Abstract
Thiamine (Vitamin B1) is an essential nutrient and indispensable for normal growth and development of the organism due to its multilateral participation in key biochemical and physiological processes. Humans must obtain thiamine from their diet since it is synthesized only in bacteria, fungi and plants. Thiamine deficiency (TD) can result from inadequate intake, increased requirement, excessive deletion and chronic alcohol consumption. TD affects multiple organ systems, including the cardiovascular, muscular, gastrointestinal, and central and peripheral nervous systems. In the brain, TD causes a cascade of events including mild impairment of oxidative metabolism, neuroinflammation and neurodegeneration, which are commonly observed in neurodegenerative diseases, such as Alzheimer’s disease (AD), Parkinson’s disease (PD) and Huntington’s disease (HD). Thiamine metabolites may serve as promising biomarkers for neurodegenerative diseases and thiamine supplementations exhibit therapeutic potential for patients of some neurodegenerative diseases. Experimental TD has been used to model aging-related neurodegenerative diseases. However, to date, the cellular and molecular mechanisms underlying TD-induced neurodegeneration are not clear. Recent research evidence indicates that TD causes oxidative stress, endoplasmic reticulum (ER) stress and autophagy in the brain, which are known to contribute to the pathogenesis of various neurodegenerative diseases. In this review, we discuss the role of oxidative stress, ER stress and autophagy in TD-mediated neurodegeneration. We propose that it is the interplay of oxidative stress, ER stress and autophagy that contributes to TD-mediated neurodegeneration.
Keywords: Alcoholism, protein degradation, reactive oxygen species, unfolded protein response, vitamin B1
Introduction
Thiamine, also referred to as thiamin and vitamin B1, is a water-soluble and colorless vitamin with a characteristic odor and slightly bitter taste [1]. Thiamine was first discovered by two Dutch chemists, Dr. B. C. P. Jansen and his closest collaborator Dr. W. Donath in 1926, and it was isolated, purified and synthesized by Dr. R. R. Williams in 1936 [2]. Thiamine is an essential micronutrient for humans, which is supplied from a diet rich in thiamine [3]. Thiamine is enriched in lean pork, beef, wheat germ and whole grains, organ meats, eggs, fish, legumes, and nuts [4]. In humans, thiamine exists in 4 forms: unphosphorylated thiamine, thiamine monophosphate, thiamine diphosphate, and thiamine triphosphate [5]. Thiamine plays an essential role for life and health for humans since thiamine derivatives and thiamine-dependent enzymes play a key role in cell metabolism [6, 7].
Thiamine is a dicyclic compound with thiazole and aminopyrimidine rings joined by a methylene bridge. Thiamine is absorbed into the body in the upper small intestine by two transporters, THTR1 and THTR2, the products of the SLC19A2 and SLC19A3 genes, respectively [8]. Thiamine diphosphate is also known as thiamine pyrophosphate (TPP), which is the biologically active form of thiamine and serves as a cofactor for four major enzyme systems: pyruvate dehydrogenase (EC 1.2.4.1) complex, an organized enzyme assembly that connects glycolysis with the tricarboxylic acid (TCA) cycle; α-ketoglutarate dehydrogenase (EC 1.2.4.2) complex (KGDHC), a multicomponent enzyme complex associated with the TCA cycle; transketolase (EC 2.2.1.1), a key participant in the pentose phosphate shunt involved in nucleic acid and lipid biosynthesis; and branched-chain alpha-ketoacid dehydrogenase (EC 1.2.4.4) complex which is required for the metabolism of branched-chain amino acids [9]. Thiamine concentrations are measured either by direct measurement via HPLC in plasma, whole blood, erythrocytes, or by determination of the activity of a thiamine-dependent enzyme in erythrocytes called transketolase.
Thiamine deficiency (TD) can be caused by a variety of reasons, such as inadequate intake, increased requirement, excessive loss, consumption of foods containing anti-thiamine components, or a combination of these factors [10-12]. Decreased thiamine levels are correlated with the aging process [13]. The recommended daily allowance for thiamine in most countries is set at about 1.4 mg [14], while chronic alcohol consumption is the primary cause of TD in the United States and other developed countries [15, 16]. Evidence suggests that TD occurs in chronic alcoholics at a frequency of at least 25–31 % and up to 80 % [17]. TD adversely affects many organ systems including the cardiovascular [18], muscular [19], gastrointestinal [20], and central and peripheral nervous systems [21]. The central nervous system (CNS) is particularly sensitive to TD, because of its dependence on TPP-mediated metabolic pathways involved in energy metabolism and neurotransmitter synthesis [22]. Insufficient levels of thiamine causes neurological and metabolic disorders which may be corrected by thiamine administration.
TD disease remains a significant world health problem. Because thiamine is an important cofactor involved in processes associated with the metabolism of lipids, glucose, amino acids, and neurotransmitters, its deficiency can lead to severe complications in both the nervous and cardiovascular systems. Generally, cardiac effects caused by TD are referred to as wet beriberi, the peripheral neurological manifestations are called dry beriberi, and delirium or mental confusion are termed Wernicke’s encephalopathy [23]. Beriberi was thought of as a disease of the peripheral nervous system, heart, and muscles until the 1930s when it was found that Wernicke’s encephalopathy was the acute cerebral manifestation of severe thiamin deficiency[24]. In humans, the neurological disorder that is most clearly associated with TD is Wernicke-Korsakoff syndrome (WKS), characterized by behavioral, cognitive and neuropathological deficits [25]. Administration of thiamine is able to prevent, reduce or reverse several symptoms in WKS patients, whereas some deficits persist permanently [26]. TD is also implicated in several neurodegenerative diseases, such as Alzheimer’s disease (AD), Parkinson’s disease(PD), and alcohol-induced dementia [27]. Therapeutic effects of thiamine supplementation have been observed in some neurodegenerative diseases and alcohol-induced dementia [28-30]. In addition, accumulating evidence has been documented that TD is able to cause regionally specific neuronal death in animal and human brains which is accompanied with a mild chronic impairment of oxidative metabolism [31-33]. TD also causes the activation of microglia, astrocytes and endothelial cells [34] as well as abnormalities of cerebral glucose metabolism [35]. These features are commonly observed in neurodegenerative diseases and make TD a feasible model to investigate the mechanisms of neurodegeneration. Although TD-induced brain damages have been well documented, the underlying cellular and molecular mechanisms are still unclear.
Oxidative stress, endoplasmic reticulum (ER) stress and autophagy have been implicated in pathogenesis of various neurodegenerative diseases, such as AD, PD, Huntington’s disease (HD), and Amyotrophic Lateral Sclerosis (ALS) [36, 37]. Increasing evidence demonstrates that TD also causes oxidative stress, ER stress and autophagy in the brain. In this review, we discuss the recent progress in this line of research. We propose that it is the interplay of oxidative stress, ER stress and autophagy that contributes to TD-mediated neurodegeneration.
