Abstract
Objective:
Diabetes mellitus causes a decrease in cardiac output, arterial blood pressure, and heart rate. In this study, we aimed to investigate, at the molecular level, the effect of nitric oxide synthase (NOS) on heart pathology in type 1 diabetes and look at the therapeutic effect of pentoxifylline on this pathology.
Methods:
In this experimental study, 50 Wistar albino male rats were used. The rats were divided into 5 groups: group C, control; group D, only diabetes; group D+PI and D+PII, diabetes + pentoxifylline; group P, only pentoxifylline. Group D+PI rats received 50 mg/kg/day pentoxifylline over two months. However, group D+PII rats received saline in the first month and 50 mg/kg/day of pentoxifylline over the following month. At the end of two months, NOS expressions in heart tissue were assessed through immunohistochemistry analysis. The data were compared by one-way ANOVA.
Results:
At the end of the experiments, there was increased cytoplasmic vacuolization, myofibrillar loss, cytoplasmic eosinophilia, and degeneration of cardiomyocytes; nNOS and iNOS expressions in group D decreased compared with that in group C. In group D+PI and group D+PII, nNOS and iNOS expressions improved compared with group D.
Conclusion:
As a result, we found that diabetes, a known chronic disease, causes serious damage in heart tissue. NOS plays a role in this damage, and pentoxifylline aided in improving nNOS and iNOS expression in this damage.
Keywords: diabetes mellitus, eNOS, iNOS, nNOS, heart, rat
Introduction
Diabetes mellitus (DM) is one of the most serious diseases threatening human health in the modern world. According to the World Health Organization’s forecast, the number of diabetic patients worldwide will reach up to 366 million by 2030. DM is a major chronic diseases that occurs as a result of insufficient insulin production or insulin resistance in peripheral tissues (1).
It has been shown that DM causes a decrease in cardiac output, arterial blood pressure, and heart rate due to hyperglycemia, particularly in animal models. In addition, it causes endothelial dysfunction and response inhibition to vasoactive agents. As a result, it causes an almost five-fold increase in cardiovascular disease risk (2, 3). Diabetic cardiomyopathy (DCM) occurs independently of coronary artery disease and hypertension in diabetic patients. The following develop in DCM: myocardial fibrosis, interstitial inflammation, endothelial dysfunction, and weakened cardiac function (4, 5). Therefore, DCM is one of the major complications of DM (6).
Nitric oxide (NO) is produced by nitric oxide synthase (NOS). NOS converts the terminal guanidine group of L-arginine amino acid to NO (7, 8). Three forms of NOS are expressed in mammalian cells: neuronal NOS (nNOS, NOS1), inducible NOS (iNOS, NOS2), and endothelial NOS (eNOS, NOS3) (9, 10). In cardiac tissue, nNOS, iNOS, and eNOS are present in subcellular locations (11).
Pentoxifylline (PTX) is a methyl xanthine derivative used in the treatment of peripheral vascular diseases; it improves erythrocyte deformity and capillary circulation (12). PTX inhibits transcription of some cytokines, such as tumor necrosis factor-alpha (TNF-a), adhesion of platelets, and maintains endothelium derived vascular relaxation (12, 13). It also has antioxidant properties (14, 15).
Induction of NOS and oxidative stress originating from an imbalance in the production of reactive oxygen species (ROS) are thought to be responsible for the cardiovascular complications of DM (16). Therefore, in this study, we aimed to investigate the role of NOS in myocardial damage caused by streptozotocin (STZ)-induced type 1 DM and the effects of PTX, as an antioxidant in this damage and NO system.
