Abstract
In order to combat the on-going malaria epidemic, discovery of new drug targets remains vital. Proteins that are essential to survival and specific to malaria parasites are key candidates. To survive within host cells, the parasites need to acquire nutrients and dispose of waste products across multiple membranes. Additionally, like all eukaryotes, they must redistribute ions and organic molecules between their various internal membrane bound compartments. Membrane transport proteins mediate all of these processes and are considered important mediators of drug resistance as well as drug targets in their own right. Recently, using advanced experimental genetic approaches and streamlined life cycle profiling, we generated a large collection of Plasmodium berghei gene deletion mutants and assigned essential gene functions, highlighting potential targets for prophylactic, therapeutic, and transmission-blocking anti-malarial drugs. Here, we present a comprehensive orthology assignment of all Plasmodium falciparum putative membrane transport proteins and provide a detailed overview of the associated essential gene functions obtained through experimental genetics studies in human and murine model parasites. Furthermore, we discuss the phylogeny of selected potential drug targets identified in our functional screen. We extensively discuss the results in the context of the functional assignments obtained using gene targeting available to date.
Keywords: drug target, experimental genetics, malaria parasite, membrane transport protein, orthology, phylogeny, Plasmodium
INTRODUCTION
The malaria parasite has adopted a highly complex life cycle involving a continuous switching between vertebrate hosts and anopheline mosquitoes. Within humans, Plasmodium species are obligate intracellular parasites moving through three different life-cycle stages. After an infectious mosquito bite, a single phase of preclinical growth within the host liver cells commences 1. Next, fast proliferating blood-stage parasites are the cause of malaria-associated pathology and severe disease outcome 2. Finally, some of the asexual blood-stage parasites are triggered to develop into male or female gametocytes, which are required for sexual reproduction following a mosquito blood meal 3,4.
Despite a gradual decline in annual malaria cases and deaths, the parasite remains one of the largest global killers. In 2015, WHO reported 150-300 million cases and 438,000 deaths 5. Difficulty in combating the disease is exacerbated by the growing resistance to anti-malarial drugs and the absence of an effective vaccine. The continuous need for new therapeutic anti-malarial drugs is unquestioned. Yet, due to limited availability, there is a much more pressing need for drugs that can prevent infections by acting prophylactically on the liver stage of the parasite, and transmission-blocking compounds, which kill the gametocytes thus helping to prevent the spread of the disease. Chemoprophylaxis is not only important for travellers 6 but also particularly for women in endemic areas in their first or second pregnancy 7. Thus, there is a critical need for novel drugs that may be used prophylactically, therapeutically, or to block transmission 8.
As defined by the Medicines for Malaria Venture in there 2015 annual report 9, ideal medicines for treatment and protection would both be suitable for mass drug administration programs and require a single encounter treatment, or better still a single exposure treatment, to help improve compliance. Treatment of infection should be effective against all life-cycle stages of all five malaria species infecting humans, including resistant strains, and resistance against the new drug should be difficult to achieve.
Generally, it is believed that these features may be best obtained by the combination of at least two active compounds, one fast acting for immediate clearance of the infection and a second, slower-acting compound providing long duration of efficacy. Furthermore, an ideal treatment would be gametocytocidal to prevent the spread of infection to mosquitoes, while sporontocidal or liver-stage activity would provide a prophylactic component. To complete the wish list of the ideal anti-malarial drug, it should also be active against the so-called hypnozoites, the dormant stages of certain malaria parasite species (most notably P. vivax) that are the cause of relapse infection even many years after the infectious mosquito bite. It is obvious that the development of the ideal anti-malarial drug will not be straightforward and will result in a compromise between the long list of desirable attributes and features. In this light, it is particularly important that on-going functional studies of malaria parasite biology continue to highlight potential new drug targets that have important roles in the different life-cycle stages of the parasite and are conserved among all malaria parasite species but are absent from or have diverged significantly in humans, such that compounds acting on these important Plasmodium proteins may do so effectively as well as selectively.
Plasmodium membrane transport proteins (MTP), such as the chloroquine resistance transporter (CRT) and the ATP-binding cassette (ABC) transporter family, including the multidrug resistance proteins (MDR) and the multidrug resistance-associated proteins (MRP), are well known for their roles in anti-malarial drug resistance 10,11. MTPs are also generally considered potential drug targets in their own right 12,13. Spiroindolones and dihydroisoquinolones are new classes of potent anti-malarial drugs, currently under clinical testing, that have both been shown to act via the Plasmodium falciparum cation ATPase, ATP4, causing severe disturbance of Na+ homeostasis in the parasite 14,15,16,17.
To survive and thrive, malaria parasites utilize a range of transport processes to import nutrients, export waste, and redistribute ions and small organic molecules between different sites and organelles 12,18. MTPs of different classes facilitate these processes. Following the functional and phylogenetic classification of MTPs from the Transporter Classification Database (http://www.tcdb.org 19, Plasmodium MTPs can be classified as: α-type channels and β-barrel porins (TCDB Class 1.A/B); P-P-bond hydrolysis-driven transporters, here referred to as pumps (TCDB Class 3.A); porters, including uniporters, symporters, and antiporters (TCDB Class 2.A); and unclassified, putative MTPs.
Experimental genetics is a powerful means to further explore critical functions of Plasmodium MTPs for parasite survival throughout its complex life cycle 20,21. Due to more efficient experimental and computational methods and access to the entire in vivo life cycle, the majority of such studies have been performed using murine malaria model species, notably Plasmodium berghei, and have focussed on single MTPs (Table S1). Two of our most recent studies have more than doubled the number of targeted genes, and have generated loss-of-function mutants in both P. falciparum and P. berghei 22,23. A systematic study of the MDR family demonstrated that four of seven members fulfil essential functions during blood-stage development, highlighting these as potential drug targets 22. While targeting 35 orphan MTPs, Kenthirapalan et al. produced the largest collection of P. berghei knock-out parasites available to date 23. The 29 available mutant lines provide a powerful resource for further studies of malaria parasite transport processes. In addition to highlighting six genes essential for blood-stage survival including five pumps, they also revealed potential prophylactic (MFS6) and transmission-blocking (ZIP1) drug targets 23.
