Abstract
Objective:
Low-molecular-weight heparins (LMWHs) are commonly used to prevent and manage postoperative thromboembolism. In general, monitoring of anticoagulant activity by anti-Xa testing is not done properly. Thromboelastography (TEG) evaluates the viscoelastic properties of blood during coagulation. The clinical application of TEG variables in monitoring LMWH treatment is not yet well defined.
Methods:
This prospective study was designed to systematically examine the correlation between anti-Xa and basic TEG parameters in monitoring LMWH treatment. We furthermore evaluated for the first time the usefulness of a composite TEG parameter, coagulation index (CI). Thirty patients undergoing unilateral or bilateral total knee replacement, admitted to the intensive care unit on a therapeutic dosage of subcutaneous enoxaparin (30-mg injections administered twice daily), were included into the study. TEG parameters and anti-Xa levels were measures at baseline and 4, 12, and 24 hours after the injection.
Results:
This study demonstrates a significant correlation between CI and plasma anti-Xa activity in surgical patients treated with enoxaparin. Although the correlation was significant between r time and anti-Xa level only at Hour 4, CI was significant for each time interval (p<0.05). CI increased immediately after T0, peaking at Hour 4, and remained elevated (relative to baseline) at Hour 24 but still did not return to admission levels.
Conclusion:
The current study may be an important first step in order to use CI to measure LMWH activity. Meanwhile, the value and usefulness of TEG in predicting bleeding or thrombotic complications following major orthopedic surgery merit further investigation.
Keywords: anti-factor Xa, coagulation index, thromboelastography
Introduction
Patients undergoing major elective lower extremity orthopedic surgery are in the highest risk category for venous thromboembolism (VTE) (1). Individuals undergoing major surgery exhibit a prothrombotic state (2). Therefore, they have a shorter duration of measurable heparin levels (3), as well as a partial thromboplastin time (aPTT), taken to assess heparin action (4). As a result, these patients have periods of unknown or insufficient anticoagulation effects during the trough periods of heparin concentration when the drug is given only once every 12 hours.
LMWHs offer a stable and predictable anticoagulant doseresponse and a great anti-Xa/anti-IIa ratio, resulting in less thrombin generation and activation (5). Accordingly, the standard assay used for monitoring enoxaparin is via the inhibition of Xa. Standard perioperative tests of coagulation do not reflect the extent of anticoagulation resulting from LMWH (6). A number of kits are available for measuring anti-Xa activity, although there is some concern relating to interassay variability; therefore, measuring this activity is not effective (7). Unfortunately, alternative tests have not yet been validated. Due to the absence of a practical measure of anticoagulation, the lack of consistent effectiveness of heparin in orthopedic surgery is still a problem.
The underpinnings of postoperative hypercoagulative conditions are not yet fully appreciated. Although the anti-Xa effect predominates, enoxaparin influences the coagulation cascade in other areas, including tissue factor pathway inhibitor (TFPI) and direct inhibition of thrombin and platelet function, and as such, a more global assessment of coagulation might provide a better guide to monitoring patients (8-10). Major orthopedic surgery leads to massive tissue factor release and hefty triggering of coagulation (11, 12). At this point, a point-of-care test reflecting the antithrombotic effect of LMWH was investigated in order to fulfill an important clinical function.
Thromboelastography (TEG) is a bedside blood test that measures the viscoelastic properties of whole blood, as it clots under a low-sheer stress environment, thus providing a comprehensive evaluation of the process of clot initiation, formation, and stability (13). Abnormal TEG data may predict patients who will bleed. Spiess et al. (14) found that TEG correlated well with activated clotting time (ACT) and coagulation profiles, and whilst no coagulation test was consistently abnormal, the TEG was the most accurate predictor (87% accuracy) of bleeding. Whilst platelet-activated clotting test (PACT) sensitivity and specificity were comparable to conventional coagulation tests in predicting blood loss, TEG was more predictive for both blood loss levels, showing 83% sensitivity and 79% specificity (15).
