Abstract
Immune-mediated drug-induced hepatotoxicity is often unrecognized as a potential mode of action due to the lack of appropriate in vitro models. We have established an in vitro rat donor-matched hepatocyte and Kupffer cell co-culture (HKCC) model to study immune-related responses to drug exposure. Optimal cell culture conditions were identified for the maintenance of co-cultures based on cell longevity, monolayer integrity, and cytokine response after lipopolysaccharide (LPS) exposure. Hepatocyte monocultures and HKCCs were then used to test a subset of compounds associated with hepatotoxic effects with or without LPS. Cytokine levels and metabolic activity (cytochrome P450 3A [Cyp3A]) were measured after a 48-h exposure to monitor endotoxin-induced changes in acute phase and functional end points. LPS-activated HKCCs, but not hepatocyte monocultures, treated with trovafloxacin or acetaminophen, compounds associated with immune-mediated hepatotoxicity, showed LPS-dependent decreases in interleukin-6 production with concomitant increases in Cyp3A activity. Differential endotoxinand model-dependent alterations were observed in cytokine profiles and Cyp3A activity levels that corresponded to specific compounds. These results indicate the utility of the HKCC model system to discern compound-specific effects that may lead to enhanced or mitigate hepatocellular injury due to innate or adaptive immune responses.
Keywords: Kupffer cells, hepatocytes, in vitro model systems, hepatotoxicity, co-culture
Introduction
Unexpected and unexplained hepatotoxicity continues to be one of the main adverse outcomes observed in humans during clinical trials and after-market withdrawals.1-3 Drug-induced hepatotoxic modes of action are often complex, involving metabolic activation, multiple cell types, and perturbation of biochemical pathways involving both hepatocytes and resident macrophages (i.e., Kupffer cells [KCs]).1,4-6 Many complex, immune-mediated hepatocellular responses, such as reactive metabolite formation, infectious disease, circulating cytokines, and gut-derived endotoxin inflammation, require interactions among hepatocytes, endothelial cells, and KCs.1,4,6,7
KCs constitute the largest resident macrophage population in the body and are crucial for the regulation of immune-mediated hepatotoxicity and liver injury.8 In their primary scavenger role, KCs endocytose foreign particles and bacterial endotoxins, which causes their activation and production of a number of cell signaling and stress pathway modulators, such as reactive oxygen species and cytokines, including tumor necrosis factor (TNF)-α and interleukin (IL)-1.9 Cell damage and soluble stress signals during drug-induced hepatocellular injury cause a similar KC activation, which modulates hepatocyte and nonparenchymal cell death by apoptosis.8-10 In addition, cytokines and chemokines secreted by KCs during injury modulate the metabolic activity of hepatocytes and induce the expression of acute phase proteins, such as C-reactive protein and NOS2, whereas causing the suppression of genes involved in the metabolism and clearance of xenobiotics, including cytochrome P-450 enzymes, uridine 5′ -diphospho-glucuronosyl transferase systems, and uptake and efflux transporters.1,11-14
These complex and dynamic cellular interactions are not adequately captured in traditional hepatocyte monocultures. As such, indirect hepatocellular toxicity caused by immune cell activation and hepatic inflammation is often overlooked as a potential mode of action.6 Development of an optimized co-culture system that incorporates KCs will be imperative to study these events. In addition, there is a growing body of evidence for the role of genetic variability and adaptive immune responses within the human population that accounts for some idiosyncratic drug-induced liver injury and its severity in some patients.2 This also becomes even more important when using inbred strains of rodents or animal models with rare or unique genetic backgrounds.15
Clearly, to better assess the potential of compounds to cause immune-mediated hepatotoxicity, more reliable and predictive culture models that incorporate donor-matched cell types are needed. Previously, we developed a rodent-based co-culture model using a commercial source of cryopreserved KCs and observed enhanced drug-induced hepatotoxicity under glucocorticoid-free medium conditions.16 In the present study, we present a more metabolically competent in vitro rat hepatic co-culture system that incorporates donor-matched primary hepatocytes and Kupffer cell co-cultures (HKCCs) to assess immune-mediated hepatotoxicity over an extended culture period. The effects of different medium formulations and glucocorticoid levels were investigated to define suitable experimental conditions before compound testing.17 Validation studies were conducted under predefined culture conditions using a set of hepatotoxic compounds, including trovafloxacin (TVX), an antibiotic associated with immune-related hepatotoxicity, and acetaminophen (APAP), an analgesic and antipyretic agent associated with reactive metabolite formation and oxidative stress. As part of these studies, we determined the concentration-dependent responses of a set of test compounds on functional end points (cytochrome P450 3A [Cyp3A] activity) and cytokine profiles (IL-6 and TNF-α) in the presence and absence of lipopolysaccharide (LPS). Our results indicate that this enhanced co-culture model system provides a more stable and physiologically relevant platform by which to investigate drug-induced, immune-mediated reactions that can lead to acute hepatotoxic effects.
Materials and Methods
Reagents
Trovafloxacin, levofloxacin, allyl alcohol (AA), and APAP were purchased from Sigma-Aldrich (St. Louis, MO). Triclosan (Irgacare MP) was obtained from Ciba-Geigy AG (Basel, Switzerland). Propiconazole and acetochlor were purchased from Chem Service (West Chester, PA). All other compounds were purchased from Sigma-Aldrich and were of the highest grade available. Compound stocks were prepared in dimethyl sulfoxide (Sigma-Aldrich) so that the final concentration did not exceed 0.1%. LPS from Escherichia coli strain 0127:B8 (Sigma-Aldrich, Cat # L4516) was dissolved in sterile phosphate-buffered saline (PBS).
Isolation of Primary Hepatocytes and KCs
Donor-matched hepatocytes and KCs were co-isolated from individual adult (200-300 g) male Sprague-Dawley rats (Charles River Laboratories, Raleigh, NC) using differential, density-gradient, and counterflow elutriation centrifugation methods.18,19 Briefly, in situ perfusions were performed following a modified 2-step digestion method using a collagenase/protease enzyme blend according to the manufacturer's recommendations (Vitacyte, Indianapolis, IN). After digestion, the liver tissue was dissected, placed in a sterile 150-mm petri dish containing Hank's balanced salt solution (HBSS) supplemented with 1% bovine serum albumin (BSA), 15 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, and 1 g/L glucose (ThermoFisher Life Technologies, Grand Island, NY), and transferred to a sterile biosafety cabinet. After removing the outer membrane (Glisson's capsule) with sterile forceps, the digested liver tissue was gently shaken to remove the parenchymal cells from the vascular tree without direct mechanical manipulation. The crude cell slurry was poured through a mesh filter (105 μm) into 50-mL tubes and centrifuged at 70 g for 3 min, which enriched for the mature hepatocytes in the pellet and nonparenchymal cells (NPCs) in the supernatant. Supernatants were collected for KC isolation, and the hepatocyte pellets were further subjected to Percoll density-gradient centrifugation (GE Healthcare, Marlborough, MA) which yielded >99% pure population of hepatocytes.18 Final hepatocyte yields and viabilities were determined using a Vision CBA Image Cytometry System (Nexcelom Bioscience, Lawrence, MA) and seeded as described subsequently.
For the isolation of KCs, the liver remnant with residual parenchymal and NPCs was placed in a 250-mL specimen cup containing 100 mL of supplemented HBSS with 1% BSA and 100 μg/mL DNase and further dissociated by gentle stirring for 5 min. Afterward, the cell slurry was filtered through a 297-micron mesh and centrifuged, along with the supernatants from the initial hepatocyte low-speed spin, at 500 × g for 6 min at 4°C.20 Crude NPC pellets were resuspended in supplemented HBSS, filtered using a 70-micron snap-cap filter, and loaded into a sterile 10-mL syringe. KCs were then further separated from other NPCs by a combination of Optiprep (Sigma-Aldrich) density-gradient and counterflow elutriation centrifugation using an Avanti J-26XP centrifuge (Beckman-Coulter, Brea, CA) and a JE-5.0 rotor equipped with a 5-mL standard chamber following a modified version of the methods described by Valatas et al.19 Adjustments were made to the elutriation protocol as necessary until KC purity was determined to be >90% based on flow cytometry results (see Flow Cytometry section).
Plating of Primary Cell Co-cultures
HKCCs were plated at a 2:1 (H:K) ratio onto 48-well type I collagen-coated plates. The ratio of KC and hepatocytes was initially optimized as part of the development of the co-culture system with the goal to achieve maximum cytokine production in the presence of LPS.16 Whereas normal KC-to-hepatocyte ratios are on average closer to 1:4 across the liver acinar structure, the 1:2 ratio that was used for the co-culture model does represent a near physiological condition when considering their enrichment in the periportal region under inflammatory conditions in vivo.21
For initial studies exploring effects of dexamethasone (Dex), co-cultures were plated in advanced Dulbecco's modified Eagle's medium (DMEM) supplemented with penicillin–streptomycin, GlutaMax, and 10% fetal bovine serum (Life Technologies). For later studies investigating effects of hydrocortisone (HC) and screening compounds, co-cultures were plated in DMEM-high glucose supplemented with MEM nonessential amino acids, penicillin–streptomycin, 10% fetal bovine serum (Life Technologies), and insulin (10 mg/mL; Sigma-Aldrich). Hepatocytes were plated at 375,000 cells per well and allowed to attach for approximately 1 h at 37°C/5% CO2 with manual gentle shaking every 15 minutes. After hepatocyte attachment, medium with unattached cells was removed and donor-matched KCs in supplemented plating medium were added at 187,500 cells per well. KCs were allowed to attach for approximately 1 h in the incubator with gentle shaking every 15 min and transferred into maintenance medium before use.