1. TD and neurodegeneration
Although many organ systems are affected by TD, the CNS is particularly vulnerable to TD. This may be attributed to thiamine having lower levels in the human brain than in other organs [38]. TPP, the main active form of thiamine, functions as an essential co-factor for a number of thiamine-dependent enzymes, such as transketolase, pyruvate dehydrogenase complex (PDHC), and α-ketoglutarate dehydrogenase complex (KGDHC). Once the levels of thiamine are diminished in the human brain, the activity of these enzymes is affected, leading to alterations in mitochondrial activity, impairment of oxidative metabolism, decreased energy status and brain damage [39].
TD is implicated in a number of neurodegenerative diseases and is also involved in alcohol abuse-induced brain damages. WKS is a neuropsychiatric disorder caused by TD, which is composed of two clinical stages. The acute stage, Wernicke’s encephalopathy, has historically been associated with a classic triad of acute neurological symptoms: confusion, ataxia, and ophthalmoplegia. The chronic stage, Korsakoff syndrome, follows those who survive the acute stage and is a life-altering, permanent neuropsychiatric condition characterized by anterograde and retrograde amnesia as well as frontal lobe dysfunction and affective disturbance [40, 41]. The first observation showing the association between TD and the etiology of WKS could be traced back to the 1930s [42]. It has now been established that TD is the primary cause of WKS. Clinically, WKS is usually associated with chronic alcoholism which depletes thiamine in the human body, but it can also be caused by malnourishment [43, 44].
AD is an age-related and progressive neurodegenerative disease characterized by the deposition of β-amyloid senile plaques and tau-associated neurofibrillary tangles [45]. Patients with AD exhibit memory impairment and cognitive decline due to neuronal loss mainly in the neocortex and the hippocampus [46]. AD currently affects over 36 million people worldwide with an estimated global economic impact of approximately $605 billion dollars in 2010 in addition to incalculable social and emotional burdens [47, 48]. Several lines of evidence have shown an association between TD and AD. For example, plasma thiamine levels were reduced in patients diagnosed with AD, suggesting that some AD patients have TD [49]. In addition, one of the main features of AD is the severe reduction of glucose metabolism in the brain of AD patients [50-52]. There are three thiamine-dependent enzymes, including transketolase, PDHC, and KGDHC, which function at key steps of glucose metabolism. It has been documented that the activities of these thiamine-dependent enzymes were also diminished in AD brains [53-55]. Moreover, the reductions in thiamine-dependent activity are associated with the severity of the dementia in AD patients [56]. In animal models, TD increased the accumulation of beta-amyloid, exacerbates plaque formation, promotes phosphorylation of tau and impairs memory, suggesting TD might play a role in the pathology of AD [57, 58]. Treatment of benfotiamine, a thiamine derivative with better bioavailability than thiamine, could reduce plaques, decrease phosphorylation of tau and reverse memory deficits in a mouse model of AD [59]. However, in the clinical trials, the therapeutic effects of thiamine supplementation are inconsistent in AD patients. One report suggests that 3 to 8 g/day of thiamine administered orally have a mild beneficial effect in dementia of AD [60]. On the contrary, some other studies indicate that there is no significant improvement by thiamine supplementation in AD patients [61, 62]. Therefore, the contribution of TD in the pathophysiology of AD remains to be clarified.
PD is the second most common form of neurodegeneration in the elderly population, characterized by resting tremor, rigidity, slowness of movement, and postural imbalance [63]. It affects more than 1% of individuals older than 55 years of age and more than 3% of those older than 75 years of age [64]. The loss of dopaminergic neurons in the substantia nigra is a major pathophysiological feature of patients with PD [65]. Several lines of evidence have shown that thiamine or thiamine-dependent processes may be involved in the pathogenesis of PD. For example, lower free thiamine levels in the cerebrospinal fluid were found in PD patients in comparison to normal individuals, while levels of thiamine-derivatives, such as thiamine-diphosphate and thiamine-monophosphate, did not differ significantly [66]. Additionally, the immunoreactivity of KGDHC, one of the most important thiamine-dependent enzymes, was decreased in the substantia nigra of patients with PD, and the reduction seemed correlated with the severity of degeneration [67, 68]. In some studies, thiamine supplementation seemed to improve the outcomes for PD patients. For example, administration of parenteral high-dose thiamine was effective in reversing motor and non-motor symptoms in PD patients [28, 30]. These findings suggest that TD may be involved in the pathophysiology of PD.
It has been well established that chronic alcohol abuse causes both dementia and TD. Chronic alcohol consumption can lead to at least two long-lasting neurological disorders associated with severe cognitive dysfunction: alcohol-associated dementia (AAD) and WKS [69]. For example, it has been estimated that greater than 10% of alcoholic patients have symptoms of either AAD or WKS [70]. Wernicke's encephalopathy and Korsakoff's psychosis in alcoholics are thought to be caused by TD. When the process goes untreated, patients may develop alcohol-induced persisting dementia [27]. Poor dietary patterns, along with the impact of alcohol consumption on absorption, storage, activation and excretion of thiamine are thought to be the mechanisms for TD among alcohol-dependent individuals [71].
Since TD is associated with various neurodegenerative diseases and shares many common pathological features, experimental TD has been extensively used to investigate the mechanisms of neurodegeneration [32, 72, 73].
2. Mechanisms underlying TD-mediated neurodegeneration
2.1. Oxidative stress
Oxidative stress is defined as disequilibrium between reactive oxygen species (ROS) production and the ability to detoxify the reactive oxygen intermediates [74]. The brain is particularly vulnerable to oxidative stress due to its high oxygen consumption and high production of reactive oxygen and nitrogen species. Accumulating evidence has demonstrated that oxidative stress plays an important role in excitotoxicity, axonal damage and neuronal death [75], and is involved in the onset and progression of several neurodegenerative diseases, such as AD, PD, HD, and ALS [76, 77].
Like in neurodegenerative diseases, oxidative stress seems to play an important role in TD-induced brain damage. Both animal and cellular models have clearly shown that TD increases ROS production and causes oxidative stress in the brain and neuronal cells. For example, in TD animal model, a wide spectrum of markers of oxidative stress, such as heme oxygenase-1, superoxide dismutase, ferritin, and malondialdehyde are increased in the brain [32, 78, 79]. In addition, lipid peroxidation product, 4-hydroxynonenal (HNE), are also increased in neurons of the CNS [80]. The changes in the levels of antioxidant enzymes, lipid peroxidation, and protein carbonyl content in brain mitochondria have been reported in the brains of TD animals [81]. Moreover, it has also been demonstrated that TD selectively causes oxidative stress in neurons in vitro [58, 82].
