Abstract
Missed lung cancer is a source of concern among radiologists and an important medicolegal challenge. In 90% of the cases, errors in diagnosis of lung cancer occur on chest radiographs. It may be challenging for radiologists to distinguish a lung lesion from bones, pulmonary vessels, mediastinal structures, and other complex anatomical structures on chest radiographs. Nevertheless, lung cancer can also be overlooked on computed tomography (CT) scans, regardless of the context, either if a clinical or radiologic suspect exists or for other reasons. Awareness of the possible causes of overlooking a pulmonary lesion can give radiologists a chance to reduce the occurrence of this eventuality. Various factors contribute to a misdiagnosis of lung cancer on chest radiographs and on CT, often very similar in nature to each other. Observer error is the most significant one and comprises scanning error, recognition error, decision-making error, and satisfaction of search. Tumor characteristics such as lesion size, conspicuity, and location are also crucial in this context. Even technical aspects can contribute to the probability of skipping lung cancer, including image quality and patient positioning and movement. Albeit it is hard to remove missed lung cancer completely, strategies to reduce observer error and methods to improve technique and automated detection may be valuable in reducing its likelihood.
Lung cancer accounts for 1.59 million deaths per year worldwide (1). It has one of the poorest survival outcomes of all cancers, with over two-thirds of patients diagnosed at an advanced stage, when curative treatment is no longer feasible. Early diagnosis of lung cancer is the main goal to improve survival. Patients with non-small cell lung cancer (NSCLC) at an operable stage have higher survival rates than those presenting with metastatic disease, with five-year survival of 71%–77% for stage IA and 58% for stage IB (2).
Initial identification of lung cancer in asymptomatic patients usually occurs on chest radiography or chest computed tomography (CT). When missed on imaging, lung cancer is inclined to progress from early-stage to advanced-stage disease, particularly if many years pass between radiologic exams (3), with potential medicolegal consequences.
Legal actions involving malignancies of the bronchus or lung represent the sixth most common medicolegal issue, and among radiologists it is the second most common cause for litigation (4). About 90% of presumed mistakes in pulmonary tumor diagnosis occurred on chest radiography, only 5% on CT examinations, and the remaining 5% on other imaging studies (4).
Awareness of the possible causes for overlooking a pulmonary lesion can help radiologists to reduce the occurrence of this eventuality. In this review, we analyze factors leading to a misdiagnosis of lung cancer mainly on chest radiography, and we discuss the impact of misdiagnosis on prognosis, its medicolegal implications, and methods to reduce the incidence of missed lung cancer. Finally, we briefly analyze the possible causes of errors on CT scans and potential aids.
Factors leading to missed lung cancer on chest radiography
Formerly, different authors recognized the burden of missed lung cancer on radiography of the thorax. Indeed, early studies on the analysis of factors leading to overlooked lung lesions date back to the middle of last century. Despite extensive technological advancement, this issue is currently present and not much has changed since then. Factors that contribute to missed lung cancer on chest X-ray can be classified as deriving from observer error, tumor characteristics, and technical considerations.
Observer error
Observer error is likely the biggest cause of misdiagnosis of lung cancer. It is not unusual to discover chest abnormalities at retrospective observation of previous radiologic exams, in people who are later diagnosed having pulmonary malignancy (5).
The perception of pulmonary lesions is dependent on the performance of the observer and it can be modified by the reader’s skills and external factors (6). Confounding factors are partially understood, even though a whole awareness of the nature of observer error has not been reached. Kundel et al. (7), analyzing the identification of lung nodules on chest radiography, classified observer errors in three categories: scanning error, recognition error, and decision-making error.
Scanning error accounts for about 30% of missed lung lesions and occurs predominantly when the nodule is not fixated on the fovea during image analysis. The human field of view covers an average angle of 180°, but only the central part of this angle provides sharp details and covers a 1° angle that corresponds to a circle sized 2.5 cm in diameter, when viewing an image at a distance of 40 cm (8). Furthermore, the foveal fixation over a point of interest lasts for maximum 200–300 milliseconds. After that, eyes lose their sensitivity and move to a new location. More fixations are necessary to center the fovea on a target (9). An average of 300 fixations are required to cover an image area of 14×17 cm with the fovea, while in medical practice only 80–120 fixations are used in the average of 20–30 seconds spent on a chest radiogram, meaning that large parts of a chest X-ray are often not scanned with fovea.
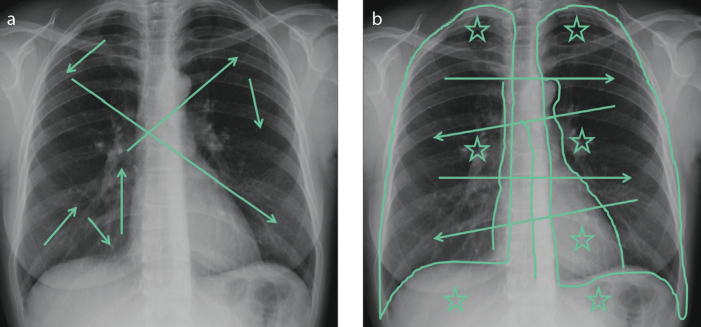
Experience of the reader also plays a role in this context. Expert observers develop their own specific scanning pattern; instead, a resident or untrained radiologist generally searches randomly, “jumping around” on the radiogram (Fig. 1). The development of an effective scan path is dependent on the level of consciousness acquired on the typical appearance of normal and pathologic findings of the chest (10).
Figure 1. a, b.
Frontal view chest X-rays showing examples of different scanning pathways. A messy scanning pattern (a) in which the reader’s gaze jumps to different areas of the lungs without any method (arrows). An ordered scan path (b), which covers all the lung zones symmetrically (arrows). The “blind zones” (apices, hila, retro-cardiac and sub-diaphragmatic spaces) (stars) and the mediastinal lines and stripes (lines) should always be checked carefully.
The recognition error, accounting for about 25% of missed lesions, consists of missed detection of lesions adequately scanned by the observer. An important element of this error is the observer’s mind-set (6). Many factors can influence the vigilance and the attitude of the radiologist in searching for lung nodules, such as work conditions and human factors like fatigue, bias and suspect index, but also patient age, presence of other abnormalities, and clinical history. The availability of clinical data improves the overall diagnostic accuracy. This is related to the concept of vigilance and the “state of alert” regarding the search for pulmonary nodules, which is higher in high risk patients than in low risk ones.
The decision-making error is responsible for 45% of errors. This mistake is due to the inaccurate interpretation of an identified anomaly to be normal (Fig. 2). One of the most important parameters influencing this kind of error is the observer’s experience (10).