Here, we performed comprehensive orthology profiling by reciprocal Blast of all identified 139 Plasmodium MTPs (consisting of the list published by Martin et al. 24 expanded with newly identified candidates) against a selection of 41 species from the entire breadth of the eukaryotic kingdom. To further validate the potential of the eight newly identified drug targets, we have explored their phylogenetic relationships in detail. These results are discussed extensively in the context of available insights from functional genetics studies of malaria parasite MTPs.
RESULTS AND DISCUSSION
Comprehensive orthology assignments of Plasmodium MTPs
To identify the levels of conservation of the 139 P. falciparum MTPs (Table S1) within the eukaryotic domain, we first performed an extensive orthology assignment using Blast against a set of 41 species (Table S2). We selected a subset of species to represent the enormous diversity in the eukaryotic domain by including sequences from all kingdoms, with an emphasis on protozoan species, commonly used model species, and parasite species of great medical or veterinary importance. These include the clinically second most important human malaria parasite Plasmodium vivax and the murine malaria model parasite P. berghei. Plasmodium species belong to a large monophyletic group of largely obligate intracellular parasites, the Apicomplexa. We included six additional apicomplexan parasites in our analysis: two piroplasms (Babesia bovis and Theileria annulata) that cause cattle fever and like Plasmodium species belong to the Aconoidasida, and four Conoidasida, including parasites of medical (Toxoplasma gondii and Cryptosporidium parvum) and veterinary (Neospora caninum and Eimeria tenella) importance.
Apicomplexan parasites are characterized by a complex at the apical end that is used to penetrate and enter a wide variety of host cells. However, this is not the only peculiar subcellular structure of these eukaryotic single cell parasites. Being eukaryotes, one would naturally expect most of the commonly shared organelles to be present, but while most do retain a single and thoroughly reduced mitochondrion, some have only vague remnants of the cell’s power house in the form of mitosomes 25. An additional organelle of endosymbiotic heritage present in the majority of apicomplexan parasites (though it has been lost in Cryptosporidium species) is the former-photosynthetic apicoplast 26. This plastid is of red algal origin, a trait shared across a variety of highly divergent species, including other alveolates, such as Chromerida and dinoflagellates, but not ciliates. For our analysis, we included four alveolates, including two ciliates and one of the closest relatives of the Apicomplexa, Vitrella brassicaformis.
The true evolutionary relationships between highly diverse protozoan lineages are unresolved and still a matter of dispute but many interesting parallels can be observed. Thus Cryptomonads, Haptophyta, and a significant proportion of Stramenopiles, all harbour plastids of red algal origin, although at present it is not clear whether these plastids originate from single or multiple secondary endosymobiotic events 27,28,29. In our analysis, we included one cryptomonad, Guillardia theta, as well as four stramenopiles including the oomycete Phytophthora infestans. The latter is a plant parasite causing potato blight, which does not harbour a plastid, but it does share another interesting feature with malaria parasites. Both oomycetes and malaria parasites extensively remodel the host they infect by exporting a large repertoire of effector proteins using comparable strategies 30. Further protist representative sequences were taken from two Rhizaria, six Excavata, including three kinetoplastid parasites, and two Amoebozoa.
From the plant kingdom, we took a green and a red alga and the most commonly used model plant Arabidopsis thaliana. Ophistokonts were represented by two fungi (baker’s yeast and the pathogenic Cryptococcus neoformans) and by nine animals, including the parasitic flatworm Schistosoma haematobium, the malaria mosquito Anopheles gambiae, a number of widely used model organisms, and of course mouse and human.
Potential prophylactic drug targets
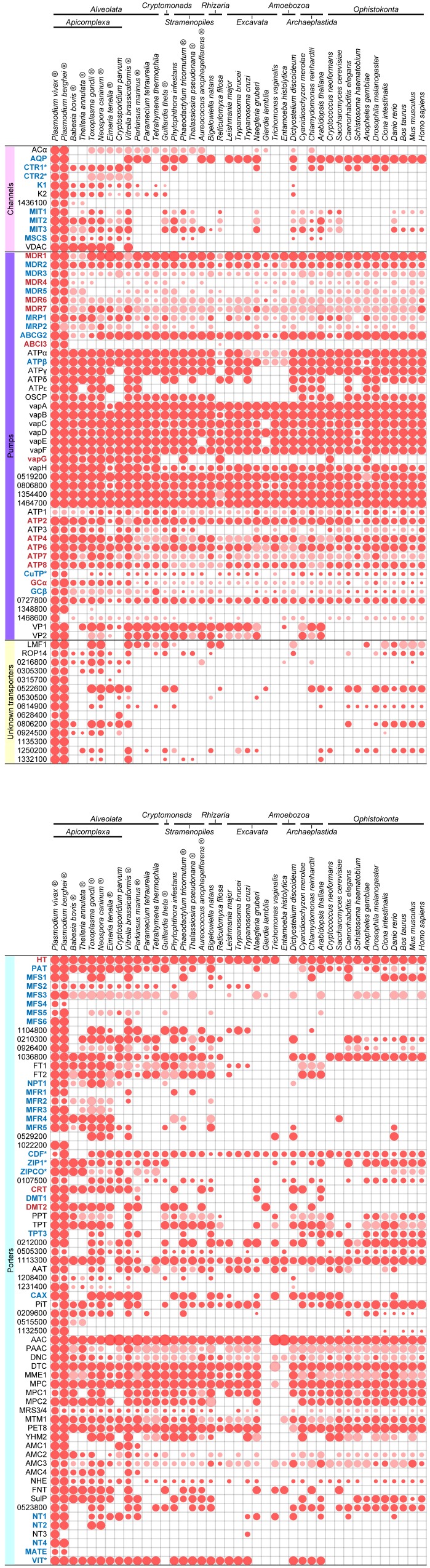
In our recent functional screen of orphan MTPs, we identified a critical role for a major facilitator superfamily member, MFS6, during liver-stage development, in addition to important functions in the blood 23. Though we identified a putative orthologue in V. brassicaformis (Figure 1), MFS6 is largely Plasmodium-specific and appears absent from humans. Its important functions during both liver- and blood-stage development justify further exploration of the protein as a target for compounds with combined prophylactic and therapeutic activity. P. berghei MRP2-deficient parasites demonstrate a complete arrest in the liver, a phenotype that was also observed in P. falciparum counterpart MRPs 31. Like MFS6, MRP2 appears to be largely Plasmodium-specific (Figure 1) providing another promising drug target. However, for these two MTPs both their function and their respective substrates remain unresolved.