TEG evaluates global coagulation, and the effect of heparin on the trace has been shown to be a more sensitive method of assessing the degree of anticoagulation compared with conventional methods of assessing clotting status (16). Enoxaparin causes a dose-dependent inhibition of the native TEG trace, with an abnormal coagulation trace apparent at plasma drug levels of >0.1 anti-Xa IU/ml (17). Low doses of enoxaparin prolong the r time, whereas therapeutic doses prolong both the r and k times and also cause a reduction in the alpha angle (17). The peak and trough levels of anti-factor Xa activity correlate with the r-value of the thromboelastograph (18).
The aim of this study was to evaluate whether TEG® variables correlate with the activity of anti-Xa in knee replacement surgery patients treated with the LMWH enoxaparin. We suggested that the reaction (r) time coagulation index (CI) values of TEG® would correlate with enoxaparin and could be used as a guide for enoxaparin therapy as early as to take precautions for heparin action.
Methods
Thirty patients, aged over 50 years [age range, 52-87 years; mean age, 58.12±6.68 years, males, 17 (62.87±8.07); females, 13 (64±7.12)] participated in this single-site, prospective clinical trial. The study was approved by the hospital ethics committee and was carried out in accordance with the standards stipulated by the World Medical Association Declaration of Helsinki. All participants granted informed consent. All patients undergoing unilateral or bilateral total knee replacement, admitted to the intensive care unit on a therapeutic dosage of subcutaneous enoxaparin (30-mg injections administered twice daily), were included. Patients were excluded on the following basis: (1) known cancer, (2) hepatic or renal dysfunction, (3) history of hematologic disorder, (4) preoperative treatment with anti-aggregative or anticoagulant drugs (other than antiplatelet drugs, including concurrent use of nonsteroidal anti-inflammatory drugs), (5) enoxaparin only for prophylaxis against deep vein thrombosis, and (6) platelet counts below 150x103 cells/mL and above 450x103 cells/mL. Samples were taken perioperatively (on admission to the hospital) and after the subcutaneous injection of enoxaparin at 4, 12, and 24 hours (symbolized from the baseline as T0, T4, T12, T24, respectively). T0 is the time point of the first (of two) daily doses of LMWH. Venous blood was collected by venipuncture from an antecubital vein by using a tourniquet and the two-syringe collection technique. The blood samples were collected into Vacutainer tubes (Becton Dickinson, Rutherford, NJ) containing sodium citrate 3.8% (9:1 vol/vol) and allowed to completely fill the tube. Venous samples were obtained simultaneously for TEG®, anti-Xa analysis, and aPTT at each of the time intervals. Platelets were quantified using a Sysmex XT-2000i automated hematologic analyzer (Sysmex Corp., Kobe, Japan).
TEG® assays were processed using the TEG 5000 Thromboelastograph hemostasis analyzer (Haemonetics Corporation, Skokie, Illinois, USA). The TEG® measures the physical properties of the clot as it forms between the testing cup and pin and produces a graphical representation (Fig. 1), which allows one to evaluate the different phases of clotting and deduce the adequacy of the coagulation and fibrinolytic pathways. All thromboelastographic tests were performed in duplicate. Routine machine quality control and standard calibrations were maintained. An electrical internal quality control (e-test) was performed prior to each assay. Haemoscope TEG® disposable cups and pins (Haemonetics Corporation) were placed in the analyzer and prewarmed to 37°C. Briefly, 340 pL of citrated blood was mixed with 20 pL of calcium chloride (a 0.2-mol/L concentration) in a plain plastic cup or a cup coated with 2 U of heparinase (the final concentration of heparinase would be 5.6 U/mL).
Figure 1.