Flow Cytometry
Cell sample preparation and flow cytometry measurements were performed using a modification of methods described previously.22 Small aliquots of NPC fractions were collected from the final cell pellets after elutriation and density-gradient separation as described earlier. Cells were then washed with PBS containing 2.5 mM EDTA and pelleted by centrifugation at 300 × g for 5 minutes. Cells were then resuspended at 1 × 106 per 100 μL staining buffer (BD Biosciences, San Jose, CA) containing 2.5 mM EDTA. To measure viability, cells were stained with a fixable LIVE/DEAD stain (Thermo Fisher, Waltham, MA) according to the manufacturer's recommendations. Cells were then blocked with Fcγ receptor (BD Biosciences) for 15 minutes and stained with AlexaFluor 488-conjugated CD163 (AbD Serotec, Raleigh, NC) for 30 min at 4°C. For intracellular staining of CD68, cells were fixed and permeabilized using BD IntraSure Kit (BD Biosciences). AlexaFluor 647-conjugated CD68 antibody (AbD Serotec) was added in 100 μL of staining buffer and incubated for 30 min at 4°C.
Flow cytometry and cell fluorescence measurements were performed using a BD Cantos II flow cytometer (BD Biosciences) and analyzed with FlowJo software (version 10.0.6, Tree Star, Ashland, OR). Samples were analyzed using 100,000 events based on NPC population. Parameters for forward- and side-scatter (FSC/SSC) measurements were set to include all cell types potentially present in liver cell isolation and elutriation fractions (i.e., endothelial cells, stellate cells, KCs, and hepatocytes). Populations were first interpreted using relative cell size and complexity in FSC/SSC, viability, and the presence or absence of cellular markers. Calculations for the percentage of each cell type were based on live cell populations as determined by viability analysis. KCs were identified as CD68+, with CD163 determining maturation. Mature KCs were defined as CD68+/CD163+. Liver sinusoidal endothelial cells were identified by exclusion.
Immunocytochemistry
To visually confirm KC enrichment in elutriated fractions and KC localization within HKCCs, KC monocultures and HKCCs were stained with antibodies against CD68 and CD163. Cultures were activated with LPS (1 (μg/mL) for 48 h after 24 h acclimation. Cells were fixed and permeabilized with ice-cold methanol. Cultures were washed 2 times with Dulbecco's PBS and blocked using BSA Stain Buffer (BD Pharminogen) containing diluted Hoechst 33342 nuclear dye (Thermo Fisher). Individual wells were probed for CD68 or CD163 using mouse anti-rat primary antibodies (AbD Serotec) incubated for 6 h at 4°C. Cultures were washed and incubated with a goat anti-mouse AlexaFluor 488 secondary antibody (Thermo Fisher) for 1 h at room temperature. After washing, cells were visualized with the EVOS FL Cell Imaging System (Thermo Fisher).
Comparison of Maintenance Media
To extend HKCC longevity, 4 different maintenance medium formulations were evaluated: Advanced DMEM (A-DMEM) (Life Technologies), modified Chee's medium (MCM) (Life Technologies), Williams' E Medium (WEM) (Life Technologies), or hepatocyte maintenance media (HMM) (Lonza, Walkersville, MD). HKCCs were plated as previously described earlier and transitioned to 1 of 4 maintenance medium formulations supplemented with ITS+ and penicillin-streptomycin (Thermo Fisher) 30 min after KC attachment. After a 24-h acclimation period, cultures were exposed to LPS (1 (μg/mL) for 48 h. Cell viability after 48 h LPS exposure was assessed by measuring cellular ATP levels as described subsequently.
Glucocorticoid Effects on LPS-Mediated Responses
To identify a plating and treatment regimen that would provide HKCCs with sufficient glucocorticoids to sustain hepatocytes over a 3- to 4-day culture period with minimal suppression of LPS-induced cytokine production, pretreatment of hepatocytes and/or KCs with normal and reduced levels of Dex and HC were evaluated. Pretreatment with Dex was initially evaluated by exposing both hepatocytes and KCs during the respective attachment stages to supplemented advanced DMEM containing 1 μM Dex. After attachment, the cells were washed, medium replaced with Dex-free MCM, and HKCCs were treated with LPS (1 (μg/mL) for a 48-h period followed by assessment of cytokine production levels and morphologic integrity. Additional experiments in which only the hepatocyte fraction was initially plated in advanced DMEM containing Dex (20 nM) were conducted. Before adding KCs in Dex-free medium, hepatocyte monolayers were washed with Dex-free medium. HKCCs were then treated with LPS in Dex-free MCM as described in the previous section followed by evaluation of cytokine production levels and morphologic integrity.
To ensure appropriate KC-mediated events were recapitulated in HKCCs exposed to HC, effects of TVX and levofloxacin (LVX), a toxic and nontoxic fluoroquinolone antibiotic, respectively, on cytokine release and HKCC viability were determined. Hepatocytes were allowed to attach (approximately 1 h) in plating medium containing 1 μM HC before addition of KC. HKCCs were maintained in MCM as described earlier and co-exposed to TVX or LVX (0-200 μM) in the presence or absence of LPS on culture day 1 for 48 h.
In a separate set of experiments, the concentration- and time-dependent effects of HC on LPS-mediated cytokine production in HKCCs were examined by exposing hepatocytes and KCs to plating medium containing 1 or 10 μM HC for a 2 or 24 h duration, before changing cells into Dex-free maintenance medium (MCM). HKCCs were then treated with LPS (1 μg/mL), and samples were collected for determination of cytokine production, Cyp3A activity, and gene expression as described subsequently.
Measurement of Cytokine Production
Cytokine levels were measured as described previously.16 Briefly, cell culture supernatant samples were collected at indicated time points after treatment with compounds in the presence or absence of 1 (μg/mL LPS and stored at −80°C until analysis. Individual cytokine levels were measured using rat IL-6 and TNF-α ELISA kits according to the manufacturer's recommendations (Life Technologies/Thermo Fisher) and quantified using a SpectroMax M5 plate reader and using SoftMax Pro software (Molecular Devices, Sunnyvale, CA). Additional multiplex measurements were performed on selected medium samples using the Cytokine Rat 10-Plex Panel (Thermo Fisher) for the Luminex® platform. Data were collected and analyzed using the Bio-Plex System (Bio-Rad, Hercules, CA).
Measurement of Cyp3A Activity
Cyp3A metabolic activity was measured in intact cultures as described previously.16 Briefly, HKCCs were cultured and treated with compounds in the presence or absence of LPS for 48 h as indicated earlier. Cyp3A activity was measured directly in wells using the P450-Glo™ CYP3A4 assay with Luciferin-IPA (Cat # V9001; Promega, Madison, WI) according to the manufacturer's instructions for cultured cells and using a Luciferin standard curve (Beetle Luciferin, Cat # E1601; Promega). Plates were analyzed using a SpectroMax M5 plate reader and using SoftMax Pro software. Values are reported as units of Luciferin (nM).
Gene Expression Profiling
At the end of the 48-h treatment period, RNA was extracted from HKCCs using the RNeasy Miniprep Kit (Qiagen, Valencia, CA) according to the manufacturer's protocol. RNA samples were reverse transcribed with the High Capacity RNA-to-cDNA Kit following the manufacturer's instructions (Applied Biosystems, ThermoFisher, Grand Island, NY). Quantitative PCR reactions (technical duplicates) were performed with 10 ng of cDNA per well using TaqMan® Universal PCR Master Mix (Applied Biosystems) and manufacturer-recommended “best coverage” TaqMan® Gene Expression Assays for Crp, Nos2, Tat, and Alb (Applied Biosystems). Reactions were carried out using an ABI 7900HT Fast Real-Time PCR System with sample analysis performed using Sequence Detection Systems software (version 2.4; Life Technologies). Relative quantities were calculated for each gene of interest by normalizing to Hprt1 and are presented as a percent of the respective HC vehicle control group for each set of treatments.
ATP Content
For measurement of ATP levels as a representation of overall cell viability, the CellTiter-Glo® Luminescent Cell Viability Assay was performed on cell cultures maintained with different medium conditions according to manufacturer's instructions and using an ATP standard curve (Promega). Plates were analyzed using a SpectroMax M5 plate reader and using SoftMax Pro software. Values are reported as micromolar ATP concentrations.
Screening With Test Compounds
After a 24-h post-plating acclimation period in MCM with 1 μM HC, HKCC or corresponding monocultures of hepatocytes were treated with several compounds associated with liver injury over a range of concentrations in the presence or absence of LPS (1 μg/mL) for an additional 48 h. Effects on cytokine production (IL-6 and TNF-α) and Cyp3A activity were determined as described in previous sections. Compounds tested were TVX, APAP, triclosan (TCS), acetochlor, propiconazole, and AA.
Data Analysis
Each treatment condition was performed in a minimum of 2 replicate culture wells. The average or mean ± SD was calculated from the data for each treatment group unless otherwise specified. Statistical tests and significance levels used are described in individual figure legends where applicable.
Results
Establishment of HKCC Model With Donor-Matched Hepatocytes and KCs
The main goal of this project was to extend and expand on our previous efforts to develop a robust co-culture model of primary rat hepatocytes and liver-derived macrophages (KCs) that would ultimately lead to a corresponding human cell-based system for drug and chemical screening purposes.16 In this work, we developed novel methods to produce donor-matched primary hepatocyte and KC fractions from the same liver tissue that, under optimized culture conditions, allow an extended compound exposure period compared with our previous culture system.18-20 The inherent sequence of events during the isolation process allowed for the rapid preparation of hepatocyte fractions first, which were preplated at the appropriate density and allowed to attach while the NPC fractions were processed using modified density-gradient and elutriation centrifugation methods.