There are several mechanisms underlying TD-induced oxidative stress. One possible mechanism might be due to mitochondrial dysfunction. Mitochondria are a major source of ROS, and also a target of TD. Previous studies indicate that binding site densities for the translocator protein, a useful marker of mitochondrial dysfunction, are increased in the brains of TD animals [83]. TD may damage the antioxidant system in the brain. For example, antioxidant enzymes such as superoxide dismutase, catalase, and glutathione peroxidase are significantly reduced in the brain of TD animals. [81]. Another possible mechanism is neuroinflammation which may contribute TD-induced CNS damage. TD significantly increases pro-inflammatory cytokines, chemokines, interferons, as well as many transcription factors known to control inflammatory gene expression are upregulated in TD animals [33]. The neuroinflammation may alter mitochondrial membrane potential and thus its function, resulting in an increase in ROS [84]. Reversely, oxidative stress can also induce neuroinflammation. For example, there is increasing evidence that activated microglia produce excessive ROS during aging and hypoxia, resulting in the nuclear factor-κB-dependent excessive production of proinflammatory mediators, such as interleukin-1β, tumor necrosis factor-α, and interleukin-6 [85].
2.2. ER stress
ER is the largest organelle found in the cells of eukaryotic organisms. It is an interconnected network of membrane-enclosed tubules and sacs that extends from the nuclear membrane throughout the cytoplasm. ER plays a critical role in a wide range of cellular processes. It regulates protein folding, modification and transport. It also processes the synthesis and distribution of phospholipids and steroids. ER is the site of calcium ion storage and regulates intracellular calcium homeostasis [86, 87].
Under physiologic conditions, there is a balance between ER protein load and folding capacity. Alterations in ER homeostasis, such as increased protein synthesis, accumulation of misfolded proteins, or alterations in the calcium or redox balance, lead to a condition called ER stress [88]. The cellular adaptive response to cope with ER stress is known as the unfolded protein response (UPR). Initially, the UPR functions to restore ER homeostasis by increasing protein folding capacity and decreasing the accumulation of unfolded proteins. Through a complex network, UPR acts to interrupt new protein translation, assist in proper protein folding and promote the degradation of accumulated unfolded or misfolded proteins, leading to a reduction of ER stress. If these mechanisms of adaptation and correction are insufficient to restore ER homeostasis, ER stress is sustained, resulting in apoptotic cell death [89, 90]. Therefore, UPR is generally considered a cellular defense mechanism. During the UPR, three major UPR sensors, namely PERK (protein kinase RNA-like ER kinase), IRE1 (inositol requiring protein-1), and ATF6 (activating transcription factor-6), transduce information about the folding status of the ER to the cytosol and nucleus to restore folding capacity [91, 92].
Neurodegenerative diseases, such as AD, PD, HD, ALS and prion-related diseases (PRD) have in common the presence of protein aggregates in specific brain areas where significant neuronal loss is detected. Emerging evidence suggests that ER stress may contribute to the neurodegeneration in these neurodegenerative diseases [93]. For example, upregulation of UPR sensors has been observed in post mortem brain tissues and experimental models of neurodegenerative diseases, such as AD, PD, HD, ALS and PRD [94, 95]. In experimental models of neurodegenerative diseases, targeting UPR pathways appears beneficial for alleviating neuronal loss[96]. We investigated the effect of TD on the ER with transmission electron microscopy and showed that TD causes an abnormality in ER structure [97]. We further demonstrated that TD up-regulates several markers of ER stress in the brain and cultured neurons, such as GRP78, CHOP and the phosphorylation of eIF2α [97]. Furthermore, TD activates an ER stress-regulated caspase, caspase-12 and blocking the caspase-12 activation significantly alleviates TD-induced neuronal death[97]. We have also shown that TD activates double stranded RNA-activated protein kinase (PKR) which is also believed to play an important role in ER stress [97, 98]. These findings suggest that ER stress may underlie TD-induced damage to the CNS. To date, the mechanisms underlying TD-induced ER stress is unclear. It has been well documented that oxidative stress and disruption of calcium homeostasis can cause ER stress [99, 100]. TD is shown to cause oxidative stress and the disruption of intracellular calcium concentration [97, 101]. Therefore, it is likely TD may induce ER stress through these mechanisms.
2.3. Autophagy
Autophagy is a cellular catabolic process for the degradation of cytoplasmic components within lysosomes [102, 103]. Autophagy plays a major role in the elimination of cellular waste components, the renewal of intracellular proteins and the prevention of the build-up of redundant or defective material [104]. There are three types of autophagy that are commonly described, namely chaperone-mediated autophagy, microautophagy, and macroautophagy and the latter being the major and best characterized subtype of autophagy [105]. Autophagy consists of several sequential steps, including sequestration/formation of autophagosomes, transportation to lysosomes, degradation, and recycling of degraded products [106].
Autophagy is fundamental for the maintenance of homeostasis and especially important in post-mitotic neuronal cells. Autophagy sequesters damaged and dysfunctional organelles/protein, and transports them to lysosomes for degradation/recycling. This process provides nutrients for injured neurons. Without competent autophagy, abnormal protein aggregates accumulate, resulting in neurodegeneration, which is a common feature of neurodegenerative diseases [107]. Recent studies have demonstrated that impairments of the autophagic process are associated with several neurodegenerative diseases, such as AD, PD and HD [108]. The deficiency of autophagic pathways documented in neurodegenerative diseases is generally caused by three forms of dysregulations; these include insufficient autophagy activation, autophagy dysfunction due to impaired lysosomal function, and autophagic stress resulting from pathologic activation[109]. In general, autophagy is neuroprotective and deficiency in autophagy may facilitate neurodegeneration. However, dysregulated or excessive autophagy can also cause neuronal atrophy, neurite degeneration and cell death [110]. Therefore, dependent on the specific situation and model system, autophagy may have dual roles in both neuroprotection and neurodegeneration.
We investigated the effects of TD on the autophagy in the brain and cultured neurons. We demonstrated that TD induces the accumulation of autophagosomes in neurons measured by transmission electron microscopy, and the up-regulation of autophagic markers LC3-II, Atg5, and Beclin1 [73]. TD-induced expression of autophagic markers was reversed once thiamine was re-administered. Inhibition of autophagy by wortmannin and Beclin1 siRNA potentiates TD-induced death of neuronal cells. In contrast, activation of autophagy by rapamycin alleviated neuronal cell death induced by TD in vitro and in the brain. Intraperitoneal injection of rapamycin stimulates neuronal autophagy and attenuates TD-induced neuronal death and microglia activation in the submedial thalamus nucleus (SmTN) [73]. These results suggest that autophagy is neuroprotective in response to TD-induced neurodegeneration in the CNS. There are several potential mechanisms for TD-induced autophagy. For example, TD is shown to affect mTOR and AMPK pathways which are critical regulators of autophagy [73]. Additionally, TD may activate autophagy through the induction of oxidative stress and/or ER stress because both oxidative stress and ER stress are known to stimulate autophagy [111].
