Abstract
Background and Objectives
The paradoxical rise in overall and cancer-specific mortality despite increased detection and treatment of Renal Cell Carcinoma (RCC) is termed “treatment disconnect.” We reassess this phenomenon by evaluating impact of missing data and rising incidence on mortality trends.
Research Design, Subjects, and Measures
Using Surveillance, Epidemiology, and End Results (SEER) data, we identified patients with RCC diagnosis from 1973–2011. We estimated mortality rates by tumor size after accounting for lags from diagnosis to death using multiple imputations for missing data from 1983. Mortality rates were estimated irrespective of tumor size after adjustment for prior cumulative incidence using ridge regression.
Results
78,891 patients met inclusion criteria. Of 70,212 patients diagnosed since 1983, 10.4% had missing data. Significant attenuation in cancer-specific mortality was noted from 1983–2011 when comparing observed to imputed rates: Δobs0.05 versus Δimp0.10 (p=0.001, <2cm tumors); Δobs0.29 versus Δimp0.18 (p=0.005, 2–4cm tumors); Δobs0.46 versus Δimp-0.20 (p<0.001, 4–7cm tumors); Δobs0.93 versus Δimp-0.15 (p<0.001, >7 cm tumors). Holding incidence of RCC constant to 2011 rates, temporal increase in overall mortality for all patients was attenuated (p<0.001) when comparing observed estimates (3.9 to 6.8) to 2011 adjusted estimates (5.9 to 7.1), suggesting that rapidly rising incidence may influence reported overall mortality trends. These findings were supported by assessment of mortality to incidence ratio trends.
Conclusions
Missing data and rising incidence may contribute substantially to the “treatment disconnect” phenomenon when examining mortality rates in RCC using tumor registry data. Caution is advised when basing clinical and policy decisions on these data.
Keywords: Incidence, Mortality, Renal Cell Carcinoma, Treatment Disconnect
Introduction
More than 62,000 men and women will be diagnosed with kidney cancer in the United States in 2016 alone, and over 14,000 will die of their disease.[1] Cross sectional imaging has led to a rise in incidence over the last three decades,[2–4] leading to a subsequent increase in surgical interventions.[4]
Based on the assumption that early detection and treatment of early stage Renal Cell Carcinoma (RCC) should result in improved survival outcomes, one would expect a decline in population based mortality rates over time. However, examination of national overall and cancer-specific mortality trends using Surveillance, Epidemiology, and End Results (SEER) data have documented a paradoxical rise in mortality rates when accounting for stage and size migration.[4] Indeed, this phenomenon, termed “treatment disconnect,” has dramatically impacted clinical and policy perspectives in the management of kidney cancer.[5]
“Treatment disconnect” has been attributed in part to length and lead-time biases, and to the fact that the absolute number of patients with ultimately lethal lesions has not diminished. However, some investigators have noted discrepancies in cancer mortality rates between whole and restricted datasets,[6] and others have demonstrated that changes in incidence can have significant impact on mortality data interpretation.[7] As such, legitimacy of “treatment disconnect” deserves reassessment. Thus, our objective was to examine the impact of missing size data and adjustment for rising incidence on overall and cancer-specific mortality trends for patients with RCC using national tumor registry data.
Methods
The SEER program of the National Cancer Institute (NCI) assembles information on cancer incidence, local treatment, and survival in the United States (US). The SEER program registries collect data on patient demographics, including primary tumor site, tumor morphology and stage at diagnosis, first course of treatment, and follow up for survival. Registries participating in the SEER program capture approximately 97% of incident cases.[8]
We identified all patients with an incident kidney cancer diagnosis from 1983–2011 from the nine original SEER registries (Metropolitan Atlanta, Connecticut, Metropolitan Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, Utah). Based on International Classification of Diseases for Oncology, Second Edition site code C64.9, we restricted the cohort to Renal Cell Carcinoma histology codes (8032, 8041, 8240, 8260, 8270, 8290, 8310, 8312, 8317, 8318, 8319, 8320, 8960, 8963, and 8966), as well as behavior codes 2 and 3 as described in the original report. All tumor stages were included for analysis.