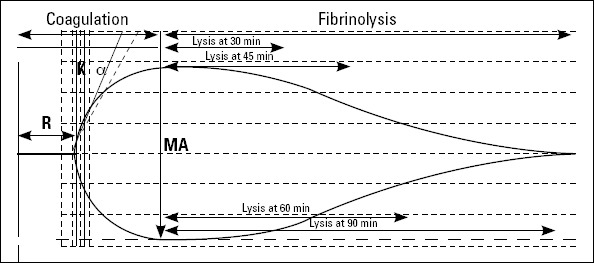
A normal thromboelastographic tracing (1). r -time is the time elapsed from placement of the sample in the cuvette until the tracing amplitude reaches 2 mm; it denotes the rate of initial fibrin formation and is functionally related to plasma clotting factors (2). K is measured from r to the point where the amplitude reaches 20 mm; k-time represents the time it takes for a fixed degree of viscoelasticity to be achieved by the forming clot and is affected by the activity of the intrinsic clotting factors, fibrinogen, and platelets (3). a-angle is the angle formed by the slope of the TEG tracing from the r to the k value; it denotes the rate at which the clot is formed (4). MA (maximum amplitude) is the greatest amplitude on the TEG tracing; it is a reflection of the absolute strength of the fibrin clot and can be altered by both qualitative and quantitative platelet abnormalities
The reaction started immediately after mixing. Variables that were recorded were r time and CI. The r time (normally between 2-8 min) reflects the latency period for blood (once placed in the analyzer) to initiate fibrin formation. A TEG coagulation index (CI) describing the subject’s overall coagulation was derived from the r-time, k-time, MA, and a-angle by the Thromboelastograph Coagulation Analyzer by means of the equation:
CI= - 0.3258R-0.1886K + 0.1224MA + 0.0759a - 7.7922
Normal values for the CI are -3.0 to +3.0 (mean 0±3 SDs). Positive values outside this range (CI >3.0) indicate that the sample is hypercoagulable; negative values outside this range (CI <3.0) indicate that the sample is hypocoagulable. CI =3 is an indicator of normal coagulation status.
The remaining blood from each sample interval underwent immediate centrifugation at 3000 g, and the plasma was stored at -70°C. Enoxaparin was quantitated using a standard chromogenic anti-Xa activity assay (Heptest, Sekisui Diagnostics, LLC) on the Amax 200 coagulation analyzer (TrinityBiotech, Berkeley Heights, New Jersey, USA) using the same manufacturer’s reagents as described by the manufacturer. The measuring range was 0.015-1.0 U/mL. The assay was calibrated using commercial heparin no less than 1000 U/mL plasma or whole blood. The coefficient of variation for the assay was 2.2%. Duplicate measures were performed each sample; the average value between the two measurements was used. aPTT assays were again performed on an AMAX 200 using TriniCLOT aPTT S (TrinityBiotech). Demographic data were collected, including participant age, sex, and weight.
Statistical analyses
All analyses were performed using the SPSS package (SPSS for Windows 17.0, Chicago, IL, USA). The Kolmogorov-Smirnov test was used to assess the normal distribution of the continuous variables. All continuous variables displaying a normal distribution were expressed as mean±standard deviation. Spearman correlation coefficient (r) was calculated for the correlation between the TEG parameters and anti-Xa and aPTT. Differences between continuous variables were compared using the paired-t test. A p value <0.05 was considered significant.
Results
The mean±SD for the TEG® variables (r time and CI), aPTT, and anti-Xa concentration before and after subcutaneous injection of enoxaparin is shown in Table 1. There were no marked variations in any parameters, as indicated by the small SDs. All parameters, except r time at hour 24, were significant when compared with the T0 values. Comparison data for anti-Xa according to T4 were also significant but decreased below the therapeutic ranges at T24 (expected therapeutic range 0.50-1.0 U/mL for the adult population).
Table 1.
TEG® variables, aPTT, and anti-Xa concentration before and after subcutaneous injection of enoxaparin
| T0 | T4 | T12 | T24 | |
|---|---|---|---|---|
| r time, min | 3.81±2.12 | 11.74±1.65* | 6.62±1.42* | 3.51±2.12 |
| CI | 0.49±2.17 | -10.36±3.28* | -8.56±3.28* | -6.36±3.28* |
| aPTT, sec | 31.10±4.84 | 52.10±2.8* | 37.10±1.44* | 35.10±1.84* |
| Anti-Xa, U/mL | 0.00±0.00 | 0.77±0.19 | 0.65±0.19* | 0.34±0.19* |
Note: Differences were significant as compared with the T0 values (p<0.001).