2.4 Interplay among oxidative stress, ER stress and autophagy
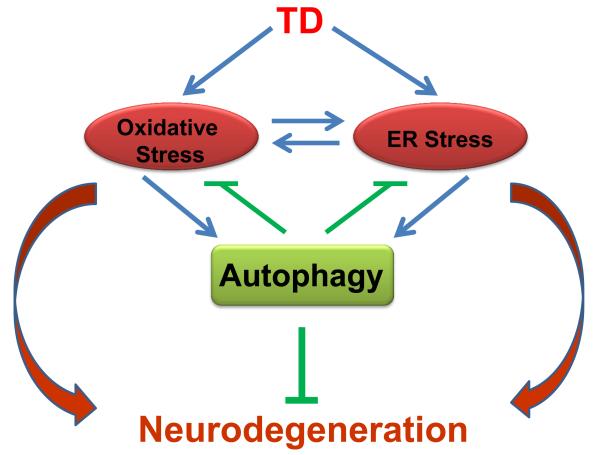
The recent research progress has clearly demonstrated that TD can activate oxidative stress, ER stress and autophagy and the activation is associated with TD-induced neurodegeneration. There is considerable interaction among oxidative stress, ER stress and autophagy. For example, oxidative stress and ER stress are interconnected; usually oxidative stress induces ER stress but sometimes ER stress can promote oxidative stress as well [112]. Both oxidative stress and ER stress may activate autophagy [111]. The interaction among oxidative stress, ER stress and autophagy is complex; so is the contribution of their interaction to TD-induced neurodegeneration. We propose a model of interaction of these three components in the context of TD-induced neurodegeneration (Fig. 1). In this model, TD first induces damages to the intracellular organelles which cause oxidative stress and ER stress, resulting in neurodegeneration. TD-induced cellular injuries and/or oxidative stress and ER stress activate autophagy which initially functions as a protective mechanism to eliminate oxidative stress- or ER stress-induced cellular damage. Autophagy can also be activated by TD through some key autophagy regulators, such as mTOR or AMPK. We have recently demonstrated that the interplay among oxidative stress, ER stress and autophagy is involved in ethanol-induced neurodegeneration [113]. Therefore, oxidative stress, ER stress and autophagy are interconnected and interdependent processes. In normal neurons, the homeostasis of these dynamic processes is well controlled and maintained. However, TD disrupts the homeostasis, eventually causing neurodegeneration in the CNS.
Figure 1.
The interplay of oxidative stress, ER stress and autophagy in TD-induced neurodegeneration. TD induces oxidative stress and ER stress in the brain, which results in neurodegeneration. Oxidative stress and ER stress can mutually regulate each other. Oxidative stress and ER stress can activate autophagy. Autophagy is activated in response to TD-induced neurotoxicity to alleviate oxidative stress and ER stress, therefore offering protection against TD-induced neurodegeneration. It is the interplay of these processes that contribute to the final outcomes of TD neurotoxicity.
3. Therapeutic implications
Experimental TD models offer an excellent opportunity to identify and evaluate therapeutic approaches to alleviate neurodegeneration. These approaches are of potential clinical significance for some neurodegenerative diseases. First, thiamine is able to across the blood-brain barrier[114]. Thiamine supplementation has been used as a therapy for patients with some neurodegenerative diseases. For example, thiamine (3-8 g/day) administered orally had a mild beneficial effect in AD patients[60]. The injection of high doses of thiamine (100 mg) parenterally twice a week was effective in reversing the symptoms of PD[28]. However, the clinical outcomes are inconsistent. A careful re-evaluation of thiamine supplementation is therefore necessary. The dosage, combination with other nutrients and the route of thiamine administration must be taken into careful consideration in clinical settings. Second, for reducing oxidative stress, the antioxidants which have potent antioxidant properties while having minimal side-effects should be evaluated. For example, natural antioxidants, such as anthocyanins, curcumin and resveratrol are suitable for screening [115]. Third, many compounds directly targeting the UPR and ER protein transportation system are under investigation [116, 117]. These compounds are worthy to be evaluated in experimental TD models because they may specifically eliminate ER stress and be beneficial to treat TD-induced ER stress and subsequent neurodegeneration with reduced side-effects. Fourth, since the activation of autophagy appears protective against ethanol neurotoxicity, pharmacological manipulation of autophagy should be considered a potential therapeutic approach. The most well studied drug that induces autophagy is rapamycin, an MTORC1 inhibitor. Rapamycin is a FDA-approved drug and a widely used immune suppressor in clinical transplant patients. Rapamycin and its analogs (CCI-779, RAD001 and AP23573) have been used to activate autophagy in vitro and in vivo[118, 119]. We showed that rapamycin alleviates TD-induced neurodegeneration [62]. In addition to directly targeting MTOR, activation of autophagy by manipulating other pathways upstream of MTORC1, such as AMPK, PI3K-AKT, GSK3B or TSC1/2 are alternative approaches. A number of studies of chemical screening have identified chemicals and small molecule enhancers that activate autophagy independent of MTOR, including some FDA approved drugs [119]. These compounds are also worthwhile to be evaluated in experimental TD models. Despite the potential beneficial effects of autophagy in neurodegenerative diseases, excessive autophagy may be detrimental and cause cell death. The experimental TD models offer a good system to develop strategies for appropriate activation of autophagy.
Conclusions
Decreased thiamine levels are associated with the aging process and TD causes neurodegeneration. TD and some neurodegenerative diseases share many common features. The mechanisms underlying TD-induced neurodegeneration, however, remain unclear. A large body of recent research evidence indicates that TD can cause oxidative stress, ER stress and autophagy in the brain. We propose that it is the interplay of oxidative stress, ER stress and autophagy that contributes to TD-mediated neurodegeneration. Therefore, therapeutic strategies simultaneously targeting oxidative stress, ER stress and autophagy may provide more effective outcomes.
Acknowledgments
We thank Jacqueline Frank for reading this manuscript. This work was supported by grants from the National Institutes of Health (NIH) (AA017226 and AA015407). It is also supported in part by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development (Biomedical Laboratory Research and Development: BX001721).
