Abstract
Several reports described an increased risk of cardiovascular (CV) events, mainly atherothrombotic, in Chronic Myeloid Leukemia (CML) patients receiving nilotinib. However, the underlying mechanism remains elusive. The objective of the current cross-sectional retrospective study is to address a potential correlation between Tyrosine Kinase Inhibitors (TKIs) treatment and CV events. One hundred and 10 chronic phase CML patients in complete cytogenetic response during nilotinib or imatinib, were screened for CV events and evaluated for: traditional CV risk factors, pro/anti-inflammatory biochemical parameters and detrimental ORL1 gene polymorphisms (encoding for altered oxidized LDL receptor-1). Multivariate analysis of the whole cohort showed that the cluster of co-existing nilotinib treatment, dyslipidaemia and G allele of LOX-1 polymorphism was the only significant finding associated with CV events. Furthermore, multivariate analysis according to TKI treatment confirmed IVS4-14 G/G LOX-1 polymorphism as the strongest predictive factor for a higher incidence of CV events in nilotinib patients. Biochemical assessment showed an unbalanced pro-inflammatory cytokines network in nilotinib vs imatinib patients. Surprisingly, pre-existing traditional CV risk factors were not always predictive of CV events. We believe that in nilotinib patients an induced “inflammatory/oxidative status”, together with a genetic pro-atherothrombotic predisposition, may favour the increased incidence of CV events. Prospective studies focused on this issue are ongoing.
Keywords: PAOD, TKI, CML, inflammation, atherothrombotic risk
INTRODUCTION
The great improvement in survival expectancy in Chronic Phase (CP) Chronic Myeloid Leukemia (CML) patients chronically treated with Tyrosine Kinase Inhibitors (TKIs) has prompted the clinical issue of long-term drug toxicity, safety and quality of life [1–3]. While imatinib has been generally well tolerated with an adverse effect profile stable over the years [1], long-term side effects of second and third generation TKIs are not completely defined yet and especially cardiovascular (CV) adverse events related to their use are a matter of concern [4–6]. Several recent retrospective clinical studies reported a higher incidence of atherothrombotic events including peripheral arterial occlusive disease (PAOD) in CML patients treated with nilotinib when compared to imatinib or dasatinib; these peculiar adverse events appear to increase over time during continuous nilotinib treatment [6–13]. Preliminary experimental data suggest that nilotinib, in contrast to imatinib, may cause direct pro-atherogenic, growth-inhibitory and thus anti-angiogenic effects on vascular endothelial cells [14]. However, the pathogenic link between nilotinib and atherothrombosis has not been fully elucidated and more work is needed to better understand the effects of nilotinib in preclinical CV models. From a clinical point of view, whether atherothrombotic events occur predominantly in CML patients with pre-existing traditional CV risk factors is a matter of debate as most studies are based on retrospective data and the risk factors rate for arterial occlusive disease varied significantly [15–19]. We have also taken into consideration that, according to various reports, the percentage of patients on nilotinib treatment with pre-existing CV risk factors who experienced atherothrombotic events ranged anywhere from over 90% down to 20% [9, 16, 17, 19, 20].
Given such heterogeneity of findings and clinical experience, we studied patients receiving TKIs treatment, in order to identify if the vascular events could be associated to a genetic predisposition and/or a drug induced pro-atherothrombotic effect, independently of the presence of traditional CV risk factors.
Arterial occlusive disease is mainly caused by atherosclerosis, a multifactorial disease of the vessels involving lipid and other metabolic disturbances, thrombogenic components, cell death and inflammatory responses of the arterial wall [21]. Metabolic and pro-inflammatory factors share a relevant role in the pathogenesis of atherothrombotic events mainly via enhanced lipid peroxidation [22]. Lectin-like oxidized-LDL (ox-LDL) receptor-1(LOX-1), is the main receptor for the pro-atherogenic ox-LDL [22–25]; it is expressed in endothelial cells, macrophages, platelets and vascular smooth muscle cells, all relevant cellular effectors of atherogenesis and atherothrombosis mechanisms [22–25]. LOX-1 gene, named OLR1, is mapped on chromosome 12p 13.2-p12.3 and different studies demonstrated that OLR1 single nucleotide polymorphisms (SNPs) may cause susceptibility to coronary artery disease (CAD), particularly in Caucasian ancestral populations [23]. In particular, the IVS4-14 A/G polymorphism, which acts in complete linkage-disequilibrium with other five polymorphisms located in the introns 4, 5 and 3′UTR region, has been implicated in geographic disparities in CV event rate [24].
Thus, to elucidate a potential pathogenic link between TKIs treatment and mechanisms underlying PAOD or other atherothrombotic events, we investigated, in a series of CML patients treated with nilotinib and imatinib, the correlation between LOX-1 polymorphisms, the presence of classic CV risk factors and vascular events. Furthermore, during treatment with either TKIs, we studied a selected pro-atherothrombotic biochemical profile evaluating the following: 1) coagulation state by Endogenous Thrombin Potential (ETP); 2) specific circulating mediators of inflammation such as IL-10, IL-6 and TNFα; 3) high-sensitivity C-reactive protein (hs-CRP) previously proven to be involved in the atherothrombosis burden [22–27]; 4) ox-LDL as metabolic effectors of atherosclerosis predisposition [22, 23, 25]; 5) soluble CD40 ligand (sCD40L) as a marker of platelet derived-inflammation and platelet activation trigger [22, 28].
RESULTS
Whole patients cohort data and multivariate analysis
One hundred and ten consequent CP CML patients in complete cytogenetic response (CCyR), willing to participate in the study, were evaluated. In order to consider a homogeneous cohort, to have the least active cancer-related thrombotic risk and to minimize any cytokine unbalance due to different CML cell burden at study evaluation, we included only CCyR patients.
Clinical characteristics, risk factors distribution and incidence of CV events are summarized in Table 1. The incidence of two or more traditional CV risk factors was 56%. LOX-1 genotype frequency in the whole cohort of patients respected the Hardy-Weinberg equilibrium that is present in ancestral Caucasian origin populations [24] (Table 2). Atherothrombotic events occurred in 17/110 (15%) patients thus counting for a consistent sample size for multivariate analysis. We took in consideration the TKI treatment at the time of any CV event or, if no event occurred, at the time of study visit: 58 patients were on imatinib and 52 on nilotinib treatment.
