Abstract
Importance
Case reports describe persistent erectile dysfunction (PED) associated with exposure to 5α-reductase inhibitors (5α-RIs). Clinical trial reports and the manufacturers’ full prescribing information (FPI) for finasteride and dutasteride state that risk of sexual adverse effects is not increased by longer duration of 5α-RI exposure and that sexual adverse effects of 5α-RIs resolve in men who discontinue exposure.
Objective
Our chief objective was to assess whether longer duration of 5α-RI exposure increases risk of PED, independent of age and other known risk factors. Men with shorter 5α-RI exposure served as a comparison control group for those with longer exposure.
Design
We used a single-group study design and classification tree analysis (CTA) to model PED (lasting ≥90 days after stopping 5α-RI). Covariates included subject attributes, diseases, and drug exposures associated with sexual dysfunction.
Setting
Our data source was the electronic medical record data repository for Northwestern Medicine.
Subjects
The analysis cohorts comprised all men exposed to finasteride or dutasteride or combination products containing one of these drugs, and the subgroup of men 16–42 years old and exposed to finasteride ≤1.25 mg/day.
Main outcome and measures
Our main outcome measure was diagnosis of PED beginning after first 5α-RI exposure, continuing for at least 90 days after stopping 5α-RI, and with contemporaneous treatment with a phosphodiesterase-5 inhibitor (PDE5I). Other outcome measures were erectile dysfunction (ED) and low libido. PED was determined by manual review of medical narratives for all subjects with ED. Risk of an adverse effect was expressed as number needed to harm (NNH).
Results
Among men with 5α-RI exposure, 167 of 11,909 (1.4%) developed PED (persistence median 1,348 days after stopping 5α-RI, interquartile range (IQR) 631.5–2320.5 days); the multivariable model predicting PED had four variables: prostate disease, duration of 5α-RI exposure, age, and nonsteroidal anti-inflammatory drug (NSAID) use. Of 530 men with new ED, 167 (31.5%) had new PED. Men without prostate disease who combined NSAID use with >208.5 days of 5α-RI exposure had 4.8-fold higher risk of PED than men with shorter exposure (NNH 59.8, all p < 0.002). Among men 16–42 years old and exposed to finasteride ≤1.25 mg/day, 34 of 4,284 (0.8%) developed PED (persistence median 1,534 days, IQR 651–2,351 days); the multivariable model predicting PED had one variable: duration of 5α-RI exposure. Of 103 young men with new ED, 34 (33%) had new PED. Young men with >205 days of finasteride exposure had 4.9-fold higher risk of PED (NNH 108.2, p < 0.004) than men with shorter exposure.
Conclusion and relevance
Risk of PED was higher in men with longer exposure to 5α-RIs. Among young men, longer exposure to finasteride posed a greater risk of PED than all other assessed risk factors.
Keywords: Finasteide, Dutasteride, Persistent sexual dysfunction, Impotence, Low libido, Pharmacoepidemiology, Drug safety
Introduction
There is conflicting information about sexual dysfunction associated with finasteride and dutasteride, the two 5α-reductase inhibitors (5α-RIs) marketed in the US. Isozymes 5α-R1 and 5α-R2 are expressed in adult male human prostate, epididymis, seminal vesicle, testis, genital skin, and brain (Aumüller et al., 1996; Hellwinkel et al., 2000; Nonomura et al., 1990; Thiele et al., 2005; Thigpen et al., 1993) and 5α-R3 is expressed ubiquitously in adult tissues (Godoy et al., 2011). Androgen receptors are expressed in both stromal and endothelial cells of adult male human corpus cavernosum (Schultheiss et al., 2003), which is functionally androgen-dependent in adult male humans (Rossi et al., 1998). Local conversion by 5α-R1 and 5α-R2 of testosterone to the more potent androgen, 5α-dihydrotestosterone (5α-DHT), is essential for development and maintenance of the normal structure and function of male reproductive tissues (Mahony et al., 1998; Oztekin et al., 2012; Tomada et al., 2013); local conversion by 5α-R3 of testosterone to 5α-DHT in adipose tissue (Fouad Mansour, Pelletier & Tchernof, 2016) contributes to systemic 5α-DHT levels.
Clinical development of finasteride followed observations that prepubertal castration prevents androgenic alopecia (Hamilton, 1942) and that male pseudohermaphrodites have nonpalpable prostates, scanty beards, and have neither androgenic alopecia nor acne (Imperato-McGinley et al., 1974; Imperato-McGinley & Peterson, 1976; Walsh et al., 1974). Pseudohermaphroditism is caused by loss-of-function mutation of the 5α-R2 gene, resulting in impaired conversion of testosterone to 5α-DHT, with varying degrees of genital ambiguity at birth. Pseudohermaphrodites are often raised as female but unexpectedly virilize at puberty, with partial masculinization of external genitalia (Imperato-McGinley et al., 1974; Sinnecker et al., 1996). The chemical composition-of-matter patent for finasteride describes finasteride and related compounds as “antiandrogenic by virtue of their ability to inhibit testosterone-5α-reductase” (Rasmusson & Reynolds, 1988). Exposure of men to finasteride mimics hereditary 5α-reductase deficiency by preferentially inhibiting 5α-R2 (Gormley et al., 1990), inducing a sex steroid profile “strikingly similar to that of pseudohermaphrodites” (Imperato-McGinley et al., 1990), with a 70% reduction in serum 5α-DHT (Drake et al., 1999; Grino, Griffin & Wilson, 1990; Marchetti & Barth, 2013; Roberts et al., 1999), corresponding to “circulating levels of 5α-DHT similar to those following castration” (Stoner, 1990). Dutasteride inhibits both 5α-R1 and 5α-R2, reducing serum 5α-DHT by 90% (Rittmaster et al., 2008).
There is conflicting information about the effect of 5α-RIs on testosterone in humans. Men exposed to finasteride had an increase over baseline of plasma testosterone levels; among men with baseline testosterone in the lowest tertile, plasma testosterone peaked at 1 year, then steadily declined, but did remain above baseline during the entire 4-year study (Roehrborn et al., 2003). Men with prostatic hyperplasia who were exposed to dutasteride had an increase over baseline of serum testosterone levels at one year (Hong et al., 2010). However, a long-term study found that men with prostatic hyperplasia who were exposed to finasteride had a progressive, and clinically significant, decline in testosterone over 45 months (Traish et al., 2015a). In men exposed to finasteride for one week, effective androgen levels in the prostate are similar to those in castrated men (Geller & Sionit, 1992). There is little available information about the effect of 5α-RI exposure on non-prostate genital tissues in humans. Chronic 5α-RI exposure in rats increases apoptosis and autophagy in corpus cavernosum smooth muscle, attenuating erectile function (Zhang et al., 2013). Given the pattern of expression and activity of androgen receptors and of 5α-reductases in multiple tissues and the marked effect of 5α-RIs on local and systemic 5α-DHT levels, it seems implausible that the anti-androgenic effects of 5α-RIs in men would be limited to prostate and scalp tissue.
The 5α-RIs also induce a global defect in C19 and C21 5α-metabolism, inhibiting 5α-reduction of progesterone, androstenedione, epitestosterone, cortisol, aldosterone, corticosterone, and deoxycorticosterone (Imperato-McGinley et al., 1985; Traish et al., 2015b) and thereby reducing levels of the brain neurosteroids that increase libido and sexual arousal (Finn et al., 2006; Melcangi et al., 2013; Torres & Ortega, 2003; Trost, 2013).
The FDA granted marketing approval for finasteride 5 mg in 1992, finasteride 1 mg in 1997, and dutasteride 0.5 mg in 2001. Per the full prescribing information (FPI), finasteride 5 mg is indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to improve symptoms, reduce the risk of acute urinary retention, and reduce the risk of the need for surgery including transurethral resection of the prostate (TURP) and prostatectomy. Per the FPI, finasteride 1 mg is indicated for the treatment of male pattern hair loss (androgenic alopecia) (Merck, 2014b). Per the FPI, dutasteride 0.5 mg is indicated for the treatment of symptomatic BPH in men with an enlarged prostate to improve symptoms, reduce the risk of acute urinary retention, and reduce the risk for the need for BPH-related surgery (GlaxoSmithKline, 2014a).
The FPI for finasteride 1 mg states that, regarding the clinical trial experience, “(t)here is no evidence of increased sexual adverse experiences with increased duration of treatment with (finasteride 5 mg)” and “resolution (of sexual adverse experiences) occurred in men who discontinued therapy with (finasteride 1 mg) due to these (sexual) side effects and in most of those who continued therapy” (Merck, 2014b). The FPI for finasteride 5 mg states that, regarding the clinical trial experience, “There is no evidence of increased sexual adverse experiences with increased duration of treatment with (finasteride 5 mg) (Merck, 2014c).” The FPI for dutasteride states, “In the three pivotal placebo-controlled BPH trials with (dutasteride), each 4 years in duration, there was no evidence of increased sexual adverse reactions…with increased duration of treatment” (GlaxoSmithKline, 2014a).
Our recent meta-analysis of reports of clinical trials of finasteride for androgenic alopecia found that adverse event reporting was of poor quality, systematically biased, not generalizable to routine practice, and that most subjects had ≤1 year of finasteride exposure (Belknap et al., 2015). Fifteen systematic reviews or meta-analyses of 5α-RI clinical trial reports each concluded that 5α-RI-associated sexual adverse events are infrequent, mild, and reversible. None of these prior meta-analyses assessed the adequacy of evaluation of adverse events in primary clinical trial reports (Boyle, Gould & Roehrborn, 1996; Chin, 2013; Edwards & Moore, 2002; Gacci et al., 2014; Gupta & Charrette, 2014; Jimenez Cruz, Quecedo Gutierrez & Del Llano Senaris, 2003; Mella et al., 2010; Oelke et al., 2015; Park & Choi, 2014; Tacklind et al., 2010; Trost, 2013; Varothai & Bergfeld, 2014; Wu et al., 2014; Yin et al., 2015; Yuan et al., 2015). A meta-analysis of clinical trials in men with prostatic hyperplasia found that the risk of ED was significantly increased with combined 5α-RI plus α2 adrenergic receptor blocker compared to α2 adrenergic receptor blocker alone (Favilla et al., 2016). However, an observational study found that the risk of ED was not increased with combined 5α-RI plus α2 adrenergic receptor blocker compared to α2 adrenergic receptor blocker alone (Hagberg et al., 2016). A case-control study found that sexual and erectile function of men exposed to finasteride 1 mg daily did not differ from controls (Tosti, Piraccini & Soli, 2001), and an observational study found that sexual function in men did not decline over the first 4–6 months of exposure to finasteride 1 mg (Tosti et al., 2004). Some commentators still assert that 5α-RIs are safe (Hirshburg et al., 2016; Mondaini et al., 2007; Singh & Avram, 2014).
There is limited information available about the effects of prolonged 5α-RI exposure on risk of persistent erectile dysfunction (PED). A 4-year study found that the rate of severe sexual adverse events was similar in subjects randomized to finasteride or placebo and that the rate of persistence of sexual dysfunction in finasteride subjects did not significantly differ from that in placebo subjects. However, this study relied on spontaneous reports of subjects for detection of sexual adverse events and did not use a validated measure of sexual function (Wessells et al., 2003). A report of a 5-year study found that about 10% of men with prostatic hyperplasia experienced erectile dysfunction (ED) with finasteride exposure but this report omitted mention of persistence of ED (Hudson et al., 1999). Another long-term study found a progressive and sustained worsening of erectile function in men with prostatic hyperplasia who had continued exposure to finasteride, with a clinically significant decline of six to eight points in international index of erectile function (IIEF)-EF score over 45 months (Traish et al., 2015a).
There are a disproportionate number of spontaneous reports of finasteride-associated persistent sexual dysfunction in the FDA’s adverse event reporting system (Ali, Heran & Etminan, 2015; Guo et al., 2016). There are also reports from uncontrolled case series of PED, low libido, loss of penile sensitivity, and lowered testosterone levels in young men that did not resolve after stopping finasteride (Chiriaco et al., 2016; Irwig, 2012a, 2012b, 2014; Irwig & Kolukula, 2011). Some reports describe men with symptoms beginning within days of initiating finasteride and persisting for years after stopping finasteride (Traish et al., 2011). PED has also been reported to occur with dutasteride exposure (Tsunemi et al., 2016). A recent functional magnetic resonance imaging (fMRI) study found that men with sexual adverse effects that persisted despite stopping finasteride did not have systemic androgen deficiency but did have fMRI responses to erotic stimuli that were consistent with diminished sexual arousal and depression (Basaria et al., 2016).
Given uncertainty about the frequency and severity of 5α-RI-associated sexual dysfunction, we analyzed existing clinical data for a large cohort of men prescribed finasteride or dutasteride to identify predictors of new PED. We hypothesized that longer 5α-RI exposure duration would increase the risk of PED. In addition to new PED, we also analyzed this data to identify predictors of new ED and new low libido.
Methods
This is a stratified, multivariable quasi-experimental cohort study of sexual dysfunction in men exposed to 5α-RIs. A cohort of men, all of whom had 5α-RI exposure, was evaluated to determine the variables that predict the occurrence of new ED, new low libido, and new PED. Those men with shorter 5α-RI exposure served as a comparison control group for men with longer 5α-RI exposure. Variables included subject attributes, diseases, and drug exposures associated with sexual dysfunction. We used existing data from electronic medical records and a single-group study design. We also performed a naïve analysis comparing men with and without exposure to 5α-RIs. Our data source was the Northwestern Medicine Enterprise Data Warehouse (NMEDW), an electronic medical record (EMR) data repository for patients of Northwestern Medicine. The Northwestern University Institutional Review Board granted approval to conduct this research and granted a waiver of informed consent (Approval reference STU00037913). The clinicians providing medical care to these subjects and the subjects themselves were unaware of this study of existing data. Eligible subjects for evaluation for new ED and new low libido were men 16–89 years old with at least one clinical encounter and one diagnosis from January 1992 to September 2015. Eligible subjects for evaluation for new PED were men 16–89 years old with at least one clinical encounter and one diagnosis from January 1992 to September 2013.
Exposure to 5α-RIs and other drugs
We used medication history and e-prescriptions to identify men prescribed 5α-RIs, non-steroidal anti-inflammatory drugs (NSAIDs), diuretics, antidepressants, acyclovir-type antivirals, and phosphodiesterase-5 inhibitors (PDE5Is), including combination forms (e.g., dutasteride + tamsulosin). Medication history was typically recorded by a mid-level practitioner with independent confirmation by a physician or other prescriber and is considered reliable at this institution. E-prescribing began in 2010; e-prescriptions were entered directly by prescribers. Prescription data included prescription dates, drug name, dose, and days of supply. Exposure duration was calculated as days from initial 5α-RI prescription to either onset of the adverse effect or last appearance in dataset, with omission of duplicate prescriptions and exclusion of intervals without documented 5α-RI exposure. The analysis included separate variables for finasteride ≤1.25 mg vs finasteride 5 mg; dutasteride vs finasteride; and finasteride ≤1.25 mg vs (finasteride >1.25 mg or dutasteride at any dose). We classified finasteride dosing as either ≤1.25 mg or >1.25 mg because tablet splitting of the 5 mg finasteride oral solid dosage form was commonly used to lower costs when prescribed for androgenic alopecia. For drugs approved after January 1992, assessment for drug exposure began with date of FDA approval for marketing.
