Abstract
Lessons Learned.
The safety profile in the patient groups who received FOLFIRI and simtuzumab did not differ from that in the FOLFIRI and placebo group.
The addition of simtuzumab to chemotherapy with FOLFIRI does not improve clinical outcomes in patients with metastatic KRAS mutant colorectal carcinoma.
Background.
Simtuzumab, a humanized IgG4 monoclonal antibody to lysyl oxidase‐like 2 (LOXL2), blocks desmoplastic reaction in colorectal carcinoma (CRC) cells in vitro.
Methods.
Patients with metastatic Kirsten rat sarcoma viral oncogene homolog (KRAS) mutant CRC were randomized to receive second‐line 5‐fluorouracil, leucovorin, and irinotecan (FOLFIRI) with either 200 or 700 mg simtuzumab or placebo every 2 weeks in cycles of 28 days. Progression‐free survival (PFS), overall survival (OS), objective response rate (ORR), and safety were assessed.
Results.
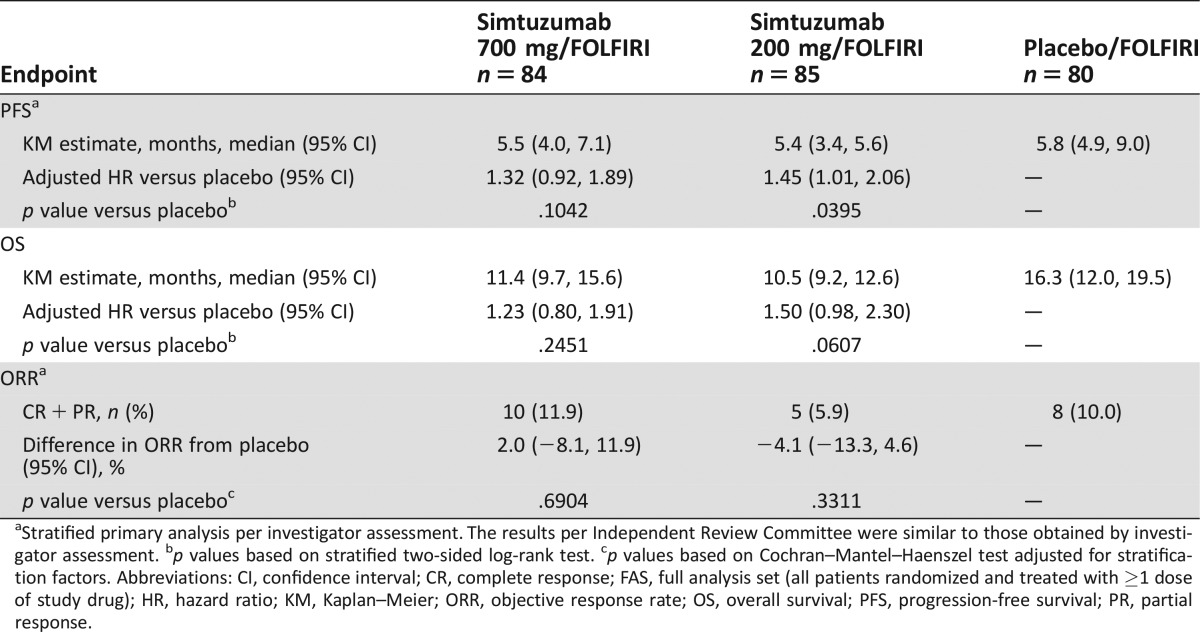
In total, 249 patients were randomized and treated with FOLFIRI/simtuzumab 700 mg (n = 84), FOLFIRI/simtuzumab 200 mg (n = 85), and FOLFIRI/placebo (n = 80). After a median follow‐up of 5.1, 3.8, and 5.5 months, respectively, median PFS for each of the respective treatment groups was 5.5 months (adjusted HR [95% CI], p value versus placebo; 1.32 [0.92, 1.89]; p = .10), 5.4 months (1.45 [1.01, 2.06]; p = .04), and 5.8 months. Median OS was 11.4 months (1.23 [0.80, 1.91]; p = .25), 10.5 months (1.50 [0.98, 2.30]; p = .06), and 16.3 months, respectively. ORR was 11.9%, 5.9%, and 10%, respectively. Simtuzumab was tolerable in metastatic KRAS mutant CRC patients.
Conclusion.
The addition of simtuzumab to FOLFIRI did not improve clinical outcomes in patients with metastatic KRAS mutant CRC.
Abstract
经验总结
• FOLFIRI+Simtuzumab组患者的安全性特征与FOLFIRI+安慰剂组没有差异。
• Simtuzumab联合FOLFIRI化疗未改善转移性KRAS突变结直肠癌患者的临床预后。
摘要
背景. Simtuzumab是赖氨酰氧化酶样蛋白‐2(LOXL2)的一种人源化IgG4单克隆抗体, 在体外可阻止结直肠癌(CRC)细胞发生促结缔组织增生性反应
方法. 携带KRAS(Kirsten大鼠肉瘤病毒癌基因同源物)突变的转移性CRC患者随机接受5‐氟尿嘧啶、甲酰四氢叶酸和伊立替康(FOLFIRI)二线治疗联合Simtuzumab 200或700 mg或者联合安慰剂治疗, 每2周给药一次, 28天为一个周期。评估无进展生存期(PFS)、总生存期(OS)、客观缓解率(ORR)和安全性
结果. 总计249例患者随机接受FOLFIRI/Simtuzumab 700 mg(n=84)、FOLFIRI/Simtuzumab 200 mg(n=85)或FOLFIRI/安慰剂(n=80)治疗。分别进行为期5.1个月、3.8个月和5.5个月的中位随访后, 相应治疗组的中位PFS分别为5.5个月[校正HR(95% CI), 与安慰剂比较的p值:1.32(0.92, 1.89), p=0.10]、5.4个月[1.45(1.01, 2.06), p=0.04]和5.8个月。中位OS分别为11.4个月[1.23(0.80, 1.91), p=0.25]、10.5个月[1.50(0.98, 2.30), p=0.06]和16.3个月。ORR分别为11.9%、5.9%和10%。转移性KRAS突变CRC患者可以耐受Simtuzumab
结论. Simtuzumab与FOLFIRI联用未改善转移性KRAS突变CRC患者的临床预后。
Discussion
Stroma in the tumor environment is associated with higher numbers of activated fibroblasts, which contribute to the desmoplastic reaction. Tumor‐associated activated fibroblasts secrete oncogenic growth factors, produce extracellular matrix (ECM), and promote epithelial cell transformation. Lysyl oxidase‐like 2 (LOXL2), an extracellular matrix enzyme that remodels ECM, is thought to contribute to the maintenance of the pathologic stromal microenvironment in cancer and fibrotic diseases and to promote tumor angiogenesis and metastatic progression. LOXL2 is expressed in desmoplastic tumors, including CRC, with no expression in the adjacent nonneoplastic areas. Simtuzumab, a humanized IgG4 monoclonal antibody to LOXL2, inhibits its enzymatic activity.
