The efficacy of the FOLFIRI (irinotecan, leucovorin, and 5‐fluorouracil) regimen was similar between elderly and nonelderly patients in both the colorectal cancer and gastric cancer cohorts. However, special attention should be paid to elderly patients because of increased risk for febrile neutropenia and asthenia.
Keywords: Irinotecan, 5‐Fluorouracil, FOLFIRI, Colorectal cancer, Gastric cancer, Elderly
Abstract
Background.
Irinotecan‐based chemotherapy is a standard backbone of therapy in patients with metastatic colorectal cancer (CRC) or gastric cancer (GC). However, there is still a paucity of information concerning the efficacy and safety of irinotecan‐based regimens in elderly patients.
Patients and Methods.
Using the patient cohort (n = 1,545) from the UGT1A1 genotype study, we compared the efficacy and safety between elderly and nonelderly patients with metastatic CRC (n = 934) or GC (n = 611) who received first‐ or second‐line FOLFIRI (irinotecan, leucovorin, and 5‐fluorouracil) chemotherapy.
Results.
Despite lower relative dose intensity in elderly patients, progression‐free survival and overall survival were similar between elderly (age ≥70 years) and nonelderly (<70 years) patients in the CRC cohort (hazard ratio [HR], 1.117; 95% confidence interval [CI], 0.927–1.345; p = .244, and HR, 0.989; 95% CI, 0.774–1.264; p = .931, respectively) and the GC cohort (HR, 1.093; 95% CI, 0.854–1.400; p = .479, and HR, 1.188; 95% CI, 0.891–1.585; p = .241, respectively). In both cohorts, febrile neutropenia (22.1% vs. 14.6% in CRC cohort and 35.2% vs. 22.5% in GC cohort) and asthenia (grade 3: 8.4% vs. 1.7% in CRC cohort and 5.5% vs. 2.9% in GC cohort) were more frequent in elderly patients. In the CRC cohort, mucositis and anorexia were more frequent in elderly patients. In the GC cohort, nausea and vomiting were less frequent in elderly patients.
Conclusion.
The efficacy of the FOLFIRI regimen was similar between elderly and nonelderly patients in both the CRC and the GC cohorts. However, special attention should be paid to elderly patients because of increased risk for febrile neutropenia and asthenia.
Implications for Practice.
The efficacy of FOLFIRI (irinotecan, leucovorin, and 5‐fluorouracil) chemotherapy in elderly patients with metastatic colorectal cancer or gastric cancer was similar to that in nonelderly patients. However, special attention should be paid to elderly patients because of the increased risk for febrile neutropenia and asthenia. These data suggest that the FOLFIRI regimen could be considered as a standard backbone of therapy in elderly patients with metastatic colorectal cancer or gastric cancer and that the clinical decision between doublet and singlet chemotherapy may not be based solely on age. However, the data require further assessment of frailty and performance status.
Introduction
Irinotecan‐based chemotherapy is an established standard backbone of therapy in patients with metastatic colorectal cancer (CRC) or gastric cancer (GC). Many clinical studies have demonstrated that irinotecan‐based regimens are active in both CRC and GC [1], [2], [3], [4], [5], [6]. The FOLFIRI regimen, consisting of irinotecan, leucovorin, and 5‐fluorouracil (5‐FU), has been widely used to treat patients with these diseases [1], [3], [4], [6]. However, there is still concern as to whether the results from these studies can be applied to the elderly subset of patients because the experience of the FOLFIRI regimen in elderly patients is limited.
For metastatic CRC, a previous analysis of patients enrolled in randomized controlled trials suggested that patients older than 70 years achieved similar benefits from irinotecan‐containing chemotherapy as younger patients, and the toxicity profile was also similar [7]. In addition, another study of irinotecan plus fluoropyrimidine combination chemotherapy showed a similar efficacy and toxicity between patients older than 70 years and those younger than 70 years [8]. However, a recent study demonstrated a conflicting result that the addition of irinotecan to infusional 5‐FU‐based chemotherapy did not significantly increase the clinical outcome in CRC patients older than 75 years of age [9]. Therefore, it is still under debate whether irinotecan plus fluoropyrimidine doublet chemotherapy could be a standard mainstay for elderly patients with metastatic CRC. In metastatic GC, previous studies support that an irinotecan‐based regimen could be an effective therapeutic option in elderly patients [10], [11]. However, the experience of irinotecan‐based chemotherapy is still limited in this subset of patients.
This study aimed to compare the efficacy and toxicity of FOLFIRI regimen as first‐line or second‐line chemotherapy between elderly patients and nonelderly patients with metastatic CRC and GC. We analyzed a patient cohort from a previous study that aimed to investigate the association between irinotecan toxicity and UGT1A1 polymorphisms in metastatic CRC and GC patients treated with the FOLFIRI regimen [12].
Materials and Methods
Patient Population
This study analyzed the aforementioned patient population (n = 1,545), which consisted of 934 CRC patients and 611 GC patients [12]. The cohort prospectively enrolled patients with metastatic CRC and GC who had not been treated previously or in whom first‐line chemotherapy had failed. Between January 2009 and April 2012, a total of 33 hospitals in Korea participated in this study. Patients were required to have an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–2. In addition, eligibility required adequate hematologic, renal, and hepatic function, defined as a neutrophil count ≥ 1.5 × 109/L, a platelet count ≥ 100 × 109/L, total bilirubin ≤ 1.5 × upper limit of normal (ULN), aspartate aminotransferase and alanine aminotransferase levels ≤ 2.5 × ULN, alkaline phosphatase levels ≤ 2.5 × ULN, and serum creatinine levels ≤ 1.5 × ULN. Patients were excluded if they were previously treated with irinotecan or had severe active infection, chronic diarrhea, paralytic ileus, pulmonary fibrosis, or interstitial pneumonia at the time of study entry. Patients who were pregnant or breastfeeding were also excluded. UGT1A1 *6 and *28 polymorphisms were analyzed in all patients in the original study [12]. Patients who had one polymorphism of *6 or *28 (i.e., *1/*6 or *1/*28) were classified as one defective allele (DA) carriers. Those who had two polymorphisms of *6 or *28 (i.e., *6/*6, *28/*28, or *6/*28) were defined as two DA carriers.
FOLFIRI Regimens
Irinotecan at a dose of 150 mg/m2 or 180 mg/m2 of body surface area (BSA) was administered on day 1. The dose of irinotecan was chosen according to the physician's preference without any guidance of the UGT1A1 genotype. Two different 5‐FU and leucovorin regimens were permitted in this study. The original de Gramont regimen comprised l‐leucovorin at a dose of 200 mg/m2 or dl‐leucovorin at a dose of 400 mg/m2 for 2 hours on days 1 and 2 and a 5‐FU bolus at a dose of 400 mg/m2, followed by 5‐FU infusion at a dose of 600 mg/m2 for 22 hours on days 1 and 2 [13]. The simplified de Gramont regimen consisted of a 2‐hour infusion of l‐leucovorin at 200 mg/m2 or dl‐leucovorin at 400 mg/m2 on day 1, followed by 5‐FU bolus at 400 mg/m2 and 5‐FU 46‐hour infusion at 2,400 mg/m2 [1]. The treatment was repeated every 2 weeks. Initial dose reduction and subsequent dose reduction and delay were based on the study protocol (supplemental online Table 1). For nonhematologic toxicity, 5‐FU dose modifications were based on the discretion of the treating physician. Relative dose intensity (RDI) (percentage) was calculated as follows: [(cumulative actual dose of a chemotherapeutic agent per BSA) × (planned treatment duration)]/[(cumulative planned dose per BSA) × (actual treatment duration)]. Planned dose of each chemotherapeutic agent was 180 mg/m2 for irinotecan, 400 mg/m2 for 5‐FU bolus, and 2,400 mg/m2 for 5‐FU infusion. Planned treatment duration was 14 days per cycle. The mean RDI for each cohort was calculated by using the mean RDIs per patient.