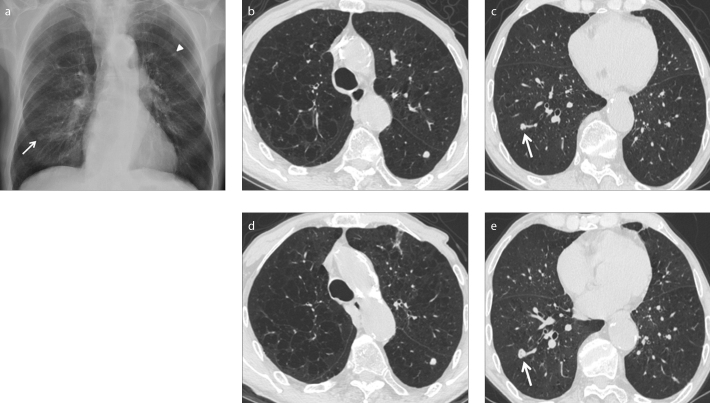
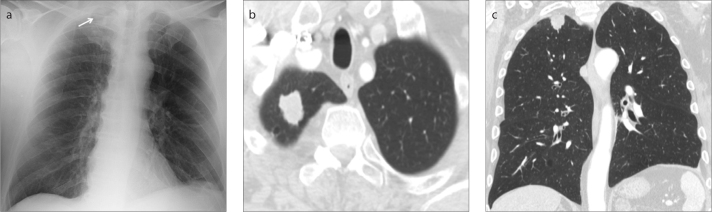
Figure 2. a–e.
Missed lung cancer in an 85-year-old patient with severe emphysema. Routine postero-anterior (PA) chest X-ray (a) shows a nodular opacity projecting upon the posterior arch of the left sixth rib (arrowhead). Another nodular opacity in the right basal region (arrow) was not reported, probably because interpreted as a nipple (decision-making error) or because of satisfaction of search. CT exam performed immediately after the chest radiograph (b, c), confirms the presence of a nodule with regular margins in the apical segment of the left lower lobe (b). A nodule with lobulated margins is also visible in the right lower lobe (c, arrow) corresponding to the nodular opacity missed on chest radiograph. CT exam performed six months later (d, e) for nodule follow-up, shows the growth of the right lower lobe nodule (arrow), suggesting malignancy, later confirmed at biopsy. The diameter of the nodule in the left lower lobe is unchanged.
Another potential observer error is related to the mechanism of “satisfaction of search”, a phenomenon consisting of “loss of interest” by radiologists after the identification of an abnormality and consequent interference with the search process and diagnosis of other lung abnormalities (Fig. 2). In lung cancer it is possible to find more than one abnormality, such as pleural effusion, thickness of the interlobular septa and lobar collapse, and the risk of skipping over a small nodule may not be irrelevant. It has been demonstrated that this phenomenon is due to two possible mechanisms: ceasing the search for other abnormalities early in a positive exam and focusing on the “wrong” part of the exam (11).
Lesion characteristics
Tumor characteristics concur significantly to the probability of identifying pulmonary lesions. Dimension, conspicuity, and location are the most important features to consider.
Size affects the identification of the lesions mainly on chest X-ray. Quekel et al. (12) reported a detection rate of 29% in tumors with a diameter ≤10 mm. For lesions measuring from 10 to 30 mm, the rate of error was 28%, from 30 to 40 mm 12%, while lesions >40 mm were not missed (12). However, in the literature the average size of missed carcinomas is quite variable and in most cases, it is more than 10 mm, specifically, 16 mm in Austin et al. (13), 25 mm in Monnier-Cholley et al. (14), and 18.1±7.7 mm in the experience of Wu et al. (15). Therefore, other than size, tumor features influence its identification.
Conspicuity is another important characteristic in determining the detection of pulmonary lesions (6). It is described as the ratio between lesion contrast and surrounding structural complexity and it is highly affected by tumor margin visibility as well as density. Many authors found that the majority of missed lesions in their series did not have sharp borders (12, 13). In the experience of Wu et al. (15), the readers qualified the opacity level of lesions detected at chest X-ray; subsequently they employed CT characteristics to describe lesion borders and density. The larger part of tumors missed on chest X-ray showed ill-defined borders and/or ground-glass density at CT examination, and among the ground-glass lesions, about 50% had attenuation values <-100 HU.
Even tumor histology may influence its contrast, attenuation and margin features (16). On the basis of histopathologic confirmation, Sone et al. (17) asserted that small adenocarcinomas showing lepidic growing pattern were less conspicuous on X-ray than similarly sized pulmonary cancer with infiltrative growing pattern.
Tumor detectability rate is also strongly influenced by its location. In the experience of Austin et al. (13), 81% of missed lung cancers located in the upper lobes, particularly on the right (56%). Other authors also registered a striking prevalence of skipped malignancy for the upper lobes, with a rate ranging from 45% to 66% (12, 14, 15, 18). The upper lobe predominance of missed lung cancer could be at least partially explained by the higher frequency of lung cancer occurrence in the upper lobes (19, 20). Nevertheless, other studies suggested that overlying anatomic structures of the thorax are far more important for missing lesions than the location (24). Anatomical structures such as ribs, lung vessels, heart, mediastinum, and diaphragm overlay each other in a pattern that can be referred to as anatomic noise (Fig. 3). A lesion is less likely to be perceived on radiography if the image contains a great deal of anatomic noise, as in some blinded areas of the chest (apices, hilar regions, and retrocardiac space).
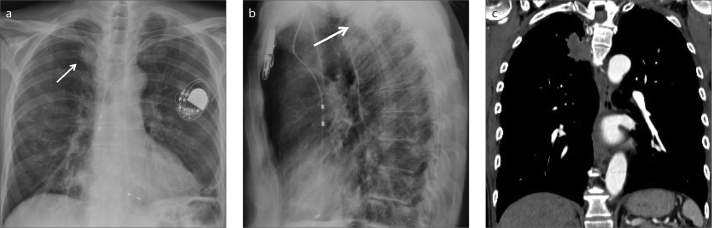
Figure 3. a–c.
Missed lung cancer in a 75-year-old man who underwent chest X-ray after pacemaker positioning. A left iatrogenic pneumothorax was reported (a). An oval opacity in the posterior segment of the right upper lobe (arrows) was not identified by the radiologist (a, b). In the PA projection (a) the opacity was missed because of the superimposition with the fifth posterior rib arch crossing the first costo-sternal joint. In the lateral projection (b), the opacity was hidden by the upper thoracic vertebral bodies. CT performed a few days later (c) to rule out a pulmonary embolism, clearly depicted a soft tissue mass in the right upper lobe adjacent to the posterior mediastinal pleura.
The predominance of overlooked pulmonary tumors in the apical zones was reported to be 72% in the study of Shah et al. (3), in which missed lung cancers were mainly localized in the apical or posterior segment (60%).
Visualization of hilar lesions is hindered by the huge variability of the hilar anatomy, as clearly demonstrated on Fig. 4, and imposes a perfect quality of the radiographs. Muhm et al. (22) showed that 65% of the pulmonary lesions originating in the hila were overlooked in a screening program; only cancers measuring >3 cm were detected in that zone. In another study by Quekel et al. (12), missed centrally located tumors were larger than the peripheral ones, meaning that superimposing structures are the main cause for overlooking a central lesion. Cardiac structures may obscure cancers localized in the lower lobes, as demonstrated by Wu et al. (15), who reported a rate of 92% of pulmonary tumors located in the lower lobes missed by the “non-thoracic” radiologists.
