Abstract
The impacts of Deepwater Horizon (DWH) oil on morphology and function during embryonic development have been documented for a number of fish species, including the economically and ecologically important pelagic species, mahi-mahi (Coryphaena hippurus). However, further investigations on molecular events and pathways responsible for developmental toxicity have been largely restricted due to the limited molecular data available for this species. We sought to establish the de novo transcriptomic database from the embryos and larvae of mahi-mahi exposed to water accommodated fractions (HEWAFs) of two DWH oil types (weathered and source oil), in an effort to advance our understanding of the molecular aspects involved during specific toxicity responses. By high throughput sequencing (HTS), we obtained the first de novo transcriptome of mahi-mahi, with 60,842 assembled transcripts and 30,518 BLAST hits. Among them, 2,345 genes were significantly regulated in 96hpf larvae after exposure to weathered oil. With comparative analysis to a reference-transcriptome-guided approach on gene ontology and tox-pathways, we confirmed the novel approach effective for exploring tox-pathways in non-model species, and also identified a list of co-expressed genes as potential biomarkers which will provide information for the construction of an Adverse Outcome Pathway which could be useful in Ecological Risk Assessments.
Crude oil spills in critical fish spawning and nursing habitats are a recurrent worldwide problem1,2,3. The Deepwater Horizon (DWH) incident in 2010, the largest marine oil spill in U.S. history, resulted in exposure of many spawning pelagic fish species of economical and ecological importance, such as mahi-mahi (Coryphaena hippurus), bluefin (Thunnus thynnus) and yellowfin tuna (Thunnus albacares)4,5,6. The DHW oil is a complex chemical mixture, with polycyclic aromatic hydrocarbons (PAHs) and their alkylated homologues being the main toxic components to fish embryos7,8. The composition and structure of individual PAHs in the water column can be significantly altered by weathering processes, and it has been shown that weathered surface slick oil (3- and 4-ring PAHs enriched) is more toxic than non-weathered source oil to mahi-mahi on a ∑PAH basis at different levels from molecular to physiological functions9,10. Numerous studies have investigated the developmental toxicity of crude oil to fish embryos and larvae and identified a variety of abnormalities such as cardiac function, kidney development, formation of the craniofacial skeleton (eye and jaw), nervous system as well as reduced swimming performance7,10,11,12,13. These impacted functions and biological processes may contribute to delayed mortality and declined population as previously observed in pink salmon (Oncorhynchus gorbuscha) exposed to crude oil14,15. Consequently, a baseline understanding of crude oil toxicity to the fish species native to the oiled pelagic zone is essential for ecological risk assessment, regulatory decision making, and spill response planning.
Despite the relatively rich literature on functional and morphological impacts of crude oil on fish embryonic development, the molecular initiating events preceding these effects are less understood. A recent study of Atlantic haddock (Melanogrammus aeglefinus) embryos exposed to crude oil found a downregulation of mRNAs encoding cardiac rapid potassium channel, as well as the sodium/calcium exchanger (ncx1) that regulates intracellular calcium levels during E-C coupling16. Another study in mahi-mahi focused on the developmental cardiotoxicity of crude oil exposure targeted ten molecular indicators of cardiac stress and injury, such as atrial and ventricular myosin heavy chains (amhc/myh6 and vhmc/myh7), cardiac myosin light chain 2 (cmlc2/myl7), B-type natriuretic peptide (nppb), as well as transcription factors nkx2.5 and tbx517. Our previous study by RNA sequencing and physiological assessment demonstrated that slick oil exposure resulted in pronounced perturbations in metabolism, steroid biosynthesis, vision, and AhR genes suggesting other targets in addition to the heart may be involved in the developmental toxicity of DHW oil10. Given the increasing evidence of morphological and physiological impacts of crude oil on a number of fish species, a comprehensive and accessible genomic database is urgently needed for target species in oiled regions to advance our understanding of the toxic molecular mechanisms of crude oil. Here, we focus on mahi-mahi, Coryphaena hippurus, a resident species in the DWH-oiled water, as a target for investigating the transcriptomic responses reflective of developmental toxicity of crude oil. This species is a highly commercially important fish widespread in tropical and temperate waters and occurs in the Atlantic, Indian, Pacific Oceans and the Mediterranean18, which thus makes an excellent candidate for understanding the impact of the recurrent oil spills worldwide.
The present study aimed to establish, for the first time, a de novo global transcriptome database from embryos and larvae of mahi-mahi exposed to water accommodated fractions (HEWAFs) of two oil types (slick/weathered oil and non-weathered source oil) using high throughput sequencing (HTS) and de novo assembly technology. In contrast, our previous study used a commercially available reference-transcriptome-guided automated OnRamp approach that failed to provide sequence information for de novo transcriptome assembly efforts for mahi-mahi10. To confirm prior molecular signatures and provide an accessible transcriptome data base, differential expression analysis was conducted in 96 hpf larvae exposed to slick oil by the de novo approach, and downstream gene ontology (GO) terms were compared to the 24, 48 and 96 hpf mahi-mahi exposed to both source and slick oil as determined by the earlier OnRamp approach. Since a number of individual genes were not commonly regulated among different developmental stages (24, 48, 96 h), oil types (slick and source oil) and transcriptome analytical approaches (de novo and OnRamp), we also considered state-of-the-art comparative toxicity pathway (tox-pathway) analysis, which closely related to the concept of adverse outcome pathways (AOP). Furthermore, the common tox-pathways and a list of common responsive genes were identified as potential biomarkers to predict crude oil toxicity or recovery in mahi-mahi.