Figure 1. FIGURE 1: Orthologies of membrane transport proteins of the malaria parasite.
Extensive amino acid based reciprocal homology searches were performed to establish orthologues of the all Plasmodium channels/pores (TCDB Class 1.A/B 19; pink), pumps (3.A; purple), porters (2.A; cyan), and unclassified, putative MTPs (yellow) in representative species of the entire breadth of the eukaryotic domain. Dot sizes indicate the fraction of the sequence length over which homology was detected; red dots indicate reciprocal orthologues. When a protein has not been given a name, these are indicated by their P. falciparum geneID number. MTPs for which the encoding genes were deleted successfully are indicated in blue, unsuccessful gene deletions suggesting essential functions during blood-stage development are highlighted in red. (®, species harbouring plastids of red algal origin; *, putative heavy metal transporting MTPs)
Evidence is mounting that transport processes of metal ions, in particular of heavy metals such as iron, copper, and zinc, could provide efficient targets for chemoprophylactic treatments. Although the exact mechanisms are unclear, P. berghei liver-stage development is influenced by host iron homeostasis 32,33. Recently, a vacuolar iron transporter was described that plays an important (although not critical) role during liver infection in vivo and in vitro 34. Parasites lacking an alternative zinc-iron permease (ZIPCO) are severely affected in liver-stage development in vitro and show a delay in prepatency of two days 35. A similar delay in prepatency was observed following needle injection of ctr2- sporozoites despite developing normally in culture 23. Interestingly, these parasites were most severely affected during natural transmission by infectious mosquito bites or following subcutaneous injection of the sporozoites. Parasites deficient in the copper channel 1 gene (CTR1) completely failed to transmit, but this was at least in part attributable to a much reduced and delayed sporozoite production, and the infectivity of these sporozoites remains to be determined in more detail to establish whether CTR1 like CTR2 plays an important role in the liver 23. Despite the fact that the putative heavy metal transporting MTPs with a demonstrated important role during the establishment of new infection are not strictly essential, they may still prove interesting targets for prophylactic interventions, since none of the MTPs discussed above appear to have a clear reciprocal orthologue in humans and show only partial sequence matches (Figure 1).
Potential transmission-blocking drug targets
The importance of heavy metal homeostasis appears not to be restricted to mosquito-to-mouse/human transition. Also sexual blood-stage parasites, parasite fertility, and effective colonization of the mosquito midgut appear to be strongly dependent on the correct distribution of these cations.
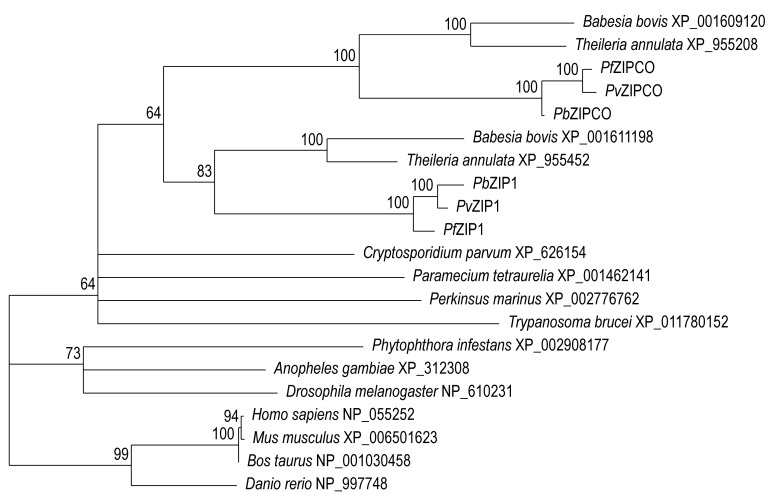
Where the two copper channels may act in copper transport in the liver stages, a copper-transporting P-type ATPase (CuTP) was shown to be central to fertility of both male and female gametes 36. Activation of cutp- male microgametes, a process known as exflagellation, is reduced to ~10% of wild-type and this reduction could be phenocopied using an intracellular copper chelator. Cross-fertilization studies and an even more pronounced reduction in oocyst numbers indicate additional important roles in the female gametes. ZIP1, a paralogue of the liver stage-specific ZIPCO, is crucial for mosquito colonization 23. Exflagellation in zip1- parasites is reduced to naught, which is directly attributable to a nearly complete absence of male gametocyte formation. Since ZIP1-deficient parasites also have a slightly reduced blood-stage multiplication rate, ZIP1 is potentially a very attractive transmission-blocking drug target. Indeed, if a compound would be able to target ZIPCO and ZIP1, this would be a triple-acting drug that could be used prophylactically, therapeutically, and to block spread of the disease. One disadvantage could be that humans harbour fourteen zinc-iron permeases 37, although only one of these bears any sequence similarity to the Plasmodium copies. Nevertheless, caution is needed to ensure that the compound is sufficiently parasite-specific. Phylogenetic profiling of the identified ZIP sequences supports a relatively recent gene duplication at the root of the Aconoidasida (Figure 2), and the relatively long distance from the vertebrate ZIPs suggest that specific targeting of the Plasmodium ZIPs might be feasible.
Figure 2. FIGURE 2: Phylogenetic tree iron-zinc permeases.
ZIPCO and ZIP1 are two zinc-iron permeases, the first is important for mosquito-to-mouse transition, while the second is vital for male gametocyte formation and transmission to the malaria mosquito. This is a maximum likelihood tree of the orthologues found in a variety of eukaryotes, including humans. Nodes with support values <50 are left unresolved. The aconoidasidan paralogues ZIPCO and ZIP1 resolve with good support values, suggesting that the genes duplicated after differentiation of the Aconoidasida.