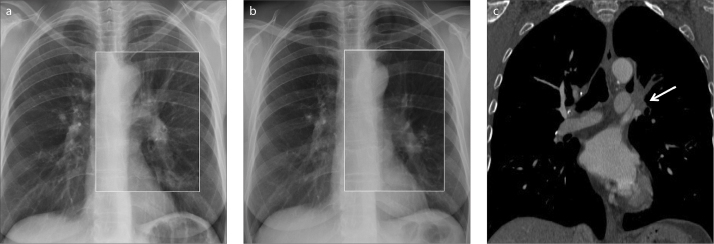
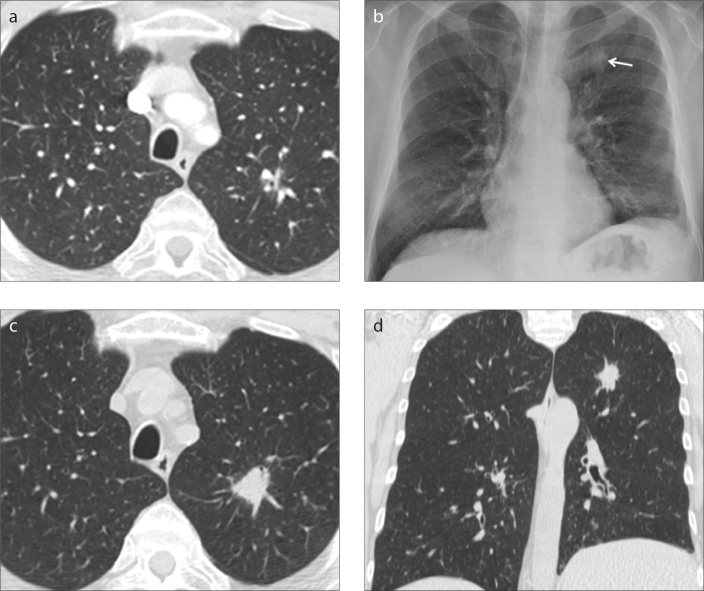
Figure 4. a–c.
Missed lung cancer in a 54-year-old heavy smoker patient with mild cough. PA chest X-ray (a) shows a slight left hilar increase in dimension and opacity with respect to the right hilum (square box), which was interpreted as negative. Comparison with a chest radiograph performed one year before (b) was of paramount importance to detect the subtle anatomic change at a second review of the images (square box). Coronal chest CT (c) at the mediastinum window setting documented solid tissue (arrow) in the left hilum occluding the apico-dorsal and anterior segmental bronchi of the left upper lobe, with subsequent segmental atelectasis and lobar volume loss.
In these cases, the lateral projection may be helpful. On a normal lateral chest radiograph, the attenuation decreases as one progresses down the thoracic vertebral bodies. If the attenuation increases, locally or diffusely, there must be a posteriorly located lesion. This is referred to as “positive spine sign”, as reported by Medjek et al. (23), who observed a good positive predictive value for this sign in detecting lower abnormalities on lateral chest X-ray. This lesion might not be seen on the frontal view, hidden by the heart or the hila.
These results reinforce the value of a solid knowledge of normal chest anatomy and its variations, particularly of certain complex areas of the thorax, and corroborate the assumption of using previous radiologic exams for comparison with current X-ray in order to detect subtle anatomic changes (Fig. 4).
Technical considerations
Image quality and patient positioning and movement are technical factors determining the probability of overlooking pulmonary malignancy (6).
Standard chest radiography is usually acquired in the postero-anterior (PA) and latero-lateral (LL) projections, with the patient standing up. Various thoughts exist on the need to perform both PA and LL X-ray. The relevance of the lateral projection has been highlighted by Chotas et al. (24), who found that frontal view represent only 73.6% of the total lung volume and 57% of the total lung area that is not obscured by the superimposed anatomical structures such as thoracic spine, mediastinum, heart, diaphragm, and blood vessels. The lateral projection may help to reduce this anatomic noise. Tala et al. (25) reported that the lateral chest X-ray allowed a confident diagnosis of lung carcinoma in 20% of patients undergoing both PA and LL projections. In the study by Wu et al. (15), thoracic radiologists found that lateral radiography was of higher quality compared with PA radiography for detectability of pulmonary lesions in 8 out of 30 patients (27%) and one nodule could be identified only on lateral projection. Other authors reported a rate of 2% to 4% of lung cancers seen only on the LL projection (13, 22, 26). On the other hand, Forrest et al. (27) and Sagel et al. (28) observed that all lung cancers were visible on frontal X-ray, suggesting that lateral radiography was unnecessary.
The LL projection remains a significant tool for lung cancer detection. The utility of an additional view should be weighed against the additional cost and radiation exposure and may depend on the clinical need.
The antero-posterior (AP) radiograph performed at patient’s bedside can be a major factor contributing to missing chest lesions (Fig. 5) (29), due to its intrinsic lower quality with respect to standard X-ray.
Figure 5. a–c.
Missed lung cancer with apical location in a bedside chest X-ray performed in a 66-year-old patient with brain metastasis. Antero-posterior (AP) radiograph (a), performed in the supine position, was reported as negative by the radiologist. The faint opacity in the right lung apex (white arrow) was hidden by the overlapping dense structures, such as ribs and right clavicle. CT exam later performed for staging purpose shows on axial (b) and coronal (c) images, a 2 cm nodular solid lesion with irregular margins in the apical segment of the right upper lobe, diagnosed as primary lung cancer.
In the past few years, extensive technological progress began the era of digital imaging. The most remarkable advancement has been the fast conversion from film-based to digital X-ray systems. Although digitized images do not contain any more information, they can be superior to film-based X-ray in the identification of lung cancer in underexposed regions of the PA view, such as the retrocardiac and apical areas, because digital radiography has the inherent capability of improving contrast through image processing (30). Digital systems have also improved the overall image quality of bedside chest X-ray and reduced the rate of exam repetition.
Impact of lung cancer misdiagnosis on prognosis and medicolegal implications
Influence of stage at diagnosis
Tumor growth and delay of the diagnosis influence prognosis. Of course, this length of time constitutes a wide possibility for an eventual radically treatable early-stage cancer to develop into a higher stage malignancy.
A recent publication on T stage revision proposals for the forthcoming 8th Edition of the TNM lung cancer classification, argued that from 1 to 5 cm, each cm separates lesions of significantly different prognosis (31). However, a missed lung cancer at X-ray does not automatically mean that the patient’s outcome will suffer, as affirmed by several studies.
There exists a big variability in the interval between the actual identification of pulmonary tumor and its first retrospective appearance on the chest radiographs, ranging from 0.2 to 48 months (13, 14, 22, 32). A number of retrospective studies discussed whether a missed diagnosis of pulmonary cancer at X-ray affects the patient outcome. In the study by Quekel et al. (12), the mean delay in diagnosis was 472 days. In 57% of patients included, the stage remained the same despite delay in the diagnosis, while T stage raised from T1 to T2 in the remaining 43%. In the study by Wu et al. (15), nearly 77% of pulmonary cancers missed by “non-thoracic” radiologists and 86% of tumors overlooked by thoracic radiologists were still in a radically treatable stage.