Results
De novo assembly of mahi-mahi transcriptome and annotation
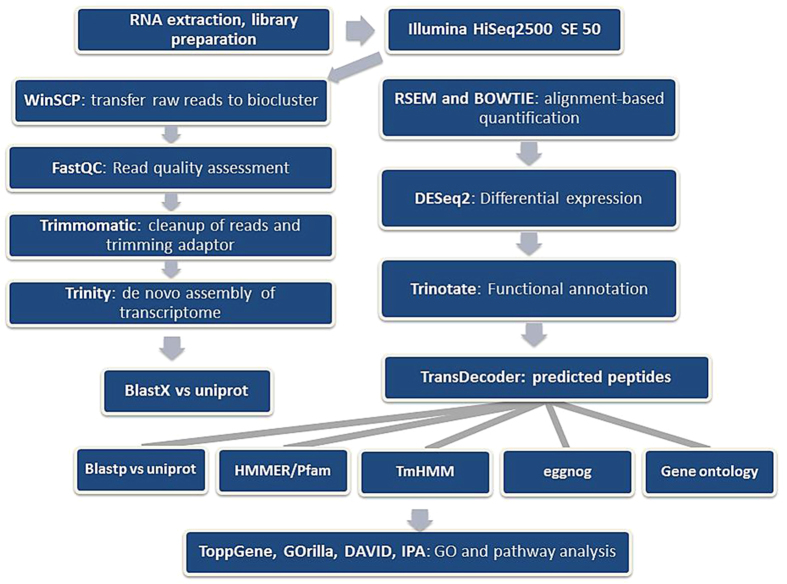
Schematic flow diagram illustrating the workflow employed for sequencing, de novo assembling and annotating the mahi-mahi transcriptome is presented in Fig. 1. A total of 1,647,902,734 Illumina HiSeq reads from mahi-mahi embryos and larvae were generated. After trimming the adapters, 42,447,657 bases were assembled with Trinity resulting in 60,842 transcript contigs with an average length of 711 bp and an N50 of 1,093 bases, 50% of which were annotated to known protein/nucleotide sequences by BLAST (e-value < 1 × 10−5) (Table 1). 17,370 HMMER/PFAM protein domains (Pfam), 3,168 predicted transmembrane regions (TmHMM), 26,444 non-supervised orthologous groups of genes (eggNOG), and 29,007 GO_blast were determined using Trinotate pipeline (Table 1). The final transcriptome assembly provided a high quality template for global gene expression profiling in this study and also provided a valuable resource for wider and deeper molecular research on mahi-mahi, which is an ecologically and economically important fish species with no existing genomic data.
Figure 1. Schematic flow diagram illustrating the workflow employed for sequencing, de novo assembling and annotating the mahi-mahi transcriptome and for determining changes in gene expression, GO terms and regulated pathways in DHW oil exposed mahi-mahi embryos and larvae.
Table 1. Summary statics of mahi-mahi transcriptome assembly and annotation.
| Total assembled bases | 42,447,657 |
|---|---|
| Total trinity transcript contigs | 60,842 |
| Percent GC | 49.6 |
| Transcrip contig N10 | 3,201 |
| Transcrip contig N20 | 2,348 |
| Transcrip contig N30 | 1,818 |
| Transcrip contig N40 | 1,417 |
| Transcrip contig N50 | 1,093 |
| Median contig length | 419 |
| Average contig | 711 |
| BLASTX_hit | 30,518 |
| BLASTP_hit | 22,302 |
| Pfam | 17,370 |
| ThHMM | 3,168 |
| eggnog | 26,444 |
| GO_blast | 29,007 |
| GO_pfam | 11,401 |
Quality of Gene Differential Expression Data
The unexposed control samples clustered separately from the slick oil treated sample, indicating global transcriptomic differences between the two sets (Fig. S1).
Differentially expressed genes (DEGs)
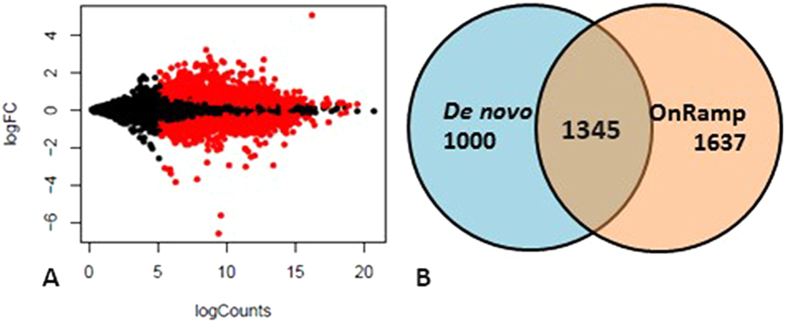
In 96 hpf mahi-mahi larvae exposed to slick oil, a total of 2,345 genes were significantly differentially expressed at a False Discovery Rate (FDR) < 0.05 (Fig. 2A), (43%) of which overlapped with the differentially expressed genes previously identified by the Onramp approach (Fig. 2B). Transcripts of the cytochrome P450 cyp1a1, a phase-I enzyme regulated by the Aryl hydrocarbon receptor pathway (AhR), catalyzes the oxidative metabolism of endogenous and exogenous substrates such as PAHs, and was consistently the most strongly upregulated gene (30 fold by de novo approach and 26.5 fold by OnRamp approach). Methylsterol monooxygenase 1 (msmo1), functions in cholesterol biosynthesis, exemplified another top upregulated gene (4.4 fold by both de novo and OnRamp approaches). The expression patterns of nine selected genes in 96 hpf larvae after slick oil exposure were verified by RT-qPCR (quantitative real-time PCR)10.
Figure 2.