Table 1. Clinical characteristics and risk factors distribution of patients during TKI treatment or at the time of cardiovascular event.
| whole cohort | imatinib cohort | nilotinib cohort | p | |
|---|---|---|---|---|
| N° Patients | 110 | 58 | 52 | 0.094 |
| M/F | 66/44 | 31/27 | 35/17 | 0.061 |
| Median age (years, range) | 61 (29-83) | 59 (33-82) | 62 (29-83) | 0.088 |
| Median duration of CML (months, range) | 60 (3-252) | 84 (12-180) | 56 (3-228) | 0.019 |
| Median TKI exposure (months, range) | n.a. | 84 (12-180) | 24 (3-84) | 0.0014 |
| First-line (%) | n.a. | 58 (100) | 29 (55) | 0.0029 |
| Second-third line TKI (%) | n.a. | 0 (0) | 23 (45) | n.a. |
| History of smoking (%) | 34 (31) | 17 (29) | 17 (33) | 0.126 |
| Arterial Hypertension (%) | 36 (33) | 17 (29) | 19 (37) | 0.054 |
| Diabetes Mellitus (%) | 19 (17) | 9 (16) | 10* (19) | 0.088 |
| BMI (range) | 26±1,4 | 26,1±1,4 | 25,9±1,3 | 0.091 |
| Dyslipidaemia (%) | 35 (32) | 15 (26) | 20 (38) | 0.047 |
| Familiarity (%) | 58 (53) | 30 (52) | 28 (54) | 0.099 |
| Number of risk factor > 2 | 62 (56) | 29 (50) | 33 (63) | 0.048 |
| Number of risk factor ≥ 3 | 44 (40) | 18 (31) | 26 (50) | 0.041 |
| Total n° of cardiovascular events (%) | 17/110 (15) | 3/58 (5) | 14/52 (27) | 0.00011 |
| First line treatment (%) | n.a. | 3/58 (5) | 6/29 (21) | n.a. |
| Second-third line (%) | n.a. | 0/0 (0) | 8/23 (35) | n.a. |
| TKI treatment time to event (months) | 125, 132, 102 | 30 (range 18-72) | n.a. |
Abbreviation: CML chronic myeloid leukemia, TKI tyrosine kinase inhibitor, BMI body mass index, n.a. not applicable *2/10 glucose intolerance
Table 2. Distribution of oxidized-LDL receptor-1 (LOX-1) polymorphisms (IVS4-14 A/G) in study population.
| all patients | imatinib whole cohort (event cohort) | nilotinib whole cohort (event cohort) | p° | |
|---|---|---|---|---|
| N° Patients | 110 | 58 (3) | 52 (14) | |
| A/A LOX-1 polymorphism | 33 | 22 (0) | 11 (0) | 0.046 |
| A/G LOX-1 polymorphism | 63 | 34 (2) | 29 (2) | 0.081 |
| G/G LOX-1 polymorphism | 14 | 2 (1) | 12 (12) | 0.00021 |
| A/A LOX-1 polymorphism (G.F. /A.F.) | *(0.300/0.195)§ | (0.379/0.270) | *(0.211/0.156) (p = 0.048) | |
| A/G LOX-1 polymorphism (G.F./A.F.) | *(0.572/0.642)§ | (0.586/0.688) | *(0.557/0.601) (p = 0.094) | |
| G/G LOX-1 polymorphism (G.F./A.F.) | *(0.128/0.163)§ | (0.034/0.042) | *(0.230/0.243) (p = 0.0038) | |
| G/G Genotype/Events relation * | p= 0.085 | p = 0.0094 |
Abbreviation : LOX-1 oxidized-LDL receptor-1; G.F. genotype frequency; A.F. allele frequency
χ2 test of independence
(p = n.s. with respect to HapMap CEU, downloaded from http://www.hapmap.org.genotypes/ and Italian population)
Mann-Whitney U test.
Whole cohort multivariate analysis
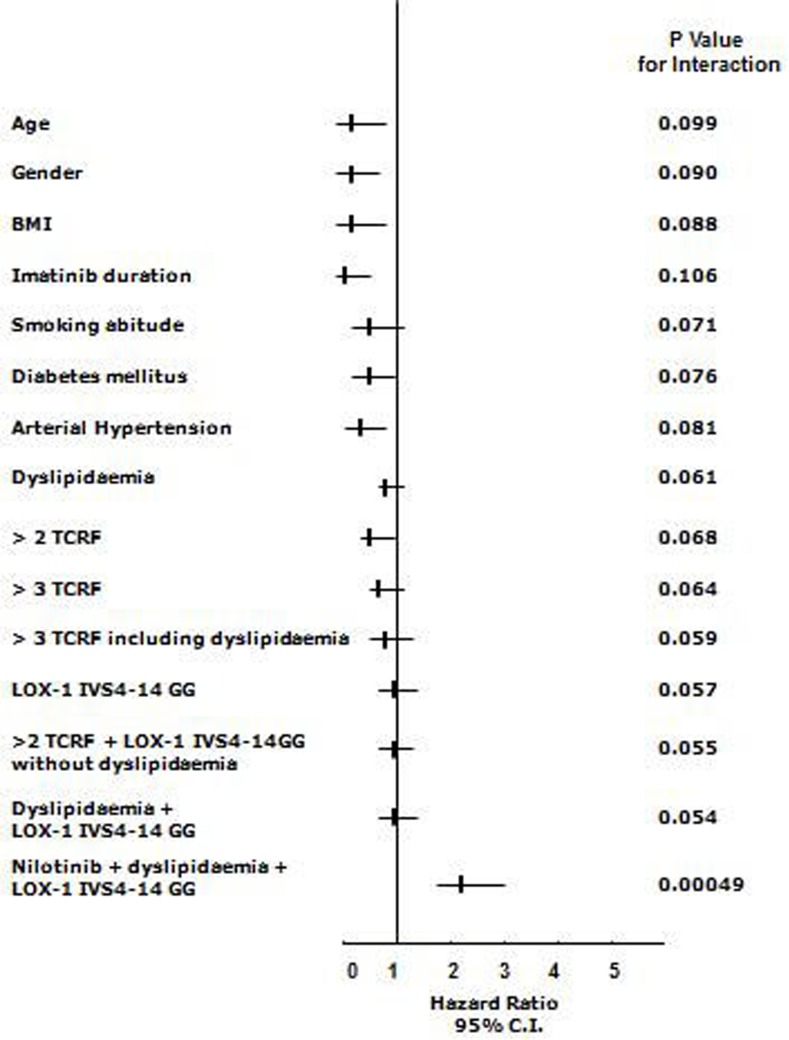
The Cox Hazard model and the Hosmer-Lemeshow confirmation test, once corrected for each applicable data (age, gender, body mass index (BMI), single or clustered risk factors including smoking habit, CV medications, LOX-1 polymorphism, TKI treatment) available at the time of occurring event or at the time of clinical observation if event-free, showed that the cluster of co-existing nilotinib treatment, dyslipidaemia and the G allele of LOX-1 polymorphisms was the only significant finding associated with events (H.R. 2.19 95% C.I. 1.66-2.99, p < 0.001) (Figure 1). Furthermore, in patients with two or three traditional risk factors, no CV events occurred during treatment with either TKIs whether not coupled with both G allele of LOX-1 and dyslipidaemia (Figure 1).