Confirmation of the identity and purity of the KC fractions was performed using a combination of flow cytometry and immunofluorescence microscopy. The isolated cells were stained for CD68 and CD163, which indicate monocyte lineage and mature macrophage phenotype, respectively, and purity was assessed using flow cytometry (Fig. 1). Flow analysis confirmed the enrichment of KCs (>90%) with very small amounts of contaminating cell types (Fig. 1d). The ratio of KCs and hepatocytes was previously optimized as part of the development of a rodent-based co-culture system with the goal to achieve maximum cytokine production after treatment with LPS.16 Phase-contrast and fluorescent images of HKCCs illustrated the localization of KCs relative to hepatocytes in these co-cultures (Supplementary Fig. 1).16 KCs were well distributed around and among the hepatocyte chords that formed as a result of the seeding conditions described in the Materials and Methods section. LPS treatment did not appear to have an impact on the localization or distribution of the KCs among the hepatocytes (Supplementary Figs. 1b,1d).
Figure 1.
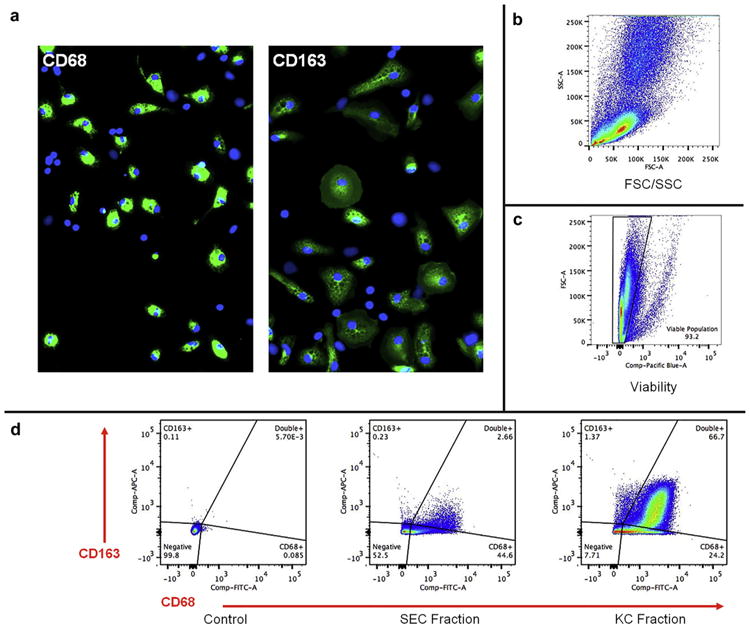
Flow cytometric analysis of freshly isolated rat NPC- and KC-containing fractions. (a) Representative fluorescent images of cells analyzed by flow cytometry and counterstained with antibodies against CD68 and CD163 for visual verification of cells with macrophage lineage (i.e., KCs). Original magnification, 100×. (b) FSC/SSC plot representing entire population present after elutriating liver cells for KC. Counts were recorded for 100,000 events within the population of interest. All cells are shown in FSC/SSC to ensure maximum collection of KC during elutriation. (c) Cell viability is represented by dye exclusion. Only viable cell populations were used for subsequent cell marker analysis. (d) Flow analysis of cell fractions using CD68 and CD163 as markers of cell lineage and relative fraction purity with corresponding gating strategy. Representative plots for unstained control sample, SEC fraction, and KC fraction are shown. Quadrants display cells that are both CD68− and CD163−, single CD68+, single CD163+, and double CD68+/CD163+. Data are representative of 3 separate elutriations. SEC, sinusoidal endothelial cells.
Effect of Medium Formulation on HKCC Integrity and Viability
As part of our model development strategy, we further explored the effects of 4 commercially available cell maintenance media (ADMEM, MCM, WEM, or Williams' E-based HMM) on HKCC viability and morphologic integrity over a 72-h period in the presence and absence of LPS. In keeping with previous study conditions, no glucocorticoids were used during these media comparison experiments.16 The results showed that MCM maintained the greatest overall ATP levels in HKCCs on culture day 3, representing contributions from both hepatocytes and KCs, followed by A-DMEM, regardless of LPS stimulation (Figs. 2a and 2b). Corresponding photomicrographs depicting the unstimulated HKCCs at 72 hours confirmed that the overall health and integrity of the co-cultures were best supported using MCM (Supplementary Fig. 2). As a result of these experiments, all subsequent experiments were performed using glucocorticoid-free MCM as the basal maintenance medium formulation.
Figure 2.
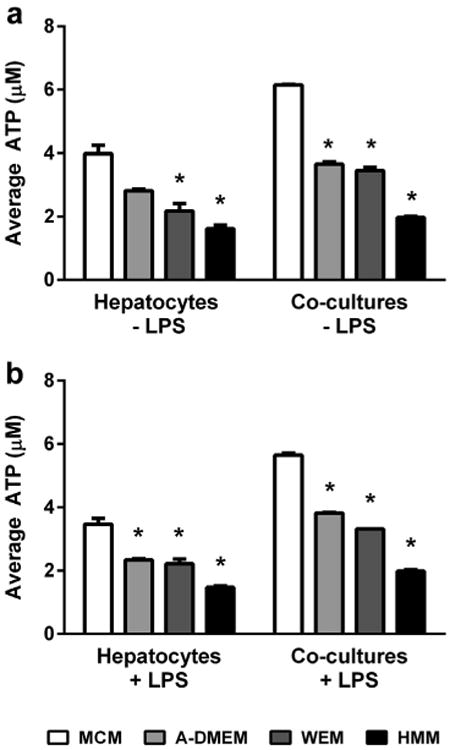
Effect of maintenance medium formulation on intracellular ATP levels in HKCCs. Hepatocyte monocultures and HKCC were cultured for 48 h with or without LPS in 4 different types of maintenance media. (a) Cell viability across medium formulations without LPS stimulation. (b) Cell viability across medium formulations with LPS stimulation. An “*” denotes a significant difference between ATP values for a given medium and those for MCM, after a Bonferroni post-test (p < 0.001).
Effects of Glucocorticoids on LPS-Induced Responses
To develop a more physiologically relevant medium formulation that included glucocorticoids to support native hepatocyte metabolic function, the impact of Dex or HC exposure on cell morphology and LPS-induced inflammatory cytokine responses in HKCCs was explored. Initial experiments were performed using Dex on account of its prevalent use and acceptance as a potent synthetic glucocorticoid for hepatocyte cell culture experimentation in the pharmaceutical and chemical industries. The impact of Dex on HKCC monolayer integrity and cytokine response was investigated after brief exposure (2 h) to medium containing 1 μM Dex during the post-isolation hepatocyte and KC attachment phase. As has previously been shown, the presence of 1 μM Dex enhanced the overall appearance of the monolayers and the cuboidal architecture of the hepatocytes (data not shown).18,23 However, the LPS-induced production of TNF-α was reduced by approximately 80% from no LPS levels, and IL-6 was completely eradicated in Dex-treated HKCCs (Figs. 3a and 3c).
Figure 3.
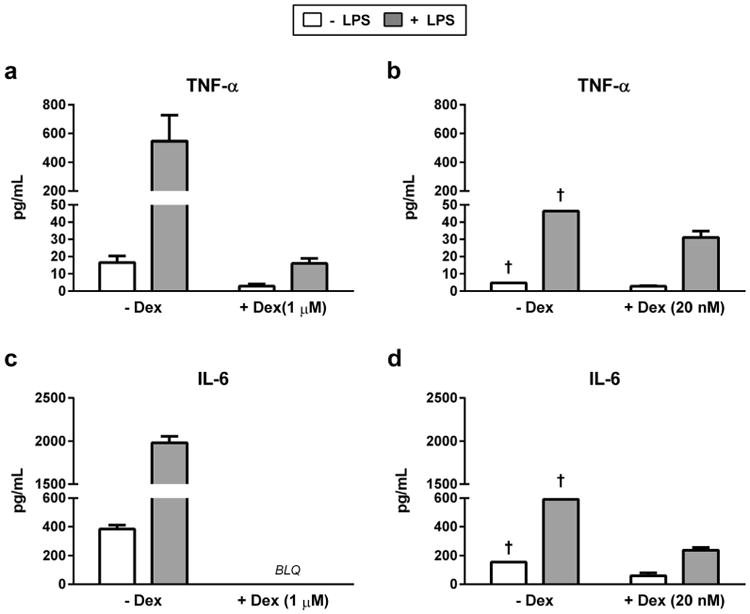
Effects of Dex supplementation in hepatocyte and KC plating medium on HKCC cytokine release. TNF-α and IL-6 release from HKCCs in which Heps and KCs were plated in media containing Dex (a and c, 1 μM; b and d, 20 nM) (†, values from biologic singlet wells). Significance of Dex treatment on cytokine release in LPS-stimulated HKCC was determined by 2-tailed Student's t-test (****p < 0.0001; ***p < 0.001; **p < 0.01). BLQ, below limit of quantification.
We further explored the tenacity of the Dex-mediated effects on LPS-induced cytokine response by exposing only the hepatocytes during the attachment step to an even reduced amount of Dex (20 nM). Upon hepatocyte attachment, the Dex-containing medium was removed and cultures were washed with fresh Dex-free medium before addition of KCs to the hepatocyte monolayers. Photomicrographs of HKCCs with and without 20 nM Dex in the plating medium during hepatocyte and KC attachment showed minor effects on cell morphology after day 1(Supplementary Figs.3a and 3c). However, there were marked differences in the overall monolayer integrity and cuboidal structure of the hepatocytes by day 3 in the absence of Dex (Supplementary Fig. 3b). LPS-induced TNF-α levels were reduced approximately 33% of those without Dex, whereas IL-6 production was reduced approximately 70% (Figs. 3b and 3d). The results from an expanded list of cytokines as represented in Table 1 confirmed that the major pro-inflammatory cytokines, for example, IL-6, IL-1α, IL-12, granulocyte-macrophage colony-stimulating factor, and IFN-γ, were drastically reduced, whereas the anti-inflammatory cytokines, for example, IL-10 and IL-4, were minimally affected. The combined results from these experiments indicated that Dex would not be a suitable glucocorticoid supplement for subsequent use in experiments to investigate compound-induced effects on adaptive immune responses, even if only used at low levels during the cell attachment stage.
