Abstract
Objective:
To evaluate the short- and mid-term effects of percutaneous mitral balloon valvuloplasty (PMBV) on right ventricular functions in mitral stenosis.
Methods:
A prospective study was conducted in 61 patients who had mitral stenosis in normal sinus rhythm (68% female, age: 42±11-16 years). Right ventricular functions were measured before, immediately after, and at 3 months and 1 year after PMBV by conventional and tissue Doppler echocardiography imaging methods. Additionally, the patients were evaluated in two groups (PAP≥40 mm Hg, n: 46; PAP<40 mm Hg, n: 15) according to the systolic pulmonary artery that was measured by echocardiography prior to PMBV.
Results:
Post-PMBV mean gradient, pulmonary artery pressure (PAP), and left atrial size decreased significantly, and the mitral valve area increased significantly in both patient groups. This significance in pulmonary artery pressure was lost at 1 year. The significant post-PMBV increase in tricuspid annular point systolic excursion (TAPSE), systolic velocity, early diastolic velocity, and peak myocardial velocity during isovolumic contraction (IVV), indicating right ventricular functions, disappeared at 1 year. The significant post-PMBV decrease in myocardial performance index (MPI) and late diastolic velocity lost its significance at 1 year. No significant change was observed in myocardial acceleration during isovolumic contraction (IVA). The group with pulmonary hypertension demonstrated significance similar to the results of the overall group. Post-PMBV TAPSE, systolic velocity, early diastolic velocity, IVV, and IVA increased significantly, and this increase was maintained up to 1 year in the group without pulmonary hypertension. MPI and late diastolic velocity maintained their significantly decreased values up to 1 year.
Conclusion:
The positive effect of PMBV on right ventricular function in the acute period decreases and even disappears in the mid-term in patients developing pulmonary hypertension. Intervention in the patients prior to the development of hypertension is very important for the improvement in right ventricular functions.
Keywords: mitral stenosis, percutaneous mitral balloon valvuloplasty, right ventricular functions
Introduction
Several diseases have been acknowledged as pathological causes of mitral valve stenosis (MS), of which rheumatic heart disease is the most prevalent. Rheumatic heart disease is a chronic manifestation of rheumatic carditis, which occurs in 60% to 90% of cases of rheumatic fever (1).
Reduced exercise capacity and fatigue are common symptoms in patients with MS; increased pulmonary venous pressure and left atrium (LA) are not the solely responsible factors for these symptoms (2). Right ventricular (RV) function plays an important role in the development of clinical symptoms, exercise capacity, prognosis, and survival in MS (3, 4).
Right ventricle dysfunction, which emerges secondary to chronic pulmonary hypertension, is accepted as an important but undesired result of mitral stenosis. Percutaneous mitral balloon valvuloplasty is the most common type of treatment used in patients with mitral stenosis. The effect of successful percutaneous mitral balloon valvuloplasty (PMBV) on global RV systolic and diastolic functions in patients with rheumatic MS has not been well defined. Conventional echocardiography, Doppler tissue imaging, radionuclide ventriculography, and magnetic resonance imaging are the methods that are used to evaluate RV functions. Conventional two-dimensional (2-D) echocardiography and Doppler tissue imaging are methods of measuring systolic and diastolic velocities of annular motions. They are also potentially non-invasive and appropriate techniques and are less expensive than the others (5).
Previous studies have investigated the effect of right ventricle functions in patients with mitral stenosis (6-10). However, few studies have examined the effect of mitral valvuloplasty on the echocardiographic markers of RV systolic and diastolic functions in the short term and mid-term (4, 11, 12). The purpose of this study was to assess the impact of PMBV on RV function in the short-term and mid-term using two-dimensional and Doppler echocardiographic indices.
Methods
Subjects
This study was performed in our clinics between April 2008 and June 2010. A prospective study was conducted in 61 patients (68% female, age: 42.7±11.6 years) with isolated rheumatic mitral valve stenosis who underwent PMBV. Indications for PMBV were New York Heart Association class ≥II, ≤IV, planimetric mitral valve area (MVA), ≤1.5 cm2, mitral regurgitation ≤2+, suitable valve morphology, and the absence of concomitant cardiovascular disease requiring surgical correction. All patients had sinus rhythm. Additionally, the patients were evaluated in two groups (PAP≥40 mm Hg, n: 46; PAP<40 mm Hg, n: 15) according to the systolic pulmonary artery pressure (PAP) that was measured by echocardiography prior to PMBV. Detailed written informed consent was obtained from each patient. Approval of the study was obtained from the local ethics committee. The exclusion criteria were as follows: left ventricular ejection fraction (LVEF) <50%; aortic regurgitation greater than mild or aortic stenosis; mitral regurgitation greater than mild; clinical, echocardiographic, or angiographic evidence of coronary artery disease; hypertension; diabetes mellitus; severe calcification of mitral valve annulus; clinical or laboratory evidence of active rheumatic disease; chronic obstructive or restrictive lung disease; chronic pulmonary thromboembolism; and low-quality echocardiographic image for tissue Doppler imaging (TDI).