Other MTPs with roles during mouse/human-to-mosquito host transition have been identified, such as the Ca2+/H+ exchanger (CAX) 38, a cation diffusion factor or putative zinc transporter (CDF) 23, and the pantothenate transporter 23,39. Unfortunately, not a single MTP has been identified that is critical for both male and female gametocyte formation, although it remains to be tested if zip1- female gametocytes remain fertile or have lost the capacity to reproduce.
Potential therapeutic drug targets
Two types of channels have been discussed as potential therapeutic drug targets. Two potassium channels (K1 and K2) were refractory to gene deletion in P. falciparum 40. However, absence of supporting evidence that this was not merely due to the technical difficulties of targeting P. falciparum genes and the fact that PbK1 could be readily deleted 41 suggest that these channels may not be strictly necessary for blood-stage survival. The suitability of aquaglyceroporin (AQP) has also been subject of controversy. Initial studies demonstrated that blood-stage growth of aqp- parasites was strongly affected 42. Independently, we were only able to replicate a minor defect in Swiss-Webster mice 43. Using a sensitive flow cytometry - based method in NMRI mice, we saw no difference in growth rates of WT and aqp- parasites either growing in direct competition or in individual mice 44. The relevant difference between NMRI and Swiss-Webster mice that may lead to this observation is unclear.
Interestingly, a vast majority of the currently identified resistance markers, e.g. the ABC family 11, as well as the single validated druggable MTP, ATP4 16,17, are primary active transporters that require ATP to fuel their activity. As the parasite invests energy in their functioning, it is perhaps not surprising that many pumps play crucial roles at some stages during the parasite’s life cycle. Indeed, of all nineteen targeted pumps, eleven were shown to be refractory to gene deletion (Table S1). In addition to the four MDRs (MDR1, MDR4, MDR6, and MDR7) 22, these include the putative cation transporting ATPase, ATP4, (S. Kenthirapalan, K. Matuschewski, T.W.A. Kooij, unpublished data), ATP6 45 and the V-type proton ATPase subunit G 46, ABCI3, and four predicted aminophospholipid transporters 23.
Our orthology profiling indicates that the ABC transporters, with the exception of MDR1 (and to lesser extent MDR2), are poorly conserved across the eukaryotic kingdom, further highlighting their potential as anti-malarial drug targets (Figure 1). ABCI3 appears to be a unique, Plasmodium-specific ABC family member and is characterized by the presence of two transmembrane domains consisting of multiple transmembrane helices interspersed by a single nucleotide-binding domain. An extensive phylogenetic profiling of all ABC transporters identified in 16 eukaryotic and 55 prokaryotic genomes assigned PfABCI3 to a poorly supported clade together with sequences from two archaea from two different kingdoms and sequences from five bacteria from four different phyli 47. Thus, the orthology and phylogeny of ABCI3, along with demonstrated essential function during blood-stage development, strengthen its potential as a therapeutic drug target.
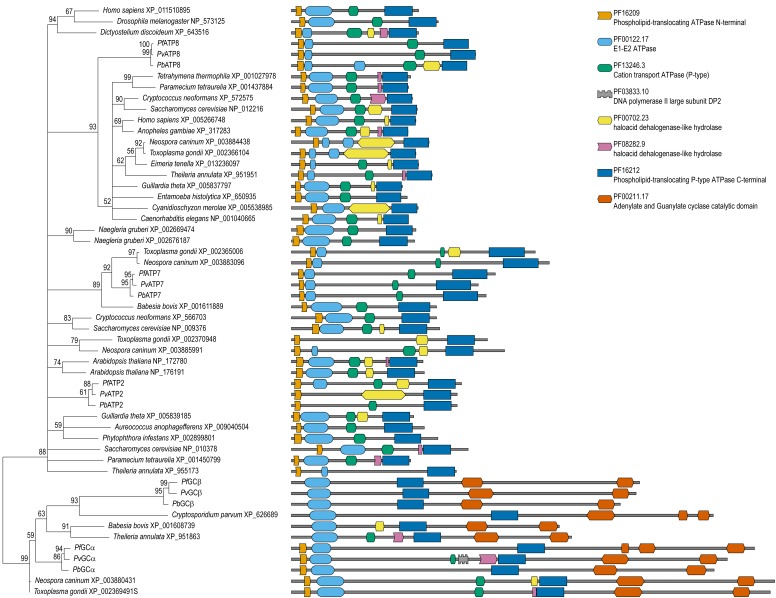
Another class of primary active transporters that was highlighted for its druggable potential consists of putative aminophospholipid-transporting P4-type ATPases, from hereon referred to as flippases 23. In addition to ATP2, ATP7, and ATP8, these also include two putatively bifunctional proteins that also harbour guanylyl cyclase (GC) activity 48. GCα, like the other flippases, was essential for blood-stage development, whereas GCβ plays a critical role in colonization of the mosquito midgut 23,49,50. While parts of the GCs are conserved, no bifunctional orthologues were found outside the apicomplexan clade (Figure 1). The phylogenetic tree of the flippases confirmed this notion (Figure 3). Orthology and phylogenetic profiles further indicate that ATP2 and ATP8 are rather well conserved including human orthologues, whereas ATP7 is largely apicomplexan-specific (Figure 1). Combined, these data suggest that ATP7 and GCα may form attractive targets for novel anti-malarial compounds. Of note, for one P. falciparum gene encoding a putative flippase (PF3D7_1468600), no orthologue exists in P. berghei and a possible essential role has not yet been established.
Figure 3. FIGURE 3: Phylogenetic tree of putative flippases.
Four of five Plasmodium berghei putative aminophospholipid-transporting P4-type ATPases were shown to be essential for blood-stage development in vivo. This is a maximum likelihood tree based on the complete sequences of the identified orthologues along with models of all the domain architectures. Nodes with support values <50 are left unresolved. This unrooted tree shows low resolution at the base, making it difficult to interpret their phylogenetic history despite the fact that the proteins share their structures. ATP8 forms a well-supported clade with orthologues from a variety of species, while the apicomplexan branches with poor orthology assignments (GCα, GCβ, and ATP7) resolve well supporting the candidacy of GCα and ATP7 as therapeutic drug targets.