Figure 1. Relations among end-points and nonparametric data in the whole cohort of CML patients treated with TKIs (n= 110).
Cox proportional-hazards modeling and formal test for interaction: starting from logistic regression analysis in which Y was the analyzed variable (both parametric such as age and BMI); the variables X1 and X2 were the presence or absence (1 or 0) of the LOX-1 polymorphism or other non-parametric variables (drugs, history of cardiovascular classical risk factors, here named TCRF, and X3 the combination). The simplified formula for calculation was: Y = β0+β1X1+β2X2+β3X3 and the null hypothesis was tested as H0: β3 = 0. Final data validation has been assessed with a resampling technique (exact tests) and discrimination analysis with the Hosmer–Lemeshow method [G2HL= Σ10J=1 (Oj – Ej)2/Ej (1-Ej/nj) ∼X28], where nj = number of observations in the jth group, Oj = Σ1yij = observed number of positive cases in the jth group, Ej = Σpij = Expected number of positive cases in the jth group. (Each reported p is obtained by this technique and considered significant if < 0.05)
Patients data and multivariate analysis according to TKI treatment
Following the evidence that nilotinib treatment was a favoring factor for vascular events in the whole cohort, we further analyzed putative differences among clinical and genetic variables according to imatinib or nilotinib treatment. As reported above, at the time of any CV event or, if no event occurred, at the time of study visit, 58 patients were on imatinib and 52 on nilotinib treatment (Table 1). As expected from a “real life” unselected study population, the duration of the disease and the TKI exposure were significantly longer in the imatinib cohort when compared to the nilotinib cohort (Table 1). Additionally, 45% of nilotinib patients were on second-third line treatment. The distribution of traditional CV risk factors showed a slightly higher prevalence of dyslipidaemia in the nilotinib cohort. Similarly, having three or more CV risk factors was a more frequent observation in such group (Table 1).
An atherothrombotic event occurred in 3/58 (5%) imatinib treated patients (1 PAOD, 2 carotid occlusion > 50%), and in 14/52 (27%) patients in the nilotinib cohort (9 PAOD, 5 acute coronary syndrome) (Table 1). All three events in the imatinib group occurred during first line treatment, while 6/14 (43%) and 8/14 (57%) of events recorded in the nilotinib cohort occurred during first and second-third line treatment, respectively. Time from TKI exposure to CV event was 125, 132 and 102 months for the three patients receiving imatinib while median time of nilotinib exposure to event was 30 months (range 18-72) (Table 1).
LOX-1 polymorphism distribution
Considering the genotype frequency according to TKI treatment, we found a slight excess of homozygotes A/A in the imatinib group and a significant excess of homozygotes G/G in the nilotinib treated cohort (Table 2). However, it has to be noted that 23 nilotinib patients switched from a previous treatment with imatinib, thus if we consider G/G frequency according to first line therapy we found a similar distribution of the LOX-1 G/G polymorphism between the two treatment groups (imatinib first line treated cohort n = 81, G/G n = 10 = 12.4%; G.F./A.F. 0.148/0.184; nilotinib first line treated cohort n = 29, G/G n = 4 = 13.8%, G.F./A.F. 0.156/0.193, no statistically significant difference). Interestingly, the homozygotes G/G clustered in the first and second line nilotinib sub-group of patients experiencing an atherothrombotic event during treatment (Table 2).
Multivariate analysis
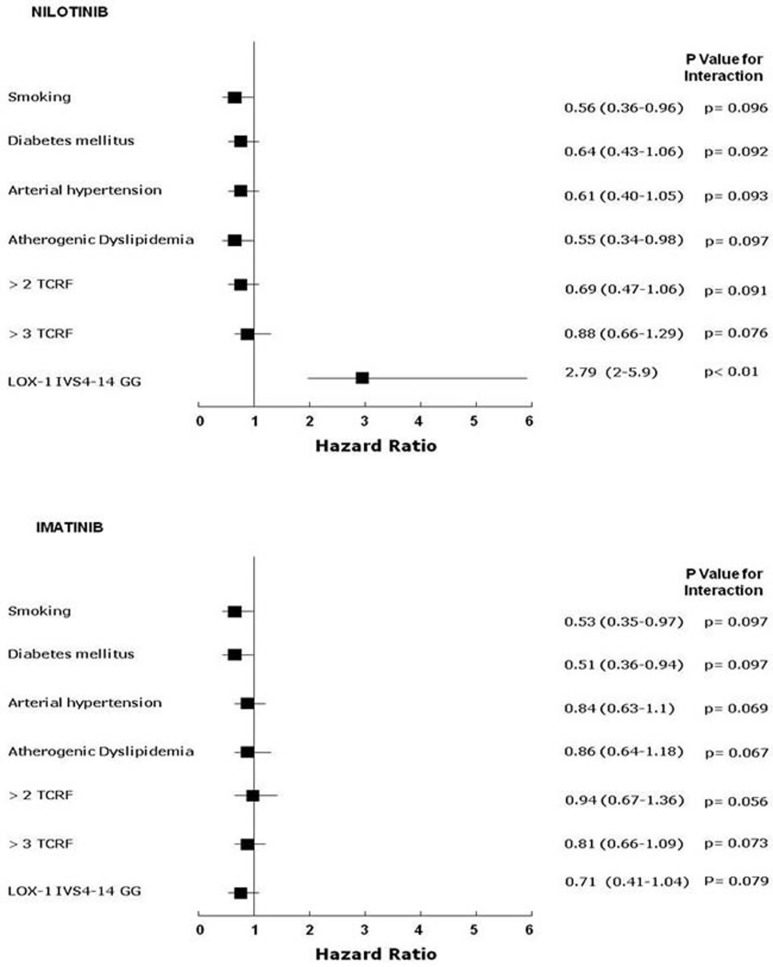
The Cox Hazard model and the Hosmer-Lemeshow confirmation test, once corrected for each applicable data (age, gender, BMI, single or clustered risk factors including smoking habit, CV medications, LOX-1 polymorphism) available at the time of event recording or at the time of clinical observation if event-free, showed that the single influencing risk factor was the G/G homozygosis for IVS4-14A/G of OLR1 in the nilotinib group (Figure 2). No significant influence on vascular event rate was detected for any single traditional CV risk factor, despite the slightly increased prevalence of dyslipidaemic subjects in the nilotinib group (Figure 2, Table 1).