Plot of normalized mean counts (expression) versus log2 fold change for untreated versus treated comparison (A). The X-axis plots normalized logCounts and the Y-axis is the log2 fold change (FC). Black dots represent non-significant genes, whereas red dots indicate significant differentially expressed genes (q < 0.05). (B) Venn diagram displaying the number of differentially expressed genes in 96 hpf larvae after slick oil exposure and the overlay between these gene lists identified by de novo and OnRamp approaches.
Gene Ontology (GO) terms analysis
de novo and OnRamp were consistent identifying significantly enriched GO categories (molecular function, biological process, pathway and phenotype) uncovered by both ToppGene and GOrilla (Tables S1 and S2). Both approaches revealed RNA binding and ATP binding as the predominant transcripts altered by slick oil exposure (>400 genes from input), which were also the most significantly enriched terms by DAVID against the zebrafish reference database (Table S3). The top overlapping biological processes and pathways impacted by slick oil exposure included transcripts involved in carboxylic acid metabolic process, cellular amino acid metabolic process, oxoacid metabolic process, ribosome biogenesis, cell development, metabolism, metabolism of amino acids, DNA replication, cholesterol biosynthesis, and synthesis of DNA. The significantly enriched common phenotypes included embryonic lethality, abnormal muscle morphology, abnormal muscle physiology, abnormal embryo development, abnormal cardiovascular system morphology, and decreased body size. Other biological processes and molecular functions that were more strongly enriched by de novo than by OnRamp included hydrolase activity, cellular lipid metabolic process, transmembrane signaling receptor activity, mitotic cell cycle process. Similar to ToppGene and GOrilla that are based on human/mouse references, DAVID topped ribonucleoprotein complex biogenesis, ribosome biogenesis, mitotic cell cycle, steroid metabolic process and heart development using zebrafish genome as background (Table S3). The top 20 GO terms by de novo approach are shown in Fig. S2.
Ingenuity pathway analyses (IPA)
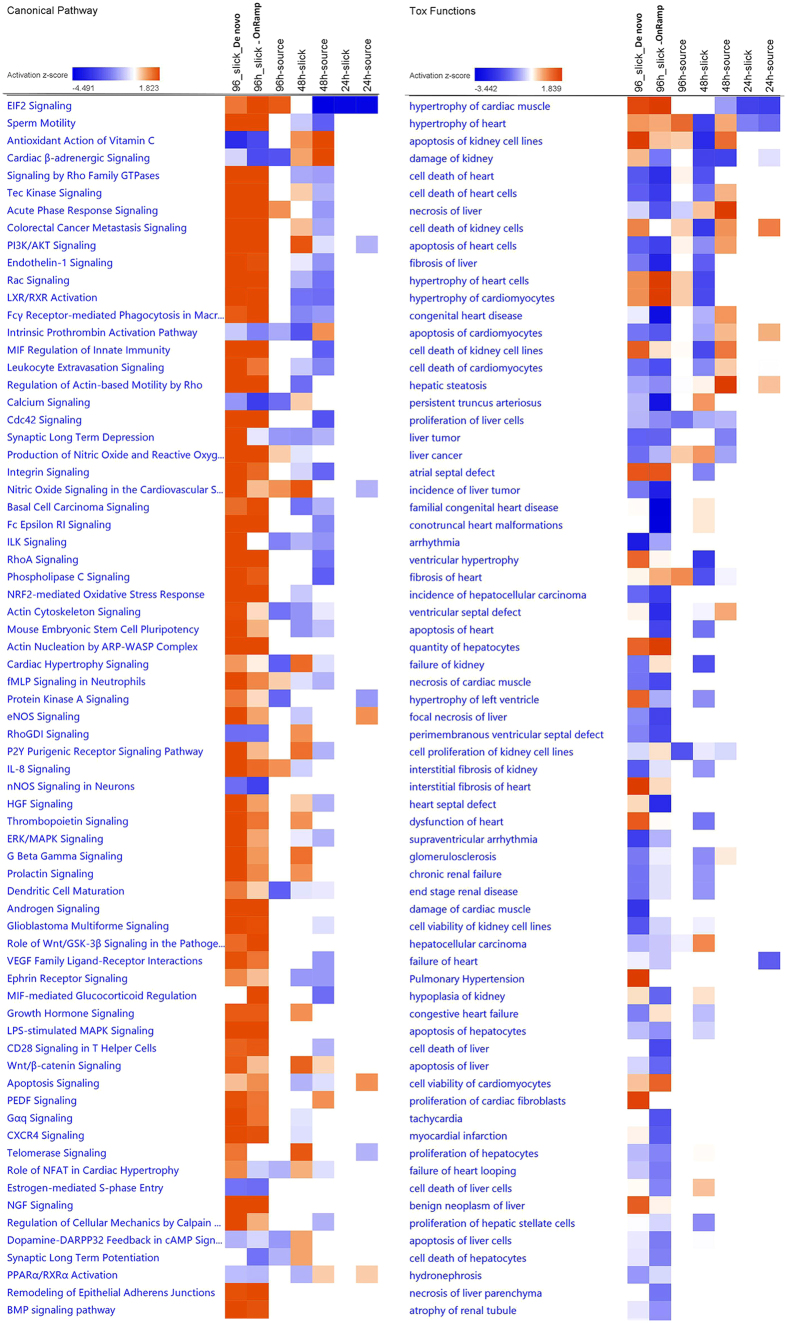
Canonical pathways and toxicity functions in mahi larvae at 96 h after slick oil exposure were analyzed by the de novo approach, and compared to responses 24, 48 and 96 h after both slick and source oil exposure by OnRamp. As expected, the 96 h_slick_De novo and the 96h_slick_OnRamp showed the highest similarity of impacted canonical pathways as well as toxicity functions (Fig. 3), reflecting similar pathway profiles with the two different approaches of analyses. The representative commonly affected canonical pathways and functions included EIF2 signaling, cardiac β-adrenergic signaling, signaling by Rho family GTPases, LXR/RXR activation, hypertrophy of cardiac muscle, apoptosis of kidney cells, and necrosis of liver. On the other hand, time (24, 48 and 96h) exhibited higher similarity than oil types, suggesting the pathway profiles might be more time-dependent than oil-type-dependent. The lists of DEGs responsible for the enrichment canonical pathways are reported in Table S4.