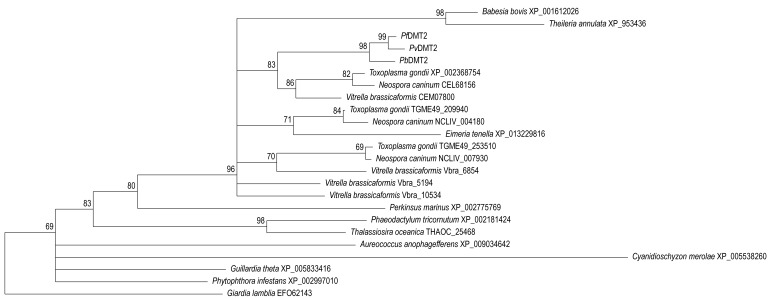
Of the largest group of MTPs, the porters (TCDB Class 2.A) 19, 28 genes have been targeted in P. berghei, only three times without success (Table S1). In addition to the well-studied CRT 51, these include a hexose transporter HT 52, which is well conserved including in humans (Figure 1), and the putative drug metabolite transporter DMT2 23. The latter presents a particularly interesting case considering the orthology discovery. Orthologues were identified in all apicomplexan parasites, with the exception of Cryptosporidium, in other alveolates, but not ciliates, and in the cryptomonad, G. theta, and most stramenopiles. Most species have a single DMT2 but T. gondii, N. caninum, and V. brassicaformis have multiple copies. While the exact phylogenetic relationships of the alveolates, cryptomonads, and stramenopiles is still a topic of debate, many of these chromalveolates, as they are commonly referred to, harbour a plastid of red algal origin. Ciliates and Cryptosporidium species do not have such a plastid and thus it is tempting to speculate that DMT2 localizes to this endosymbiotic organelle. Indeed, the red alga Cyanidioschizon merolae appears to harbour a sequence with a very weak similarity that was initially just below the cut-off E-value applied to establish significant homologies. However, the presence of a rather well conserved copy in P. infestans, that lacks a relic plastid, and presence of weak similarities in three other unrelated eukaryotes (Giardia lamblia, Dictyostelium discoideum, and Chlamydomonas reinhardtii) suggest that the evolutionary history of this gene may be more complicated. When building a maximum likelihood tree for all identified homologues, including the few hits from unrelated species, the tree does not resolve well (data not shown). However, when only including sequences from the chromalveolates and the red alga, the tree is well-supported showing phylum-specific clades with Cyanidioschizon merolae as the outmost group of the tree (data not shown). Attempts to include other sequences as outgroup, e.g. Plasmodium DMT1 sequences, were unsuccessful due to a lack in sequence similarity and the consequently poorly resolving sequence alignments. The most consistent results were obtained when using a single DMT2 homologue of one of the three unrelated species as an outgroup (Figure 4).
Figure 4. FIGURE 4: Phylogenetic tree of drug metabolite transporter 2 (DMT2).
Plasmodium berghei DMT2 was shown to be essential for blood-stage development in vivo. This is a maximum likelihood tree of all homologues found in chromalveolate species and the red alga Cyanidioschyzon merolae using the Giardia lamblia homologue as an outgroup. Nodes with support values <50 are left unresolved. Apicomplexan DMT2 sequences, including the different paralogous sequences, form a distinct and well-supported clade, while the red algal distant homologue and the majority of other chromalveolates sequences sit unresolved at the base of this clade.
Despite the significant uncertainties and remaining unresolved questions about the origin of DMT2, these data could well suggest that this essential MTP is localized in the parasite’s apicoplast. Our initial localization studies were hampered by very low expression levels of the protein and hence difficult to interpret, but appeared to indicate an intraparasitic staining including a specific, small structure that may well be the apicoplast 23. In blood-stage parasites, the single critical role of the apicoplast was shown to be the production of isopentenyl pyrophosphate (IPP) 53 and it is tempting to speculate that DMT2 is the dedicated IPP transport protein. However, DMT2 may also be involved in critical processes for the maintenance of the organelle and the IPP biosynthesis pathway, e.g. through the import of sulphur or iron into the organelle for essential iron-sulphur cluster biosynthesis 54.
Conclusions
In conclusion, our extensive orthology assignment and phylogenetic profiling, in combination with published experimental genetics studies, particularly in the murine malaria model parasite P. berghei, support the candidacy of a number of prophylactic, therapeutic, and transmission-blocking drug targets. Further studies into the biochemical and structural properties of these MTPs are required and deserve prioritization.
MATERIALS AND METHODS
Identification of putative orthologues
We first selected candidate protein sequences by homology search using Blast (blastp, version 2.2.29+) 55, against a set of pre-selected proteomes (Table S2) applying a threshold for being considered significant of E=5e-4. Next, for each candidate, we performed a reciprocal Blast search against the Plasmodium falciparum genome. To circumvent incomplete annotations of the genomes, we have additionally searched each query against the NCBI non-redundant (NR) database from March 19th, 2016. Hits on the same sequence were analysed and the overall coverage of the identified homology was calculated as the fraction of the query length. Candidates which returned the same protein as the original query (i.e., reciprocal blast hits) were kept as putative orthologues, while remaining hits were retained as unspecified homologues. We further inspected the putative homologues using the RADS algorithm 56 and the Needleman-Wunsch global alignment algorithm.
Phylogenetic reconstruction
Groups of putative orthologues for selected proteins were aligned using the programme Clustal Omega v. 1.2.1 57 with default parameters. The alignments were then manually inspected and regions of low coverage and poor conservation were masked. Maximum likelihood phylogeny reconstruction was performed with the TreePuzzle 58 with 10,000 iterations and a mixed model of rate homogeneity.
Domain analysis
Domains of protein sequences were detected with hmmscan from the HMMer package v. 3.1b1 (http://hmmer.org) and the HMM profile collection from the PfamA database (October 2015). Significant overlaps were solved by E-value precedence. Domain architectures were analysed in combination with phylogenetic trees using the DoMosaic programme 59.
SUPPLEMENTAL MATERIAL
All supplemental data for this article are also available online at http://microbialcell.com/researcharticles/phylogenetic-profiles-of-all-membrane-transport-proteins-of-the-malaria-parasite-highlight-new-drug-targets/.
