Abstract
Osteoporosis -related to various factors including menopause and aging- is the most common chronic metabolic bone disease, which is characterized by increased bone fragility. Although it is seen in all age groups, gender, and races, it is more common in Caucasians (white race), older people, and women. With an aging population and longer life span, osteoporosis is increasingly becoming a global epidemic. Currently, it has been estimated that more than 200 million people are suffering from osteoporosis. According to recent statistics from the International Osteoporosis Foundation, worldwide, 1 in 3 women over the age of 50 years and 1 in 5 men will experience osteoporotic fractures in their lifetime. Every fracture is a sign of another impending one. Osteoporosis has no clinical manifestations until there is a fracture. Fractures cause important morbidity; in men, in particular, they can cause mortality. Moreover, osteoporosis results in a decreased quality of life, increased disability-adjusted life span, and big financial burden to health insurance systems of countries that are responsible for the care of such patients. With an early diagnosis of this disease before fractures occur and by assessing the bone mineral density and with early treatment, osteoporosis can be prevented. Therefore, increasing awareness among doctors, which, in turn, facilitates increase awareness of the normal populace, will be effective in preventing this epidemic.
Keywords: Osteoporosis, osteoporosis management, bone mineral density, fracture risk
Introduction
Osteoporosis is a disease that is characterized by low bone mass, deterioration of bone tissue, and disruption of bone microarchitecture: it can lead to compromised bone strength and an increase in the risk of fractures (1).
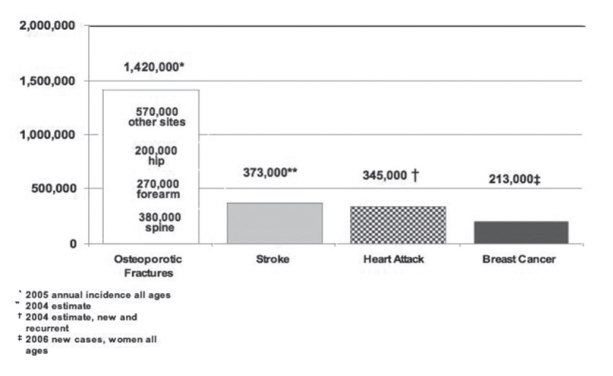
Osteoporosis is the most common bone disease in humans, representing a major public health problem. It is more common in Caucasians, women, and older people. Osteoporosis is a risk factor for fracture just as hypertension is for stroke. Osteoporosis affects an enormous number of people, of both sexes and all races, and its prevalence will increase as the population ages. It is a silent disease until fractures occur, which causes important secondary health problems and even death (Figure 1) (2).
Figure 1.
Comparative incidences of osteoporosis-related fractures, new strokes, heart attacks, and invasive breast cancer in women in the United States (6)
It was estimated that the number of patients worldwide with osteoporotic hip fractures is more than 200 million (3). It was reported that in both Europe and the United States, 30% women are osteoporotic, and it was estimated that 40% post-menopausal women and 30% men will experience an osteoporotic fracture in the rest of their lives (4–6).
Osteoporosis is also an important health issue in Turkey, because the number of older people is increasing. The incidence rate for hip fracture increases exponentially with age in all countries as well as in Turkey, which is evident in the FRACTURK study (7). It was estimated that around the age of 50 years, the probability of having a hip fracture in the remaining lifetime was 3.5% in men and 14.6% in women (7).
Bone tissue is continuously lost by resorption and rebuilt by formation; bone loss occurs if the resorption rate is more than the formation rate. The bone mass is modeled (grows and takes its final shape) from birth to adulthood: bone mass reaches its peak (referred to as peak bone mass (PBM)) at puberty; subsequently, the loss of bone mass starts. PBM is largely determined by genetic factors, health during growth, nutrition, endocrine status, gender, and physical activity. Bone remodeling, which involves the removal of older bone to replace with new bone, is used to repair microfractures and prevent them from becoming macrofractures, thereby assisting in maintaining a healthy skeleton. Menopause and advancing age cause an imbalance between resorption and formation rates (resorption becomes higher than absorption), thereby increasing the risk of fracture. Certain factors that increase resorption more than formation also induce bone loss, revealing the microarchitecture. Individual trabecular plates of bone are lost, leaving an architecturally weakened structure with significantly reduced mass; this leads to an increased risk of fracture that is aggravated by other aging-associated declines in functioning. Increasing evidence suggests that rapid bone remodeling (as measured by biochemical markers of bone resorption or formation) increases bone fragility and risk of fracture.
There are factors associated with an increased risk of osteoporosis-related fractures. These include general factors that relate to aging and sex steroid deficiency, as well as specific risk factors such as use of glucocorticoids (which cause decreased bone formation and bone loss), reduced bone quality, and disruption of microarchitectural integrity. Fractures result when weakened bone is overloaded, often by falls or certain daily chores (8).
Classification
Osteoporosis can be classified into two main groups by considering the factors affecting bone metabolism:
- Primary osteoporosis
- Primary osteoporosis can also be divided into two subgroups:
Table 1.
| Lifestyle changes | Genetic diseases | Endocrine disorders | Other |
|---|---|---|---|
| Vitamin D insufficiency | Cystic fibrosis | Central obesity | AIDS/HIV |
| High salt intake | Glycogen storage diseases | Cushing’s syndrome | Amyloidosis |
| Smoking (active or passive) | Menkes steely hair syndrome | Diabetes mellitus (types 1 and 2) | Chronic obstructive lung disease |
| Alcohol abuse | Osteogenesis imperfecta | Hyperparathyroidism | Congestive heart failure |
| Immobilizatıon | Riley–Day syndrome | Thyrotoxicosis | Chronic metabolic acidosis |
| Excessive thinness | Ehler Danlos | Hypogonadal states: | Depression |
| Frequent falling | Hemochromatosis | Androgen insensitivity | End-stage renal disease |
| Low calcium intake | Marfan syndrome | Athletic amenorrhea | Hypercalciuria |
| Inadequate physical activity | Parental history of hip fracture | Premature menopause (<40 years) | Post-transplant bone disease |
| Excess vitamin A | Gaucher’s disease | Hyperprolactinemia | Idiopathic scoliosis |
| Homocystinuria | Panhypopituitarism | Sarcoidosis | |
| Hypophosphatasia | Anorexia nervosa | Weight loss | |
| Porphyria | Turner’s and Klinefelter’s syndromes | ||
|
| |||
| Gastrointestinal disorders | Hematological disorders | Neurological and musculoskeletal factors | Rheumatologic and autoimmune diseases |
|
| |||
| Celiac disease | Hemophilia | Epilepsy | Ankylosing spondylitis |
| Gastric bypass | Leukemia and lymphomas | Multiple sclerosis | Systemic lupus |
| Gastrointestinal surgery | Sickle cell disease | Muscular dystrophy | Rheumatoid arthritis |
| Malabsorption | Multiple myeloma | Parkinson’s disease | Systemic lupus |
| Inflammatory bowel disease | Monoclonal gammopathies | Spinal cord injury | Other rheumatic and autoimmune diseases |
| Pancreatic disease | Systemic mastocytosis | Stroke | |
| Primary biliary cirrhosis | Thalassemia | Proximal myopathy | |
| Medications | |||
Aluminum (in antacids), Anticoagulants (heparin), Anticonvulsants, Aromatase inhibitors, Barbiturates, Cancer chemotherapeutic drugs, Depomedroxyprogesterone (premenopausal contraception), Glucocorticoids (≥5 mg/day prednisone or equivalent for ≥3 months), GnRH (gonadotropin-releasing hormone) agonists, Lithium Cyclosporine A, Tacrolimus, Methotrexate, Parental nutrition, Proton pump inhibitors Selective serotonin reuptake inhibitors, Tamoxifen® (premenopausal use) Thiazolidinediones, Thyroid hormones (in excess), warfarin
AIDS/HIV: acquired immune deficiency syndrome/human immunodeficiency virus
Involutional Osteoporosis Type I
It is also known as postmenopausal osteoporosis, caused by the deficiency of estrogen, mainly affecting the trabecular bone; therefore, women are more susceptible to osteoporosis than men, as evident by a men/women ratio of 4/5.7 (2).