In preclinical studies with antibody precursors to simtuzumab, inhibition of LOXL2 expression reduced the number of activated fibroblasts, decreased ECM deposition, inhibited angiogenesis, and prevented tumor cell invasion and metastases. In a phase I study in patients with advanced solid tumors, simtuzumab reduced the size of several tumors in some patients.
We assessed the additive efficacy of simtuzumab in a multicenter, randomized, double‐blind, placebo‐controlled, phase II study of simtuzumab or placebo in combination with FOLFIRI as a second‐line therapy in patients with metastatic KRAS‐mutant adenocarcinoma of the colon or rectum not amenable to complete surgical resection, who previously failed fluoropyrimidine and oxaliplatin therapy (ClinicalTrials.gov #NCT01479465). Eligible patients had metastatic disease; Eastern Cooperative Oncology Group performance status (ECOG PS) ≤2; adequate bone marrow, hepatic, and renal function; and estimated life expectancy >3 months. Patients were randomized 1:1:1 to receive intravenous FOLFIRI in combination with 700 mg simtuzumab, 200 mg simtuzumab, or placebo every 2 weeks in cycles of 28 days until disease progression or unacceptable toxicity; randomization was stratified by ECOG PS 0 versus >0. From December 2011 to March 2014, 255 patients were enrolled and 249 patients received at least 1 cycle of study drug, with a median number of 6 cycles. Overall, 231 (90.6%) patients discontinued study drug, mainly due to disease progression (185 [72.5%]) and adverse events (12 [4.7%]). The majority of patients were male (128/249, 51.4%) and white (212/249, 85.1%), with a mean (range) age of 60.5 (22.0–85.0) years and a mean 20.3 months from diagnosis. A sequential stepwise hypothesis and a Hochberg testing procedure were used to compare between the two simtuzumab dosing groups versus placebo for PFS, OS, and ORR. The efficacy analyses failed to demonstrate improvement in clinical endpoints upon the addition of simtuzumab to FOLFIRI chemotherapy (Table 1). Results from additional sensitivity analyses were consistent with the primary analyses. The safety profile in the patient groups who received FOLFIRI and simtuzumab did not differ from that in the FOLFIRI and placebo group. Among seven adverse events that led to death, a case of febrile neutropenia was deemed related to treatment with FOLFIRI.
Table 1. The efficacy endpoints (FAS analysis set).
Stratified primary analysis per investigator assessment. The results per Independent Review Committee were similar to those obtained by investigator assessment.
p values based on stratified two‐sided log‐rank test.
p values based on Cochran–Mantel–Haenszel test adjusted for stratification factors.
Abbreviations: CI, confidence interval; CR, complete response; FAS, full analysis set (all patients randomized and treated with ≥1 dose of study drug); HR, hazard ratio; KM, Kaplan–Meier; ORR, objective response rate; OS, overall survival; PFS, progression‐free survival; PR, partial response.
Trial Information
- Disease
Colorectal cancer
- Stage of disease/treatment
Metastatic/Advanced
- Prior therapy
First‐line oxaliplatin‐ and fluoropyrimidine‐containing regimen
- Type of study ‐ 1
Phase II
- Type of study ‐ 2
Randomized
- ORR
Difference in ORR from placebo, stratified (primary) analysis was 2.0% for the simtuzumab 700 mg arm (p = .69), and −4.1% for the simtuzumab 200 mg arm (p = .33).
- PFS
The median PFS was 5.5 months, (HR 1.32, 0.92, 1.89; p = .10) in the 700 mg simtuzumab arm, 5.4 months (HR 1.45, 1.01, 2.06, p = .04) in the 200 mg simtuzumab arm, and 5.8 month in the control arm
- Primary endpoint
Progression‐free survival
- Secondary endpoint
Overall response rate
- Secondary endpoint
Overall survival
- Secondary endpoint
Safety
- Additional details of endpoints or study design
- Patients had to have measurable disease per Response Evaluation Criteria In Solid Tumors (RECIST) v1.1, defined with all of following criteria: (a) lesions accurately measured in at least one dimension, (b) longest diameter in the plane of measurement was recorded, and (c) minimum size of 10 mm if computed tomography slice thickness ≤5 mm; if thickness was >5 mm, then the minimum size of measurable lesions was twice the slice thickness. Patients were excluded if they had more than one prior chemotherapy regimen for stage IV/metastatic colorectal cancer; underwent an experimental medical treatment within 30 days prior to study entry; received an antitumor therapy (chemotherapy, antibody therapy, molecular targeted therapy, retinoid therapy, hormonal therapy) within 21 days prior to randomization; or had cerebral metastases, uncontrolled hypertension, coronary heart disease, liver disease, or uncontrolled infection. Treatments were administered on day 1 and 15 of each 28‐day cycle. Simtuzumab or placebo was given by IV infusion over 30 minutes. FOLFIRI consisted of 200 mg/m2 leucovorin or 400 mg/m2 dl‐leucovorin administered as a 2‐hour infusion; 180 mg/m2 irinotecan given as a 90‐minute infusion in 500 mL dextrose 5% via a Y‐connector; and 400 mg/m2 fluorouracil bolus followed by a 46‐hour infusion of 2,400 mg/m2 fluorouracil. Efficacy was analyzed in all randomized patients who received at least one dose of study drug (full analysis set [FAS]) based on investigator assessment. Safety analysis set included patients in the FAS population grouped for analyses with treatment assignments designated according to the actual study drug received. Safety assessments included the incidence of adverse events (AEs), infusion site reactions, and clinically relevant changes in laboratory values and vital signs. AEs were coded according to MedDRA version 17.1 and graded per National Cancer Institute Common Toxicity Criteria (CTCAE version 4.03). Overall response rate (ORR) was assessed by the investigator per RECIST v1.1 as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The difference in PFS and OS from placebo was assessed using Kaplan–Meier methods and the stratified log‐rank test, adjusted for the stratification factor ECOG 0 or >0 at randomization. Cochran–Mantel–Haenszel test was used for calculating difference in ORR from placebo. A sequential stepwise hypothesis and a Hochberg testing procedure were used to compare between simtuzumab dosing groups versus placebo for PFS, OS, and ORR, per investigator assessment. A number of sensitivity analyses for PFS, OS, and ORR were also performed to confirm the results of primary analyses. A total of 185 PFS events had to be observed in this study to detect a hazard ratio of 0.6 with approximately 90% power at a two‐sided 0.05 significance level based on Hochberg procedure to claim that at least one of the simtuzumab treatment groups improved PFS significantly compared with placebo. The estimated sample size was 255 patients.