Study Objectives
The primary objective of this study was to compare the efficacy of the FOLFIRI regimen among age groups of patients in both the CRC and GC cohorts. Our primary hypothesis was that progression‐free survival (PFS) and overall survival (OS) in elderly patients (age ≥ 70 years) would be noninferior to those in nonelderly patients (<70 years) with a margin of 1.50 for the upper boundary of the 95% confidence interval (CI) of the hazard ratio (HR). In addition, the baseline demographic characteristics and treatment efficacy data were further divided according to treatment lines and the following three age groups: patients younger than 70 years, those aged 70–74 years, and those older than 75 years. The secondary aim was to analyze the toxicity profile of the FOLFIRI regimen among age groups, according to primary disease.
Acquisition of Clinical Data
Treatment response was evaluated by the Response Evaluation Criteria in Solid Tumors, version 1.0 [14]. PFS was calculated from the initiation of chemotherapy to documented disease progression or death from any cause. OS was calculated from the start of chemotherapy to death from any cause. Adverse events were assessed in all patients by using the National Cancer Institute's Common Terminology Criteria for Adverse Events version 3.0.
Statistical Analysis
Statistical analysis of categorical variables was performed by using Pearson's chi‐square test or Fisher's exact test, as appropriate. Analysis of variance test was used to compare three different groups consisting of continuous variables. Post hoc power calculation for a noninferiority log rank test was performed by using PASS software, version 11 (NCSS, LLC, Kaysville, UT, https://www.ncss.com). The median PFS and OS were calculated by using the Kaplan‐Meier method. Survival data were compared by using the log‐rank test. A Cox proportional hazard model was used to perform multivariable analysis and to calculate HRs using the survival data. The multivariable analyses were performed to adjust the effects of other baseline clinical variables, including UGT1A1 genotype, sites of metastatic disease, prior adjuvant therapy, and treatment line. All statistical tests were two‐sided, with significance defined as p < .05. All analyses were performed by using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, http:/www.sas.com) and R 3.2 (R Foundation, Vienna, Austria).
Ethical Considerations
Signed informed consent was obtained from all patients before study entry. The original trial was registered at ClinicalTrials.gov as number NCT01271582. This study protocol was reviewed and approved by the institutional review board of each participating hospital and conducted in accordance with the precepts established by the Helsinki Declaration.
Results
Patient Characteristics
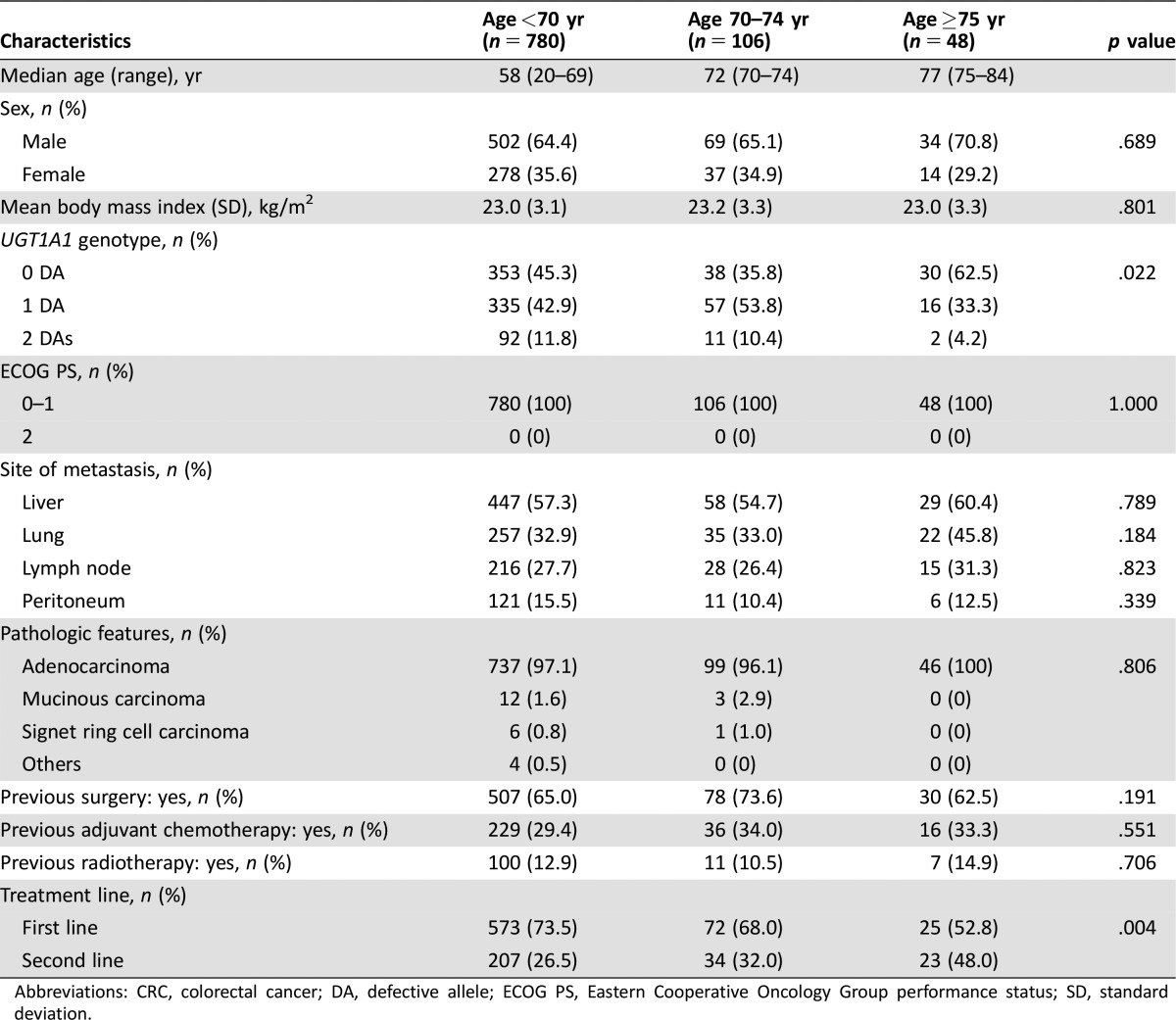
The baseline characteristics of patients in the CRC cohort are summarized in Table 1. The numbers of patients were as follows: 780 younger than 70 years, 106 aged 70–74 years, and 48 older than 75 years. Sex, body mass index, ECOG PS, site of metastasis, and histologic features were not significantly different among the age groups. The distribution of the UGT1A1 genotype, however, was significantly different among the age groups: Patients older than 75 years were less likely to have DAs in UGT1A1 gene (p = .022). Previous treatment was not significantly different among the age groups. In elderly patients, the proportion of those undergoing the FOLFIRI regimen as second‐line therapy was significantly higher compared with nonelderly patients (p = .004).
Table 1. Baseline characteristics of patients in the colorectal cancer cohort.
Abbreviations: CRC, colorectal cancer; DA, defective allele; ECOG PS, Eastern Cooperative Oncology Group performance status; SD, standard deviation.