Demographic and cancer-specific data, including age at diagnosis, race, gender, tumor laterality, organ-confined status, and tumor histology were extracted. All covariates were treated as categorical variables with the exception of age, which was treated as a continuous variable. Restricted to 1983–2011 (years with available tumor size data), patients were sorted into four tumor size categories (<2, 2–4, >4–7, and >7 cm) as previously described.[4] Patients with missing size data were categorized independently. Patient demographic and clinical characteristics were compared between size groups independently, and then between those with and without available tumor size data using ANOVA and Chi-square tests.
Age-adjusted annual mortality rates (overall and cancer-specific) for RCC were calculated by determining the number of deaths each year, dividing by the corresponding mid-year population estimates provided by SEER, and multiplying by 100,000. Rates were age adjusted to the 2000 US population using direct adjustment methods. Data was extracted using SEER*STAT software (reference: SEER*STAT Surveillance Research Program, National Cancer Institute SEER*Stat software (www.seer.cancer.gov/seerstat) version 8.1.5) and analyses were performed using STATA Version 13 software (StataCorp LP, College Station, Texas). Missing size data was estimated using multiple imputations (IVE ware). Differences between observed and imputed mortality rates when considering observed and imputed tumor size were compared between size categories using linear regression in which we included main effects and interaction terms for year and imputation status (observed versus imputed rate). In estimating the models to test for differences between observed and imputed rates, we weighted the regressions by the inverse of the variance of each estimated rate, as is appropriate when using summarized data,[9] and used robust standard errors.
In separate analyses, we estimated the overall mortality rate for all comers from 1983–2011, irrespective of tumor size, after adjustment for prior cumulative incidence using ridge regression, an estimation technique used to attain a substantial reduction in variance while increasing the stability of the coefficients. We chose to use ridge regression, since year and incidence rates are strongly correlated; that is, the incidence rates change at an almost completely linear rate over time. The ridge regression is able to provide stable estimates in the presence of such collinearity.[10] For prior adjustment, we first summed the current year and the previous ten-year incidence rate to get a cumulative incidence rate. We then entered the cumulative incidence rate as a covariate in the ridge regression using a restricted cubic spline with four knots.[11] Differences between the mortality rates estimated using observed cumulative incidence (i.e. incidence varying over time) and those estimated using incidence adjusted to the 2011 rate (i.e. holding incidence constant at 2011 rates) were assessed using linear regression with bootstrap standard errors with 10,000 iterations.[12] We also examined the mortality over incidence ratio (MOI) as another metric for investigating the relationship of yearly mortality compared to same year incidence.[13]
Results
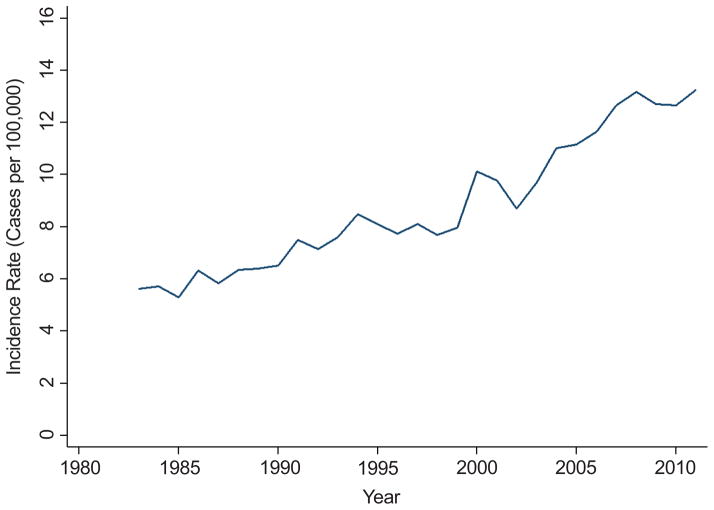
78,891 patients met inclusion criteria from 1973–2011, of which 70,212 were diagnosed in 1983 or later. Of those diagnosed from 1983, 10.4% had missing data. Demographic and clinical data stratified by tumor size is presented in Table 1. Significant differences were observed between size groups with respect to age, gender, laterality, stage, histology, and grade (all p values <0.001). Age adjusted kidney cancer incidence has uniformly increased across age categories over the study period (Figure 1).
Table 1.