- Investigator's analysis
Level of activity did not meet planned endpoint.
Drug Information Control Arm
- Generic/Working name
Folinic acid (leucovorin)
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
200 mg/m2
- Route
IV over 2 hours
- Schedule of administration
Day 1 and 15 of each 28‐day cycle
- Drug 2
- Generic/working name
5‐FU
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
2,400 mg/m2 (bolus 400 mg/m2)
- Route
Continuous intravenous infusion (CIV) over 46 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
- Drug 3
- Generic/working name
Irinotecan
- Drug type
Small molecule
- Drug class
Inhibitor of topoisomerase I
- Dose
180 mg/m2
- Route
IV over 1.5 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
Drug Information Experimental Arm A: Simtuzumab 200 mg
- Drug 1
- Generic/working name
Simtuzumab (GS‐6624)
- Company name
Gilead Sciences, Inc.
- Drug type
Antibody
- Drug class
Inhibitor of lysyl oxidase-like 2 enzyme
- Dose
200 mg per flat dose
- Route
IV
- Schedule of administration
Day 1 and 15 of each 28‐day cycle
- Drug 2
- Generic/working name
Leucovorin
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
200 mg/m2
- Route
IV over 2 hours
- Schedule of administration
Day 1 and 15 of each 28‐day cycle
- Drug 3
- Generic/working name
5‐FU
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
2,400 mg/m2 (bolus 400 mg/m2)
- Route
Continuous intravenous infusion (CIV) over 46 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
- Drug 4
- Generic/working name
Irinotecan
- Drug type
Small molecule
- Drug class
Inhibitor of topoisomerase I
- Dose
180 mg/m2
- Route
IV over 1.5 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
Drug Information Experimental Arm B: Simtuzumab 700 mg
- Drug 1
- Generic/working name
Simtuzumab (GS‐6624)
- Company name
Gilead Sciences, Inc.
- Drug type
Antibody
- Drug class
Inhibitor of lysyl oxidase-like 2 enzyme
- Dose
700 mg per flat dose
- Route
IV
- Schedule of administration
Day 1 and 15 of each 28‐day cycle
- Drug 2
- Generic/working name
Leucovorin
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
200 mg/m2
- Route
IV over 2 hours
- Schedule of administration
Day 1 and 15 of each 28‐day cycle
- Drug 3
- Generic/working name
5‐FU
- Drug type
Small molecule
- Drug class
Antimetabolite
- Dose
2,400 mg/m2 (bolus 400 mg/m2)
- Route
Continuous intravenous infusion (CIV) over 46 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
- Drug 4
- Generic/working name
Irinotecan
- Drug type
Small molecule
- Drug class
Inhibitor of topoisomerase I
- Dose
180 mg/m2
- Route
IV over 1.5 hours