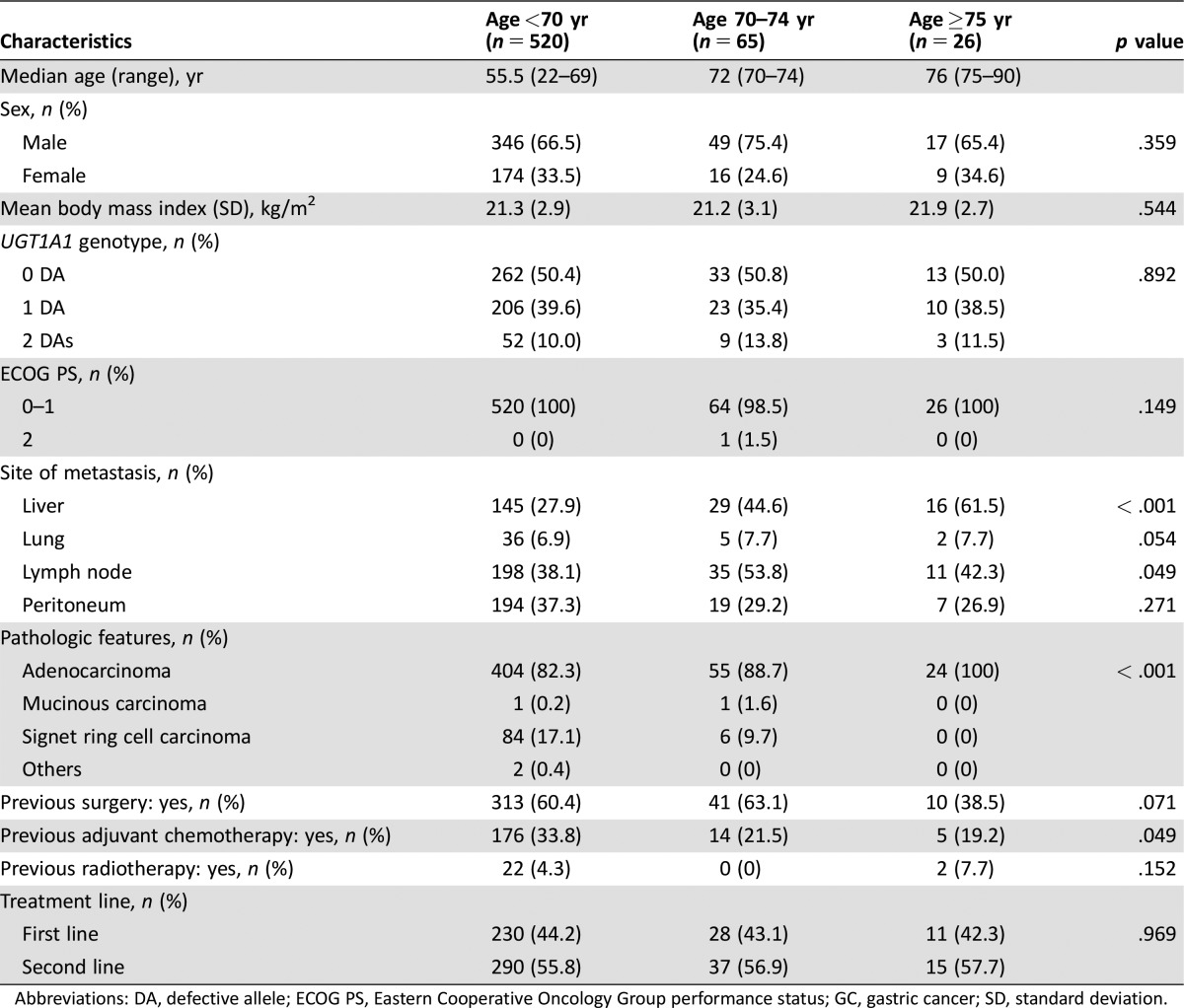
The baseline characteristics of patients in the GC cohort are listed in Table 2. The numbers of patients were as follows: 520 younger than 70 years, 65 aged 70–74 years, and 26 older than 75 years. Sex, body mass index, UGT1A1 genotype, and ECOG PS were similar among the age groups. The proportion of patients with liver metastasis was significantly higher in elderly patients (p <0.001). In contrast, signet ring cell carcinoma histologic type was significantly more frequent in nonelderly patients (p < .001). Elderly patients were less likely to receive surgery (p = .071) or adjuvant chemotherapy (p = .049) before study entry. In the GC cohort, the treatment lines of FOLFIRI chemotherapy were well balanced among the age groups.
Table 2. Baseline characteristics of patients in the gastric cancer cohort.
Abbreviations: DA, defective allele; ECOG PS, Eastern Cooperative Oncology Group performance status; GC, gastric cancer; SD, standard deviation.
RDI
In the CRC cohort, the RDI of irinotecan was significantly higher in nonelderly patients than elderly patients (p < 0.001; supplemental online Table 2A). In patients younger than 70 years, the mean ± standard deviation RDI of irinotecan was 80.9% ± 14.5%, whereas patients aged 70–74 years received 74.5% ± 15.4% dose, and those older than 75 years were treated with 75.4% ± 16.3% dose. In addition, patients younger than 70 years received significantly higher doses of infusional 5‐FU (p < .001): 71.8% ± 24.8% (<70 years), 66.6% ± 26.0% (70–74 years), and 50.3% ± 25.2% (≥75 years). In contrast, the RDI of the 5‐FU bolus was not significantly different among the age groups (p = .959).
In the GC cohort, the RDI of irinotecan tended to be higher in the younger age group (p = .060): 76.2% ± 14.3% (<70 years), 72.3% ± 12.8% (70–74 years), and 72.7% ± 15.4% (≥75 years) (supplemental online Table 2B). The RDI of the 5‐FU bolus was not significantly different among the age groups (p = .647). The RDIs of infusional 5‐FU among the age groups were as follows: 55.8% ± 27.1% (<70 years), 43.5% ± 22.8% (70–74 years), and 56.0% ± 27.2% (≥75 years) (p = .002).
Post Hoc Power Calculation for PFS and OS between Patients Younger and Older Than 70 Years
In the CRC cohort, a noninferiority log‐rank test of PFS between patients younger than 70 years and those older than 70 years achieved 99.4% power at a .050 significance level to detect an equivalence HR of 1.50. For OS, the statistical power was 98.3% to detect an equivalence HR of 1.50 at a .050 significance level. In the GC cohort, the statistical power of a noninferiority log‐rank test of PFS between patients younger than 70 years and those older than 70 years was 95.4% at a .050 significance level to detect an equivalence HR of 1.50. For OS, the statistical power was 93.9% to detect an equivalence HR of 1.50 at a .050 significance level.
