Abstract
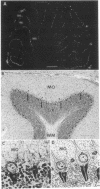
A method was developed to produce monoclonal antibodies to brain cell antigens by using Xenopus oocytes as immunological vectors. The method consists in injecting Xenopus oocytes with rat brain mRNA to express foreign proteins and using the oocytes for immunization. Immunizations were preceded by immunotolerization of mice to antigens of native oocyte membranes. With this approach we generated a set of monoclonal antibodies that are specific markers for the cerebellar "pinceau"--a unique complex synapse formed between basket cell terminals and the initial segment of the Purkinje cell axon. Our findings reveal an immunoreactivity highly localized at the pinceau and its late expression beginning at postnatal day 19 during cerebellar development.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altman J. Autoradiographic and histological studies of postnatal neurogenesis. 3. Dating the time of production and onset of differentiation of cerebellar microneurons in rats. J Comp Neurol. 1969 Jul;136(3):269–293. doi: 10.1002/cne.901360303. [DOI] [PubMed] [Google Scholar]
- Altman J. Postnatal development of the cerebellar cortex in the rat. II. Phases in the maturation of Purkinje cells and of the molecular layer. J Comp Neurol. 1972 Aug;145(4):399–463. doi: 10.1002/cne.901450402. [DOI] [PubMed] [Google Scholar]
- Altman J., Sudarshan K. Postnatal development of locomotion in the laboratory rat. Anim Behav. 1975 Nov;23(4):896–920. doi: 10.1016/0003-3472(75)90114-1. [DOI] [PubMed] [Google Scholar]
- Cuello A. C., Priestley J. V., Sofroniew M. V. Immunocytochemistry and neurobiology. Q J Exp Physiol. 1983 Oct;68(4):545–578. doi: 10.1113/expphysiol.1983.sp002748. [DOI] [PubMed] [Google Scholar]
- Galfre G., Howe S. C., Milstein C., Butcher G. W., Howard J. C. Antibodies to major histocompatibility antigens produced by hybrid cell lines. Nature. 1977 Apr 7;266(5602):550–552. doi: 10.1038/266550a0. [DOI] [PubMed] [Google Scholar]
- Gallyas F. Light insensitive physical developers. Stain Technol. 1979 Jul;54(4):173–176. doi: 10.3109/10520297909112679. [DOI] [PubMed] [Google Scholar]
- Guilbert B., Mahana W., Gilbert M., Mazie J. C., Avrameas S. Presence of natural autoantibodies in hyperimmunized mice. Immunology. 1985 Nov;56(3):401–408. [PMC free article] [PubMed] [Google Scholar]
- Hockfield S. A Mab to a unique cerebellar neuron generated by immunosuppression and rapid immunization. Science. 1987 Jul 3;237(4810):67–70. doi: 10.1126/science.3603010. [DOI] [PubMed] [Google Scholar]
- Korn H., Axelrad H. Electrical inhibition of Purkinje cells in the cerebellum of the rat. Proc Natl Acad Sci U S A. 1980 Oct;77(10):6244–6247. doi: 10.1073/pnas.77.10.6244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kusano K., Miledi R., Stinnakre J. Cholinergic and catecholaminergic receptors in the Xenopus oocyte membrane. J Physiol. 1982 Jul;328:143–170. doi: 10.1113/jphysiol.1982.sp014257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975 Aug 7;256(5517):495–497. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- Maeda N., Niinobe M., Nakahira K., Mikoshiba K. Purification and characterization of P400 protein, a glycoprotein characteristic of Purkinje cell, from mouse cerebellum. J Neurochem. 1988 Dec;51(6):1724–1730. doi: 10.1111/j.1471-4159.1988.tb01151.x. [DOI] [PubMed] [Google Scholar]
- Matthew W. D., Sandrock A. W., Jr Cyclophosphamide treatment used to manipulate the immune response for the production of monoclonal antibodies. J Immunol Methods. 1987 Jun 26;100(1-2):73–82. doi: 10.1016/0022-1759(87)90174-8. [DOI] [PubMed] [Google Scholar]
- Miledi R. A calcium-dependent transient outward current in Xenopus laevis oocytes. Proc R Soc Lond B Biol Sci. 1982 Jul 22;215(1201):491–497. doi: 10.1098/rspb.1982.0056. [DOI] [PubMed] [Google Scholar]
- Painter C. J., Monestier M., Chew A., Bona-Dimitriu A., Kasturi K., Bailey C., Scott V. E., Sidman C. L., Bona C. A. Specificities and V genes encoding monoclonal autoantibodies from viable motheaten mice. J Exp Med. 1988 Mar 1;167(3):1137–1153. doi: 10.1084/jem.167.3.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellegrino L. J., Altman J. Effects of differential interference with postnatal cerebellar neurogenesis on motor performance, activity level, and maze learning of rats: a developmental study. J Comp Physiol Psychol. 1979 Feb;93(1):1–33. doi: 10.1037/h0077589. [DOI] [PubMed] [Google Scholar]
- Seigneurin J. M., Guilbert B., Bourgeat M. J., Avrameas S. Polyspecific natural antibodies and autoantibodies secreted by human lymphocytes immortalized with Epstein-Barr virus. Blood. 1988 Mar;71(3):581–585. [PubMed] [Google Scholar]
- Slemmon J. R., Danho W., Hempstead J. L., Morgan J. I. Cerebellin: a quantifiable marker for Purkinje cell maturation. Proc Natl Acad Sci U S A. 1985 Oct;82(20):7145–7148. doi: 10.1073/pnas.82.20.7145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sotelo C., Llinás R. Specialized membrane junctions between neurons in the vertebrate cerebellar cortex. J Cell Biol. 1972 May;53(2):271–289. doi: 10.1083/jcb.53.2.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokota S. Effect of particle size on labeling density for catalase in protein A-gold immunocytochemistry. J Histochem Cytochem. 1988 Jan;36(1):107–109. doi: 10.1177/36.1.3335766. [DOI] [PubMed] [Google Scholar]
- de Blas A. L., Vitorica J., Friedrich P. Localization of the GABAA receptor in the rat brain with a monoclonal antibody to the 57,000 Mr peptide of the GABAA receptor/benzodiazepine receptor/Cl- channel complex. J Neurosci. 1988 Feb;8(2):602–614. doi: 10.1523/JNEUROSCI.08-02-00602.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de StGroth S. F., Scheidegger D. Production of monoclonal antibodies: strategy and tactics. J Immunol Methods. 1980;35(1-2):1–21. doi: 10.1016/0022-1759(80)90146-5. [DOI] [PubMed] [Google Scholar]