Table 1. Effects of Dexamethasone (20 nM) in Hepatocyte Plating Medium on Subsequent HKCC Cytokine Release.
| No LPS Activation | Analyte (pg/mL) | LPS Activation | ||
|---|---|---|---|---|
|
|
|
|||
| −Dex | +Dex | −Dex | +Dex | |
| 16.6 ± 6.5 | 3.0 ± 2.0a | TNF-α | 547.65 ± 311.3 | 16.0 ± 5.2 |
| 384.25 ± 47.6 | ND | IL-6 | 1981.1 ± 130.9 | ND |
| 5.7 ± 0.9a | 6.6 ± 1.4a | GM-CSF | 115.9 ± 12.1 | ND |
| 1.0 ± 3.8a | ND | INF-γ | 12.2 ± 34.4 | ND |
| ND | ND | IL-1α | 148.78 ± 38.0 | ND |
| 20.3 ± 1.4a | 6.4 ± 3.1a | IL-1β | 135.1 ± 26.1 | 7.0 ± 0.7a |
| 93.2 ± 2.1 | 41.6 ± 0.9a | IL-2 | 381.5 ± 11.6 | 48.4 ± 1.4a |
| 0.4 ± 1.3a | 0.4 ± 0.5a | IL-4 | 0.4 ± 0.9a | 0.4 ± 0.6a |
| 55.2 ± 1.3a | 65.6 ± 2.7a | IL-10 | 162.83 ± 5.1 | 117.3 ± 2.1 |
| 23.3 ± 1.1a | 22.0 ± 0.8a | IL-12 | 203.1 ± 8.9 | 6.0 ± 0.5a |
GM-CSF, granulocyte-macrophage colony-stimulating factor; INF-g, interferon gamma; ND, not detected.
Indicates that a value was extrapolated below the linear range of the standard curve for a given cytokine.
Hydrocortisone as a Substitute Glucocorticoid in HKCCs for Immune-Based Toxicity Testing
Given the tenacity of Dex pretreatment on cytokine response to LPS exposure in HKCCs, we explored whether brief exposure to low concentrations of HC, a glucocorticoid with lower in vitro stability and potency, would serve as a suitable substitute. For these experiments, hepatocytes and KCs were seeded in attachment medium containing 1 μM HC, which was subsequently replaced with HC-free maintenance medium. Cultures were then treated with TVX over a range of concentrations (0-200 μM) known to cause differential effects on cytokine profiles and cytotoxicity in the presence of LPS after a 48-h exposure period.24,25 As a negative control, the experiments were run in parallel using HKCCs treated with the nontoxic analog LVX with or without LPS. As anticipated, the results showed statistically significant increases in both TNF-α and IL-6 production levels in LPS-activated HKCCs compared with those that were not (Figs. 4a and 4b). TVX caused decreased ATP levels with LPS co-treatment (Fig. 4c), whereas no significant decreases in ATP levels were observed in LVX-treated HKCCs under any condition (Fig. 4d). TVX produced concentration-dependent increases in TNF-α production (Fig. 4a) and corresponding decreases in IL-6 production (Fig. 4b) at 22 μM before overt toxicity at the highest concentration tested (200 μM). TVX and LPS co-exposure significantly affected both TNF-α and IL-6 production (interaction term p < 0.0001). LPS and LVX co-exposure caused significant increases in TNF-α that were independent of LVX concentration (Fig. 4a). In contrast, LPS and LVX interact (interaction term p < 0.05) to increase IL-6 at 66 μM with respect to LVX vehicle control. These data recapitulate previous data collected from rat co-culture systems without HC.16
Figure 4.
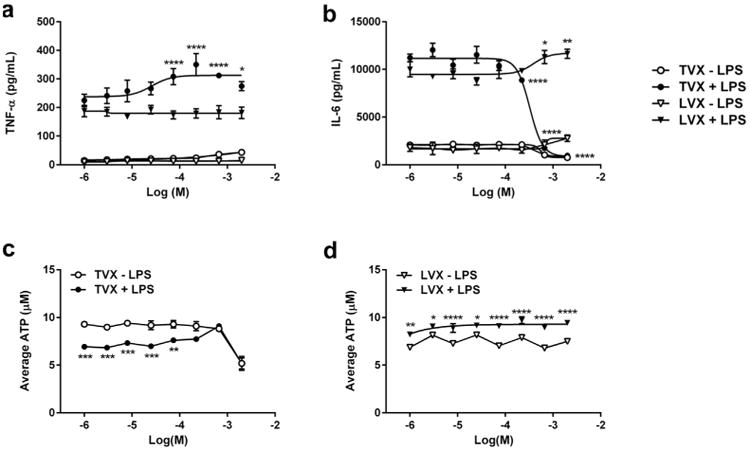
Validation of immune-mediated biology in HKCC with prototypical positive and negative control compounds. HKCCs were exposed to a concentration range (0-200 μm) of TVX or LVX in the presence and absence of LPS. Levels of TNF-α (a) and IL-6 (b) in collected medium samples were assayed by ELISA after 48 h of compound exposure. Combined effects of LPS and compound were analyzed for each cytokine by 2-way ANOVA with Dunnett's post-test and compared with compound vehicle control (*p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001). Matching data for levels of ATP in HKCC after treatment with TVX (c) or with LVX (d) in the presence or absence of LPS found that relative to no LPS control, LPS significantly increases ATP levels for LVX and significantly decreases ATP for low TVX concentrations. ATP statistics were performed by 2-way ANOVA with Bonferroni's post-test to compare no LPS to LPS (*p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001).
As a result of these initial outcomes, we further expanded on these findings by exploring the effects of low levels of HC on Cyp3A metabolic activity and cytokine levels in LPS-activated HKCCs. Inflammatory cytokines, such as IL-6, are known to reduce the drug-metabolizing capacity of the liver in both animal models and isolated primary hepatocytes.26-28 Therefore, cells were exposed to either 1 or 10 μM HC during attachment (∼2 h) or continuously for the first 24 h in culture, followed by treatment for an additional 48 h with LPS and TVX (50 μM), which causes well-defined alterations in IL-6 and Cyp3A activity levels without overt hepatocellular injury in the previous experiments. Under all HC treatment conditions tested, the expected suppression of Cyp3A activity was observed in the absence of TVX as a result of the corresponding increases in IL-6 and TNF-α production in LPS-activated HKCCs (Fig. 5). Moreover, the corresponding reversal of this suppressive effect was observed in activated co-cultures treated with 50 μM TVX regardless of the HC concentration or exposure period. These outcomes were further confirmed by the concomitant LPS-induced changes in the proinflammatory cytokine profiles of IL-6 and TNF-α in the presence and absence of TVX (Figs. 5b, 5c, 5e, and 5f).
Figure 5.
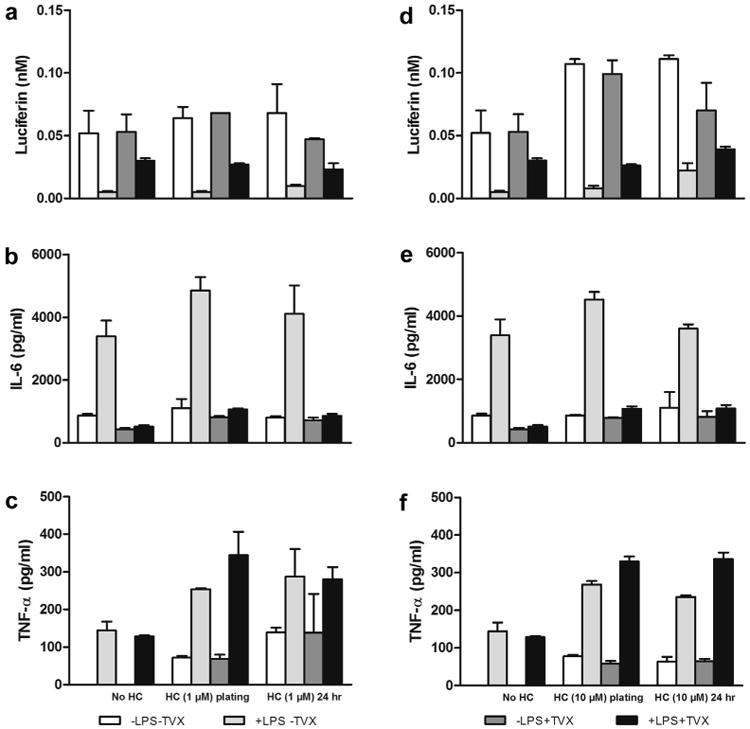
Effects of 1 and 10 μM HC on Cyp3A activity and IL-6/TNF-α levels in HKCCs. HKCCs were exposed to 1 or 10 μM HC briefly (∼2 h) at plating or for 24 h and then were treated with 1 μg/mL LPS, 50 μM TVX, and/or respective controls for 48 h. Cyp3A activity was assessed (a, 1 μM HC; d, 10 μM HC), and media was collected and frozen for IL-6 (b, 1 μM HC; e, 10 μM HC) and TNF-α (c, 1 μM HC; f, 10 μM HC) analyses as described in the Materials and Methods section.