Involutional Osteoporosis Type II
It is also called senile osteoporosis, and it is related to bone mass lost due to the aging of cortical and trabecular bones (2, 9).
- Secondary osteoporosis:
Different diseases, medications, and lifestyle changes can cause osteoporosis (Table 1).
Clinical Consequences
Clinical evaluation
Osteoporosis has been mislabeled as a women’s disease by the public, but it affects men, too: young men are afflicted by it, which usually goes undiagnosed until a fracture brings the patient to a doctor. However, delayed interventions are usually unsuccessful. The diagnosis of osteoporosis is never taken as primary osteoporosis without ruling out the secondary causes. A good history and physical examination of the patient always reveal certain clues about the presence of another disease: certain special laboratory evaluations might be needed to rule out other responsible diseases.
Clinical findings and complications
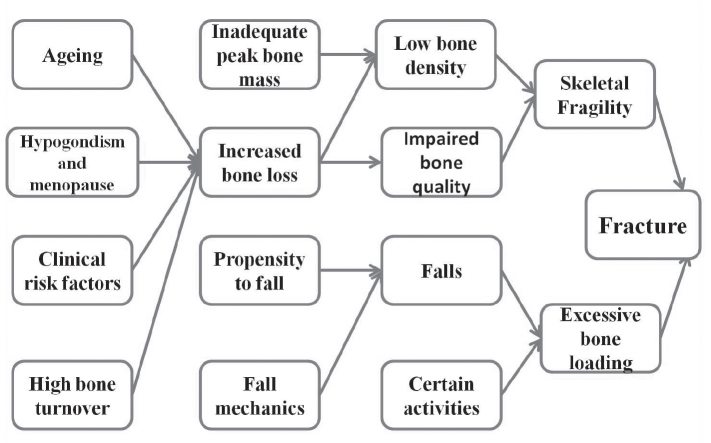
Fractures and their complications are the relevant clinical sequelae of osteoporosis. Osteoporosis is a silent disease until the patient experiences a fracture. A recent fracture at any major skeletal site, such as vertebrae (spine), proximal femur (hip), distal forearm (wrist), or shoulder in an adult older than 50 years with or without trauma, should suggest that the diagnosis of osteoporosis needs further urgent assessment involving diagnosis and treatment. Clinical risk factors are listed in (Table 2). The pathogenesis of osteoporosis-related fractures is summarized in (Figure 2) (10, 11).
Table 2.
Clinical risk factors used for the assessment of fracture probability (10)
| Age |
| Sex |
| Low body mass index (≤19 kg/m2) |
| Previous fragility fracture, particularly of the hip, wrist and spine including morphometric vertebral fracture |
| Secondary causes of osteoporosis including: |
| Parental history of hip fracture |
| Current glucocorticoid treatment (any dose, by mouth for three months or more) |
| Current smoking Alcohol intake of three or more units daily |
| Rheumatoid arthritis |
| Untreated hypogonadism in men and women |
| Prolonged immobility |
| Organ transplantation |
| Type I diabetes |
| Hyperthyroidism |
| Gastrointestinal disease |
| Chronic liver disease |
| Chronic obstructive pulmonary disease |
| Frequent falling |
Figure 2.
Fractures may cause chronic pain, disability, and death. Hip fractures are associated with 15–20% increased mortality rate within 1 year, with a higher mortality rate in men than in women, followed by a 2.5-fold increased risk of future fractures. Approximately 20–50% hip fracture patients require long-term nursing homecare and suffer from decreased quality of life, social isolation, depression, and loss of self-esteem (12). Fractures that occur spontaneously or following minor trauma (e.g., fall from a standing height or less)—called fragility fractures—are very common in osteoporotic individuals (13).
Vertebral fractures might occur during daily chores without any trauma or fall, and they are the predictors of future fracture risk: the probability is fivefold for subsequent vertebral fractures and twofold to threefold for fractures at other sites (14, 15).
The first complaint of the patient might be the loss of height caused by vertebral compression due to fractures, which is more evident in the presence of multiple fractures; this abnormality can be objectively detected by increased occiput-to-wall distance caused by dorsal kyphosis (dowager’s hump). The determination of historical height loss (difference between the current height and peak height at an age of 20 years) of 1.5 inches (4 cm) or more and prospective height loss (difference between the current height and a previously documented height measurement) of 0.8 inches (2 cm) or more is important. Multiple vertebral thoracic fractures may result in restrictive lung disease and secondary heart problems. Lumbar fractures may decrease the volumes between the ribs to the pelvis, alter abdominal anatomy, crowd internal organs (particularly the gastrointestinal system, causing gastrointestinal complaints such as premature satiety, reduced appetite, abdominal pain, constipation, and distention); further, back pain (acute and chronic), prolonged disability, poor self-image, social isolation, depression, and positional restriction are other problems created by compression fractures in addition to increased mortality (16).
Dorsal kyphosis is also seen in some older people without fractures so it is not a diagnostic criterion for osteoporosis.
There are some other factors that increase fracture risk and osteoporosis, independent of bone mineral density (BMD) (17):
- Age of the patient (18)
- A low body mass index (BMI<21 kg/m2) is a significant risk factor for hip fracture (19).
- A history of a previous osteoporotic fracture is another important factor for further fracture risk and almost doubles the risk of spinal fractures (15)
- Parental history of hip fracture (20)
- Smoking (21)
- Oral glucocorticoids ≥5 mg/d of prednisone for >3 months (ever) (22)
- There is a dose-dependent relationship between alcohol intake and fracture risk. Daily intake of 3 or more units of alcohol is associated with fracture risk (23)
- Rheumatoid arthritis increases fracture risk independently of BMD, as well as the use of glucocorticoids (24)
- Falls are an important risk factor for osteoporotic fractures
Environmental and medical factors increasing falls and measures that can be taken to prevent such falls at home are listed in (Table 3) (25).
Table 3.