Comparison data for anti-Xa were according to T4 values.
Anti-Xa-anti-Xa activity; aPTT-activated partial thromboplastin time; CI-coagulation index; min-minute; R time-reaction time; sec-seconds
Initially, the r time correlated with the expected peak and trough the levels of anti-Xa concentrations (p<0.053) (Fig. 2). An observable trend and acceleration of r time tended to normalize through T12, indicating decreased clot formation TEG. But, the mean±SD values were still between the reference values (2-8 min), showing inadequate coagulation. However, the total blood hemostatic potential and CI increased immediately after T0, peaking at hour 4, remained elevated (relative to baseline) at hour 24, and still did not return to admission levels (Fig. 3), expectedly.
Figure 2.
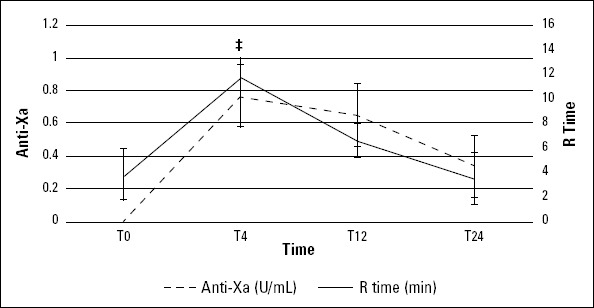
Anti-Xa concentration and r time between T0 and T24. Error bars reflect SE
*T0 vs. T4, p=0.002
Figure 3.
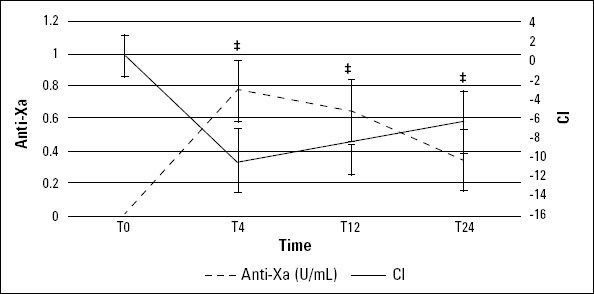
Anti-Xa concentration and CI value between T0 and T24. Error bars reflect SE
*T0 vs. T4, p<0.002, T0 vs. T12, p<0.002, T0 vs. T24, p=0.002
At each period after the completion of surgery, the calculated Spearman correlation (rs) between r time and anti-Xa was only significant at T4 (p=0.023) (Table 2). However, the correlation between CI and anti-Xa was significant for each time interval. Especially, at T24, when the anti-Xa level was below the reference ranges, CI was still a better indication of hypocoagu-lability in progress (p=0.031). There was no evident correlation between aPTT and anti-Xa levels (data not shown).
Table 2.
Correlation and P values between TEG® variables, aPTT, and anti-Xa concentration after subcutaneous injection of enoxaparin
| T4 (rs, P value) | T12 (rs, P value) | T24 (rs, P value) | |
|---|---|---|---|
| r time, min | 0.40, p=0.023* | 0.25, p=0.181 | 0.11, p=0.594 |
| Cl | -0.48, p=0.007* | -0.52, p=0.003* | -0.38, p=0.031* |
| aPTT, sec | 0.15, p=0.422 | 0.15, p=0.421 | 0.17, p=0.364 |
Note: Statistically significant of the correlation coefficient.
Anti-Xa-anti-Xa activity; aPTT-activated partial thromboplastin time; CI-coagulation index; min-minute; R time-reaction time; sec-seconds
Discussion
In this study, we have demonstrated a strong correlation between a basic TEG parameter, CI, and anti-Xa levels in patients following major orthopedic surgery receiving LMWH. Although the present studies suggested that monitoring with the thromboelastographic r value correlated significantly with the peak of anti-Xa levels, this was not demonstrated through time, with an inadequate clinical impact.