Treatment Efficacy between Patients Younger and Older Than 70 Years
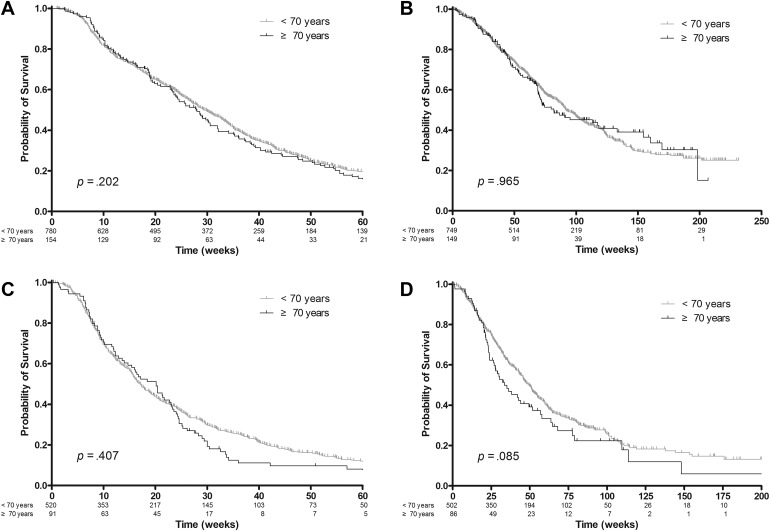
In the CRC cohort, the median follow‐up time was 69.6 weeks (range, 1.0–231.6 weeks). The treatment response was evaluable in 815 patients (87.3%). The response rate was not significantly different among the age groups (p = .720): 24.6% (<70 years) and 22.7% (≥70 years) (supplemental online Table 3A). PFS was similar between patients younger than 70 years and those older than 70 years (29.1 weeks [95% CI, 27.0–31.9 weeks] vs. 27.9 weeks [95% CI, 23.6–30.6 weeks]; p = .202; Fig. 1A). In multivariable analysis, PFS in patients older than 70 years did not differ significantly compared with those younger than 70 years (HR, 1.117 [95% CI, 0.927–1.345]; p = 0.244; supplemental online Table 4A). OS was also similar between the two age groups (91.9 weeks [95% CI, 86.0–101.7 weeks] vs. 81.9 weeks [95% CI, 69.1–133.1 weeks]; p = .965; Fig. 1B). In multivariable analysis, OS in patients older than 70 years were similar to that in those younger than 70 years (HR, 0.989 [95% CI, 0.774–1.264]; p = .931; supplemental online Table 4B).
Figure 1.
Kaplan‐Meier analysis of progression‐free survival (PFS) and overall survival (OS) according to age. (A): In the colorectal cancer (CRC) cohort, PFS was similar between patients younger than 70 years and those older than 70 years (p = .202). (B): In the CRC cohort, OS was also similar between the two age groups (p = 0.965). (C): In the gastric cancer (GC) cohort, PFS was not significantly different between the two age groups (p = .407). (D): In the GC cohort, OS tended to be inferior in elderly patients compared with nonelderly patients (p = .085).
In the GC cohort, the median follow‐up time was 36.8 weeks (range, 0.1–208.6 weeks). The treatment response was evaluable in 508 patients (83.1%). The response rate was not significantly different among the age groups (p = .749): 14.8% (<70 years) and 17.1% (≥70 years) (supplemental online Table 3B). PFS was not significantly different between the two age groups (17.0 weeks [95% CI, 15.6–18.9 weeks] vs. 20.1 weeks [95% CI, 14.0–23.0 weeks]; p = .407; Fig. 1C). In multivariable analysis, patients older than 70 years were not statistically different from those younger than 70 years in terms of PFS (HR, 1.093 [95% CI, 0.854–1.400]; p = .479; supplemental online Table 4C). OS tended to be inferior in elderly patients (33.6 weeks [95% CI, 26.6–51.9 weeks]) compared with nonelderly patients (49.3 weeks [95% CI, 44.7–54.1 weeks]) (p = .085; Fig. 1D). The multivariable analysis yielded the same result in terms of OS (HR, 1.188 [95% CI, 0.891–1.585]; p = .241; supplemental online Table 4D).
Subgroup Analysis of the CRC Cohort according to Treatment Lines and Three Age Groups
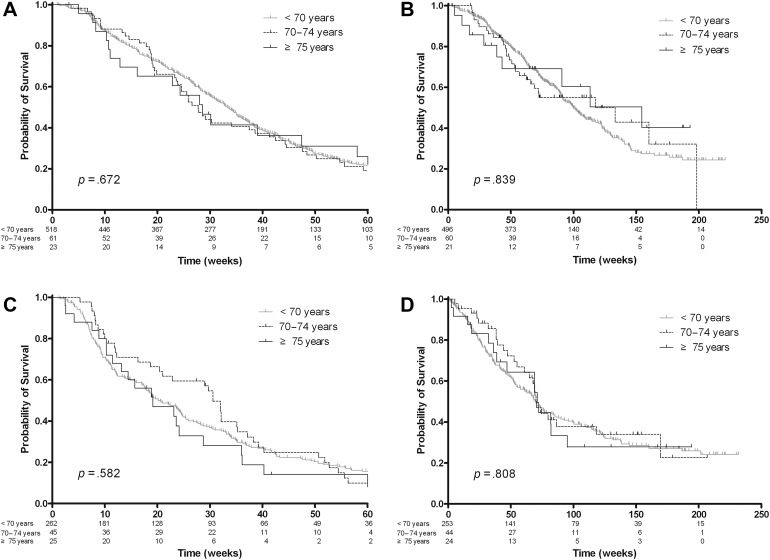
In the first‐line subgroup, the response rates were 30.4% in patients younger than 70 years, 30.2% in those aged 70–74 years, and 31.6% in those older than 75 years (p = .993). The median PFSs were 33.0 weeks (95% CI, 30.4–35.3 weeks), 27.7 weeks (95% CI, 23.3–38.6 weeks), and 28.6 weeks (95% CI, 12.9–58.0 weeks), respectively (p = .672; Fig. 2A). The median OSs were 99.7 weeks (95% CI, 91.3–109.1 weeks), 133.1 weeks (95% CI, 62.3–198.3 weeks), and 154.6 weeks (95% CI, 38.9 weeks–infinity), respectively (p = .839; Fig. 2B).
Figure 2.
Survival analysis according to treatment lines and three age groups in the colorectal cancer cohort. In the first‐line subgroup, progression‐free survival (PFS) (A) and overall survival (OS) (B) were not significantly different among the age groups (p = .672 and p = .839, respectively). In the second‐line subgroup, PFS (C) and OS (D) were also similar among the age groups (p = .582 and p = .808, respectively).
In the second‐line subgroup, the response rates were 12.6%, 13.2%, and 11.1%, respectively (p = 1.000). The median PFSs were 20.1 weeks (95% CI, 17.6–24.4 weeks), 30.6 weeks (95% CI, 20.4–35.3 weeks), and 19.1 weeks (95% CI, 11.4–28.7 weeks), respectively (p = .582; Fig. 2C). The median OSs were 69.4 weeks (95% CI, 57.6–84.0 weeks), 70.1 weeks (95% CI, 55.0–118.4 weeks), and 71.1 weeks (95% CI, 38.7–95.1 weeks), respectively (p = .808; Fig. 2D).
Subgroup Analysis of the GC Cohort according to Treatment Lines and Three Age Groups
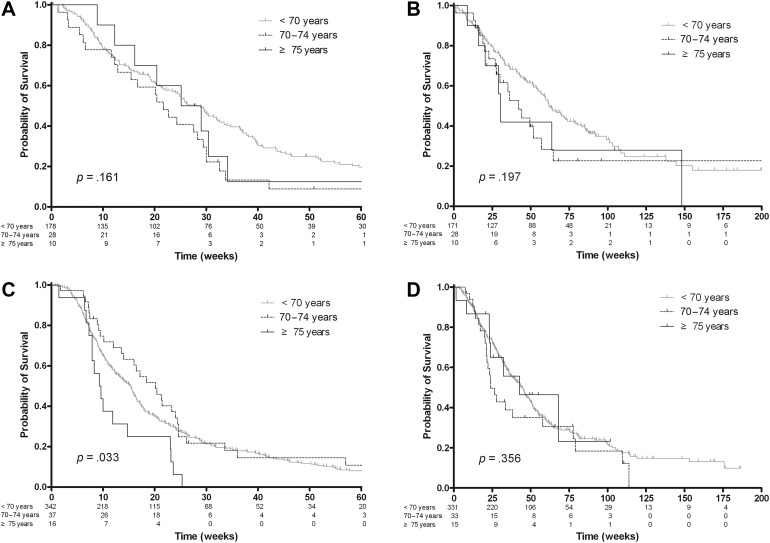
In the first‐line subgroup, the response rates were 20.5% in patients younger than 70 years, 3.3% in those aged 70–74 years, and 0% in those older than 75 years (p = .189). The median PFSs were 28.0 weeks (95% CI, 22.0–32.1 weeks), 21.7 weeks (95% CI, 12.3–29.4 weeks), and 27.1 weeks (95% CI, 8.9–34.1 weeks), respectively (p = .161; Fig. 3A). OS tended to be shorter in older patients (p = .197; Fig. 3B): The median OSs were 61.9 weeks (95% CI, 54.4–78.4 weeks), 42.1 weeks (95% CI, 26.9–57.0 weeks), and 30.4 weeks (95% CI, 8.9–148.1 weeks), respectively.