Diagnoses, adverse effects, and other attributes
We used the term “impotence” for the database searches because this is the target term to which synonyms are mapped in international classification of diseases codes (ICD-9), and was thereby the term used for encoding structured data on diagnosis into the EMR that was our primary data source. The International Conference for the Ninth Revision of the International Classification of Diseases met in 1975; final proposals for ICD-9 were ratified in 1978. Thus, the terminology for ICD-9 reflects that in use in the late 1970s. The term “impotence” is now deprecated in medical parlance and has been replaced with the term “erectile dysfunction.” “Male erectile dysfunction” is the term used in ICD-10. We used the term “prostate disease” to aggregate ICD-9 terms for hyperplasia of the prostate, including with or without urinary obstruction, and with or without other urinary symptoms.
For each subject, we identified physician-determined diagnosis of impotence (ED), low libido, alopecia, prostate disease, prostate cancer, prostate surgery, Peyronie’s disease, cardiovascular disease, hypertension, diabetes mellitus, obesity, alcoholism, tobacco use, depression, herpes simplex virus (HSV-1 or HSV-2) infection, and HIV infection using ICD-9. Surgical procedures were encoded using ICD-9 procedure codes, AMA current procedural terminology (CPT) codes, and institution-specific billing codes. For manual review of narratives, impotence (ED) was defined as “inability to initiate and maintain erection sufficient for sexual intercourse.” We calculated body mass index from measured weights (kg) and heights (m). Laboratory data included glycosylated hemoglobin A1c, low-density lipoprotein, triglyceride, and magnesium. We assessed extent of healthcare utilization as number of clinical encounters before onset of the adverse effect, and also between initial 5α-RI exposure and onset of the adverse effect.
The adverse effect of ED required both a physician-determined diagnosis of ED and a contemporaneous prescription for a PDE5I during 1998 or later—when sildenafil, the first PDE5I, became available. The adverse effect of low libido required a physician-determined diagnosis of decreased libido. Designation of either new ED or new low libido additionally required that there be no prior diagnosis of ED, nor of PDE5I use, nor of low libido before initial 5α-RI exposure. PED additionally required description by a physician in the clinical narrative of new ED lasting ≥90 days after stopping 5α-RI (per FDA criterion for PED; Kothary, Diak & McMahon, 2011), as determined by manual review; a second reviewer independently assessed the relevant text and rare differences between reviewers were reconciled by consensus. The date of resolution of ED was that reported by the subject and recorded by the physician in the clinical narrative or the first encounter where the physician documented resolution, or the last encounter where the physician recorded that the subject continued to experience ED. Thus, PED required the simultaneous presence of a new diagnosis of an adverse effect (ED), discontinuation of the suspect drug (5α-RI), new use of an antidote (PDE5I), and documented persistence of the adverse effect after stopping the suspect drug (5α-RI). This case definition of 5α-RI-associated PED is analogous to trigger tools that have demonstrated high reliability in drug safety studies (Classen et al., 2011; Naessens et al., 2009; Resar, Rozich & Classen, 2003).
Definition of cohorts of men without prior sexual dysfunction and exposed to a 5α-RI
For the cohort of all men, we identified men prescribed a 5α-RI with no recorded diagnosis of ED nor of low libido nor a record of PDE5I use prior to initial prescription of a 5α-RI. For the cohort of young men, we identified men with 5α-RI exposure who were 16–42 years old, had exposure to finasteride ≤1.25 mg/day, had neither exposure to finasteride >1.25 mg/day nor exposure to dutasteride, nor prostate surgery, nor ED, nor low libido, nor PDE5I use prior to finasteride exposure.
Statistical analysis
We used a single-group study design for the main analysis. The source data did not satisfy the assumptions underlying analytical methods that are based on the general linear model or maximum likelihood function (Grimm & Yarnold, 1995, 2000). Accordingly, all analyses used optimal discriminant analysis, an exact, non-parametric statistical method (Arozullah et al., 2003; Belknap et al., 2008; Nebeker et al., 2007; Smart et al., 2008) to model ED, low libido, and PED. Use of these non-parametric methods simplifies and standardizes presentation and interpretation of statistical findings, avoids certain ambiguities that arise with alternative multivariable analytic methods, ensures valid p-values, and also identifies models that maximize predictive accuracy (i.e., as opposed to models that maximize explained variation or that maximize the value of the likelihood function; Grimm & Yarnold, 1995, 2000; Linden & Yarnold, 2016a, 2016b, 2016c, 2016d, 2016e; Linden, Yarnold & Nallamothu, 2016; Yarnold & Soltysik, 2005). All calculations were either computed exactly or were estimated using Monte Carlo simulation. Univariable analyses were conducted using optimal data analysis (ODA) software (Yarnold & Soltysik, 2005), and multivariable analyses using classification tree analysis (CTA) software (Ostrander et al., 1998; Yarnold & Soltysik, 2016; Yarnold, Soltysik & Bennett, 1997). These analyses identify the model that explicitly maximizes predictive accuracy as indexed by the effect strength for sensitivity (ESS) statistic—a chance-corrected and maximum-corrected measure of classification accuracy for which 0 is the discrimination accuracy expected by chance and 100 is perfect intergroup discrimination (Linden & Yarnold, 2016c; Yarnold, Soltysik & Bennett, 1997). As ODA analyses require no distributional assumptions about the data, permutation probability is used to compute statistical significance as exact p-values. Where multiple statistical hypotheses were tested, the Šidák multiple comparisons method was used to ensure the statistical reliability at the experimentwise (p ≤ Šidák criterion) or the generalized (per-comparison p ≤ 0.05) criterion (Yarnold & Soltysik, 2005, 2016).
Results for univariable analyses of the relationship between adverse effects and exposure variables are presented in descending order by ESS. The multivariable relationship between adverse effects and exposure variables was modeled using hierarchically optimal CTA, an algorithm that chains ODA analyses over all strata and over each branch of the classification tree to explicitly maximize ESS for the overall model. As with ODA, CTA analyses also require no distributional assumptions about the data, so permutation probability is used to compute statistical significance as exact p-values (Yarnold & Soltysik, 2016; Yarnold, Soltysik & Martin, 1994). Multivariable models identified by CTA drew potential predictors from a pool of demographic variables, subject attributes, healthcare utilization measures, disease classifications, and drug exposures (Table 3). Multivariable model endpoints were constrained a priori to be at least 10% of overall cohort size to insure adequate statistical power as well as to inhibit overfitting (i.e., identifying strata with insufficiently large sample size) and thereby improve reproducibility of the findings (Linden & Yarnold, 2016c; Yarnold & Soltysik, 2016; Yarnold, Soltysik & Martin, 1994). Under this minimum sample size constraint, the CTA algorithm uses a search procedure that explicitly assures that the reported classification tree achieves greater accuracy than any other possible alternative classification tree. We prospectively validated the CTA models of ED and low libido in the subcohort of 5α-RI exposed men who had no identified sexual dysfunction during the main study interval by using data from the 6-month interval immediately following the end of the main study interval.
Table 3. Univariate risk factors for new erectile dysfunction, new low libido, and new persistent erectile dysfunction.
(A). For men exposed to 5α-RIs, there were 29 statistically significant risk factors (p < 0.05) predicting new erectile dysfunction after exposure to 5α-RIs. Number of days of 5α-RI exposure was the fifth most important risk factor for new erectile dysfunction. Men with >90.5 days of 5α-RI exposure had a 2.2-fold higher risk of new erectile dysfunction compared with men with ≤90.5 days of 5α-RI exposure. There were nine statistically significant risk factors (p < 0.05) predicting new low libido after exposure to 5α-RIs. Number of days of 5α-RI exposure was the most important risk factor for new low libido. Men with >96.5 days of 5α-RI exposure had a three-fold higher risk of new low libido compared with men with ≤96.5 days of 5α-RI exposure. (B). For men exposed to 5α-RIs, there were 26 statistically significant risk factors (p < 0.05) predicting new persistent erectile dysfunction after exposure to 5α-RIs. Number of days of 5α-RI exposure was the third most important risk factor for new persistent erectile dysfunction. Men with >179.5 days of 5α-RI exposure had a 2.3-fold higher risk of new persistent erectile dysfunction compared with men with ≤179.5 days of 5α-RI exposure. For men younger than 42 years and exposed to 5α-RIs, there were nine statistically significant risk factors (p < 0.05) predicting new persistent erectile dysfunction after exposure to 5α-RIs. Number of days of 5α-RI exposure was the most important risk factor for new persistent erectile dysfunction. Men with >205 days of 5α-RI exposure had a 4.9-fold higher risk of new erectile dysfunction compared with men with ≤205 days of 5α-RI exposure.
| RIsk factor | N (exposure/effect) | NNH | Risk ratio | Specificity (%) | Sensitivity (%) | NPV (%) | PPV (%) | ESS (%) | p value | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| +/+ | +/− | −/+ | |||||||||
| A. (i) Univariable predictors of new erectile dysfunctiona in men prescribed 5α-reductase inhibitorsc 699 of 15,634 (4.5%) | |||||||||||
| Prostate diseased | 403 | 4,792 | 296 | 20.3 | 2.7 | 67.9 | 57.7 | 97.2 | 7.8 | 25.6 | <0.0001 |
| Number of encounters >27.5 | 506 | 7,312 | 193 | 25.0 | 2.6 | 51.0 | 72.4 | 97.5 | 6.5 | 23.4 | <0.0001 |
| Encounters after 5α-RI exposure >12.5e | 534 | 8,129 | 165 | 26.3 | 2.6 | 45.6 | 76.4 | 97.6 | 6.2 | 22.0 | <0.0001 |
| Prostate surgeryf | 203 | 1,300 | 496 | 10.0 | 3.8 | 91.3 | 29.0 | 96.5 | 13.5 | 20.3 | <0.0001 |
| Days of exposure to 5α-RI >90.5e | 497 | 7,685 | 202 | 29.7 | 2.2 | 48.5 | 71.1 | 97.3 | 6.1 | 19.6 | <0.0001 |
| Encounters prior to 5α-RI exposure >8.5e | 426 | 6,174 | 273 | 29.1 | 2.1 | 58.7 | 60.9 | 97.0 | 6.5 | 19.6 | <0.0001 |