Methods
Sexually mature 8-weeks-old male Wistar rats, obtained from the Hakan Çetinsaya Experimental and Clinic Research Center, Erciyes University, Kayseri, Turkey, were used for this study. They were housed in plastic cages, placed in a well-ventilated rat house, allowed ad libitum access to rat chow and water, and were subjected to a natural photoperiod of 12-h light/dark cycle. This study was carried out in strict accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The study protocol was approved by the Committee on the Ethics of Animal Experiments of the University of Erciyes (Permit Number: 12/32). The rats were randomly assigned to five groups of ten rats per group. The animals received 40 mg/kg intraperitoneal streptozotosin (sc-200719, Santa Cruz Biotechnology, CA, USA) to induce diabetes (17). Seventy-two hours following this application, blood glucose levels were measured from the tails of the rat. The rats whose measured blood glucose levels were over 250 mg/dL were considered to be diabetic. PTX (Trental 100 mg/5 mL, Sanofi Aventis, Paris, France) 50 mg/kg/day was administered by intraperitoneal injection to the rats (18). Group C (control) and group D (diabetic) were administered intraperitoneal saline. Group D+PI was administered PTX from the beginning of the experiment. Group D+PII received saline for the first month and then PTX was injected during the subsequent month. Group P received only PTX from beginning of the experiment.
At the end of the second month, before the rats were decapitated, their weights and blood glucose levels from their tails were measured again. Then the subjects were decapitated under ketamine (75 mg/kg) + xylazine (10 mg/kg) anesthesia, their blood was collected, and heart was removed and weighted. Heart tissues were placed in formalin fixation solution for histological examination.
Immunohistochemistry
To determine the differences in expression with eNOS (1/250 dilution, PA3-031A, Pierce antibody product, Rockford, IL USA), iNOS (1/200 dilution, PA3- 030A, Pierce antibody product, Rockford, IL USA) and nNOS (1/200 dilution, PA3-032A, Pierce antibody product, Rockford, IL USA) and nNOS (Pierce antibody product, PA3-032A, 1/200 dilution) primary antibodies in heart tissue with the streptavidin-biotin-peroxidase technique, as previously described (19). Under the light microscope Under the light microscope (Olympus BX51, Center Valley, PA, USA) and with a digital camera (DP71, Olympus, Center Valley, PA, USA), images were obtained. From each of the subjects, five different areas were evaluated in terms of the expression differences using the image J program.
Apoptosis (TUNEL)
The Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) method was utilized to show apoptosis of heart tissue, as previously described (20). An in situ Cell Death Detection Kit Fluorescein Kit (11684795910, Roche, Mannheim, Germany) was used. After casing the tissues with solution containing glycerol, they were all examined with the Olympus BX51 fluorescence microscope at 450-500 nm wavelength. Cells were considered to be apoptotic when the cell nuclei demonstrated positive TUNEL staining. For quantification of TUNEL-positive cells, 10 fields per section were examined and counted at 400-fold magnification.
Statistical analysis
All statistical analyses were carried out using SPSS statistical software (SPSS for windows, SPSS Inc, Chicago, IL, USA, version 15.0). The Kolmogorov-Smirnov test was used to identify normal distribution of data. In case of normal distribution, quantitative variables were compared using one-way analysis of variance (ANOVA) and posthoc Tukey test. Results are presented as mean±SEM. A p value of <0.05 was considered statistically significant.
Results
At the beginning of experiment, the body weights of the rats were similar to each other and blood sugar levels were normal. However, by the end of the experiment, in groups D, D+PI, and D+PII, the average body weights had significantly decreased compared with that of the control group. Similarly, in rats belonging to the diabetes groups (group D, group D+PI, and group D+PII), the blood sugar levels had significantly increased compared with that of the control group. When heart weight was considered, it was found that groups D, D+PI and D+PII had decreased heart weights compared to group C. The body weight and blood sugar level of the group P was similar group C (Table 1).
Table 1.
Body weight, heart weight, blood glucose levels, and apoptotic index of control and streptozotocin (STZ)-induced diabetic rats
| Body weight (g) (n=10) | Heart weight (g) (n=10) | Blood glucose (mg/dL) (n=10) | Apoptotic index (n=100) | |
|---|---|---|---|---|
| Group C | 282.12±9.74 | 0.88±0.04 | 119.25±3.84 | 0.30±0.11 |
| Group D | 190.20±13.23* | 0.72±0.04* | 565.50±23.11* | 0.63±0.24 |
| Group D+PI | 221.77±19.43* | 0.73±0.04 | 545.44±25.54* | 0.73±0.25 |
| Group D+PII | 198.22±14.00* | 0.68±0.04* | 531.88±36.30* | 0.40±0.24 |
| Group P | 300.00±12.35 | 0.80±0.01 | 122.00±8.69 | 0.06±0.04 |
Values are expressed as mean±SEM.