Figure 3.
Survival analysis according to treatment lines and three age groups in the gastric cancer cohort. (A): In the first‐line subgroup, progression‐free survival (PFS) was not significantly different among the age groups (p = .161). (B): In the first‐line subgroup, overall survival (OS) tended to be shorter in older patients (p = .197). (C): In the second‐line subgroup, PFS was significantly shorter in patients older than 75 years (p = .033). (D): In the second‐line subgroup, OS was similar among the age groups (p = .356).
In the second‐line subgroup, the response rates were 12.0%, 11.1%, and 38.5%, respectively (p = .032). The median PFSs were significantly shorter (p = .033; Fig. 3C) in patients older than 75 years: 15.3 weeks (95% CI, 12.6–16.1 weeks), 20.1 weeks (95% CI, 13.6–24.0 weeks), and 9.4 weeks (95% CI, 7.3–14.7 weeks), respectively. OS was similar among the age groups (p = .356; Fig. 3D): The median OSs were 44.1 weeks (95% CI, 37.6–49.3 weeks), 24.0 weeks (95% CI, 21.1–57.9 weeks), and 42.7 weeks (95% CI, 23.0–infinity weeks), respectively.
Toxicity
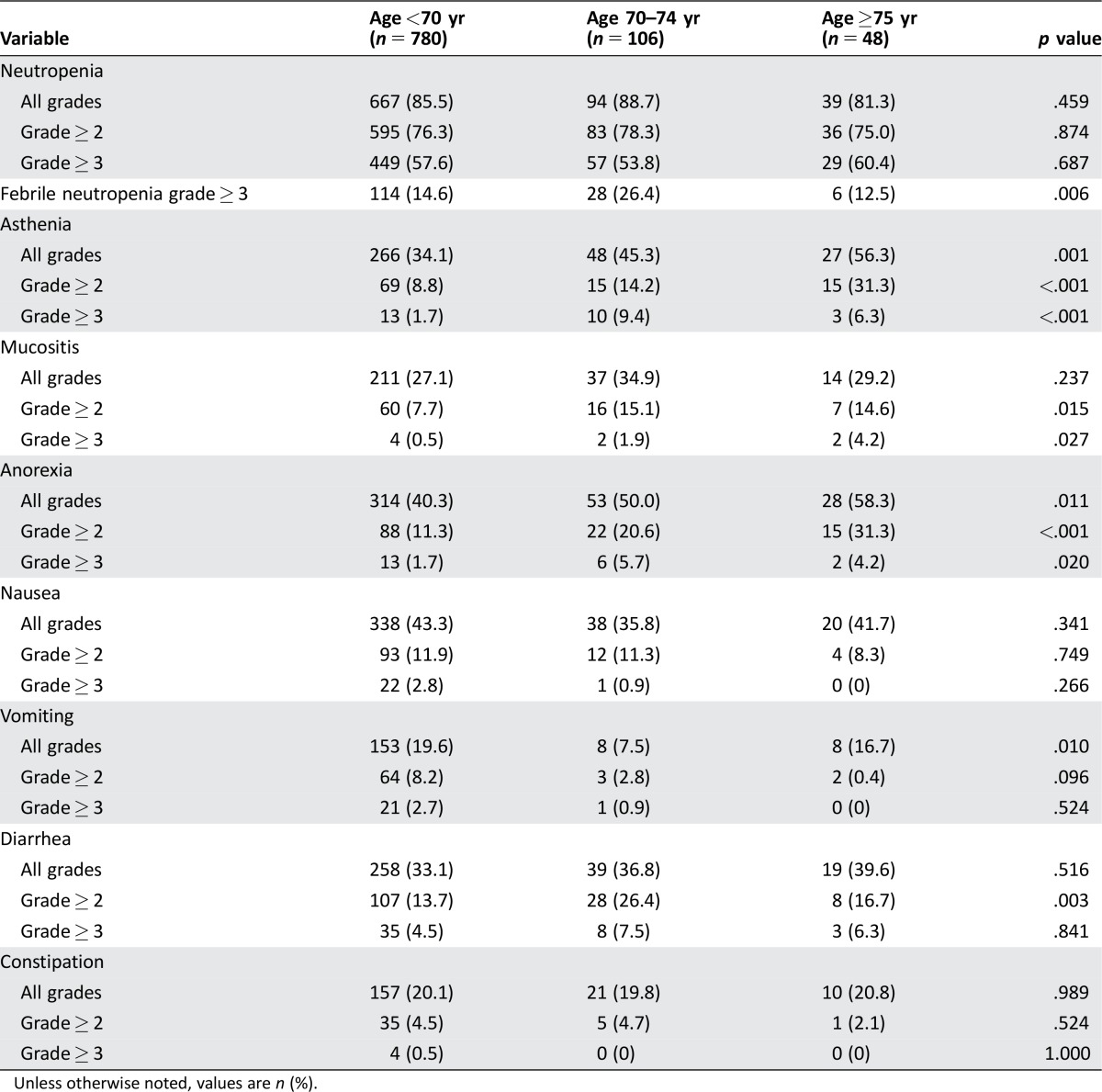
Toxicity during FOLFIRI chemotherapy in the CRC cohort is summarized in Table 3 (per patient) and supplemental online Table 5A (per treatment cycle). Febrile neutropenia was significantly more frequent in patients aged 70–74 years (p = .006). Asthenia, mucositis, and anorexia were also significantly more frequent in elderly patients. Nausea, vomiting, diarrhea, and constipation tended to be similar among the age groups.
Table 3. Treatment‐related toxicity per patient in the colorectal cancer cohort.
Unless otherwise noted, values are n (%).
