Abstract
Renal artery aneurysms (RAAs) and renal arteriovenous fistulas (RAVFs) are rare lesions occurring in less than 1% of the population. Understanding treatment options is important in the care of these patients. Depending on the type of disease process, endovascular techniques, vascular bypass, renal autotransplantation, or nephrectomy may be indicated. The case reported describes a combined RRA and RAVF treated with nephrectomy.
Keywords: aneurysm, renal disease, resection, arteriovenous malformation, fistula, arteriovenous fistula, excision
Renal artery aneurysms (RAAs) and renal arteriovenous fistulas (RAVFs) are rare lesions that may be congenital or acquired.1 2 RAA may be treated conservatively with observation and serial evaluation, or surgically with nephrectomy, stent exclusion, vascular bypass, renal autotransplantation, or transarterial embolization, which is becoming the gold standard.3 4 5 6 The case reported is anRAA with a high-flow renal arteriovenous malformation of the right kidney with ureteral duplication; this was successfully treated with radical nephrectomy after evaluation for a potential endovascular repair.
Case Presentation
A 75-year-old woman with past medical history of hypertension and chronic kidney disease stage III (CKD III) presented to the emergency department with complaints of cough, hypertension, and light-headedness. On presentation, blood pressure was 157/78, heart rate was 107, and the patient was afebrile. Laboratory studies included a basic metabolic panel, complete blood count, urinalysis, and cardiac enzymes; results were significant only for a creatinine of 1.38, which was slightly elevated from her known baseline of 1.2 mg/dL, and blood count and cardiac enzymes were normal. Urinalysis was positive for a small amount of blood but gross hematuria was not present. Physical examination did not reveal any abnormal findings. No abdominal bruit or mass was noted. The patient was evaluated with computed tomography (CT) to rule out a pulmonary embolism (PE). The CT study incidentally demonstrated a 2.1 cm renal anomaly suspected of being an RAA with arteriovenous malformation (Fig. 1). The patient was discharged from the emergency department with instructions to follow up with her primary care physician (PCP) for further workup of the RAA.
Fig. 1.

Computerized tomography with intravenous contrast pulmonary embolism study that incidentally found the renal artery aneurysms (RAA).This is the most inferior slice in the series and the true size of the RAA would likely have been demonstrated on a full abdomen scan.
On outpatient evaluation with the PCP, there was concern of renal malignancy and the patient was evaluated with magnetic resonance imaging (MRI) that further characterized the malformation as a 5.2 cm RAA with anRAVF (Fig. 2). She had no history of abdominal surgery, renal biopsy, abdominal or flank trauma, or other history of vascular disease or intervention to the renal vascular system. Two weeks later, the patient was admitted to the hospital for hypertension and palpitations. It was suspected that the RAVF was responsible for the symptoms and the patient was subsequently taken to the angiography suite for definitive evaluation and possible intervention of the RAVF. Her baseline creatinine was of concern, but preoperative hydration and limited use of contrast during the angiography prevented a rise in creatinine postoperatively (1.3 mg/dLpreangiography and 1.2 mg/dL on postprocedure day 1).
Fig. 2.
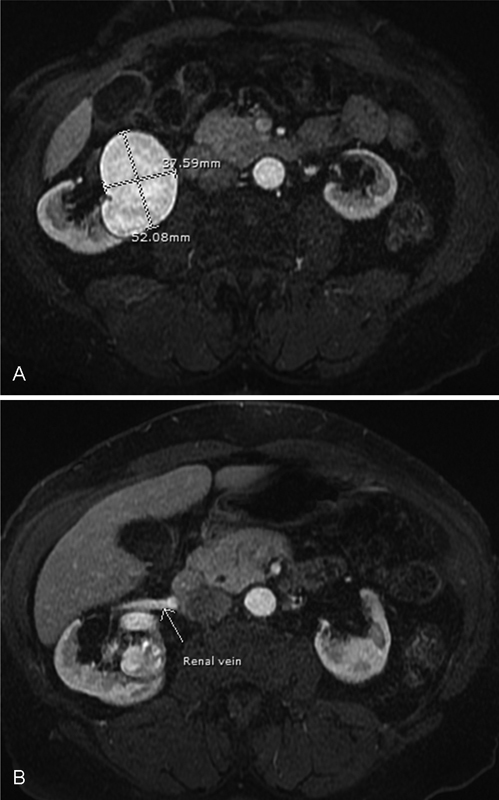
(A) T1 weighted MRI demonstrating a renal artery lesion with isoenhancement of the aorta. (B) T1 weighted MRI demonstrating an early enhancing right renal vein consistent with arteriovenous fistula. The left renal vein did not enhance in the same series of images.