P<0.05 compared to group C. Statistical analysis was used: one-way ANOVA and posthoc Tukey test
Histological findings
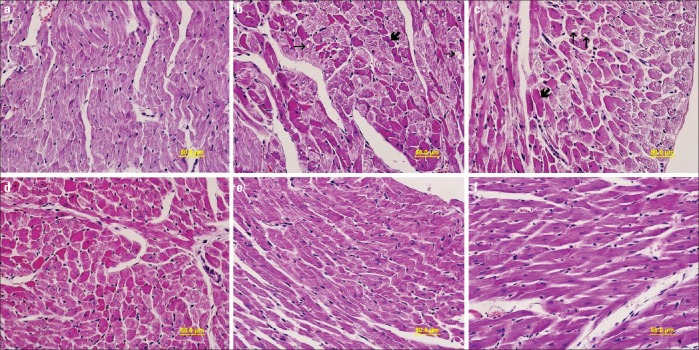
Microscopic examination of heart tissue belonging to the control group subjects exhibited normal histomorphology. In group D, which comprised of heart tissues of diabetic rats and in whom diabetes was induced with STZ, increased cytoplasmic vacuolization, myofibrillar loss, cytoplasmic eosinophilia of some cells, and degeneration of some cardiomyocytes were observed. These damages tended to be decreased in groups D+PI and D+PII, which had been treated with PTX. The heart histology of the group treated only with PTX was similar to that of the control group (Fig. 1).
Figure 1.
a–f. Light microscopy of heart tissue in different groups: (a) group C, normal heart architecture was observed; (b) group D, increased cytoplasmic vacuolization (thin arrow) and cytoplasmic eosinophilia of some cells (thick arrow) were exhibited; (c) group D, cytoplasmic vacuolization (thin arrow), cytoplasmic eosinophilia of some cells (thick arrow), and myofibrillar loss (red arrow) were particularly increased in the peripheral myocardium; (d) group D+PI, mild heart damage was observed; (e) group D+PII, normal heart architecture was observed; and (f) group P, normal heart architecture was observed. Hematoxylin & eosin
Immunohistochemistry findings
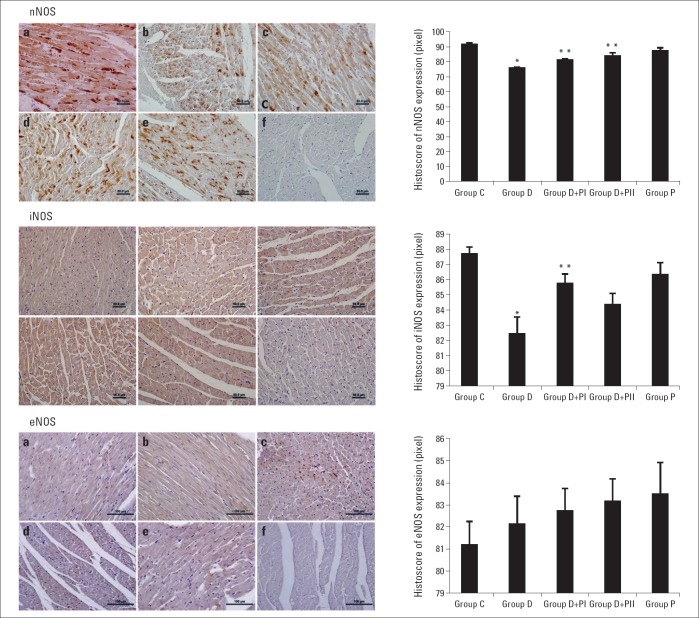
Immunohistochemical staining was performed using the avidin-biotin method to determine the heart tissue expression of NOS. Each of the three NOS (iNOS, eNOS, and nNOS) expressions were observed in cardiomyocytes. iNOS and eNOS immunoreactivity showed homogeneous distribution pattern; however, nNOS immunoreactivity showed heterogeneous distribution pattern. In group D, nNOS (p=0.000) and iNOS (p=0.000)expressions had statistically decreased compared with that in group C. In groups D+PI and D+PII, nNOS (p=0.049, p=0.001 respectively) and iNOS expressions (p=0.039, p=0.472 respectively) had significantly improved compared to that in group D. The nNOS and iNOS expressions of group P were similar to group C. eNOS expressions were similar in all groups. (Fig. 2, Table 2).