Risk factors for falls and multifactorial interventions to prevent falls in community-dwelling older people (1, 6, 25)
| Environmental risk factors | |
|---|---|
| Lack of assistive devices | Obstacles in the walking paths |
| Loose throw rugs | Slippery conditions |
| Low level lighting | |
|
| |
| Medical risk factors | |
|
| |
| Age | Arrhythmias |
| Anxiety and agitation | Depression |
| Poor vision | Orthostatic hypotension |
| Dehydration | Previous falls or fear of falling |
| Medications causing sedation (narcotic analgesics, anticonvulsants, psychotropics | Reduced problem solving or mental acuity and diminished cognitive skills |
| Vitamin D insufficiency [serum 25-hydroxyvitamin D (25 (OH) D) <30 ng/mL (75 nmol/L)] | Urgent urinary incontinence |
| Malnutrition | |
|
| |
| Neurological and musculoskeletal risk factors | |
|
| |
| Kyphosis | Reduced proprioception |
| Impaired transfer and mobility | Weak muscles/sarcopenia |
| Poor balance | |
| Deconditioning | Diseases listed in Table 1 |
|
| |
| Multifactorial interventions to prevent of falls in community-dwelling elderly people | |
|
| |
| Appropriate exercise program after evaluation of the multifactorial fall risk assessment | |
| Withdrawal or minimization of psychoactive medications (e.g., sedative hypnotics, anxiolytics, antidepressants) and antipsychotics | |
| Reduced visual acuity due to either cataracts, macular degeneration, glaucoma are very common in elderly and increases risk of falling, should be treated orderly | |
| Treatment of postural hypotension | |
| Treatment of cardiac problems especially arrhythmias | |
| Exercise, in the form of strength training and balance, gait, and coordination training, is effective in reducing falls | |
| Selective use of orthotics could help reduce discomfort, prevent falls and fractures, and improve quality of life | |
Management of foot problems and footwear
| |
Adaptation or modification of home environment
| |
Prevention of falls
Falls are the cause of a majority of osteoporotic fractures; therefore, a program needs to be structured for the effective treatment of osteoporosis in order to prevent falls (Table 3). Older and frail persons and those who have had a stroke or are taking medications that decrease mental alertness are particularly predisposed toward falls. Although several interventions reduce the risk of falling, none of them apparently reduce the risk of fractures. Hip protectors do not reduce the risk of falling, but they reduce the risk of fracture. Patients at an older age with severe kyphosis, back discomfort, and gait instability could benefit from weight-bearing exercises, back strengthening, and balance training therapies.
Diagnosis of osteoporosis
Bone strength can be defined using BMD (70%) and bone quality (20%). It is easy to measure BMD, but, in clinical settings, bone quality is not measurable yet. The diagnosis of osteoporosis is established by the measurement of BMD or by the occurrence of a fragility fracture of the hip or vertebra or in the absence of major trauma (e.g., motor vehicle accident or fall from multiple stories). Laboratory testing revealed no secondary causes of osteoporosis (Table 1).
Bone mineral density is measured by means of dual X-ray absorptiometry (DXA); it is the actual expression of the bone in absolute terms of grams of mineral (primarily, as g/cm2 of calcium) per square centimeter of the scanned bone. BMD measurements of the hip and spine are used to establish or confirm the diagnosis of osteoporosis to predict future fracture risk and monitor patients. The difference between the patient’s BMD and mean BMD of young females aged in the range of 20–29 years (divided by the standard deviation (SD) of the reference population) yields the T-score; comparing the BMD of a particular age, sex, and ethnicity-matched adult reference population is called the Z-score. As defined by the World Health Organization (WHO), osteoporosis is present when BMD is 2.5 SD or more below the average value for young healthy women (a T-score of <−2.5 SD). A second, higher threshold describes “low bone mass” or osteopenia as a T-score that lies between −1 and −2.5 SD. “Severe” or “established” osteoporosis denotes osteoporosis that has been defined in the presence of one or more documented fragility fractures (Table 4) (26).
Table 4.
WHO definitions of osteoporosis based on BMD (26)
| Classification | Bone Mineral Density | T Score |
|---|---|---|
| Normal | Within 1 SD of the mean level for a young adult reference population | T score at −1.0 and above |
| Low bone mass (Osteopenia) | Between 1 and 2.5 SD below that of the mean level for a young adult reference population | T score between −1.0 and −2.5 |
| Osteoporosis | 2.5 or more below that of the mean level for a young adult reference population | T score at or below −2.5 |
| Severe or established osteoporosis | 2.5 or more below that of the mean level for a young adult reference population with fractures | T score at or below −2.5 with one or more fractures |
WHO: World Health Organization; BMD: bone mineral density; SD: standard deviation
Bone mineral density can be easily measured to detect bone density, but the degree of deterioration of the bone tissue cannot be measured in clinical settings, except for the biochemical markers of bone tissue (27).
Fracture risk is closely correlated with bone strength and increases exponentially as the BMD decreases. Dual energy X-ray absorptiometry (DXA) measurements of hips are the best predictors of hip fracture risks. The measurements of the total hip, femoral neck, or total lumbar spine (or a combination of these sites) are the preferred measurement sites. If the hip and/or lumbar spine sites cannot be measured or become unusable (e.g., hyperparathyroidism or very obese patients), one-third (33%) of the site radius can be used. Other hip regions, including Ward’s area and the greater trochanter, should not be used for diagnosis. BMD may be measured at either hip.
T-score criteria are applied for the BMD measured by means of central DXA at the femoral hip and lumbar spine for postmenopausal women and men aged 50 years and older. For premenopausal women, men less than 50 years of age, and children, the BMD diagnostic classification as defined by the WHO should not be applied. The International Society for Clinical Densitometry (ISCD) recommends using ethnic- or race-adjusted Z-scores: Z-scores of −2.0 or lower are defined as “low bone mineral density for chronological age” or “below the expected range for age” and those above −2.0 are defined as “within the expected range for age” (28).
The United States Preventive Services Task Force (USPSTF) recommends the testing of all women aged 65 years and above and younger women whose fracture risk is equal to or greater than that of a 65-year-old white woman who exhibits no additional risk factors (29). Indications for measuring BMD are listed at (Table 5) (13).
Table 5.
Indications for measuring BMD (13)
| Older adults (age >50 yr) | Younger adults (age <50 yr) |
|---|---|
| Age ≥65 yr (both women and men) Clinical risk factors for fracture (menopausal women, men aged 50–64 yr)
|
Fragility fracture Hypogonadism or premature menopause (age<45 yr) Malabsorption syndrome Prolonged use of glucocorticoids* Use of other high-risk medications** Primary hyperparathyroidism Other disorders strongly associated with rapid bone loss and/or Fracture |
BMD: bone mineral density; yr: years; kg: kilogram
At least 3 months cumulative therapy in the previous year at a prednisone-equivalent dose of 7.5 mg
Aromatase inhibitors or androgen deprivation therapy use of thiazolidinediones, proton pump inhibitors
Fracture Risk Assessment Tool Model (FRAX)
The most important health consequence of osteoporosis is fractures. Recently, algorithms have been developed to predict the risk of fracture in individuals that incorporate significant predictors of fracture risk in addition to BMD. Estimating the 10-year risk of a major osteoporotic fracture (i.e., fracture of the hip, vertebra (clinical), forearm, or proximal humerus) is possible with algorithms that integrate the weight of clinical risk fractures for fracture risk with or without information on the BMD have been developed (http://www.shef.ac.uk/FRAX). They can be used to compute the 10-year probability of hip fracture or a major osteoporotic fracture (clinical spine, hip, forearm, or humerus).
Clinical risk factors used in FRAX are as follows (26):
- Current age
- Sex
- A prior osteoporotic fracture (including clinical and asymptomatic vertebral fractures)
- BMD of femur neck
- Low BMI (BMI<21 kg/m2)
- Oral glucocorticoids ≥5 mg/d of prednisone for >3 months (ever)
- Rheumatoid arthritis
- Parental history of hip fracture
- Secondary causes of osteoporosis: Type-1 DM, early menopause <40 years, etc.