Turkington et al. (5) reviewed previous chest X-rays of 28 patients with lung cancer and, among them, 14 were recognized to be positive. No significant difference was found between the median survival of patients with no abnormality at previous chest radiography compared with those with a previously positive radiography (260 vs. 228 days; P = 0.7). On the other hand, a significantly higher median length of time from first positive radiograph to the time of starting treatment was found in patients with overlooked lesions than in those without preceding abnormalities (155 vs. 51 days; P = 0.001).
Kashiwabara et al. (33) enrolled 143 asymptomatic patients with lung cancer detected during an eight-year period of screening performed by X-ray, who had undergone chest X-ray one year before the disease was found. The authors divided patients into two groups: patients with tumor that could not be detected on previous radiography and patients with tumor that could be seen on a previous chest exam but had not been detected. No significant differences were reported between the five-year survival rates between the two groups. However, according to the tumor size of the overlooked lesions at chest X-ray, the outcome in stage I–II patients with missed tumors measuring >20 mm was worse than those with missed tumors ≤20 mm (P = 0.0047), demonstrating that one-year delayed detection of lung cancer ≤20 mm will not affect the prognosis.
Medicolegal implications
Missed diagnosis is one of the leading causes of malpractice actions against radiologists. Diagnostic errors in lung cancer detection are the second cause of legal litigation after malignant neoplasm of the breast (4), and among radiologists specialized on thorax imaging, failure to diagnose lung cancer is by far the most common reason for initiating a malpractice suit (42.5% of cases) (34).
On chest radiography, missed detection, identification, or description of a lung lesion that is subsequently shown to be a cancer could cause severe consequences in medical malpractice litigations. Fortunately it does not always result in litigation, particularly when it is not a cause of patient’s injury.
Analyzing the possible causes of missed lung cancer, we can affirm that radiographic diagnosis of lung cancer is not always achievable and even the most prudent and responsible radiologist cannot eliminate all possible sources of error.
It is essential to define when there is malpractice in missing a lung cancer on chest radiographs, proving elements of negligence. Negligence is demonstrated if standard of care is breached, causing remarkable damage to the patient. Since there is no rigorous definition of standard of care in the diagnosis of pulmonary malignancy, the last judgment often depends on the capacity of the experts of the two parties to persuade the magistrate and the jury that they are right. In this context, if a lesion is easily detectable it must be detected. However, errors in detection and recognition are often due to the diagnostic method used rather than medical malpractice.
Methods to reduce the incidence of missed lung cancer on chest radiography
Methods to potentially decrease the incidence of overlooked pulmonary malignancy at radiography of the thorax include strategies to reduce observer error and improve techniques.
Reducing observer error
A possible strategy for reducing observer error requires optimization of perception and interpretation. Optimal viewing state, comprising scarce surrounding illumination, excellent condition of the screen, modifying viewing distances, employment of a reducing lens and quiet conditions, are of great importance (35). Failure to recognize a lesion can never be completely avoided, although previous exams as well as pertinent anamnestic data can alert the physician and should always be available. It has also been shown that training the radiologists in chest reading methodology (Fig. 1b) and in chest diseases reduces observer error (26). A short checklist of steps that can be of help in reducing the observer errors when reading a chest X-ray is proposed in the Table.
Table.
Short checklist of steps for reducing observer errors in lung cancer identification on chest radiography
| CHECKLIST |
|---|
| Spend necessary time (more than 30 seconds) to carefully look at a chest X-ray, ensuring that every part of the radiogram has been scrutinized |
| Do not “jump around” on the chest radiogram (example in Fig. 1a), but develop a methodologic and symmetrical scan path in order not to miss any zones (example in Fig. 1b) |
| Always check the blind zones (apices, hila, retro-cardiac and sub-diaphragmatic spaces) and the mediastinal lines and stripes (Fig. 1b) |
| Use lateral projection if it will be of any help |
| Use the inverse-intensity image as an additional tool to increase confidence in identifying lesions |
| Compare with previous chest X-ray |
| Use patients’ clinical history to improve your accuracy and maintain a constant state of alert |
Recently, Howarth and Tack (36) suggested that in cases of missed lung lesion on chest radiograph, a side-by-side comparison of chest X-ray with CT scan may allow the radiologist to understand the reasons for missing lesions. By adopting an inquisitive approach, errors could be reduced.
Improving techniques
Regarding technical considerations, radiographies of thorax should use a wide-latitude screen-film system, anti-scattered grids and low-contrast parameters (130–140 kVp) to achieve a uniform film exposure (6). Images should be acquired with the patient standing up and with optimal inspiration, usually in two projections. X-ray must be checked habitually directly after accomplishment to evaluate consistency to technical parameters and quality. It should be repeated if needed (37). Sometimes additional views can be used, such as oblique projections or lordotic view to visualize the lung apices.
In the 1980s, the arrival of digital systems allowed the development of novel tools to ameliorate the identification of faint nodules; lung lesions became better detectable using image manipulation, which became possible with digitized X-ray. By comparing the ability to identify pulmonary nodules on conventional plain chest radiographs, standard (bones white) digitized images and inverse-intensity (bones black) images, Sheline et al. (38) found that inverse-intensity image can have some benefits in lesion recognition.
New algorithms may increase lesion conspicuity using image subtraction strategies. Dual-energy subtraction imaging is employed to produce illustrations of two different tissues, such as calcified structures and soft tissue, by using the known energy dependence of X-ray attenuation of both materials. Once two images are generated, it is possible to remove the overlying anatomic noise from the bones; consequently lung lesion conspicuity results are improved (39).
An additional system to facilitate the visualization of new faint opacities is the temporal subtraction imaging, which selectively highlights areas of interval change by subtracting the patient’s previous radiograph from the current one (40) thus improving the visual perception of subtle new abnormalities (41).
Recently, an advance technological system has been developed aiming to detect lung nodules that might be overlooked by the reader on X-ray: computer aided detection (CAD) system. The use of CAD as a second reader has been demonstrated to improve sensitivity of chest radiography. Detection accuracy is higher when CAD is used (42), particularly for less experienced readers (43). White et al. (44) studied the CAD’s capacity to identify a pulmonary nodule that has been skipped at first human reading. Each radiograph was analyzed by a commercial CAD system, which detected roughly 50% of the nodules missed by the radiologists.
Another way to increase recognition of small pulmonary nodules is the use of section imaging. While it does not have the resolution of CT, digital tomosynthesis represents an evolution of the conventional tomography at lower cost and radiation dose, allowing the production of several section images at different depths from a single set of acquisition. This method of processing images increases the visualization of anatomical structures such as vessels, airway and spine compared with the traditional radiograph, enabling better visibility of small parenchymal abnormalities. Two large clinical trials demonstrated that sensitivity for pulmonary lesion recognition is superior when tomosynthesis is used instead of traditional X-ray (45).
Missed lung cancer on CT
Chest CT is much more accurate than conventional radiography in the identification of pulmonary abnormalities because anatomical structures are depicted in a two-dimensional picture, without superimposition, and because of its greater contrast and spatial resolution. The probability of having a missed lung cancer on CT is therefore much lower than on chest X-ray. Nevertheless, a pulmonary tumor may be missed on CT scans, regardless of the context, whether a clinical or radiologic suspect exists or not. Various factors contribute to a misdiagnosis of lung cancer at CT, often very similar in nature to those occurring on chest radiography (46).