- Schedule of administration
Day 1 and 15 of each 28-day cycle
Patient Characteristics
- Number of patients, male
128
- Number of patients, female
121
- Stage
Advanced, metastatic, stage IV
- Age
Median (range), years: 700 mg simtuzumab, 61 (22–78); 200 mg simtuzumab, 64 (31–83); placebo, 60.5 (32–85)
- Number of prior systemic therapies
1, first‐line oxaliplatin‐ and fluoropyrimidine‐containing regimen
- Performance status: ECOG
-
0 — 118
1 — 126
2 — 5
Primary Assessment Method
- Control Arm:
- Number of patients enrolled
84
- Number of patients evaluable for toxicity
80
- Number of patients evaluated for efficacy
80
- Response assessment CR
n = 0 (0%)
- Response assessment PR
n = 8 (10%)
- Response assessment SD
n = 53 (66%)
- Response assessment PD
n = 17 (21%)
- Response assessment OTHER
n = 2 (3%)
- (Median) duration assessments PFS
5.8 months, CI: 4.9–9.0
- (Median) duration assessments OS
16.3 months, CI: 12.0–19.5
- Experimental Arm A: Simtuzumab 200 mg
- Number of patients enrolled
86
- Number of patients evaluable for toxicity
85
- Number of patients evaluated for efficacy
85
- Response assessment CR
n = 0 (0%)
- Response assessment PR
n = 5 (6%)
- Response assessment SD
n = 50 (59%)
- Response assessment PD
n = 23 (27%)
- Response assessment OTHER
n = 7 (8%)
- (Median) duration assessments PFS
5.4 months, CI: 3.4–5.6
- (Median) duration assessments OS
10.5 months, CI: 9.2–12.6
- Experimental Arm B: Simtuzumab 700 mg
- Number of patients enrolled
85
- Number of patients evaluable for toxicity
84
- Number of patients evaluated for efficacy
84
- Response assessment CR
n = 0 (0%)
- Response assessment PR
n = 10 (12%)
- Response assessment SD
n = 49 (58%)
- Response assessment PD
n = 23 (27%)
- Response assessment OTHER
n = 2 (2%)
- (Median) duration assessments PFS
5.5 months, CI: 4.0–7.1
- (Median) duration assessments OS
11.4 months, CI: 9.7–15.6
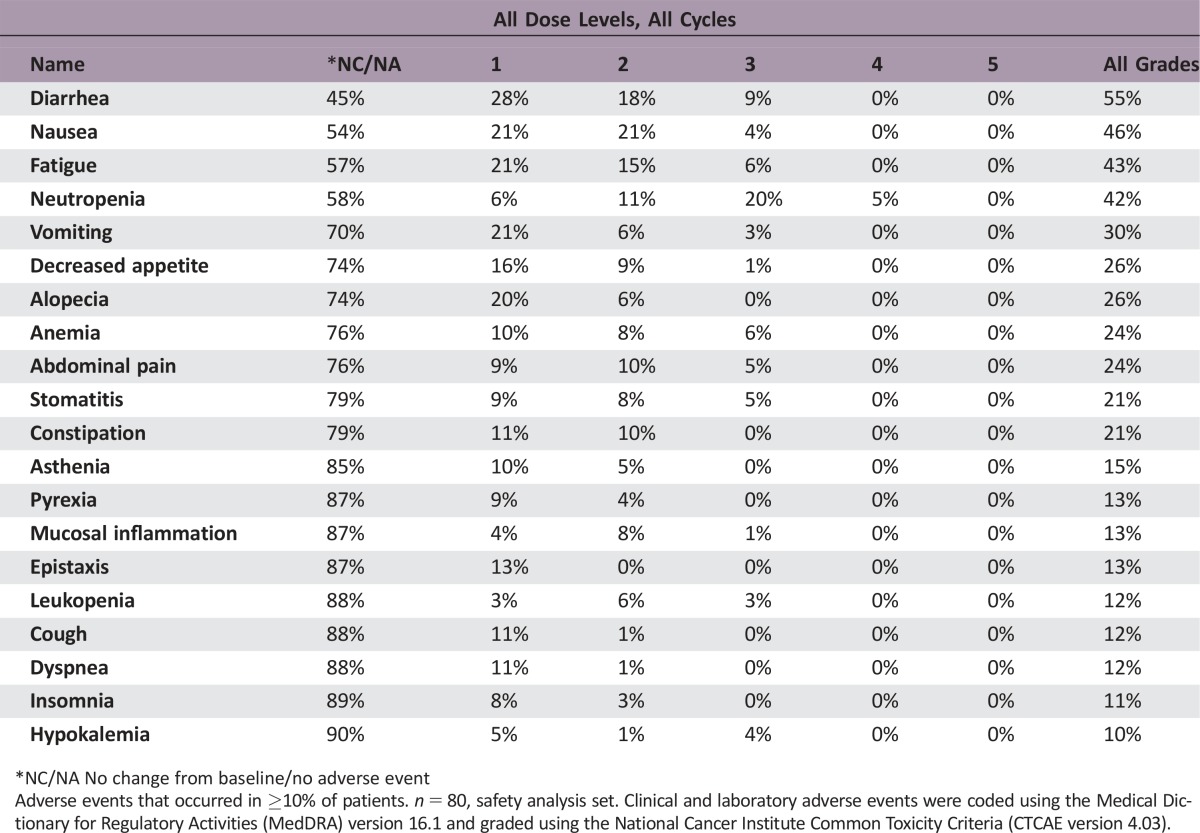
Adverse Events Control Arm
*NC/NA No change from baseline/no adverse event
Adverse events that occurred in ≥10% of patients. n = 80, safety analysis set. Clinical and laboratory adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 16.1 and graded using the National Cancer Institute Common Toxicity Criteria (CTCAE version 4.03).
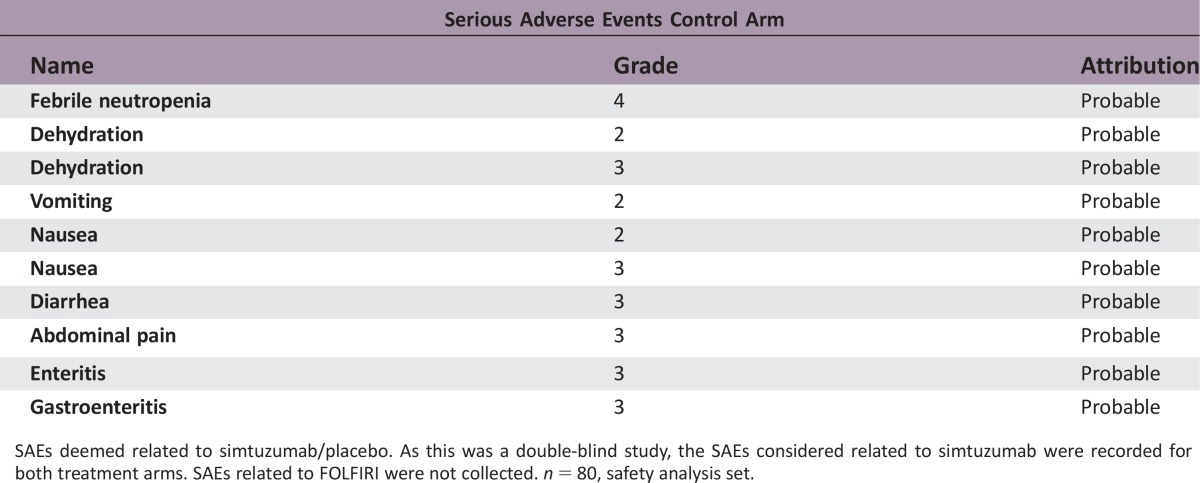
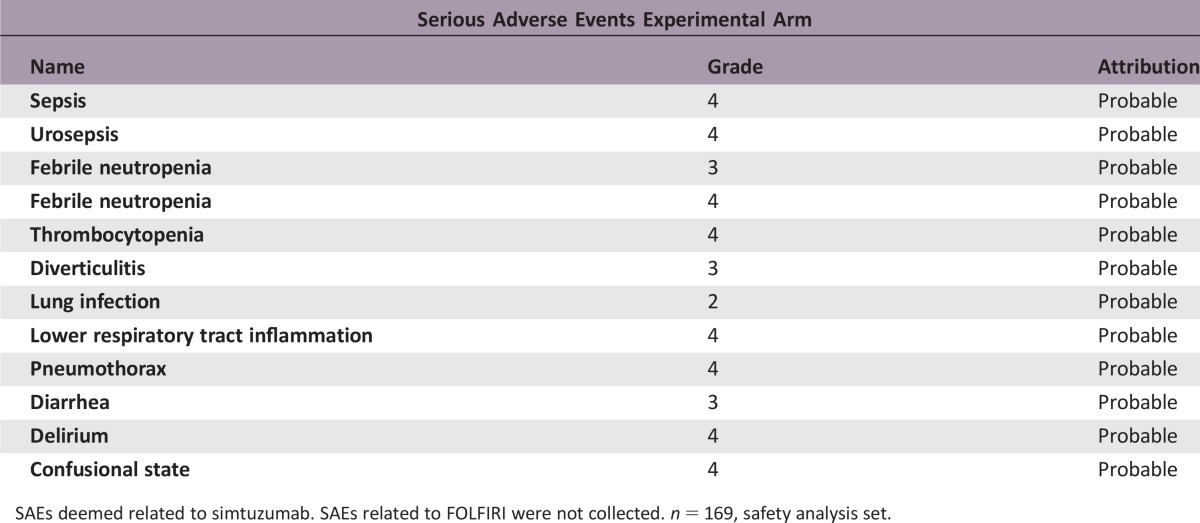
SAEs deemed related to simtuzumab/placebo. As this was a double‐blind study, the SAEs considered related to simtuzumab were recorded for both treatment arms. SAEs related to FOLFIRI were not collected. n = 80, safety analysis set.