Figure 2. Relations among end-points and nonparametric data in imatinib treated (n=58) and nilotinib treated (n=52) CML patients.
Cox proportional-hazards modeling and formal test for interaction: starting from logistic regression analysis in which Y was the analyzed variable; the variable X1 and X2 were the presence or not (1 or 0) of the LOX-1 polymorphism or other non-parametric variables (history of cardiovascular classical risk factors, here named TCRF, and X3 the combination). The simplified formula for calculation was: Y = β0+β1X1+β2X2+β3X3 and the null hypothesis was tested as H0: β3 = 0. Final validation of data was assessed by a resampling technique (exact tests) and discrimination analysis by the Hosmer–Lemeshow method [G2HL= Σ10J=1 (Oj – Ej)2/Ej (1-Ej/nj) ∼X28], where nj = number of observations in the jth group, Oj = Σ1yij = observed number of positive cases in the jth group, Ej = Σpij = Expected number of positive cases in the jth group. (Each reported p is that obtained by this technique and significant if < 0.05)
Biochemical analysis according to TKI treatment
The biochemical analysis was performed during follow-up visit in all 110 patients, yet it has to be underlined that after an atherothrombotic event some clinicians decided to switch TKI due to the adverse event. Thus biochemical assessment was performed in 65 patients on imatinib (7 of them has been previously treated with nilotinib) and in 45 patients on nilotinib treatment. Patients with a history of CV events underwent biochemical analysis after at least 6 months from the event (median time 12 months, range 6-60 months). As shown in Table 3, ox-LDL, sCD40L level and ETP, in close linear relation among each other, (Supplemental Table S1), were significantly higher in nilotinib vs. imatinib treated group, while IL-10 level, inversely related to ox-LDL, sCD40L and ETP (Supplemental Table S1), was significantly lower.
Table 3. Biochemical pro-inflammatory/pro-atherothrombotic evaluation according to TKI treatment at study visit.
| imatinib whole cohort | nilotinib whole cohort | p | imatinib (previously treated with nilotinib) | P (vs. always treated with imatinib n. 58) | |
|---|---|---|---|---|---|
| N° Patients | 65 | 45 | 7 | ||
| oxLDL (UI/L) | 69.9±7.1 | 92.2±9.9 | 0.0022 | 71.4±8.8 | 0.097 |
| IL6 (pg/ml) | 8.8±1.33 | 9.9±1.44 | 0.091 | 8.5±1.67 | 0.094 |
| IL10 (pg/ml) | 4.86±1.17 | 1.06±0.58 | 0.00012 | 4.59±1.23 | 0.081 |
| TNFα (pg/ml) | 9.6±1.8 | 10.8±1.93 | 0.094 | 9.9±1.9 | 0.088 |
| sCD40L (pg/ml) | 330.5±59.2 | 513.3±91.9 | 0.0023 | 336.4±55.2 | 0.090 |
| ETP (%) | 7.3±1.82 | 14.7±3.73 | 0.0019 | 7.4±1.93 | 0.104 |
| hs-CRP (mg/dl) | 1.02±0.17 | 1.14±0.21 | 0.076 | 1.09±0.14 | 0.070 |
Abbreviation: oxLDL oxidized-LDL, IL interleukin, TNFα tumor necrosis factor alpha, sCD40L soluble CD40 ligand, ETP Endogenous Thrombin Potential, hs-CRP high-sensibility C reactive protein.
No statistically significant differences were found in the expression of IL-6 and TNF-α between the two groups (Table 3). Interestingly, we observed no statistical difference in the cytokine profile between patients who had always been treated with imatinib and those 7 patients that were on imatinib at the time of biochemical assessment but experienced a CV event during nilotinib (Table 3).
The influence of CV risk factors and ongoing medications for CV risk control on the biochemical profile of all CML patients was also assessed. In the nilotinib cohort ox-LDL, sCD40L, ETP and IL-10 levels showed a similar pattern in patients with CV risk factors and concomitant CV drugs as well as in those without risk factors and no related treatment (Supplemental Table S1 and S2). Thus, the detrimental biochemical profile observed in nilotinib treated patients was unrelated to the underlying CV risk and remained significantly different from that observed both in the imatinib treated cohort and in the non-CML matched controls (Supplemental Table S1 and S2).
Conversely, fasting plasma glucose, lipid profile (including LDL) and hs-CRP were not significantly different between imatinib and nilotinib treated group, regardless of the presence of CV risk factors and related treatments (Table S1).
DISCUSSION
Recently, the high occurrence of CV toxicity mainly related to atherothrombotic events during second and third generation TKIs treatment has prompted the interest and concern of clinicians caring for CML patients [13, 29]. However, the trigger of these serious adverse events has not fully clarified yet. In this preliminary cross-sectional study including a series of 110 CML patients treated with imatinib or nilotinib, we found that the cluster of co-existing nilotinib treatment, dyslipidaemia and the G allele of LOX-1 polymorphism was the only significant finding associated with atherothrombotic events.
As nilotinib appeared to be a possible causing factor for vascular events of the overall population, we further performed a multivariate analysis according to TKI treatment including age, gender, BMI, single or clustered risk factors, CV medications, LOX-1 polymorphism. The latter confirmed that the detrimental IVS4-14 G/G LOX-1 polymorphism was the strongest predictive factor for a higher incidence of atherothrombotic events in nilotinib treated patients (i.e. 14/52 (27%) vs. 3/58 (5%) events in nilotinib and imatinib treated patients, respectively). To our surprise, we found no clear correlation of traditional CV risk factors other than dyslipidaemia with atherothrombotic events. Indeed, we found that 12/14 (86%) of nilotinib patients with vascular events had G/G variant genotype, whereas among 26 nilotinib patients with ≥3 traditional risk factors, only 9/26 (35%) experienced an atherothrombotic event, thus implying a possible role for a different genetic profile. The distribution of LOX-1 IVS4-14 A/G polymorphisms of the whole CML population showed a close correlation with previously described Caucasian populations epidemiologic data [24]. However, with respect to treatment, the G/G distribution apparently clustered in the nilotinib treated cohort (Table 2) thus inferring a possible bias in the patients' selection. Against this hypothesis is the lack of significant between-group difference in the allelic and genetic frequency of LOX-1 when we considered patients during first line treatment (81 imatinib, 29 nilotinib) (Table 2). The apparent segregation of G/G in the nilotinib group may be due to the fact that eight G/G patients treated in first line with imatinib without evidence of any vascular event, were then switched to nilotinib (due to intolerance or no response) and only afterward experienced an event.