Funding Statement
This work was supported by the Max Planck Society and by the Netherlands Organisation for Scientific Research (NWO-VIDI 864.13.009). We would like to thank Gayle McEwen for her help with the manuscript.
References
- 1.Lindner SE, Miller JL, Kappe SHI. Malaria parasite pre-erythrocytic infection: preparation meets opportunity. Cell Microbiol. 2012;14(3):316–324. doi: 10.1111/j.1462-5822.2011.01734.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Miller LH, Ackerman HC, Su X-Z, Wellems TE. Malaria biology and disease pathogenesis: insights for new treatments. Nat Med. 2013;19(2):156–167. doi: 10.1038/nm.3073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Josling GA, Llinás M. Sexual development in Plasmodium parasites: knowing when it's time to commit. Nat Rev Microbiol. 2015;13(9):573–587. doi: 10.1038/nrmicro3519. [DOI] [PubMed] [Google Scholar]
- 4.Kooij TWA, Matuschewski K. Triggers and tricks of Plasmodium sexual development. Curr Opin Microbiol. 2007;10(6):547–553. doi: 10.1016/j.mib.2007.09.015. [DOI] [PubMed] [Google Scholar]
- 5.World Health Organization. World malaria report 2015. 2015 Available at: http://www.who.int/malaria/publications/world-malaria-report- 2015/report/en/. [Google Scholar]
- 6.Schlagenhauf P, Petersen E. Malaria chemoprophylaxis: strategies for risk groups. Clin Microbiol Rev. 2008;21(3):466–472. doi: 10.1128/CMR.00059-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Radeva-Petrova D, Kayentao K, Kuile ter FO, Sinclair D, Garner P. Drugs for preventing malaria in pregnant women in endemic areas: any drug regimen versus placebo or no treatment. Cochrane Database Syst Rev. 2014;10:CD000169. doi: 10.1002/14651858.CD000169.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wells TNC, van Huijsduijnen RH, Van Voorhis WC. Malaria medicines: a glass half full? Nat Rev Drug Discov. 2015;14(6):424–442. doi: 10.1038/nrd4573. [DOI] [PubMed] [Google Scholar]
- 9.Medicines for Malaria Venture. MMV annual report 2015. 2015 Available at: http://www.mmv.org/newsroom/publications/annual-report-2015. [Google Scholar]
- 10.Ecker A, Lehane AM, Clain J, Fidock DA. PfCRT and its role in antimalarial drug resistance. Trends Parasitol. 2012;28(11):504–514. doi: 10.1016/j.pt.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koenderink JB, Kavishe RA, Rijpma SR, Russel FGM. The ABCs of multidrug resistance in malaria. Trends Parasitol. 2010;26(9):440–446. doi: 10.1016/j.pt.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 12.Kirk K, Lehane AM. Membrane transport in the malaria parasite and its host erythrocyte. Biochem J. 2014;457(1):1–18. doi: 10.1042/BJ20131007. [DOI] [PubMed] [Google Scholar]
- 13.Kirk K. Channels and transporters as drug targets in the Plasmodium-infected erythrocyte. Acta Trop. 2004;89(3):285–298. doi: 10.1016/j.actatropica.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 14.Rottmann M, McNamara CW, Yeung BKS, Lee MCS, Zou B, Russell B, Seitz P, Plouffe DM, Dharia NV, Tan J, Cohen SB, Spencer KR, González-Páez GE, Lakshminarayana SB, Goh A, Suwanarusk R, Jegla T, Schmitt EK, Beck H-P, Brun R, Nosten F, Renia L, Dartois V, Keller TH, Fidock DA, Winzeler EA, Diagana TT. Spiroindolones, a potent compound class for the treatment of malaria. Science. 2010;329(5996):1175–1180. doi: 10.1126/science.1193225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yeung BKS, Zou B, Rottmann M, Lakshminarayana SB, Ang SH, Leong SY, Tan J, Wong J, Keller-Maerki S, Fischli C, Goh A, Schmitt EK, Krastel P, Francotte E, Kuhen K, Plouffe D, Henson K, Wagner T, Winzeler EA, Petersen F, Brun R, Dartois V, Diagana TT, Keller TH. Spirotetrahydro β-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J Med Chem. 2010;53(14):5155–5164. doi: 10.1021/jm100410f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Spillman NJ, Allen RJW, McNamara CW, Yeung BKS, Winzeler EA, Diagana TT, Kirk K. Na+ regulation in the malaria parasite Plasmodium falciparum involves the cation ATPase PfATP4 and is a target of the spiroindolone antimalarials. Cell Host Microbe. 2013;13(2):227–237. doi: 10.1016/j.chom.2012.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jiménez-Díaz MB, Ebert D, Salinas Y, Pradhan A, Lehane AM, Myrand-Lapierre M-E, O'Loughlin KG, Shackleford DM, Justino de Almeida M, Carrillo AK, Clark JA, Dennis ASM, Diep J, Deng X, Duffy S, Endsley AN, Fedewa G, Guiguemde WA, Gómez MG, Holbrook G, Horst J, Kim CC, Liu J, Lee MCS, Matheny A, Martínez MS, Miller G, Rodríguez-Alejandre A, Sanz L, Sigal M, Spillman NJ, Stein PD, Wang Z, Zhu F, Waterson D, Knapp S, Shelat A, Avery VM, Fidock DA, Gamo F-J, Charman SA, Mirsalis JC, Ma H, Ferrer S, Kirk K, Angulo-Barturen I, Kyle DE, Derisi JL, Floyd DM, Guy RK. (+)-SJ733, a clinical candidate for malaria that acts through ATP4 to induce rapid host-mediated clearance of Plasmodium. Proc Natl Acad Sci USA. 2014;111(50):E5455–E5462. doi: 10.1073/pnas.1414221111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kirk K. Ion regulation in the malaria parasite. Annu Rev Microbiol. 2015;69(1):341–359. doi: 10.1146/annurev-micro-091014-104506. [DOI] [PubMed] [Google Scholar]