| Prescription NSAIDg | 489 | 7,953 | 210 | 34.8 | 2.0 | 46.7 | 70.0 | 97.1 | 5.8 | 16.7 | <0.0001 |
| Age ≤72.6h | 608 | 1,0544 | 91 | 29.2 | 2.7 | 29.4 | 87.0 | 98.0 | 5.5 | 16.4 | <0.0001 |
| Hypertensiond | 371 | 5,507 | 328 | 33.9 | 1.9 | 63.1 | 53.1 | 96.6 | 6.3 | 16.2 | <0.0001 |
| Depressiond | 162 | 1,591 | 537 | 18.6 | 2.4 | 89.3 | 23.2 | 96.1 | 9.2 | 12.5 | <0.0001 |
| Number of progress notes >8.5 | 343 | 5,470 | 356 | 43.9 | 1.6 | 63.4 | 49.1 | 96.4 | 5.9 | 12.4 | <0.0001 |
| High-dose finasteride (>1.25 mg/day)g | 393 | 6,557 | 306 | 46.9 | 1.6 | 56.1 | 56.2 | 96.5 | 5.7 | 12.3 | <0.0001 |
| Progress notes after 5α-RI exposure >2.5e | 473 | 8,346 | 226 | 48.8 | 1.6 | 44.1 | 67.7 | 96.7 | 5.4 | 11.8 | <0.0001 |
| Smokingd | 284 | 4,561 | 415 | 49.6 | 1.5 | 69.5 | 40.6 | 96.2 | 5.9 | 10.1 | <0.0001 |
| Prostate cancerd | 108 | 816 | 591 | 13.0 | 2.9 | 94.5 | 15.5 | 96.0 | 11.7 | 10.0 | <0.0001 |
| Androgen drugg | 64 | 229 | 635 | 5.6 | 5.3 | 98.5 | 9.2 | 95.9 | 21.8 | 7.6 | <0.0001 |
| Diagnosis of obesityd | 104 | 1,140 | 595 | 23.7 | 2.0 | 92.4 | 14.9 | 95.9 | 8.4 | 7.2 | <0.0001 |
| Vascular diseased | 260 | 4,500 | 439 | 70.2 | 1.4 | 69.9 | 37.2 | 96.0 | 5.5 | 7.1 | <0.0001 |
| SSRI drugg | 147 | 2,106 | 552 | 41.7 | 1.6 | 85.9 | 21.0 | 95.9 | 6.5 | 6.9 | <0.0001 |
| Body mass index >27.8i | 298 | 4,863 | 396 | 72.5 | 1.3 | 63.9 | 42.9 | 95.6 | 5.8 | 6.9 | 0.0038 |
| Cyclovir drugg | 100 | 1,140 | 599 | 25.6 | 1.9 | 92.4 | 14.3 | 95.8 | 8.1 | 6.7 | <0.0001 |
| Triglyceride level >71.8i | 420 | 5,553 | 140 | 54.6 | 1.4 | 31.5 | 75.0 | 94.8 | 7.0 | 6.5 | 0.0176 |
| Diuretic drugg | 210 | 3,818 | 489 | 100.0 | 1.2 | 74.4 | 30.0 | 95.8 | 5.2 | 4.5 | 0.0097 |
| Dutasterideg | 137 | 2,333 | 562 | 78.3 | 1.3 | 84.4 | 19.6 | 95.7 | 5.5 | 4.0 | 0.0052 |
| History of herpes infectiond | 47 | 412 | 652 | 16.8 | 2.4 | 97.2 | 6.7 | 95.7 | 10.2 | 4.0 | <0.0001 |
| Diabetes mellitusd | 113 | 1,852 | 586 | 68.3 | 1.3 | 87.6 | 16.2 | 95.7 | 5.8 | 3.8 | 0.0030 |
| Anti-androgen drugg | 22 | 208 | 677 | 19.3 | 2.2 | 98.6 | 3.1 | 95.6 | 9.6 | 1.8 | 0.0005 |
| Peyronie’s diseased | 10 | 55 | 689 | 9.1 | 3.5 | 99.6 | 1.4 | 95.6 | 15.4 | 1.1 | 0.0007 |
| (ii) Univariable predictors of new low libidob in men prescribed 5α-reductase inhibitorsc 210 of 15,634 (1.34%) | |||||||||||
| HIVd | 13 | 145 | 686 | 26.3 | 1.9 | 99.0 | 1.9 | 95.6 | 8.2 | 0.9 | 0.0313 |
| Days of exposure to 5α-RI >96.5e | 161 | 8,006 | 49 | 76.0 | 3.0 | 48.1 | 76.7 | 99.3 | 2.0 | 24.8 | <0.0001 |
| Age ≤67.4h | 179 | 9,361 | 31 | 73.1 | 3.7 | 39.3 | 85.2 | 99.5 | 1.9 | 24.6 | <0.0001 |
| Encounters after 5α-RI exposure >9.5e | 170 | 9,532 | 40 | 92.8 | 2.6 | 38.2 | 81.0 | 99.3 | 1.8 | 19.1 | <0.0001 |
| Number of encounters >34.5 | 133 | 6,871 | 77 | 99.3 | 2.1 | 55.5 | 63.3 | 99.1 | 1.9 | 18.8 | <0.0001 |
| SSRI drugg | 68 | 2,185 | 142 | 51.1 | 2.8 | 85.8 | 32.4 | 98.9 | 3.0 | 18.2 | <0.0001 |
| Androgen drugg | 39 | 254 | 171 | 8.2 | 11.9 | 98.4 | 18.6 | 98.9 | 13.3 | 16.9 | <0.0001 |
| Depressiond | 58 | 1,695 | 152 | 45.2 | 3.0 | 89.0 | 27.6 | 98.9 | 3.3 | 16.6 | <0.0001 |
| Prescription NSAIDg | 139 | 8,303 | 71 | 151.7 | 1.7 | 46.2 | 66.2 | 99.0 | 1.6 | 12.4 | 0.0002 |
| LDL cholesterol >106.8i | 96 | 3,432 | 85 | 113.2 | 1.5 | 56.9 | 53.0 | 98.2 | 2.7 | 10.0 | 0.0438 |
| Encounters prior to 5α-RI exposure >11.5e | 98 | 5677 | 112 | 178.3 | 1.5 | 63.2 | 46.7 | 98.9 | 1.7 | 9.9 | 0.0192 |
| Diagnosis of obesityd | 34 | 1,210 | 176 | 66.2 | 2.2 | 92.2 | 16.2 | 98.8 | 2.7 | 8.3 | 0.0002 |
| Cyclovir drugg | 33 | 1,207 | 177 | 69.9 | 2.2 | 92.2 | 15.7 | 98.8 | 2.7 | 7.9 | 0.0003 |
| History of herpes infectiond | 18 | 441 | 192 | 37.6 | 3.1 | 97.1 | 8.6 | 98.7 | 3.9 | 5.7 | <0.0001 |
| Alcohol abused | 15 | 422 | 195 | 46.5 | 2.7 | 97.3 | 7.1 | 98.7 | 3.4 | 4.4 | 0.0012 |
| Peyronie’s diseased | 5 | 60 | 205 | 15.7 | 5.8 | 99.6 | 2.4 | 98.7 | 7.7 | 2.0 | 0.0020 |
| B. Univariable predictors of persistent erectile dysfunctionj in men prescribed 5α-reductase inhibitorsk 167 of 11,909 (1.40%) | |||||||||||
| Prostate surgeryf | 66 | 120 | 101 | 2.9 | 41.2 | 99.0 | 39.5 | 99.1 | 35.5 | 38.5 | <0.0001 |
| Prostate diseased | 103 | 3,950 | 64 | 57.9 | 3.1 | 66.4 | 61.7 | 99.2 | 2.5 | 28.0 | <0.0001 |
| 5α-RI exposure >179.5 dayse | 113 | 5,555 | 54 | 88.6 | 2.3 | 52.7 | 67.7 | 99.1 | 2.0 | 20.4 | <0.0001 |
| Prescription NSAIDg | 121 | 6,173 | 46 | 90.6 | 2.3 | 47.4 | 72.5 | 99.2 | 1.9 | 19.9 | <0.0001 |
| Hypertensiond | 94 | 4,392 | 73 | 89.9 | 2.1 | 62.6 | 56.3 | 99.0 | 2.1 | 18.9 | <0.0001 |
| Age ≤71.8h | 148 | 8,212 | 19 | 81.0 | 3.3 | 30.1 | 88.6 | 99.5 | 1.8 | 18.7 | <0.0001 |
| Encounters prior to 5α-RI exposure >2.5e | 128 | 6,842 | 39 | 95.5 | 2.3 | 41.7 | 76.6 | 99.2 | 1.8 | 18.4 | <0.0001 |
| Age at earliest 5α-RI exposure ≤70.3h | 150 | 8,509 | 17 | 82.7 | 3.3 | 27.5 | 89.8 | 99.5 | 1.7 | 17.4 | 0.0004 |
| Number of encounters >13.5 | 125 | 6,758 | 42 | 102.0 | 2.2 | 42.4 | 74.9 | 99.2 | 1.8 | 17.3 | <0.0001 |
| Prostate cancerd | 37 | 739 | 130 | 27.8 | 4.1 | 93.7 | 22.2 | 98.8 | 4.8 | 15.9 | <0.0001 |
| Any high-dose 5α-reductase inhibitorg | 114 | 6,400 | 53 | 130.3 | 1.8 | 45.5 | 68.3 | 99.0 | 1.8 | 13.8 | 0.0004 |
| High-dose finasteride (>1.25 mg/day)g | 91 | 5,047 | 76 | 154.2 | 1.6 | 57.0 | 54.5 | 98.9 | 1.8 | 11.5 | 0.0030 |
| Encounters after 5α-RI exposure >3.5e | 140 | 8,522 | 27 | 127.4 | 1.9 | 27.4 | 83.8 | 99.2 | 1.6 | 11.3 | 0.0162 |
| Smokingd | 67 | 3,464 | 100 | 142.1 | 1.6 | 70.5 | 40.1 | 98.8 | 1.9 | 10.6 | 0.0042 |
| Androgen drugg | 20 | 220 | 147 | 14.1 | 6.6 | 98.1 | 12.0 | 98.7 | 8.3 | 10.1 | <0.0001 |
| Depressiond | 34 | 1,239 | 133 | 70.4 | 2.1 | 89.4 | 20.4 | 98.7 | 2.7 | 9.8 | <0.0001 |
| SSRI drugg | 39 | 1,618 | 128 | 90.5 | 1.9 | 86.2 | 23.4 | 98.8 | 2.4 | 9.6 | 0.0004 |
| Dutasterideg | 40 | 1,902 | 127 | 127.3 | 1.6 | 83.8 | 24.0 | 98.7 | 2.1 | 7.8 | 0.0096 |
| Vascular diseased | 66 | 3,765 | 101 | 211.6 | 1.4 | 67.9 | 39.5 | 98.7 | 1.7 | 7.5 | 0.0487 |
| Diuretic drugg | 55 | 2,998 | 112 | 186.3 | 1.4 | 74.5 | 32.9 | 98.7 | 1.8 | 7.4 | 0.0340 |
| Diabetes mellitusd | 31 | 1,429 | 136 | 121.7 | 1.6 | 87.8 | 18.6 | 98.7 | 2.1 | 6.4 | 0.0121 |
| Cyclovir drugg | 20 | 865 | 147 | 107.9 | 1.7 | 92.6 | 12.0 | 98.7 | 2.3 | 4.6 | 0.0294 |
| Diagnosis of obesityd | 19 | 827 | 148 | 110.1 | 1.7 | 93.0 | 11.4 | 98.7 | 2.2 | 4.3 | 0.0333 |
| Alcohol abused | 10 | 307 | 157 | 55.5 | 2.3 | 97.4 | 6.0 | 98.6 | 3.2 | 3.4 | 0.0142 |
| HIVd | 7 | 128 | 160 | 26.1 | 3.8 | 98.9 | 4.2 | 98.6 | 5.2 | 3.1 | 0.0035 |
| Anti-androgen drugg | 7 | 168 | 160 | 37.9 | 2.9 | 98.6 | 4.2 | 98.6 | 4.0 | 2.8 | 0.0103 |
| C. Univariable risk factors for persistent erectile dysfunctionj in men <42 years old exposed to finasteride 1.25 mg/dayl 34 of 4,284 (0.79%) | |||||||||||
| Finasteride exposure >205.0 dayse | 30 | 2,557 | 4 | 108.2 | 4.9 | 39.8 | 88.2 | 99.8 | 1.2 | 28.1 | 0.0039 |
| Cyclovir drugg | 12 | 325 | 22 | 33.3 | 6.4 | 92.4 | 35.3 | 99.4 | 3.6 | 27.6 | <0.0001 |
| SSRI drugg | 13 | 478 | 21 | 47.8 | 4.8 | 88.8 | 38.2 | 99.4 | 2.6 | 27.0 | <0.0001 |
| Depressiond | 11 | 329 | 23 | 37.7 | 5.5 | 92.3 | 32.4 | 99.4 | 3.2 | 24.6 | <0.0001 |
| Prescription NSAIDg | 16 | 1,181 | 18 | 132.7 | 2.3 | 72.2 | 47.1 | 99.4 | 1.3 | 19.3 | 0.0180 |
| Smokingd | 9 | 582 | 25 | 118.2 | 2.2 | 86.3 | 26.5 | 99.3 | 1.5 | 12.8 | 0.0447 |
| Hypertensiond | 7 | 340 | 27 | 75.1 | 2.9 | 92.0 | 20.6 | 99.3 | 2.0 | 12.6 | 0.0159 |
| HIVd | 4 | 38 | 30 | 11.3 | 13.5 | 99.1 | 11.8 | 99.3 | 9.5 | 10.9 | 0.0004 |
| Diabetes mellitusd | 2 | 39 | 32 | 24.2 | 6.5 | 99.1 | 5.9 | 99.2 | 4.9 | 5.0 | 0.0436 |
Notes:
Excludes risk factors with p ≥ 0.05.
NNH, Number Needed to Harm = 1/attributable risk; NPV, Negative Predictive Value; PPV, Positive Predictive Value; ESS, Effect Strength for Sensitivity (defined in text).
The adverse effect of erectile dysfunction is defined as the earliest occurrence of ICD-9 code 607.84 or v41.7 with a concurrent prescription for any PDE-5 inhibitor drug, after exposure to 5α-RI and not present prior to exposure.
New low libido is defined based on relevant ICD-9 codes present after exposure to 5-αRI drugs but not present prior to exposure.
Selection criteria for the cohort: exposed to one or more 5α-RI drugs; no diagnoses of erectile dysfunction or low libido prior to 5α-RI exposure; no use of PDE-5 inhibitors prior to 5α-RI exposure.
Based on the presence of relevant ICD-9 codes in the medical record, without regard to diagnosis date.
Exposure is based on prescription dates in the electronic medical record.
Excludes surgery performed after the earliest occurrence of erectile dysfunction.
Prescription issued at any time in the medical record.
Age as of earliest diagnosis of sexual dysfunction; or, if no dysfunction, then age at last encounter in the medical record.
Median values over the course of the medical record.
New persistent erectile dysfunction is defined as erectile dysfunction persisting at least 90 days after discontinuation of 5α-RI drugs, based on manual review of the electronic medical record.
New persistent erectile dysfunction is defined as erectile dysfunction persisting at least 90 days after discontinuation of 5-αRI drugs, based on manual review of the medical record.
Selection criteria for the cohort: exposed to finasteride with dosage ≤1.25 mg/day; not exposed to finasteride with dosage >1.25 mg/day; and not exposed to dutasteride; no diagnoses of erectile dysfunction or low libido prior to 5α-RI exposure; no use of PDE-5 inhibitors prior to 5α-RI exposure; no prostate disease, prostate surgery, or prostate cancer; and age <42 years at time of first prescription for finasteride.
Results
Demographics and naïve analysis in exposed vs non-exposed men
The repository contained medical records for 691,268 men (Table 1) of whom 17,475 (2.5%) had 5α-RI exposure; of these, 15,634 (89.5%) had no prior diagnosis of ED nor of low libido, nor of PDE5I use. Men exposed to 5α-RIs were more likely than unexposed men to have the diagnosis of ED (number needed to harm (NNH) 17.3, ESS 6.4%, p < 0.0001) and of low libido (NNH 73.5, ESS 4.0%, p < 0.0001) and to have been prescribed a PDE5I (NNH 10.6, ESS 5.8%, p < 0.0001) (Table 2A). There were 327,437 men 16–42 years old, with 743 exposed either to finasteride >1.25 mg/day or to dutasteride, and 5,582 (1.7%) exposed to finasteride ≤1.25 mg/day. Compared to young men without 5α-RI exposure, those young men with exposure to finasteride ≤1.25 mg/day were more likely to have the adverse effect of ED (NNH 31.1, ESS 6.7%, p < 0.0001) and of low libido (NNH 51.0, ESS 7.2%, p < 0.0001) (Table 2B). Among 16,032 men with prostatic hyperplasia and with an encounter and a diagnosis recorded during 2014 (the last complete year in the dataset), 3,890 (24.3%) had 5α-RI exposure.
Table 1. Baseline characteristics of cohort membersa (total N = 691,268).