The corresponding morphologic effects of short-term (2 h) versus long-term (24 h) exposure to HC before LPS stimulation were also examined on HKCCs using phase-contrast microscopy. The results indicated that treatment with 1 or 10 μM HC sustained or improved the over morphologic integrity and longevity of the cultures compared with those maintained in HC-free medium over the course of the study period (Supplementary Fig. 4).
HC Effects on LPS- and TVX-Induced Gene Expression in HKCCs
To further elucidate the impact of brief versus prolonged exposure of HKCCs to 1 or 10 μM HC on liver-specific gene expression, we investigated the changes in the mRNA levels of 2 glucocorticoid receptor-dependent genes (Alb and Tat) and 2 oxidative stress and acute phase-response genes (Crp and Nos2). The results, represented as a percentage of respective HC-free controls, indicate that brief exposure to 1 μM HC did not cause significant changes in the expression levels of the 4 genes under any treatment condition (Figs. 6a-6d). However, 24-h exposure to 1 μM HC caused a decrease in the acute-phase response gene Crp in the absence of LPS, whereas it caused an overall decrease in the expression of Nos2 under all treatment conditions (Figs. 6a and 6b). Addition of LPS reversed Crp suppression brought about by24 h of 1 μM HC exposure. HKCCs exhibited a global increase for all 4 genes when exposed to 10 μM HC at plating, regardless of LPS or TVX treatments (Figs. 6e-6h). As seen with 24 h-1 μM HC, LPS-induced Crp expression increased with 24 h of 10 μM HC (Fig. 6e). Another similarity to 24 h of 1 μM HC results is the overall decrease in expression of Nos2 under all treatment conditions when combined with 24 h of 10 μM HC (Fig. 6f). Although no differences in Tat expression were observed between the different 10 μM HC exposure times tested, a trend in the upregulation of Alb was observed in HKCCs exposed to 10 μM HC for 24 h (Figs. 6g and 6h).
Figure 6.
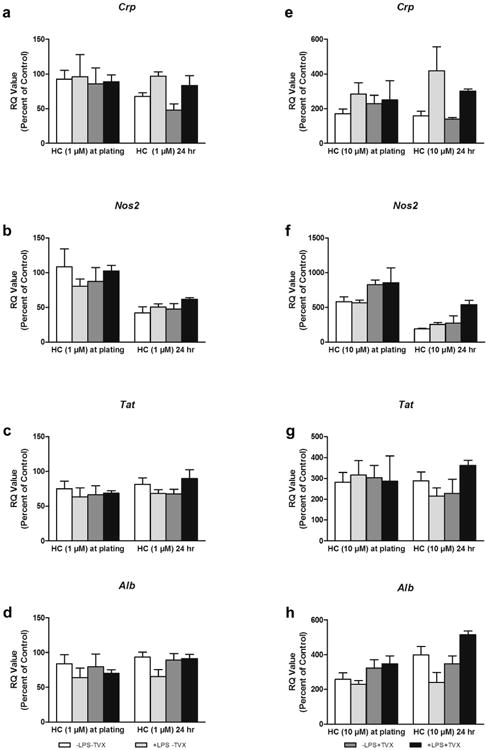
Gene expression in HKCC after exposure to 1 or 10 μM HC. HKCCs were exposed to 1 or 10 μM HC briefly (∼2 h) at plating or for 24 h and then were treated with 1 μg/mL LPS and 50 μM TVX and/or respective controls for 48 h; (a-d) represent levels of gene expression after 1 μM HC exposure and (e-h) after 10 μM HC exposure. Crp expression is shown in (a) and (e), Nos2 in (b) and (f), Tat in (c) and (g), and Alb in (d) and (h).
Immune-Mediated Toxicity Testing Using the Modified HKCC System
Overall, the combined results from the previous experiments demonstrated that HKCCs exposed to HC in the first 24 h of culture exhibited better viability, morphology, and hepatic gene expression profiles while successfully recapitulating the in vitro changes in metabolic function, cytokine profiles, and hepatotoxic effects known to occur under glucocorticoid-free conditions.16 As a result of these findings, MCM medium supplemented with 1 μM HC for a 24-h period before treatment with LPS was selected as our system of choice for screening additional compounds for immune-mediated changes in cytokine profiles and hepatocellular functions. For these experiments,a subset of compounds with known associations with liver injury and inflammation was selected from a list of hepatotoxic compounds published previously.29 After a 24-h post-plating acclimation period, monocultures of hepatocytes and matching HKCCs were treated with compounds over a range of concentrations in the presence or absence of LPS to assess effects on cytokine production and metabolic (Cyp3A) activity. These experiments were designed to show the impact of KC activation in the presence of LPS on the concentration-dependent effects of 6 hepatotoxic compounds (Fig. 8). Matching monocultures of hepatocytes alone (Fig. 7) were treated in an identical fashion to discriminate between intrinsic compound-dependent hepatocellular injury in the absence of a KC-mediated inflammatory response.
Figure 8.
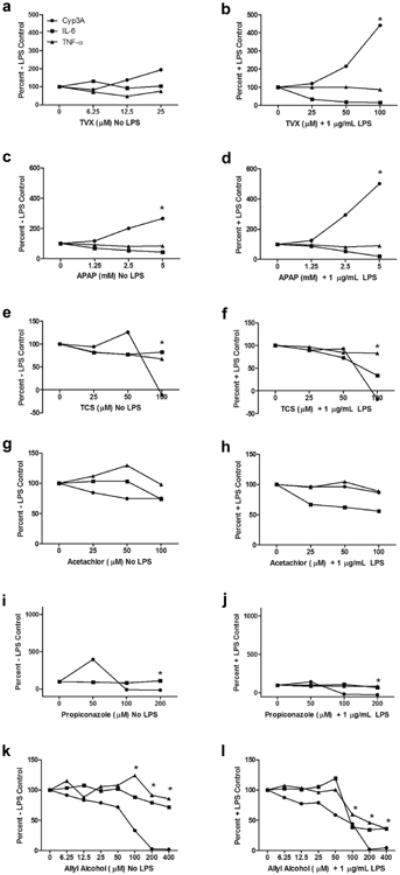
Effects of known hepatotoxic compounds on Cyp3A activity and IL-6/TNF-α levels in HKCCs with or without LPS; data expressed as % of +/− LPS controls. HKCCs were treated with varying doses of TVX (a, b), APAP(c, d), TCS (e, f), acetochlor (g, h), propiconazole(ij), orAA(k, l) with or without 1 μg/mL LPS for 48 h. Cyp3A activity was assessed and media was collected and frozen for IL-6 and TNF-α analyses as described in the Materials and Methods section. Data are representative of a single experiment and expressed as a percentage of the respective LPS-free or LPS-containing vehicle controls as shown on the y-axis. “*,” Indicates toxicity evident in photomicrographs or ATP studies for this compound (data not shown).
Figure 7.
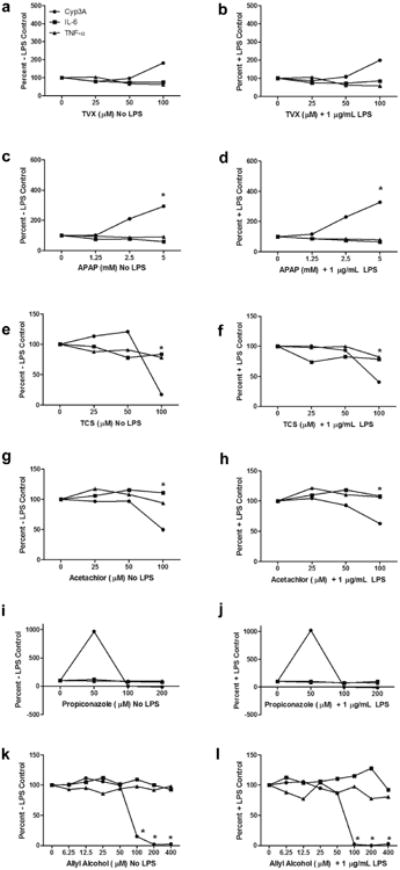
Effects of known hepatotoxic compounds on Cyp3A activity and IL-6/TNF-α levels in monocultures of hepatocytes with or without LPS; data expressed as % +/− LPS controls; 24 h post-plating, hepatocytes were treated with varying doses of TVX (a, b), APAP (c, d), TCS (e, f), acetochlor (g, h), propiconazole (i, j), or AA (k, l) with or without 1 μg/mL LPS for 48 h. Cyp3A activity was assessed, and media was collected and frozen for IL-6 and TNF-α analyses as described in the Materials and Methods section. Data are representative of a single experiment and expressed as a percentage of the respective LPS-free or LPS-containing vehicle controls as shown on the y-axis. *Toxicity evident in photomicrographs or ATP studies for this compound (data not shown).
The results are represented in Figures 7 (monocultures) and 8 (co-cultures) as a percent change in each end point at each concentration compared with the initial vehicle control value from the corresponding groups treated with or without LPS. Corresponding actual values for each end point in monocultures and co-cultures treated with each compound are included as supplementary tables (Supplementary Tables 1 and 2). In monocultures of hepatocytes, TVX caused a 2-fold increase in Cyp3A activity at the highest concentration tested but no net change in cytokine production regardless of LPS treatment (Figs. 7a and 7b). This same overall pattern was observed in HKCCs in the absence of LPS (Fig. 8a). However, in the presence of LPS (i.e., KC activation), TVX caused a concentration-dependent decrease in IL-6 levels that was concomitant with a pronounced approximately 5-fold increase in Cyp3A activity and enhanced hepatotoxicity (at the highest concentration) compared with the other treatment groups (Fig. 8b). Little or no concentration-dependent effects were observed on TNF-α levels. On closer inspection of the absolute values of the end point data for the HKCCs with and without LPS, it was apparent that the activation of KCs in the vehicle control HKCCs treated with LPS caused a 50% reduction of Cyp3A activity concomitant with a marked increase in both IL-6 and TNF-α levels (Supplementary Table 2A). The data also showed that the TVX-mediated increase in the Cyp3A activity correlated with a concentration-dependent decrease in IL-6 production without effect on the corresponding production of TNF-α.