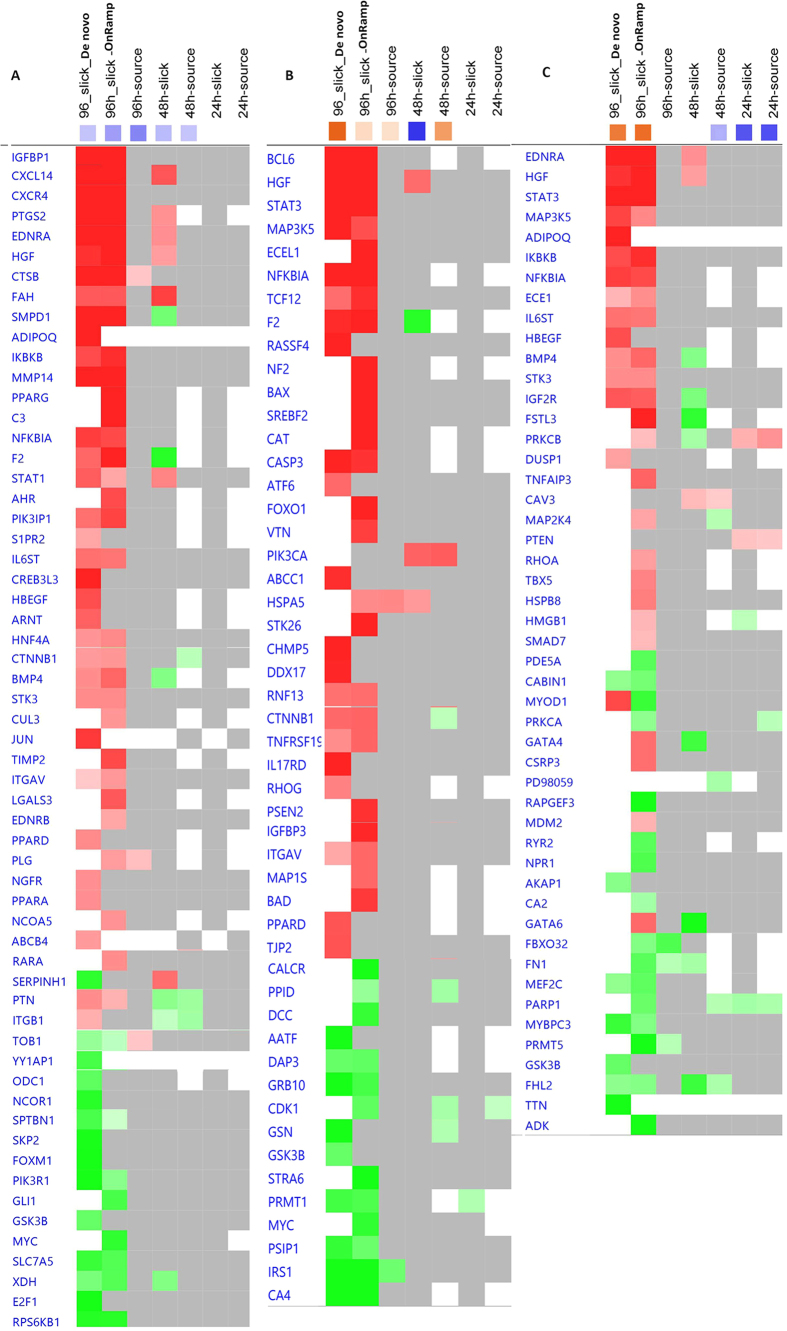
Figure 3. Heat maps clustering de novo (96h_slick) and OnRamp approaches (96h_slick, 96h_source, 48h_slick, 48h_source, 24h_slick, 24h_source) in canonical pathways and tox functions.
Orange indicates the z-score is positive. IPA predicts that the biological process or function is trending towards an increase when Z-scores ≥ 1.3. Blue indicates z-score is negative. IPA predicts that the biological process or function is trending towards a decrease when Z-scores ≤ −1.3.