ABBREVIATIONS
- AD
Alzheimer’s disease
- ALS
Amyotrophic lateral sclerosis
- ATF6
activating transcription factor-6
- ER
endoplasmic reticulum
- HD
Huntington’s disease
- HNE
4-hydroxynonenal
- IRE1
inositol requiring protein-1
- KGDHC
α-ketoglutarate dehydrogenase complex
- PD
Parkinson’s disease
- PDHC
pyruvate dehydrogenase complex
- PERK
protein kinase RNA-like ER kinase
- PRD
prion-related diseases
- ROS
reactive oxygen species
- TD
thiamine deficiency
- TPP
thiamine pyrophosphate
- UPR
unfolded protein response
- WKS
Wernicke-Korsakoff syndrome
Footnotes
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Sica DA. Loop diuretic therapy, thiamine balance, and heart failure. Congest Heart Fail. 2007;13(4):244–7. doi: 10.1111/j.1527-5299.2007.06260.x. [DOI] [PubMed] [Google Scholar]
- 2.Wooley JA. Characteristics of thiamin and its relevance to the management of heart failure. Nutr Clin Pract. 2008;23(5):487–93. doi: 10.1177/0884533608323430. [DOI] [PubMed] [Google Scholar]
- 3.Pacal L, Kuricova K, Kankova K. Evidence for altered thiamine metabolism in diabetes: Is there a potential to oppose gluco- and lipotoxicity by rational supplementation? World J Diabetes. 2014;5(3):288–95. doi: 10.4239/wjd.v5.i3.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kerns JC, Arundel C, Chawla LS. Thiamin deficiency in people with obesity. Adv Nutr. 2015;6(2):147–53. doi: 10.3945/an.114.007526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Saif MW. Is there a role for thiamine in the management of congestive heart failure? South Med J. 2003;96(1):114–5. doi: 10.1097/01.SMJ.0000049859.82132.20. [DOI] [PubMed] [Google Scholar]
- 6.Osiezagha K, et al. Thiamine deficiency and delirium. Innov Clin Neurosci. 2013;10(4):26–32. [PMC free article] [PubMed] [Google Scholar]
- 7.Balakumar P, et al. The multifaceted therapeutic potential of benfotiamine. Pharmacol Res. 2010;61(6):482–8. doi: 10.1016/j.phrs.2010.02.008. [DOI] [PubMed] [Google Scholar]
- 8.Zhao R, Goldman ID. Folate and thiamine transporters mediated by facilitative carriers (SLC19A1-3 and SLC46A1) andfolate receptors. Mol Aspects Med. 2013;34(2-3):373–85. doi: 10.1016/j.mam.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Alstrup Lie M, Schiott B. A DFT study of solvation effects on the tautomeric equilibrium and catalytic ylide generation of thiamin models. J Comput Chem. 2008;29(7):1037–47. doi: 10.1002/jcc.20860. [DOI] [PubMed] [Google Scholar]
- 10.Moulin P, et al. [Thiamine deficiency in infants: a case report] Arch Pediatr. 2014;21(4):392–5. doi: 10.1016/j.arcped.2014.01.009. [DOI] [PubMed] [Google Scholar]
- 11.Vimokesant SL, et al. Effects of betel nut and fermented fish on the thiamin status of northeastern Thais. Am J Clin Nutr. 1975;28(12):1458–63. doi: 10.1093/ajcn/28.12.1458. [DOI] [PubMed] [Google Scholar]
- 12.Boniol S, et al. Wernicke encephalopathy complicating lymphoma therapy: case report and literature review. South Med J. 2007;100(7):717–9. doi: 10.1097/SMJ.0b013e318061920a. [DOI] [PubMed] [Google Scholar]
- 13.Zhang G, et al. Thiamine nutritional status and depressive symptoms are inversely associated among older Chinese adults. J Nutr. 2013;143(1):53–8. doi: 10.3945/jn.112.167007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Afadlal S, Labetoulle R, Hazell AS. Role of astrocytes in thiamine deficiency. Metab Brain Dis. 2014;29(4):1061–8. doi: 10.1007/s11011-014-9571-y. [DOI] [PubMed] [Google Scholar]
- 15.Hoyumpa AM., Jr. Mechanisms of thiamin deficiency in chronic alcoholism. Am J Clin Nutr. 1980;33(12):2750–61. doi: 10.1093/ajcn/33.12.2750. [DOI] [PubMed] [Google Scholar]
- 16.Martin PR, Singleton CK, Hiller-Sturmhofel S. The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health. 2003;27(2):134–42. [PMC free article] [PubMed] [Google Scholar]
- 17.Abdou E, Hazell AS. Thiamine deficiency: an update ofpathophysiologic mechanisms and future therapeutic considerations. Neurochem Res. 2015;40(2):353–61. doi: 10.1007/s11064-014-1430-z. [DOI] [PubMed] [Google Scholar]
- 18.Ahmed M, et al. Thiamin deficiency and heart failure: the current knowledge and gaps in literature. Heart Fail Rev. 2015;20(1):1–11. doi: 10.1007/s10741-014-9432-0. [DOI] [PubMed] [Google Scholar]
- 19.Koike H, et al. Myopathy in thiamine deficiency: analysis of a case. J Neurol Sci. 2006;249(2):175–9. doi: 10.1016/j.jns.2006.06.016. [DOI] [PubMed] [Google Scholar]
- 20.Francini-Pesenti F, et al. Wernicke's encephalopathy during parenteral nutrition. JPEN J Parenter Enteral Nutr. 2007;31(1):69–71. doi: 10.1177/014860710703100169. [DOI] [PubMed] [Google Scholar]
- 21.Nardone R, et al. Thiamine deficiency induced neurochemical, neuroanatomical, and neuropsychological alterations: a reappraisal. ScientificWorldJournal. 2013;2013:309143. doi: 10.1155/2013/309143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vernau K, et al. Thiamine Deficiency-Mediated Brain Mitochondrial Pathology in Alaskan Huskies with Mutation in SLC19A3.1. Brain Pathol. 2014;25(4):441–53. doi: 10.1111/bpa.12188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rao SN, Chandak GR. Cardiac beriberi: often a missed diagnosis. J Trop Pediatr. 2010;56(4):284–5. doi: 10.1093/tropej/fmp108. [DOI] [PubMed] [Google Scholar]
- 24.Haas RH. Thiamin and the brain. Annu Rev Nutr. 1988;8:483–515. doi: 10.1146/annurev.nu.08.070188.002411. [DOI] [PubMed] [Google Scholar]
- 25.Dror V, et al. Rasagiline prevents neurodegeneration in thiamine deficient rats-a longitudinal MRI study. Brain Res. 2014;1557:43–54. doi: 10.1016/j.brainres.2013.12.030. [DOI] [PubMed] [Google Scholar]
- 26.Sechi G, Serra A. Wernicke's encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007;6(5):442–55. doi: 10.1016/S1474-4422(07)70104-7. [DOI] [PubMed] [Google Scholar]
- 27.Tanev KS, Roether M, Yang C. Alcohol dementia and thermal dysregulation: a case report and review of the literature. Am J Alzheimers Dis Other Demen. 2008;23(6):563–70. doi: 10.1177/1533317508323479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Costantini A, et al. High-dose thiamine as initial treatment for Parkinson's disease. BMJ Case Rep. 2013;2013 doi: 10.1136/bcr-2013-009289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu'o'ng K, Nguyen LT. Role of thiamine in Alzheimer's disease. Am J Alzheimers Dis Other Demen. 2011;26(8):588–98. doi: 10.1177/1533317511432736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Costantini A, et al. Long-Term Treatment with High-Dose Thiamine in Parkinson Disease: An Open-Label Pilot Study. J Altern Complement Med. 2015;21(12):740–7. doi: 10.1089/acm.2014.0353. [DOI] [PubMed] [Google Scholar]