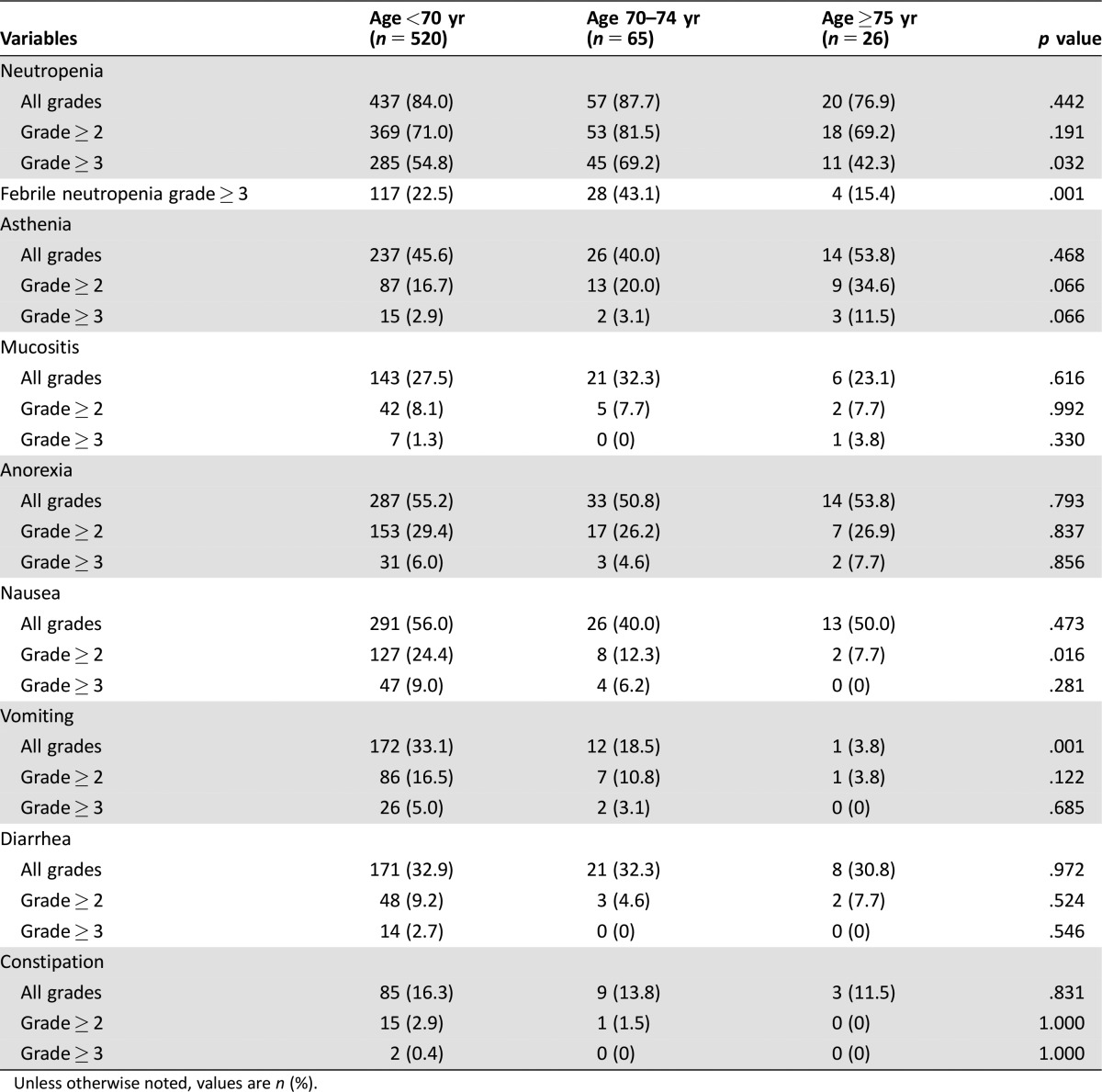
Toxicity in the GC cohort is listed in Table 4 (per patient) and supplemental online Table 5B (per treatment cycle). Neutropenia of grade 3 or more and febrile neutropenia were significantly more frequent in patients aged 70–74 years (p = .032 and p = .001, respectively). Asthenia tended to be more frequent in elderly patients. Mucositis and anorexia were similar among the age groups. Nausea and vomiting were less frequent in elderly patients. Diarrhea and constipation were not significantly different among the age groups.
Table 4. Treatment‐related toxicity per patient in the gastric cancer cohort.
Unless otherwise noted, values are n (%).
Discussion
This study used previous study data consisting of CRC and GC patients who underwent palliative first‐ or second‐line FOLFIRI chemotherapy. We analyzed the effect of advanced age on efficacy and toxicity of the FOLFIRI regimen. In both cohorts, elderly patients received significantly lower doses of chemotherapeutic agents. On the basis of the post hoc power calculation data, in the CRC cohort, the treatment efficacy in elderly patients (≥70 years) was similar to that in nonelderly patients (<70 years) in terms of PFS and OS. In the GC cohort, PFS in elderly patients was similar to that in nonelderly patients. However, elderly patients in the GC cohort tended to have inferior OS compared with nonelderly patients in our data. In addition, because of limited statistical power, we could not draw any meaningful conclusion when comparing PFS and OS between patients younger than 75 years and those older than 75 years. Nevertheless, we performed additional subgroup analysis according to treatment lines and the following three age groups: patients younger than 70 years, those aged 70–74 years, and those older than 75 years. In toxicity analysis, febrile neutropenia and asthenia were more frequent in elderly patients in both CRC and GC cohorts.
For metastatic CRC, some previous studies showed that elderly patients could benefit from doublet chemotherapy consisting of fluoropyrimidine combined with irinotecan or oxaliplatin [7], [8], [15], [16], [17]. However, several randomized controlled studies performed in elderly or frail patients failed to demonstrate the superiority of doublet chemotherapy to fluoropyrimidine monotherapy [9], [18], [19]. These conflicting results might be attributed to the inclusion of frail patients because age and frailty may not always be linked. The aforementioned studies with negative results included approximately 30% of patients with a Karnofsky index of 60%–70% [9] or an ECOG PS of 2 [18, 19]. Therefore, it is still unclear whether the doublet chemotherapy could be a standard treatment in old but not frail patients. Our cohort consisted of patients with excellent PS: only 1 of 1,545 patients had an ECOG PS of 2. Therefore, our study could not evaluate the effect of poor PS on treatment efficacy or toxicity. However, our data indicate that the efficacy of FOLFIRI regimen may be similar between elderly and nonelderly patients with CRC if they have good PS.
In metastatic GC, previous studies suggest that irinotecan‐based doublet regimens could be an active treatment option in elderly patients [10], [11]. Fonck et al. reported that patients with advanced or metastatic GC aged 70 years or older showed a response rate of 26%, a median PFS of 7 months, and a median OS of 10 months [10]. Most patients in this study had an ECOG PS of 0–1 (83.4%) and were in a first‐line setting (92.9%). In another study reported by Kim and colleagues, 75% of patients were ECOG PS 2, and 45.8% were older than 65 years [11]. The response rate was 12.5%, the median time to progression was 2 months, and the median OS was 5.4 months. With these limited data, one can speculate that poor PS rather than advanced age might have been a more important predictor of clinical outcome in GC patients receiving irinotecan‐based doublet chemotherapy. In contrast to CRC, the experience of irinotecan‐based chemotherapy in elderly patients is still limited in GC. Some studies indicated that oxaliplatin‐based doublet regimens could also be an option in these patients [20], [21], [22], [23].
In contrast, a retrospective analysis showed that doublet chemotherapy was not superior to monotherapy and adverse events were more frequent in doublet group [24]. In addition, a recent population‐based outcome research by Lee and colleagues [25] revealed that elderly GC patients received significantly fewer lines of chemotherapy, leading to significantly shorter survival, and that first‐line combination chemotherapy did not provide an additional benefit compared with monotherapy in elderly GC patients.
In our data for GC patients, although the FOLFIRI regimen demonstrated considerable efficacy even in elderly patients, the treatment efficacy in these patients seems to be compromised. Especially in the first‐line subgroup, although the median PFS was relatively similar according to age, the median OS in patients older than 75 years was almost half that of those younger than 70 years. It might be attributed to limited eligibility to second or later lines of chemotherapy after progression in elderly patients. In addition, in the second‐line subgroup, PFS in patients older than 75 years was significantly shorter than in the others. Thus, it is possible that patients older than 75 years might not have undergone a significant benefit from doublet chemotherapy despite relatively good PS. These findings need to be investigated in further prospective studies.