- Being a past or current smoker
-
- Alcohol intake (3 or more drinks/day)
Probabilities have been computed for several countries (30).
A vertebral fracture is consistent with the diagnosis of osteoporosis, even in the absence of a bone density diagnosis; it is an indication for pharmacologic treatment with osteoporosis medication to reduce subsequent fracture risk. Unrecognized vertebral fractures may change the diagnostic classification, alter future fracture risk calculations, and affect treatment decisions. Regardless of BMD, age, and other clinical risk factors, radiographically confirmed vertebral fractures are a sign of impaired bone quality and strength and strong predictor of new vertebral and other fractures (15).
Vertebral imaging (Vertebral fracture assessment)
Asymptomatic vertebral fractures are very common in older patients necessitating vertebral imaging, which can be performed using a lateral thoracic and lumbar spine X-ray or lateral Vertebral Fracture Assessment (VFA) available on most DXA machines; this can be performed at the time of BMD assessment. Lateral spine imaging using conventional radiography or densitometric VFA is indicated when the T-score <–1.0 at the spine, total hip, femoral neck, and other sites is present (2) (Table 6).
Table 6.
Indications for vertebral imaging (2)
| Indications for Vertebral Imaging |
|---|
| Women aged ≥70 years or men aged ≥80 years |
| Difference between the current height and peak height at age 20 (historical height loss) ≥4 cm (>1.5 inches) |
| Self-reported but undocumented prior vertebral fracture |
| Prospective height loss (difference between the current height and a previously documented height measurement) of 2 cm (0.8 inches) or more |
| Recent or ongoing long-term glucocorticoid treatment (Glucocorticoid therapy equivalent to ≥ 5 mg of prednisone or equivalent per day for ≥3 months) |
| If bone density measurement is not available, vertebral imaging may be considered based on age alone |
cm: Centimeter; mg: milligram
Biochemical bone turnover marker (BTM)
Bone remodeling (or turnover) occurs throughout life to repair fatigue damage and microfractures in the bone and to maintain mineral homeostasis. Biochemical markers of bone remodeling include resorption markers, namely serum C-terminal telopeptide type-I collagen (s-CTX) and urinary N-telopeptide (NTX), and formation markers, such as serum procollagen type-I N-terminal propeptide (s-PINP), which may provide information on fracture risk independent of BMD and predict the rapidity of bone loss in untreated patients. Therefore, the fracture risk prediction might get enhanced by their inclusion in assessment algorithms. Further, they can be used to predict the response to treatments. It was shown that there is a significant relationship between the reduction in BTMs following antiresorptive therapy and reduction in vertebral and non-vertebral fracture risk. In general, these studies showed that the greater the decrease in BTM, larger is the reduction in fracture risk.
International Osteoporosis Foundation (IOF) and International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Working Group evaluated BTMs in the prediction of fracture risk and for monitoring treatment; it was recommended that bone formation markers (s-PINP) and bone resorption markers (s-CTX) should be used as reference markers and measured by standardized assays in observational and intervention studies (31).
Following studies are necessary to rule out secondary osteoporosis (32):
Complete blood count (CBC)
Serum creatinine, calcium, phosphorus, and magnesium
Alanin aminotransferase (ALT), aspartat aminotransferase (AST), and alkaline phosphatase (AP)
Thyroid-stimulating hormone (TSH) and free T4
Vitamin D (V-D) (25 (OH) D)
Parathyroid hormone (PTH)
Total testosterone and gonadotropin in younger men
BTMs
Considered in select patients
Serum protein electrophoresis (SPEP), serum immunofixation, and serum-free light chains
Tissue transglutaminase antibodies (IgA and IgG)
Iron and ferritin levels
Homocysteine
Prolactin
Tryptase
24-h urinary calcium
Urinary protein electrophoresis (UPEP)
Urinary-free cortisol level
Urinary histamine
Approach to a patient with osteoporosis
A detailed history and physical examination together with BMD assessment, vertebral imaging to diagnose vertebral fractures (when appropriate), and the WHO-defined 10-year estimated fracture probability test are utilized to establish an individual patient’s fracture risk (30).
All postmenopausal women and men aged 50 years and above should be evaluated for osteoporosis risk in order to determine the need for BMD testing and/or vertebral imaging. In general, the more the risk factors, larger is the risk of fracture. Osteoporosis is preventable and treatable, but because there are no warning signs prior to a fracture, many people are not being diagnosed in time to receive effective therapy during the early phase of this disease. The factors that increase the risk of osteoporosis-related fractures are listed in (Table 2).
Universal recommendations for all patients
Several interventions, including an adequate intake of calcium and V-D, are fundamental aspects for any osteoporosis prevention or treatment program, including lifelong regular weight-bearing and muscle-strengthening exercises, cessation of tobacco use and excess alcohol intake, and treatment of risk factors for falling (Table 2, 3).
In order to maintain serum calcium at a constant level, an external supply of adequate calcium is necessary; otherwise, low serum calcium levels promote bone resorption to bring the calcium levels to normal. Calcium requirements increase among older persons; thus, the older population is particularly susceptible to calcium deficiency. The Institute of Medicine (IOM) recommends a daily intake of 1000 mg/day for men aged 50–70 years and 1200 mg of calcium for women aged over 50 years and men aged over 70 years (33, 34).
All calcium preparations are better absorbed when taken with food, particularly in the absence of the secretion of gastric acid. For optimal absorption, the amount of calcium should not exceed 500–600 mg per dose. Calcium carbonate is the least expensive and necessitates the use of the fewest number of tablets, but it may cause gastrointestinal (GI) complaints. Calcium citrate is more expensive, and a larger number of tablets are needed to achieve the desired dose; however, its absorption is not dependent on gastric acid and it does not cause GI complaints. Some food products contain excess oxalate, which prevents absorption of calcium by binding with it. Intakes in excess of 1200–1500 mg/day may increase the risk of developing kidney stones, cardiovascular diseases, and strokes.
Vitamin D is necessary for calcium absorption, bone health, muscle performance, and balance. The IOM recommends a dose of 600 IU/day until the age of 70 years in adults and 800 IU/day thereafter (34–36). Chief dietary sources of V-D include V-D–fortified milk, juices and cereals, saltwater fish, and liver. Supplementation with V-D2 (ergocalciferol) or V-D3 (cholecalciferol) may be used.
Many older patients are at a high risk for V-D deficiency, which include the following: patients with malabsorption issues (e.g., celiac disease) or other intestinal diseases (e.g., inflammatory bowel disease, gastric bypass surgery); gastric acidity; pernicious anemia; proton pump inhibitors; chronic renal or liver insufficiency; patients on medications that increase the breakdown of V-D (e.g., some anticonvulsive drugs); or glucocorticoids, which decrease calcium absorption; housebound and chronically ill patients; persons with limited sun exposure; individuals with very dark skin; and obese individuals.
Serum 25 (OH) D levels should be measured in patients at the risk of V-D deficiency. V-D supplements should be recommended in amounts sufficient to bring the serum 25 (OH) D level to approximately 30 ng/mL (75 nmol/L). Many patients with osteoporosis will need more than the general recommendation of 800–1000 IU/day. The safe upper limit for V-D intake for the general adult population was increased to 4000 IU/day in 2010.