From a pathogenic point of view, it is known that dysfunctional LOX-1 derived from specific detrimental SNPs in the OLR-1 gene, such as the IVS4-14 A/G evaluated in our study, strongly exerts pro-atherothrombotic effects via disturbances of ox-LDL metabolism [23]. Indeed ox-LDL is the most active pro-atherogenic and pro-thrombotic form of lipoproteins and the process of lipid peroxidation is induced mainly by inflammatory stimuli [22, 30–33]. Therefore, in this study we have also performed a cross-sectional analysis of a selected pro-inflammatory/pro-oxidative biochemical profile during TKIs treatment. Firstly, we provided biochemical evidence of a pro-atherothrombotic profile in patients on nilotinib treatment, as highlighted by the increased sCD40L levels, and a concurrent (and correlated) enhancement of thrombin generation as compared to imatinib treated patients and matched controls. Indeed sCD40L increase was significantly associated with enhanced lipid peroxidation, as reflected by higher ox-LDL levels, suggesting that lipid peroxidation can be a relevant molecular link between inflammation and thrombosis [33]. Secondly, systemic inflammation also appeared to be enhanced, as reflected by higher hs-CRP levels in nilotinib treated subjects, although not reaching a statistical between-group significance. The evidence of an underlying pro-inflammatory state was also documented by the reduction of the anti-inflammatory cytokine IL-10 in the nilotinib cohort. In fact, the reduction of IL-10 has been described as a pro-atherogenic inflammatory condition [34] whereas hs-CRP is not a universal predictor of CV risk [35] and its levels are strictly related to IL-6 rather than other cytokines production [36]. Our data also showed a significant increase of ox-LDL levels in the nilotinib cohort of patients; this may not be due to higher circulating LDL levels but to an increased pro-oxidative, inflammatory- and genetically-driven state [37]. The finding of an increased ox-LDL level is of interest because it is independently associated with atherothrombotic events, regardless of classical parameters of lipid profile. In the general population, ox-LDL levels improved the reclassification capacity of Framingham-derived CAD risk functions [31, 32], however in our experimental model we could not estimate CV risk with this or other specific algorithms [38, 39] due to the lack of baseline data. Further evidence from specifically-designed prospective studies are needed to better clarify the involvement of traditional risk factors clustering in the development of atherothrombotic events during TKIs. An additional suggestion emerges from our study: in nilotinib patients with ongoing treatment for CV risk factors there was no apparent positive effect of CV and anti-atherothrombotic drugs on the altered ox-LDL, sCD40L, ETP and IL-10 levels.
Increase of IL-10 levels may induce an anti-atherogenic profile of LOX-1 in terms of reduction of ox-LDL uptake and minor vascular damage [40]. Thus, we have hypothesized that the pro-atherothrombotic risk during nilotinib could arise from the combination of increased lipid peroxidation due to detrimental LOX-1 polymorphism and an imbalance in cytokine-driven inflammation, mainly exerted by the strong reduction of IL-10 levels [40]. Furthermore, the activation of LOX-1 by oxLDL induces up-regulation of monocyte chemotactic factor (MCP)-1, intercellular adhesion molecule (ICAM)-1 and vascular cell adhesion molecule (VCAM)-1 [25, 41]. In this regard, Valent et al. suggest that other mechanisms potentially are involved in vascular disease during TKIs treatment [14]. They demonstrated that high levels of VCAM-1 molecules are expressed in vascular cells in nilotinib treated mice, providing a possible nilotinib “off target” effect [14].
If confirmed, this direct effect could be involved in the acceleration of plaque formation and/or destabilization and could synergize with the endothelial dysfunction induced by the altered LOX-1 activity and inflammatory state [25, 41].
CONCLUSIONS
The combination of genetic and biochemical data here reported may provide a possible clue to unravel the underlying mechanism of atherosclerotic vascular events occurring in some nilotinib treated patients. To further confirm our pathogenic hypothesis we started a large multicenter prospective study in CP CML patients treated with any of the first line approved TKI in which pro-atherothrombotic genetic status, traditional CV risk factors and biochemical profile, will be evaluated from baseline and monitored during treatment. The ultimate goal of this study is to provide a genetic and biochemical tool that will aid clinical evaluation in identifying patients with relevant atherothrombotic risk earlier and better, in order to offer them the most effective, yet safest, personalized TKI treatment.
MATERIAL AND METHODS
Patient population
Between May 2013 and October 2013, all consecutive patients with CP-CML referring to our out-patient clinic for a routine follow-up visit during imatinib or nilotinib treatment, with CCyR and with various degree of molecular response, were recruited for the study. Written informed consent was obtained in all subjects. Each participant was evaluated for: presence and onset-time of traditional CV risk factors (diabetes mellitus, dyslipidaemia, arterial hypertension, BMI, smoking habit, family history for vascular events); concurrent medications; presence of PAOD and other CV events occurring during TKIs treatment. An atherothrombotic event was taken into consideration if any of the following occurred: PAOD, acute cardiac syndrome, atherosclerotic cerebral ischemia and carotid atherosclerosis (stenosis > 50%). At the same time peripheral blood and serum samples were obtained to assess the following: intron 4 IVS4-14 A > G polymorphisms of OLR1 (rs3736235), distribution of genotypes AA (low risk), AG and GG (high risk); sCD40L, ox-LDL, IL-6, IL-10, TNF-α, hs-CRP, ETP, complete lipid profile (including LDL calculated by the Friedewald's formula) and fasting plasma glucose.
Genetic analysis
Genomic DNA from peripheral blood leukocytes was extracted using the NucleoSpin Blood QuickPure Kit (MACHEREY-NAGEL). All samples were genotyped for the OLR1/Intron4 IVS4-14 A > G polymorphism by allelic discrimination assay using the TaqMan SNP Genotyping Assay (rs3736235; Applied Biosystem)
Biochemical analysis
Plasma samples for biochemical analysis were harvested after centrifugation within two hours from blood collection and immediately stored at −80°C until processing.