Echocardiographic study
A Vingmed System Five Doppler echocardiographic unit (GE Vingmed Ultrasound, Horten, Norway) with a 2.5-MHz FPA probe was used. Two-dimensional and pulse-wave Doppler echocardiographic studies were performed in the left lateral decubitus position with conventional views (parasternal long- and shortaxis, apical four-chamber) and the in the supine position for the subxiphoid approach. An electrocardiogram was recorded simultaneously with the M-mode and Doppler tracings on the same monitor, and a 50-mm/s M-mode sweeping speed was used for the M-mode trace recording. Maximum RA volumes were calculated by 2-D apical 2- and 4-chamber views using the area/length method. Tricuspid annular plane systolic excursion (TAPSE) was determined by the difference in the displacement of the RV base during systole and diastole (13). RV end-diastolic and end-systolic areas were measured from the apical 4-chamber view to calculate RV fractional area change (RVFAC) (14). Tricuspid inflow velocity was recorded from the apical 4-chamber view by pulsed-wave Doppler sample volume, positioned at the tips of the tricuspid leaflets during diastole. Peak early (E) and late (A) tricuspid inflow velocity and deceleration time of E velocity were obtained. The RV outflow velocity was recorded from the parasternal short-axis view with the pulsed-wave Doppler sample volume positioned just below the pulmonary valve. Pre-ejection period (PEP) was measured from the onset of the QRS wave to the onset of RV ejection flow. RV ejection time (RVET) was measured from the onset to the end of RV outflow. Isovolumetric relaxation time (IRT) was obtained as the time interval from the cessation of RV outflow to the onset of tricuspid valve inflow. Isovolumetric contraction time (ICT) was determined from the cessation of tricuspid inflow to the onset of RV outflow (15). Myocardial performance index (MPI) was calculated by the formula (ICT+IRT)/RVET (Fig. 1) (15, 16). Tissue Doppler imaging (TDI) was applied in the pulse-Doppler mode of the tricuspid annulus velocity at its lateral corners with the same echocardiographic unit, and systolic (S), early diastolic (E), and late diastolic (A) velocity; peak myocardial velocity during isovolumic contraction (IVV, cm/sec); and myocardial acceleration during isovolumic contraction (IVA, m/sec2; defined as the ratio of IVV divided by the acceleration time) were measured. All measurements were calculated from three consecutive cycles, and the average of the three measurements was recorded.
Figure 1.
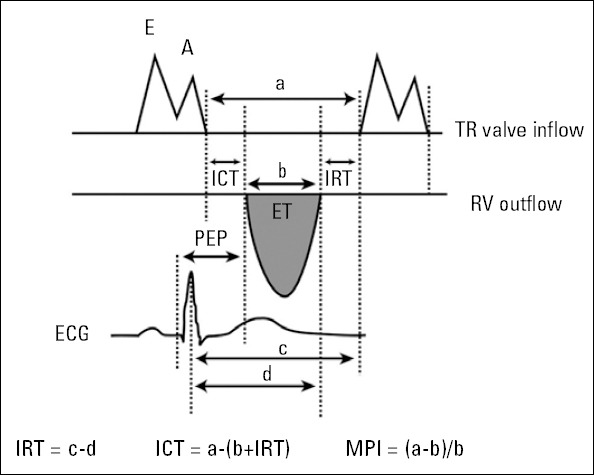
Measurement of right ventricular pre-ejection period (PEP), ejection time (ET), isovolumetric contraction time (ICT), isovolumetric relaxation time (IRT), and calculation of myocardial performance index (MPI)
Percutaneous balloon valvuloplasty
MS patients underwent PMBV, which was performed by three investigators. The mitral valve area was calculated using the Gorlin equation (17). Balloon dilation of the mitral valve was performed using a single-balloon dilating technique. Pre-procedural and post-procedural mitral insufficiency was evaluated based on Sellers classification on left ventriculography (18). The success of the procedure was defined as post-procedural planimetered mitral valve area (MVA) >1.5 cm2 echocardiographically and/or a 50% increase over the pre-procedural value and non-development of 3+ or 4+ mitral insufficiency.
Follow-up
Clinical and echocardiographic evaluations were performed before, 24-48 hours after, 3 months after, and 1 year after percutaneous mitral balloon valvuloplasty. Recurrent stenosis was defined as >50% loss of planimetered MVA calculated after the procedure and/or a valve area of 1.5 cm2. A major cardiovascular event was defined as death, repeat of balloon valvuloplasty, and the need for mitral valve replacement during the follow-up period.
Statistical analysis
The statistical evaluation was performed using SPSS 15.0 (Statistical package for the social sciences, Chicago, IL, USA). Categorical variables were presented as frequencies and percentages and were compared with the χ2 test. Continuous variables were expressed as means and SD. The normal distribution of continuous variables was tested with the Kolmogorov- Smirnov test. The Friedman test was used to compare consecutive measurements, and the Wilcoxon signed-rank test was used for the post-hoc analysis. A value of p<0.05 was considered significant.
Results
The baseline clinical and demographic properties of all study subjects are presented in Table 1. Forty-two of the 61 patients that were enrolled in the study were female (68%). The average age of the patients was 42±11-16 years. NYHA functional capacity was class 3 in 43 cases, class 2 in 14 cases, and class 4 in 4 cases before PMBV. Of the patients, 19 developed right heart failure, and 28 patients were receiving diuretics. The number of patients receiving diuretics decreased to 22 during the follow-ups.
Table 1.
Clinical characteristics of study patients
| Mean | |||||
|---|---|---|---|---|---|
| Age, year | 42.11±11.16 | ||||
| Female | 42 (68%) | ||||
| Systolic blood pressure, mm Hg | 109.8±7.8 | ||||
| Diastolic blood pressure, mm Hg | 71.6±4.3 | ||||
| Heart rate, bpm | 82.9±5.27 | ||||
| NYHA functional class | Before PMBV | After PMBV | 3rd month | 1st year | |
| Class 2 | 14 (23%) | 32 (53%) | 28 (45%) | 24 (39%) | |
| Class 3 | 43 (70%) | 28 (45%) | 32 (53%) | 34 (56%) | |
| Class 4 | 4 (7%) | 1 (2%) | 1 (2%) | 3 (5%) | |
| LVDD, mm | 47.4±3.2 | ||||
| LVSD, mm | 31.3±3.8 | ||||
| RA maximal volume, mL | 35.1±3.03 | ||||
| RVFAC, % | 37.7±1.8 | ||||
| PAP, mm Hg | 47.2±15.4 | ||||
| MVA PHT, cm2 | 1.2±0.14 | ||||
| Mean gradient, mm Hg | 11.3±2.5 | ||||
| Right heart failure, % | 19 (31%) | ||||
Data expressed as mean±SD or percentage. Variables were recorded before the procedure.