Alcohol
Excessive intake of alcohol has detrimental effects on bones, so it should be avoided. The mechanisms are multifactorial and include predisposition to falls, calcium deficiency, and chronic liver disease, which, in turn, results in predisposition toward V-D deficiency. Persons predisposed toward osteoporosis should be advised against consuming more than 7 drinks/week, 1 drink being equivalent to 120 mL of wine, 30 mL of liquor, or 260 mL of beer (23).
Caffeine
Patients should be advised to limit their caffeine intake to less than 1 to 2 servings (8 to 12 ounces in each serving) of caffeinated drinks per day. Some studies showed that there is a relationship between caffeine consumption and fracture risk (37).
Exercise
A regular weight-bearing exercise regimen (for example, walking 30–40 min per session) along with back and posture exercises for a few minutes on most days of the week should be advocated throughout life. Children and young adults who are active reach a higher peak bone mass than those who are not (38).
Among older patients, these exercises help slow bone loss attributable to disuse, improve balance, and increase muscle strength, ultimately reducing the risk of falls (39). Patients should avoid forward flexion, side-bending exercises, or lifting heavy objects because pushing, pulling, lifting, and bending activities compress the spine, leading to fractures.
Prevention of falls
Falls are the precipitating cause for most osteoporotic fractures; some measures should be taken to prevent falls in the household, particularly for patients who are frail, taking some medications that affect mental alertness, thereby resulting in a stroke (40) (Table 3).
Pharmacologic therapy
All osteoporotic patients should be evaluated for secondary causes of osteoporosis before starting treatment and subjected to BMD measurements using central DXA, including vertebral imaging studies when appropriate: the BTM levels should be obtained if monitoring of the treatment is planned.
Who should be considered for treatment?
Postmenopausal women and men aged 50 years and above who present with the following should be considered for treatment:
- A hip or vertebral fracture (clinically apparent or found on vertebral imaging) because it was shown that the patients with spine and hip fractures had reduced fracture risk with pharmacologic therapy, irrespective of the T score.
- Determination of fractures when the T-score ≤−2.5 at the femoral neck, total hip, or lumbar spine
- Low bone mass (T-score between −1.0 and −2.5 at the femoral neck or lumbar spine) and
- 10-year probability of a hip fracture ≥3% or a 10-year probability of a major osteoporosis-related fracture ≥20%
The anti-fracture benefits of Food and Drug Administration (FDA)-approved drugs have mostly been studied in women with postmenopausal osteoporosis. There are limited fracture data for men and cases involving glucocorticoid-induced osteoporosis.
Pharmacologic agents for treatment of osteoporosis
The main goals of therapy in osteoporotic patients are as follows:
- To prevent fractures by improving bone strength and reducing the risk of falling and injury
- To relieve symptoms of fractures and skeletal deformity
- To maintain normal physical function
Most of the current therapies in the prevention of osteoporosis and fractures are designed to decrease bone resorption and they are known as antiresorptive agents. They include estrogen; bisphosphonates (BPs) such as alendronate, risedronate, ibandronate, and zoledronic acid; selective estrogen receptor modulators (SERM) raloxifene; human monoclonal antibody against receptor activator of NF-κB ligand (RANKL) denosumab; and strontium ranelate (SR). The agents used for the prevention or treatment of osteoporosis and drugs that decrease the risk of fractures are shown in (Table 7, 8) respectively.
Table 7.
Drugs used for prevention and treatment of osteoporosis
| Medication | Postmenopausal osteoporosis | Glucocorticoid induced osteoporosis | In Men | ||
|---|---|---|---|---|---|
|
|
|
|
|||
| Prevention | Treatment | Prevention | Treatment | Treatment | |
| Estrogen | Multiple regimens | ||||
| Denosumab | 60 mg sc/6mo | ||||
| Raloxifene | 60mg po/d | 60 mg po/d | |||
| Ibandronate | 2.5 mg po/d | 2.5 mg po/d | |||
| 150 mg/mo | 150 mg po/mo | ||||
| 3 mg iv/3mo | |||||
| Alendronate 5–10–70 mg | 5 mg po/d | 10 mg po/d | 5 mg po/d | 10 mg po/d | |
| 35 mg po/wk | 70 mg po/wk | 10mg po/d | 70 mg po/wk | ||
| 70mg po+VD | |||||
| Risedronate 5–35–75 mg | 5 mg po/d | 5 mg po/d | 5 mg po/d | 5 mg po/d | 35 mg po/wk |
| 35 mg po/wk | 150 mg po/mo | ||||
| 150 mg/po/mo | |||||
| Stronsium Ranelate 2gr po, eff | 2 gr/d | 2 gr/d | 2 gr/d | ||
| Zoledronic acid | 5mg iv/2 yr | 5 mg iv/yr | 5 mg iv/yr | 5 mg iv/yr | 5 mg iv/yr |
| Teriparatid | 20 ug sc/d | 20 ug sc/yr | 20 ug sc/d | ||
iv:intravenous; po: oral; sc: subcutaneous; eff: effervescent yr: year; wk: week; mo: month; d: day
Table 8.
Drugs that decreases the risk of fractures
| Drug | Vertebral | Nonvertebral | Hip |
|---|---|---|---|
| Alendronate | Yes | Yes | Yes |
| Risedronate | Yes | Yes | Yes |
| Zolendronic Acid | Yes | Yes | Yes |
| Denosumab | Yes | Yes | Yes |
| Teriparatid demonstrated | Yes | Yes | No effect |
| Ibandronat demonstrated | Yes | Yes | No effect |
| Raloxifene demonstrated | Yes | Yes | No effect |
All antiresorptive agents increase bone mass, but only alendronate, risedronate, zoledronic acid, and SR are shown to decrease both vertebral and hip fractures. However, the efficacy of the remaining is only for vertebral fractures. Nonetheless, hip fractures cause increased morbidity, mortality, and healthcare costs as compared to vertebral fractures. All of these antiresorptive therapies do not stimulate bone formation, except currently available anabolic agents (namely, PTH), which increase bone strength and bone formation. They are particularly helpful for those having severe or established osteoporosis. Unfortunately, the adherence to and persistence toward these techniques are poor toward all osteoporosis therapies, which decrease the success of treatment.
Antiresorptive agents
Bisphosphonates are the most widely used drugs for the treatment of osteoporosis. Alendronate is used in the prevention and treatment of postmenopausal, glucocorticoid-induced, and male osteoporosis cases. Trials lasting up to 10 years have been reported for postmenopausal osteoporosis (41). Efficacy and safety beyond 10 years have not yet been established. A drug holiday of 1 or 2 years is suggested after 4–5 years of therapy (and longer for those with severe osteoporosis).
Risedronate is used in men and women for the prevention and treatment of osteoporosis and glucocorticoid-induced osteoporosis. A drug holiday of up to 1 year after 7 years of therapy has been suggested (42).
Ibandronate is another BP used for the prevention and treatment of postmenopausal osteoporosis, which has proven efficacy in reducing the risk of spinal fractures of postmenopausal women suffering from osteoporosis, but it is not proven in reducing non-vertebral or hip fractures except for higher-risk subgroup. Ibandronate has been studied in trials of up to 3 years and its efficacy and safety beyond 3 years is not known (43).
Zoledronic acid is used for the prevention and treatment of postmenopausal osteoporosis and osteoporosis in men as well as glucocorticoid-induced osteoporosis. It is given once yearly, administered intravenously, as a 5-mg infusion over a minimum of 15 min (44).