- 31.Karuppagounder SS, et al. Translocation of amyloid precursor protein C-terminal fragment(s) to the nucleus precedes neuronal death due to thiamine deficiency-induced mild impairment of oxidative metabolism. Neurochem Res. 2008;33(7):1365–72. doi: 10.1007/s11064-008-9594-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mouton-Liger F, et al. PKR downregulation prevents neurodegeneration and beta-amyloid production in a thiamine-deficient model. Cell Death Dis. 2015;6:e1594. doi: 10.1038/cddis.2014.552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hazell AS, et al. The impact of oxidative stress in thiamine deficiency: a multifactorial targeting issue. Neurochem Int. 2013;62(5):796–802. doi: 10.1016/j.neuint.2013.01.009. [DOI] [PubMed] [Google Scholar]
- 34.Ke ZJ, Bowen WM, Gibson GE. Peripheral inflammatory mechanisms modulate microglial activation in response to mild impairment of oxidative metabolism. Neurochem Int. 2006;49(5):548–56. doi: 10.1016/j.neuint.2006.04.011. [DOI] [PubMed] [Google Scholar]
- 35.Chen Z, Zhong C. Decoding Alzheimer's disease from perturbed cerebral glucose metabolism: implications for diagnostic and therapeutic strategies. Prog Neurobiol. 2013;108:21–43. doi: 10.1016/j.pneurobio.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 36.Doyle KM, et al. Unfolded proteins and endoplasmic reticulum stress in neurodegenerative disorders. J Cell Mol Med. 2011;15(10):2025–39. doi: 10.1111/j.1582-4934.2011.01374.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tanida I. Autophagosome formation and molecular mechanism of autophagy. Antioxid Redox Signal. 2011;14(11):2201–14. doi: 10.1089/ars.2010.3482. [DOI] [PubMed] [Google Scholar]
- 38.Gangolf M, et al. Thiamine status in humans and content of phosphorylated thiamine derivatives in biopsies and cultured cells. PLoS One. 2010;5(10):e13616. doi: 10.1371/journal.pone.0013616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thomson AD, Guerrini I, Marshall EJ. The evolution and treatment of Korsakoff's syndrome: out of sight, out of mind? Neuropsychol Rev. 2012;22(2):81–92. doi: 10.1007/s11065-012-9196-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Parkin AJ, et al. Neuropsychological sequelae of Wernicke's encephalopathy in a 20-year-old woman: selective impairment of a frontal memory system. Brain Cogn. 1993;21(1):1–19. doi: 10.1006/brcg.1993.1001. [DOI] [PubMed] [Google Scholar]
- 41.Sullivan EV, Fama R. Wernicke's encephalopathy and Korsakoff's syndrome revisited. Neuropsychol Rev. 2012;22(2):69–71. doi: 10.1007/s11065-012-9205-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jhala SS, Hazell AS. Modeling neurodegenerative disease pathophysiology in thiamine deficiency: consequences of impaired oxidative metabolism. Neurochem Int. 2011;58(3):248–60. doi: 10.1016/j.neuint.2010.11.019. [DOI] [PubMed] [Google Scholar]
- 43.McCormick LM, et al. Beyond alcoholism: Wernicke-Korsakoff syndrome in patients with psychiatric disorders. Cogn Behav Neurol. 2011;24(4):209–16. doi: 10.1097/WNN.0b013e31823f90c4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu M, et al. Thiamine deficiency induces anorexia by inhibiting hypothalamic AMPK. Neuroscience. 2014;267:102–13. doi: 10.1016/j.neuroscience.2014.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hohsfield LA, Humpel C. Intravenous infusion of monocytes isolated from 2-week-old mice enhances clearance of Beta-amyloid plaques in an Alzheimer mouse model. PLoS One. 2015;10(4):e0121930. doi: 10.1371/journal.pone.0121930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gross AL, et al. Cortical signatures of cognition and their relationship to Alzheimer's disease. Brain Imaging Behav. 2013;6(4):584–98. doi: 10.1007/s11682-012-9180-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rafii MS, Aisen PS. Advances in Alzheimer's disease drug development. BMC Med. 2015;13:62. doi: 10.1186/s12916-015-0297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wimo A, et al. The worldwide economic impact of dementia 2010. Alzheimers Dement. 2013;9(1):1–11. e3. doi: 10.1016/j.jalz.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 49.Gold M, Hauser RA, Chen MF. Plasma thiamine deficiency associated with Alzheimer's disease but not Parkinson's disease. Metab Brain Dis. 1998;13(1):43–53. doi: 10.1023/a:1020678912330. [DOI] [PubMed] [Google Scholar]
- 50.Furst AJ, et al. Cognition, glucose metabolism and amyloid burden in Alzheimer's disease. Neurobiol Aging. 2012;33(2):215–25. doi: 10.1016/j.neurobiolaging.2010.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mosconi L, Pupi A, De Leon MJ. Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer's disease. Ann N Y Acad Sci. 2008;1147:180–95. doi: 10.1196/annals.1427.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yao J, Brinton RD. Targeting mitochondrial bioenergetics for Alzheimer's prevention and treatment. Curr Pharm Des. 2011;17(31):3474–9. doi: 10.2174/138161211798072517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gibson GE, et al. Reduced activities of thiamine-dependent enzymes in the brains and peripheral tissues of patients with Alzheimer's disease. Arch Neurol. 1988;45(8):836–40. doi: 10.1001/archneur.1988.00520320022009. [DOI] [PubMed] [Google Scholar]
- 54.Gibson GE, Blass JP. Thiamine-dependent processes and treatment strategies in neurodegeneration. Antioxid Redox Signal. 2007;9(10):1605–19. doi: 10.1089/ars.2007.1766. [DOI] [PubMed] [Google Scholar]
- 55.Butterworth RF, Besnard AM. Thiamine-dependent enzyme changes in temporal cortex of patients with Alzheimer's disease. Metab Brain Dis. 1990;5(4):179–84. doi: 10.1007/BF00997071. [DOI] [PubMed] [Google Scholar]
- 56.Gibson GE, et al. Abnormal thiamine-dependent processes in Alzheimer's Disease. Lessons from diabetes. Mol Cell Neurosci. 2013;55:17–25. doi: 10.1016/j.mcn.2012.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Karuppagounder SS, et al. Thiamine deficiency induces oxidative stress and exacerbates the plaque pathology in Alzheimer's mouse model. Neurobiol Aging. 2009;30(10):1587–600. doi: 10.1016/j.neurobiolaging.2007.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang Q, et al. Thiamine deficiency increases beta-secretase activity and accumulation of beta-amyloid peptides. Neurobiol Aging. 2011;32(1):42–53. doi: 10.1016/j.neurobiolaging.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 59.Pan X, et al. Powerful beneficial effects of benfotiamine on cognitive impairment and beta-amyloid deposition in amyloid precursor protein/presenilin-1 transgenic mice. Brain. 2011;133(Pt 5):1342–51. doi: 10.1093/brain/awq069. [DOI] [PubMed] [Google Scholar]