Endogenous thrombin potential (ETP)
ETP was measured in platelet-poor plasma using a commercially available assay (Siemens, Marburg, Germany) in a BCS-XP System (Siemens, Marburg, Germany) according to manufacturer instructions. Coagulation activation was initiated by incubation of plasma with phospholipids, human recombinant tissue factor (Innovin; Siemens, Marburg, Germany), and calcium ions in the absence of thrombomodulin. The concentration of phospholipids and tissue factor is confidential to the manufacturer. Thrombin generation and subsequent inactivation was recorded by monitoring conversion of a specific slow reacting chromogenic substrate at a wavelength of 405 nm over time.
Soluble CD40 ligand
sCD40L was evaluated according to manufacturer instructions as previously reported and specific recommendations regarding the appropriate specimen and preparation for laboratory evaluation by Platinum ELISA kits from eBioscience, Ltd. (Hatfield, Ireland, United Kingdom) were used.
IL-6, TNF alpha, IL-10, ox-LDL
According to each manufacturer's specifications, IL-6 and TNF-α plasma levels were determined by using the high sensitivity Quantikine ELISA assays (R&D systems, Minneapolis, Minnesota); the Platinum ELISA kit from eBioscience, Ltd. (Hatfield, Ireland, United Kingdom) was employed for IL-10; oxLDL levels were determined by using ox-LDL Immundiagnostik AG - Bensheim, Germany ELISA Kit.
High-sensitivity C-reactive protein
Serum hs-CRP levels were measured using an available high sensitivity-CRP commercial kit (Siemens, CardioPhase, Marburg, Germany) based on an immunonephelometry assay.
ELISA equipment
Each ELISA test was performed on a Bio-Rad iMark Microplate Absorbance Reader (Biorad, Hercules, CA) except for sCD40, IL-10 and ox-LDL assays that were done on DSX automatized processor (DSX™, Bouty-Technogenetics Milan, Italy), provided by a specific software to perform the entire analytical process. All ELISA samples were assayed in duplicates.
Statistical analysis
The Cox proportional-hazards modelling was used to evaluate the putative relations among end-points and the nonparametric data such as the genetic trait in the whole cohort of 110 subsequent CP CML patients in CCyR. For this purpose, the calculated number to detect a significant difference in parametric and not parametric variables, including genetic trait, evaluated, with 90% power at p = 0.01 was 96, if at least 14 events would occur. Accordingly, the statistical model consisting of a formal test for interaction was employed to determine the putative relation for each single variable. Final validation of data was assessed by a resampling technique (exact tests in SPSS 2003 module) and discrimination analysis by the Hosmer-Lemeshow method assuming a p < 0.05 as indicating a statistical significance. Furthermore, assuming a putative 10% difference in the frequency of events in association with each analyzed variable (single or cluster), according to previous data [42] and reported incidence and prevalence of PAOD in the general Caucasian population affected by traditional CV risk factors stratified for age [43], the calculated sample size to detect a significant difference in the genetic variables evaluated, with 90% power at p = 0.01, was 51 for each putative arm emerging from the whole cohort analysis.
To estimate putative differences in the levels of each single biochemical variable we employed the Mann Whitney U-test and the Wilcoxon test for comparisons between and within groups. The Kendall rank correlation coefficient was used to measure the relationship among measurable variables. Differences in genotype and allele proportion of the population and differences between observed and expected genotype frequencies (assuming Hardy-Weinberg equilibrium) were evaluated by χ2 test of independence. All calculations were performed using the SPSS library version 13 (SPSS Inc. Chicago, IL).
SUPPLEMENTARY MATERIALS TABLES
Acknowledgments
The authors would like to thank Dr. Carlo Scapellato, Dr. Lucia Terzuoli and Dr. Antonella Tabucchi of the Azienda Ospedaliera Universitaria Senese (Siena, Italy) for their support in the biochemical tests and Dr. Ruggiero Mango of Tor Vergata University (Rome, Italy) for his expertise, technical support and data revising for LOX-1 polymorphism analysis.
Footnotes
CONFLICTS OF INTEREST
EA has consultancy and advisory board with Novartis, BMS, Pfizer and Ariad. The authors declare that they have no conflict of interest.
GRANT SUPPORT
The study was partially founded by a grant from Sienail onlus (Associazione Italiana Leucemie, Linfomi e Mieloma, sezione di Siena)
REFERENCES
- 1.Kalmanti L, Saussele S, Lauseker M, Müller MC, Dietz CT, Heinrich L, Hanfstein B, Proetel U, Fabarius A, Krause SW, Rinaldetti S, Dengler J, Falge C, et al. Safety and efficacy of imatinib in CML over a period of 10 years: data from the randomized CML-study IV. Leukemia. 2015;29:1123–32. doi: 10.1038/leu.2015.36. [DOI] [PubMed] [Google Scholar]
- 2.Breccia M, Colafigli G, Molica M, Alimena G. Adverse events associated with tyrosine kinase inhibitors for the treatment of chronic myeloid leukemia. Expert Opin Drug Saf. 2016;16:1–9. doi: 10.1517/14740338.2016.1145654. [DOI] [PubMed] [Google Scholar]
- 3.Caldemeyer L, Dugan M, Edwards J, Akard L. Long-Term Side Effects of Tyrosine Kinase Inhibitors in Chronic Myeloid Leukemia. Curr Hematol Malig Rep. 2016;11:71–9. doi: 10.1007/s11899-016-0309-2. [DOI] [PubMed] [Google Scholar]