Angiography revealed a dilated renal artery measuring up to 14mm with high output RAVF with direct flow from the renal artery to caval system (Fig. 3). Due to the size of the aneurysm and high flow of the RAVF, no endovascular procedures such as embolization or exclusion were attempted as there was concern of emboli migration into the venous system.
Fig. 3.

Aortogram demonstrating markedly enlarged renal artery measuring 14.58 mm with early filling of the renal vein and enhancement of the inferior vena cava within seconds of contrast administration.
The patient was discharged from the hospital stay and followed up with an urologist regarding candidacy for nephrectomy. One month later, the patient underwent a radical right nephrectomy via midline laparotomy. As expected, intraoperatively there was a very large renal aneurysm, and a dilated renal artery and vein consistent with the angiographic findings. Massive bleeding from the high-flow AVF was of concern but limited by first isolating and ligating the renal artery with a vascular stapling device; the renal vein was then separately ligated. The AVF connection was directly between the renal artery and the renal vein, and ligation of these vessels near their origin limited blood loss and isolated the AVF,thus preventing excessive blood loss. Total blood loss for the procedure was approximately 300 mL.
Pathological evaluation showed a 4.6 cm RAA with a direct renal artery to renal vein connection and ureteral duplication. Probing the vessels demonstrated both vessels communicating through the AVF at 4.5 cm from the renal artery resection margin and 2.0 cm from the renal vein resection margin. Microscopically, the specimen exhibited arteriolonephrosclerosis with 20% globally sclerotic glomeruli.
Preoperatively, the patient had a creatinine value of 1.3 mg/dL with an expected elevation postoperatively to 2.06 mg/dL. Recovery was uneventful and the patient was discharged on postoperative day 6 without complication with a creatinine of 2.8 mg/dL.
The patient was seen 6 months later. Her antihypertensive regimen, Lisinopril-Hydrochlorothiazide and Amlodipine, remains unchanged from the preoperative regimen, but the blood pressure is now well controlled in the 110s/70s and she has recovered fully without complication. At 1 year follow-up, her creatinine is 1.64 mg/dL and she remains in CKD III and free of renal replacement therapy.
Discussion
RAA is defined as dilation twice the diameter of a normal renal artery.7 The detection of RAA during angiography has been estimated to be between 0.3 and 0.7%, and in one series it was nearly 1%.8 Symptoms of RAA consist of the following: flank pain, hematuria, renal infarction secondary to embolization from the aneurysm, and uncontrolled hypertension refractory to medical management from associated renal artery stenosis.7 Rundback et al developed an angiographic classification system describing three types of RAAs: type 1 are saccular aneurysms arising from either the main renal artery or from a proximal large segmental artery,type II aneurysms are fusiform and occur at the main renal artery or proximal segmental arteries, andtype III are intraparenchymal aneurysms affecting the small segmental or accessory arteries.5 6 7
Indications for treatment of RAAs are rupture, rapid expansion, females who are pregnant or contemplating pregnancy, symptomatic RAA, or diameter >2 cm.5 6 7 Though rupture of RAA is well documented, Tham et al performed an analysis of 36,656 autopsies with a total of 19 ruptured arterial aneurysms discovered, none of which involved the renal artery. They concluded that in a patient with an incidentally detected RAA, surgery cannot be justified if only for the risk of rupture.4 8 However, intervention for RAA is justified if symptomatic or if associated with an RAVF. If an RAA is discovered and the patient has confirmed renovascular hypertension, the aneurysm must be excised and the associated stenosis be surgically treated.7 Treatment options include nephrectomy, vascular bypass, coil embolization, and stent-graft placement. Nephrectomy with ex vivo angioplasty and autotransplantation has also been reported.9 10