- 60.Meador K, et al. Preliminary findings of high-dose thiamine in dementia of Alzheimer's type. J Geriatr Psychiatry Neurol. 1993;6(4):222–9. doi: 10.1177/089198879300600408. [DOI] [PubMed] [Google Scholar]
- 61.Blass JP, et al. Thiamine and Alzheimer's disease. A pilot study. Arch Neurol. 1988;45(8):833–5. doi: 10.1001/archneur.1988.00520320019008. [DOI] [PubMed] [Google Scholar]
- 62.Nolan KA, et al. A trial of thiamine in Alzheimer's disease. Arch Neurol. 1991;48(1):81–3. doi: 10.1001/archneur.1991.00530130093025. [DOI] [PubMed] [Google Scholar]
- 63.K LN, Nguyen L. Role of vitamin d in Parkinson's disease. ISRN Neurol. 2012;2012:134289. doi: 10.5402/2012/134289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.de Rijk MC, et al. Prevalence of parkinsonism and Parkinson's disease in Europe: the EUROPARKINSON Collaborative Study. European Community Concerted Action on the Epidemiology of Parkinson's disease. J Neurol Neurosurg Psychiatry. 1997;62(1):10–5. doi: 10.1136/jnnp.62.1.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Grenier K, Kontogiannea M, Fon EA. Short mitochondrial ARF triggers Parkin/PINK1-dependent mitophagy. J Biol Chem. 2014;289(43):29519–30. doi: 10.1074/jbc.M114.607150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jimenez-Jimenez FJ, et al. Cerebrospinal fluid levels of thiamine in patients with Parkinson's disease. Neurosci Lett. 1999;271(1):33–6. doi: 10.1016/s0304-3940(99)00515-7. [DOI] [PubMed] [Google Scholar]
- 67.Mizuno Y, et al. An immunohistochemical study on alpha-ketoglutarate dehydrogenase complex in Parkinson's disease. Ann Neurol. 1994;35(2):204–10. doi: 10.1002/ana.410350212. [DOI] [PubMed] [Google Scholar]
- 68.Gibson GE, Zhang H. Interactions of oxidative stress with thiamine homeostasis promote neurodegeneration. Neurochem Int. 2002;40(6):493–504. doi: 10.1016/s0197-0186(01)00120-6. [DOI] [PubMed] [Google Scholar]
- 69.Vetreno RP, Hall JM, Savage LM. Alcohol-related amnesia and dementia: animal models have revealed the contributions of different etiological factors on neuropathology, neurochemical dysfunction and cognitive impairment. Neurobiol Learn Mem. 2011;96(4):596–608. doi: 10.1016/j.nlm.2011.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Parsons OA, Nixon SJ. Neurobehavioral sequelae of alcoholism. Neurol Clin. 1993;11(1):205–18. [PubMed] [Google Scholar]
- 71.Pitel AL, et al. Signs of preclinical Wernicke's encephalopathy and thiamine levels as predictors of neuropsychological deficits in alcoholism without Korsakoff's syndrome. Neuropsychopharmacology. 2011;36(3):580–8. doi: 10.1038/npp.2010.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhang Q, et al. Senescence accelerated mouse strain is sensitive to neurodegeneration induced by mild impairment of oxidative metabolism. Brain Res. 2009;1264:111–8. doi: 10.1016/j.brainres.2009.02.005. [DOI] [PubMed] [Google Scholar]
- 73.Meng Y, et al. Autophagy alleviates neurodegeneration caused by mild impairment of oxidative metabolism. J Neurochem. 2013;126(6):805–18. doi: 10.1111/jnc.12268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fischer R, Maier O. Interrelation of oxidative stress and inflammation in neurodegenerative disease: role of TNF. Oxid Med Cell Longev. 2015;2015:610813. doi: 10.1155/2015/610813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chiurchiu V, Orlacchio A, Maccarrone M. Is Modulation of Oxidative Stress an Answer? The State of the Art of Redox Therapeutic Actions in Neurodegenerative Diseases. Oxid Med Cell Longev. 2015;2016:7909380. doi: 10.1155/2016/7909380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Radi E, et al. Apoptosis and oxidative stress in neurodegenerative diseases. J Alzheimers Dis. 2014;42(Suppl 3):S125–52. doi: 10.3233/JAD-132738. [DOI] [PubMed] [Google Scholar]
- 77.Cobb CA, Cole MP. Oxidative and nitrative stress in neurodegeneration. Neurobiol Dis. 2015;84:4–21. doi: 10.1016/j.nbd.2015.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Calingasan NY, et al. Induction of nitric oxide synthase and microglial responses precede selective cell death induced by chronic impairment of oxidative metabolism. Am J Pathol. 1998;153(2):599–610. doi: 10.1016/S0002-9440(10)65602-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Langlais PJ, et al. Increased cerebral free radical production during thiamine deficiency. Metab Brain Dis. 1997;12(2):137–43. [PubMed] [Google Scholar]
- 80.Calingasan NY, et al. Oxidative stress is associated with region-specific neuronal death during thiamine deficiency. J Neuropathol Exp Neurol. 1999;58(9):946–58. doi: 10.1097/00005072-199909000-00005. [DOI] [PubMed] [Google Scholar]
- 81.Sharma A, Bist R, Bubber P. Thiamine deficiency induces oxidative stress in brain mitochondria of Mus musculus. J Physiol Biochem. 2013;69(3):539–46. doi: 10.1007/s13105-013-0242-y. [DOI] [PubMed] [Google Scholar]
- 82.Yang G, et al. Neuronal MCP-1 mediates microglia recruitment and neurodegeneration induced by the mild impairment of oxidative metabolism. Brain Pathol. 2011;21(3):279–97. doi: 10.1111/j.1750-3639.2010.00445.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Leong DK, et al. Increased densities of binding sites for the "peripheral-type" benzodiazepine receptor ligand [3HJPK11195 in vulnerable regions of the rat brain in thiamine deficiency encephalopathy. J Cereb Blood Flow Metab. 1994;14(1):100–5. doi: 10.1038/jcbfm.1994.14. [DOI] [PubMed] [Google Scholar]
- 84.Terlecky SR, Terlecky LJ, Giordano CR. Peroxisomes, oxidative stress, and inflammation. World J Biol Chem. 2012;3(5):93–7. doi: 10.4331/wjbc.v3.i5.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wu Z, et al. Nutrients, Microglia Aging, and Brain Aging. Oxid Med Cell Longev. 2016;2016:7498528. doi: 10.1155/2016/7498528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Schroder M. Endoplasmic reticulum stress responses. Cell Mol Life Sci. 2008;65(6):862–94. doi: 10.1007/s00018-007-7383-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bravo R, et al. Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int Rev Cell Mol Biol. 2013;301:215–90. doi: 10.1016/B978-0-12-407704-1.00005-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ozcan L, Tabas I. Role of endoplasmic reticulum stress in metabolic disease and other disorders. Annu Rev Med. 2012;63:317–28. doi: 10.1146/annurev-med-043010-144749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Morishima N, et al. Translocation of Bim to the endoplasmic reticulum (ER) mediates ER stress signaling for activation of caspase-12 during ER stress-induced apoptosis. J Biol Chem. 2004;279(48):50375–81. doi: 10.1074/jbc.M408493200. [DOI] [PubMed] [Google Scholar]