The original aim of this study was to determine the association between UGT1A1 polymorphisms and grade 3 or 4 neutropenia during FOLFIRI chemotherapy [12]. In this primary analysis, UGT1A1 DA carriers had a significantly higher risk for grade 3 or 4 neutropenia, which was in agreement with the results of previous studies [26], [27]. Interestingly, in our CRC cohort, the proportion of UGT1A1 wild‐type carriers was significantly higher in patients older than 75 years, possibly by chance. In a previous study by Jackson et al. that was performed in patients with metastatic CRC receiving irinotecan plus fluoropyrimidine chemotherapy, the incidence of grade 3 or 4 neutropenia tended to be higher in patients older than 70 years than those younger than 70 years (38%–54% vs. 30%–40% of patients) [8]. However, the incidence of febrile neutropenia was less than 10% in both elderly and nonelderly patients, and the difference was not significant between the two groups.
In our CRC and GC cohorts, the incidence of febrile neutropenia was significantly higher in elderly patients. Moreover, 22.1% of patients older than 70 years in the CRC cohort and 35.2% of patients older than 70 years in the GC cohort experienced febrile neutropenia during FOLFIRI chemotherapy. Because our study was conducted in Korea, the difference in the incidence febrile neutropenia across studies might have been attributed to ethnic differences. Therefore, more attention should be given to elderly patients receiving FOLFIRI chemotherapy because of the increased risk for febrile neutropenia.
In both the CRC and GC cohorts, asthenia was more frequent in elderly patients. This finding is in line with the aforementioned study by Jackson et al., which reported significantly higher grade 3 or 4 asthenia in patients older than 70 years (7%–14% vs. <5%) [8]. In the CRC cohort, the incidence of mucositis and anorexia was significantly higher in elderly patients. However, mucositis and anorexia were similar among the age groups in the GC cohort. In the CRC cohort, nausea, vomiting, diarrhea, and constipation tended to be similar among the age groups. In contrast, in the GC cohort, nausea and vomiting were less frequent in elderly patients. The difference in toxicity profile between elderly and nonelderly patients should be considered when FOLFIRI chemotherapy is administered.
This study has at least four clear limitations. First, the original objective of this study did not include a subgroup analysis based on age. Thus, in both CRC and GC cohorts, some baseline characteristics were not well balanced among the age groups. To minimize the effects of the imbalance, we performed multivariable analysis using the Cox proportional hazard model. Second, the proportion of patients older than 75 years was low in both cohorts, which may result in insufficient statistical power in the subgroup analyses. Third, the current standard regimen for metastatic CRC patients is FOLFIRI or FOLFOX (oxaliplatin, 5‐FU, and leucovorin) combined with cetuximab, panitumumab, or bevacizumab [3], [28], [29]. Our data cannot provide information for the addition of these targeted agents to doublet regimen in elderly patients with CRC. Fourth, our results may not be generalized to those with poor PS as well as old age because the proportion of patients with ECOG PS 2 was very low in this study. Therefore, further studies should stratify elderly patients by their age and frailty using a comprehensive geriatric assessment to identify a subgroup of patients who may not benefit from doublet chemotherapy because of limited efficacy and serious toxicity [30]. Nevertheless, our data are meaningful because our results were drawn from a prospective cohort consisting of a relatively large number of patients. In addition, even though we analyzed the data according to primary disease or treatment lines, some clinical findings in our study were uniformly observed across patient populations regardless of primary disease or treatment lines, which may provide stronger clinical evidence.
Conclusion
Despite relatively lower dose intensity in elderly patients, the efficacy of FOLFIRI chemotherapy in elderly patients was similar to that in nonelderly patients. However, special attention should be paid to elderly patients because of increased risk for febrile neutropenia and asthenia. Our data suggest that the clinical decision between doublet and singlet chemotherapy may not be based solely on age but requires further assessment of frailty and PS. The FOLFIRI regimen could be considered as a standard backbone of therapy in elderly patients with metastatic CRC or GC and good PS.
See http://www.TheOncologist.com for supplemental material available online.
Acknowledgments
This study was funded by grant no. 1320370 from the National R & D Program for Cancer Control, Ministry for Health and Welfare, Korea, and was supported in part by CJ Korea, Seoul, Korea.
Contributed equally to this work.
Contributor Information
Jee Hyun Kim, Email: jhkimmd@snu.ac.kr.
Tae Won Kim, Email: twkimmd@amc.seoul.kr.
Author Contributions
Conception/Design: Kyu‐Pyo Kim, Byung‐Ho Nam, Jee Hyun Kim, Tae Won Kim
Provision of study material or patients: Keun‐Wook Lee, Kyu‐Pyo Kim, Yong Sang Hong, Sun Young Kim, Sook Ryun Park, Sang‐Hee Cho, Ik‐Joo Chung, Young Suk Park, Ho‐Suk Oh, Myung‐Ah Lee, Hye Jin Kang, Young Lee Park, Hye Sook Han, Kyu Taeg Lee, Dong Bok Shin, Jung Hun Kang, Dae Young Zang, Jee Hyun Kim, Tae Won Kim
Collection and/or assembly of data: Kyu‐Pyo Kim, Byung‐Ho Nam, Eun‐Kee Song, Jee Hyun Kim, Tae Won Kim
Data analysis and interpretation: Ji‐Won Kim, Keun‐Wook Lee, Kyu‐Pyo Kim, Ju Hyun Lee, Yong Sang Hong, Sun Young Kim, Sook Ryun Park, Byung‐Ho Nam, Sang‐Hee Cho, Ik‐Joo Chung, Young Suk Park, Ho‐Suk Oh, Myung‐Ah Lee, Hye Jin Kang, Young Lee Park, Eun‐Kee Song, Hye Sook Han, Kyu Taeg Lee, Dong Bok Shin, Jung Hun Kang, Dae Young Zang, Jee Hyun Kim, Tae Won Kim
Manuscript writing: Ji‐Won Kim, Keun‐Wook Lee, Ju Hyun Lee, Jee Hyun Kim, Tae Won Kim
Final approval of manuscript: Ji‐Won Kim, Keun‐Wook Lee, Kyu‐Pyo Kim, Ju Hyun Lee, Yong Sang Hong, Sun Young Kim, Sook Ryun Park, Byung‐Ho Nam, Sang‐Hee Cho, Ik‐Joo Chung, Young Suk Park, Ho‐Suk Oh, Myung‐Ah Lee, Hye Jin Kang, Young Lee Park, Eun‐Kee Song, Hye Sook Han, Kyu Taeg Lee, Dong Bok Shin, Jung Hun Kang, Dae Young Zang, Jee Hyun Kim, Tae Won Kim
Disclosures
The authors indicated no financial relationships.
