Abstract
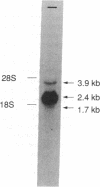
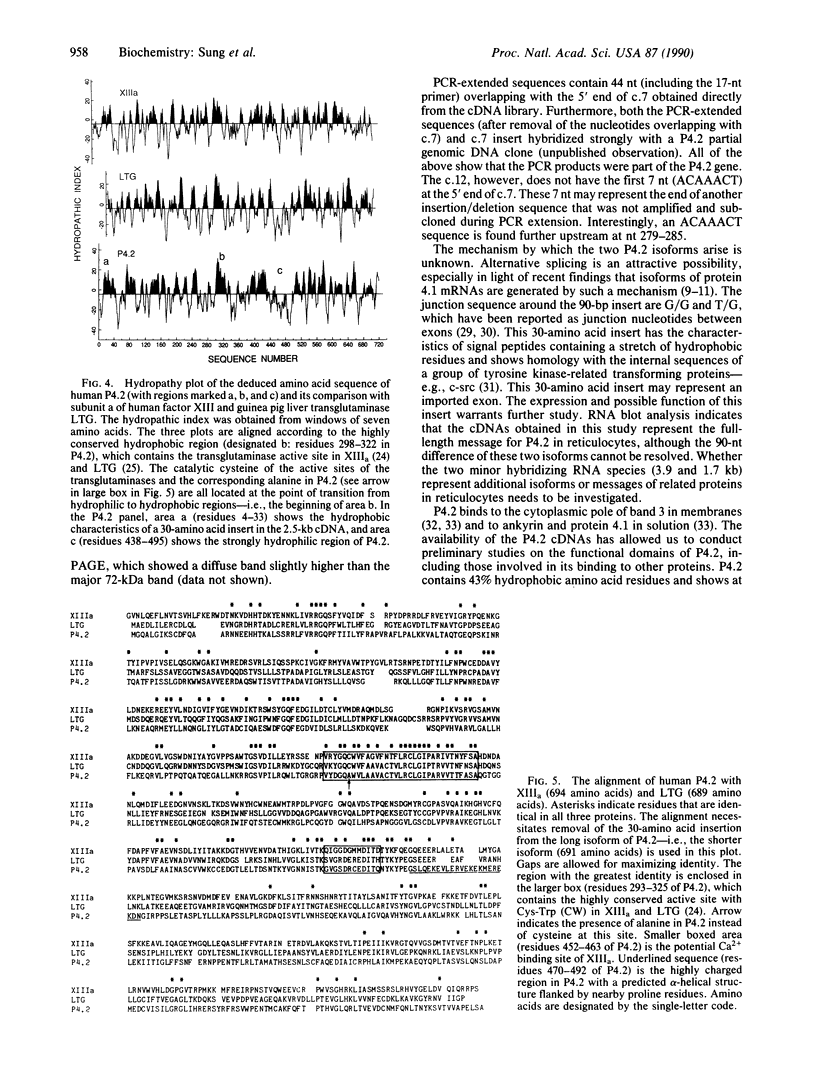
Protein 4.2 (P4.2) comprises approximately 5% of the protein mass of human erythrocyte (RBC) membranes. Anemia occurs in patients with RBCs deficient in P4.2, suggesting a role for this protein in maintaining RBC stability and integrity. We now report the molecular cloning and characterization of human RBC P4.2 cDNAs. By immunoscreening a human reticulocyte cDNA library and by using the polymerase chain reaction, two cDNA sequences of 2.4 and 2.5 kilobases (kb) were obtained. These cDNAs differ only by a 90-base-pair insert in the longer isoform located three codons downstream from the putative initiation site. The 2.4- and 2.5-kb cDNAs predict proteins of approximately 77 and approximately 80 kDa, respectively, and the authenticity was confirmed by sequence identity with 46 amino acids of three cyanogen bromide-cleaved peptides of P4.2. Northern blot analysis detected a major 2.4-kb RNA species in reticulocytes. Isolation of two P4.2 cDNAs implies existence of specific regulation of P4.2 expression in human RBCs. Human RBC P4.2 has significant homology with human factor XIII subunit a and guinea pig liver transglutaminase. Sequence alignment of P4.2 with these two transglutaminases, however, revealed that P4.2 lacks the critical cysteine residue required for the enzymatic crosslinking of substrates.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bennett V. The membrane skeleton of human erythrocytes and its implications for more complex cells. Annu Rev Biochem. 1985;54:273–304. doi: 10.1146/annurev.bi.54.070185.001421. [DOI] [PubMed] [Google Scholar]
- Chang H., Langer P. J., Lodish H. F. Asynchronous synthesis of erythrocyte membrane proteins. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3206–3210. doi: 10.1073/pnas.73.9.3206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Prediction of protein conformation. Biochemistry. 1974 Jan 15;13(2):222–245. doi: 10.1021/bi00699a002. [DOI] [PubMed] [Google Scholar]
- Conboy J. G., Chan J., Mohandas N., Kan Y. W. Multiple protein 4.1 isoforms produced by alternative splicing in human erythroid cells. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9062–9065. doi: 10.1073/pnas.85.23.9062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conboy J., Kan Y. W., Shohet S. B., Mohandas N. Molecular cloning of protein 4.1, a major structural element of the human erythrocyte membrane skeleton. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9512–9516. doi: 10.1073/pnas.83.24.9512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curtis P. J., Palumbo A., Ming J., Fraser P., Cioe L., Meo P., Shane S., Rovera G. Sequence comparison of human and murine erythrocyte alpha-spectrin cDNA. Gene. 1985;36(3):357–362. doi: 10.1016/0378-1119(85)90191-x. [DOI] [PubMed] [Google Scholar]
- Drickamer L. K. Fragmentation of the band 3 polypeptide from human erythrocyte membranes. Identification of regions likely to interact with the lipid bilayer. J Biol Chem. 1977 Oct 10;252(19):6909–6917. [PubMed] [Google Scholar]