Exposed to 5α-RI drugs vs unexposed. Men exposed to 5α-RIs differed from men unexposed to 5α-RIs for several characteristics relevant to the frequency or to the detection of sexual dysfunction. Notably, exposed men had more years in the cohort (i.e., longer duration of time represented in the medical record). Men exposed to finasteride ≤1.25 mg daily were younger and were less likely to have prostate disease than men exposed to finasteride >1.25 mg daily or exposed to dutasteride.
| Characteristic | Cohort | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Unexposed (N = 673,793) | Finasteride ≤1.25 mg (N = 7,419) | Finasteride >1.25 mg (N = 7,187) | Dutasteride (N = 1,921) | All exposed (N = 17,475) | |||||||||||
| Mean year of cohort entry | 2005 ± 6.00 | 2006 ± 5.42 | 2004 ± 6.93 | 2005 ± 7.04 | 2005 ± 6.42 | ||||||||||
| Median year of cohort entry | 2006 | 2006 | 2004 | 2007 | 2005 | ||||||||||
| Median age at cohort entry | 38.9 | 30.4 | 63.8 | 64.3 | 51.0 | ||||||||||
| IQR of age at cohort entry | 28.7–53.6 | 26.2–36.8 | 53.6–71.5 | 56.2–72.4 | 30.8–66.3 | ||||||||||
| Median years in cohort (IQR) | 0.9 (0.0–5.6) | 6.1 (2.1–11.3) | 8.5 (2.2–14.6) | 5.4 (1.2–13.0) | 7.1 (2.1–13.1) | ||||||||||
| Attribute | N | % | N | % | N | % | N | % | N | % | |||||
| Use of prescription NSAIDb | 180,375 | 26.8 | 2,604 | 35.1 | 5,037 | 70.1 | 1,346 | 70.1 | 9,759 | 55.8 | |||||
| Hypertensionc | 117,585 | 17.5 | 945 | 12.7 | 4,285 | 59.6 | 1,035 | 53.9 | 6,942 | 39.7 | |||||
| Smokingc | 111,798 | 16.6 | 1,336 | 18.0 | 3,033 | 42.2 | 798 | 41.5 | 5,663 | 32.4 | |||||
| Vascular diseasec | 88,752 | 13.2 | 538 | 7.3 | 3,644 | 50.7 | 835 | 43.5 | 5,623 | 32.2 | |||||
| Use of diuretic drugb | 60,680 | 9.0 | 418 | 5.6 | 3,074 | 42.8 | 726 | 37.8 | 4,696 | 26.9 | |||||
| Diabetes mellitusc | 45,431 | 6.7 | 150 | 2.0 | 1,599 | 22.2 | 352 | 18.3 | 2,348 | 13.4 | |||||
| Use of SSRI drugb | 35,570 | 5.3 | 966 | 13.0 | 1,183 | 16.5 | 269 | 14.0 | 2,614 | 15.0 | |||||
| Depressionc | 34,586 | 5.1 | 673 | 9.1 | 1,070 | 14.9 | 195 | 10.2 | 2,101 | 12.0 | |||||
| Diagnosis of alcoholismc | 31,310 | 4.6 | 186 | 2.5 | 264 | 3.7 | 36 | 1.9 | 535 | 3.1 | |||||
| Diagnosis of obesityc | 30,069 | 4.5 | 365 | 4.9 | 797 | 11.1 | 188 | 9.8 | 1,504 | 8.6 | |||||
| Prostate diseasec | 24,936 | 3.7 | 181 | 2.4 | 4,438 | 61.8 | 940 | 48.9 | 6,349 | 36.3 | |||||
| Prostate cancerc | 18,207 | 2.7 | 57 | 0.8 | 770 | 10.7 | 187 | 9.7 | 1,155 | 6.6 | |||||
| Use of cyclovir drugb | 17,850 | 2.6 | 673 | 9.1 | 591 | 8.2 | 113 | 5.9 | 1,468 | 8.4 | |||||
| History of prostate surgeryd | 13,096 | 1.9 | 65 | 0.9 | 1,428 | 19.9 | 291 | 15.1 | 2,073 | 11.9 | |||||
| HSVc | 6,647 | 1.0 | 327 | 4.4 | 169 | 2.4 | 25 | 1.3 | 543 | 3.1 | |||||
| Alopeciac | 2,938 | 0.4 | 3,078 | 41.5 | 457 | 6.4 | 9 | 0.5 | 3,574 | 20.5 | |||||
| Peyronie’s diseasec | 840 | 0.1 | 41 | 0.6 | 47 | 0.7 | 18 | 0.9 | 111 | 0.6 | |||||
| Unexposed | Finasteride ≤1.25 mg | Finasteride >1.25 mg | Dutasteride | All exposed | |||||||||||
| Attributee | N | Median | IQR | N | Median | IQR | N | Median | IQR | N | Median | IQR | N | Median | IQR |
| Body mass index | 335,057 | 26.9 | 24.3–30.4 | 6,503 | 25.8 | 24.0–28.1 | 6,762 | 26.9 | 24.4–30.2 | 1,732 | 27.6 | 24.9–30.8 | 15,933 | 26.4 | 24.3–29.3 |
| Triglycerides | 177,526 | 100.0 | 69.0–149.0 | 4,535 | 88.0 | 63.0–127.0 | 4,057 | 95.5 | 68.0–136.0 | 792 | 99.0 | 73.0–141.1 | 10,009 | 92.0 | 66.0–132.0 |
| LDL cholesterol | 154,340 | 107.0 | 85.0–129.5 | 4,384 | 112.0 | 93.0–132.0 | 3,722 | 88.0 | 69.0–112.0 | 737 | 90.0 | 70.0–113.0 | 9,437 | 100.0 | 78.0–123.0 |
| Magnesium | 117,488 | 2.0 | 1.9–2.1 | 763 | 2.0 | 1.9–2.2 | 3,677 | 2.0 | 1.9–2.1 | 741 | 2.0 | 1.9–2.1 | 5,824 | 2.0 | 1.9–2.1 |
| Hemoglobin A1c | 48,722 | 5.6 | 5.3–6.4 | 786 | 5.3 | 5.1–5.6 | 1,862 | 5.8 | 5.5–6.5 | 412 | 5.8 | 5.4–6.5 | 3,396 | 5.7 | 5.3–6.3 |
Notes:
Selection criteria for cohort: all men with at least one diagnosis in the medical record and between 16 and 89 years old as of their last encounter.
Based on prescription data in the electronic medical record.
Based on ICD-9 codes in the electronic medical record.
Based on ICD-9 procedure codes, AMA current procedural terminology (CPT) codes, and institution-specific billing codes.
Value for each individual is the median of all values in the electronic medical record.
Table 2. Naïve analysis men exposed vs unexposed to 5α-reductase inhibitors.
(A). Men exposed to 5α-RIs had a higher risk of erectile dysfunction (based solely on ICD-9 code), PDE5I use, and low libido in the cohort of all men. The higher risk was found for all men exposed to 5α-RIs vs unexposed, men exposed to either finasteride >1.25 mg daily or to dutasteride vs unexposed, finasteride >1.25 daily vs unexposed, finasteride ≤1.25 mg vs unexposed, or dutasteride vs unexposed. (B). Men younger than 42 exposed to finasteride ≤1.25 mg/day had a higher risk of erectile dysfunction (based on ICD-9 code alone), PDE5I use, and low libido compared with unexposed men.
| Effect variable | N (exposure/effect) | NNH | Risk ratio | Specificity (%) | Sensitivity (%) | NPV (%) | PPV (%) | ESS (%) | p value | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| +/+ | +/− | −/+ | ||||||||||
| A. Univariate predictors for all men (N = 691,268): cohorts exposed to 5α-RI drugs compared to unexposed cohort | ||||||||||||
| Drug exposure | ||||||||||||
| All 5α-RIs exposed (N = 17,475) vs unexposed (N = 673,793) | Erectile dysfunctiona | 1,381 | 16,094 | 14,367 | 17.3 | 3.706 | 97.6 | 8.8 | 97.9 | 7.9 | 6.4 | <0.0001 |
| PDE-5 inhibitorb | 2,346 | 15,129 | 26,823 | 10.6 | 3.372 | 97.7 | 8.0 | 96.0 | 13.4 | 5.8 | <0.0001 | |
| Low libidoc | 381 | 17,094 | 5,520 | 73.5 | 2.661 | 97.5 | 6.5 | 99.2 | 2.2 | 4.0 | <0.0001 | |
| High-dose 5α-RIs exposed (N = 9363) vs unexposed (N = 673,793) | Erectile dysfunctiona | 915 | 8,448 | 14,367 | 13.1 | 4.583 | 98.7 | 6.0 | 97.9 | 9.8 | 4.7 | <0.0001 |
| PDE-5 inhibitorb | 1,506 | 7,857 | 26,823 | 8.3 | 4.040 | 98.8 | 5.3 | 96.0 | 16.1 | 4.1 | <0.0001 | |
| Low libidoc | 182 | 9,181 | 5,520 | 88.9 | 2.373 | 98.6 | 3.2 | 99.2 | 1.9 | 1.8 | <0.0001 | |
| High-dose finasteride exposed (N = 6,547) vs unexposed (N = 673,793) | Erectile dysfunctiona | 628 | 5,919 | 14,367 | 13.4 | 4.499 | 99.1 | 4.2 | 97.9 | 9.6 | 3.3 | <0.0001 |
| PDE-5 inhibitorb | 1,027 | 5,520 | 26,823 | 8.5 | 3.940 | 99.2 | 3.7 | 96.0 | 15.7 | 2.8 | <0.0001 | |
| Low libidoc | 123 | 6,424 | 5,520 | 94.4 | 2.293 | 99.0 | 2.2 | 99.2 | 1.9 | 1.2 | <0.0001 | |
| Low-dose finasteride exposed (N = 7,419) vs unexposed (N = 673,793) | Erectile dysfunctiona | 383 | 7,036 | 14,367 | 33.0 | 2.421 | 98.9 | 2.6 | 97.9 | 5.2 | 1.5 | <0.0001 |
| PDE-5 inhibitorb | 707 | 6,712 | 26,823 | 18.0 | 2.394 | 99.0 | 2.6 | 96.0 | 9.5 | 1.5 | <0.0001 | |
| Low libidoc | 175 | 7,244 | 5,520 | 65.0 | 2.879 | 98.9 | 3.1 | 99.2 | 2.4 | 2.0 | <0.0001 | |
| Dutasteride exposed (N = 1,921) vs unexposed (N = 673,793) | Erectile dysfunctiona | 162 | 1,759 | 14,367 | 15.9 | 3.955 | 99.7 | 1.1 | 97.9 | 8.4 | 0.9 | <0.0001 |
| PDE-5 inhibitorb | 280 | 1,641 | 26,823 | 9.4 | 3.661 | 99.7 | 1.0 | 96.0 | 14.6 | 0.8 | <0.0001 | |
| Low libidoc | 37 | 1,884 | 5,520 | 90.3 | 2.351 | 99.7 | 0.7 | 99.2 | 1.9 | 0.4 | <0.0001 | |
| B. Univariate predictors for men <42d (N = 326,694): cohort exposed to low-dose finasteride (≤1.25 mg/day), compared to unexposed cohort | ||||||||||||
| Drug exposure | ||||||||||||
| Low-dose finasteride exposed (N = 5,582) vs unexposed (N = 321,112) | Erectile dysfunctiona | 222 | 5,360 | 2,447 | 31.1 | 5.219 | 98.3 | 8.3 | 99.2 | 4.0 | 6.7 | <0.0001 |
| PDE-5 inhibitorb | 386 | 5,196 | 3,787 | 17.4 | 5.864 | 98.4 | 9.2 | 98.8 | 6.9 | 7.6 | <0.0001 | |
| Low libidoc | 133 | 5,449 | 1,359 | 51.0 | 5.630 | 98.3 | 8.9 | 99.6 | 2.4 | 7.2 | <0.0001 | |
Notes:
NNH, Number Needed to Harm = 1/attributable risk; NPV, Negative Predictive Value; PPV, Positive Predictive Value; ESS, Effect Strength for Sensitivity (defined in text).
Adverse event of erectile dysfunction is the earliest occurrence of ICD-9 code 607.84 or v41.7 after exposure to 5α-RI with a concurrent prescription for a PDE5I, and not present prior to exposure.
Based on prescription dates in the electronic medical record.
Based on ICD-9 codes in the electronic medical record.
Men <42: Males between 16 and 42 years old (at time of first exposure to any 5-αRI), either not exposed to any 5-α reductase inhibitor or exposed to low-dose finasteride, excluding high-dose 5α-RIs.
New erectile dysfunction and new low libido in men exposed to a 5α-RI
Among the 15,634 men with 5α-RI exposure and without prior sexual dysfunction, 699 (4.5%) developed new ED and 210 (1.3%) developed new low libido.
Univariable predictors for new erectile dysfunction and new low libido in all men exposed
5α-Reductase inhibitor exposure duration was a significant predictor of new ED (cutpoint >90.5 days of 5α-RI exposure, NNH 29.7, ESS 19.6%, p < 0.0001). Of the 29 significant predictors of new ED, four were more accurate predictors than 5α-RI exposure duration: prostate disease, prostate surgery, number of encounters, and number of encounters after 5α-RI exposure (Table 3A). Of the 15 significant predictors of new low libido, 5α-RI exposure duration was the most accurate predictor (cutpoint >96.5 days of 5α-RI exposure, NNH 76, ESS 24.8%, p < 0.0001) (Table 3A).
Multivariable models for new erectile dysfunction and new low libido in all men exposed
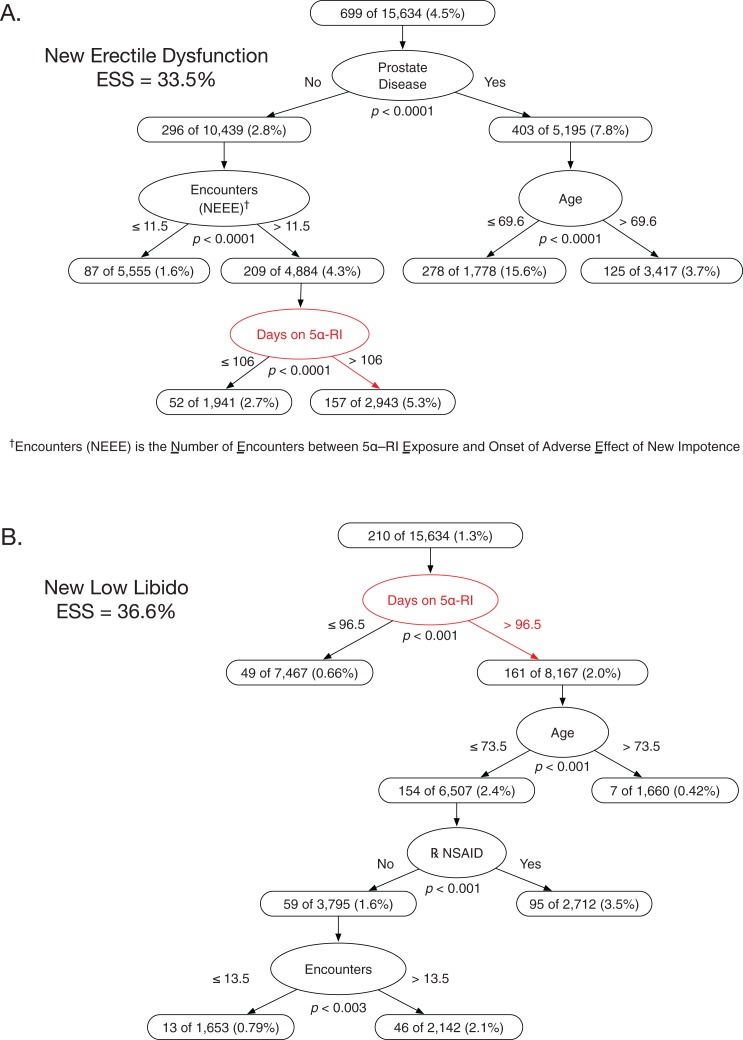
The best multivariable model predicting new ED had four variables: prostate disease, number of encounters after initial 5α-RI exposure, 5α-RI exposure duration, and age (ESS 33.5%, all p ≤ 0.0001; prospective validity on 6-month holdout sample ESS 30.8%) (Fig. 1A). Among men without prostate disease and with > 11.5 encounters after 5α-RI exposure, the NNH for new ED was 37.7 for longer vs shorter 5α-RI exposure duration (cutpoint >106 days of 5α-RI exposure). The multivariable model predicting new low libido had four attributes: 5α-RI exposure duration, age, NSAID exposure (Y/N), and total number of clinical encounters (ESS 36.6%, all p ≤ 0.003; prospective validity on 6-month holdout sample ESS 34.2%) (Fig. 1B).