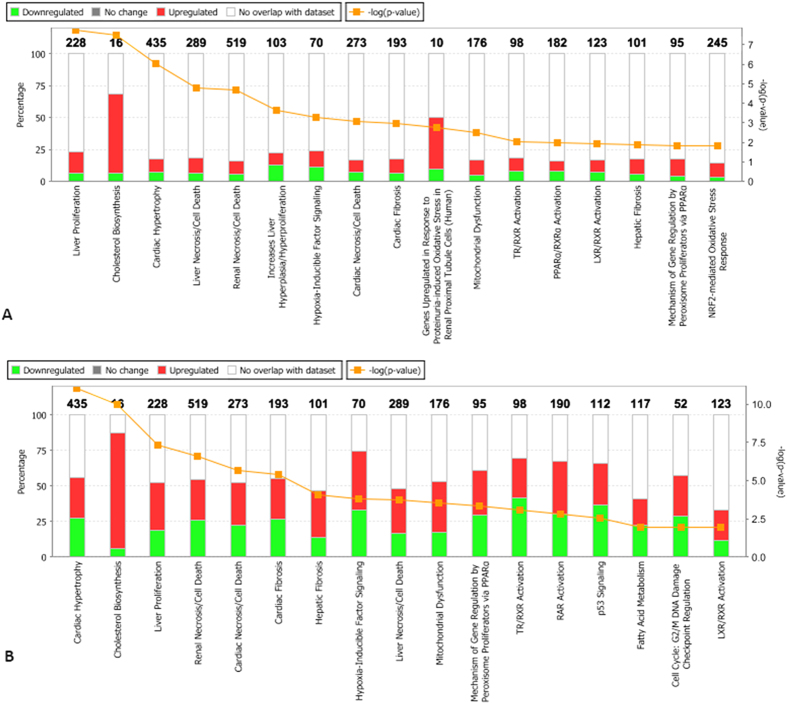
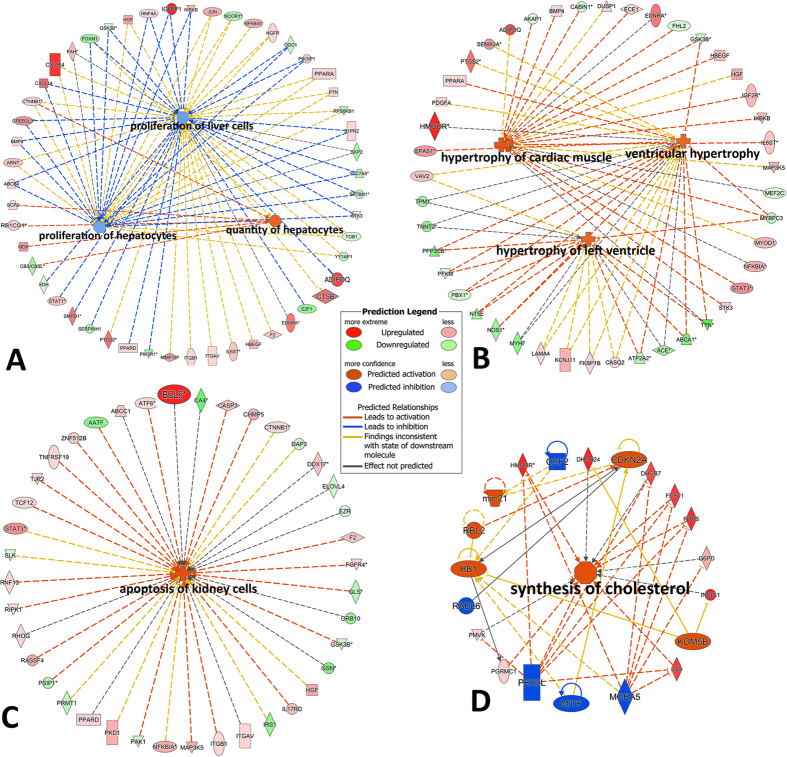
Furthermore, we also examined toxicity pathways which were enriched using the IPA-Tox tool (p < 0.05), focusing toxicity assessment of input molecules using toxicogenomic approaches. The toxicities of the slick oil-interacting DEGs identified by de novo and OnRamp approaches were analyzed and compared to each other. The two approaches shared 20 common toxicity pathways among the top 30 significant ones. Liver proliferation (p-value: 1.84E-08), cholesterol biosynthesis (p-value: 3.17E-08) and cardiac hypertrophy (p-value: 9.52E-07) topped the list generated from the de novo approach, while cardiac hypertrophy (p-value: 9.01E-12), cholesterol biosynthesis (p-value: 9.85E-11) and liver proliferation (p-value: 4.85E-08) topped the list from OnRamp (Fig. 4). The DEGs responsible for liver proliferation, cardiac hypertrophy and apoptosis of kidney were identified, and the common DEGs may be used as biomarkers for detecting DHW oil toxicity (Fig. 5; Table S5). Again, the 96h_slick_De novo and the 96h_slick_OnRamp showed the highest similarity of DEG profiles in each tox function. A full list of toxicity-pathways and DEGs by IPA-Tox are provided in Table S6.
Figure 4.
Stacked Bar Charts display the number of up-regulated (red), down-regulated (green) in each tox-pathway as a percentage of the total number of dataset molecules overlapping with the Tox Lists by de novo (A) and OnRamp (B) approaches.
Figure 5.
Heatmaps of genes in inhibited proliferation of liver cells (A), apoptosis of kidney cells (B) and hypertrophy of cardiac muscle (C) across different transcriptome analytic approaches, oil types and time. Heatmap colors: Red, up-regulated in the dataset; Green, down-regulated in the dataset; Gray; in the dataset but not analysis-ready (i.e. did not pass cutoffs and filters); White, not in the dataset. The orange or blue-colored squares in the column headers indicate the z-score (activity prediction) for each analysis. Orange shading indicates predicted activation and blue shading indicates predicted inhibition.