Factors leading to missed lung cancer on chest CT
In 1996, two papers focused on the characteristics of pulmonary lesions missed at CT have been published. White et al. (47) described location, shape and cell type of 15 lung cancers overlooked at CT and observed that the majority of these cancers had endobronchial location with lower lobe predominance. Gurney et al. (48) documented nine lung tumors missed at CT, five of which were peripheral and four were central. Davis et al. (49) analyzed these two studies, underlining the remarkable percentage of high-risk patients in these populations and identifying three possible causes of error on CT, similar to those previously described for chest X-rays, as related to lesion characteristics, observer performance, and technique.
The most frequent lesion characteristics that led to miss lung cancers were small dimension and poor conspicuity (Fig. 6), ill-defined margins and central location. Regarding observer performance, diagnostic failures may be due to decision-making error, as in the case of inaccurate interpretation of the characteristics of a lung cancer as a benign process, and to recognition error (46). The mechanism of “satisfaction of search” could be another source of observer’s false negative result, also in CT scans (49, 50). Attention to technical parameters, such as thin-slice thickness (≤ 1.25 mm), is an achievable way to avoid errors (49). More than one factor may contribute to a misdiagnosis simultaneously.
Figure 6. a–d.
Missed lung cancer on CT in a 63-year-old patient with hepatic cirrhosis hospitalized for ascites and complaining of cough and dyspnea. CT axial image (a) at lung window setting shows a small and poorly conspicuous lesion close to sub-segmental vessels of the apico-dorsal segment of the left upper lobe, that was not identified by the radiologist at the time of the exam, probably for its characteristics and location. Routine PA chest X-ray (b), performed two years later, shows a nodular opacity with irregular margins in the left apical region (arrow), identified by the radiologist. Axial (c) and coronal (d) CT images confirmed the presence of a 2 cm solid and not calcified nodule, with spiculated margins, located in the apico-dorsal segment of the left upper lobe, increased in diameter with respect to the previous CT exam and diagnosed as lung cancer.
After these first reports, other authors dealt with the issue of missed lung cancer on CT, especially because technological progresses led to the diffusion of lung cancer screening programs using CT as diagnostic tool, long before the publication of the National Lung Screening Trial’s results in 2011 (51), which currently represents the biggest study on this topic.
Kakinuma et al. (52) analyzed the data of 1443 subjects screened for lung cancer; 22 tumors were recognized and seven of them were visible on the images from an earlier spiral CT examination. Small diameter (<7 mm), juxtavascular location, and presence of confounding factors such as tuberculosis-related alterations were the causes of failed diagnosis.
In another study, Li et al. (53) concluded that low-dose CT generally fails in the identification of small faint tumors mainly located in the context of other lung disease or superimposed to normal anatomical structures.
More recently, the possible causes of missed lung tumors in the NELSON screening program have been analyzed (54). Causes of failure in detecting pulmonary tumors were human error, interpretation error, and detection error, owing to lesions’ location (endobronchial, iuxtapleural, adjacent to bullous structure or within extensive areas of honeycombing).
Veronesi et al. (55) described the causes of false negatives in the COSMOS lung cancer screening study. Centrally located or endobronchial lesions were disproportionately represented in missed lung cancers.
By analyzing existing studies, Deveraj (56) concluded that many causes exist for missed lung cancers in CT screening trials; however, certain anatomic chest locations such as endobronchial, hilar, and mediastinal are blind spots for radiologists on screening CT scans.
Moreover, it is worth to note that the development of multislice CT technology has increased the probability of detecting even very small nodules, particularly in the context of screening of smoker patients who may have a significant number of them. However, the high number of images produced by multidetector CT scanners has an impact on the workload of radiologists, which can lead to overlook of a lesion or misinterpretation of the findings (inability to properly diagnose a tumor) (57).
Malpractice cases regarding missing pulmonary tumor on CT of the thorax have been reported (48, 58). Most cases were related to recognition error. A remarkable case involved a lawsuit in a patient with advanced lung cancer that had been overlooked on a previous lung cancer screening CT, when the lesion measured 8 mm. A settlement of $1,000,000 was made, of which the radiologist assumed 90% (59).
Methods to reduce the incidence of missed lung cancer on chest CT
Modern technologies let radiologists detect focal lung lesions more efficiently, using appropriate postprocessing advances, such as off-axial reformatting, maximum intensity projection, and volumetric imaging. The majority of these systems allow a more confident recognition of the parenchymal lesions from the adjacent normal structures like vessels (60).
CAD is another helpful method for the detection of small lung lesions on CT scans and can assist the reader in the identification of early-stage pulmonary cancer. Armato et al. (61) used a database of 38 low-dose CT scans with 50 lung nodules, 38 of which were biopsy-proved lung cancer, to estimate the success of a completely automated system to recognize focal pulmonary lesions. Using this method, 80% of nodules were identified and 84% of missed lung cancers were correctly identified. Using a different CAD system, Li et al. (62) analyzed a group of 27 patients, 17 of them with a missed peripheral lung cancer. The adopted CAD scheme improved radiologists’ performance and, in particular, sensitivity in detection of missed lung cancers raised from 52% to 71%.
Although CAD software shows high false-positivity, which represents a major limit in wider use of the system, in recent years the number of false-positive nodules decreased thanks to advances in CAD technology. However, more developments in CAD algorithms are necessary to further reduce false-positivity (62).
Several studies have also demonstrated that CAD as second reader significantly increases sensitivity in the identification of lung nodules on both standard dose (63) and low dose (64) CT scans, particularly when using CAD detection system in combination with thin-slice (1.25 mm) CT scans compared with medium-slice (2.5 mm) scans (65).
Overlooked lung cancer on CT is challenging and clinically important, occurring both in clinical and screening settings. Failures are ascribed to missing identification or to misinterpretation of the lesion as a normal structure or a benign finding. Technical advances are useful in the detection of pulmonary nodules. CAD is a promising aid, improving radiologists’ performance in recognition of even very small tumors.
Conclusion
Missed lung cancer has potentially serious medicolegal implications for radiologists, despite the widespread awareness of this issue. The reasons for a misdiagnosis on chest radiography, and less frequently on CT scans, are many but frequently related to observer error (scanning, recognition, and decision-making error), specific characteristics of the undetected lesion (size, conspicuity, and location), or technical inaccuracies. Possible methods to decrease the incidence of missed lung cancer include strategies to reduce observer error, ameliorating lesion detection and interpretation, as well as methods to improve technique and automated detection.
Main points.
Approximately 90% of missed lung cancer cases occur on chest X-ray.
Although CT is much more sensitive than chest radiography, lung cancer may still be missed.
Observer error, lesion characteristics, and technical defects are the main causes of missed lung cancer.
Optimization of observer perception and skills in interpreting images, as well as advances in technology may help reduce diagnostic errors.
Acknowledgements
The authors thank Lucio Calandriello, MD, Alessandra Farchione, MD, and Fabio Maggi, MD, for their contribution.
Footnotes
Conflict of interest disclosure
The authors declared no conflicts of interest.
References
- 1.World Health Organization. Cancer. Fact Sheet Number 297. [Accessed July 10, 2015]. Available at: www.who.int/mediacentre/factsheets/fs297/en/Last updated July 2015.