Supplementary Information
References
- 1. Tournigand C, Andre T, Achille E et al. Folfiri followed by folfox6 or the reverse sequence in advanced colorectal cancer: A randomized gercor study. J Clin Oncol 2004;22:229–237. [DOI] [PubMed] [Google Scholar]
- 2. Grothey A, Sargent D, Goldberg RM et al. Survival of patients with advanced colorectal cancer improves with the availability of fluorouracil‐leucovorin, irinotecan, and oxaliplatin in the course of treatment. J Clin Oncol 2004;22:1209–1214. [DOI] [PubMed] [Google Scholar]
- 3. Heinemann V, von Weikersthal LF, Decker T et al. Folfiri plus cetuximab versus folfiri plus bevacizumab as first‐line treatment for patients with metastatic colorectal cancer (FIRE‐3): A randomised, open‐label, phase 3 trial. Lancet Oncol 2014;15:1065–1075. [DOI] [PubMed] [Google Scholar]
- 4. Bouche O, Raoul JL, Bonnetain F et al. Randomized multicenter phase II trial of a biweekly regimen of fluorouracil and leucovorin (LV5FU2), LV5FU2 plus cisplatin, or LV5FU2 plus irinotecan in patients with previously untreated metastatic gastric cancer: A Federation Francophone de Cancerologie Digestive Group Study–FFCD 9803. J Clin Oncol 2004;22:4319–4328. [DOI] [PubMed] [Google Scholar]
- 5. Kang JH, Lee SI, Lim do H et al. Salvage chemotherapy for pretreated gastric cancer: A randomized phase III trial comparing chemotherapy plus best supportive care with best supportive care alone. J Clin Oncol 2012;30:1513–1518. [DOI] [PubMed] [Google Scholar]
- 6. Guimbaud R, Louvet C, Ries P et al. Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: A French intergroup (Fédération Francophone de Cancérologie Digestive, Fédération Nationale des Centres de Lutte Contre le Cancer, and Groupe Coopérateur Multidisciplinaire en Oncologie) study. J Clin Oncol 2014;32:3520–3526. [DOI] [PubMed] [Google Scholar]
- 7. Folprecht G, Seymour MT, Saltz L et al. Irinotecan/fluorouracil combination in first‐line therapy of older and younger patients with metastatic colorectal cancer: Combined analysis of 2,691 patients in randomized controlled trials. J Clin Oncol 2008;26:1443–1451. [DOI] [PubMed] [Google Scholar]
- 8. Jackson NA, Barrueco J, Soufi‐Mahjoubi R et al. Comparing safety and efficacy of first‐line irinotecan/fluoropyrimidine combinations in elderly versus nonelderly patients with metastatic colorectal cancer: Findings from the bolus, infusional, or capecitabine with camptostar‐celecoxib study. Cancer 2009;115:2617–2629. [DOI] [PubMed] [Google Scholar]
- 9. Aparicio T, Lavau‐Denes S, Phelip JM et al. Randomized phase iii trial in elderly patients comparing LV5FU2 with or without irinotecan for first‐line treatment of metastatic colorectal cancer (FFCD 2001‐02). Ann Oncol 2016;27:121–127. [DOI] [PubMed] [Google Scholar]
- 10. Fonck M, Brunet R, Becouarn Y et al. Evaluation of efficacy and safety of FOLFIRI for elderly patients with gastric cancer: A first‐line phase II study. Clin Res Hepatol Gastroenterol 2011;35:823–830. [DOI] [PubMed] [Google Scholar]
- 11. Kim JH, Kim HS, Han AR et al. Irinotecan, leucovorin and 5‐fluorouracil (modified FOLFIRI) as salvage chemotherapy for frail or elderly patients with advanced gastric cancer. Oncol Lett 2012;4:751–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kim KP, Kim TW, Hong YS et al. Modified FOLFIRI (irinotecan 150 mg/m2) compared to FOLFIRI (irinotecan 180 mg/m2) in Korean patients with gastrointestinal cancer. J Clin Oncol 2015;33(suppl): abstr 3600. [Google Scholar]
- 13. de Gramont A, Figer A, Seymour M et al. Leucovorin and fluorouracil with or without oxaliplatin as first‐line treatment in advanced colorectal cancer. J Clin Oncol 2000;18:2938–2947. [DOI] [PubMed] [Google Scholar]
- 14. Therasse P, Arbuck SG, Eisenhauer EA et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 2000;92:205–216. [DOI] [PubMed] [Google Scholar]
- 15. Sastre J, Marcuello E, Masutti B et al. Irinotecan in combination with fluorouracil in a 48‐hour continuous infusion as first‐line chemotherapy for elderly patients with metastatic colorectal cancer: A Spanish cooperative group for the treatment of digestive tumors study. J Clin Oncol 2005;23:3545–3551. [DOI] [PubMed] [Google Scholar]
- 16. Rosati G, Cordio S, Bordonaro R et al. Capecitabine in combination with oxaliplatin or irinotecan in elderly patients with advanced colorectal cancer: Results of a randomized phase II study. Ann Oncol 2010;21:781–786. [DOI] [PubMed] [Google Scholar]
- 17. Goldberg RM, Tabah‐Fisch I, Bleiberg H et al. Pooled analysis of safety and efficacy of oxaliplatin plus fluorouracil/leucovorin administered bimonthly in elderly patients with colorectal cancer. J Clin Oncol 2006;24:4085–4091. [DOI] [PubMed] [Google Scholar]
- 18. Seymour MT, Thompson LC, Wasan HS et al. Chemotherapy options in elderly and frail patients with metastatic colorectal cancer (MRC FOCUS2): An open‐label, randomised factorial trial. Lancet 2011;377:1749–1759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hong YS, Jung KH, Kim HJ et al. Randomized phase II study of capecitabine with or without oxaliplatin as first‐line treatment for elderly or fragile patients with metastatic colorectal cancer: A prospective, multicenter trial of the Korean Cancer Study Group CO06‐ 01 Am J Clin Oncol 2013;36:565–571. [DOI] [PubMed] [Google Scholar]
- 20. Bando H, Yamada Y, Tanabe S et al. Efficacy and safety of S‐1 and oxaliplatin combination therapy in elderly patients with advanced gastric cancer. Gastric Cancer 2016;19:919–926. [DOI] [PubMed] [Google Scholar]
- 21. Catalano V, Bisonni R, Graziano F et al. A phase II study of modified FOLFOX as first‐line chemotherapy for metastatic gastric cancer in elderly patients with associated diseases. Gastric Cancer 2013;16:411–419. [DOI] [PubMed] [Google Scholar]
- 22. Cho YH, Kim SY, Hong Lee M et al. Comparative analysis of the efficacy and safety of chemotherapy with oxaliplatin plus fluorouracil/leucovorin between elderly patients over 65 years and younger patients with advanced gastric cancer. Gastric Cancer 2012;15:389–395. [DOI] [PubMed] [Google Scholar]
- 23. Choi IS, Oh DY, Kim BS et al. Oxaliplatin, 5‐FU, folinic acid as first‐line palliative chemotherapy in elderly patients with metastatic or recurrent gastric cancer. Cancer Res Treat 2007;39:99–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sun DS, Jeon EK, Won HS et al. Outcomes in elderly patients treated with a single‐agent or combination regimen as first‐line chemotherapy for recurrent or metastatic gastric cancer. Gastric Cancer 2015;18:644–652. [DOI] [PubMed] [Google Scholar]
- 25. Lee KW, Lee JH, Kim JW et al. Population‐based outcomes research on treatment patterns and impact of chemotherapy in older patients with metastatic gastric cancer. J Cancer Res Clin Oncol 2016;142:687–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Innocenti F, Undevia SD, Iyer L et al. Genetic variants in the UDP‐glucuronosyltransferase 1a1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol 2004;22:1382–1388. [DOI] [PubMed] [Google Scholar]
- 27. Han JY, Lim HS, Shin ES et al. Comprehensive analysis of UGT1A polymorphisms predictive for pharmacokinetics and treatment outcome in patients with non‐small‐cell lung cancer treated with irinotecan and cisplatin. J Clin Oncol 2006;24:2237–2244. [DOI] [PubMed] [Google Scholar]
- 28. Douillard JY, Siena S, Cassidy J et al. Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first‐line treatment in patients with previously untreated metastatic colorectal cancer: The PRIME study. J Clin Oncol 2010;28:4697–4705. [DOI] [PubMed] [Google Scholar]
- 29. Saltz LB, Clarke S, Diaz‐Rubio E et al. Bevacizumab in combination with oxaliplatin‐based chemotherapy as first‐line therapy in metastatic colorectal cancer: A randomized phase III study. J Clin Oncol 2008;26:2013–2019. [DOI] [PubMed] [Google Scholar]
- 30. Kim JW, Kim YJ, Lee KW et al. The early discontinuation of palliative chemotherapy in older patients with cancer. Support Care Cancer 2014;22:773–781. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.