Patient characteristics by tumor size
| Characteristic | Tumor Size (cm) | P Value | ||||
|---|---|---|---|---|---|---|
|
| ||||||
| <2 | ≥2 to ≤4 | >4 to ≤7 | >7 | Missing | ||
| Number | 4,184 | 20,015 | 19,518 | 19,226 | 7,269 | |
| Age (mean±SD) | 59.9±14.3 | 62.9±13.5 | 63.2±13.8 | 59.6±17.7 | 65.8±15.9 | <0.001* |
| Gender (%) | <0.001* | |||||
|
| ||||||
| Female | 40.9 | 38.7 | 37.4 | 35.4 | 37.8 | |
| Male | 59.1 | 61.3 | 62.6 | 64.6 | 62.2 | |
|
| ||||||
| Race (%) | <0.001† | |||||
|
| ||||||
| White | 80.3 | 82.2 | 83.7 | 83.6 | 83.7 | |
| Black | 12.1 | 11.0 | 9.4 | 9.7 | 10.5 | |
| Other | 7.6 | 6.8 | 6.9 | 6.7 | 5.8 | |
|
| ||||||
| Tumor side (%) | <0.001* | |||||
|
| ||||||
| Right | 50.4 | 52.2 | 50.7 | 48.5 | 41.5 | |
| Left | 48.6 | 47.7 | 49.0 | 50.8 | 42.9 | |
| Bilateral | 0.1 | 0.1 | 0.2 | 0.5 | 1.8 | |
| Unknown | 0.9 | 0.1 | 0.1 | 0.2 | 13.8 | |
|
| ||||||
| Tumor stage (%) | <0.001* | |||||
|
| ||||||
| Localized | 89.2 | 85.5 | 66.0 | 38.0 | 19.0 | |
| Regional/distant | 9.1 | 13.5 | 32.1 | 60.5 | 56.1 | |
| Unstaged | 1.7 | 2.0 | 1.9 | 1.5 | 24.9 | |
|
| ||||||
| Histology (%) | <0.001* | |||||
|
| ||||||
| Clear cell | 84.3 | 86.8 | 90.2 | 86.6 | 94.3 | |
| Papillary | 12.6 | 8.8 | 5.5 | 3.7 | 1.9 | |
| Chromophobe | 3.9 | 3.4 | 2.5 | 2.8 | 0.5 | |
| Oncocytoma | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | |
| Other/Missing | 1.1 | 0.9 | 1.8 | 6.9 | 3.2 | |
|
| ||||||
| Fuhrman grade (%) | <0.001* | |||||
|
| ||||||
| 1 | 17.9 | 14.3 | 9.3 | 4.9 | 3.9 | |
| 2 | 35.4 | 35.3 | 28.8 | 20.1 | 6.6 | |
| 3 | 9.3 | 11.4 | 15.6 | 19.2 | 6.1 | |
| 4 | 1.1 | 1.3 | 3.3 | 7.6 | 1.9 | |
| Unknown | 36.3 | 37.8 | 43.0 | 48.2 | 81.4 | |
Only patients from 1983 to 20011 included
Comparison between patients with missing size data and all patients with reported size data
p<0.001
p=0.001
Figure 1.
Age adjusted kidney cancer incidence has uniformly increased across age categories over the study period of 1983–2011
Corresponding with increasing rates of surgeries performed (data not presented), the proportion of patients with missing tumor data (21.6% in 1983 to 4.2% in 2011, p<0.001) significantly decreased over the study period. Patients with missing tumor size significantly differed from patients with documented tumor size with respect to age, race, laterality, stage, histology, and grade (Table I). Patients with missing size data tended to be older with more advanced stage (or unstaged with missing histology).