LVDD - left ventricular diastolic diameter; LVSD - left ventricular systolic diameter; MVA PHT - pressure half-time mitral valve area; PAP - pulmonary artery pressure; RA - right atrium; RVFAC - RV fractional area change; RV - right ventricle diameter
The procedure failed in 4 (6.1%) patients enrolled in the study. Serious mitral insufficiency due to chorda rupture occurred in 3 patients after the procedure, and valve replacement was performed. Cardiac tamponade due to myocardial rupture occurred in another patient in whom the procedure failed. This patient underwent pericardiocentesis in the catheter laboratory and had no problems during the follow-up in the intensive care unit. The patient was then referred to the surgery department under elective conditions. Clinically insignificant pericardial effusion was found after the procedure in 1 patient who had a successful valvuloplasty. Other major cardiovascular events (death, repeated balloon valvuloplasty) and restenosis were not observed throughout the follow-up.
A comparison of pre- and post-PMBV (at 48 hours, 3 months, and 1 year) values measured by transthoracic echocardiography is presented in Table 2. The mitral valve area that was measured increased significantly after successful PMBV and in the follow-up period (p<0.01). The value of the mean gradient, pulmonary artery pressure (PAP), and RA maximal volume decreased significantly after successful PMBV. The maximal PAP and RA volumes began to increase again in the third-month and first-year follow-up measurements and lost significance at the end of the first year. The Wilkins score decreased significantly after successful PMBV and in the follow-up period (p<0.01). TAPSE and RVFAC increased significantly after PMBV but reached their basal levels in the follow-up measurements and lost significance at the end of the first year. Deceleration time, pre-ejection period, A peak, and myocardial performance index decreased significantly, and ejection time and E peak increased significantly after successful PMBV (p<0.01). The MPI lost its significance at the end of the first year. The changes in TAPSE and MPI in the follow-up measurements are presented in Figure 2. Systolic velocity and early diastolic velocity increased significantly, and late diastolic velocity decreased significantly after successful PMBV (p<0.01). However, at 1 year, the statistical significance disappeared. A comparison of pre- and post-PMBV (at 48 hours, 3 months, and 1 year) did not differ significantly with respect to right ventricular myocardial acceleration during isovolumic contraction (IVA) (p=0.23, 0.37, 0.14), respectively, whereas peak myocardial velocity during isovolumic contraction (IVV) increased significantly after successful PMBV (p<0.01). However, at 1 year, the statistical significance disappeared (Table 3). The subgroup analysis that was conducted showed similar results in the pulmonary hypertension group compared to the overall group. TAPSE, systolic velocity, early diastolic velocity, IVV, and IVA increased significantly after PMBV in the group without pulmonary hypertension, and this increase was maintained during their 1-year follow-ups. Late diastolic velocity and MPI decreased significantly after PMBV, and this significance continued at 1 year (Table 4).
Table 2.
Comparison of pre- and post-PMBV (at 48 hours, 3 months, and 1 year) values measured by transthoracic echocardiography
| Before PMBV | After PMBV | 3rd month | 1styear | |
|---|---|---|---|---|
| LVDD, mm | 47.44 (41-52) | 47.32 (42-52) | 47.15 (42-52) | 48.33 (43-54) |
| LVSD, mm | 31.30 (25-37) | 31.15 (25-39) | 32.22 (27-41) | 32.36 (27-42) |
| Mean GR, mm Hg | 11.3 (17-8) | 4.3 (2-6)** | 4.3 (2-6)** | 4.4 (2-6)** |
| MVA PHT, cm2 | 1.1 (0.9-1.6) | 2.2 (1.8-2.8)** | 2.2 (1.7-2.9)** | 2.1 (1.8-2.7)** |
| MVA plan, cm2 | 1.1 (1-1.6) | 2.2 (1.9-2.7)** | 2.2 (1.9-2.7)** | 2.2 (1.9-2.7)** |
| PAP mm Hg | 47.20 (25-75) | 39.49 (25-65)** | 39.26 (25-70)** | 47.29 (25-75) |
| RA maximum volume, mL | 35.13 (30-42) | 33.22 (25-39)* | 33.86 (27-39)* | 35.16 (28-41) |
| LA, mm | 48 (42-57) | 46 (39-55)* | 44 (38-52)** | 43 (38-52)** |
| RVFAC, % | 37.71 (35-41) | 42.26 (38-45)** | 40.59 (35-45)* | 37.63 (33-42) |
| TAPSE, mm | 17.24 (15-20) | 18.68 (17-22)** | 18.06 (16-21)* | 17.56 (15-20) |
| TR jet area/RAA | 0.23 (0.07-0.43) | 0.17 (0.05-0.32)** | 0.18 (0.05-0.35)* | 0.21 (0.06-0.41) |
| Wilkins score | 8 (6-10) | 6 (4-8) | 6 (4-8) | 6 (4-8) |
| E peak, cm/s | 41.98 (35-55) | 50.03 (42-56)** | 44.40 (35-56)* | 43.49 (35-55)* |
| A peak, cm/s | 45.41 (40-50) | 39.61 (35-48)** | 43.89 (37-49)* | 44.48 (34-50) |
| Deceleration time, ms | 247.47 (220-282) | 214.16 (160-260)** | 238.44 (216-280)* | 245.32 (216-285)* |
| Pre-ejection period/ejection time | 0.62 (0.52-0.68) | 0.39 (0.35-0.47)** | 0.55 (0.42-0.64)* | 0.58 (0.45-0.55)* |
| Myocardial performance index | 0.48 (0.43-0.55) | 0.42 (0.36-0.46)** | 0.44 (0.40-0.50)* | 0.48 (0.43-0.55) |
Results are shown as rank (min-max) value.