Treatment of RAA differs depending on the type of aneurysm utilizing Rundback's classification. Type I aneurysms can be treated with stent grafts or coil embolization.6 Open repair of type I aneurysms includes tangential excision with primary repair or patch angioplasty with autologous vein or graft material.7 Coil embolization involves engagement of the aneurysm with a catheter and deployment of coils with or without n-butyl-2-cyanoacrylate (N-BCA) glue. In reported series, any combination of these two techniques results in a 98% success rate of exclusion with embolization within the aneurysm and no flow through aneurysm after coil deployment.11 However, no series was found demonstrating this technique in the treatment of aneurysms with high-flow AVF. Type II aneurysms are best treated surgically with segmental excision of the aneurysm and arterial reconstruction utilizing an autologous vein or aortorenal bypass graft, preferably with an autologous vein.7 Type III aneurysms, due to their intraparenchymal location, are best treated with coil embolization.6 7 Any type of RAA may be treated with nephrectomy if the above options fail or are not feasible. After the treatment of RAAs, it is generally accepted to follow the patient with ultrasound initially at 1 month, and then 6 months and yearly after that. Any abnormality may be further classified with CT, MRA, or catheter arteriography.7
Rarely, as in this case, an RAA may be associated with an RAVF. RAVFs account for less than 1% of arteriovenous fistulas.1 Symptoms associated with RAVF have been reported as abdominal bruit, hypertension, cardiomegaly, congestive heart failure, and hematuria.2 There are two types of renal arteriovenous anomalies: cirsoid type, which have multiple small and dilated arteriovenous communications, and aneurysmal type, which consists of a solitary arterial connection with a solitary vein.3 12 Cirsoid-type is considered an arteriovenous malformation and are congenital, whereas aneurysmal type are considered arteriovenous fistulas and may either be idiopathic or secondary. Idiopathic RAVFs develop when pre-existing aneurysms form shunts between adjacent segments of renal artery and vein. Secondary RAVFs occur after an intervention such as a percutaneous renal biopsy or after penetrating flank trauma.2 13 Idiopathic RAVF is presumed to be an eroding RAA into an adjacent vein.2 14 Most RAAs are discovered incidentally and may be further evaluated with duplex ultrasound, spiral computerized tomography, contrast enhanced magnetic resonance angiography, or digital subtraction arteriography.7 Arteriography is the ideal study to evaluate both RAA and RAVFs with the hallmark of RAVFs being visualization of the inferior vena cava within seconds of contrast injection.2 7
Treatment of RAAs usually consists of transcatheter arterial embolization (TAE), but high-flow fistulas may preclude this technique due to the risk of emboli migration into the venous system and resultant PE.2 3 4 Embolic materials for TAE may include gelatin sponge, metallic coil, absolute ethanol, lipiodol, and N-BCA.3 Treatment should be pursued for symptomatic RAVFs or when significant hemodynamic effects are present. The goal of treatment of RAVFs is the maximal preservation of renal function and eradication of hemodynamic effects with minimization of procedural complications. For large aneurysms with associated RAVF, nephrectomy is the most common surgical treatment though ligation of feeding vessels and partial nephrectomy are also viable options.2
The aneurysm reported here was a type I lesion with associated idiopathic aneurysmal arteriovenous fistula. The patient had no history of neither intervention to the renal vasculature nor flank trauma; thus, the RAA and RAVF are either idiopathic or congenital lesions. Type I lesions, as stated above, are usually amenable to surgical repair. However, due to the large size of the RAA and the high-flow characteristics of the RAVF, nephrectomy was determined to be the optimal surgical solution. Pathological examination of the fistulous connection and size of the aneurysm confirmed that embolization was not an option because of the high flow of the fistula and subsequent high risk of emboli migration into the venous system. On follow up at 1 year, the patient's hypertension improved and her renal function has remained stable.
The improvement in blood pressure is hypothesized to be due to a decrease in activation of the renin–angiotensin–aldosterone system (RAAS) after removal of the kidney. The high-flow state through the RAVF likely resulted in the kidney sensing a low-flow state and activated RAAS in an effort to increase local renal perfusion. Once the kidney was removed, the renin levels would have decreased, and the hypertension became more easily controlled with an oral antihypertensive regimen. Though no renal vein renin levels were collected, this hypothesis is the most likely cause of improvement in the patient's hypertension postoperatively.