Figure 1. Classification tree analyses for erectile dysfunction and low libido in men prescribed 5α-reductase inhibitors.
The two classification trees shown are those that predicted, respectively, new erectile dysfunction or new low libido after 5α-RI exposure with greater accuracy than any other possible alternative classification tree given the pool of exposure variables in Table 3. (A) This optimally predictive multivariable model for new erectile dysfunction had four variables: prostate disease, number of encounters between 5α-RI exposure and onset of impotence, age, and number of days on 5α-RIs. Men with no prostate disease, >11.5 clinical encounters, and >106 days of 5α-RI exposure had a 5.3% rate of new erectile dysfunction. (B) This optimally predictive multivariable model for new low libido had four variables: number of days of 5α-RI exposure, age, use of prescribed NSAIDs, and total number of clinical encounters. Men with >96.5 days of 5α-RI exposure, age ≤73.5 years, and use of NSAIDs had a 3.5% rate of new low libido.
New persistent erectile dysfunction in men exposed to a 5α-RI
Of the 11,909 men with 5α-RI exposure and without prior sexual dysfunction and who were evaluated for new PED, 167 (1.4%) developed new PED lasting for ≥90 days after stopping the 5α-RI (median 1,348 days after stopping 5α-RI, interquartile range (IQR) 631.5–2,320.5 days). Of the 530 men with new ED, 167 (31.5%) had new PED.
Univariable predictors for new persistent erectile dysfunction in all men exposed
5α-Reductase inhibitor exposure duration was the third most accurate predictor of PED (cutpoint >179.5 days of 5α-RI exposure, NNH 88.6, ESS 20.4%, p < 0.0001). Of the 26 statistically significant predictors of PED, only prostate surgery and prostate disease were more accurate predictors than 5α-RI exposure duration (Table 3B).
Multivariable model for new persistent erectile dysfunction in all men exposed
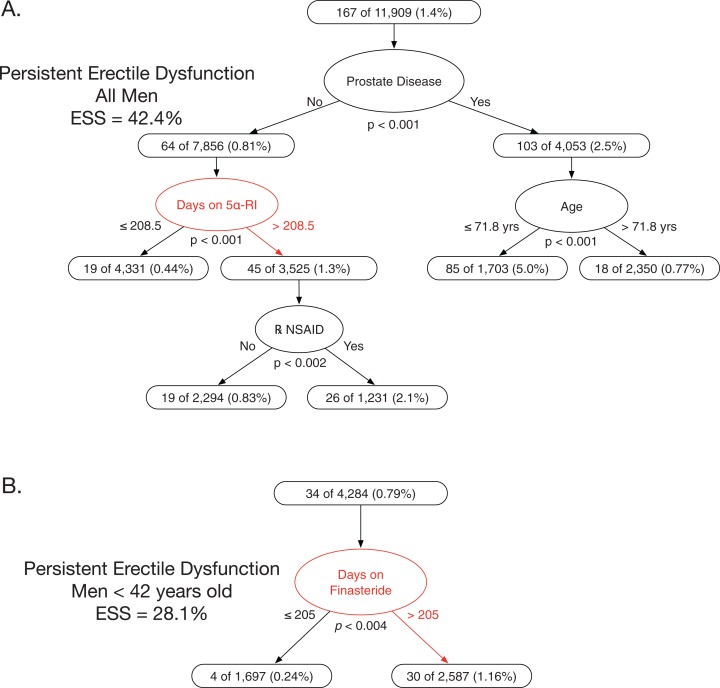
The best multivariable model predicting new PED had four variables: prostate disease, 5α-RI exposure duration, age, and NSAID use (ESS 42.4%, all p ≤ 0.002) (Fig. 2A). Among men with no prostate disease, those with longer 5α-RI exposure plus concomitant NSAID exposure had a risk of new PED that was 4.8-fold higher than men with shorter exposure (cutpoint >208.5 days of 5α-RI exposure, NNH = 59.8, all p < 0.002).
Figure 2. Classification tree analyses for persistent erectile dysfunction in men prescribed 5α-reductase inhibitors.
The two classification trees shown are those that predicted new persistent erectile dysfunction in, respectively, all 5α-RI exposed men or in all 5α-RI exposed men <42 years old with greater accuracy than any other possible alternative classification tree given the pool of exposure variables in Table 3. (A) This optimally predictive multivariable model for new persistent erectile dysfunction in all men had four variables: prostate disease, number of days on 5α-RIs, age, and use of prescribed NSAIDs. Men with no prostate disease, >208.5 days on 5α-RIs, and use of prescribed NSAIDs had a 2.1% rate of new persistent erectile dysfunction. The median duration of new persistent erectile dysfunction was 1,348 days. (B) This optimally predictive multivariable model for new persistent erectile dysfunction in finasteride exposed men <42 years old had one variable: number of days on finasteride. Men with >205 days on finasteride had a 1.16% rate of new persistent erectile dysfunction. The median duration of new persistent erectile dysfunction was 1,534 days.
New persistent erectile dysfunction in men 16–42 years old and exposed only to finasteride ≤1.25 mg/day
Of 4,284 young men exposed to finasteride ≤1.25 mg/day, and without prior sexual dysfunction and who were evaluated for new PED, 34 (0.79%) developed PED (median 1,534 days after stopping 5α-RI, IQR 651–2,351 days). Of 103 young men with new ED, 34 (33%) had new PED.
Univariable predictors for new persistent erectile dysfunction in young men exposed to finasteride ≤1.25
Of the nine significant predictors of new PED in young men, duration of finasteride exposure was the most accurate predictor (cutpoint >205 days of finasteride exposure, NNH 108.2, ESS 28.1%, p < 0.004) (Table 3C).
Multivariable model for new persistent erectile dysfunction in young men exposed to finasteride ≤1.25
The best multivariable model predicting new PED had one variable: duration of finasteride exposure. Compared to young men with shorter exposure, those young men with longer 5α-RI exposure had a 4.9-fold higher risk (cutpoint >205 days of finasteride exposure, NNH 108.2, ESS 28.1%, p < 0.004) (Fig. 2B).
Discussion
Among an estimated 14 million US men with symptomatic prostatic hyperplasia (National Institute of Diabetes and Digestive and Kidney Diseases, 2015), 5α-RI exposure rose from 4.3% in 1993 to 15.2%, or 2.1 million men, in 2010 (Filson, Wei & Hollingsworth, 2013). An estimated half-million additional men were prescribed finasteride for androgenic alopecia in 2011 (Merck, 2011). Prescribers and patients might reasonably expect that accurate information would be available about the frequency, severity, and persistence of a common adverse effect of a drug approved for marketing more than two decades ago and prescribed to an estimated 2.6 million men annually. However, a meta-analysis of 34 reports of clinical trials of finasteride for androgenic alopecia found inadequate safety reporting and systematic underreporting of adverse events (Belknap et al., 2015), exemplifying a known flaw in the detection or reporting of adverse drug effects in the medical literature (Kostoff, 2016). While a few reports of trials of 5α-RIs for prostatic hyperplasia and lower urinary tract symptoms provide assessments of sexual dysfunction using universal evaluation and validated instruments (Fwu et al., 2014); most rely on spontaneous voluntary reporting for adverse event detection and on global introspection for causality assessment (Belknap et al., 2013); these methods are considered unreliable for detecting and evaluating adverse events in general (Arimone et al., 2007; Koch-Weser, Sellers & Zacest, 1977; Kramer et al., 1985), and sexual dysfunction in particular (Althof et al., 2013; Moore, 2015). Since the introduction of the IIEF in 1997 (Rosen et al., 1997, 1999), the IIEF has been routinely used in clinical trials of PDE5Is to assess their efficacy for treatment of sexual dysfunction (Vardi & Nini, 2007). In contrast, the IIEF has rarely been used for assessing sexual adverse effects in clinical trials of 5α-RIs, although many such trials occurred after 1997. An uncontrolled study in 55 men reported that moderate to severe ED (IIEF score <17) was present in 38% of men after 1 month of dutasteride exposure and in 22% of men after 12 months of dutasteride exposure (Chi & Kim, 2011). Despite assertions to the contrary (Singh & Avram, 2014), there is scant available clinical trial data bearing on persistence of sexual dysfunction after 5α-RI exposure. In an oft-cited report on the Prostate Cancer Prevention Trial, some sexual dysfunction variables apparently were not analyzed at all, and adverse effect outcomes were not reported for those subjects who temporarily or permanently discontinued study drug, even though sexual dysfunction was the most common reason for early termination of subjects (Moinpour et al., 2007). In our previous study, we found that 20% of serious adverse events that occur during cancer clinical trials are not reported to an Institutional Review Board and likely were not detected at all by the investigators (Belknap et al., 2013). Such flaws in the design and analysis of clinical trials of 5α-RIs have created a knowledge gap regarding risk of 5α-RI-associated severe sexual dysfunction. Similar flaws in clinical trials resulted in multi-decade delay in recognition of the high frequency of sexual dysfunction associated with thiazide diuretics (Langford et al., 1990), β-adrenergic antagonists (Doumas et al., 2006), and antidepressants (Khazaie et al., 2015).
Our data show that, in a cohort of men exposed to 5α-RIs, the duration of 5α-RI exposure was a more accurate predictor of PED than many known risk factors, including age, hypertension, diabetes mellitus, cigarette smoking, ethanol abuse, obesity, and depression. In our data, confounding by age or extent of healthcare utilization did not account for the increased risk of PED associated with longer 5α-RI exposure duration (Table 3, Figs. 2A and 2B). Also, duration of finasteride exposure proved to be a more accurate predictor of sexual dysfunction than higher dose vs lower dose of finasteride, likely reflecting that finasteride exerts near-maximal inhibition of 5α-DHT synthesis at a dose of 1 mg (Drake et al., 1999; Roberts et al., 1999; Shukla, 2011).
A limitation of our study is the potential for confounding by factors associated with 5α-RI exposure. To address this limitation, our experimental design and statistical analyses included provisions to avoid or mitigate such confounding. We included an extensive set of potential confounders in the analyses, including measures of extent of healthcare utilization, age, BMI, comorbid conditions, and concomitant drugs. We made no a priori assumption about the structure of the predictive multivariable models. To reduce heterogeneity, we used a single-group experimental design (Corrao et al., 2014), excluding men who were not prescribed a 5α-RI. We required both diagnosis of ED and prescription of a PDE5I for designation of ED as well as physician description in the narrative for designation of PED. While unmeasured clinical or behavioral attributes may exist, our finding of a consistent effect provides evidence of an intrinsic relationship between duration of 5α-RI exposure and PED. We did not evaluate the extent to which NSAID-associated sexual dysfunction represents either an adverse drug effect (Gleason et al., 2011; Shiri et al., 2006) or confounding by indication for NSAIDs (Patel et al., 2015). We did not evaluate other reported adverse effects associated with 5α-RI exposure, including other sexual effects (infertility, anorgasmia, and sexual anhedonia), genital disorders (Peyronie’s Disease, penile or scrotal numbness, penile or scrotal shrinkage, and infertility), physical effects (gynecomastia, muscle atrophy, thinning, and drying of skin), cognitive disorders (memory impairment, slowed cognition, and confusion), or psychological disorders (anxiety, depression, anhedonia, and insomnia) (Ganzer, Jacobs & Iqbal, 2015).
The lower rate of detection of sexual dysfunction in older men likely reflects diminished disclosure by older men and lower likelihood of inquiry about sexual health by their physicians (Loeb et al., 2011). The predictor cutpoints for 5α-RI exposure duration do not establish a safe threshold for exposure duration. Our use of a single-group design for the primary analysis means that the observed NNH must be considered as an upper bound, as all men in the study cohort were exposed to 5α-RIs. As our data source was derived from an existing EMR system, detection of sexual dysfunction was necessarily dependent on what clinicians entered into the medical record. Evaluations using standardized instruments, such as the IIEF (introduced in 1997), were not routinely recorded in the source medical record and thus was not reliably available for our study. We expect that a clinical trial using randomization, placebo-control, universal evaluation, and a validated measure of ED would give a higher attributable risk and therefore a lower NNH.
Androgenic alopecia and prostatic hyperplasia are chronic, non-life-threatening conditions. In 1994, the International Conference on Harmonization (ICH) provided a guideline for assessment of the safety of drugs being developed to treat chronic, non-life-threatening conditions. This guideline recommends that a cohort of 300–600 subjects be exposed to the new drug for six months, and that 100 subjects be exposed for 1 year. This guideline does not directly address the evaluation of resolution or persistence of an adverse drug event (ICH, 1994). Although severe sexual dysfunction was a foreseeable consequence of 5α-reductase inhibition, it is not clear if the pivotal clinical trials for finasteride and dutasteride included assessment for persistent sexual dysfunction or other severe sexual adverse effects. If there was meaningful assessment of persistent sexual dysfunction in humans during the clinical development of the 5α-RIs, this does not appear to have been reported in the medical literature, the FPI, or other publicly accessible sources.
In conclusion, among men with 5α-RI exposure, duration of 5α-RI exposure was a more accurate predictor of PED than all other assessed risk factors except prostate disease and prostate surgery. Among young men with 5α-RI exposure, duration of 5α-RI exposure was a more accurate predictor of PED than all other assessed risk factors. For each 108 young men exposed for >205 days to the finasteride dose typically used for androgenic alopecia (≤1.25 mg/day), one additional young man experienced PED when compared to those men with shorter exposure. The median duration of PED in young men was 1,534 days. We expect that our finding of an association between debilitating sexual dysfunction and exposure to finasteride or dutasteride will be of particular interest to prescribers and patients considering medical management of androgenic alopecia or symptomatic treatment of prostatic hyperplasia.
Acknowledgments
This work was a collaborative project of the research on adverse drug events and reports (RADAR) Group. We thank Jeffrey Barsuk, MD for asking whether finasteride is safe in men with androgenic alopecia.
Technical Support: We thank Dan Schneider, Jon Tavernier, Andrew Winter, and the staff of the Northwestern University Enterprise Data Warehouse for their extensive advice and assistance.
Funding Statement
This work was supported by National Institutes of Health grants 5R01CA102713-04 (Dr. Belknap and Dr. West) and 1R01 CA125077-01A1 (Dr. Belknap and Dr. West). This work was also supported, in part, by the National Institutes of Health’s National Center for Advancing Translational Sciences, Grant Numbers UL1TR001422, UL1TR000150, and UL1RR025741. Additional funding was provided by a gift from the Post-Finasteride Syndrome Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
Dr. Yarnold is an employee of Optimal Data Analysis, LLC.
Author Contributions
Tina Kiguradze performed the experiments, analyzed the data, wrote the paper, and reviewed drafts of the paper, data abstraction.
William H. Temps performed the experiments, analyzed the data, contributed reagents/materials/analysis tools, wrote the paper, prepared figures and/or tables, and reviewed drafts of the paper.