LVDD -left ventricular diastolic diameter; LVSD - left ventricular systolic diameter; Mean GR - mitral mean gradient; MVA - planimetric mitral valve area; PAP - pulmonary artery pressure; PHT - pressure half-time; PMBV - percutaneous balloon mitral valvuloplasty; RA - right atrium; RV - right ventricle; RVFAC - RV fractional area change; TAPSE - tricuspid annular plane systolic excursion; TR jet area/RAA - tricuspid valve regurgitation area/right atrial area
P<0.05 comparison with baseline after PMBV and at 3 months and 1 year
P<0.01 comparison with baseline after PMBV and at 3 months and 1 year
Figure 2.
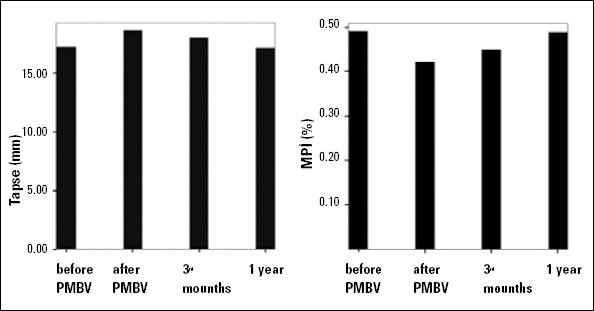
Changes in right ventricular myocardial performance index (MPI) and tricuspid annular plane systolic excursion (TAPSE) before and 3 months and 1 year after percutaneous mitral balloon valvuloplasty
Table 3.
Comparison of pre- and post-PMBV (at 48 hours, 3 months, and 1 year) pulsed tissue Doppler
| Before PMBV | After PMBV | 3rd month | 1st year | |
|---|---|---|---|---|
| Systolic velocity, cm/s | 12.29 (9-17) | 15.03 (11-17)** | 13.08 (10-16)* | 12.50 (10-15) |
| Early diastolic velocity, cm/s | 10.14 (8-14) | 13.08 (10-16)** | 11.14 (8-16) | 11.06 (8-15) |
| Late diastolic velocity, cm/s | 12.88 (8-16) | 11.14 (8-16)** | 11.06 (8-15)** | 11.45 (8-15)* |
| Right ventricular IVV, cm/sec | 0.11 (0.06-0.16) | 0.14 (0.08-0.19)** | 0.13 (0.07-0.18)* | 0.12 (0.07-0.16) |
| Right ventricular IVA, m/sec2 | 2.21 (1.60-2.90) | 2.19 (1.80-2.80) | 2.20 (1.70-2.90) | 2.22 (1.60-2.90) |
IVA - myocardial acceleration during isovolumic contraction; IVV - peak myocardial velocity during isovolumic contraction
P<0.05 comparison with basaline after PMBV and at 3 months and 1 year.
P<0.01 comparison with basaline after PMBV and at 3 months and 1 year.
Table 4.
Comparison of pre- and post-PMBV (at 48 hours, 3 months, and 1 year) echocardiographic variables in patients with or without baseline pulmonary hypertension
| Pulmonary hypertension (+) (n:46) | Pulmonary hypertension (-) (n:15) | |||||||
|---|---|---|---|---|---|---|---|---|
| Before | After | 3rd | 1st | Before | After | 3rd | 1st | |
| PMBV | PMBV | months | years | PMBV | PMBV | months | years | |
| TAPSE, mm | 17.19 | 18.6 | 18 | 17.21 | 17.4 | 18.8 | 18.5 | 18.3 |
| (13-24) | (15-24)** | (14-24)** | (13-24) | (13-25) | (15-27)** | (15-27)** | (15-26)** | |
| MPI | 0.49 | 0.42 | 0.45 | 0.49 | 0.48 | 0.41 | 0.42 | 0.42 |
| (0.42-0.58) | (0.36-0.54)** | (0.39-0.56)** | (0.42-0.59) | (0.42-0.59) | (0.34-0.55)** | (0.34-0.56)** | (0.35-0.56)** | |
| PAP mm Hg | 55.6 | 43.73 | 43.36 | 54.13 | 29.53 | 26.46 | 26.66 | 27.4 |
| (40-75) | (30-65)** | (30-70)** | (40-75) | (25-39) | (25-32) | (25-35) | (25-42) | |
| Tricuspid annulus | ||||||||
| Sv, cm/s | 12.15 | 15.15 | 13.08 | 12.21 | 12.7 | 14.6 | 14.06 | 13.67 |
| (9-17) | (11-19)** | (10-16)* | (9-16) | (10-16) | (11-18)** | (10-17)* | (10-17)* | |
| Ev, cm/s | 10.02 | 13.08 | 11.15 | 10.75 | 10.60 | 13.06 | 12.26 | 12.20 |
| (8-14) | (11-17)** | (8-16)* | (8-15) | (8-14) | (11-18)** | (11-17)** | (11-17)** | |
| Av, cm/s | 12.8 | 11.15 | 12.02 | 12.6 | 13.06 | 11.13 | 11.13 | 11.20 |
| (10-16) | (8-16)** | (9-17)* | (10-17) | (11-17) | (9-15)** | (8-15)** | (8-14)** | |
| IVV, cm/sec | 0.11 | 0.12 | 0.12 | 0.11 | 0.11 | 0.12 | 0.13 | 0.13 |
| (0.06-0.16) | (0.07-0.17)* | (0.07-0.16)* | (0.06-0.16) | (0.06-0.15) | (0.07-0.16)* | (0.07-0.17)** | (0.08-0.18)** | |
| IVA, m/sec2 | 2.19 | 2.16 | 2.18 | 2.20 | 2.28 | 2.34 | 2.38 | 2.40 |
| (1.6-2.9) | (1.6-2.8) | (1.7-2.9) | (1.6-2.9) | (1.9-2.6) | (1.9-2.6) | (2-2.7)* | (2-2.8)* | |
*Results are shown as rank (min-max) values
Av - late diastolic velocity; Ev - early diastolic velocity; IVA - myocardial acceleration during isovolumic contraction; IVV - peak myocardial velocity during isovolumic contraction; MPI - myocardial performance index; Sv - systolic velocity; TAPSE - tricuspid annular plane systolic excursion.