- 19.Saier MH, Reddy VS, Tamang DG, Västermark A. The transporter classification database. Nucleic Acids Res. 2014;42(Database issue):D251–D258. doi: 10.1093/nar/gkt1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matz JM, Kooij TWA. Towards genome-wide experimental genetics in the in vivo malaria model parasite Plasmodium berghei. Pathog Glob Health. 2015;109(2):46–60. doi: 10.1179/2047773215Y.0000000006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.de Koning-Ward TF, Gilson PR, Crabb BS. Advances in molecular genetic systems in malaria. Nat Rev Microbiol. 2015;13(6):373–387. doi: 10.1038/nrmicro3450. [DOI] [PubMed] [Google Scholar]
- 22.Rijpma SR, van der Velden M, Annoura T, Matz JM, Kenthirapalan S, Kooij TWA, Matuschewski K, van Gemert G-J, van de Vegte-Bolmer M, Siebelink-Stoter R, Graumans W, Ramesar J, Klop O, Russel FGM, Sauerwein RW, Janse C, Franke-Fayard BMD, Koenderink JB. Vital and dispensable roles of Plasmodium multidrug resistance transporters during blood- and mosquito-stage development. Mol Microbiol. 2016;101(1):78–91. doi: 10.1111/mmi.13373. [DOI] [PubMed] [Google Scholar]
- 23.Kenthirapalan S, Waters AP, Matuschewski K, Kooij TWA. Functional profiles of orphan membrane transporters in the life cycle of the malaria parasite. Nat Commun. 2016;7:10519. doi: 10.1038/ncomms10519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Martin RE, Ginsburg H, Kirk K. Membrane transport proteins of the malaria parasite. Mol Microbiol. 2009;74(3):519–528. doi: 10.1111/j.1365-2958.2009.06863.x. [DOI] [PubMed] [Google Scholar]
- 25.Vaidya AB, Mather MW. Mitochondrial evolution and functions in malaria parasites. Annu Rev Microbiol. 2009;63:249–267. doi: 10.1146/annurev.micro.091208.073424. [DOI] [PubMed] [Google Scholar]
- 26.van Dooren GG, Striepen B. The algal past and parasite present of the apicoplast. Annu Rev Microbiol. 2013;67(1):271–289. doi: 10.1146/annurev-micro-092412-155741. [DOI] [PubMed] [Google Scholar]
- 27.Archibald JM. Endosymbiosis and Eukaryotic Cell Evolution. Curr Biol. 2015;25(19):R911–R921. doi: 10.1016/j.cub.2015.07.055. [DOI] [PubMed] [Google Scholar]
- 28.Zimorski V, Ku C, Martin WF, Gould SB. Endosymbiotic theory for organelle origins. Curr Opin Microbiol. 2014;22:38–48. doi: 10.1016/j.mib.2014.09.008. [DOI] [PubMed] [Google Scholar]
- 29.Keeling PJ. The number, speed, and impact of plastid endosymbioses in eukaryotic evolution. Annu Rev Plant Biol. 2013;64(1):583–607. doi: 10.1146/annurev-arplant-050312-120144. [DOI] [PubMed] [Google Scholar]
- 30.Jiang RHY, Stahelin RV, Bhattacharjee S, Haldar K. Eukaryotic virulence determinants utilize phosphoinositides at the ER and host cell surface. Trends Microbiol. 2013;21(3):145–156. doi: 10.1016/j.tim.2012.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rijpma SR, van der Velden M, González-Pons M, Annoura T, van Schaijk BCL, van Gemert G-J, van den Heuvel JJMW, Ramesar J, Chevalley-Maurel S, Ploemen IHJ, Khan SM, Franetich J-F, Mazier D, de Wilt JHW, Serrano AE, Russel FGM, Janse C, Sauerwein RW, Koenderink JB, Franke-Fayard BMD. Multidrug ATP-binding cassette transporters are essential for hepatic development of Plasmodium sporozoites. Cell Microbiol. 2016;18(3):369–383. doi: 10.1111/cmi.12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Portugal S, Carret C, Recker M, Armitage AE, Gonçalves LA, Epiphanio S, Sullivan D, Roy C, Newbold CI, Drakesmith H, Mota MM. Host-mediated regulation of superinfection in malaria. Nat Med. 2011;17(6):732–737. doi: 10.1038/nm.2368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ferrer P, Castillo-Neyra R, Roy CN, Sullivan DJ. Dynamic control of hepatic Plasmodium numbers by hepcidin despite elevated liver iron during iron supplementation. Microbes Infect. 2016;18(1):48–56. doi: 10.1016/j.micinf.2015.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Slavic K, Krishna S, Lahree A, Bouyer G, Hanson KK, Vera I, Pittman JK, Staines HM, Mota MM. A vacuolar iron-transporter homologue acts as a detoxifier in Plasmodium. Nat Commun. 2016;7:10403. doi: 10.1038/ncomms10403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sahu T, Boisson B, Lacroix C, Bischoff E, Richier Q, Formaglio P, Thiberge S, Dobrescu I, Ménard R, Baldacci P. ZIPCO, a putative metal ion transporter, is crucial for Plasmodium liver-stage development. EMBO Mol Med. 2014;6(11):1387–1397. doi: 10.15252/emmm.201403868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kenthirapalan S, Waters AP, Matuschewski K, Kooij TWA. Copper-transporting ATPase is important for malaria parasite fertility. Mol Microbiol. 2014;91(2):315–325. doi: 10.1111/mmi.12461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kambe T, Hashimoto A, Fujimoto S. Current understanding of ZIP and ZnT zinc transporters in human health and diseases. Cell Mol Life Sci. 2014;71(17):3281–3295. doi: 10.1007/s00018-014-1617-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Guttery DS, Pittman JK, Frénal K, Poulin B, McFarlane LR, Slavic K, Wheatley SP, Soldati-Favre D, Krishna S, Tewari R, Staines HM. The Plasmodium berghei Ca2+/H+ exchanger, PbCAX, is essential for tolerance to environmental Ca2+ during sexual development. PLoS Pathog. 