Cautions to be taken, contraindications, and complications to BP therapy
The absorption of oral BPs is less than 1% with any food, beverage other than plain water, or medications within 2 h after drug administration. Oral BPs should be used with caution: they should not be used in patients with active upper GI diseases, inability to remain upright for 30–60 min, anatomic or functional esophageal abnormalities that might delay the transit of the tablet (achalasia, stricture, etc.), V-D deficiency, hypocalcemia, and hypersensitivity to drug and kidney failure (should be used with caution when the glomerular filtration rate is below 30 ml/min for risedronate and ibandronate or below 35 mL/min for alendronate and zoledronate). Intravenous administration of BPs, such as ibandronate and zoledronate, causes acute phase reactions (e.g., fever, muscle aches, etc.) with the first dose and it can last several days in some patients; this can be prevented by administering acetaminophen. BPs have been reported to cause bone, joint, or muscle complaints; osteonecrosis of the jaw (ONJ); and alendronate causing atypical fractures (45).
Raloxifene has been shown to reduce the risk of fractures of the spine in women with postmenopausal osteoporosis, but its efficacy in reducing non-vertebral or hip fractures has not been demonstrated. Increases in hot flushes are contraindicated in fertile women and those who have had venous thromboembolic disease (46).
Estrogen replacement therapy (ERT) is used for the prevention of postmenopausal osteoporosis for those showing a significant risk of osteoporosis and for whom non-estrogen medications are not considered to be appropriate, but estrogen has never been approved for the treatment of osteoporosis.
Strontium ranelate reduces vertebral and non-vertebral fracture risk in postmenopausal osteoporotic women, irrespective of age or severity of the underlying disease (47, 48). It was demonstrated that SR increases BMD and reduces fracture risk in postmenopausal women and in men with osteoporosis. Although SR has not been approved by the FDA, it was approved by the European Medicines Agency (EMA) in the treatment of osteoporosis in men with an increased risk of fracture.
Denosumab (human monoclonal antibody against RANKL) is used in the treatment of postmenopausal women at a high risk of fracture, patients having a history of osteoporotic fractures, or patients who have failed or are intolerant to other available osteoporosis therapies. It has been shown to reduce the risk of fractures of the spine, hip, and non-vertebral sites. Hypocalcemia must be corrected before the initiation of therapy. Serious infections, dermatitis, rashes, and eczema may occur. ONJ has been reported; drug discontinuation is suggested with severe symptoms. Efficacy and safety beyond 6 years with denosumab have not yet been established (49).
Teriparatide (recombinant human PTH 1–34) is used in the treatment of postmenopausal osteoporosis with a high risk of fracture, those who have failed or are intolerant to previous osteoporosis therapies, and to increase bone mass in men with idiopathic or hypogonadal osteoporosis. Teriparatide is also approved for the treatment of men and women with glucocorticoid-induced osteoporosis. Serum calcium, PTH, and 25 (OH) D levels should be checked before treatment (50). Teriparatide has a “black box” warning because of the occurrence of osteosarcomas in rats treated with very high doses of teriparatide; it is contraindicated in patients at increased risk of osteosarcoma (those with Paget’s disease of the bone), having open epiphyses, history of irradiation involving the skeleton, unexplained elevation of alkaline phosphatase levels of skeletal origin, or patients with primary or any other form of secondary untreated or unresolved hyperparathyroidism.
The drugs decreasing the fracture risk in vertebral and non-vertebral sites are listed in (Table 8).
Combination or sequential therapies
There is no evidence showing that a combination treatment with 2 or more osteoporosis drugs or sequential use of therapeutically agents shows any benefit on the natural history of osteoporosis or fracture risk reduction. In most patients, long-term treatment needs to be continued. The beneficial effects of treatment with drugs other than BPs wear off soon after the therapy is discontinued, but they may be maintained for longer periods of time after the cessation of BP therapy.
It is recommended that treatment review should be performed after 5 years for alendronate, risedronate, or ibandronate and after 3 years for zoledronic acid. Individuals with fracture risk should continue osteoporosis therapy without a drug holiday until we have convincing new data on the subject.
Conclusion
Osteoporosis is a common and silent disease until it is complicated by fractures that become common. It was estimated that 50% women and 20% of men over the age of 50 years will have an osteoporosis-related fracture in their remaining life. These fractures are responsible for lasting disability, impaired quality of life, and increased mortality, with enormous medical and heavy personnel burden on both the patient’s and nation’s economy. Osteoporosis can be diagnosed and prevented with effective treatments, before fractures occur. Therefore, the prevention, detection, and treatment of osteoporosis should be a mandate of primary healthcare providers.
Footnotes
Ethics Committee Approval: N/A.
Informed Consent: N/A.
Peer-review: Externally peer-reviewed.
Author Contributions: Concept - T.S., L.O., N.C.B.; Design - T.S., L.O.; Supervision - T.S., L.O.; Resources - T.S., L.O.; Materials - T.S., L.O.; Data Collection and/or Processing - T.S., N.C.B.; Analysis and/or Interpretation - T.S., N.C.B.; Literature Search - T.S., N.C.B., L.O.; Writing Manuscript - T.S., L.O., N.C.B.; Critical Review - T.S., L.O., N.C.B.; Other - T.S., L.O., N.C.B.
Conflict of Interest: No conflict of interest was declared by the authors.
Financial Disclosure: The authors declared that this study has received no financial support.