P<0.05 comparison with basaline-after PMBV, at 3 months and 1 year
P<0.01 comparison with basaline-after PMBV, at 3 months and 1 year
Discussion
This study evaluated the acute and mid-term effects of PMBV on RV functions by echocardiographic tissue Doppler technique with the intent to investigate whether this acute improvement is a progressive process or an acute response to changes in the cardiopulmonary system. In this study, the improved right ventricular functions in mitral stenosis patients with pulmonary hypertension in the post-PMBV acute period were shown to decrease in the following period and disappeared at the end of 1 year. The improvement in the group without pulmonary hypertension in the acute period was also shown to be sustained at 1 year.
The trapezoidal anatomy of the ventricle makes the quantitative echocardiographic evaluation extremely difficult. Up to date many different assessment methods has been investigated but none of them appears to be the gold standard at this era. New methods have been evaluated in recent years. Cardiac catheterization, MR, radionuclide ventriculography, and 3D- echocardiography have shown that right ventricle functions can be used reliably (19-21). On the other hand, these methods are not readily accessible and can not be performed in a short time. In practice, clinicians largely rely on two modalities: two-dimensional echocardiography and TDI echocardiography. Conventional M-mode, Doppler echocardiography evaluation, and TDI echocardiography, which is used to evaluate right and left ventricle functions, are preferred, because they are less affected by physiologic changes in flow velocities and indicate subclinical functional effects. In typical pulse-wave Doppler imaging obtained with Doppler echocardiography, it is possible to measure the duration of the systolic and diastolic waves. In the case of systolic dysfunction, isovolumetric contraction time increases, whereas ejection time decreases. However, in the case of diastolic dysfunction, in which flexibility decreases, isovolumet- ric relaxation time increases. MPI (Tei index), which is calculated using these three indices of time, is a reliable parameter evaluating both systolic and diastolic functions (22). The Tei index is not greatly influenced by changes in blood pressure, afterload, heart rate, preload, RV pressure, dilatation, or tricuspid regurgitation in the clinical setting (11). Several studies have been published regarding the use of the Tei index and pulsed TDI to identify patients with impaired systolic and diastolic function (23-27). Recently, a new TDI-derived index of myocardial acceleration during isovolumic contraction (IVA) has been shown to be a reliable and relatively load-independent measure of RV systolic function (28). Despite certain limitations, TAPSE, which can be performed using echocardiography (14, 29), can be readily used in daily practice. Comparative studies on the right ventricle have shown that TAPSE is correlated with magnetic resonance imaging and radionuclide ventriculography (30).
MS is a deteriorating clinical outcome that causes RV failure. As a result of this one can determine clinical symptoms, exercise capacity, and survival via RV function. Increased pulmonary wedge pressure is associated with a boost in pulmonary artery pressure, increased afterload, and, consequently, failure in RV ejection fraction. The systolic wall stress is caused by the afterload volume changes due to RV functions (31). RV is sensitive to changes in afterload because of smaller mass and higher wall stress (32). In this study, the recovery of right ventricle function (a decrease in RV, Tei index, and pulmonary arterial pressure and an increase in TAPSE, Sa, and IVV) right after PMBV can be explained by the recovery of the RV outflow tract systolic functions due to the acute decrease in RV afterload (12). However, no significant change was observed in IVA in the acute period or mid-term follow-ups. This is an expected outcome, since the IVA is not specifically affected by hemodynamic changes. Borgers et al. (33) showed recovery in the Tei index after vasodilator therapy in patients who had chronic pulmonary hypertension. Vogel et al. (34) demonstrated that IVA was an accurate parameter to assess RV systolic dysfunction and was able to measure the force-frequency relation. Previous studies also showed that RV function decreased in MS due to increased RV afterload (6, 8, 9) and demonstrated a positive effect of PMBV on RV functions in the acute period (11, 12, 35). However, to our knowledge, there are no studies that demonstrate whether this effect is carried into the mid-term.