2013;9(2):e1003191. doi: 10.1371/journal.ppat.1003191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hart RJ, Lawres L, Fritzen E, Ben Mamoun C, Aly ASI. Plasmodium yoelii vitamin B5 pantothenate transporter candidate is essential for parasite transmission to the mosquito. Sci Rep. 2014;4:5665. doi: 10.1038/srep05665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Waller KL, McBride SM, Kim K, McDonald TV. Characterization of two putative potassium channels in Plasmodium falciparum. Malar J. 2008;7(1):19. doi: 10.1186/1475-2875-7-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ellekvist P, Maciel J, Mlambo G, Ricke CH, Colding H, Klaerke DA, Kumar N. Critical role of a K+ channel in Plasmodium berghei transmission revealed by targeted gene disruption. Proc Natl Acad Sci USA. 2008;105(17):6398–6402. doi: 10.1073/pnas.0802384105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Promeneur D, Liu Y, Maciel J, Agre P, King LS, Kumar N. Aquaglyceroporin PbAQP during intraerythrocytic development of the malaria parasite Plasmodium berghei. Proc Natl Acad Sci USA. 2007;104(7):2211–2216. doi: 10.1073/pnas.0610843104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kenthirapalan S, Waters AP, Matuschewski K, Kooij TWA. Flow cytometry-assisted rapid isolation of recombinant Plasmodium berghei parasites exemplified by functional analysis of aquaglyceroporin. Int J Parasitol. 2012;42(13-14):1185–1192. doi: 10.1016/j.ijpara.2012.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Matz JM, Matuschewski K, Kooij TWA. Two putative protein export regulators promote Plasmodium blood stage development in vivo. Mol Biochem Parasitol. 2013;191(1):44–52. doi: 10.1016/j.molbiopara.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 45.Pulcini S, Staines HM, Pittman JK, Slavic K, Doerig C, Halbert J, Tewari R, Shah F, Avery MA, Haynes RK, Krishna S. Expression in yeast links field polymorphisms in PfATP6 to in vitro artemisinin resistance and identifies new inhibitor classes. J Infect Dis. 2013;208(3):468–478. doi: 10.1093/infdis/jit171. [DOI] [PubMed] [Google Scholar]
- 46.Fonager J, Pasini EM, Braks JAM, Klop O, Ramesar J, Remarque EJ, Vroegrijk IOCM, van Duinen SG, Thomas AW, Khan SM, Mann M, Kocken CHM, Janse C, Franke-Fayard BMD. Reduced CD36-dependent tissue sequestration of Plasmodium-infected erythrocytes is detrimental to malaria parasite growth in vivo. J Exp Med. 2012;209(1):93–107. doi: 10.1084/jem.20110762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xiong J, Feng J, Yuan D, Zhou J, Miao W. Tracing the structural evolution of eukaryotic ATP binding cassette transporter superfamily. Sci Rep. 2015;5:16724. doi: 10.1038/srep16724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Carucci DJ, Witney AA, Muhia DK, Warhurst DC, Schaap P, Meima M, Li JL, Taylor MC, Kelly JM, Baker DA. Guanylyl cyclase activity associated with putative bifunctional integral membrane proteins in Plasmodium falciparum. J Biol Chem. 2000;275(29):22147–22156. doi: 10.1074/jbc.M001021200. [DOI] [PubMed] [Google Scholar]
- 49.Moon RW, Taylor CJ, Bex C, Schepers R, Goulding D, Janse C, Waters AP, Baker DA, Billker O. A cyclic GMP signalling module that regulates gliding motility in a malaria parasite. PLoS Pathog. 2009;5(9):e1000599. doi: 10.1371/journal.ppat.1000599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hirai M, Arai M, Kawai S, Matsuoka H. PbGCβ is essential for Plasmodium ookinete motility to invade midgut cell and for successful completion of parasite life cycle in mosquitoes. J Biochem. 2006;140(5):747–757. doi: 10.1093/jb/mvj205. [DOI] [PubMed] [Google Scholar]
- 51.Ecker A, Lakshmanan V, Sinnis P, Coppens I, Fidock DA. Evidence that mutant PfCRT facilitates the transmission to mosquitoes of chloroquine-treated Plasmodium gametocytes. J Infect Dis. 2011;203(2):228–236. doi: 10.1093/infdis/jiq036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Slavic K, Straschil U, Reininger L, Doerig C, Morin C, Tewari R, Krishna S. Life cycle studies of the hexose transporter of Plasmodium species and genetic validation of their essentiality. Mol Microbiol. 2010;75(6):1402–1413. doi: 10.1111/j.1365-2958.2010.07060.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yeh E, Derisi JL. Chemical rescue of malaria parasites lacking an apicoplast defines organelle function in blood-stage Plasmodium falciparum. PLoS Biol. 2011;9(8):e1001138. doi: 10.1371/journal.pbio.1001138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Haussig JM, Matuschewski K, Kooij TWA. Identification of vital and dispensable sulfur utilization factors in the Plasmodium apicoplast. PLoS ONE. 2014;9(2):e89718. doi: 10.1371/journal.pone.0089718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL. BLAST+: architecture and applications. BMC Bioinformatics. 2009;10(1):421. doi: 10.1186/1471-2105-10-421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Terrapon N, Weiner J, Grath S, Moore AD, Bornberg-Bauer E. Rapid similarity search of proteins using alignments of domain arrangements. Bioinformatics. 2014;30(2):274–281. doi: 10.1093/bioinformatics/btt379. [DOI] [PubMed] [Google Scholar]
- 57.Sievers F, Wilm A, Dineen D, Gibson TJ, Karplus K, Li W, Lopez R, McWilliam H, Remmert M, Söding J, Thompson JD, Higgins DG. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol. 2011;7(1):539–539. doi: 10.1038/msb.2011.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Schmidt HA, Strimmer K, Vingron M, Haeseler von A. TREE-PUZZLE: maximum likelihood phylogenetic analysis using quartets and parallel computing. Bioinformatics. 2002;18(3):502–504. doi: 10.1093/bioinformatics/18.3.502. [DOI] [PubMed] [Google Scholar]
- 59.Moore AD, Held A, Terrapon N, Weiner J, Bornberg-Bauer E. DoMosaics: software for domain arrangement visualization and domain-centric analysis of proteins. Bioinformatics. 2014;30(2):282–283. doi: 10.1093/bioinformatics/btt640. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.