References
- 1.NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA. 2001;285:785–95. doi: 10.1001/jama.285.6.785. [DOI] [PubMed] [Google Scholar]
- 2.Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359–81. doi: 10.1007/s00198-014-2794-2. https://doi.org/10.1007/s00198-014-2794-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cooper C, Campion G, Melton LJ., 3rd Hip fractures in the elderly: a world-wide projection. Osteoporos Int. 1992;2:285–9. doi: 10.1007/BF01623184. https://doi.org/10.1007/BF01623184. [DOI] [PubMed] [Google Scholar]
- 4.Reginster JY, Burlet N. Osteoporosis: a still increasing prevalence. Bone. 2006;38(Suppl 1):S4–9. doi: 10.1016/j.bone.2005.11.024. https://doi.org/10.1016/j.bone.2005.11.024. [DOI] [PubMed] [Google Scholar]
- 5.Wright NC, Looker AC, Saag KG, Curtis JR, Delzell ES, Randall S, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29:2520–6. doi: 10.1002/jbmr.2269. https://doi.org/10.1002/jbmr.2269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Watts NB, Bilezikian JP, Camacho PM, Greenspan SL, Harris ST, Hodgson SF, et al. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for the diagnosis and treatment of postmenopausal osteoporosis. Endocr Pract. 2010;16(Suppl 3):1–37. doi: 10.4158/ep.16.s3.1. https://doi.org/10.4158/10024.GL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tuzun S, Eskiyurt N, Akarirmak U, Saridogan M, Senocak M, Johansson H, et al. Incidence of hip fracture and prevalence of osteoporosis in Turkey: the FRACTURK study. Osteoporos Int. 2012;23:949–55. doi: 10.1007/s00198-011-1655-5. https://doi.org/10.1007/s00198-011-1655-5. [DOI] [PubMed] [Google Scholar]
- 8.Riggs BL, Wahner HW, Seeman E, Offord KP, Dunn WL, Mazess RB, et al. Changes in bone mineral density of the proximal femur and spine with aging. Differences between the postmenopausal and senile osteoporosis syndromes. J Clin Invest. 1982;70:716–23. doi: 10.1172/JCI110667. https://doi.org/10.1172/JCI110667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hannan MT, Felson DT, Dawson-Hughes B, Tucker KL, Cupples LA, Wilson PW, et al. Risk factors for longitudinal bone loss in elderly men and women: the Framingham Osteoporosis Study. J Bone Miner Res. 2000;15:710–20. doi: 10.1359/jbmr.2000.15.4.710. https://doi.org/10.1359/jbmr.2000.15.4.710. [DOI] [PubMed] [Google Scholar]
- 10.Compston J, Bowring C, Cooper A, Cooper C, Davies C, Francis R, et al. Diagnosis and management of osteoporosis in postmenopausal women and older men in the UK: National Osteoporosis Guideline Group (NOGG) update 2013. Maturitas. 2013;75:392–6. doi: 10.1016/j.maturitas.2013.05.013. https://doi.org/10.1016/j.maturitas.2013.05.013. [DOI] [PubMed] [Google Scholar]
- 11.Cooper C, Melton LJ., 3rd Epidemiology of osteoporosis. Trends Endocrinol Metab. 1992;3:224–9. doi: 10.1016/1043-2760(92)90032-v. https://doi.org/10.1016/1043-2760(92)90032-V. [DOI] [PubMed] [Google Scholar]
- 12.Melton LJ, 3rd, Achenbach SJ, Atkinson EJ, Therneau TM, Amin S. Long-term mortality following fractures at different skeletal sites: a population-based cohort study. Osteoporos Int. 2013;24:1689–96. doi: 10.1007/s00198-012-2225-1. https://doi.org/10.1007/s00198-012-2225-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Papaioannou A, Morin S, Cheung AM, Atkinson S, Brown JP, Feldman S, et al. 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ. 2010;182:1864–73. doi: 10.1503/cmaj.100771. https://doi.org/10.1503/cmaj.100771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ensrud KE, Ewing SK, Taylor BC, Fink HA, Stone KL, Cauley JA, et al. Frailty and risk of falls, fracture, and mortality in older women: the study of osteoporotic fractures. J Gerontol A Biol Sci Med Sci. 2007;62:744–51. doi: 10.1093/gerona/62.7.744. https://doi.org/10.1093/gerona/62.7.744. [DOI] [PubMed] [Google Scholar]
- 15.Kanis JA, Johnell O, De Laet C, Johansson H, Oden A, Delmas P, et al. A meta-analysis of previous fracture and subsequent fracture risk. Bone. 2004;35:375–82. doi: 10.1016/j.bone.2004.03.024. https://doi.org/10.1016/j.bone.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 16.Siminoski K, Warshawski RS, Jen H, Lee K. The accuracy of historical height loss for the detection of vertebral fractures in postmenopausal women. Osteoporos Int. 2006;17:290–6. doi: 10.1007/s00198-005-2017-y. https://doi.org/10.1007/s00198-005-2017-y. [DOI] [PubMed] [Google Scholar]
- 17.Kanis JA, Oden A, Johnell O, Johansson H, De Laet C, Brown J, et al. The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int. 2007;18:1033–46. doi: 10.1007/s00198-007-0343-y. https://doi.org/10.1007/s00198-007-0343-y. [DOI] [PubMed] [Google Scholar]
- 18.D’Amelio P, Isaia GC. Male Osteoporosis in the Elderly. Int J Endocrinol. 2015;2015:907689. doi: 10.1155/2015/907689. https://doi.org/10.1155/2015/907689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.De Laet C, Kanis JA, Oden A, Johanson H, Johnell O, Delmas P, et al. Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporos Int. 2005;16:1330–8. doi: 10.1007/s00198-005-1863-y. https://doi.org/10.1007/s00198-005-1863-y. [DOI] [PubMed] [Google Scholar]
- 20.Fox KM, Cummings SR, Powell-Threets K, Stone K. Family history and risk of osteoporotic fracture. Study of Osteoporotic Fractures Research Group. Osteoporos Int. 1998;8:557–62. doi: 10.1007/s001980050099. https://doi.org/10.1007/s001980050099. [DOI] [PubMed] [Google Scholar]
- 21.Kanis JA, Johnell O, Oden A, Johansson H, De Laet C, Eisman JA, et al. Smoking and fracture risk: a meta-analysis. Osteoporos Int. 2005;16:155–62. doi: 10.1007/s00198-004-1640-3. https://doi.org/10.1007/s00198-004-1755-6. [DOI] [PubMed] [Google Scholar]
- 22.Kanis JA, Stevenson M, McCloskey EV, Davis S, Lloyd-Jones M. Glucocorticoid-induced osteoporosis: a systematic review and cost-utility analysis. Health Technol Assess. 2007;11:1–231. doi: 10.3310/hta11070. https://doi.org/10.3310/hta11070. [DOI] [PubMed] [Google Scholar]
- 23.Kanis JA, Johansson H, Johnell O, Oden A, De Laet C, Eisman JA, et al. Alcohol intake as a risk factor for fracture. Osteoporos Int. 2005;16:737–42. doi: 10.1007/s00198-004-1734-y. https://doi.org/10.1007/s00198-004-1734-y. [DOI] [PubMed] [Google Scholar]
- 24.Sarkis KS, Salvador MB, Pinheiro MM, Silva RG, Zerbini CA, Martini LA. Association between osteoporosis and rheumatoid arthritis in women: a cross-sectional study. Sao Paulo Med J. 2009;127:216–22. doi: 10.1590/S1516-31802009000400007. https://doi.org/10.1590/S1516-31802009000400007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Panel on Prevention of Falls in Older Persons, American Geriatrics Society and British Geriatrics Society. Summary of the Updated American Geriatrics Society/British Geriatrics Society clinical practice guideline for prevention of falls in older persons. J Am Geriatr Soc. 2011;59:148–57. doi: 10.1111/j.1532-5415.2010.03234.x. https://doi.org/10.1111/j.1532-5415.2010.03234.x. [DOI] [PubMed] [Google Scholar]
- 26.Kanis JA on behalf of the World Health Organization Scientific Group. Technical Report. World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield; UK: 2007. 2007. Assessment of osteoporosis at the primary health-care level. [Google Scholar]