- Folk J. E. Transglutaminases. Annu Rev Biochem. 1980;49:517–531. doi: 10.1146/annurev.bi.49.070180.002505. [DOI] [PubMed] [Google Scholar]
- Friedrichs B., Koob R., Kraemer D., Drenckhahn D. Demonstration of immunoreactive forms of erythrocyte protein 4.2 in nonerythroid cells and tissues. Eur J Cell Biol. 1989 Feb;48(1):121–127. [PubMed] [Google Scholar]
- Grundmann U., Amann E., Zettlmeissl G., Küpper H. A. Characterization of cDNA coding for human factor XIIIa. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8024–8028. doi: 10.1073/pnas.83.21.8024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanks S. K., Quinn A. M., Hunter T. The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science. 1988 Jul 1;241(4861):42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- Ichinose A., Hendrickson L. E., Fujikawa K., Davie E. W. Amino acid sequence of the a subunit of human factor XIII. Biochemistry. 1986 Nov 4;25(22):6900–6906. doi: 10.1021/bi00370a025. [DOI] [PubMed] [Google Scholar]
- Ikura K., Nasu T., Yokota H., Tsuchiya Y., Sasaki R., Chiba H. Amino acid sequence of guinea pig liver transglutaminase from its cDNA sequence. Biochemistry. 1988 Apr 19;27(8):2898–2905. doi: 10.1021/bi00408a035. [DOI] [PubMed] [Google Scholar]
- Kopito R. R., Lodish H. F. Primary structure and transmembrane orientation of the murine anion exchange protein. Nature. 1985 Jul 18;316(6025):234–238. doi: 10.1038/316234a0. [DOI] [PubMed] [Google Scholar]
- Korsgren C., Cohen C. M. Associations of human erythrocyte band 4.2. Binding to ankyrin and to the cytoplasmic domain of band 3. J Biol Chem. 1988 Jul 25;263(21):10212–10218. [PubMed] [Google Scholar]
- Korsgren C., Cohen C. M. Purification and properties of human erythrocyte band 4.2. Association with the cytoplasmic domain of band 3. J Biol Chem. 1986 Apr 25;261(12):5536–5543. [PubMed] [Google Scholar]
- Korsgren C., Lawler J., Lambert S., Speicher D., Cohen C. M. Complete amino acid sequence and homologies of human erythrocyte membrane protein band 4.2. Proc Natl Acad Sci U S A. 1990 Jan;87(2):613–617. doi: 10.1073/pnas.87.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. At least six nucleotides preceding the AUG initiator codon enhance translation in mammalian cells. J Mol Biol. 1987 Aug 20;196(4):947–950. doi: 10.1016/0022-2836(87)90418-9. [DOI] [PubMed] [Google Scholar]
- Krebs E. G., Beavo J. A. Phosphorylation-dephosphorylation of enzymes. Annu Rev Biochem. 1979;48:923–959. doi: 10.1146/annurev.bi.48.070179.004423. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lorand L., Downey J., Gotoh T., Jacobsen A., Tokura S. The transpeptidase system which crosslinks fibrin by gamma-glutamyle-episilon-lysine bonds. Biochem Biophys Res Commun. 1968 Apr 19;31(2):222–230. doi: 10.1016/0006-291x(68)90734-1. [DOI] [PubMed] [Google Scholar]
- McMahon A. P., Giebelhaus D. H., Champion J. E., Bailes J. A., Lacey S., Carritt B., Henchman S. K., Moon R. T. cDNA cloning, sequencing and chromosome mapping of a non-erythroid spectrin, human alpha-fodrin. Differentiation. 1987;34(1):68–78. doi: 10.1111/j.1432-0436.1987.tb00052.x. [DOI] [PubMed] [Google Scholar]