APAP also caused a concentration-dependent increase (∼3-fold) in Cyp3A activity in monocultures of hepatocytes relative to the vehicle controls without a major change in the corresponding cytokine levels regardless of LPS treatment (Figs. 7c and 7d). This pattern of effects also occurred on Cyp3A and cytokine profiles in APAP-treated HKCCs in the absence of LPS (Fig. 8c). In APAP-treated HKCCs in the presence of LPS, there was a concomitant concentration-dependent reduction in IL-6 levels and >5-fold change in Cyp3A activity at the highest concentration without a corresponding effect on TNF-α levels (Fig. 8d). The shift in Cyp3A activity was again associated with the reversal of IL6-mediated suppression of Cyp3A similar to what was observed with TVX (Supplementary Table 2A).
TCS caused no significant changes in Cyp3A activity or cytokine levels in monocultures of hepatocytes regardless of LPS treatment before observing overt toxicity, which occurred at the highest concentration tested (100 μM) (Figs. 7e and 7f). In HKCCs treated with LPS, TCS also did not cause a significant change in Cyp3A activity or cytokine levels at subtoxic concentrations. A slight reduction in IL-6 levels was observed in the presence of LPS, but it was less pronounced compared with TVX and did not result in reversing the IL-6 suppression of Cyp3A activity levels (Supplementary Table 2A). Acetochlor also did not cause marked changes in Cyp3A activity or cytokine levels in monocultures of hepatocytes relative to vehicle controls before overt toxicity, which also occurred at the highest concentration tested (100 μM) (Figs. 7g and 7h). In HKCCs, acetochlor caused a slight increase in TNF-α which resulted in a slight decrease in Cyp3A activity but no net effect on IL-6 levels until the highest concentration (Fig. 8g). In contrast, it caused a reduction in IL-6 levels in HKCCs treated with LPS, without concomitant effects on TNF-α or Cyp3A (Fig. 8g), which was not observed in corresponding monocultures of hepatocytes (Supplementary Tables 1 and 2). Notably, the overt cytotoxicity observed in acetochlor-treated monocultures at the highest concentration was attenuated in HKCCs (data not shown).
Propiconazole treatment of monocultures caused a large ∼10-fold increase in Cyp3A activity at the lowest concentration tested, followed by a complete inhibition of Cyp3A activity at the higher concentrations (Figs. 7i and 7j). In contrast, it had minimal effects on cytokine levels that were related to the LPS treatment (Supplementary Table 1A). Propiconazole-treated HKCCs in the absence of LPS exhibited a reduced 4-fold increase in Cyp3A activity at the lowest concentration followed by complete inhibition of metabolic activity at the higher concentrations (Fig. 8i; Supplementary Table 2A). However, the induction of Cyp3A activity at the lower concentration was nearly completely attenuated in LPS-activated HKCCs (Fig. 8j). Little or no effects were observed on the cytokine levels in HKCCs regardless of the LPS treatment with the exception of LPS-treated co-cultures at the highest propiconazole concentration tested, where reduced IL-6, but not TNF-α, and enhanced cytotoxicity were observed (Supplementary Table 2A).
AA caused little or no changes in IL-6 or TNF-α levels in monocultures of hepatocytes in the absence of LPS (Fig. 7k). However, there was a slight concentration-dependent increase in IL-6 in LPS-treated monocultures (Fig. 7l). Regardless of the LPS treatment, a marked reduction in Cyp3A activity was observed in monocultures at concentrations ≥100 μM, which coincided with the onset of overt cytotoxicity (Figs. 7k and 7l; Supplementary Table 1B). Treatment of HKCCs with AA caused more pronounced changes in cytokine levels in the presence of LPS at concentrations ≥100 μM (Figs. 8k and 8l). Notably, there were more pronounced concentration-dependent decreases in Cyp3A activity at the lower concentrations of AA in HKCCs compared with monocultures but less pronounced decreases in Cyp3A activity at higher AA concentrations (Figs. 8k and 8l). On closer inspection of the separate end point values, AA caused a concentration-dependent decrease in Cyp3A activity that was independent of LPS treatment (Supplementary Table 2B).
Discussion
Drug- and chemical-induced hepatotoxicity continues to be a major concern during development and compound prioritization in the pharmaceutical and agrochemical industries, despite the scientific advancements in our understanding of the clearance, disposition, and exposure for most compounds. Many hepatotoxic responses are caused or exacerbated by an immune system component, which cannot be mimicked in simple monocultures of hepatocytes.3,6 More advanced, multicellular models incorporating both primary adult hepatocytes and resident immune cells, i.e., KCs, recapitulate the interaction between parenchymal and immune cells under controlled conditions.30 LPS- and treatment-mediated KC activation also has been shown to contribute to a number of adverse effects produced by hepatotoxic compounds.21,30,31 Hypotheses for the key molecular events and cellular pathways that may be involved in the mechanisms of action and progression of the hepatotoxic effects in vivo have been elegantly described and illustrated previously.10,32 Accordingly, the direct and indirect effects of compounds on important cell–cell interactions, shifts in cytokine levels, and changes in metabolic clearance can only be examined more systematically in vitro using more sophisticated co-culture systems that incorporate the key cell types that possess biochemical pathways.
In a previous study, we described an in vitro co-culture model using rat primary hepatocytes and cryopreserved KCs.16 We established conditions that facilitated short-term culture of both cell types while maintaining responsiveness to LPS activation and TVX-dependent hepatocellular injury. TVX is one of the most highly studied and best described examples of immune-mediated enhanced hepatotoxicity.33,34 Previous work has shown that TNF-α is a key mediator of TVX effects on caspase-dependent hepatocellular injury by direct effects on resident macrophages (e.g., KCs) and increasing its biosynthesis and slowing its elimination.32,35 LVX, a structural analog of TVX, does not exhibit the direct effects on KCs nor the corresponding hepatotoxic properties in vivo or in vitro.
In our previous work, we used KCs from a commercial source (Life Technologies/Thermo Fisher) and avoided the use of glucocorticoids due to their known anti-inflammatory effects. As an extension of our previous work, our goal was to establish methods of isolating donor-matched hepatocytes and KCs from the same tissue specimen to establish a model for studying the role of innate and adaptive immunity in idiosyncratic drug-induced liver injury (IDILI). The ability to isolate and culture donor-matched cell types also would allow studies to be performed using the matched cell types from knock-out animals and specific strains from murine genetic diversity panels.15,36 Moreover, we are exploring the genetic variability that accounts for the idiosyncratic drug-induced liver injury and its severity in some patient populations.2 To conduct these studies, it is important to eliminate any artifactual immune response that could be due to differences in the human leukocyte antigen system and major histocompatibility complex proteins or mismatches across donors. This becomes even more important when using inbred strains of rodents or precious human tissue samples from patients that have very rare or unique genetic backgrounds. In addition to having donor-matched liver cells, we also intended to identify a more suitable medium formulation incorporating low levels of glucocorticoids, if possible, to prolong the useful life span of HKCCs and to better understand drug-induced hepatotoxic events under more physiologic conditions.
To isolate liver cells from the same tissue, we combined methods from the literature that had been originally designed to isolate individual cell types and modified them until we were able to obtain suitable quantities of hepatocytes and KCs with acceptable purity and viability.18-20 As demonstrated in Figure 1, we were able to obtain relatively pure populations of KCs that exhibited >90% enrichment of macrophage-specific markers, CD68 and CD163. Further examination by phase-contrast and immunofluorescence microscopy confirmed the presence of KCs that stained positive for these same markers. We also established a protocol for plating the hepatocytes and KCs sequentially, which followed the natural progression of the isolation procedure, such that the hepatocytes were isolated first and allowed to attach for 45-60 min, whereas the KCs were purified from the NPC-enriched supernatant fractions. Freshly isolated KCs were then added to the attached hepatocyte monolayer, after which they became integrated in between individual hepatocytes, as illustrated in Figure 2.
Following the establishment of our isolation protocols, we explored different formulations of culture media that had been used for the maintenance of primary hepatocytes.18,23 The results from the medium experiments indicated that MCM provided greater support for optimal co-culture morphology and viability over a 72-h culture period, which was deemed suitable for investigating compound effects on immune-related responses and hepatocellular injury. Notably, MCM had been identified previously as a “preferred” medium for maintaining primary hepatocyte function and viability over longer culture periods, but this is the first report to show evidence of its beneficial effects on maintenance of HKCC monolayer integrity and functionality.17,23
We then attempted to identify suitable conditions under which we could incorporate glucocorticoids to sustain hepatocyte gene expression and metabolic capacity, while retaining the HKCC cytokine response to LPS.17,30 Our initial experiments confirmed that the use of potent and stable glucocorticoids, such as Dex, completely abolish the activation of KC by LPS in both KC monocultures and co-cultures even after limited exposure during the initial attachment phase. However, subsequent results suggest that HKCCs, exposed to low levels of HC (1 μM) for up to 24 h after plating, could still respond normally to LPS treatment, recapitulating key events such as TNF-α and IL-6 cytokine production and concentration-dependent TVX toxicity. Importantly, cytokine production directly resulted in the downregulation of CYP activity, which was reversed by TVX treatment, a benchmark result from a prototype compound that is known to elicit adverse effects in vitro and in vivo, especially under inflammatory conditions.10,12,16,25 Moreover, gene expression levels of liver-specific and oxidative stress and acute phase-response genes were maintained at higher levels in HKCCs with HC pretreatment compared with those without it.