It was surprising to the researchers that pulmonary artery pressure decreased after PMBV, and this decrease reached the basal level at the 1-year follow-up, although it was maintained during the 3-month follow-ups. The subgroup evaluation revealed that there was a similar case in the group with pulmonary hypertension; however, the decrease that occurred in the acute period in the group without pulmonary hypertension continued during the 1-year follow-up, as well. Given the re-elevation of PAP at the 1-year follow-ups, especially in the pulmonary hypertension group, and the non-observation of clinical conditions that might cause this during the follow-up, such as restenosis, development of paroxysmal AF, and mitral regurgitation, it is difficult to explain the underlying mechanism. It may be that the irreversible changes in the pulmonary vascular bed in the group with pulmonary hypertension presented a pseudo-improvement for a given time due to the decreased post-PMBV afterload. The study by Mahfouz et al. (36) analyzed the long-term effect of pulmonary artery stiffness on right ventricular functions and tricuspid regurgitation. Based on the evaluations before, immediately after, and at 6 months and 12 months after the procedure, the investigator demonstrated that pulmonary artery stiffness was significantly lower in patients who had permanent improvement in right ventricular functions and regression of tricuspid regurgitation. The investigator argued that the tricuspid regurgitation and the continued right ventricular dysfunction in some patients, even though a sufficient mitral valve area opening could be ensured after PMBV, may be the increased pulmonary artery stiffness in this patient group and highlighted the importance of early intervention.
In the present study, the recovery of RV functions decreased in the mid-term and disappeared at the end of the first year (an increase in the Tei index, and pulmonary arterial pressure and a reduction in TAPSE). Although similar results were achieved in the group with pulmonary hypertension, the improved right ventricular functions that occurred in the acute period in the group without pulmonary hypertension were maintained at 1 year. High wall stress due to ventricular dilatation that affects RV myocardium or rheumatic conditions that occur as a secondary result of myocardial dysfunction can elaborate this situation (37). In a previous study by Malhotra et al. (38), it is shown that intra myocardial branches of myocardial vessels were also involved in a form of active rheumatic vasculitis or inactive lesions characterized by medial hypertrophy and replacement fibrosis in patients with rheumatic heart disease affecting. The study of Mohan et al. (39) showed that pulmonary artery pressure decreased immediately after balloon valvuloplasty and that right ventricular functions, as assessed by the Tei index, returned to normal values within 1 year in 65% of such patients. It was shown that there was right ventricular fractional shortening and improvement in systolic functions, as assessed by the Tei index, after balloon valvuloplasty in patients with mitral stenosis, whereas a decrease was observed in right ventricular contraction, as assessed by IVA (12). The lack of observed improvement in right ventricular function despite the improved hemodynamic status suggested irreversible myocardial damage due to rheumatic pathology or long-lasting hemodynamic burden in these patients. Arat et al. (11) evaluated RV functions in the early (first 48 hours) and mid (3rd month)-term after PMBV and did not observe a significant difference in the Tei index. The authors determined that RV functions increased significantly in the early period in the group without pulmonary HT and maintained their high level in the mid-term. Mahfouz et al. (35) determined a significant decrease in pulmonary arterial pressure and a significant increase in TAPSE in the post-PMBV evaluation. As demonstrated in these literature findings, during the acute phase and short term, there are no clear data showing the effect of PMBV on RV functions, and follow-up studies with larger numbers of patients are needed to assess whether this finding has any prognostic implications. Although echocardiography is a noninvasive and reproducible method to evaluate cardiac functions, it should be kept in mind that RV function parameters are also not fully independent parameters.
Study limitations
One of the limitations of this study is that it was a singlecenter study and not randomized, and the study population was relatively small. The parameters used to predict RV dysfunction were not independent parameters. In the current study, invasive measurements were not made, and RV ejection fraction was not measured. Additionally, strain imaging has a lower temporal resolution compared to tissue Doppler-derived deformation indices, resulting in less reliable estimates. Because of the complex geometry of the RV, real-time 3-D echocardiography is estimated to accurately evaluate the morphology and function of this chamber. However, none of the methods used to evaluate right cardiac function is free of limitations (40, 41). Tissue Doppler imaging is a method with proven efficacy and relative reliability. Also, these limitations were minimized because of this being a follow-up study.
Conclusion
The data of the present study revealed that right ventricle functions improved significantly immediately after PMBV, but the observed recovery decreased and even disappeared in patients with pulmonary hypertension in the early- and mid-term follow-ups. This condition may indicate the importance of intervention in MS patients prior to the development of latent right ventricle myocardial dysfunction.
Footnotes
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept - S.İ., H.H.; Design - M.K.E., S.İ., H. Duman; Supervision - M.K.E., E.M.B., Ş.K.; Resource - S.İ., H. Değirmenci; Materials - S.İ., H.H., E.M.B.; Data collection &/or processing - S.İ., M.K.E., H. Duman; Literature search - S.İ., H. Değirmenci, H.H.; Writing - S.İ., H. Değirmenci, H. Duman; Critical review - Ş.K.