Figure 2.
a-f. Immunohistochemical localization of neuronal, inducible, and endothelial NOS expression of the heart tissue and semiquantitative results of NOS content obtained by densitometric analysis of immunohistochemistry in different groups: (a) group C, (b) group D, (c) group D+PI, (d) group D+PII, (e) group P, and (f) negative controls. (Statistical analysis was used One-way ANOVA, posthoc Tukey test)
Table 2.
nNOS, iNOS, and eNOS immunoreactivity scores of control and streptozotocin (STZ)-induced diabetic rats
| nNOS (n=50) | iNOS (n=50) | eNOS (n=50) | |
|---|---|---|---|
| Group C | 91.05±1.47 | 87.69±0.51 | 81.16±1.09 |
| Group D | 75.54±0.65* | 82.51±1.10* | 82.11±1.28 |
| Group D+PI | 81.32±0.92** | 85.79±0.62** | 82.70±1.05 |
| Group D+PII | 84.06±2.23** | 84.38±0.74 | 83.17±1.01 |
| Group P | 87.85±2.19 | 86.28±0.87 | 83.46±1.43 |
Values are expressed as mean ± SEM.
p<0.05 compared to group C.
p<0.05 compared to group D. Statistical analysis was used: one-way ANOVA and posthoc Tukey test
Apoptosis (TUNEL) findings
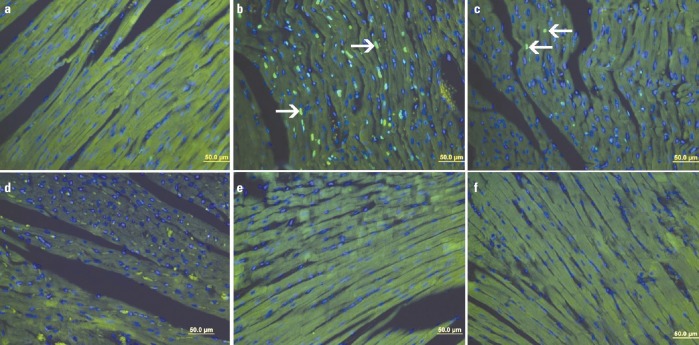
TUNEL staining was performed to determine apoptotic cells in heart tissue (Fig. 3). The apoptotic index results are given in Table 1. The numbers of apoptotic cells in heart tissues increased in group D and decreased in group D+PII. However, the apoptotic index in the heart tissues tended to decrease in group P compared to group C.
Figure 3.
a-f. TUNEL staining of heart tissue in different groups: (a) group C, (b) group D, (c) group D+PI, (d) group D+PII, (e) group P, and (f) negative controls. TUNEL-positive cells (arrow) were observed in the heart. (Statistical analysis was used One-way ANOVA, posthoc Tukey test)
Discussion
In the present study, NOS level changes caused by myocardial damage due to DM and effect of PTX on these changes, and the diabetic myocardium were investigated using histopathological and immunohistochemical techniques. According to the findings of the present study, there was damage in the diabetic myocardium and the NO system was affected in these tissues. PTX improved the damage and restored NOS levels.