After establishing the culture conditions for maintaining cell longevity and improved hepatocyte functionality while retaining LPS responsiveness, a subset of known hepatotoxicants was tested using the modified HKCC model system.29,37 The compounds were tested over a 48-h exposure period at several concentrations that were relevant to their exposure in vivo or that had been shown to cause hepatocellular injury in vitro. We then examined their immune-mediated (LPS induced) and direct effects (without LPS) on the production of key cytokines, namely TNF-α and IL-6, and metabolic capacity as represented by Cyp3A activity. Cytotoxicity was monitored either visually by phase-contrast microscopy and/or biochemically by measuring changes in intracellular ATP levels.
First, the data confirmed the fidelity of the modified HKCC model to recapitulate the key events exhibited by the benchmark compound TVX, including the concentration-dependent alteration in IL-6/TNF-α ratios and corresponding shift in Cyp3A suppression that has been described previously.16 HKCCs treated with APAP also exhibited a similar trend in LPS-mediated effects on cytokine profiles and CYP activity. In addition, APAP caused a greater cellular ATP decrease in LPS-treated HKCCs relative to co-cultures not exposed to LPS, which was similar to what was observed with TVX (data not shown). Notably, APAP is another compound known to be associated with immune-mediated enhanced hepatotoxic events leading to severe liver injury and liver failure in some individuals.37 Although it is well established in the literature that APAP is metabolically activated to a reactive metabolite (N-acetyl-p-benzoquinone imine), which depletes glutathione as a key initiating event leading to oxidative stress and hepatocellular injury, our results provide confirmation that there may be secondary pathways involved in its hepatotoxic mechanism, such as changes in cytokine profiles and metabolic capacity, that may be responsible for enhancement of toxicity in LPS-activated HKCCs, outside the classical APAP-induced cytotoxic effects from N-acetyl-p-benzoquinone imine, formation. Notably, the “idiosyncratic” nature of the severity of APAP toxicity in some patients has been linked to the patient's adaptive immune responses, suggesting that it too may fall into a category of a “TVX-like” mechanism.15,37
Other hepatotoxic compounds that were tested using the enhanced HKCC system exhibited unique trends in their effects on either cytokine profiles and/or CYP activity that seemed to lead to exacerbated or attenuated cytotoxicity. For the most part, the compound-induced effects observed in the HKCC model system are likely to be a reflection of their mode of action on KC activation and/or hepatocellular functions. For example, it has been shown that TCS causes LPS-independent perturbations in thyroxine disposition.38 Chloroacetanilide pesticides, such as acetochlor and alachlor, exhibit metabolism-dependent hepatotoxicity.29,39 Many antifungals, such as propiconazole, are known to be potent inhibitors and inducers of liver CYP enzymes and nongenotoxic tumor promoters, events which are exacerbated by inflammatory responses.40 AA has been shown to exhibit selective hepatocellular damage around the periportal region that was shown to be related to localized gradients in oxygen and inflammatory responses.41
Taken together, these results suggest that the HKCC model could provide additional information and insights into the innate potential of compounds in development to cause hepatocellular injury and toxicity. We propose that the HKCC model system could potentially be used to stratify toxic compounds into subcategories of immune-mediated hepatotoxic responses as observed in this study, for example, (1) compounds that caused KC-mediated decreases in IL-6 levels with concomitant metabolic activity leading to enhanced cytotoxicity (e.g., TVX, APAP); (2) compounds that caused hepatocellular effects and cytotoxicity that were independent of inflammatory mediators (e.g., TCS); (3) compounds exhibiting KC-mediated attenuation of hepatocellular effects and cytotoxicity (e.g., acetochlor, AA); and (4)compoundsthatcausemarked changes in metabolic capacity (i.e., induction and inhibition of Cyp3A), with enhanced KC-mediated cytotoxicity in the absence of distinct cytokine effects (e.g., propiconazole). Moreover, our findings underscore the need for more biologically relevant, multicellular culture models to study these complex biologic and toxicologic events. Further research is warranted to explore the mechanisms of the LPS-enhanced and KC-mediated secondary and adaptive events leading to enhanced or diminished hepatocellular injury.
Conclusions
We have demonstrated the utility of an optimized donor-matched HKCC model for exploring possible mechanisms of immune-mediated drug responses. Our HKCC system responds to prototype effector molecules of inflammation and toxicity and corresponds closely with results observed previously in vitro and in vivo for prototype hepatotoxic compounds. Application of this model system to screening a limited set of compounds associated with liver injury indicated that APAP displayed enhanced immune-mediated effects similar to that of TVX, a known IDILI compound. Other compounds had unique direct or indirect effects on cytokine levels and metabolic capacity that appeared to reflect known in vitro and in vivo effects or modes of action.29,37,42,43 Overall, these findings support the use of HKCCs, with proper medium formulation, as a valuable in vitro tool to evaluate and stratify compounds with hepatotoxic liability that would otherwise be overlooked using hepatocytes alone. Moreover, this novel approach can be extended to development of a human-based co-culture model to explore population diversity in innate immunity and its role in IDILI.
Supplementary Material
Acknowledgments
This project was supported in part by funding from the Long-Range Research Initiative of the American Chemistry Council, Washington, DC. The authors would also like to acknowledge the contribution and leadership of Dr. Ronald T. Borchardt toward advancing the field of in vitro biomedical and pharmaceutical research, and the ongoing vision, guidance, and support that he has provided over the years to the numerous students and affiliates of the Departments of Pharmaceutical Chemistry and Biochemistry at the University of Kansas.
Footnotes
This article contains supplementary material available from the authors by request or via the Internet at http://dx.doi.org/10.1016/S0022-3549(15)00192-6.
References
- 1.Sunman JA, Hawke RL, LeCluyse EL, Kashuba AD. Kupffer cell-mediated IL-2 suppression of CYP3A activity in human hepatocytes. Drug Metab Dispos. 2004;32(3):359–363. doi: 10.1124/dmd.32.3.359. [DOI] [PubMed] [Google Scholar]
- 2.Corsini A, Ganey P, Ju C, et al. Current challenges and controversies in drug-induced liver injury. Drug Saf. 2012;35(12):1099–1117. doi: 10.1007/BF03261997. [DOI] [PubMed] [Google Scholar]
- 3.Kaplowitz N. Idiosyncratic drug hepatotoxicity. Nat Rev Drug Discov. 2005;4(6):489–499. doi: 10.1038/nrd1750. [DOI] [PubMed] [Google Scholar]
- 4.DeLeve LD, Wang X, Kaplowitz N, Shulman HM, Bart JA, van der Hoek A. Sinusoidal endothelial cells as a target for acetaminophen toxicity Direct action versus requirement for hepatocyte activation in different mouse strains. Biochem Pharmacol. 1997;53(9):1339–1345. doi: 10.1016/s0006-2952(97)00048-8. [DOI] [PubMed] [Google Scholar]
- 5.Kmiec Z. Cooperation of liver cells in health and disease. Adv Anat Embryol Cell Biol. 2001;161:III–XIII. 1–151. doi: 10.1007/978-3-642-56553-3. [DOI] [PubMed] [Google Scholar]
- 6.Evers R, Dallas S, Dickmann LJ, et al. Critical review of preclinical approaches to investigate cytochrome p450-mediated therapeutic protein drug-drug interactions and recommendations for best practices: a white paper. Drug Metab Dispos. 2013;41(9):1598–1609. doi: 10.1124/dmd.113.052225. [DOI] [PubMed] [Google Scholar]
- 7.Liu ZX, Kaplowitz N. Role of innate immunity in acetaminophen-induced hepatotoxicity. Expert Opin Drug Metab Toxicol. 2006;2(4):493–503. doi: 10.1517/17425255.2.4.493. [DOI] [PubMed] [Google Scholar]
- 8.Dixon LJ, Barnes M, Tang H, Pritchard MT, Nagy LE. Kupffer cells in the liver. Compr Physiol. 2013;3(2):785–797. doi: 10.1002/cphy.c120026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kolios G, Valatas V, Kouroumalis E. Role of Kupffer cells in the pathogenesis of liver disease. World J Gastroenterol. 2006;12(46):7413–7420. doi: 10.3748/wjg.v12.i46.7413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roberts RA, Ganey PE, Ju C, Kamendulis LM, Rusyn I, Klaunig JE. Role of the Kupffer cell in mediating hepatic toxicity and carcinogenesis. Toxicol Sci. 2007;96(1):2–15. doi: 10.1093/toxsci/kfl173. [DOI] [PubMed] [Google Scholar]