- 2.Groome PA, Bolejack V, Crowley JJ, et al. The IASLC Lung Cancer Staging Project: validation of the proposals for revision of the T, N, and M descriptors and consequent stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J Thorac Oncol. 2007;2:694–705. doi: 10.1097/JTO.0b013e31812d05d5. https://doi.org/10.1097/JTO.0b013e31812d05d5. [DOI] [PubMed] [Google Scholar]
- 3.Shah PK, Austin JH, White CS, et al. Missed non-small cell lung cancer: radiographic findings of potentially resectable lesions evident only in retrospect. Radiology. 2003;226:235–241. doi: 10.1148/radiol.2261011924. https://doi.org/10.1148/radiol.2261011924. [DOI] [PubMed] [Google Scholar]
- 4.White CS, Salis AI, Meyer CA. Missed lung cancer on chest radiography and computed tomography: imaging and medicolegal issue. J Thorac Imaging. 1999;14:63–68. doi: 10.1097/00005382-199901000-00006. https://doi.org/10.1097/00005382-199901000-00006. [DOI] [PubMed] [Google Scholar]
- 5.Turkington PM, Kennan N, Greenstone MA. Misinterpretation of the chest x ray as a factor in delayed diagnosis of lung cancer. Postgrad Med J. 2002;78:158–160. doi: 10.1136/pmj.78.917.158. https://doi.org/10.1136/pmj.78.917.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brogdon BG, Kelsey CA, Moseley RD. Factors affecting perception of pulmonary lesions. Radiol Clin North Am. 1983;21:633–654. [PubMed] [Google Scholar]
- 7.Kundel HL, Nodine CF, Carmody D. Visual scanning, pattern recognition, and decision making in pulmonary nodule detection. Invest Radiol. 1978;13:175–181. doi: 10.1097/00004424-197805000-00001. https://doi.org/10.1097/00004424-197805000-00001. [DOI] [PubMed] [Google Scholar]
- 8.Manning D, Barker-Mill SC, Donovan T, Crawford T. Time-dependent observer errors in pulmonary nodule detection. Br J Radiol. 2006;79:342–346. doi: 10.1259/bjr/13453920. https://doi.org/10.1259/bjr/13453920. [DOI] [PubMed] [Google Scholar]
- 9.Manning DJ, Ethell SC, Donovan T. Detection or decision errors? Missed lung cancer from the postero-anterior chest radiograph. Br J Radiol. 2004;77:231–235. doi: 10.1259/bjr/28883951. https://doi.org/10.1259/bjr/28883951. [DOI] [PubMed] [Google Scholar]
- 10.Kundel HL, La Follette PS. Visual search patterns and experience with radiological images. Radiology. 1972;103:523–528. doi: 10.1148/103.3.523. https://doi.org/10.1148/103.3.523. [DOI] [PubMed] [Google Scholar]
- 11.Samuel S, Kundel HL, Nodine CF, Toto LC. Mechanism of satisfaction of search: eye position recordings in the reading of chest radiographs. Radiology. 1995;194:895–902. doi: 10.1148/radiology.194.3.7862998. https://doi.org/10.1148/radiology.194.3.7862998. [DOI] [PubMed] [Google Scholar]
- 12.Quekel LG, Kessels AG, Goei R, van Engelshoven JM. Miss rate of lung cancer on the chest radiograph in clinical practice. Chest. 1999;115:720–724. doi: 10.1378/chest.115.3.720. https://doi.org/10.1378/chest.115.3.720. [DOI] [PubMed] [Google Scholar]
- 13.Austin JH, Romney BM, Goldsmith LS. Missed bronchogenic carcinoma: radiographic findings in 27 patients with a potentially resectable lesion evident in retrospect. Radiology. 1992;182:115–122. doi: 10.1148/radiology.182.1.1727272. https://doi.org/10.1148/radiology.182.1.1727272. [DOI] [PubMed] [Google Scholar]
- 14.Monnier-Cholley L, Arrivè L, Porcel A, et al. Characteristics of missed lung cancer on chest radiographs: a French experience. Eur Radiol. 2001;11:597–605. doi: 10.1007/s003300000595. https://doi.org/10.1007/s003300000595. [DOI] [PubMed] [Google Scholar]
- 15.Wu MH, Gotway MB, Lee TJ, et al. Features of non-small cell lung carcinomas overlooked at digital chest radiography. Clin Radiol. 2008;63:518–528. doi: 10.1016/j.crad.2007.09.011. https://doi.org/10.1016/j.crad.2007.09.011. [DOI] [PubMed] [Google Scholar]
- 16.Yang ZG, Sone S, Li F, et al. Visibility of small peripheral lung cancers on chest radiographs: influence of densitometric parameters, CT values and tumour type. Br J Radiol. 2001;74:32–41. doi: 10.1259/bjr.74.877.740032. https://doi.org/10.1259/bjr.74.877.740032. [DOI] [PubMed] [Google Scholar]
- 17.Sone S, Li F, Yang ZG, et al. Characteristics of small lung cancers invisible on conventional chest radiography and detected by population based screening using spiral CT. Br J Radiol. 2000;73:137–145. doi: 10.1259/bjr.73.866.10884725. https://doi.org/10.1259/bjr.73.866.10884725. [DOI] [PubMed] [Google Scholar]
- 18.Theros EG. 1976 Caldwell Lecture: varying manifestation of peripheral pulmonary neoplasms: a radiologic-pathologic correlative study. AJR Am J Roentgenol. 1977;128:893–914. doi: 10.2214/ajr.128.6.893. https://doi.org/10.2214/ajr.128.6.893. [DOI] [PubMed] [Google Scholar]
- 19.Horeweg N, van der Aalst CM, Thunnissen E, et al. Characteristics of lung cancers detected by computer tomography screening in the randomized NELSON trial. Am J Respir Crit Care Med. 2013;187:848–854. doi: 10.1164/rccm.201209-1651OC. https://doi.org/10.1164/rccm.201209-1651OC. [DOI] [PubMed] [Google Scholar]
- 20.Byers T, Vena J, Rzepka T. Predilection of lung cancer for the upper lobes: An epidemiologic inquiry. J Natl Cancer Inst. 1984;72:1271–1275. [PubMed] [Google Scholar]
- 21.Samei E, Eyler W, Baron L. Effects of anatomical structure on signal detection. In: Beutel J, Kundel HL, Van Metter RL, editors. Handbook of medical imaging: physics and psychophysics. Bellingham: Washington; 2000. pp. 655–682. https://doi.org/10.1117/3.832716.ch12. [Google Scholar]
- 22.Muhm JR, Miller WE, Fontana RS, Sanderson DR, Uhlenhopp MA. Lung cancer detected during a screening program using four-month chest radiographs. Radiology. 148:609–615. doi: 10.1148/radiology.148.3.6308709. 983. https://doi.org/10.1148/radiology.148.3.6308709. [DOI] [PubMed] [Google Scholar]