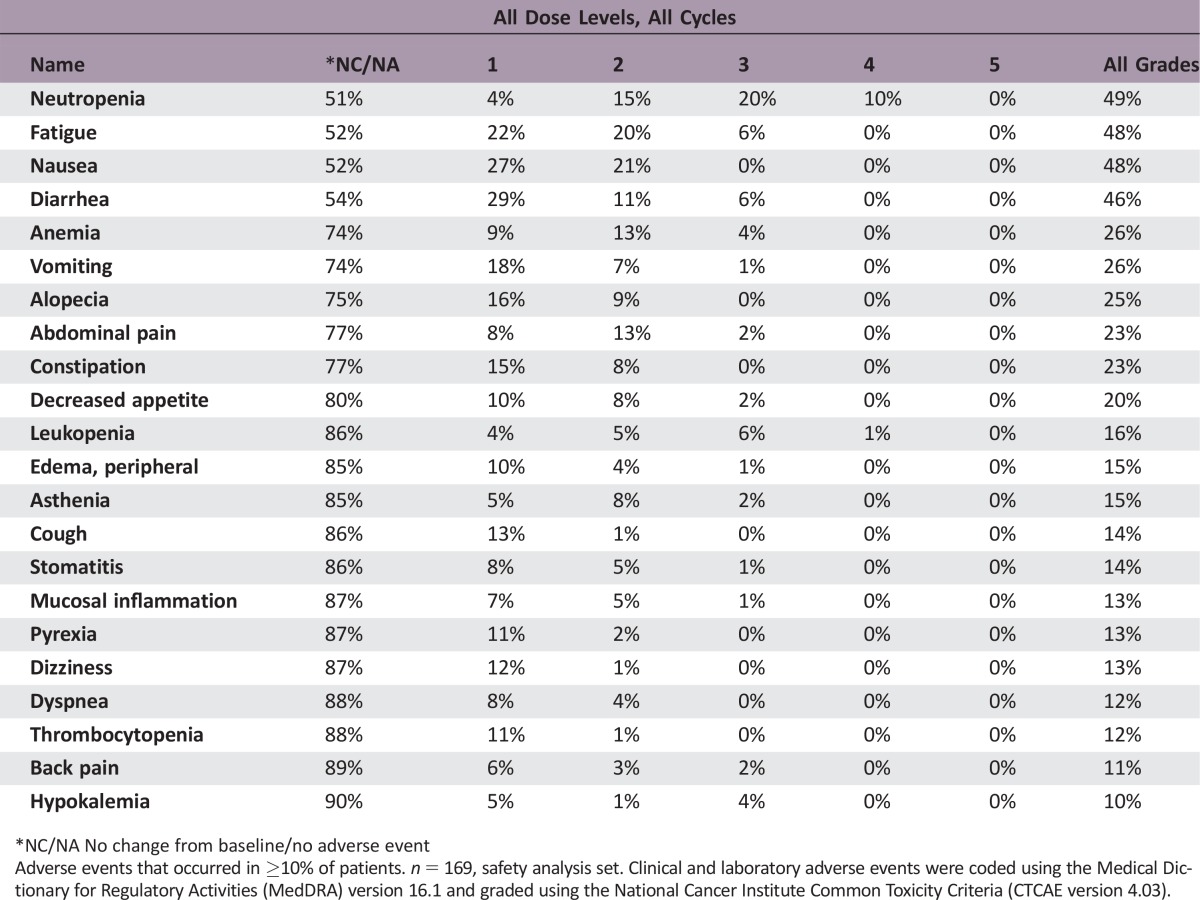
Adverse Events Both Experimental Arms
*NC/NA No change from baseline/no adverse event
Adverse events that occurred in ≥10% of patients. n = 169, safety analysis set. Clinical and laboratory adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 16.1 and graded using the National Cancer Institute Common Toxicity Criteria (CTCAE version 4.03).
SAEs deemed related to simtuzumab. SAEs related to FOLFIRI were not collected. n = 169, safety analysis set.
Assessment, Analysis, and Discussion
- Completion
Study completed
- Pharmacokinetics/Pharmacodynamics
Not Collected
- Investigator's Assessment
Level of activity did not meet planned endpoint
Colorectal cancer (CRC) accounts for 9.7% of all incident cancers worldwide and is the fourth leading cause of cancer mortality [1]. One of the frequent genetic mutations in CRC is in the KRAS gene in the chromosomal instability pathway [2]. KRAS mutation‐positive patients do not respond to anti‐epidermal growth factor receptor antibody therapy [3] and have poorer survival than patients with the KRAS wild‐type gene [4]. The currently available therapies for advanced KRAS‐mutated CRC include chemotherapy alone or in combination with antiangiogenic antibodies such as bevacizumab, aflibercept, or ramucirumab [5]. New therapies are urgently needed for this patient population.