There are many studies on the myocardial damage caused by DM. Li et al. (6) reported the cardiac muscle fibers derangement, widening gap, pyknotic and fragmented nuclei, and more apoptosis in the diabetic heart. They also reported increased serum malondialdehyde (MDA) levels. Abdel-Raheem et al. (21) reported atrophy in some cardiomyocytes and an increase in TUNEL-positive cell number in diabetic cardiac tissue. TUNEL-positive cell number increase was reported in another study together with an increase in MDA content in the myocardial mitochondria and in collagen density in the interstitium (22). Parallel to the above studies, due to the unfavorable microenvironment caused by DM, cytoplasmic vacuolization, myofibril loss in the myocytes, and an increase in apoptotic cell number were observed in group D cardiac tissues in the present study. Also, there was an increase in cytoplasmic eosinophilia and degeneration in some myocytes in this group.
Oxidative stress is thought to be responsible for the cardiovascular complications of DM and has been investigated in many studies. There are three major sources of free radicals in the diabetic myocardium: mitochondria, NADPH oxidase, and NOS (3). NOS is responsible for NO synthesis from L-arginine in mammalian cells. There are three isoforms of NOS: neuronal NOS (nNOS, NOS1), inducible NOS (iNOS, NOS2), and endothelial NOS (eNOS, NOS3). nNOS and eNOS are calmodulin-dependent enzymes, whereas iNOS is not (23). Changes in NOS enzymes in the diabetic myocardium have been investigated in many studies. Increased myocardial nNOS levels were observed in STZ-induced diabetic rats when compared with that of the control group (24). However, no amounts of nNOS mRNA were detected in another study (25). In Nagareddy et al.’s (26) study, although there was no difference in expression of iNOS and eNOS between diabetic and control groups in the first week, eNOS expression later decreased significantly while iNOS expression increased. Similarly, in another study, an increase in iNOS and a decrease in eNOS expression in the superior mesenteric artery and myocardium of diabetic rats, respectively, were shown (2). On the other hand, Felaco et al. (27) showed that there was no significant difference in eNOS expression in cardiomyocytes and the vessel wall between the diabetic and control groups, although there was positive immunoreactivity in eNOS expression. Considering the above studies, it is clear that the findings on NOS expression in the diabetic myocardium are incompatible. In the present study, all isoforms of NOS were detected in the cardiomyocytes. In all sections, iNOS and eNOS staining were similar in all cardiomyocytes, i.e., the stainings showed homogeneity. However, in some cells nNOS staining was denser, although all cardiomyocytes showed staining. In the diabetic group, nNOS and iNOS immunoreactivity were significantly decreased compared with that of the control group, whereas eNOS immunoreactivity was similar.
In DM, oxidative stress-induced myocardial damage is caused by an imbalance in the production of reactive oxygen and nitrogen species and insufficient antioxidant defense (3). It is known that an increase in ROS in the diabetic heart contributes to the pathophysiology of DCM (28, 29). In many studies, the administration of antioxidant agents, such as vitamin E, carvedilol (21), alpha-lipoic acid (22), and caffeic acid (30), was investigated to relieve the harmful effects of DM on cardiac tissue.
PTX has been used in peripheral and cerebral vascular diseases to improve erythrocyte deformity and capillary circulation (12, 31). Myocardial damage is caused by increased calcium concentration due to ischemia and production of endotoxins and TNF-a (32). PTX inhibits the transcription of TNF-a and other cytokines. It also inhibits the adhesion of platelets and maintains endothelium-derived vascular relaxation (6, 13). As a result, it increases blood flow and improves tissue oxygenation (33). In addition, it has been used as an antioxidant to heal damage in several tissues (15, 34). In accordance with these effects, in the present study, there was an improvement in cardiomyocytes in groups P+I and P+II compared with that in group D. Also, there was a significant an improvement in nNOS and iNOS immunoreactivity in these groups compared to that in group D. The nNOS, iNOS, and eNOS expressions of group P were similar to those of group C. These results indicate that PTX decreases nitrosative stress by endothelial-dependent vascular relaxation and provides healing.
Study limitations
The major limitation of the present study was not confined to NOS expression in heart tissue by Western blot or real-time PCR. The other limitations were not considered to assess the oxidative parameters in the heart tissue. Further studies will be conducted to measure NOS expression by Western blot and real-time PCR. Also, the possible mechanism of PTX that ameliorates diabetic cardiac damage will be clarified in further studies.