- 4.Cortes J, Mauro M, Steegmann JL, Saglio G, Malhotra R, Ukropec JA, Wallis NT. Cardiovascular and pulmonary adverse events in patients treated with BCR-ABL inhibitors: Data from the FDA Adverse Event Reporting System. Am J Hematol. 2015;90:66–72. doi: 10.1002/ajh.23938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chai-Adisaksopha C, Lam W, Hillis C. Major arterial events in patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors: a meta-analysis. Leuk Lymphoma. 2015;20:1–11. doi: 10.3109/10428194.2015.1091929. [DOI] [PubMed] [Google Scholar]
- 6.Moslehi JJ, Deininger M. Tyrosine Kinase Inhibitor-Associated Cardiovascular Toxicity in Chronic Myeloid Leukemia. J Clin Oncol. 2015;33:4210–8. doi: 10.1200/JCO.2015.62.4718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Aichberger KJ, Herndlhofer S, Schernthaner GH, Schillinger M, Mitterbauer-Hohendanner G, Sillaber C, Valent P. Progressive peripheral arterial occlusive disease and other vascular events during nilotinib therapy in CML. Am J Hematol. 2011;86:533–9. doi: 10.1002/ajh.22037. [DOI] [PubMed] [Google Scholar]
- 8.Le Coutre P, Rea D, Abruzzese E, Dombret H, Trawinska MM, Herndlhofer S, Dörken B, Valent P. Severe peripheral arterial disease during nilotinib therapy. J Natl Cancer Inst. 2011;103:1347–8. doi: 10.1093/jnci/djr292. [DOI] [PubMed] [Google Scholar]
- 9.Le Coutre P, Hughes TP, Mahon FX, Kim DW, Steegmann JL, Shah NP, Wallis N, Cortes J. Arterial occlusive disease (PAOD) in patients (Pts) receiving dasatinib: experience across multiple clinical trials. Blood ASH annual meeting abstract. 2013;122:1489. [Google Scholar]
- 10.Giles FJ, Mauro MJ, Hong F, Ortmann CE, McNeill C, Woodman RC, Hochhaus A, le Coutre PD, Saglio G. Rates of peripheral arterial occlusive disease in patients with chronic myeloid leukemia in the chronic phase treated with imatinib, nilotinib, or non-tyrosine kinase therapy: a retrospective cohort analysis. Leukemia. 2013;27:1310–5. doi: 10.1038/leu.2013.69. [DOI] [PubMed] [Google Scholar]
- 11.Kim TD, Rea D, Schwarz M, Grille P, Nicolini FE, Rosti G, Levato L, Giles FJ, Dombret H, Mirault T, Labussière H, Lindhorst R, Haverkamp W, et al. Peripheral artery occlusive disease in chronic phase chronic myeloid leukemia patients treated with nilotinib or imatinib. Leukemia. 2013;27:1316–21. doi: 10.1038/leu.2013.70. [DOI] [PubMed] [Google Scholar]
- 12.Gora-Tybor J, Medras E, Calbecka M, Kolkowska-Leśniak A, Ponikowska-Szyba E, Robak T, Jamroziak K. Real-life comparison of severe vascular events and other non-hematological complications in patients with chronic myeloid leukemia undergoing second-line nilotinib or dasatinib treatment. Leuk Lymphoma. 2015;5:1–6. doi: 10.3109/10428194.2014.994205. [DOI] [PubMed] [Google Scholar]
- 13.Valent P, Hadzijusufovic E, Schernthaner GH, Wolf D, Rea D, le Coutre P. Vascular safety issues in CML patients treated with BCR/ABL1 kinase inhibitors. Blood. 2015;125:901–6. doi: 10.1182/blood-2014-09-594432. [DOI] [PubMed] [Google Scholar]
- 14.Hadzijusufovic E, Albrecht-Schgoer K, Huber K, Grebien F, Eisenwort G, Schgoer W, Kaun C, Herndlhofer S, Theurl M, Cerny-Reiterer S, Hoermann G, Sperr WR, Rix U. Further Evaluation of Pro-Atherogenic and Anti-Angiogenic Effects of Nilotinib in Mice and in Patients with Ph-Chromosome+ CML. Blood. 2014:124. (abstract 1800) [Google Scholar]
- 15.Quintás-Cardama A, Kantarjian H, Cortes J. Nilotinib-associated vascular events. Clin Lymphoma Myeloma Leuk. 2012;12:337–40. doi: 10.1016/j.clml.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 16.Hekmatjou H, Roboz GJ, Ritchie EK, Lee S, Desai P, Scandura JM, Allen-Bard S, Christos P, Silver RT, Feldman EJ. Arterial Thrombotic Complications Are Uncommon in Patients without Cardiovascular Risk Factors and Occur at Equivalent Rates in Chronic Myeloid Leukemia (CML) Patients Treated with Imatinib and Nilotinib. Blood. 2014:124. (abstract 1811) [Google Scholar]
- 17.Gilbert J, Deplano S, Szydlo R, Palanicawandar R, Gerrard G, Foroni L, Apperley JF, Milojkovic D. Incidence of Vascular Thrombotic Events in 183 Consecutive Patients Treated with Nilotinib: A Single Centre Experience. Blood. 2014:124. (abstract 3147) [Google Scholar]
- 18.Breccia M1, Molica M, Zacheo I, Serrao A, Alimena G. Application of systematic coronary risk evaluation chart to identify chronic myeloid leukemia patients at risk of cardiovascular diseases during nilotinib treatment. Ann Hematol. 2015;94:393–7. doi: 10.1007/s00277-014-2231-9. [DOI] [PubMed] [Google Scholar]
- 19.Bondon-Guitton E, Combret S, Pérault-Pochat MC, Stève-Dumont M, Bagheri H, Huguet F, Despas F, Pathak A, Montastruc JL. Cardiovascular risk profile of patients with peripheral arterial occlusive disease during nilotinib therapy. Target Oncol. 2016;11:549–52. doi: 10.1007/s11523-016-0417-x. [DOI] [PubMed] [Google Scholar]
- 20.Mirault T, Rea D, Azarine A, Messas E. Rapid onset of peripheral artery disease in a chronic myeloid leukemia patient without prior arterial disorder: direct relationship with nilotinib exposure and clinical outcome. Eur J Haematol. 2015;94:363–7. doi: 10.1111/ejh.12367. [DOI] [PubMed] [Google Scholar]
- 21.Libby P. Inflammation in atherosclerosis. Arterioscler Thromb Vasc Biol. 2012;32:2045–51. doi: 10.1161/ATVBAHA.108.179705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wolf D, Stachon P, Bode C, Zirlik A. Inflammatory mechanisms in atherosclerosis. Hamostaseologie. 2014;34:63–71. doi: 10.5482/HAMO-13-09-0050. [DOI] [PubMed] [Google Scholar]
- 23.Mehta JL, Khaidakov M, Hermonat PL, Mitra S, Wang X, Novelli G, Sawamura T. LOX-1: a new target for therapy for cardiovascular diseases. Cardiovasc Drugs Ther. 2011;25:495–500. doi: 10.1007/s10557-011-6325-5. [DOI] [PubMed] [Google Scholar]