- Moon R. T., Lazarides E. Biogenesis of the avian erythroid membrane skeleton: receptor-mediated assembly and stabilization of ankyrin (goblin) and spectrin. J Cell Biol. 1984 May;98(5):1899–1904. doi: 10.1083/jcb.98.5.1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mount S. M. A catalogue of splice junction sequences. Nucleic Acids Res. 1982 Jan 22;10(2):459–472. doi: 10.1093/nar/10.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ngai J., Stack J. H., Moon R. T., Lazarides E. Regulated expression of multiple chicken erythroid membrane skeletal protein 4.1 variants is governed by differential RNA processing and translational control. Proc Natl Acad Sci U S A. 1987 Jul;84(13):4432–4436. doi: 10.1073/pnas.84.13.4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rybicki A. C., Heath R., Wolf J. L., Lubin B., Schwartz R. S. Deficiency of protein 4.2 in erythrocytes from a patient with a Coombs negative hemolytic anemia. Evidence for a role of protein 4.2 in stabilizing ankyrin on the membrane. J Clin Invest. 1988 Mar;81(3):893–901. doi: 10.1172/JCI113400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slife C. W., Dorsett M. D., Tillotson M. L. Subcellular location and identification of a large molecular weight substrate for the liver plasma membrane transglutaminase. J Biol Chem. 1986 Mar 5;261(7):3451–3456. [PubMed] [Google Scholar]
- Steck T. L. The organization of proteins in the human red blood cell membrane. A review. J Cell Biol. 1974 Jul;62(1):1–19. doi: 10.1083/jcb.62.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki K., Ikebuchi H., Terao T. Mercuric and cadmium ions stimulate phosphorylation of band 4.2 protein on human erythrocyte membranes. J Biol Chem. 1985 Apr 10;260(7):4526–4530. [PubMed] [Google Scholar]
- Takahashi N., Takahashi Y., Putnam F. W. Primary structure of blood coagulation factor XIIIa (fibrinoligase, transglutaminase) from human placenta. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8019–8023. doi: 10.1073/pnas.83.21.8019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang T. K., Leto T. L., Correas I., Alonso M. A., Marchesi V. T., Benz E. J., Jr Selective expression of an erythroid-specific isoform of protein 4.1. Proc Natl Acad Sci U S A. 1988 Jun;85(11):3713–3717. doi: 10.1073/pnas.85.11.3713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodgett J. R., Gould K. L., Hunter T. Substrate specificity of protein kinase C. Use of synthetic peptides corresponding to physiological sites as probes for substrate recognition requirements. Eur J Biochem. 1986 Nov 17;161(1):177–184. doi: 10.1111/j.1432-1033.1986.tb10139.x. [DOI] [PubMed] [Google Scholar]
- Yamada Y., Avvedimento V. E., Mudryj M., Ohkubo H., Vogeli G., Irani M., Pastan I., de Crombrugghe B. The collagen gene: evidence for its evolutinary assembly by amplification of a DNA segment containing an exon of 54 bp. Cell. 1980 Dec;22(3):887–892. doi: 10.1016/0092-8674(80)90565-6. [DOI] [PubMed] [Google Scholar]