- 23.Medjek M, Hackx M, Ghaye B, De Maertelaer V, Gevenois PA. Value of the “spine sign” on lateral chest views. Br J Radiol. 2015;88:20140378. doi: 10.1259/bjr.20140378. https://doi.org/10.1259/bjr.20140378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chotas HG, Ravin CE. Chest radiography: estimated lung volume and projected area obscured by the heart, mediastinum, and diaphragm. Radiology. 1994;193:403–404. doi: 10.1148/radiology.193.2.7972752. https://doi.org/10.1148/radiology.193.2.7972752. [DOI] [PubMed] [Google Scholar]
- 25.Tala E. Carcinoma of the lung. A retrospective study with special reference to pre-diagnosis period and roentgenographic signs. Acta Radiol Diagn (Stockh) 1967;(Suppl 268):1–127. [PubMed] [Google Scholar]
- 26.Stitik FP, Tockman MS. Radiographic screening in the early detection of lung cancer. Radiol Clin North Am. 1978;16:347–366. [PubMed] [Google Scholar]
- 27.Forrest JV, Sagel SS. The lateral radiograph for early diagnosis of lung cancer. Radiology. 1979;131:309–310. doi: 10.1148/131.2.309. https://doi.org/10.1148/131.2.309. [DOI] [PubMed] [Google Scholar]
- 28.Sagel SS, Evens RG, Forrest JV, Bramson RT. Efficacy of routine screening and lateral chest radiographs in a hospital-based population. N Engl J Med. 1974;291:1001–1004. doi: 10.1056/NEJM197411072911904. https://doi.org/10.1056/NEJM197411072911904. [DOI] [PubMed] [Google Scholar]
- 29.Latief KH, White CS, Protopapas Z, Attar S, Krasna MJ. Search for a primary lung neoplasm in patients with brain metastasis: is the chest radiograph sufficient? AJR Am J Roentgenol. 1997;168:1339–1344. doi: 10.2214/ajr.168.5.9129439. https://doi.org/10.2214/ajr.168.5.9129439. [DOI] [PubMed] [Google Scholar]
- 30.Sherrier RH, Chiles C, Wilkinson WE, Johnson GA, Ravin CE. Effects of image processing on nodule detection rates in digitized chest radiographs: ROC study of observer performance. Radiology. 1988;166:447–450. doi: 10.1148/radiology.166.2.3336719. https://doi.org/10.1148/radiology.166.2.3336719. [DOI] [PubMed] [Google Scholar]
- 31.Rami-Porta R, Bolejack V, Giroux DJ, et al. The IASLC lung cancer staging project: the new database to inform the eighth edition of the TNM classification of lung cancer. J Thorac Oncol. 2014;9:1618–1624. doi: 10.1097/JTO.0000000000000334. https://doi.org/10.1097/JTO.0000000000000334. [DOI] [PubMed] [Google Scholar]
- 32.Forrest JV, Friedman PJ. Radiologic errors in patients with lung cancer. West J Med. 1981;134:485–490. [PMC free article] [PubMed] [Google Scholar]
- 33.Kashiwabara K, Koshi S, Ota K, Tanaka M, Toyonaga M. Outcome in patients with lung cancer found retrospectively to have had evidence of disease on past lung cancer mass screening roentgenograms. Lung Cancer. 2002;35:237–241. doi: 10.1016/s0169-5002(01)00444-5. https://doi.org/10.1016/S0169-5002(01)00444-5. [DOI] [PubMed] [Google Scholar]
- 34.Baker SR, Patel RH, Yang L, Lelkes VM, Castro A. Malpractice suits in chest radiology: an evaluation of the histories of 8265 radiologists. J Thorac Imaging. 2013;28:388–391. doi: 10.1097/RTI.0b013e3182a21be2. https://doi.org/10.1097/RTI.0b013e3182a21be2. [DOI] [PubMed] [Google Scholar]
- 35.Shea FJ, Ziskin MC. Visual system transfer function and optimal viewing distance for radiologists. Invest Radiol. 1972;7:147–151. doi: 10.1097/00004424-197205000-00003. https://doi.org/10.1097/00004424-197205000-00003. [DOI] [PubMed] [Google Scholar]
- 36.Howarth N, Track D. Missed lung lesions: side by side comparision of chest radiography with MDCT. In: Hodler J, Kubik-Huch RA, von Schulthess GK, Zollikofer C, editors. Diseases of the chest and heart Diagnostic imaging and interventional techiques. Springer; Milan: 2015. pp. 80–87. https://doi.org/10.1007/978-88-470-5752-4_10. [PubMed] [Google Scholar]
- 37.Hayabuchi N, Russell WJ, Murakami J, Nishitani H. Screening for lung cancer in a fixed population by biennial chest radiography. Radiology. 1983;148:369–373. doi: 10.1148/radiology.148.2.6306720. https://doi.org/10.1148/radiology.148.2.6306720. [DOI] [PubMed] [Google Scholar]
- 38.Sheline ME, Brikman I, Epstein DM, Mezrich JL, Kundel HL, Arenson RL. The diagnosis of pulmonary nodules: comparison between standard and inverse digitized images and conventional chest radiographs. AJR Am J Roentgenol. 1989;152:261–263. doi: 10.2214/ajr.152.2.261. https://doi.org/10.2214/ajr.152.2.261. [DOI] [PubMed] [Google Scholar]
- 39.Rühl R, Wozniak MM, Werk M, et al. Csl-detector-based dual-exposure dual energy in chest radiography for lung nodule detection: results of an international multicenter trial. Eur Radiol. 2008;18:1831–1839. doi: 10.1007/s00330-008-0945-6. https://doi.org/10.1007/s00330-008-1160-1. [DOI] [PubMed] [Google Scholar]
- 40.Kano A, Doi K, MacMahon H, Hassell D, Giger M. Digital image subtraction of temporally sequential chest images for detection of interval change. Med Phys. 1994;21:453–461. doi: 10.1118/1.597308. https://doi.org/10.1118/1.597308. [DOI] [PubMed] [Google Scholar]
- 41.Okazaki H, Nakamura K, Watanabe H, et al. Improved detection of lung cancer arising in diffuse lung diseases on chest radiographs using temporal subtraction. Acad Radiol. 2004;11:498–505. doi: 10.1016/S1076-6332(03)00820-1. https://doi.org/10.1016/S1076-6332(03)00820-1. [DOI] [PubMed] [Google Scholar]
- 42.Li F, Engelmann R, Metz CE, Doi K, MacMahon H. Lung cancers missed on chest radiographs: results obtained with a commercial computer-aided detection program. Radiology. 2008;246:273–280. doi: 10.1148/radiol.2461061848. https://doi.org/10.1148/radiol.2461061848. [DOI] [PubMed] [Google Scholar]
- 43.Kakeda S, Moriya J, Sato H, et al. Improved detection of lung nodules on chest radiographs using a commercial computer-aided diagnosis system. AJR Am J Roentgenol. 2004;182:505–510. doi: 10.2214/ajr.182.2.1820505. https://doi.org/10.2214/ajr.182.2.1820505. [DOI] [PubMed] [Google Scholar]
- 44.White CS, Flukinger T, Jeudy J, Chen JJ. Use of a computer-aided detection system to detect missed lung cancer at chest radiography. Radiology. 2009;252:273–281. doi: 10.1148/radiol.2522081319. https://doi.org/10.1148/radiol.2522081319. [DOI] [PubMed] [Google Scholar]