- 24.Predazzi IM, Martinez-Labarga C, Vecchione L, Mango R, Ciccacci C, Amati F, Ottoni C, Crawford MH, Rickards O, Romeo F, Novelli G. Population differences in allele frequencies at the OLR1 locus may suggest geographic disparities in cardiovascular risk events. Ann Hum Biol. 2010;37:137–49. doi: 10.3109/03014460903393857. [DOI] [PubMed] [Google Scholar]
- 25.Lubrano V, Balzan S. Roles of LOX-1 in microvascular dysfunction. Microvasc Res. 2016;18:30012–7. doi: 10.1016/j.mvr.2016.02.006. [DOI] [PubMed] [Google Scholar]
- 26.Emerging Risk Factors Collaboration. Kaptoge S, Di Angelantonio E, Pennells L, Wood AM, White IR, Gao P, Walker M, Thompson A, Sarwar N, Caslake M, Butterworth AS, Amouyel P, Assmann G, et al. C-reactive protein, fibrinogen, and cardiovascular disease prediction. N Engl J Med. 2012;367:1310–20. doi: 10.1056/NEJMoa1107477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Eichinger S, Hron G, Kollars M, Kyrle PA. Prediction of recurrent venous thromboembolism by endogenous thrombin potential and D-dimer. Clin Chem. 2008;54:2042–8. doi: 10.1373/clinchem.2008.112243. [DOI] [PubMed] [Google Scholar]
- 28.Riondino S, Martini F, La Farina F, Spila A, Guadagni F, Ferroni P. Increased plasma levels of soluble CD40 ligand correlate with platelet activation markers and underline the need for standardized pre-analytical conditions. Clin Biochem. 2010;43:666–70. doi: 10.1016/j.clinbiochem.2009.12.021. [DOI] [PubMed] [Google Scholar]
- 29.Tefferi A. Nilotinib treatment-associated accelerated atherosclerosis: when is the risk justified? Leukemia. 2013;27:1939–40. doi: 10.1038/leu.2013.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mango R, Predazzi IM, Romeo F, Novelli G. LOX-1/LOXIN: the yin/yang of atheroscleorosis. Cardiovasc Drug Ther. 2011;25:489–94. doi: 10.1007/s10557-011-6333-5. [DOI] [PubMed] [Google Scholar]
- 31.Linna M, Ahotupa M, Löppönen MK, Irjala K, Vasankari T. Circulating oxidised LDL lipids, when proportioned to HDL-c, emerged as a risk factor of all-cause mortality in a population-based survival study. Age Ageing. 2013;42:110–3. doi: 10.1093/ageing/afs074. [DOI] [PubMed] [Google Scholar]
- 32.Gómez M, Vila J, Elosua R, Molina L, Bruguera J, Sala J, Masià R, Covas MI, Marrugat J, Fitó M. Relationship of lipid oxidation with subclinical atherosclerosis and 10-year coronary events in general population. Atherosclerosis. 2014;232:134–40. doi: 10.1016/j.atherosclerosis.2013.10.026. [DOI] [PubMed] [Google Scholar]
- 33.Santilli F, Basili S, Ferroni P, Davì G. CD40/CD40L system and vascular disease. Intern Emerg Med. 2007;2:256–68. doi: 10.1007/s11739-007-0076-0. [DOI] [PubMed] [Google Scholar]
- 34.Kleemann R, Zadelaar S, Kooistra T. Cytokines and atherosclerosis: a comprehensive review of studies in mice. Cardiovasc Res. 2008;79:360–76. doi: 10.1093/cvr/cvn120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sever PS, Poulter NR, Chang CL, Hingorani A, Thom SA, Hughes AD, Welsh P, Sattar N, ASCOT Investigators Evaluation of C-reactive protein prior to and on-treatment as a predictor of benefit from atorvastatin: observations from the Anglo-Scandinavian Cardiac Outcomes Trial. Eur Heart J. 2012;33:486–94. doi: 10.1093/eurheartj/ehr262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Blake GJ, Ridker PM. C-reactive protein: A surrogate risk marker or mediator of atherothrombosis? Am J Physiol Regul Integr Comp Physiol. 2003;285:R1250–2. doi: 10.1152/ajpregu.00227.2003. [DOI] [PubMed] [Google Scholar]
- 37.Violi F, Pignatelli P. Clinical Application of NOX Activity and Other Oxidative Biomarkers in Cardiovascular Disease: A Critical Review. Antioxid Redox Signal. 2015;23:514–32. doi: 10.1089/ars.2013.5790. [DOI] [PubMed] [Google Scholar]
- 38.Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) the European Atherosclerosis Society (EAS) Catapano AL. Reiner Z, De Backer G, Graham I, Taskinen MR, Wiklund O, Agewall S, Alegria E, Chapman MJ, Durrington P, Erdine S, Halcox J, Hobbs R, et al. ESC/EAS Guidelines for the management of dyslipidaemias: the Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS) Atherosclerosis. 2011;217(Suppl 1):S1–44. doi: 10.1016/j.atherosclerosis.2011.06.012. [DOI] [PubMed] [Google Scholar]
- 39.Stone NJ, Robinson J, Lichtenstein AH, Bairey Merz CN, Blum CB, Eckel RH, Schwartz JS, Goldberg AC, Shero ST, Gordon D, Smith SC, Jr, Levy D, Watson K, et al. ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;2015;23:514–32. doi: 10.1016/j.jacc.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 40.Arjuman A, Chandra NC. Effect of IL-10 on LOX-1 expression, signaling and functional activity: an atheroprotective response. Diab Vasc Dis Res. 2013;10:442–51. doi: 10.1177/1479164113489042. [DOI] [PubMed] [Google Scholar]
- 41.Gorga E, Vizzardi E, Bonadei I, Piovanelli B, Della Pina P, Raddino R, Dei Cas L. LOX-1 and its implications on cardiovascular diseases a possible new perspective target on atherosclerosis. J Clin Exp Cardiol. 2013;4:1. [Google Scholar]
- 42.Fowkes FG, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, Norman PE, Sampson UK, Williams LJ, Mensah GA, Criqui MH. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet. 2013;382:1329–40. doi: 10.1016/S0140-6736(13)61249-0. [DOI] [PubMed] [Google Scholar]
- 43.Achterberg S, Cramer MJ, Kappelle LJ, de Borst GJ, Visseren FL, van der Graaf Y, Algra A, SMART study Group Patients with coronary, cerebrovascular or peripheral arterial obstructive disease differ in risk for new vascular events and mortality: the SMART study. Eur J Cardiovasc Prev Rehabil. 2010;17:424–30. doi: 10.1097/HJR.0b013e3283361ce6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.