Conclusion
There are structural and functional changes in the myocardium of STZ-induced diabetic rats. There are also changes in the expressions of NOS, particularly iNOS and nNOS. Therefore, these enzymes may play a role in the pathophysiology of myocardial damage. PTX has therapeutic effect in this damage by particularly affecting nNOS and iNOS expression.
Footnotes
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept - M.F.S.; Design - M.F.S., H.B.U.; Supervision - M.F.S.; Materials - M.F.S., H.B.U.; Research - D.K., E.K.; Data collection &/or processing - D.K., E.K.; Literature search - D.K., E.K.; Writing - D.K., H.B.U., M.F.S.; Critical review - H.B.U., M.F.S.
References
- 1.Roessner C, Paasch U, Kratzsch J, Glander HJ, Grunewald S. Sperm apoptosis signalling in diabetic men. Reprod Biomed Online. 2012;25:292–9. doi: 10.1016/j.rbmo.2012.06.004. [DOI] [PubMed] [Google Scholar]
- 2.Nagareddy PR, McNeill JH, MacLeod KM. Chronic inhibition of inducible nitric oxide synthase ameliorates cardiovascular abnormalities in streptozotocin diabetic rats. Eur J Pharmacol. 2009;611:53–9. doi: 10.1016/j.ejphar.2009.03.061. [DOI] [PubMed] [Google Scholar]
- 3.Ansley DM, Wang B. Oxidative stress and myocardial injury in the diabetic heart. J Pathol. 2013;229:232–41. doi: 10.1002/path.4113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Soliman H, Craig GP, Nagareddy P, Yuen VG, Lin G, Kumar U, et al. Role of inducible nitric oxide synthase in induction of RhoA expression in hearts from diabetic rats. Cardiovasc Res. 2008;79:322–30. doi: 10.1093/cvr/cvn095. [DOI] [PubMed] [Google Scholar]
- 5.Van Linthout S, Riad A, Dhayat N, Spillmann F, Du J, Dhayat S, et al. Anti-inflammatory effects of atorvastatin improve left ventricular function in experimental diabetic cardiomyopathy. Diabetologia. 2007;50:1977–86. doi: 10.1007/s00125-007-0719-8. [DOI] [PubMed] [Google Scholar]
- 6.Li J, Peng L, Du H, Wang Y, Lu B, Xu Y, et al. The protective effect of beraprost sodium on diabetic cardiomyopathy through the Inhibition of the p38 MAPK signaling pathway in high-fat-induced SD Rats. Int J Endocrinol. 2014;2014:901437. doi: 10.1155/2014/901437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sönmez MF, Narin F, Akkuş D, Özdamar S. Effect of melatonin and vitamin C on expression of endothelial NOS in heart of chronic alcoholic rats. Toxicol Ind Health. 2009;25:385–93. doi: 10.1177/0748233709106444. [DOI] [PubMed] [Google Scholar]
- 8.Liu VW, Huang PL. Cardiovascular roles of nitric oxide: a review of insights from nitric oxide synthase gene disrupted mice. Cardiovasc Res. 2008;77:19–29. doi: 10.1016/j.cardiores.2007.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sönmez MF, Narin F, Balcıoğlu E. Melatonin and vitamin C attenuates alcohol-induced oxidative stress in aorta. Basic Clin Pharmacol Toxicol. 2009;105:410–5. doi: 10.1111/j.1742-7843.2009.00469.x. [DOI] [PubMed] [Google Scholar]
- 10.Sönmez MF, Narin F, Akkuş D, Türkmen AB. Melatonin and vitamin C ameliorate alcohol-induced oxidative stress and eNOS expression in rat kidney. Ren Fail. 2012;34:480–6. doi: 10.3109/0886022X.2011.649678. [DOI] [PubMed] [Google Scholar]