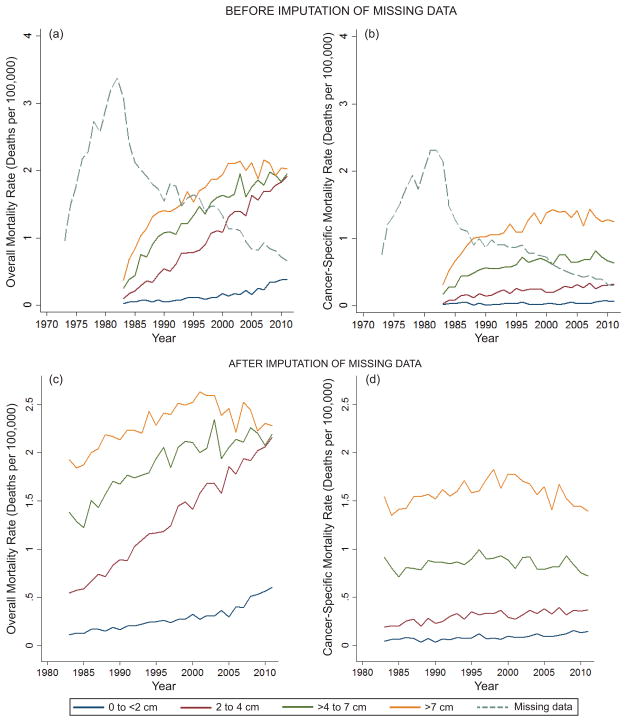
When evaluating temporal changes in age adjusted overall mortality between 1983 and 2011, significant increases for all sizes were noted for <2 cm tumors (0.11 to 0.61, p<0.001), 2–4cm tumors (0.54 to 2.16, p<0.001), >4–7cm tumors (1.38 to 2.19, p<0.001), and >7cm tumors (1.93 to 2.28, p<0.001) (Figure 2a). In comparison, small but statistically significant increases in age adjusted cancer-specific mortality for tumors less than 2 cm (0.05 to 0.14, p<0.001) and 2–4cm tumors (0.19 to 0.37, p<0.001), but not for >4–7cm tumors (0.92 to 0.72, p=0.45), and >7cm tumors (1.54 to 1.40, p=0.98) were demonstrated (Figure 2b).
Figure 2.
Age adjusted (a) overall and (b) kidney cancer-specific mortality (# of deaths per 100,000) by tumor size shows attenuation of mortality rates when accounting for missing data (c, d).
Compared to observed rates, following missing data imputation, significant attenuation in the change in overall mortality rate per 100,000 US population from 1983–2011 were noted (Figure 2c) for tumors <2cm (Δobs0.35 versus Δimp0.49, p=0.006), 2–4cm tumors (Δobs1.82 versus Δimp1.61, p<0.001), >4–7cm tumors (Δobs1.70 versus Δimp0.81, p<0.001), and >7cm tumors (Δobs1.65 versus Δimp0.35, p<0.001). Similarly, comparing observed to imputed rates, significant differences in the cancer-specific morality rates were noted (Figure 2d) for tumors <2cm (Δobs0.05 versus Δimp0.10, p=0.001) 2–4cm tumors (Δobs0.29 versus Δimp0.18, p=0.005), >4–7cm tumors (Δobs0.46 versus Δimp-0.20, p<0.001), and >7cm tumors (Δobs0.93 versus Δimp-0.15, p<0.001).
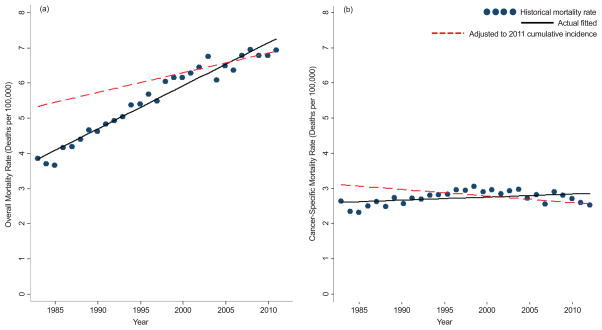
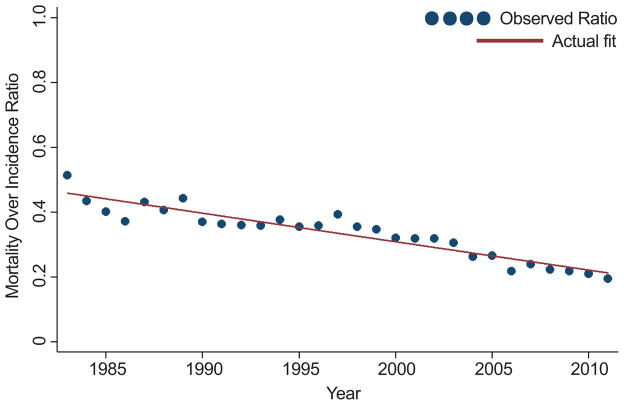
For the entire cohort irrespective of tumor size, holding the incidence of kidney cancer constant at 2011 rates, the temporal increase from 1983–2011 in overall mortality for all patients was attenuated (p<0.001 for test of difference in slope) when comparing model estimates using historical incidence (3.8 to 7.3) and adjusted to 2011 incidence (5.3 to 6.9) rates (Figure 3a), suggesting that rapidly rising incidence may also influence reported overall mortality trends. Comparing estimates using historical incidence (2.6 to 2.9) and estimated (3.1 to 2.6) cancer-specific mortality rates (Figure 3b), no significant differences were observed (p=0.97). The MOI metric for each year was also plotted, demonstrating a reduction in kidney cancer-specific mortality over the study period (Figure 4).