The accuracy of the assay rests on the correct timing of the sample in relation to dose administration. In this study, the mean time from dose to sampling was 4.13±0.1 h, consistent with recommendations for anti-Xa sampling.
Earlier studies have shown a correlation between TEG variables and routine coagulation tests (19, 20). There are only a few in vivo studies available on monitoring LMWH therapy by TEG in humans (21-23). Results, however, have been mixed, with some demonstrating a good correlation with TEG and less so in others. Many studies utilize healthy volunteers to provide samples for in vitro analysis (6, 24-26). Results from these investigations may not be relevant to patients who might display altered hemostasis changes, such as major surgery.
While LMWH seems to alter all TEG parameters, r time has been the most investigated parameter. R time is prolonged by clotting factor deficiencies, warfarin, unfractionated heparin, and LMWH treatment (23, 24) and has a tendency to decrease in thrombophilic conditions (25). Therefore, the fact that the r time is closely associated with the level of anti-Xa activity is not surprising. In this study, the r time correlated with the expected peak of anti-Xa concentrations but tended to normalize after hour 12. Surgical intervention stimulates platelets to release granular factors (26). Antropova et al. (27) suggested that active secretion of ß-thromboglobulin by platelets greatly impacts reaction time, peaking at the completion of surgery. The release of this factor might be as quick as surgical induction and end at the completion of surgery, causing the rapid normalization of r time just before the therapeutic LMWH interval. However, Louis et al. (28) studied TEG-adjusted enoxaparin dosing, which led to significant increases in anti-Xa activity but did not correlate with a decreased VTE rate. Failure to reduce the DVT rate and increase AR, despite increased dosing and increased anti-Xa activity, was consistent with the high rate of AT-III deficiency. They suggested evaluating the AT-III status before the optimization of LMWH.
Mainly, tests examining a single point of the coagulation cascade may not represent the overall coagulation picture or patient outcomes. Hayden et al. (29) found no evidence of association between r time and anti-Xa in hospitalized patients. Artang et al. (30) compared anti-Xa activity to a composite TEG parameter, called the thrombodynamic ratio, and noted a good correlation (using healthy male volunteers), whereas in vivo studies in monitoring hemostasis in patients undergoing major surgery were not investigated in detail. Therefore, we attempted to introduce additional TEG parameters to monitor anti-Xa activity. We studied the other TEG variable, coagulation index (CI), as a single integrated TEG parameter consisting of several basic parameters, for the assessment of LMWH treatment. CI has no units. CI describes the patient’s overall coagulation, derived from TEG variables. Normal values for the CI lie between -3.0 and +3.0. In essence, CI portrays an amplified profile of the prothrombotic or antithrombotic state in the sample. As a consequence, hypocoagulability is defined as a CI of less than -3. This means that patients with reaction time (r), coagulation time (k), alpha angle, or maximum amplitude on the ‘hypocoagulable side’ may still fall within the normal CI range and not meet the criteria to be called ‘hypocoagulable.’ CI is a unique variable that provides an overall global assessment of the clotting process, from fibrin formation to clot lysis, in real time. CI increased after LMWH injection, peaking at hour 4 and remaining elevated until the end of the study. The result suggested that thromboelastographic CI value correlated significantly with the peak of anti-Xa activity after the administration of enoxaparin (p<0.05), and this correlation between CI and anti-Xa activity was attributable to the influence of anticoagulant therapy within 2-4 hours after subcutaneous administration (31).