Paul R. Yarnold conceived and designed the experiments, analyzed the data, contributed reagents/materials/analysis tools, wrote the paper, prepared figures and/or tables, and reviewed drafts of the paper.
John Cashy analyzed the data, reviewed drafts of the paper, consultation on data queries.
Robert E. Brannigan reviewed drafts of the paper, expertise in Sexual Medicine.
Beatrice Nardone performed the experiments, analyzed the data, and reviewed drafts of the paper, consultation on data queries.
Giuseppe Micali reviewed drafts of the paper, expertise in dermatology.
Dennis Paul West performed the experiments, analyzed the data, wrote the paper, and reviewed drafts of the paper, expertise in dermatopharmacology.
Steven M. Belknap conceived and designed the experiments, performed the experiments, analyzed the data, wrote the paper, prepared figures and/or tables, and reviewed drafts of the paper.
Human Ethics
The following information was supplied relating to ethical approvals (i.e., approving body and any reference numbers):
The Northwestern University Institutional Review Board approved conduct of this study with a waiver of consent. The relevant approval number is STU00037913.
Data Deposition
The following information was supplied regarding data availability:
The raw data was provided for review, but cannot be made publicly available because, though it has been de-identified, it is possible to re-identify the patients and would compromise their privacy.
References
- Ali, Heran & Etminan (2015).Ali AK, Heran BS, Etminan M. Persistent sexual dysfunction and suicidal ideation in young men treated with low-dose finasteride: a pharmacovigilance study. Pharmacotherapy: The Journal ofHuman Pharmacology and Drug Therapy. 2015;35(7):687–695. doi: 10.1002/phar.1612. [DOI] [PubMed] [Google Scholar]
- Althof et al. (2013).Althof SE, Rosen RC, Perelman MA, Rubio-Aurioles E. Standard operating procedures for taking a sexual history. Journal of Sexual Medicine. 2013;10(1):26–35. doi: 10.1111/j.1743-6109.2012.02823.x. [DOI] [PubMed] [Google Scholar]
- Arimone et al. (2007).Arimone Y, Miremont-Salame G, Haramburu F, Molimard M, Moore N, Fourrier-Reglat A, Begaud B. Inter-expert agreement of seven criteria in causality assessment of adverse drug reactions. British Journal of Clinical Pharmacology. 2007;64(4):482–488. doi: 10.1111/j.1365-2125.2007.02937.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arozullah et al. (2003).Arozullah AM, Parada J, Bennett CL, Deloria-Knoll M, Chmiel JS, Phan L, Yarnold PR. A rapid staging system for predicting mortality from HIV-associated community-acquired pneumonia. Chest. 2003;123(4):1151–1160. doi: 10.1378/chest.123.4.1151. [DOI] [PubMed] [Google Scholar]
- Aumüller et al. (1996).Aumüller G, Eicheler W, Renneberg H, Adermann K, Vilja P, Forssmann WG. Immunocytochemical evidence for differential subcellular localization of 5α-reductase isoenzymes in human tissues. Cells Tissues Organs. 1996;156(4):241–252. doi: 10.1159/000147852. [DOI] [PubMed] [Google Scholar]
- Basaria et al. (2016).Basaria S, Jasuja R, Huang G, Wharton W, Pan H, Pencina K, Li Z, Travison TG, Bhawan J, Gonthier R, Labrie F, Dury AY, Serra C, Papazian A, O’Leary M, Amr S, Storer TW, Stern E, Bhasin S. Characteristics of men who report persistent sexual symptoms after finasteride use for hair loss. Journal of Clinical Endocrinology and Metabolism. 2016;101(12):4669–4680. doi: 10.1210/jc.2016-2726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belknap et al. (2015).Belknap SM, Aslam I, Kiguradze T, Temps WH, Yarnold PR, Cashy J, Brannigan RE, Micali G, Nardone B, West DP. Adverse event reporting in clinical trials of finasteride for androgenic alopecia: a meta-analysis. JAMA Dermatology. 2015;151(6):600–606. doi: 10.1001/jamadermatol.2015.36. [DOI] [PubMed] [Google Scholar]
- Belknap et al. (2013).Belknap SM, Georgopoulos CH, Lagman J, Weitzman SA, Qualkenbush L, Yarnold PR, Edwards BJ, McKoy JM, Trifilio SM, West DP. Reporting of serious adverse events during cancer clinical trials to the institutional review board: an evaluation by the research on adverse drug events and reports (RADAR) project. Journal of Clinical Pharmacology. 2013;53(12):1334–1340. doi: 10.1002/jcph.177. [DOI] [PubMed] [Google Scholar]
- Belknap et al. (2008).Belknap SM, Moore H, Lanzotti SA, Yarnold PR, Getz M, Deitrick DL, Peterson A, Akeson J, Maurer T, Soltysik RC, Storm GA, Brooks I. Application of software design principles and debugging methods to an analgesia prescription reduces risk of severe injury from medical use of opioids. Clinical Pharmacology and Therapeutics. 2008;84(3):385–392. doi: 10.1038/clpt.2008.24. [DOI] [PubMed] [Google Scholar]
- Boyle, Gould & Roehrborn (1996).Boyle P, Gould AL, Roehrborn CG. Prostate volume predicts outcome of treatment of benign prostatic hyperplasia with finasteride: meta-analysis of randomized clinical trials. Urology. 1996;48(3):398–405. doi: 10.1016/s0090-4295(96)00353-6. [DOI] [PubMed] [Google Scholar]
- Chi & Kim (2011).Chi BH, Kim SC. Changes in sexual function in benign prostatic hyperplasia patients taking dutasteride: 1-year follow-up results. Korean Journal of Urology. 2011;52(9):632–636. doi: 10.4111/kju.2011.52.9.632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin (2013).Chin EY. Androgenetic alopecia (male pattern hair loss) in the United States: what treatments should primary care providers recommend? Journal of the American Association of Nurse Practitioners. 2013;25(8):395–401. doi: 10.1111/1745-7599.12030. [DOI] [PubMed] [Google Scholar]
- Chiriaco et al. (2016).Chiriaco G, Cauci S, Mazzon G, Trombetta C. An observational retrospective evaluation of 79 young men with long-term adverse effects after use of finasteride against androgenetic alopecia. Andrology. 2016;4(2):245–250. doi: 10.1111/andr.12147. [DOI] [PubMed] [Google Scholar]
- Classen et al. (2011).Classen DC, Resar R, Griffin F, Federico F, Frankel T, Kimmel N, Whittington JC, Frankel A, Seger A, James BC. ‘Global trigger tool’ shows that adverse events in hospitals may be ten times greater than previously measured. Health Affairs. 2011;30(4):581–589. doi: 10.1377/hlthaff.2011.0190. [DOI] [PubMed] [Google Scholar]
- Corrao et al. (2014).Corrao G, Ghirardi A, Segafredo G, Zambon A, Della Vedova G, Lapi F, Cipriani F, Caputi A, Vaccheri A, Gregori D, Gesuita R, Vestri A, Staniscia T, Mazzaglia G, Di Bari M, On behalf of the BEST investigators User-only design to assess drug effectiveness in clinical practice: application to bisphosphonates and secondary prevention of fractures. Pharmacoepidemiology and Drug Safety. 2014;23:859–867. doi: 10.1002/pds.3650. [DOI] [PubMed] [Google Scholar]
- Doumas et al. (2006).Doumas M, Tsakiris A, Douma S, Grigorakis A, Papadopoulos A, Hounta A, Tsiodras S, Dimitriou D, Giamarellou H. Factors affecting the increased prevalence of erectile dysfunction in Greek hypertensive compared with normotensive subjects. Journal of Andrology. 2006;27(3):469–477. doi: 10.2164/jandrol.04191. [DOI] [PubMed] [Google Scholar]
- Drake et al. (1999).Drake L, Hordinsky M, Fiedler V, Swinehart J, Unger WP, Cotterill PC, Thiboutot DM, Lowe N, Jacobson C, Whiting D, Stieglitz S, Kraus SJ, Griffin EI, Weiss D, Carrington P, Gencheff C, Cole GW, Pariser DM, Epstein ES, Tanaka W, Dallob A, Vandormael K, Geissler L, Waldstreicher J. The effects of finasteride on scalp skin and serum androgen levels in men with androgenetic alopecia. Journal of the American Academy of Dermatology. 1999;41(4):550–554. doi: 10.1016/s0190-9622(99)70295-1. [DOI] [PubMed] [Google Scholar]
- Edwards & Moore (2002).Edwards JE, Moore RA. Finasteride in the treatment of clinical benign prostatic hyperplasia: a systematic review of randomised trials. BMC Urology. 2002;2(1):14. doi: 10.1186/1471-2490-2-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favilla et al. (2016).Favilla V, Russo GI, Privitera S, Castelli T, Giardina R, Calogero AE, Condorelli RA, La Vignera S, Cimino S, Morgia G. Impact of combination therapy 5-alpha reductase inhibitors (5-ARI) plus alpha-blockers (AB) on erectile dysfunction and decrease of libido in patients with LUTS/BPH: a systematic review with meta-analysis. Aging Male. 2016;9(3):175–181. doi: 10.1080/13685538.2016.1195361. [DOI] [PubMed] [Google Scholar]
- Filson, Wei & Hollingsworth (2013).Filson CP, Wei JT, Hollingsworth JM. Trends in medical management of men with lower urinary tract symptoms suggestive of benign prostatic hyperplasia. Urology. 2013;82(6):1386–1392. doi: 10.1016/j.urology.2013.07.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finn et al. (2006).Finn DA, Beadles-Bohling AS, Beckley EH, Ford MM, Gililland KR, Gorin-Meyer RE, Wiren KM. A new look at the 5α-reductase inhibitor finasteride. CNS Drug Reviews. 2006;12:53–76. doi: 10.1111/j.1527-3458.2006.00053.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fouad Mansour, Pelletier & Tchernof (2016).Fouad Mansour M, Pelletier M, Tchernof A. Characterization of 5α-reductase activity and isoenzymes in human abdominal adipose tissues. Journal of Steroid Biochemistry Molecular Biology. 2016;161:45–53. doi: 10.1016/j.jsbmb.2016.02.003. [DOI] [PubMed] [Google Scholar]
- Fwu et al. (2014).Fwu CW, Eggers PW, Kirkali Z, McVary KT, Burrows PK, Kusek JW. Change in sexual function in men with lower urinary tract symptoms/benign prostatic hyperplasia associated with long-term treatment with doxazosin, finasteride and combined therapy. Journal of Urology. 2014;191(6):1828–1834. doi: 10.1016/j.juro.2013.12.014. [DOI] [PubMed] [Google Scholar]
- Gacci et al. (2014).Gacci M, Ficarra V, Sebastianelli A, Corona G, Serni S, Shariat SF, Maggi M, Zattoni F, Carini M, Novara G. Impact of medical treatments for male lower urinary tract symptoms due to benign prostatic hyperplasia on ejaculatory function: a systematic review and meta-analysis. Journal of Sexual Medicine. 2014;11(6):1554–1566. doi: 10.1111/jsm.12525. [DOI] [PubMed] [Google Scholar]
- Ganzer, Jacobs & Iqbal (2015).Ganzer CA, Jacobs AR, Iqbal F. Persistent sexual, emotional, and cognitive impairment post-finasteride: a survey of men reporting symptoms. American Journal of Men’s Health. 2015;9(3):222–228. doi: 10.1177/1557988314538445. [DOI] [PubMed] [Google Scholar]
- Geller & Sionit (1992).Geller J, Sionit L. Castration-like effects on the human prostate of a 5α-reductase inhibitor, finasteride. Journal of Cellular Biochemistry. 1992;15(S16H):109–112. doi: 10.1002/jcb.240501225. [DOI] [PubMed] [Google Scholar]
- GlaxoSmithKline (2014a).GlaxoSmithKline Dutasteride full prescribing information. 2014a. https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Avodart/pdf/AVODART-PI-PIL.PDF https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Avodart/pdf/AVODART-PI-PIL.PDF
- Gleason et al. (2011).Gleason JM, Slezak JM, Jung H, Reynolds K, Van den Eeden SK, Haque R, Quinn VP, Loo RK, Jacobsen SJ. Regular nonsteroidal anti-inflammatory drug use and erectile dysfunction. Journal of Urology. 2011;185(4):1388–1393. doi: 10.1016/j.juro.2010.11.092. [DOI] [PubMed] [Google Scholar]
- Godoy et al. (2011).Godoy A, Kawinski E, Li Y, Oka D, Alexiev B, Azzouni F, Titus MA, Mohler JL. 5α-reductase type 3 expression in human benign and malignant tissues: a comparative analysis during prostate cancer progression. Prostate. 2011;71(10):1033–1046. doi: 10.1002/pros.21318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gormley et al. (1990).Gormley GJ, Stoner E, Rittmaster RS, Gregg H, Thompson DL, Lasseter KC, Vlasses PH, Stein EA. Effects of finasteride (MK-906), a 5α-reductase inhibitor, on circulating androgens in male volunteers. Journal of Clinical Endocrinology and Metabolism. 1990;70(4):1136–1141. doi: 10.1210/jcem-70-4-1136. [DOI] [PubMed] [Google Scholar]
- Grimm & Yarnold (1995).Grimm LG, Yarnold PR. Reading and Understanding Multivariate Statistics. Washington, DC: American Psychological Association; 1995. [Google Scholar]
- Grimm & Yarnold (2000).Grimm LG, Yarnold PR. Reading and Understanding More Multivariate Statistics. Washington, DC: American Psychological Association; 2000. [Google Scholar]
- Grino, Griffin & Wilson (1990).Grino PB, Griffin JE, Wilson JD. Testosterone at high concentrations interacts with the human androgen receptor similarly to dihydrotestosterone. Endocrinology. 1990;126(2):1165–1172. doi: 10.1210/endo-126-2-1165. [DOI] [PubMed] [Google Scholar]
- Guo et al. (2016).Guo M, Heran B, Flannigan R, Kezouh A, Etminan M. Persistent sexual dysfunction with finasteride 1 mg taken for hair loss. Journal of Human Pharmacology and Drug Therapy. 2016;36(11):1180–1184. doi: 10.1002/phar.1837. [DOI] [PubMed] [Google Scholar]
- Gupta & Charrette (2014).Gupta AK, Charrette A. The efficacy and safety of 5α-reductase inhibitors in androgenetic alopecia: a network meta-analysis and benefit-risk assessment of finasteride and dutasteride. Journal of Dermatological Treatment. 2014;25(2):156–161. doi: 10.3109/09546634.2013.813011. [DOI] [PubMed] [Google Scholar]