Figure 3.
Actual and adjusted (a) overall and (b) kidney cancer-specific mortality demonstrating that, when adjusted to 2011 cumulative incidence, the observed rise in mortality over time is attenuated.
Figure 4.
Kidney cancer-specific mortality over incidence ratio trended from 1983 to 2011.
Discussion
To better understand the “treatment disconnect” phenomenon, our aim was to investigate factors that affect interpretation of epidemiologic data for patients diagnosed with renal cell carcinoma, which has wide applicability across the spectrum of cancer care. Specifically, we hypothesized that missing tumor size data, and the rapidly increasing incidence of RCC, affect interpretation of RCC mortality trends using SEER data.[4] We found that imputation of missing size data significantly attenuates the previously documented rise in both overall and cancer-specific mortality rates when stratified by tumor size. Furthermore, when adjusting for 2011 cumulative incidence, the temporal increase in overall mortality was significantly less than unadjusted estimates, irrespective of tumor size. Tracking the MOI over time, a recently suggested metric to normalize mortality rates,[13] further supported our findings that the rise in RCC mortality is likely significantly smaller than previously described and its precise quantification is markedly confounded by rising RCC incidence.
Over the last 3 decades, kidney cancer incidence has increased uniformly across age categories in the SEER dataset, and has been predominantly due to increased detection of localized disease (Figure 1).[3] Similar trends have been observed in European registry databases, albeit granular data have demonstrated regional variations in detection rates.[14] Although the reasons are multifactorial, increased performance of routine abdominal imaging is the most likely cause.[15] However, while the rates of renal surgery have risen simultaneously with increased tumor detection, conflicting RCC survival and mortality trends have been reported, and it remains unclear if the dramatic increase in early detection and treatment over the past two decades has meaningfully impacted survival in patients with kidney cancer. As over-diagnosis of clinically insignificant malignancy, along with receipt of unneeded treatment as well as its attendant risks, are arguably the most important harms associated with early cancer detection,[16] this uncertainty has led to the reassessment of contemporary practice patterns for incidentally diagnosed lesions.[17]
Tumor size remains one the most influential clinical factor impacting provider treatment decisions in the management of kidney cancer.[18] Not recorded in SEER until 1983, findings from our study demonstrate that tumor size information was missing in 22.7% of cases in the SEER cohort, and that the proportion of patients with missing tumor size is inversely proportional to the rate of renal procedures performed. As a result, the proportion of cases with missing tumor data early in the study period significantly outweighed that of the latter period. Imputing tumor size for these cases significantly attenuated the rise in mortality for tumors when stratified by tumor size. Reasons for this are multifactorial, but may reflect the fact that patients early in the study period presented with larger more aggressive tumors, given the well described stage migration towards earlier stage disease due to the increased use of routine abdominal imaging in the later years of the cohort.[15] These findings are significant, suggesting that the “treatment disconnect” phenomenon that has been previously described when evaluating SEER data segregated by tumor size may be largely explained by accounting for missing size data.[4, 6]
Rapid concurrent rises in tumor incidence and mortality have been demonstrated in other tumor types, most notably in prostate cancer cohorts following the advent of PSA screening; however, kinetics of disease progression and intensity of screening markedly differ between malignancies.[19] A rapid peak followed by a decline in mortality after the initiation of cancer screening can be influenced by lead and length time biases. The influence of increasing incidence on mortality rates can significantly influence data interpretation, yet this fact has not received significant attention in the urologic literature.[7, 13, 19] When examining overall and cancer-specific mortality rates per 100,000 persons, a rapid influx of newly documented incidental cancer diagnoses of uncertain clinical significance concurrently increases the pool of patients eligible to die with (not likely from) that given cancer. This phenomenon artificially raises the mortality rate in a given population, resulting in a form of allocation bias. For instance, if data collection is initiated in a particular year, only patients diagnosed in that year are captured as dying from kidney cancer. Meanwhile the following year a larger pool of patients, those diagnosed in the initial year and the following year, become “eligible” for being indexed as dying with the disease, thus significantly confounding data interpretation. As such, our analyses holding the cumulative incidence of kidney cancer constant significantly attenuated the observed increase in overall survival rates over time, while cancer-specific survival remained essentially unchanged.