References
- 1.Kawakita S. Rheumatic fever and rheumatic heart disease in Japan. Jpn Circ J. 1986;50:1241–5. doi: 10.1253/jcj.50.1241. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 2.Rigolin VH, Higgenbotham MB, Robiolio PA, Hearne SE, Baker WA, Kisslo KB, et al. Effect of inadequate cardiac output reserve on exercise tolerance in patients with moderate mitral stenosis. Am J Cardiol. 1997;80:236–40. doi: 10.1016/s0002-9149(97)00333-0. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 3.Tayyareci Y, Nişancı Y, Umman B, Öncül A, Yurdakul S, Altun I, et al. Early detection of right ventricular systolic dysfunction by using myocardial acceleration during isovolumic contraction in patients with mitral stenosis. Eur J Echocardiogr. 2008;9:516–21. doi: 10.1016/j.euje.2007.08.007. [DOI] [PubMed] [Google Scholar]
- 4.Yıldırımtürk O, Helvacıoğlu FF, Tayyareci Y, Yurdakul S, Aytekin S. Assessment of right ventricular endocardial dysfunction in mild- to-moderate mitral stenosis patients using velocity vector imaging. Echocardiography. 2012;29:25–33. doi: 10.1111/j.1540-8175.2011.01529.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 5.Sanchez Lazaro JI, Almenar Bonet L, Igual Munoz B, Rueda-Soriano J, Martinez-Dolz L, Zorio-Grima E, et al. Phenotypic patterns of right ventricular dysfunction: analysis by cardiac magnetic imaging. Heart Int. 2013;22:e3. doi: 10.4081/hi.2013.e3. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burger W, Brinkies C, Illert S, Teupe C, Kneissl GD, Schrader R. Right ventricular function before and after percutaneous balloon mitral valvuloplasty. Int J Cardiol. 1997;58:7–15. doi: 10.1016/s0167-5273(96)02860-4. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 7.Burger W, Illert S, Teupe C, Kneissl GD, Kober G, Schrader R. Right ventricular function in patients with rheumatic mitral valve stenosis. Effect of balloon mitral valvuloplasty. Z Kardiol. 1993;82:545–51. [PubMed] [Google Scholar]
- 8.Özdemir K, Altunkeser BB, Gök H, İçli A, Temizhan A. Analysis of the myocardial velocities in patients with mitral stenosis. J Am Soc Echocardiogr. 2002;15:1472–8. doi: 10.1067/mje.2002.128645. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 9.Özdemir K, Altunkeser BB, Gök H, İçli A. Does the myocardial performance index affect pulmonary artery pressure in patients with mitral stenosis? A tissue Doppler imaging study. Echocardiography. 2003;20:249–56. doi: 10.1046/j.1540-8175.2003.03022.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 10.Hirata N, Sakakibara T, Shimazaki Y, Watanabe S, Nomura F, Akamatsu H, et al. Preoperative and postoperative right ventricular function during exercise in patients with mitral stenosis. J Thorac Cardiovasc Surg. 1992;104:1029–34. [PubMed] [Google Scholar]
- 11.Arat N, Altay H, Korkmaz S, Ilkay E. The effect of baseline pulmonary artery pressure on right ventricular functions after mitral balloon valvuloplasty for rheumatic mitral stenosis: a tissue Doppler imaging study. Turk Kardiyol Dern Ars. 2008;36:223–30. [PubMed] [Google Scholar]
- 12.Drighil A, Bennis A, Mathewson JW, Lancelotti P, Rocha P. Immediate impact of successful percutaneous mitral valve com- missurotomy on right ventricular function. Eur J Echocardiogr. 2008;9:536–41. doi: 10.1093/ejechocard/jen126. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 13.Özcan F, Şen N, Arat N, Turak O, Akpek M, Kaya MG, et al. Evaulation of right ventricular functions with TAPSE and tissue Doppler in mitral stenotic patients. Tıp Araştırmaları Dergisi. 2013;11:12–6. [Google Scholar]
- 14.Schenk P, Globits S, Koller J, Brunner C, Artemiou O, Klepetko W, et al. Accuracy of echocardiographic right ventricular parameters in patients with different end-stage lung diseases prior to lung transplantation. J Heart Lung Transplant. 2000;19:145–54. doi: 10.1016/s1053-2498(99)00121-7. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 15.Eidem BW, O’Leary PW, Tei C, Seward JB. Usefulness of the myocardial performance index for assessing right ventricular function in congenital heart disease. Am J Cardiol. 2000;86:654–8. doi: 10.1016/s0002-9149(00)01047-x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 16.Tei C, Dujardin KS, Hodge DO, Bailey KR, McGoon MD, Tajik AJ, et al. Doppler echocardiographic index for assessment of global right ventricular function. J Am Soc Echocardiogr. 1996;9:838–47. doi: 10.1016/s0894-7317(96)90476-9. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 17.Gorlin R, Gorlin SG. Hydraulic formula for calculation of the area of the stenotic mitral valve, other cardiac valves and central circulatory shunts. Am Heart J. 1951;41:1–29. doi: 10.1016/0002-8703(51)90002-6. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 18.Sellers RD, Levy MJ, Amplatz K, Lillehei CW. Left retrograde cardio- angiography in acquired cardiac disease: technic, indications and interpretations in 700 cases. Am J Cardiol. 1964;14:437–47. doi: 10.1016/0002-9149(64)90027-x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 19.Brookes CI, White PA, Bishop AJ, Oldershaw PJ, Redington AN, Moat NE. Validation of a new intraoperative technique to evaluate load-independent indices of right ventricular performance in patients undergoing cardiac operations. J Thorac Cardiovasc Surg. 1998;116:468–76. doi: 10.1016/S0022-5223(98)70013-3. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 20.Helbing WA, Bosch HG, Maliepaard C, Rebergen SA, van der Geest RJ, Hansen B, et al. Comparison of echocardiographic methods with magnetic resonance imaging for assessment of right ventricular function in children. Am J Cardiol. 1995;76:589–94. doi: 10.1016/s0002-9149(99)80161-1. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 21.Rumberger JA, Behrenbeck T, Bell MR, Breen JF, Johnston DL, Holmes DR, Jr, et al. Determination of ventricular ejection fraction: a comparison of available imaging methods. The Cardiovascular Imaging Working Group. Mayo Clin Proc. 1997;72:860–70. doi: 10.4065/72.9.860. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 22.Harada K, Tamura M, Toyono M, Oyama K, Takada G. Assessment of global left ventricular function by tissue Doppler imaging. Am J Cardiol. 2001;88:927–32. doi: 10.1016/s0002-9149(01)01912-9. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 23.Waggoner AD, Bierig SM. Tissue Doppler imaging: a useful echocardiographic method for the cardiac sonographer to assess systolic and diastolic ventricular function. J Am Soc Echocardiogr. 2001;14:1143–52. doi: 10.1067/mje.2001.115391. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 24.Rodriguez L, Garcia M, Ares M, Griffin BP, Nakatani S, Thomas JD. Assessment of mitral annular dynamics during diastole by Doppler tissue imaging: comparison with mitral Doppler inflow in subjects without heart disease and in patients with left ventricular hypertrophy. Am Heart J. 1996;131:982–7. doi: 10.1016/s0002-8703(96)90183-0. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 25.Oki T, Tabata T, Yamada H, Wakatsuki T, Mishiro Y, Abe M, et al. Left ventricular diastolic properties of hypertensive patients measured by pulsed tissue Doppler imaging. J Am Soc Echocardiogr. 1998;11:1106–12. doi: 10.1016/s0894-7317(98)80005-9. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 26.Severino S, Caso P, Galderisi M, De Simone L, Petrocelli A, de Divitiis O, et al. Use of pulsed Doppler tissue imaging to assess regional left ventricular diastolic dysfunction in hypertrophic cardiomyopathy. Am J Cardiol. 1998;82:1394–8. doi: 10.1016/s0002-9149(98)00648-1. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 27.Garcia MJ, Rodriguez L, Ares M, Griffin BP, Thomas JD, Klein AL. Differentiation of constrictive pericarditis from restrictive cardio- myopathy: assessment of left ventricular diastolic velocities in longitudinal axis by Doppler tissue imaging. J Am Coll Cardiol. 1996;27:108–14. doi: 10.1016/0735-1097(95)00434-3. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 28.Tayyareci Y, Tayyareci G, Nisançı Y, Umman B, Buğra Z. Evaluation of the severity of mitral stenosis with a new index: isovolumic myocardial acceleration. Turk Kardiyol Dern Ars. 2008;36:388–94. [PubMed] [Google Scholar]
- 29.Miller D, Farah MG, Liner A, Fox K, Schluchter M, Hoit BD. The relation between quantitative right ventricular ejection fraction and indices of tricuspid annular motion and myocardial performance. J Am Soc Echocardiogr. 2004;17:443–7. doi: 10.1016/j.echo.2004.01.010. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 30.Kjaergaard J, Petersen CL, Kjaer A, Schaadt BK, Oh JK, Hassager C. Evaluation of right ventricular volume and function by 2-D and 3-D echocardiography compared to MRI. Eur J Echocardiogr. 2006;7:430–8. doi: 10.1016/j.euje.2005.10.009. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 31.Nagel E, Stuber M, Hess OM. Importance of the right ventricle in valvular heart disease. Eur Heart J. 1996;17:829–36. doi: 10.1093/oxfordjournals.eurheartj.a014963. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 32.Morrison D, Goldman S, Wright AL, Henry R, Sorenson S, Caldwell J, et al. The effect of pulmonary hypertension on systolic function of right ventricle. Chest. 1983;84:250–7. doi: 10.1378/chest.84.3.250. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 33.Borges AC, Knebel F, Eddicks S, Panda A, Schattke S, Witt C, et al. Right ventricular function assessed by two-dimensional strain and tissue Doppler echocardiography in patient with pulmonary arterial hypertension and effect of vasodilatator therapy. Am J Cardiol. 2006;98:530–4. doi: 10.1016/j.amjcard.2006.02.060. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 34.Vogel M, Schmidt MR, Kristiansen SB, Cheung M, White PA, Sorensen K, et al. Validation of myocardial acceleration during isovolumic contraction as a novel noninvasive index of right ventricular contractility: comparison with ventricular pressure-volume relations in an animal model. Circulation. 2002;105:1693–9. doi: 10.1161/01.cir.0000013773.67850.ba. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 35.Mahfouz RA, Elawady W, Hossein E, Yosri A. Impact of atrioventricular compliance on clinical outcome of patients undergoing successful percutaneous balloon mitral valvuloplasty. Echocardiography. 2013;30:1187–93. doi: 10.1111/echo.12256. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 36.Mahfouz RA. Impact of pulmonary artery stiffness on right ventricular function and tricuspid regurgitation after successful percutaneous balloon mitral valvuloplasty: the importance of early intervention. Echocardiography. 2012;29:1157–63. doi: 10.1111/j.1540-8175.2012.01785.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 37.Lee YS, Lee CP. Ultrastructural pathological study of left ventricular myocardium in patients with isolated mitral stenosis with normal or abnormal left ventricular function. Jpn Heart J. 1990;31:435–48. doi: 10.1536/ihj.31.435. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 38.Malhotra V, Beohar PC, Gondal R, Kaul UA, Khanna SK. An autopsy study of rheumatic heart disease. Part II. Associated findings. Jpn Heart J. 1987;28:7–14. doi: 10.1536/ihj.28.7. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 39.Mohan JC, Sengupta PP, Arora R. Immediate and delayed effects of successful percutaneous transvenous mitral commissurotomy on global right ventricular function in patients with isolated mitral stenosis. Int J Cardiol. 1999;68:217–23. doi: 10.1016/s0167-5273(98)00358-1. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 40.Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23:685–713. doi: 10.1016/j.echo.2010.05.010. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 41.Küçükdurmaz Z, Karapınar H, Karavelioğlu Y, Açar G, Gül I, Emiroglu MY, et al. Effect of blood donation mediated volume reduction on right ventricular function parameters in healthy subjects. Echocardiography. 2012;29:451–4. doi: 10.1111/j.1540-8175.2011.01614.x. [CrossRef] [DOI] [PubMed] [Google Scholar]