- Hagberg et al. (2016).Hagberg KW, Divan HA, Persson R, Nickel JC, Jick SS. Risk of erectile dysfunction associated with use of 5-alpha reductase inhibitors for benign prostatic hyperplasia or alopecia: population based studies using the Clinical Practice Research Datalink. BMJ. 2016;354:i4823. doi: 10.1136/bmj.i4823. [DOI] [PubMed] [Google Scholar]
- Hamilton (1942).Hamilton J. Male hormone stimulation is prerequisite and an incitant in common baldness. American Journal of Anatomy. 1942;71(3):451–480. doi: 10.1002/aja.1000710306. [DOI] [Google Scholar]
- Hellwinkel et al. (2000).Hellwinkel OJ, Muller A, Struve D, Hiort O. Influence of androgens and age on androgen receptor and 5α-reductase II transcription. European Journal of Endocrinology. 2000;143(2):217–225. doi: 10.1530/eje.0.1430217. [DOI] [PubMed] [Google Scholar]
- Hirshburg et al. (2016).Hirshburg JM, Kelsey PA, Therrien CA, Gavino AC, Reichenberg JS. Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. Journal of Clinical and Aesthetic Dermatology. 2016;9:56–62. [PMC free article] [PubMed] [Google Scholar]
- Hong et al. (2010).Hong SK, Min GE, Ha SB, Doo SH, Kang MY, Park HJ, Yoon CY, Jeong SJ, Byun SS, Lee SE. Effect of the dual 5α-reductase inhibitor, dutasteride, on serum testosterone and body mass index in men with benign prostatic hyperplasia. BJU International. 2010;105(7):970–974. doi: 10.1111/j.1464-410X.2009.08915.x. [DOI] [PubMed] [Google Scholar]
- Hudson et al. (1999).Hudson PB, Boake R, Trachtenberg J, Romas NA, Rosenblatt S, Narayan P, Geller J, Lieber MM, Elhilali M, Norman R, Patterson L, Perreault JP, Malek GH, Bruskewitz RC, Roy JB, Ko A, Jacobsen CA, Stoner E. Efficacy of finasteride is maintained in patients with benign prostatic hyperplasia treated for 5 years. The North American Finasteride Study Group. Urology. 1999;53:690–695. doi: 10.1016/s0090-4295(98)00666-9. [DOI] [PubMed] [Google Scholar]
- Imperato-McGinley et al. (1974).Imperato-McGinley J, Guerrero L, Gautier T, Peterson RE. Steroid 5α-reductase deficiency in man: an inherited form of male pseudohermaphroditism. Science. 1974;186(4170):1213–1215. doi: 10.1126/science.186.4170.1213. [DOI] [PubMed] [Google Scholar]
- Imperato-McGinley & Peterson (1976).Imperato-McGinley J, Peterson RE. Male pseudohermaphroditism: the complexities of male phenotypic development. American Journal of Medicine. 1976;61(2):251–272. doi: 10.1016/0002-9343(76)90175-3. [DOI] [PubMed] [Google Scholar]
- Imperato-McGinley et al. (1985).Imperato-McGinley J, Peterson RE, Gautier T, Arthur A, Shackleton C. Decreased urinary C19 and C21 steroid 5α-metabolites in parents of male pseudohermaphrodites with 5α-reductase deficiency: detection of carriers. Journal of Clinical Endocrinology and Metabolism. 1985;60(3):553–558. doi: 10.1210/jcem-60-3-553. [DOI] [PubMed] [Google Scholar]
- Imperato-McGinley et al. (1990).Imperato-McGinley J, Shackleton C, Orlic S, Stoner E. C19 and C21 5β/5α metabolite ratios in subjects treated with the 5α-reductase inhibitor finasteride: comparison of male pseudohermaphrodites with inherited 5α-reductase deficiency. Journal of Clinical Endocrinology and Metabolism. 1990;70(3):777–782. doi: 10.1210/jcem-70-3-777. [DOI] [PubMed] [Google Scholar]
- ICH (1994).International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Geneva: International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH); 1994. [Google Scholar]
- Irwig (2012a).Irwig MS. Depressive symptoms and suicidal thoughts among former users of finasteride with persistent sexual side effects. Journal of Clinical Psychiatry. 2012a;73(9):1220–1223. doi: 10.4088/JCP.12m07887. [DOI] [PubMed] [Google Scholar]
- Irwig (2012b).Irwig MS. Persistent sexual side effects of finasteride: could they be permanent? Journal of Sexual Medicine. 2012b;9(11):2927–2932. doi: 10.1111/j.1743-6109.2012.02846.x. [DOI] [PubMed] [Google Scholar]
- Irwig (2014).Irwig MS. Androgen levels and semen parameters among former users of finasteride with persistent sexual adverse effects. JAMA Dermatology. 2014;150(2):1361. doi: 10.1001/jamadermatol.2014.1830. [DOI] [PubMed] [Google Scholar]
- Irwig & Kolukula (2011).Irwig MS, Kolukula S. Persistent sexual side effects of finasteride for male pattern hair loss. Journal of Sexual Medicine. 2011;8(6):1747–1753. doi: 10.1111/j.1743-6109.2011.02255.x. [DOI] [PubMed] [Google Scholar]
- Jimenez Cruz, Quecedo Gutierrez & Del Llano Senaris (2003).Jimenez Cruz JF, Quecedo Gutierrez L, Del Llano Senaris J. Finasteride: 10 years of clinical use. Systematic review of the literature. Actas Urologicas Españolas. 2003;27:202–215. doi: 10.1016/s0210-4806(03)72906-4. [DOI] [PubMed] [Google Scholar]
- Khazaie et al. (2015).Khazaie H, Rezaie L, Rezaei Payam N, Najafi F. Antidepressant-induced sexual dysfunction during treatment with fluoxetine, sertraline and trazodone; a randomized controlled trial. General Hospital Psychiatry. 2015;37(1):40–45. doi: 10.1016/j.genhosppsych.2014.10.010. [DOI] [PubMed] [Google Scholar]
- Koch-Weser, Sellers & Zacest (1977).Koch-Weser J, Sellers EM, Zacest R. The ambiguity of adverse drug reactions. European Journal of Clinical Pharmacology. 1977;11:75–78. doi: 10.1007/BF00562895. [DOI] [PubMed] [Google Scholar]
- Kostoff (2016).Kostoff RN. Under-reporting of adverse events in the biomedical literature. Journal of Data and Information Science. 2016;1(4):10–32. doi: 10.20309/jdis.201623. [DOI] [Google Scholar]
- Kothary, Diak & McMahon (2011).Kothary N, Diak I-L, McMahon A. Pharmacovigilance Review; Propecia (Finasteride 1 mg); Sexual Dysfunction. Silver Spring: Department of Health and Human Services, Public Health Service, Food and Drug Administration, Center for Drug Evaluation and Research, Office of Surveillance and Epidemiology; 2011. pp. 1–20. [Google Scholar]
- Kramer et al. (1985).Kramer MS, Hutchinson TA, Naimark L, Contardi R, Flegel KM, Leduc DG. Antibiotic-associated gastrointestinal symptoms in general pediatric outpatients. Pediatrics. 1985;76:365–370. [PubMed] [Google Scholar]
- Langford et al. (1990).Langford HG, Rockhold RW, Wassertheil-Smoller S, Oberman A, Davis BR, Blaufox MD. Effect of weight loss on thiazide produced erectile problems in men. Transactions of the American Clinical and Climatological Association. 1990;101:190–194. [PMC free article] [PubMed] [Google Scholar]
- Linden & Yarnold (2016a).Linden A, Yarnold PR. Combining machine learning and matching techniques to improve causal inference in program evaluation. Journal of Evaluation in Clinical Practice. 2016a;22(6):868–874. doi: 10.1111/jep.12592. [DOI] [PubMed] [Google Scholar]
- Linden & Yarnold (2016b).Linden A, Yarnold PR. Combining machine learning and propensity score weighting to estimate causal effects in multivalued treatments. Journal of Evaluation in Clinical Practice. 2016b;22(6):875–885. doi: 10.1111/jep.12610. [DOI] [PubMed] [Google Scholar]
- Linden & Yarnold (2016c).Linden A, Yarnold PR. Using data mining techniques to characterize participation in observational studies. Journal of Evaluation in Clinical Practice. 2016c;22(6):839–847. doi: 10.1111/jep.12515. [DOI] [PubMed] [Google Scholar]
- Linden & Yarnold (2016d).Linden A, Yarnold PR. Using machine learning to assess covariate balance in matching studies. Journal of Evaluation in Clinical Practice. 2016d;22(6):848–854. doi: 10.1111/jep.12538. [DOI] [PubMed] [Google Scholar]
- Linden & Yarnold (2016e).Linden A, Yarnold PR. Using machine learning to identify structural breaks in single-group interrupted time series designs. Journal of Evaluation in Clinical Practice. 2016e;22(6):855–859. doi: 10.1111/jep.12544. [DOI] [PubMed] [Google Scholar]
- Linden, Yarnold & Nallamothu (2016).Linden A, Yarnold PR, Nallamothu BK. Using machine learning to model dose-response relationships. Journal of Evaluation in Clinical Practice. 2016;22(6):860–867. doi: 10.1111/jep.12573. [DOI] [PubMed] [Google Scholar]
- Loeb et al. (2011).Loeb DF, Lee RS, Binswanger IA, Ellison MC, Aagaard EM. Patient, resident physician, and visit factors associated with documentation of sexual history in the outpatient setting. Journal of General Internal Medicine. 2011;26:887–893. doi: 10.1007/s11606-011-1711-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahony et al. (1998).Mahony MC, Swanlund DJ, Billeter M, Roberts KP, Pryor JL. Regional distribution of 5α-reductase type 1 and type 2 mRNA along the human epididymis. Fertility and Sterility. 1998;69(6):1116–1121. doi: 10.1016/s0015-0282(98)00094-6. [DOI] [PubMed] [Google Scholar]
- Marchetti & Barth (2013).Marchetti PM, Barth JH. Clinical biochemistry of dihydrotestosterone. Annals of Clinical Biochemistry. 2013;50(2):95–107. doi: 10.1258/acb.2012.012159. [DOI] [PubMed] [Google Scholar]
- Melcangi et al. (2013).Melcangi RC, Caruso D, Abbiati F, Giatti S, Calabrese D, Piazza F, Cavaletti G. Neuroactive steroid levels are modified in cerebrospinal fluid and plasma of post-finasteride patients showing persistent sexual side effects and anxious/depressive symptomatology. Journal of Sexual Medicine. 2013;10(10):2598–2603. doi: 10.1111/jsm.12269. [DOI] [PubMed] [Google Scholar]
- Mella et al. (2010).Mella JM, Perret MC, Manzotti M, Catalano HN, Guyatt G. Efficacy and safety of finasteride therapy for androgenetic alopecia: a systematic review. Archives of Dermatology. 2010;146(10):1141–1150. doi: 10.1001/archdermatol.2010.256. [DOI] [PubMed] [Google Scholar]
- Merck (2011).Merck Merck form 10-K. 2011. http://www.merck.com/investors/financials/form-10-k-2011.pdf http://www.merck.com/investors/financials/form-10-k-2011.pdf
- Merck (2014b).Merck Finasteride 1 mg full prescribing information. 2014b. https://www.merck.com/product/usa/pi_circulars/p/propecia/propecia_pi.pdf https://www.merck.com/product/usa/pi_circulars/p/propecia/propecia_pi.pdf
- Merck (2014c).Merck Finasteride 5 mg full prescribing information. 2014c. https://www.merck.com/product/usa/pi_circulars/p/proscar/proscar_pi.pdf https://www.merck.com/product/usa/pi_circulars/p/proscar/proscar_pi.pdf
- Moinpour et al. (2007).Moinpour CM, Darke AK, Donaldson GW, Thompson IM, Jr, Langley C, Ankerst DP, Patrick DL, Ware JE, Jr, Ganz PA, Shumaker SA, Lippman SM, Coltman CA., Jr Longitudinal analysis of sexual function reported by men in the Prostate Cancer Prevention Trial. Journal of the National Cancer Institute. 2007;99(13):1025–1035. doi: 10.1093/jnci/djm023. [DOI] [PubMed] [Google Scholar]
- Mondaini et al. (2007).Mondaini N, Gontero P, Giubilei G, Lombardi G, Cai T, Gavazzi A, Bartoletti R. Finasteride 5 mg and sexual side effects: how many of these are related to a nocebo phenomenon? Journal of Sexual Medicine. 2007;4(6):1708–1712. doi: 10.1111/j.1743-6109.2007.00563.x. [DOI] [PubMed] [Google Scholar]
- Moore (2015).Moore TJ. Finasteride and the uncertainties of establishing harms. JAMA Dermatology. 2015;151(6):585–586. doi: 10.1001/jamadermatol.2015.37. [DOI] [PubMed] [Google Scholar]
- Naessens et al. (2009).Naessens JM, Campbell CR, Huddleston JM, Berg BP, Lefante JJ, Williams AR, Culbertson RA. A comparison of hospital adverse events identified by three widely used detection methods. International Journal of Quality in Health Care. 2009;21(4):301–307. doi: 10.1093/intqhc/mzp027. [DOI] [PubMed] [Google Scholar]
- NIDDK (2015).National Institute of Diabetes and Digestive and Kidney Diseases . Washington, D.C.: US Department of Health and Human Services; 2015. Benign prostatic hyperplasia. [Google Scholar]
- Nebeker et al. (2007).Nebeker JR, Yarnold PR, Soltysik RC, Sauer BC, Sims SA, Samore MH, Rupper RW, Swanson KM, Savitz LA, Shinogle J, Xu W. Developing indicators of inpatient adverse drug events through nonlinear analysis using administrative data. Medical Care. 2007;45(Suppl 2):S81–S88. doi: 10.1097/MLR.0b013e3180616c2c. [DOI] [PubMed] [Google Scholar]
- Nonomura et al. (1990).Nonomura K, Sakakibara N, Demura T, Mori T, Koyanagi T. Androgen binding activity in the spongy tissue of mammalian penis. Journal of Urology. 1990;144:152–155. doi: 10.1016/s0022-5347(17)39399-0. [DOI] [PubMed] [Google Scholar]
- Oelke et al. (2015).Oelke M, Becher K, Castro-Diaz D, Chartier-Kastler E, Kirby M, Wagg A, Wehling M. Appropriateness of oral drugs for long-term treatment of lower urinary tract symptoms in older persons: results of a systematic literature review and international consensus validation process (LUTS-FORTA 2014) Age and Ageing. 2015;44(5):745–755. doi: 10.1093/ageing/afv077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostrander et al. (1998).Ostrander R, Weinfurt KP, Yarnold PR, August GJ. Diagnosing attention deficit disorders with the behavioral assessment system for children and the child behavior checklist: test and construct validity analyses using optimal discriminant classification trees. Journal of Consulting and Clinical Psychology. 1998;66(4):660–672. doi: 10.1037//0022-006x.66.4.660. [DOI] [PubMed] [Google Scholar]