In this study, the peak plasma anti-Xa levels occurred 4 hours after the subcutaneous administration and decreased to 15% of peak values at hour 12 and to 55% of peak values at hour 24 after injection. Meanwhile, bleeding risk can not be predicted from available standard laboratory tests. Lack of point-of-care measurements for LMWH anticoagulant effects makes it difficult to predict which patients may be at particularly high risk for bleeding complications. In this study, there was a group of patients (45%) with a CI value still less than -3 at hour 24, whose anti-Xa activity was below the therapeutic range. However, r time was between the reference values at this time. This long-term hypocoagulability result given only by CI value may reflect an exaggerated response to LMWH and create caution for increased bleeding risk in these patients.
aPTT is the assay for monitoring unfractionated heparin (UFH). Patients receiving therapeutic doses of enoxaparin rarely have an aPTT value that exceeds the upper limit of the normal control. In this study, although anti-Xa activity was below the therapeutic ranges, aPTT progressively increased at each of the postoperative sample intervals measured. However, CI did not show apparent correlations with aPTT from T0 to T24. The total coagulation potential of blood showed a peak at 30 minutes after total hip arthroplasty and remained elevated at least until postoperative Day 14, correlating with The activity of the external coagulation pathway including aPTT did not show an apparent correlation with CI from T0 to T24. The mechanism underlying the uncorrelated relationship was not clear but might be related to patient-to-patient variability of the effects of LMWH.
Study limitations
One of the limitation of this study was that we did record the body mass index of patients. In our study, preoperative conditions were optimal, but we did not follow up the patients according to basic laboratory analysis for 24 hours. A reevaluation of the potentials and limitations of the method with the effects on the hemostatic system should be undertaken.
Conclusion
This study demonstrated a correlation between plasma anti-Xa activity and a composite parameter, CI, as determined by thromboelastography, in surgical patients treated with enoxaparin. However, the reaction time has a substantial correlation with anti-factor Xa levels. The current study may be an important first step in order to use CI to measure LMWH activity. Meanwhile, the value and usefulness of TEG in predicting bleeding or thrombotic complications following major orthopedic surgery merit further investigation. Larger trials on patients treated with different antithrombotic agents, including different LMWH preparations, are needed to correlate abnormal CI values with anti-Xa activity.
Footnotes
Conflict of interest: None declared.
Peer-review: Partially external peer-reviewed.
Authorship contributions: Concept - N. T., G.K., M. T.; Design - N. T., G.K., M. T.; Supervision - N. T.; Data collection&/or processing - N. T., G.K., M. T.; Analysis &/or interpretation - N. T., G.K., M. T.; Literature search - N. T., G.K., M. T.; Writing - N. T., G.K., M. T.; Critical review - N. T.
References
- 1.Geerts WH, Pineo GF, Heit JA, Bergqvist D, Lassen MR, Colwell CW, et al. Prevention of venous thromboembolism: The seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest. 2004;126:338–40. doi: 10.1378/chest.126.3_suppl.338S. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 2.Salzman EW. Heparin for prophylaxis of venous thromboembolism. Ann NY Acad Sci. 1989;556:371–7. doi: 10.1111/j.1749-6632.1989.tb22518.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 3.Kakkar VV, Corrigan T, Spindler J, Fossard DIP, Flute PT, Crellin RQ, et al. Efficacy of low doses of heparin in prevention of deep-vein thrombosis after major surgery. A double-blind, randomised trial. Lancet. 1972;2:101–6. doi: 10.1016/s0140-6736(72)91595-4. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 4.Gallus AS, Hirsh J, Tuttle RJ, Trebilcock R, O’Brien SE, Carroll JJ, et al. Small subcutaneous doses of heparin in prevention of venous thrombosis. New Eng J Med. 1973;288:545–51. doi: 10.1056/NEJM197303152881103. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 5.Cohen M. The role of low-molecular-weight heparins in the management of unstable angina and non-ST-segment elevation myocardial infarction. J Thromb Thrombolysis. 2001;11:171–4. doi: 10.1023/a:1011284902244. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 6.Amarigiri SV, Lees TA. Elastic compression stockings for prevention of deep vein thrombosis. Cochrane Database Syst Rev. 2000;3:CD001484. doi: 10.1002/14651858.CD001484. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 7.Kitchen S, Iampietro R, Woolley AM, Preston FE. Anti Xa monitoring during treatment with low molecular weight heparin or danaparoid: inter-assay variability. Thromb Haemost. 1999;82:1289–93. [PubMed] [Google Scholar]
- 8.Van PY, Cho SD, Underwood SJ, Morris MS, Watters JM, Schreiber MA. Thrombelastography versus anti-Factor Xa levels in the assessment of prophylactic-dose enoxaparin in critically ill patients. J Trauma. 2009;66:1509–15. doi: 10.1097/TA.0b013e3181a51e33. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 9.Gori AM, Fedi S, Pepe G, Falciani M, Rogolino A, Prisco D, et al. Tissue factor and tissue factor pathway inhibitor levels in unstable angina patients during short-term low-molecular-weight heparin administration. Br J Haematol. 2002;117:693–8. doi: 10.1046/j.1365-2141.2002.03522.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 10.Green B, Greenwood M, Saltissi D, Westhuyzen J, Kluver L, Rowell J, et al. Dosing strategy for enoxaparin in patients with renal impairment presenting with acute coronary syndromes. Br J Clin Pharmacol. 2005;59:281–90. doi: 10.1111/j.1365-2125.2004.02253.x. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rosencher N, Vassilieff N, Guigonis V, Toulon P, Conseiller C. Comparison of effects of elohes and albumin on hemostasis in orthopedic surgery. Ann Fr Anesth Reanim. 1992;11:526–30. doi: 10.1016/s0750-7658(05)80758-0. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 12.Wei KL, Lin CJ, Lai KA. Changes in coagulatory profile after orthopedic surgery. J Formos Med Assoc. 1995;94:541–7. [PubMed] [Google Scholar]
- 13.Rogers MC. Principles and practice as anesthesiology. In: Murray DJ, editor. Monitoring of hemostasis. Boston: Mosby Year Book; 1993. p. 853. [Google Scholar]
- 14.Spiess BD, Tuman KJ, McCarthy RJ, DeLaria GA, Schillo R, Ivankovich AD. Thromboelastography as an indicator of post-cardiopulmonary bypass coagulopathies. J Clin Monito. 1987;3:25–30. doi: 10.1007/BF00770880. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 15.Ereth MH, Nuttall GA, Klindworth JT, Mac Veigh I, Santrach PJ, Orszulak TA, et al. Does the platelet-activated clotting test (HemoSTATUS) predict blood loss and platelet dysfunction associated with cardiopulmonary bypass? Anesth Analg. 1997;85:259–64. doi: 10.1097/00000539-199708000-00004. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 16.Coppell JA, Thalheimer U, Zambruni A, Triantos CK, Riddell AF, Burroughs AK, et al. The effects of unfractionated heparin, low molecular weight heparin and danaparoid on the thromboelastogram (TEG): an in-vitro comparison of standard and heparinase-modified TEGs with conventional coagulation assays. Blood Coagul Fibinrolysis. 2006;17:97–104. doi: 10.1097/01.mbc.0000203859.62739.25. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 17.Zmuda K, Neofotistos D, Ts’ao CH. Effects of unfractionated heparin, low-molecular-weight heparin, and heparinoid on thromboelasto-graphic assay of blood coagulation. Am J Clin Pathol. 2000;113:725–31. doi: 10.1309/Q4AE-BMCW-CQ7J-NUVT. [DOI] [PubMed] [Google Scholar]