The case reported here demonstrates the wide array of treatment options available for the rare RAA and RAVF. In the age of endovascular intervention, it is important to evaluate all surgical options and to realize when a minimally invasive approach is and is not appropriate. In this case, coil embolization had an unacceptably high risk of central venous embolization, and nephrectomy was ultimately the appropriate treatment of this patient.
References
- 1.Cura M, Elmerhi F, Suri R, Bugnone A, Dalsaso T. Vascular malformations and arteriovenous fistulas of the kidney. ActaRadiol. 2010;51(2):144–149. doi: 10.3109/02841850903463646. [DOI] [PubMed] [Google Scholar]
- 2.Crotty K L Orihuela E Warren M M Recent advances in the diagnosis and treatment of renal arteriovenous malformations and fistulas J Urol 1993150(5 Pt 1):1355–1359. [DOI] [PubMed] [Google Scholar]
- 3.Takeuchi N, Nomura Y. Ruptured renal arteriovenous malformation successfully treated by catheter embolization: a case report. BMC Res Notes. 2014;7:19. doi: 10.1186/1756-0500-7-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Obara H, Tanaka K, Matsubara K. et al. Large renal arteriovenous fistula with left-sided inferior vena cava. Ann VascSurg. 2013;27(8):1.185E12–1.185E15. doi: 10.1016/j.avsg.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 5.Rundback J H, Rizvi A, Rozenblit G N. et al. Percutaneous stent-graft management of renal artery aneurysms. J Vasc IntervRadiol. 2000;11(9):1189–1193. doi: 10.1016/s1051-0443(07)61362-1. [DOI] [PubMed] [Google Scholar]
- 6.Kalva S P, Wu S, Irani Z. Endovascular treatment of renal artery aneurysms. Endovascular Today. 2011:58–62. [Google Scholar]
- 7.González J, Esteban M, Andrés G, Linares E, Martínez-Salamanca J I. Renal artery aneurysms. Curr Urol Rep. 2014;15(1):376. doi: 10.1007/s11934-013-0376-z. [DOI] [PubMed] [Google Scholar]
- 8.Tham G, Ekelund L, Herrlin K, Lindstedt E L, Olin T, Bergentz S E. Renal artery aneurysms. Natural history and prognosis. Ann Surg. 1983;197(3):348–352. doi: 10.1097/00000658-198303000-00016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ogawa S, Yanagida T, Kataoka M. et al. Laparoscopic nephrectomy, ex vivo angioplasty, and renal autotransplant for a renal artery aneuryms: A case report. ExpClin Transplant. 2012;10(1):67–69. doi: 10.6002/ect.2011.0072. [DOI] [PubMed] [Google Scholar]
- 10.Eisenberg M L, Lee K L, Zumrutbas A E, Meng M V, Freise C E, Stoller M L. Long-term outcomes and late complications of laparoscopic nephrectomy with renal autotransplantation. J Urol. 2008;179(1):240–243. doi: 10.1016/j.juro.2007.08.135. [DOI] [PubMed] [Google Scholar]
- 11.Tulsyan N Kashyap V S Greenberg R K et al. The endovascular management of visceral artery aneurysms and pseudoaneurysms J Vasc Surg 2007452276–283., discussion 283 [DOI] [PubMed] [Google Scholar]
- 12.Iwazu Y, Muto S, Miyata Y. et al. The origin of idiopathic renal arteriovenous malformation with giant twin aneurysms. ClinNephrol. 2013;79(1):81–84. doi: 10.5414/cn107228. [DOI] [PubMed] [Google Scholar]
- 13.Andreev A P, Guirov K G. Post-traumatic giant fistula between the aorta and the left renal vein: a case report. Int J Angiol. 2008;17(4):217–219. doi: 10.1055/s-0031-1278314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kato T, Takagi H, Ogaki K, Oba S, Umemoto T. Giant renal aneurysm with arteriovenous fistula. Heart Vessels. 2006;21(4):270–272. doi: 10.1007/s00380-005-0888-7. [DOI] [PubMed] [Google Scholar]