Discussion
The effects of crude oil on fish development have been well established using conventional morphological assessment. However, morphological evaluation can be subjective and subtle/delayed effects (such as delayed mortality or reduced swimming performance) may remain unnoticed. Moreover, classic morphological assessment using microscopy is relatively labor intensive and therefore a challenge for large samples during rapid embryonic development17. Global gene expression using transcriptomic tools can aid in detecting subsequent phenotypic effects and also anchoring multiple morphological impacts at one time of assessment, which may improve predictability, sensitivity and efficiency of the toxicity assessment. Yang19 revealed barcode-like toxicogenomic responses in zebrafish embryos exposed to eleven compounds prior to the onset of phenotypic effects. A recent study in mahi-mahi focused on the developmental cardiotoxicity of crude oil exposure and targeted ten molecular indicators of cardiac stress and suggested the necessity to design species-specific molecular markers17. To our knowledge, the present study is the first to de novo assemble the mahi-mahi transcriptome. An extensive list of phenotype-associated gene markers for mahi-mahi was identified from commonly differentially expressed genes between different oil types and different developmental stages. This comparative DEG analysis illustrated similarity and differences in responses to different oil types at different developmental times at individual gene levels. However, it is reasonable to expect that the numbers of common DEGs used for biomarkers will decrease when more developmental stages, more oil types, more concentrations of oil, and more species are included. Therefore, comparative pathway analysis was also employed in the present study. Similar to previous studies on embryonic stem cell20 and zebrafish embryo21, we found that whereas individual genes within a pathway were not significantly altered, significant activation/inhibition of common toxicity pathways still occurred. This comparative toxicity-pathway analysis also provided additional information on modes of action of crude oil, closely relating to the concept of the adverse outcome pathway22. Moreover, as most enriched GO and proposed pathways were from vertebrates, notably humans, it may also enable an extrapolation of crude oil toxicity to the human situation.
One of our objectives was to determine whether there were commonalities in the gene expression profiles generated by the Trinity de novo approach and the reference-transcriptome-guided automatic OnRamp approach. The results indicated a large proportion of DEGs, and GO terms common to both approaches, although OnRamp exhibited relatively more extensive changes (more enriched DEGs, GOs and pathways). We found that whereas individual genes within a gene pathway were not significantly regulated, significant common regulation at pathway categories still occurred. In this regard, we explored and focused on toxicity pathways, which were not limited by differences on the level of individual gene expression. Given that DWH oil is a complex chemical mixture, developmental toxicity is likely to arise from multiple mechanisms and pathways. IPA-Tox examination pointed to high commonalities between the two approaches, reflecting highly similar transcriptional responses to slick oil toxicity. The following discussion emphasizes the most significant enriched toxicity pathways and their responsive genes that were commonly observed in the two approaches, including liver proliferation, cholesterol biosynthesis, cardiac hypertrophy, renal necrosis/apoptosis, TR/RXR and LXR/RXR activation (Fig. 6).
Figure 6.
Top significant toxicity pathways through IPA-Tox showing how slick oil may impact/lead to (A) inhibition of liver proliferation; (B) cardiac hypertrophy; (C) apoptosis of kidney cells; (D) cholesterol biosynthesis.
Liver proliferation, the highest enriched toxicity function (Fig. 4A), occurs in response to partial hepatectomy and also after chemical injury23. Exposure to benzo[a]pyrene (BaP), a model polycyclic aromatic hydrocarbon (PAH), leads to increased liver tumorigenesis24 and adiposity as well as hepatic steatosis25 in animals. Liver proliferation was identified in the top three most significantly affected mechanistic responses in Atlantic cod (Gadus morhua) larvae exposed to mechanically dispersed oil for four days26. Liver proliferation is associated with signaling cascades involving growth factors, cytokines, hormones, nuclear receptors and alterations of growth related signals27. The changes of 55 interacting molecules were predicted to affect liver proliferation by IPA (Fig. 6A). Significant upregulation of cxcl14, adipoq, bmp4, ppard, and pik3ip genes may be associated with inhibited growth and proliferation of liver in mice. For example, cxcl14 over-expression aggravated CCl4-induced liver injuries, evidenced by enhanced acidophilic change and necrosis as well as increased fat deposition in hepatocytes, resulting in inhibited hepatocyte proliferation28. Adiponectin (adipoq) decreases proliferation of activated rat hepatic stellate cells29. In mouse, homozygous mutant mouse peroxisome proliferator activated receptor delta (ppard gene knockout) increases proliferation of mouse hepatic stellate cells in cell culture30, and a negative regulator of PI3K (pik3ip1) decreases proliferation of hepatocytes in liver31. More recently, Do32 found bone morphogenetic protein 4 gene (bmp4) inhibits proliferation of primary hepatocytes and HepG2 cells in culture. Work on zebrafish hepatogenesis has revealed that similar genes regulate hepatic patterning in both fish and mice33. The ability for the liver to regenerate is central to liver homeostasis. Because the liver is the main site of chemical detoxification, it is exposed to many chemicals (oil-derived PAHs in this case) in the body, which may potentially induce injury in cells other than hepatocytes. It is possible that the DHW oil exposure inhibits proliferation of damaged hepatocytes (as predicted by IPA in Fig. 6A) thereby diminishing detoxification and recovery.