- 11.Umar S, van der Laarse A. Nitric oxide and nitric oxide synthase isoforms in the normal, hypertrophic, and failing heart. Mol Cell Biochem. 2010;333:191–201. doi: 10.1007/s11010-009-0219-x. [DOI] [PubMed] [Google Scholar]
- 12.Wang W, Zolty E, Falk S, Basava V, Reznikov L, Schrier R. Pentoxifylline protects against endotoxin-induced acute renal failure in mice. Am J Physiol Renal Physiol. 2006;291:F1090–5. doi: 10.1152/ajprenal.00517.2005. [DOI] [PubMed] [Google Scholar]
- 13.Motawi TK, Darwish HA, Abd El Tawab AM. The relative efficacy of aminoguanidine and pentoxifylline in modulating endotoxin-induced cardiac stress. Cell Biochem Funct. 2011;29:694–702. doi: 10.1002/cbf.1809. [DOI] [PubMed] [Google Scholar]
- 14.Stosic-Grujicic S, Maksimovic D, Badovinac V, Samardzic T, Trajkovic V, Lukic M, et al. Antidiabetogenic effect of pentoxifylline is associated with systemic and target tissue modulation of cytokines and nitric oxide production. J Autoimmun. 2001;16:47–58. doi: 10.1006/jaut.2000.0456. [DOI] [PubMed] [Google Scholar]
- 15.Garcia FA, Pinto SF, Cavalcante AF, Lucetti LT, Menezes SM, Felipe CF, et al. Pentoxifylline decreases glycemia levels and TNF-alpha, iNOS and COX-2 expressions in diabetic rat pancreas. Springerplus. 2014;3:283. doi: 10.1186/2193-1801-3-283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jo H, Otani H, Jo F, Shimazu T, Okazaki T, Yoshioka K, et al. Inhibition of nitric oxide synthase uncoupling by sepiapterin improves left ventricular function in streptozotocin-induced diabetic mice. Clin Exp Pharmacol Physiol. 2011;38:485–93. doi: 10.1111/j.1440-1681.2011.05535.x. [DOI] [PubMed] [Google Scholar]
- 17.Rathinam A, Pari L, Chandramohan R, Sheikh BA. Histopathological findings of the pancreas, liver, and carbohydrate metabolizing enzymes in STZ-induced diabetic rats improved by administration of myrtenal. J Physiol Biochem. 2014;70:935–46. doi: 10.1007/s13105-014-0362-z. [DOI] [PubMed] [Google Scholar]
- 18.Queiroz-Junior CM, Bessoni RL, Costa VV, Souza DG, Teixeira MM, Silva TA. Preventive and therapeutic anti-TNF-alpha therapy with pentoxifylline decreases arthritis and the associated periodontal co-morbidity in mice. Life Sci. 2013;93:423–8. doi: 10.1016/j.lfs.2013.07.022. [DOI] [PubMed] [Google Scholar]
- 19.Ozan E, Sönmez MF, Ozan S, Çolakoğlu N, Yılmaz S, Kuloğlu T. Effects of melatonin and vitamin C on cigarette smoke-induced damage in the kidney. Toxicol Ind Health. 2007;23:479–85. doi: 10.1177/0748233708089023. [DOI] [PubMed] [Google Scholar]
- 20.Bayatlı F, Akkuş D, Kılıç E, Saraymen R, Sönmez MF. The protective effects of grape seed extract on MDA, AOPP, apoptosis and eNOS expression in testicular torsion: an experimental study. World J Urol. 2013;31:615–22. doi: 10.1007/s00345-013-1049-8. [DOI] [PubMed] [Google Scholar]
- 21.Abdel-Raheem MH, Salim SU, Mosad E, Al-Rifaay A, Salama HS, Hasan-Ali H. Antiapoptotic and antioxidant effects of carvedilol and vitamin E protect against diabetic nephropathy and cardiomyopathy in diabetic wistar albino rats. Horm Metab Res. 2015;47:97–106. doi: 10.1055/s-0034-1385855. [DOI] [PubMed] [Google Scholar]