Activated stroma in the tumor environment is associated with higher numbers of activated fibroblasts, which contribute to the desmoplastic reaction [6], [7]. Tumor‐associated activated fibroblasts secrete oncogenic growth factors, produce extracellular matrix (ECM), and promote epithelial cell transformation [7], [8]. Lysyl oxidase‐like 2 (LOXL2), an enzyme that remodels the ECM, contributes to the maintenance of the pathologic stromal microenvironment in cancer and fibrotic diseases [9] and promotes tumor angiogenesis [10] and metastatic progression [11]. LOXL2 is expressed in desmoplastic tumors, including CRC, with no expression in the adjacent nonneoplastic areas [8], [12].
Simtuzumab, a humanized IgG4 monoclonal antibody against LOXL2, inhibits its enzymatic activity [13]. In preclinical studies with antibody precursors to simtuzumab, inhibition of LOXL2 expression reduced numbers of activated fibroblasts, decreased ECM deposition [9], inhibited angiogenesis [14], and prevented tumor cell invasion and metastases [15]. Of note, in preclinical models, simtuzumab had better efficacy in KRAS mutant cell lines (Gilead Sciences, Inc., data on file). In a phase I study in patients with advanced solid tumors, single agent simtuzumab reduced the size of several tumors in select patients [16].
In a multicenter, randomized, double‐blind, placebo‐controlled phase II study, we evaluated the efficacy of simtuzumab or placebo in combination with FOLFIRI as a second‐line therapy in patients with metastatic KRAS‐mutant CRC who progressed following oxaliplatin‐ and fluoropyrimidine‐containing first‐line therapy (ClinicalTrials.gov #NCT01479465).
The key endpoints included progression‐free survival (PFS), overall survival (OS), objective response rate (ORR) per RECIST v1.1, and safety. Enrolled patients had stage IV disease; ECOG PS ≤2; adequate bone marrow, hepatic, and renal function; and estimated life expectancy >3 months. Patients were randomized 1:1:1 to receive intravenous FOLFIRI in combination with simtuzumab or placebo until disease progression or unacceptable toxicity; randomization was stratified by Eastern Cooperative Oncology Group performance status (ECOG PS) 0 vs >0. Patients were scheduled for 2 visits per cycle and a computed tomography (CT) or magnetic resonance imaging (MRI) scan every 8 weeks.
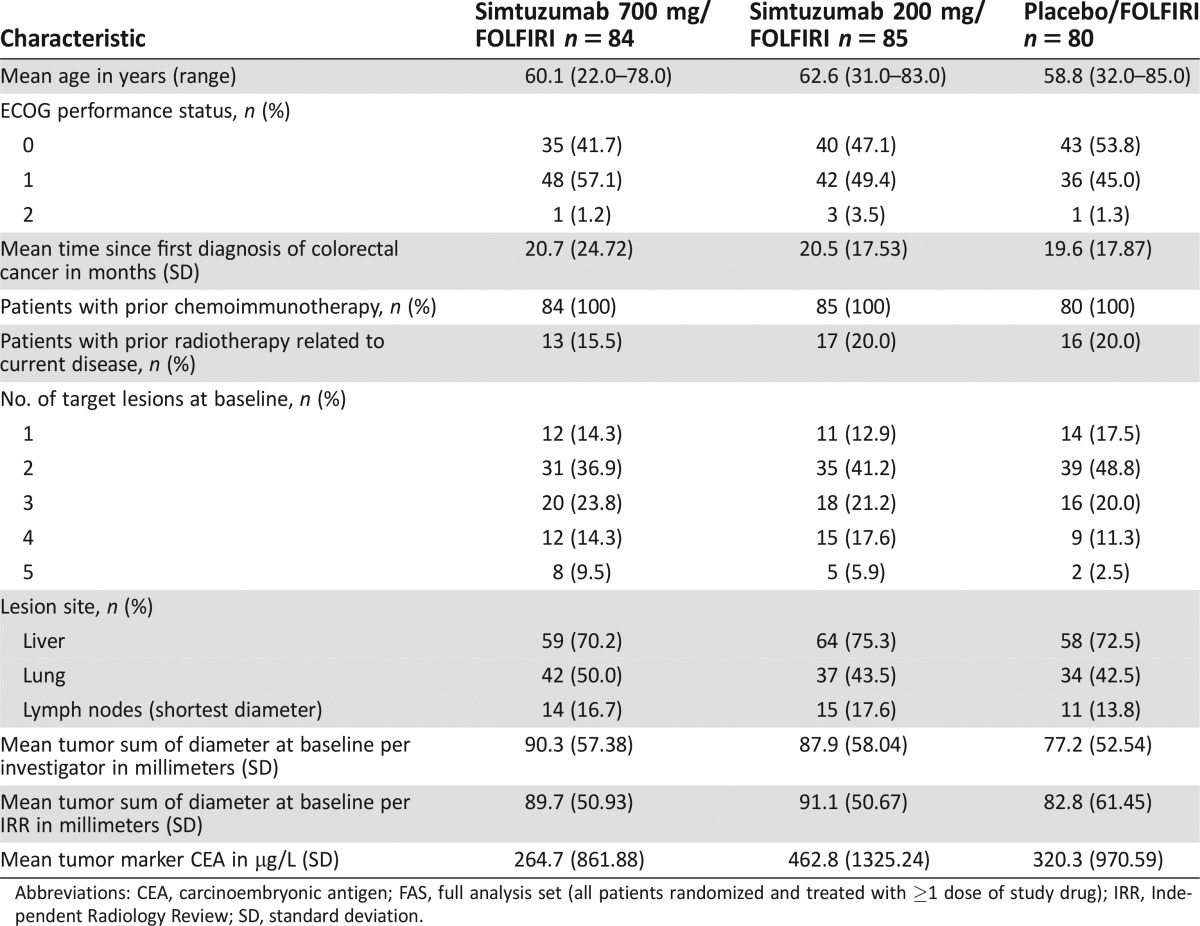
From December 2011 to March 2014, 255 patients were randomized, 249 patients received at least one 28‐day cycle of study drug (full analysis set [FAS]), and 233 (94%) patients received ≥2 cycles, with a median number of 6 cycles. The majority of enrolled patients were male (128/249, 51.4%) and white (212/249, 85.1%), with a mean (range) age of 60.5 (22.0–85.0) years (Table 2). The mean (SD) time since first diagnosis of colorectal cancer was 20.3 (20.31) months. In total, 42% of patients had 3 or more target lesions at baseline. The most common target lesion sites included liver (73%), lung (45%), and lymph nodes (16%) (Table 2).