While the impact of lack of recurrence data, reliance on death certificate cause of death to determine cancer-specific survival, and lack of co-morbidity assessment significantly impacts the use of SEER data for unbiased comparative effectiveness assessment,[20, 21] these limitations are likely less important for measuring general population level trends in mortality over time that are blinded to treatment type.[22, 23] To evaluate the influence of rising incidence on observed mortality over time, we elected to use ridge regression. Although this approach avoids the exaggerated impact on variance observed with linear regression analyses, the interpretation of the actual magnitude of measured effects remains limited using this methodology due to inherent biases.
Pitfalls and challenges associated with understanding underpinnings of population-based survival, mortality, and incidence data trends have been described.[19] Supplementary Table A summarizes expected data trends under various ideal scenarios if they were to occur non-concurrently. Real-world data represents an amalgam of concurrent processes and as such, as our analyses demonstrate, are extremely challenging to interpret. Some investigators have proposed that normalizing mortality by incidence may provide insights into progress in cancer management.[13] We have included MOI analysis for patients with RCC, which indeed suggests improvement in treatment effect over time (Figure 4). Nevertheless, these data are likely overly optimistic given that MOI analyses are disproportionally affected by over diagnosis phenomena, which undoubtedly plague kidney cancer management.[7, 13]
In conclusion, our analyses suggest that missing data and rising incidence rates contribute substantially to the “treatment disconnect” phenomenon observed when examining kidney cancer mortality rates using SEER data. As these adjusted changes in mortality appear to be substantively less pronounced than previously demonstrated, “treatment disconnect” should not temper our enthusiasm for definitive treatment in appropriate candidates. We hope that our work puts into better focus the limitations of existing datasets and serves as a catalyst for improving epidemiologic data gathering and interpretation. Nevertheless, as the evidence base to support shared decision making for treatment of kidney cancer is limited, we will continue to rely on tumor registry data to evaluate changes in mortality trends as the treatment paradigm evolves. However, these trends must be contextualized clinically and should be used to inform clinical trial development as opposed to direct evidence to implement changes in practice.
Supplementary Material
Change in observed mortality and incidence if ideal clinical scenarios occur non-concurrently (adapted from [19])
Acknowledgments
Funded by Fox Chase Cancer Center via institutional support of the Kidney Cancer Keystone Program (National Cancer Institute P30 CA006927). The authors were supported in part through the National Institutes of Health R03CA152388 (BLE)
Footnotes
IRB Approved Protocol Number: 06-840
Disclosure of potential conflict of interest:
Marc C Smaldone, Brent K. Hollenbeck, John M. Hollingsworth, David Miller, Todd Morgan, Simon Kim, Aseem Malhotra, Robert Uzzo, and Alexander Kutikov have no conflicts of interest to disclose.
Brian Egleston serves as a consultant/advisor for Teva and receives salary support via institution (Janssen and Verilogue).
Elizabeth Handorf and Yu-Ning Wong receive research support paid to the institution via Pfizer. Yu-Ning Wong receives travel support from Tokia.