- 22.Li CJ, Zhang QM, Li MZ, Zhang JY, Yu P, Yu DM. Attenuation of myocardial apoptosis by alpha-lipoic acid through suppression of mitochondrial oxidative stress to reduce diabetic cardiomyopathy. Chin Med J (Engl) 2009;122:2580–6. [PubMed] [Google Scholar]
- 23.Dixit VD, Parvizi N. Nitric oxide and the control of reproduction. Anim Reprod Sci. 2001;65:1–16. doi: 10.1016/s0378-4320(00)00224-4. [DOI] [PubMed] [Google Scholar]
- 24.Le Douairon Lahaye S, Rebillard A, Zguira MS, Malarde L, Saiag B, Gratas-Delamarche A, et al. Effects of exercise training combined with insulin treatment on cardiac NOS1 signaling pathways in type 1 diabetic rats. Mol Cell Biochem. 2011;347:53–62. doi: 10.1007/s11010-010-0611-6. [DOI] [PubMed] [Google Scholar]
- 25.Stockklauser-Farber K, Ballhausen T, Laufer A, Rosen P. Influence of diabetes on cardiac nitric oxide synthase expression and activity. Biochim Biophys Acta. 2000;1535:10–20. doi: 10.1016/s0925-4439(00)00078-8. [DOI] [PubMed] [Google Scholar]
- 26.Nagareddy PR, Xia Z, McNeill JH, MacLeod KM. Increased expression of iNOS is associated with endothelial dysfunction and impaired pressor responsiveness in streptozotocin-induced diabetes. Am J Physiol Heart Circ Physiol. 2005;289:H2144–52. doi: 10.1152/ajpheart.00591.2005. [DOI] [PubMed] [Google Scholar]
- 27.Felaco M, Grilli A, De Lutiis MA, Patruno A, Libertini N, Taccardi AA, et al. Endothelial nitric oxide synthase (eNOS) expression and localization in healthy and diabetic rat hearts. Ann Clin Lab Sci. 2001;31:179–86. [PubMed] [Google Scholar]
- 28.Cai L, Wang Y, Zhou G, Chen T, Song Y, Li X, et al. Attenuation by metallothionein of early cardiac cell death via suppression of mitochondrial oxidative stress results in a prevention of diabetic cardiomyopathy. J Am Coll Cardiol. 2006;48:1688–97. doi: 10.1016/j.jacc.2006.07.022. [DOI] [PubMed] [Google Scholar]
- 29.Cai L. Suppression of nitrative damage by metallothionein in diabetic heart contributes to the prevention of cardiomyopathy. Free Radic Biol Med. 2006;41:851–61. doi: 10.1016/j.freeradbiomed.2006.06.007. [DOI] [PubMed] [Google Scholar]
- 30.Ho YJ, Lee AS, Chen WP, Chang WL, Tsai YK, Chiu HL, et al. Caffeic acid phenethyl amide ameliorates ischemia/reperfusion injury and cardiac dysfunction in streptozotocin-induced diabetic rats. Cardiovasc Diabetol. 2014;13:98. doi: 10.1186/1475-2840-13-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lin SL, Chiang WC, Chen YM, Lai CF, Tsai TJ, Hsieh BS. The renoprotective potential of pentoxifylline in chronic kidney disease. J Chin Med Assoc. 2005;68:99–105. doi: 10.1016/S1726-4901(09)70228-X. [DOI] [PubMed] [Google Scholar]
- 32.Shaw SM, Shah MK, Williams SG, Fildes JE. Immunological mechanisms of pentoxifylline in chronic heart failure. Eur J Heart Fail. 2009;11:113–8. doi: 10.1093/eurjhf/hfn040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aydın K, Şahin V, Gürsu S, Mercan AS, Demir B, Yıldırım T. Effect of pentoxifylline on fracture healing: an experimental study. Eklem Hastalık Cerrahisi. 2011;22:160–5. [PubMed] [Google Scholar]
- 34.Zhang J, Ling Y, Tang L, Luo B, Chacko BK, Patel RP, et al. Pentoxifylline attenuation of experimental hepatopulmonary syndrome. J Appl Physiol (1985) 2007;102:949–55. doi: 10.1152/japplphysiol.01048.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]