Table 2. Demographic and baseline characteristics (FAS analysis set).
Abbreviations: CEA, carcinoembryonic antigen; FAS, full analysis set (all patients randomized and treated with ≥1 dose of study drug); IRR, Independent Radiology Review; SD, standard deviation.
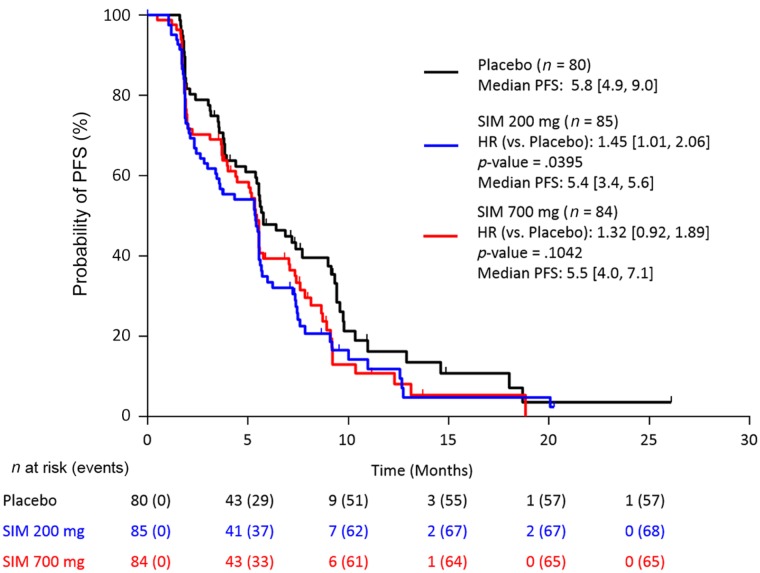
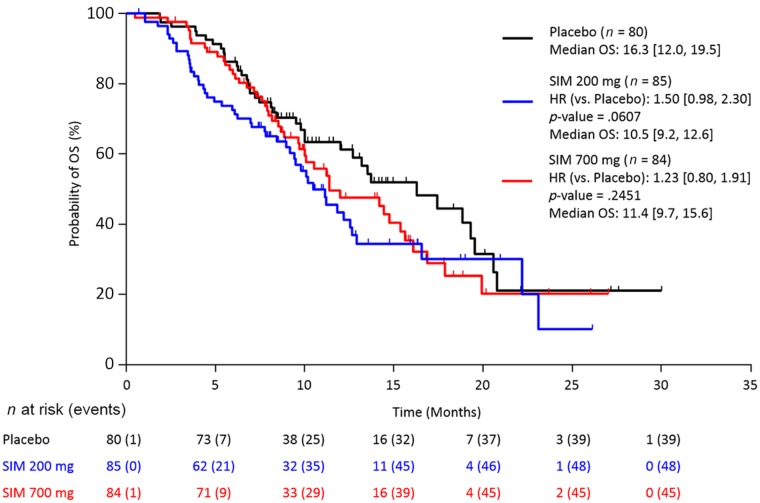
Overall, 231 (90.6%) patients had discontinued study, mainly due to disease progression (185 [72.5%]) and adverse events (12 [4.7%]). A sequential stepwise hypothesis and a Hochberg testing procedure were used to compare between the two simtuzumab dosing groups versus placebo for PFS, OS, and ORR. A number of sensitivity analyses were also conducted for PFS, OS, and ORR to confirm the results of the primary analyses. The study failed to reach any of the prespecified efficacy endpoints. Median PFS for FOLFIRI/simtuzumab 700 mg, FOLFIRI/simtuzumab 200 mg, and FOLFIRI/placebo was 5.5 months (adjusted hazard ratios [HRs] with 95% confidence interval [CIs] for the stratified primary analysis, p value versus placebo; 1.32 [0.92, 1.89]; p = .10), 5.4 months (1.45 [1.01, 2.06] p = .04), and 5.8 months, respectively (Fig. 1), after a median follow‐up of 5.1, 3.8, and 5.5 months, respectively. Median OS for the FOLFIRI/simtuzumab 700 mg, FOLFIRI/simtuzumab 200 mg, and FOLFIRI/placebo was 11.4 months (adjusted HR, [95% CIs] for the stratified OS primary analysis, p value versus placebo; 1.23 [0.80, 1.91]; p = .25), 10.5 months (1.50 [0.98, 2.30]; p = .06), and 16.3 months, respectively (Fig. 2). The adjusted HRs for PFS and OS for the simtuzumab arms compared with placebo were all greater than 1. The ORRs in FOLFIRI/simtuzumab 700 mg, FOLFIRI/simtuzumab 200 mg, and FOLFIRI/placebo were 11.9%, 5.9%, and 10.0%, respectively. The difference (95% CI) in ORR for patients treated with 700 mg FOLFIRI plus simtuzumab versus patients treated with FOLFIRI plus placebo was 2.0% (−8.1, 11.9; p = .69) and −4.1% (−13.3, 4.6; p = .33) for patients treated with 200 mg FOLFIRI plus simtuzumab compared with FOLFIRI plus placebo. Results from sensitivity analyses were consistent with the primary analyses.
Figure 1.
Stratified Kaplan–Meier plot of progression‐free survival by investigator assessment (FAS population).
Abbreviations: CI, confidence interval; FAS, full analysis set (all patients randomized and treated with ≥1 dose of study drug); HR, hazard ratio (numbers in brackets are 95% CIs); PFS, progression‐free survival; SIM, simtuzumab.
Figure 2.
Stratified Kaplan–Meier plot of overall survival (FAS population).
Abbreviations: CI, confidence interval; FAS, full analysis set (all patients randomized and treated with ≥1 dose of study drug); HR, hazard ratio (numbers in brackets are 95% CIs); OS, overall survival; SIM, simtuzumab.
The safety profile in the patient groups who received FOLFIRI combined with simtuzumab was not different from patients who received FOLFIRI with placebo. The most common adverse events (AEs) deemed by the investigator to be related to study treatment were fatigue, diarrhea, nausea and neutropenia. AEs greater than or equal to grade 3 were reported in 59.5% in the FOLFIRI/simtuzumab 700 mg group, 67.1% in the FOLFIRI/simtuzumab 200 mg group, and 61.3% in the FOLFIRI/placebo group. Seven AEs leading to death occurred during this study; one case of febrile neutropenia was deemed related to FOLFIRI treatment. None of the deaths on study were considered related to simtuzumab.