- Oztekin et al. (2012).Oztekin CV, Gur S, Abdulkadir NA, Lokman U, Akdemir AO, Cetinkaya M, Hellstrom WJ. Incomplete recovery of erectile function in rat after discontinuation of dual 5-alpha reductase inhibitor therapy. Journal of Sexual Medicine. 2012;9(7):1773–1781. doi: 10.1111/j.1743-6109.2012.02759.x. [DOI] [PubMed] [Google Scholar]
- Park & Choi (2014).Park T, Choi JY. Efficacy and safety of dutasteride for the treatment of symptomatic benign prostatic hyperplasia (BPH): a systematic review and meta-analysis. World Journal of Urology. 2014;32(4):1093–1105. doi: 10.1007/s00345-014-1258-9. [DOI] [PubMed] [Google Scholar]
- Patel et al. (2015).Patel DP, Schenk JM, Darke A, Myers JB, Brant WO, Hotaling JM. Non-steroidal anti-inflammatory drug (NSAID) use is not associated with erectile dysfunction risk: results from the Prostate Cancer Prevention Trial. BJU International. 2015;117(3):500–506. doi: 10.1111/bju.13264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmusson & Reynolds (1988).Rasmusson GHW, Reynolds GF. 17β-N-Monosubstituted Carbamoyl-4-Aza-5α-Androst-1-en-3-Ones which are Active as Testosterone 5α-Reductase Inhibitors. Rahway, NJ: Merck & Co., Inc; 1988. [Google Scholar]
- Resar, Rozich & Classen (2003).Resar RK, Rozich JD, Classen D. Methodology and rationale for the measurement of harm with trigger tools. Quality and Safety in Health Care. 2003;12(Suppl 2):ii39–ii45. doi: 10.1136/qhc.12.suppl_2.ii39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rittmaster et al. (2008).Rittmaster R, Hahn RG, Ray P, Shannon JB, Wurzel R. Effect of dutasteride on intraprostatic androgen levels in men with benign prostatic hyperplasia or prostate cancer. Urology. 2008;72(4):808–812. doi: 10.1016/j.urology.2008.06.032. [DOI] [PubMed] [Google Scholar]
- Roberts et al. (1999).Roberts JL, Fiedler V, Imperato-McGinley J, Whiting D, Olsen E, Shupack J, Stough D, DeVillez R, Rietschel R, Savin R, Bergfeld W, Swinehart J, Funicella T, Hordinsky M, Lowe N, Katz I, Lucky A, Drake L, Price VH, Weiss D, Whitmore E, Millikan L, Muller S, Gencheff C, Carrington P, Binkowitz B, Kotey P, He W, Bruno K, Jacobsen C, Terranella L, Gormley GJ, Kaufman KD. Clinical dose ranging studies with finasteride, a type 2 5α-reductase inhibitor, in men with male pattern hair loss. Journal of American Academy of Dermatology. 1999;41(4):555–563. doi: 10.1016/s0190-9622(99)70296-3. [DOI] [PubMed] [Google Scholar]
- Roehrborn et al. (2003).Roehrborn CG, Lee M, Meehan A, Waldstreicher J, Group PS. Effects of finasteride on serum testosterone and body mass index in men with benign prostatic hyperplasia. Urology. 2003;62(5):894–899. doi: 10.1016/s0090-4295(03)00661-7. [DOI] [PubMed] [Google Scholar]
- Rosen et al. (1999).Rosen RC, Cappelleri JC, Smith MD, Lipsky J, Pena BM. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. International Journal of Impotence Research. 1999;11(6):319–326. doi: 10.1038/sj.ijir.3900472. [DOI] [PubMed] [Google Scholar]
- Rosen et al. (1997).Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology. 1997;49:822–830. doi: 10.1016/s0090-4295(97)00238-0. [DOI] [PubMed] [Google Scholar]
- Rossi et al. (1998).Rossi P, Menchini Fabris F, Fiorini I, Palego P, Simi S, Rossi B, Sarteschi LM, Carpi A. Comparison between plasma concentrations of testosterone, nitric oxide and endothelin 1-2 in penile and brachial venous blood: preliminary results in men with psychogenic impotence. Biomedicine and Pharmacotherapy. 1998;52(7–8):308–310. doi: 10.1016/s0753-3322(98)80026-7. [DOI] [PubMed] [Google Scholar]
- Schultheiss et al. (2003).Schultheiss D, Badalyan R, Pilatz A, Gabouev AI, Schlote N, Wefer J, von Wasielewski R, Mertsching H, Sohn M, Stief CG, Jonas U. Androgen and estrogen receptors in the human corpus cavernosum penis: immunohistochemical and cell culture results. World Journal of Urology. 2003;21(5):320–324. doi: 10.1007/s00345-003-0371-y. [DOI] [PubMed] [Google Scholar]
- Shiri et al. (2006).Shiri R, Koskimaki J, Hakkinen J, Tammela TL, Auvinen A, Hakama M. Effect of nonsteroidal anti-inflammatory drug use on the incidence of erectile dysfunction. Journal of Urology. 2006;175(5):1812–1815. doi: 10.1016/S0022-5347(05)01000-1. Discussion 1815–1816. [DOI] [PubMed] [Google Scholar]
- Shukla (2011).Shukla C. Dose Response Data for Finasteride. Silver Spring: Department of Health and Human Services PHS, Food and Drug Administration, Clinical Pharmacology and Biopharmaceutics; 2011. [Google Scholar]
- Singh & Avram (2014).Singh MK, Avram M. Persistent sexual dysfunction and depression in finasteride users for male pattern hair loss: a serious concern or red herring? Journal of Clinical Aesthetic Dermatology. 2014;7:51–55. [PMC free article] [PubMed] [Google Scholar]
- Sinnecker et al. (1996).Sinnecker GH, Hiort O, Dibbelt L, Albers N, Dorr HG, Hauss H, Heinrich U, Hemminghaus M, Hoepffner W, Holder M, Schnabel D, Kruse K. Phenotypic classification of male pseudohermaphroditism due to steroid 5α-reductase 2 deficiency. American Journal of Medical Genetics. 1996;63(1):223–230. doi: 10.1002/(SICI)1096-8628(19960503)63:1<223::AID-AJMG39>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- Smart et al. (2008).Smart CM, Nelson NW, Sweet JJ, Bryant FB, Berry DT, Granacher RP, Heilbronner RL. Use of MMPI-2 to predict cognitive effort: a hierarchically optimal classification tree analysis. Journal of International Neuropsychological Society. 2008;14(5):842–852. doi: 10.1017/S1355617708081034. [DOI] [PubMed] [Google Scholar]
- Stoner (1990).Stoner E. The clinical development of a 5α-reductase inhibitor, finasteride. Journal of Steroid Biochemistry and Molecular Biology. 1990;37(3):375–378. doi: 10.1016/0960-0760(90)90487-6. [DOI] [PubMed] [Google Scholar]
- Tacklind et al. (2010).Tacklind J, Fink HA, Macdonald R, Rutks I, Wilt TJ. Finasteride for benign prostatic hyperplasia. Cochrane Database of Systematic Reviews. 2010 doi: 10.1002/14651858.CD006015.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thiele et al. (2005).Thiele S, Hoppe U, Holterhus PM, Hiort O. Isoenzyme type 1 of 5α-reductase is abundantly transcribed in normal human genital skin fibroblasts and may play an important role in masculinization of 5α-reductase type 2 deficient males. European Journal of Endocrinology. 2005;152(6):875–880. doi: 10.1530/eje.1.01927. [DOI] [PubMed] [Google Scholar]
- Thigpen et al. (1993).Thigpen AE, Silver RI, Guileyardo JM, Casey ML, McConnell JD, Russell DW. Tissue distribution and ontogeny of steroid 5α-reductase isozyme expression. Journal of Clinical Investigation. 1993;92(2):903–910. doi: 10.1172/JCI116665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomada et al. (2013).Tomada I, Tomada N, Almeida H, Neves D. Androgen depletion in humans leads to cavernous tissue reorganization and upregulation of Sirt1-eNOS axis. AGE. 2013;35(1):35–47. doi: 10.1007/s11357-011-9328-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres & Ortega (2003).Torres JM, Ortega E. Differential regulation of steroid 5α-reductase isozymes expression by androgens in the adult rat brain. FASEB Journal. 2003;17(11):1428–1433. doi: 10.1096/fj.02-1119com. [DOI] [PubMed] [Google Scholar]
- Tosti et al. (2004).Tosti A, Pazzaglia M, Soli M, Rossi A, Rebora A, Atzori L, Barbareschi M, Benci M, Voudouris S, Vena GA. Evaluation of sexual function with an international index of erectile function in subjects taking finasteride for androgenetic alopecia. Archives of Dermatology. 2004;140(7):857–858. doi: 10.1001/archderm.140.7.857. [DOI] [PubMed] [Google Scholar]
- Tosti, Piraccini & Soli (2001).Tosti A, Piraccini BM, Soli M. Evaluation of sexual function in subjects taking finasteride for the treatment of androgenetic alopecia. Journal of the European Academy of Dermatology and Venereology. 2001;15(5):418–421. doi: 10.1046/j.1468-3083.2001.00315.x. [DOI] [PubMed] [Google Scholar]
- Traish et al. (2015a).Traish AM, Haider KS, Doros G, Haider A. Finasteride, not tamsulosin, increases severity of erectile dysfunction and decreases testosterone levels in men with benign prostatic hyperplasia. Hormone Molecular Biology and Clinical Investigation. 2015a;23(3):85–96. doi: 10.1515/hmbci-2015-0015. [DOI] [PubMed] [Google Scholar]
- Traish et al. (2015b).Traish AM, Melcangi RC, Bortolato M, Garcia-Segura LM, Zitzmann M. Adverse effects of 5α-reductase inhibitors: what do we know, don’t know, and need to know? Reviews in Endocrine and Metabolic Disorders. 2015b;16(3):177–198. doi: 10.1007/s11154-015-9319-y. [DOI] [PubMed] [Google Scholar]
- Traish et al. (2011).Traish AM, Hassani J, Guay AT, Zitzmann M, Hansen ML. Adverse side effects of 5α-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and depression in a subset of patients. Journal of Sexual Medicine. 2011;8(3):872–884. doi: 10.1111/j.1743-6109.2010.02157.x. [DOI] [PubMed] [Google Scholar]
- Trost (2013).Trost L. Side effects of 5-alpha reductase inhibitors: a comprehensive review. Sexual Medicines Review. 2013;1(1):24–41. doi: 10.1002/smrj.3. [DOI] [PubMed] [Google Scholar]
- Tsunemi et al. (2016).Tsunemi Y, Irisawa R, Yoshiie H, Brotherton B, Ito H, Tsuboi R, Kawashima M, Manyak M, Group ARIS. Long-term safety and efficacy of dutasteride in the treatment of male patients with androgenetic alopecia. Journal of Dermatology. 2016;43(9):1051–1058. doi: 10.1111/1346-8138.13310. [DOI] [PubMed] [Google Scholar]
- Vardi & Nini (2007).Vardi M, Nini A. Phosphodiesterase inhibitors for erectile dysfunction in patients with diabetes mellitus. Cochrane Database of Systematic Reviews. 2007 doi: 10.1002/14651858.CD002187.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varothai & Bergfeld (2014).Varothai S, Bergfeld WF. Androgenetic alopecia: an evidence-based treatment update. American Journal of Clinical Dermatology. 2014;15(3):217–230. doi: 10.1007/s40257-014-0077-5. [DOI] [PubMed] [Google Scholar]
- Walsh et al. (1974).Walsh PC, Madden JD, Harrod MJ, Goldstein JL, MacDonald PC, Wilson JD. Familial incomplete male pseudohermaphroditism, type 2. Decreased dihydrotestosterone formation in pseudovaginal perineoscrotal hypospadias. New England Journal of Medicine. 1974;291(18):944–949. doi: 10.1056/NEJM197410312911806. [DOI] [PubMed] [Google Scholar]
- Wessells et al. (2003).Wessells H, Roy J, Bannow J, Grayhack J, Matsumoto AM, Tenover L, Herlihy R, Fitch W, Labasky R, Auerbach S, Parra R, Rajfer J, Culbertson J, Lee M, Bach MA, Waldstreicher J, Group PS. Incidence and severity of sexual adverse experiences in finasteride and placebo-treated men with benign prostatic hyperplasia. Urology. 2003;61(3):579–584. doi: 10.1016/s0090-4295(02)02401-9. [DOI] [PubMed] [Google Scholar]
- Wu et al. (2014).Wu XJ, Zhi Y, Zheng J, He P, Zhou XZ, Li WB, Zhou ZS. Dutasteride on benign prostatic hyperplasia: a meta-analysis on randomized clinical trials in 6460 patients. Urology. 2014;83(3):539–543. doi: 10.1016/j.urology.2013.10.007. [DOI] [PubMed] [Google Scholar]
- Yarnold & Soltysik (2005).Yarnold PR, Soltysik RC. Optimal Data Analysis: A Guidebook with Software for Windows. Washington, DC: American Psychological Association; 2005. [Google Scholar]
- Yarnold & Soltysik (2016).Yarnold PR, Soltysik RC. Maximizing Classification Accuracy. Chicago, IL: ODA Books; 2016. [Google Scholar]
- Yarnold, Soltysik & Bennett (1997).Yarnold PR, Soltysik RC, Bennett CL. Predicting in-hospital mortality of patients with AIDS-related Pneumocystis carinii pneumonia: an example of hierarchically optimal classification tree analysis. Statistics in Medicine. 1997;16(3):1451–1463. doi: 10.1002/(sici)1097-0258(19970715)16:13<1451::aid-sim571>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- Yarnold, Soltysik & Martin (1994).Yarnold PR, Soltysik RC, Martin GJ. Heart rate variability and susceptibility for sudden cardiac death: an example of multivariable optimal discriminant analysis. Statistics in Medicine. 1994;13(10):1015–1021. doi: 10.1002/sim.4780131004. [DOI] [PubMed] [Google Scholar]
- Yin et al. (2015).Yin T, Qiao Z, Li Y, Li D, Jiang M, An C, Wang F, Zuo M, Hu K, Li Q. Comparisons of the efficacy and safety of finasteride and dutasteride for benign prostatic hyperplasia: a network meta-analysis. American Journal of Therapeutics. 2015 doi: 10.1097/MJT.0000000000000326. [Epub ahead of print 3 February 2017] [DOI] [PubMed] [Google Scholar]
- Yuan et al. (2015).Yuan JQ, Mao C, Wong SY, Yang ZY, Fu XH, Dai XY, Tang JL. Comparative effectiveness and safety of monodrug therapies for lower urinary tract symptoms associated with benign prostatic hyperplasia: a network meta-analysis. Medicine. 2015;94(27):e974. doi: 10.1097/MD.0000000000000974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang et al. (2013).Zhang MG, Wang XJ, Shen ZJ, Gao PJ. Long-term oral administration of 5α-reductase inhibitor attenuates erectile function by inhibiting autophagy and promoting apoptosis of smooth muscle cells in corpus cavernosum of aged rats. Urology. 2013;82(3):743.e9–743.e15. doi: 10.1016/j.urology.2013.02.045. [DOI] [PubMed] [Google Scholar]