD, Doganay M. 2013. Brucella as a potential agent of bioterrorism. Recent Pat Antiinfect Drug Discov 8:27–33. doi: 10.2174/1574891X11308010006. [DOI] [PubMed] [Google Scholar]
- 4.Alton GG, Jones LM. 1967. Laboratory techniques in brucellosis. Monogr Ser World Health Organ 55:1–92. [PubMed] [Google Scholar]
- 5.Hausker WJ Jr, Moyer NP, Holcomb LA. 1985. Brucella, p 382–386. In Lennette EH, Ballows A, Hausler WJ Jr, Shadomy EH (ed), Manual of clinical microbiology, 4th ed American Society for Microbiology, Washington, DC. [Google Scholar]
- 6.Yagupsky P. 1999. Detection of brucellae in blood cultures. J Clin Microbiol 37:3437–3442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gamazo C, Vitas AI, Lopez-Goñi I, Diaz R, Moriyon I. 1993. Factors affecting detection of Brucella melitensis by BACTEC NR730, a nonradiometric system for hemocultures. J Clin Microbiol 31:3200–3203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Solomon HM, Jackson D. 1992. Rapid diagnosis of Brucella melitensis in blood: some operational characteristics of the BACT/ALERT. J Clin Microbiol 30:222–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yagupsky P, Nolte FS, Menegus MA. 1990. Quantitative aspects of septicemia. Clin Microbiol Rev 3:269–279. doi: 10.1128/CMR.3.3.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shemesh AA, Yagupsky P. 2013. Increasing incidence of human brucellosis in southern Israel after the cessation of a veterinarian control campaign. Air Water Borne Dis 2:112. [Google Scholar]
- 11.Shemesh AA, Yagupsky P. 2011. Limitations of the standard agglutination test for detecting patients with Brucella melitensis bacteremia. Vector Borne Zoonotic Dis 11:1599–1601. doi: 10.1089/vbz.2011.0704. [DOI] [PubMed] [Google Scholar]
- 12.Bouza E, Sousa D, Rodríguez-Creixéms M, Lechuz JG, Muñoz P. 2007. Is the volume still a significant factor in the diagnosis of bloodstream infections? J Clin Microbiol 45:2765–2769. doi: 10.1128/JCM.00140-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shemesh AA, Yagupsky P. 2012. Isolation rates of Brucella melitensis in an endemic area and implications for laboratory safety. Eur J Clin Microbiol Infect Dis 31:441–443. doi: 10.1007/s10096-011-1325-8. [DOI] [PubMed] [Google Scholar]
- 14.Banai M, Mayer I, Cohen A. 1990. Isolation, identification, and characterization in Israel of Brucella melitensis biovar 1 atypical strains susceptible to dyes and penicillin, indicating the evolution of a new variant. J Clin Microbiol 28:1057–1059. [DOI] [PMC free article] [PubMed] [Google Scholar]