- 27.Seeman E, Delmas PD. Bone quality--the material and structural basis of bone strength and fragility. N Engl J Med. 2006;354:2250–61. doi: 10.1056/NEJMra053077. https://doi.org/10.1056/NEJMra053077. [DOI] [PubMed] [Google Scholar]
- 28.Schousboe JTSJ, Bilezikian JP, Baim S. Executive Summary of the 2013 ISCD Position Development Conference on Bone Densitometry. J Clin Densitom. 2013;16:455–66. doi: 10.1016/j.jocd.2013.08.004. https://doi.org/10.1016/j.jocd.2013.08.004. [DOI] [PubMed] [Google Scholar]
- 29.Nelson HD, Haney EM, Chou R, Dana T, Fu R, Bougatsos C. Evidence Synthesis No 77. Rockville, Maryland: Agency for Healthcare Research and Quality; Jul, 2010. Screening for osteoporosis: systematic review to update the 2002. U.S Preventive Services Task Force Recommendation. AHRQ Publication 10-05145-EF-1. [PubMed] [Google Scholar]
- 30.Kanis JA, McCloskey EV, Johansson H, Strom O, Borgstrom F, Oden A. Case finding for the management of osteoporosis with FRAX--assessment and intervention thresholds for the UK. Osteoporos Int. 2008;19:1395–408. doi: 10.1007/s00198-008-0712-1. https://doi.org/10.1007/s00198-008-0712-1. [DOI] [PubMed] [Google Scholar]
- 31.Vasikaran S, Eastell R, Bruyere O, Foldes AJ, Garnero P, Griesmacher A, et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int. 2011;22:391–420. doi: 10.1007/s00198-010-1501-1. https://doi.org/10.1007/s00198-010-1501-1. [DOI] [PubMed] [Google Scholar]
- 32.Tannenbaum C, Clark J, Schwartzman K, Wallenstein S, Lapinski R, Meier D, et al. Yield of laboratory testing to identify secondary contributors to osteoporosis in otherwise healthy women. J Clin Endocrinol Metab. 2002;87:4431–7. doi: 10.1210/jc.2002-020275. https://doi.org/10.1210/jc.2002-020275. [DOI] [PubMed] [Google Scholar]
- 33.Ross AC, Taylor CL, Yaktine AL, et al., editors. Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Dietary Reference Intakes for Calcium and Vitamin D. Washington (DC): National Academies Press (US); 2011. Available from: http://www.ncbi.nlm.nih.gov/books/NBK56070/ [DOI] [PubMed] [Google Scholar]
- 34.Moyer VA. Vitamin D and calcium supplementation to prevent fractures in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;158:691–6. doi: 10.7326/0003-4819-158-9-201305070-00603. https://doi.org/10.7326/0003-4819-158-6-201303190-00588. [DOI] [PubMed] [Google Scholar]
- 35.Bischoff-Ferrari HA, Willett WC, Wong JB, Giovannucci E, Dietrich T, Dawson-Hughes B. Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA. 2005;293:2257–64. doi: 10.1001/jama.293.18.2257. https://doi.org/10.1001/jama.293.18.2257. [DOI] [PubMed] [Google Scholar]
- 36.Bischoff-Ferrari HA, Dawson-Hughes B, Willett WC, Staehelin HB, Bazemore MG, Zee RY, et al. Effect of Vitamin D on falls: a meta-analysis. JAMA. 2004;291:1999–2006. doi: 10.1001/jama.291.16.1999. https://doi.org/10.1001/jama.291.16.1999. [DOI] [PubMed] [Google Scholar]
- 37.Hallström H, Wolk A, Glynn A, Michaëlsson K. Coffee, tea and caffeine consumption in relation to osteoporotic fracture risk in a cohort of Swedish women. Osteoporos Int. 2006;17:1055–64. doi: 10.1007/s00198-006-0109-y. https://doi.org/10.1007/s00198-006-0109-y. [DOI] [PubMed] [Google Scholar]
- 38.Rizzoli R, Bianchi ML, Garabédian M, McKay HA, Moreno LA. Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone. 2010;46:294–305. doi: 10.1016/j.bone.2009.10.005. https://doi.org/10.1016/j.bone.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 39.Kelley GA, Kelley KS, Tran ZV. Exercise and lumbar spine bone mineral density in postmenopausal women: a meta-analysis of individual patient data. J Gerontol A Biol Sci Med Sci. 2002;57:M599–604. doi: 10.1093/gerona/57.9.m599. https://doi.org/10.1093/gerona/57.9.M599. [DOI] [PubMed] [Google Scholar]
- 40.Robertson MC, Campbell AJ, Gardner MM, Devlin N. Preventing injuries in older people by preventing falls: a meta-analysis of individual-level data. J Am Geriatr Soc. 2002;50:905–11. doi: 10.1046/j.1532-5415.2002.50218.x. https://doi.org/10.1046/j.1532-5415.2002.50218.x. [DOI] [PubMed] [Google Scholar]
- 41.Bone HG, Hosking D, Devogelaer JP, Tucci JR, Emkey RD, Tonino RP, et al. Ten years’ experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med. 2004;350:1189–99. doi: 10.1056/NEJMoa030897. https://doi.org/10.1056/NEJMoa030897. [DOI] [PubMed] [Google Scholar]
- 42.Eastell R, Devogelaer JP, Peel NF, Chines AA, Bax DE, Sacco-Gibson N, et al. Prevention of bone loss with risedronate in glucocorticoid-treated rheumatoid arthritis patients. Osteoporos Int. 2000;11:331–7. doi: 10.1007/s001980070122. https://doi.org/10.1007/s001980070122. [DOI] [PubMed] [Google Scholar]
- 43.Chesnut CH, 3rd, Skag A, Christiansen C, Recker R, Stakkestad JA, Hoiseth A, et al. Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004;19:1241–9. doi: 10.1359/JBMR.040325. https://doi.org/10.1359/JBMR.040325. [DOI] [PubMed] [Google Scholar]
- 44.Reid DM, Devogelaer JP, Saag K, Roux C, Lau CS, Reginster JY, et al. Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (Horizon): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet. 2009;373:1253–63. doi: 10.1016/S0140-6736(09)60250-6. https://doi.org/10.1016/S0140-6736(09)60250-6. [DOI] [PubMed] [Google Scholar]
- 45.Shane E, Burr D, Ebeling PR, Abrahamsen B, Adler RA, Brown TD, et al. Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2010;25:2267–94. doi: 10.1002/jbmr.253. https://doi.org/10.1002/jbmr.253. [DOI] [PubMed] [Google Scholar]
- 46.Siris ES, Harris ST, Eastell R, Zanchetta JR, Goemaere S, Diez-Perez A, et al. Skeletal effects of raloxifene after 8 years: results from the continuing outcomes relevant to Evista (Core) study. J Bone Miner Res. 2005;20:1514–24. doi: 10.1359/JBMR.050509. https://doi.org/10.1359/JBMR.050509. [DOI] [PubMed] [Google Scholar]
- 47.Seeman E. Strontium ranelate: vertebral and non-vertebral fracture risk reduction. Curr Opin Rheumatol. 2006;18(Suppl 1):S17–20. doi: 10.1097/01.bor.0000229523.89546.32. https://doi.org/10.1097/01.bor.0000229523.89546.32. [DOI] [PubMed] [Google Scholar]
- 48.Committee for Medicinal Products for Human Use. Summary of opinion (post authorisation): treatment of osteoporosis in men at increased risk of fracture. European Medicines Agency Website. [Accessed June 12, 2012]. http://www.ema.europa.eu/docs/en_GB/document_library/Summary_of_opinion/human/000560/WC500127765.pdfPublished May 24, 2012. - See more at: http://press.endocrine.org/doi/full/10.1210/jc.2012-3048#sthash.M7MrgBEV.dpuf.
- 49.Bone HG, Chapurlat R, Brandi ML, Brown JP, Czerwinski E, Krieg MA, et al. The effect of three or six years of denosumab exposure in women with postmenopausal osteoporosis: results from the freedom extension. J Clin Endocrinol Metab. 2013;98:4483–92. doi: 10.1210/jc.2013-1597. https://doi.org/10.1210/jc.2013-1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, et al. Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001;344:1434–41. doi: 10.1056/NEJM200105103441904. https://doi.org/10.1056/NEJM200105103441904. [DOI] [PubMed] [Google Scholar]