- 45.Vikgren J, Zachrisson S, Svalkvist A, et al. Comparison of chest tomosynthesis and chest radiography for detection of pulmonary nodules: human observer study of clinical cases. Radiology. 2008;249:1034–1041. doi: 10.1148/radiol.2492080304. https://doi.org/10.1148/radiol.2492080304. [DOI] [PubMed] [Google Scholar]
- 46.Fardanesh M, White C. Missed lung cancer on chest radiography and computed tomography. Semin Ultrasound CT MR. 2012;33:280–287. doi: 10.1053/j.sult.2012.01.006. https://doi.org/10.1053/j.sult.2012.01.006. [DOI] [PubMed] [Google Scholar]
- 47.White CS, Romney BM, Mason AC, Austin JH, Miller BH, Protopapas Z. Primary carcinoma of the lung overlooked at CT: analysis of findings in 14 patients. Radiology. 1996;199:109–115. doi: 10.1148/radiology.199.1.8633131. https://doi.org/10.1148/radiology.199.1.8633131. [DOI] [PubMed] [Google Scholar]
- 48.Gurney JW. Missed lung cancer at CT: imaging findings in nine patients. Radiology. 1996;199:117–122. doi: 10.1148/radiology.199.1.8633132. https://doi.org/10.1148/radiology.199.1.8633132. [DOI] [PubMed] [Google Scholar]
- 49.Davis SD. Through the “retrospectoscope”: a glimpse of missed lung cancer at CT. Radiology. 1996;199:23–24. doi: 10.1148/radiology.199.1.8633150. https://doi.org/10.1148/radiology.199.1.8633150. [DOI] [PubMed] [Google Scholar]
- 50.Berbaum KS, Schartz KM, Caldwell RT, et al. Satisfaction of search from detection of pulmonary nodules in computed tomography of the chest. Acad Radiol. 2013;20:194–201. doi: 10.1016/j.acra.2012.08.017. https://doi.org/10.1016/j.acra.2012.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.National Lung Screening Trial Research Team. Aberle DR, Adams AM, Berg CD, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395–409. doi: 10.1056/NEJMoa1102873. https://doi.org/10.1056/NEJMoa1102873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kakinuma R, Ohmatsu H, Kaneko M, et al. Detection failures in spiral CT screening for lung cancer: analysis of CT findings. Radiology. 1999;212:61–66. doi: 10.1148/radiology.212.1.r99jn1461. https://doi.org/10.1148/radiology.212.1.r99jn1461. [DOI] [PubMed] [Google Scholar]
- 53.Li F, Sone S, Abe H, MacMahon H, Armato SG, 3rd, Doi K. Lung cancers missed at low-dose helical CT screening in a general population: comparison of clinical, histopathologic, and imaging findings. Radiology. 2002;225:673–683. doi: 10.1148/radiol.2253011375. https://doi.org/10.1148/radiol.2253011375. [DOI] [PubMed] [Google Scholar]
- 54.Horeweg N, Scholten ET, de Jong PA, et al. Detection of lung cancer through low-dose CT screening (NELSON): a prespecified analysis of screening test performance and interval cancers. Lancet Oncol. 2014;15:1342–1350. doi: 10.1016/S1470-2045(14)70387-0. https://doi.org/10.1016/S1470-2045(14)70389-4. [DOI] [PubMed] [Google Scholar]
- 55.Veronesi G, Maisonneuve P, Spaggiari L, et al. Diagnostic performance of low-dose computed tomography screening for lung cancer over five years. J Thorac Oncol. 2014;9:935–939. doi: 10.1097/JTO.0000000000000200. https://doi.org/10.1097/JTO.0000000000000200. [DOI] [PubMed] [Google Scholar]
- 56.Devaraj A. Missed cancers in lung cancer screening--more than meets the eye. Eur Radiol. 2015;25:89–91. doi: 10.1007/s00330-014-3395-3. https://doi.org/10.1007/s00330-014-3395-3. [DOI] [PubMed] [Google Scholar]
- 57.Firmino M, Morais AH, Mendoça RM, Dantas MR, Hekis HR, Valentim R. Computer-aided detection system for lung cancer in computed tomography scans: review and future prospects. Biomed Eng Online. 2014;13:41. doi: 10.1186/1475-925X-13-41. https://doi.org/10.1186/1475-925X-13-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Berlin L. Failure to diagnose lung cancer: anatomy of a malpractice trial. AJR Am J Roentgenol. 2003;180:37–45. doi: 10.2214/ajr.180.1.1800037. https://doi.org/10.2214/ajr.180.1.1800037. [DOI] [PubMed] [Google Scholar]
- 59.Berlin L. Liability of performing CT screening for coronary artery disease and lung cancer. AJR Am J Roentgenol. 2002;179:837–842. doi: 10.2214/ajr.179.4.1790837. https://doi.org/10.2214/ajr.179.4.1790837. [DOI] [PubMed] [Google Scholar]
- 60.Jankowski A, Martinelli T, Timsit JF, et al. Pulmonary nodule detection on MDCT images: evaluation of diagnostic performance using thin axial images, maximum intensity projections, and computer-assisted detection. Eur Radiol. 2007;17:3148–3156. doi: 10.1007/s00330-007-0727-6. https://doi.org/10.1007/s00330-007-0727-6. [DOI] [PubMed] [Google Scholar]
- 61.Armato SG, 3rd, Li F, Giger ML, MacMahon H, Sone S, Doi K. Lung cancer: performance of automated lung nodule detection applied to cancers missed in a CT screening program. Radiology. 2002;225:685–692. doi: 10.1148/radiol.2253011376. https://doi.org/10.1148/radiol.2253011376. [DOI] [PubMed] [Google Scholar]
- 62.Li F, Arimura H, Suzuki K, et al. Computer-aided detection of peripheral lung cancers missed at CT: ROC analyses without and with localization. Radiology. 2005;237:684–690. doi: 10.1148/radiol.2372041555. https://doi.org/10.1148/radiol.2372041555. [DOI] [PubMed] [Google Scholar]
- 63.Foti G, Faccioli N, D’Onofrio M, Contro A, Milazzo T, Pozzi Mucelli R. Evaluation of a method of computer-aided detection (CAD) of pulmonary nodules with computed tomography. Radiol Med. 2010;115:950–961. doi: 10.1007/s11547-010-0556-6. https://doi.org/10.1007/s11547-010-0556-6. [DOI] [PubMed] [Google Scholar]
- 64.Zhao Y, de Bock GH, Vliegenthart R, et al. Performance of computer-aided detection of pulmonary nodules in low-dose CT: comparison with double reading by nodule volume. Eur Radiol. 2012;22:2076–2084. doi: 10.1007/s00330-012-2437-y. https://doi.org/10.1007/s00330-012-2437-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yuan R, Vos PM, Cooperberg PL. Computer-aided detection in screening CT for pulmonary nodules. AJR Am J Roentgenol. 2006;186:1280–1287. doi: 10.2214/AJR.04.1969. https://doi.org/10.2214/AJR.04.1969. [DOI] [PubMed] [Google Scholar]