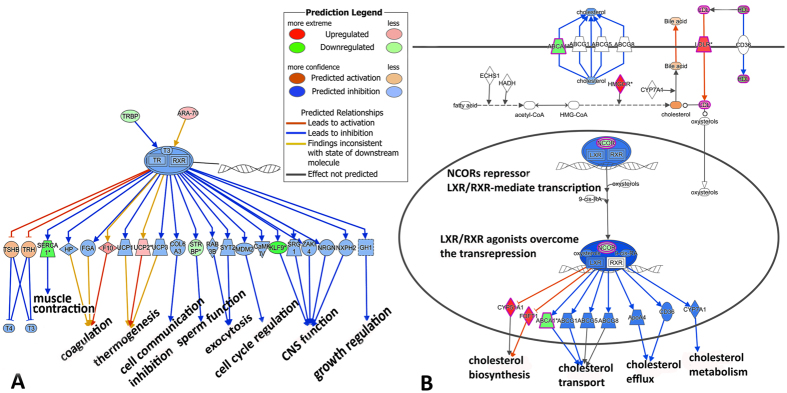
Slick oil had a profound impact on the expression of genes involved in cholesterol biosynthesis in mahi-mahi larvae as indicated by both GO and pathway analyses (Figs S2 and 6). Consistent with our mahi-mahi data, Olsvik26 identified cholesterol biosynthesis as one of the five most significant impacted pathways in Altantic cod Gadus morhua larvae exposed to chemically and mechanically dispersed oil. Cholesterol is a major structural component of the plasma membrane of eukaryotes and determines important biophysical characteristics of the cell surface bilayer. Membrane cholesterol dynamics also plays a key role in other cellular processes, such as intracellular sorting and vesicle transport. Due to the importance of cholesterol in these functions, cholesterol concentration within the cell is tightly regulated34. Cholesterol is either derived from the diet or from de novo synthesis occurring mainly in the liver through the mevalonate pathway. This pathway comprises several critical enzymes such as 3-hydroxy-3-methylglutaryl-coenzyme A reductase (hmgcr), farnesyl diphosphate synthase (fdps) farnesyl-diphosphate farnesyltransferase (fdft1), and 2,3-oxidosqualene-lanosterol cyclase (lss) and 7-dehydrocholesterol reductase (dhcr7)35,36,37,38,39. All of these genes were significantly downregulated in 96hpf mahi-mahi larvae by slick oil exposure (Fig. 6D). The inhibition of cholesterol transport, efflux and metabolism also affects the LXR/RXR pathway which also regulates ATP binding cassette transporters (abca1, abcg1, abcg5 and abcg8), cytochrome P450 51A1 and 7A140,41,42,43 (Fig. 7). Interestingly, Tanos44 established an important relationship between aryl hydrocarbon receptor (AHR) and cholesterol biosynthesis pathway independent of its dioxin response element (DRE) in both mouse liver and primary human hepatocytes. A coordinate repression of genes involved in cholesterol biosynthesis, namely hmgcr, fdft1, sqle and lss, and subsequently decreased cholesterol secretion were observed following AHR activation.
Figure 7.
A series of functions (e.g. muscle contraction, cell cycle regulation, CNS funtion, cholesterol biosynthesis and metabolism) pertubated through TR/RXR (A) and LXR/RXR (B) pathways predicted by IPA.
Approximately 80 genes differentially expressed at 96 hpf mahi-mahi larvae after slick oil exposure were involved in cardiac toxicity as determined by the de novo approach (Fig. 6B). The OnRamp approach also identified genes associated with cardiac toxicity as well as cardiac muscle development, contraction, K+ and Ca2+ homeostasis. For example, calsequestrin 2 (casq2), plays important roles in maintaining the cellular Ca2+ levels modulating contraction of cardiac muscle45, and was significantly upregulated. Bmp4 was also upregulated which is consistent with induced cardiomyocyte hypertrophy, apoptosis, and cardiac fibrosis46. Interestingly, pathological consequences were inhibited in mice treated with the bmp4 inhibitors noggin and DMH146. Troponin T type 2 (tnnt2) was significantly downregulated 96 hpf after slick oil exposure, which would also result in changes of cardiac muscle contraction in response to alterations in intracellular calcium ion concentration47. The inhibition of muscle contraction can also be mediated through the thyroid receptor/retinoid receptor (TR/RXR) pathway (Fig. 7), or downregulation of sarcoplasmic-endoplasmic reticulum calcium ATPase gene (serca1)48. Each of these was diminished by oil exposure. Another mechanism of crude oil-caused cardiotoxicity is a blockade of the IKr potassium current, which drives the repolarization phase of the cardiac action potential49. We also found potassium voltage-dependent potassium channel genes (kcnj11 and kcnq1) were significantly downregulated in 96 hpf after slick oil exposure. A recent study of Altantic haddock embryos exposed to crude oil also found a reduction of mRNA encoding cardiac ion channels16. In mahi-mahi, crude oil exposures significantly reduced the expression of genes related to cardiomyocyte contractility and cardiac defects, such as atrial and ventricular myosin heavy chain and cardiac myosin light chain genes17. In the present study, myh6, myh7, myl4, and myl6 were significantly downregulated. Two sarcoplasmic-endoplasmic reticulum calcium ATPase genes (serca1 and sercal2) and sodium/potassium/calcium exchanger (nckx) were also significantly downregulated. This suggests that slick oil can modify cardiac ion channels, pumps, and exchangers. The relationship between ion signaling and transcription of ion channel genes most likely reflects the recently characterized process of excitation-transcription (ET) coupling50. It was suggested that crude oil may act through a dual mechanism by blocking both ion channel function and also feedback to transcriptional regulation of ion channel genes16. These affected cardiac genes have been anchored to the higher order cardiac syndrome characteristics of crude oil exposure in mahi-mahi larvae (heart rate, pericardial and yolk sac edema)10.