- 90.Tabas I, Ron D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol. 2011;13(3):184–90. doi: 10.1038/ncb0311-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hetz C, et al. The unfolded protein response: integrating stress signals through the stress sensor IRE1alpha. Physiol Rev. 2011;91(4):1219–43. doi: 10.1152/physrev.00001.2011. [DOI] [PubMed] [Google Scholar]
- 92.Yang F, Luos J. Endoplasmic Reticulum Stress and Ethanol Neurotoxicity. Biomolecules. 2015;5(4):2538–53. doi: 10.3390/biom5042538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Perri ER, et al. The Unfolded Protein Response and the Role of Protein Disulfide Isomerase in Neurodegeneration. Front Cell Dev Biol. 2016;3:80. doi: 10.3389/fcell.2015.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Matus S, Glimcher LH, Hetz C. Protein folding stress in neurodegenerative diseases: a glimpse into the ER. Curr Opin Cell Biol. 2011;23(2):239–52. doi: 10.1016/j.ceb.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 95.Homma K, et al. Targeting ASK1 in ER stress-related neurodegenerative diseases. Expert Opin Ther Targets. 2009;13(6):653–64. doi: 10.1517/14728220902980249. [DOI] [PubMed] [Google Scholar]
- 96.Placido AI, et al. Modulation of endoplasmic reticulum stress: an opportunity to prevent neurodegeneration? CNS Neurol Disord Drug Targets. 2015;14(4):518–33. doi: 10.2174/1871527314666150429112353. [DOI] [PubMed] [Google Scholar]
- 97.Wang X, et al. Thiamine deficiency induces endoplasmic reticulum stress in neurons. Neuroscience. 2007;144(3):1045–56. doi: 10.1016/j.neuroscience.2006.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ke ZJ, et al. Ethanol promotes thiamine deficiency-induced neuronal death: involvement of double-strandedRNA-activatedprotein kinase. Alcohol Clin Exp Res. 2009;33(6):1097–103. doi: 10.1111/j.1530-0277.2009.00931.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Mota SI, et al. Oxidative stress involving changes in Nrf2 and ER stress in early stages of Alzheimer's disease. Biochim Biophys Acta. 2015;1852(7):1428–41. doi: 10.1016/j.bbadis.2015.03.015. [DOI] [PubMed] [Google Scholar]
- 100.Begum G, et al. ER stress and effects of DHA as an ER stress inhibitor. Transl Stroke Res. 2013;4(6):635–42. doi: 10.1007/s12975-013-0282-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lee S, et al. ADAR2-dependent RNA editing of GluR2 is involved in thiamine deficiency-induced alteration of calcium dynamics. Mol Neurodegener. 2010;5:54. doi: 10.1186/1750-1326-5-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell. 2004;6(4):463–77. doi: 10.1016/s1534-5807(04)00099-1. [DOI] [PubMed] [Google Scholar]
- 103.Mizushima N, Klionsky DJ. Protein turnover via autophagy: implications for metabolism. Annu Rev Nutr. 2007;27:19–40. doi: 10.1146/annurev.nutr.27.061406.093749. [DOI] [PubMed] [Google Scholar]
- 104.Jia G, Sowers JR. Autophagy: a housekeeper in cardiorenal metabolic health and disease. Biochim Biophys Acta. 2015;1852(2):219–24. doi: 10.1016/j.bbadis.2014.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Osna NA, Thomes PG, T.M. Involvement of autophagy in alcoholic liver injury and hepatitis Cpathogenesis. World J Gastroenterol. 2011;17(20):2507–14. doi: 10.3748/wjg.v17.i20.2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Mizushima N. Autophagy: process and function. Genes Dev. 2007;21(22):2861–73. doi: 10.1101/gad.1599207. [DOI] [PubMed] [Google Scholar]
- 107.Navone F, Genevini P, Borgese N. Autophagy and Neurodegeneration: Insights from a Cultured Cell Model of ALS. Cells. 2015;4(3):354–86. doi: 10.3390/cells4030354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Shibata M, et al. Regulation of intracellular accumulation of mutant Huntingtin by Beclin 1. J Biol Chem. 2006;281(20):14474–85. doi: 10.1074/jbc.M600364200. [DOI] [PubMed] [Google Scholar]
- 109.Nah J, Yuan J, Jung YK. Autophagy in neurodegenerative diseases: from mechanism to therapeutic approach. Mol Cells. 2015;38(5):381–9. doi: 10.14348/molcells.2015.0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Cherra SJ, 3rd, Chu CT. Autophagy in neuroprotection and neurodegeneration: A question of balance. Future Neurol. 2008;3(3):309–323. doi: 10.2217/14796708.3.3.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Luo J. Autophagy and ethanol neurotoxicity. Autophagy. 2014;10(12):2099–108. doi: 10.4161/15548627.2014.981916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhang K. Integration of ER stress, oxidative stress and the inflammatory response in health and disease. Int J Clin Exp Med. 2010;3(1):33–40. [PMC free article] [PubMed] [Google Scholar]
- 113.Chen G, et al. Autophagy is a protective response to ethanol neurotoxicity. Autophagy. 2012;8(11):1577–89. doi: 10.4161/auto.21376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Greenwood J, Love ER, Pratt OE. Kinetics of thiamine transport across the blood-brain barrier in the rat. J Physiol. 1982;327:95–103. doi: 10.1113/jphysiol.1982.sp014222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen G, Luo J. Anthocyanins: are they beneficial in treating ethanol neurotoxicity? Neurotox Res. 2010;17(1):91–101. doi: 10.1007/s12640-009-9083-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Rivas A, Vidal RL, Hetz C. Targeting the unfolded protein response for disease intervention. Expert Opin Ther Targets. 2015;19(9):1203–18. doi: 10.1517/14728222.2015.1053869. [DOI] [PubMed] [Google Scholar]
- 117.Scheper W, Hoozemans JJ. The unfolded protein response in neurodegenerative diseases: a neuropathologicalperspective. Acta Neuropathol. 2015;130(3):315–31. doi: 10.1007/s00401-015-1462-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Tan CC, et al. Autophagy in aging and neurodegenerative diseases: implications for pathogenesis and therapy. Neurobiol Aging. 2013;35(5):941–57. doi: 10.1016/j.neurobiolaging.2013.11.019. [DOI] [PubMed] [Google Scholar]
- 119.Sarkar S. Regulation of autophagy by mTOR-dependent and mTOR-independent pathways: autophagy dysfunction in neurodegenerative diseases and therapeutic application of autophagy enhancers. Biochem Soc Trans. 2013;41(5):1103–30. doi: 10.1042/BST20130134. [DOI] [PubMed] [Google Scholar]