- 18.Gerotziafas GT, Chakroun T, Samama MM, Elalamy I. In vitro comparison of the effect of fondaparinux and enoxaparin on whole blood tissue factor-triggered thromboelastography profile. Thromb Haemost. 2004;92:1296–302. doi: 10.1160/TH03-11-0694. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 19.Chandler WL. The thromboelastography and the thromboelastograph technique. Semin Thromb Hemost. 1995;21:1–6. [PubMed] [Google Scholar]
- 20.Zuckerman L, Cohen E, Vagher JP, Woodward E, Caprini JA. Comparison of thrombelastography with common coagulation tests. Thromb Haemost. 1981;46:752–6. [PubMed] [Google Scholar]
- 21.Shinoda T, Arakura H, Katakura M, Shirota T, Nakagawa S. Usefulness of thrombelastography for dosage monitoring of low molecular weight heparin and unfractionated heparin during hemodialysis. Artif Organs. 1990;14:413–5. doi: 10.1111/j.1525-1594.1990.tb02996.x. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 22.Klein SM, Slaughter TF, Vail PT, Ginsberg B, El-Moalem HE, Alexander R, et al. Thromboelastography as a perioperative measure of anticoagulation resulting from low molecular weight heparin: a comparison with anti-Xa concentrations. Anesth Analg. 2000;91:1091–5. doi: 10.1097/00000539-200011000-00009. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 24.Traverso CI, Caprini JA, Arcelus JI, Arcelus IM. Thromboelastographic modifications induced by intravenous and subcutaneous heparin administration. Semin Thromb Hemost. 1995;21:53–8. doi: 10.1055/s-0032-1313622. [DOI] [PubMed] [Google Scholar]
- 25.Traverso CI, Arcelus JI, Caprini JA. Role of thromboelastography in evaluating other anticoagulant agents: warfarin and heparin-dihydroergotamine. Semin Thromb Hemost. 1995;21:59–62. doi: 10.1055/s-0032-1313623. [DOI] [PubMed] [Google Scholar]
- 26.Arcelus JI, Traverso CI, Caprini JA. Thromboelastography for the assessment of hypercoagulability during general surgery. Semin Thromb Hemost. 1995;21:21–6. doi: 10.1055/s-0032-1313617. [DOI] [PubMed] [Google Scholar]
- 27.Bunescu A, Widman J, Lenkei R, Menyes IP, Levin K, Egberg N. Increases in circulating levels of monocyte-platelet and neutrophil-platelet complexes following hip arthroplasty. Clin Sci (Lond) 2002;102:279–86. [CrossRef] [PubMed] [Google Scholar]
- 28.Antropova IP, Reino EV, Yushkov BG, Shlykov IL, Varaksin AN. Blood coagulation at major orthopaedic surgery. J Blood Disorders Transf. 2013;5:1. [Google Scholar]
- 29.Louis SG, Van PY, Riha GM, Barton JS, Kunio NR, Underwood SJ, et al. Thromboelastogram-guided enoxaparin dosing does not confer protection from deep venous thrombosis: a randomized controlled pilot trial. J Trauma Acute Care Surg. 2014;76:937–42. doi: 10.1097/TA.0000000000000165. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 30.White H, Sosnowski K, Bird R, Jones M, Solano C. The utility of thromboelastography in monitoring low molecular weight heparin therapy in the coronary care unit. Blood Coagul Fibrinolysis. 2012;23:304–10. doi: 10.1097/MBC.0b013e32835274c0. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 31.Artang R, Frandsen NJ, Nielsen JD. Application of basic and composite thrombelastography parameters in monitoring of the antithrombotic effect of the low molecular weight heparin dalteparin: an in vivo study. Thromb J. 2009;7:14. doi: 10.1186/1477-9560-7-14. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Horlocker TT, Heit JA. Low molecular weight heparin: biochemistry, pharmacology, perioperative prophylaxis regimens, and guidelines for regional anesthetic management. Anesth Analg. 1997;85:87485. doi: 10.1097/00000539-199710000-00031. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 33.Sharma S, Uprichard J, Moretti A, Boyce H, Szydlo R, Stocks G. Use of thromboelastography to assess the combined role of pregnancy and obesity on coagulation: a prospective study. Int J Obstet Anesth. 2013;22:113–8. doi: 10.1016/j.ijoa.2012.12.004. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 34.Muller MC, Meijers JC, Vroom MB, Juffermans NR. Utility of thromboelastography and/or thromboelastometry in adults with sepsis: a systematic review. Critical Care. 2014;18:R30. doi: 10.1186/cc13721. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]