- 11.Hoebe KH, Witkamp RF, Fink-Gremmels J, Van Miert AS, Monshouwer M. Direct cell-to-cell contact between Kupffer cells and hepatocytes augments endotoxin-induced hepatic injury. Am J Physiol Gastrointest Liver Physiol. 2001;280(4):G720–728. doi: 10.1152/ajpgi.2001.280.4.G720. [DOI] [PubMed] [Google Scholar]
- 12.Morgan ET. Impact of infectious and inflammatory disease on cytochrome P450-mediated drug metabolism and pharmacokinetics. Clin Pharmacol Ther. 2009;85(4):434–438. doi: 10.1038/clpt.2008.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu R, Cui X, Dong W, Zhou M, Simms HH, Wang P. Suppression of hepatocyte CYP1A2 expression by Kupffer cells via AhR pathway: the central role of proinflammatory cytokines. IntJ Mol Med. 2006;18(2):339–346. [PubMed] [Google Scholar]
- 14.Higuchi S, Wu R, Zhou M, Ravikumar TS, Wang P. Downregulation of hepatic cytochrome P-450 isoforms and PPAR-gamma: their role in hepatic injury and proinflammatory responses in a double-hit model of hemorrhage and sepsis. J Surg Res. 2007;137(1):46–52. doi: 10.1016/j.jss.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 15.Harrill AH, Watkins PB, Su S, et al. Mouse population-guided resequencing reveals that variants in CD44 contribute to acetaminophen-induced liver injury in humans. Genome Res. 2009;19(9):1507–1515. doi: 10.1101/gr.090241.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bonzo JA, Rose KA, Freeman K, et al. Differential effects of trovafloxacin on TNF-alpha and IL-6 profiles in a rat hepatocyte-Kupffer cell co-culture system. Appl In Vitro Toxicol. 2015;1(1):45–54. [Google Scholar]
- 17.Waxman DJ, Morrissey JJ, Naik S, Jauregui HO. Phenobarbital induction of cytochromes P-450. High-level long-term responsiveness of primary rat hepatocyte cultures to drug induction, and glucocorticoid dependence of the phenobarbital response. Biochem J. 1990;271(1):113–119. doi: 10.1042/bj2710113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.LeCluyse EL, Bullock PL, Parkinson A, Hochman JH. Cultured rat hepatocytes. Pharm Biotechnol. 1996;8:121–159. doi: 10.1007/978-1-4899-1863-5_9. [DOI] [PubMed] [Google Scholar]
- 19.Valatas V, Xidakis C, Roumpaki H, Kolios G, Kouroumalis EA. Isolation of rat Kupffer cells: a combined methodology for highly purified primary cultures. Cell Biol Int. 2003;27(1):67–73. doi: 10.1016/s1065-6995(02)00249-4. [DOI] [PubMed] [Google Scholar]
- 20.DeLeve LD, Wang X, McCuskey MK, McCuskey RS. Rat liver endothelial cells isolated by anti-CD31 immunomagnetic separation lack fenestrae and sieve plates. Am J Physiol Gastrointest Liver Physiol. 2006;291(6):G1187–1189. doi: 10.1152/ajpgi.00229.2006. [DOI] [PubMed] [Google Scholar]
- 21.Jaeschke H. Kupffer cells. In: Rodes J, Benhamou J, Blei A, Reichen J, Rizzetto M, editors. Textbook of Hepatology: From Basic Science to Clinical Practice. 3rd. Malden: Wiley-Blackwell Publishing; 2007. pp. 36–42. [Google Scholar]
- 22.Hanna RN, Carlin LM, Hubbeling HG, et al. The transcription factor NR4A1 (Nur77) controls bone marrow differentiation and the survival of Ly6C-monocytes. Nat Immunol. 2011;12(8):778–785. doi: 10.1038/ni.2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.LeCluyse E, Bullock P, Madan A, Carroll K, Parkinson A. Influence of extracellular matrix overlay and medium formulation on the induction of cytochrome P-450 2B enzymes in primary cultures of rat hepatocytes. Drug Metab Dispos. 1999;27(8):909–915. [PubMed] [Google Scholar]
- 24.Waring JF, Liguori MJ, Luyendyk JP, et al. Microarray analysis of lipopolysaccharide potentiation of trovafloxacin-induced liver injury in rats suggests a role for proinflammatory chemokines and neutrophils. J Pharmacol Exp Therapeutics. 2006;316(3):1080–1087. doi: 10.1124/jpet.105.096347. [DOI] [PubMed] [Google Scholar]
- 25.Shaw PJ, Hopfensperger MJ, Ganey PE, Roth RA. Lipopolysaccharide and trovafloxacin coexposure in mice causes idiosyncrasy-like liver injury dependent on tumor necrosis factor-alpha. Toxicol Sci. 2007;100(1):259–266. doi: 10.1093/toxsci/kfm218. [DOI] [PubMed] [Google Scholar]
- 26.Abdel-Razzak Z, Loyer P, Fautrel A, et al. Cytokines down-regulate expression of major cytochrome P-450 enzymes in adult human hepatocytes in primary culture. Mol Pharmacol. 1993;44(4):707–715. [PubMed] [Google Scholar]
- 27.Poüs C, Giroud JP, Damais C, Raichvarg D, Chauvelot-Moachon L. Effect of recombinant human interleukin-1 beta and tumor necrosis factor alpha on liver cytochrome P-450 and serum alpha-1-acid glycoprotein concentrations in the rat. Drug Metabol Dispos. 1990;18(4):467–470. [PubMed] [Google Scholar]
- 28.Ashino T, Oguro T, Shioda S, et al. Involvement of interleukin-6 and tumor necrosis factor a in CYP3A11 and 2C29 down-regulation by Bacillus Calmette-Guérin and lipopolysaccharide in mouse liver. Drug Metabol Dispos. 2004;32(7):707–714. doi: 10.1124/dmd.32.7.707. [DOI] [PubMed] [Google Scholar]
- 29.Rodrigues RM, Bouhifd M, Bories G, et al. Assessment of an automated in vitro basal cytotoxicity test system based on metabolically-competent cells. Toxicol In Vitro. 2013;27(2):760–767. doi: 10.1016/j.tiv.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 30.LeCluyse EL, Witek RP, Andersen ME, Powers MJ. Organotypic liver culture models: meeting current challenges in toxicity testing. Crit Rev Toxicol. 2012;42(6):501–548. doi: 10.3109/10408444.2012.682115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jaeschke H, Gores GJ, Cederbaum AI, Hinson JA, Pessayre D, Lemasters JJ. Mechanisms of hepatotoxicity. Toxicol Sci. 2002;65(2):166–176. doi: 10.1093/toxsci/65.2.166. [DOI] [PubMed] [Google Scholar]
- 32.Shaw PJ, Ganey PE, Roth RA. Idiosyncratic drug-induced liver injury and the role of inflammatory stress with an emphasis on an animal model of trovafloxacin hepatotoxicity. Toxicol Sci. 2010;118(1):7–18. doi: 10.1093/toxsci/kfq168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shaw PJ, Ganey PE, Roth RA. Tumor necrosis factor alpha is a proximal mediator of synergistic hepatotoxicity from trovafloxacin/lipopolysaccharide coexposure. J Pharmacol Exp Ther. 2009;328(1):62–68. doi: 10.1124/jpet.108.143792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Beggs KM, Fullerton AM, Miyakawa K, Ganey PE, Roth RA. Molecular mechanisms of hepatocellular apoptosis induced by trovafloxacin-tumor necrosis factor-alpha interaction. Toxicol Sci. 2014;137(1):91–101. doi: 10.1093/toxsci/kft226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Poulsen KL, Albee RP, Ganey PE, Roth RA. Trovafloxacin potentiation of lipopolysaccharide-induced tumor necrosis factor release from RAW 264.7 cells requires extracellular signal-regulated kinase and c-Jun N-terminal kinase. J Pharmacol Exp Ther. 2014;349(2):185–191. doi: 10.1124/jpet.113.211276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Harrill AH, Ross PK, Gatti DM, Threadgill DW, Rusyn I. Population-based discovery of toxicogenomics biomarkers for hepatotoxicity using a laboratory strain diversity panel. Toxicol Sci. 2009;110(1):235–243. doi: 10.1093/toxsci/kfp096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Borlak J, Chatterji B, Londhe KB, Watkins PB. Serum acute phase reactants hallmark healthy individuals at risk for acetaminophen-induced liver injury. Genome Med. 2013;5(9):86. doi: 10.1186/gm493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Paul KB, Hedge JM, Bansal R, et al. Developmental triclosan exposure decreases maternal, fetal, and early neonatal thyroxine: a dynamic and kinetic evaluation of a putative mode-of-action. Toxicology. 2012;300(1-2):31–45. doi: 10.1016/j.tox.2012.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kale VM, Miranda SR, Wilbanks MS, Meyer SA. Comparative cytotoxicity of alachlor, acetochlor, and metolachlor herbicides in isolated rat and cryopreserved human hepatocytes. J Biochem Mol Toxicol. 2008;22(1):41–50. doi: 10.1002/jbt.20213. [DOI] [PubMed] [Google Scholar]
- 40.Wolf DC, Allen JW, George MH, et al. Toxicity profiles in rats treated with tumorigenic and nontumorigenic triazole conazole fungicides: propiconazole, triadimefon, and myclobutanil. Toxicol Pathol. 2006;34(7):895–902. doi: 10.1080/01926230601047808. [DOI] [PubMed] [Google Scholar]
- 41.Przybocki JM, Reuhl KR, Thurman RG, Kauffman FC. Involvement of nonparenchymal cells in oxygen-dependent hepatic injury by allyl alcohol. Toxicol Appl Pharmacol. 1992;115(1):57–63. doi: 10.1016/0041-008x(92)90367-2. [DOI] [PubMed] [Google Scholar]
- 42.Rotroff DM, Beam AL, Dix DJ, et al. Xenobiotic-metabolizing enzyme and transporter gene expression in primary cultures of human hepatocytes modulated by ToxCast chemicals. J Toxicol Environ Health B Crit Rev. 2010;13(2-4):329–346. doi: 10.1080/10937404.2010.483949. [DOI] [PubMed] [Google Scholar]
- 43.Liu J, Mansouri K, Judson RS, et al. Predicting hepatotoxicity using ToxCast in vitro bioactivity and chemical structure. Chem Res Toxicol. 2015;28(4):738–751. doi: 10.1021/tx500501h. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