In conclusion, the addition of simtuzumab to FOLFIRI did not improve PFS, OS, or ORR in patients with metastatic KRAS mutant CRC.
Figures and Tables
Acknowledgments
Medical writing and editorial support was provided by Ewa Wandzioch Ph.D., of AlphaBioCom, LLC, King of Prussia, Pennsylvania, and was funded by Gilead Sciences, Inc.
Footnotes
ClinicalTrials.gov Identifier: NCT01479465
Sponsor(s): Gilead Sciences, Inc., Foster City, California, USA
Principal Investigator: J. Randolph Hecht
IRB Approved: Yes
Disclosures
J. Randolph Hecht: Amgen, Genentech, Boston Biomedical (C/A), Amgen, Symphogen,Merrimack Pharmaceuticals (RF); Al B. Benson III: Genentech/Roche, Sanofi, Bristol‐Myers Squibb, Merck Serono, Merck/Schering Plough, Spectrum Pharmaceuticals, Lilly/ImClone, Celgene, Genomic Health, Vicus Therapeutics, Pharmacyclics, Precision Therapeutics, Taiho Pharmaceutical, Bayer, Alchemia, Infinity Pharmaceuticals, Boehringer Ingelheim, Astellas Pharma, EMD Serono (C/A), Genentech/Roche, Sanofi, Bristol‐Myers Squibb, Merck Serono, Merck/Schering Plough, Spectrum Pharmaceuticals, Lilly/ImClone, Celgene, Genomic Health, Vicus Therapeutics, Pharmacyclics, Precision Therapeutics, Taiho Pharmaceutical, Bayer, Alchemia, Infinity Pharmaceuticals, Boehringer Ingelheim, Astellas Pharma, EMD Serono, AVEO, Gilead Sciences (H), Genentech, Gilead Sciences, Amgen, Astellas Pharma, Bayer/Onyx, Novartis, Alchemia, AVEO, Infinity Pharmaceuticals, Merck Serono, EMD Serono (RF); Yingsi Yang: Gilead Sciences, Inc. (E). The other authors indicated no financial relationships.
(C/A) Consulting/advisory relationship; (RF) Research funding; (E) Employment; (ET) Expert testimony; (H) Honoraria received; (OI) Ownership interests; (IP) Intellectual property rights/inventor/patent holder; (SAB) Scientific advisory board
References
- 1. Ferlay J, Soerjomataram I, Dikshit R et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359–E386. [DOI] [PubMed] [Google Scholar]
- 2. Armaghany T, Wilson JD, Chu Q et al. Genetic alterations in colorectal cancer. Gastrointest Cancer Res 2012;5:19–27. [PMC free article] [PubMed] [Google Scholar]
- 3. Benvenuti S, Sartore‐Bianchi A, Di Nicolantonio F et al. Oncogenic activation of the RAS/RAF signaling pathway impairs the response of metastatic colorectal cancers to anti‐epidermal growth factor receptor antibody therapies. Cancer Res 2007;67:2643–2648. [DOI] [PubMed] [Google Scholar]
- 4. Phipps AI, Buchanan DD, Makar KW et al. KRAS‐mutation status in relation to colorectal cancer survival: The joint impact of correlated tumour markers. Br J Cancer 2013;108:1757–1764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.NCCN Clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. 2016. Available at https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Accessed May 12, 2016.
- 6. Bremnes RM, Dønnem T, Al‐Saad S et al. The role of tumor stroma in cancer progression and prognosis: Emphasis on carcinoma‐associated fibroblasts and non‐small cell lung cancer. J Thorac Oncol 2011;6:209–217. [DOI] [PubMed] [Google Scholar]
- 7. Bhowmick NA, Neilson EG, Moses HL. Stromal fibroblasts in cancer initiation and progression. Nature 2004;432:332–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Fong SF, Dietzsch E, Fong KS et al. Lysyl oxidase‐like 2 expression is increased in colon and esophageal tumors and associated with less differentiated colon tumors. Genes Chromosomes Cancer 2007;46:644–655. [DOI] [PubMed] [Google Scholar]
- 9. Barry‐Hamilton V, Spangler R, Marshall D et al. Allosteric inhibition of lysyl oxidase‐like‐2 impedes the development of a pathologic microenvironment. Nat Med 2010;16:1009–1017. [DOI] [PubMed] [Google Scholar]
- 10. Zaffryar‐Eilot S, Marshall D, Voloshin T et al. Lysyl oxidase‐like‐2 promotes tumour angiogenesis and is a potential therapeutic target in angiogenic tumours. Carcinogenesis 2013;34:2370–2379. [DOI] [PubMed] [Google Scholar]
- 11. Payne SL, Hendrix MJ, Kirschmann DA. Paradoxical roles for lysyl oxidases in cancer–a prospect. J Cell Biochem 2007;101:1338–1354. [DOI] [PubMed] [Google Scholar]
- 12. Hecht JR, Bendell JC, Vyushkov D et al. A phase II, randomized, double‐blinded, placebo‐controlled study of simtuzumab or placebo in combination with FOLFIRI for the second line treatment of metastatic KRAS mutant colorectal adenocarcinoma. J Clin Oncol 2015;33:abstr 3537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Rodriguez HM, Vaysberg M, Mikels A et al. Modulation of lysyl oxidase‐like 2 enzymatic activity by an allosteric antibody inhibitor. J Biol Chem 2010;285:20964–20974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Van Bergen T, Marshall D, Van de Veire S et al. The role of LOX and LOXL2 in scar formation after glaucoma surgery. Invest Ophthalmol Vis Sci 2013;54:5788–5796. [DOI] [PubMed] [Google Scholar]
- 15. Peng L, Ran YL, Hu H et al. Secreted LOXL2 is a novel therapeutic target that promotes gastric cancer metastasis via the Src/FAK pathway. Carcinogenesis 2009;30:1660–1669. [DOI] [PubMed] [Google Scholar]
- 16. LoRusso P, Hecht J, Thai D et al. Phase I and IIa studies of simtuzumab alone and in combination with FOLFIRI in patients with advanced solid tumors. J Clin Oncol 2014;32:abstr 554. [Google Scholar]