References
- 1.Siegel R, Miller K, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.Chow WH, et al. Rising incidence of renal cell cancer in the United States. JAMA. 1999;281(17):1628–31. doi: 10.1001/jama.281.17.1628. [DOI] [PubMed] [Google Scholar]
- 3.Simard EP, et al. Cancers with increasing incidence trends in the United States: 1999 through 2008. CA Cancer J Clin. 2012;62(2):118–28. doi: 10.3322/caac.20141. [DOI] [PubMed] [Google Scholar]
- 4.Hollingsworth JM, et al. Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst. 2006;98(18):1331–4. doi: 10.1093/jnci/djj362. [DOI] [PubMed] [Google Scholar]
- 5.Parsons JK, Schoenberg MS, Carter HB. Incidental renal tumors: casting doubt on the efficacy of early intervention. Urology. 2001;57(6):1013–5. doi: 10.1016/s0090-4295(01)00991-8. [DOI] [PubMed] [Google Scholar]
- 6.Chow WH, Linehan WM, Devesa SS. Re: Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst. 2007;99(7):569–70. doi: 10.1093/jnci/djk114. author reply 570–1. [DOI] [PubMed] [Google Scholar]
- 7.Asadzadeh Vostakolaei F, et al. The validity of the mortality to incidence ratio as a proxy for site-specific cancer survival. Eur J Public Health. 2011;21(5):573–7. doi: 10.1093/eurpub/ckq120. [DOI] [PubMed] [Google Scholar]
- 8.Zippin C, Lum D, Hankey BF. Completeness of hospital cancer case reporting from the SEER Program of the National Cancer Institute. Cancer. 1995;76(11):2343–50. doi: 10.1002/1097-0142(19951201)76:11<2343::aid-cncr2820761124>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 9.Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135(11):1301–9. doi: 10.1093/oxfordjournals.aje.a116237. [DOI] [PubMed] [Google Scholar]
- 10.Hadgu A. An application of ridge regression analysis in the study of syphilis data. Stat Med. 1984;3(3):293–9. doi: 10.1002/sim.4780030311. [DOI] [PubMed] [Google Scholar]
- 11.Harrell F. Regression Modeling Strategies. New York: Springer; 2001. [Google Scholar]
- 12.Efron B, Tishshirani R. CRC Monographs on Statistics & Applied Probability. Chapman & Hall; 1993. An Introduction to the Bootstrap. [Google Scholar]
- 13.Maruvka YE, Tang M, Michor F. On the validity of using increases in 5-year survival rates to measure success in the fight against cancer. PLoS One. 2014;9(7):e83100. doi: 10.1371/journal.pone.0083100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li P, et al. Regional geographic variations in kidney cancer incidence rates in European countries. Eur Urol. 2015;67(6):1134–41. doi: 10.1016/j.eururo.2014.11.001. [DOI] [PubMed] [Google Scholar]
- 15.Cooperberg MR, et al. Decreasing size at diagnosis of stage 1 renal cell carcinoma: analysis from the National Cancer Data Base, 1993 to 2004. J Urol. 2008;179(6):2131–5. doi: 10.1016/j.juro.2008.01.097. [DOI] [PubMed] [Google Scholar]
- 16.Welch HG, Black WC. Overdiagnosis in cancer. J Natl Cancer Inst. 2010;102(9):605–13. doi: 10.1093/jnci/djq099. [DOI] [PubMed] [Google Scholar]
- 17.Kunkle DA, Egleston BL, Uzzo RG. Excise, ablate or observe: the small renal mass dilemma--a meta-analysis and review. J Urol. 2008;179(4):1227–33. doi: 10.1016/j.juro.2007.11.047. discussion 1233–4. [DOI] [PubMed] [Google Scholar]
- 18.Smaldone MC, et al. Clinical Characteristics Associated With Treatment Type for Localized Renal Tumors: Implications for Practice Pattern Assessment. Urology. 81(2):269–276. doi: 10.1016/j.urology.2012.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Welch HG, Schwartz LM, Woloshin S. Are increasing 5-year survival rates evidence of success against cancer? JAMA. 2000;283(22):2975–8. doi: 10.1001/jama.283.22.2975. [DOI] [PubMed] [Google Scholar]
- 20.Kutikov A, et al. Evaluating overall survival and competing risks of death in patients with localized renal cell carcinoma using a comprehensive nomogram. J Clin Oncol. 2010;28(2):311–7. doi: 10.1200/JCO.2009.22.4816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hollingsworth JM, et al. Five-year survival after surgical treatment for kidney cancer: a population-based competing risk analysis. Cancer. 2007;109(9):1763–8. doi: 10.1002/cncr.22600. [DOI] [PubMed] [Google Scholar]
- 22.Scosyrev E, et al. Surveillance Epidemiology and End Results (SEER) program and population-based research in urologic oncology: an overview. Urol Oncol. 2012;30(2):126–32. doi: 10.1016/j.urolonc.2009.11.005. [DOI] [PubMed] [Google Scholar]
- 23.Hershman DL, Wright JD. Comparative effectiveness research in oncology methodology: observational data. J Clin Oncol. 2012;30(34):4215–22. doi: 10.1200/JCO.2012.41.6701. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Change in observed mortality and incidence if ideal clinical scenarios occur non-concurrently (adapted from [19])