In addition to cardiac toxicity, PAHs have been shown to cause significant renal toxicity in both freshwater and marine fish species. For example, Incardona11 have demonstrated that embryonic exposure to PAHs can cause primary effects on cardiac conduction that have secondary consequences for late stages of cardiac morphogenesis, kidney development, neural tube structure and formation of the craniofacial skeleton. Head kidney was also a sensitive target of exposure to DWH oil on resident gulf killifish (Fundulus grandis)51. Benzanthrone showed evidence of patchy glomerular congestion, tubular lesions, and damaged epithelial cells in kidney52. Incardona and Scholz53 suggested the most likely cause for edema in pink salmon larvae exposed to oil-derived PAHs was the inability of a failing heart and kidney. While PAHs are shown to cause nephrotoxicity and there are many know mechanisms of renal toxicity from membrane disruption to metabolic inhibition54, the underlying molecular mechanisms in fish are largely unknown. In the current study with mahi-mahi, renal necrosis/apoptosis was ranked No. 5 (p-value: 2.12E-05) and No. 4 (p-value: 3.69E-07) by de novo and OnRamp approaches, respectively (Fig. 4). The predicted induction of apoptosis of kidney cells are based on the expression profile of a number of important kinases, including mitogen-activated protein kinase (MAP3K)55, receptor interacting serine/threonine kinase (RIPK1)56, tight junction protein (TJP2)57, p21 protein (Cdc42/Rac)-activated kinase (PAK1)58, and phosphatidylinositol-4,5-bisphosphate 3-kinase (FGFR4)59 (Fig. 6C). Although functional effects of renal toxicity of crude oil have been rarely reported, it is reasonable to expect that exposure to crude oil that induce apoptosis in kidney may create a critical disturbance in kidney homeostasis, injury to renal tubular epithelial cells, and affect glomerular filtration, active secretion and urine formation, contributing to acute or chronic renal failure54,60,61.
In summary, the first de novo approach applied in this study, was used to determine the global transcriptomic responses of mahi-mahi larvae exposed to DWH oil. Compared with the reference-transcriptome-guided automated OnRamp approach, a large proportion of DEGs, GO terms and toxicity pathways were common, with genes involved in several toxicity-pathways (liver proliferation, cholesterol biosynthesis, cardiac hypertrophy, renal necrosis/apoptosis, TR/RXR and LXR/RXR activation). Genes involved in these pathways may serve as biomarkers to predict toxicities and/or recovery of DWH oil. There is a need to conduct focused follow-up studies to confirm the initial transcriptomic results with additional methods (e.g. dose-response assays) at an expanded range of oil concentrations in multiple fish species. Overall, the present study has provided valuable molecular resources for facilitating further investigations on the holistic understanding of the developmental toxicity of DWH oil in mahi-mahi. The developed de novo and comparative toxicity pathway methods will also be useful for toxicogenomic study on other non-model fish species.
Methods
Animals and DHW oil exposure
Mahi-mahi (Coryphaena hippurus) broodstock were caught off the coast of South Florida using hook and line angling techniques and then directly transferred to University of Miami Experimental Hatchery (UMEH). Broodstock were acclimated in 80 m3 fiberglass maturation tanks equipped with recirculating and temperature controlled water. All embryos used in the experiments described here were collected within 2–10 h following a volitional (non-induced) spawn using standard UMEH methods62. Two sources of crude oil from the DWH spill that varied with respect to the state of weathering were obtained from British Petroleum under chain of custody for testing purposes: (1) slick oil collected from surface skimming operations (sample ID: OFS-20100719-Juniper-001 A0091G) and (2) oil from the Massachusetts barge (sample ID: SO-20100815-Mass-001 A0075K) which received oil collected from the subsea containment system positioned directly over the well (referred to herein as slick and source oil, respectively). Preparation of water accommodated fractions (HEWAFs) of two oil types, and oil exposures were performed at the University of Miami as outlined previously13. Time course (24, 48 and 96 hpf) exposures were performed using the nominal LC25s determined from the aforementioned bioassays for RNA sequencing. The LC25 concentration (2% HEWAF of slick oil or 0.09% HEWAF of source oil) was chosen as a compromise between attempting to capture initiating events as well as significant cascade effects while ensuring that a sufficient signal was observed. Three replicates were used per time point with 25 embryos per replicate. Slick and source oil HEWAF exposures were run concurrently using the same batch of embryos and a shared set of controls for both sets of experiments. A full description of the samples collection and exposure is presented in Xu10. All animal experiments were performed ethically and in accordance with Institutional Animal Care and Use Committee (IACUC protocol number 15–019) approved by the University of Miami IACUC committee, and the institutional assurance number is A-3224–01.
RNA isolation, cDNA library construction and sequencing
The surviving embryos or larvae from each replicate were pooled and RNA was isolated and purified with RNeasy Mini Kit (Qiagen, Valencia, California). The total RNA sample was quantified by NanoDrop ND-1000 Spectrophotometer (Nanodrop Technologies, Wilmington, DE, USA). 200 ng of total RNA was used to prepare RNA-Seq libraries using the TruSeq RNA Sample Prep kit following the protocol described by the manufacturer (Illumina, San Diego, CA). Single Read 1 × 50 sequencing was performed on an Illumina HiSeq 2500 at the Center for Genomics Medicine, Medical University of South Carolina, Charleston, SC, with each individual sample sequenced to a minimum depth of ~50 million reads. Data were subjected to Illumina quality control (QC) procedures (>80% of the data yielded a Phred score of 30). A detailed description of these methods is presented in Xu10. The read data for the 32 samples was deposited in the NCBI database (Accession Number: GSE79675).
De novo assembly and annotation of the Mahi-mahi transcriptome: de novo approach
Raw sequences were trimmed off Illumina adapter sequences and filtered out sequences that did not meet the quality thresholds using Trimmomatic (version 0.33)63. All reads were pooled and assembled de novo using Trinity (version 2.2.0)64 with k-mer length set at 25. According to Grabherr64, Trinity is a de novo algorithm recently developed for reconstructing transcriptome using de Bruijn graphs. Transcriptome assembly is challenging mainly due to the unevenly distributed RNA-Seq data coverage and alternative splicing in individual genes. Trinity consists of three parts: Inchworm, Chrysalis, and Butterfly. Trinity firstly makes linear contigs from RNA-Seq reads, and Chrysalis, the second step of Trinity, clusters minimally overlapping Inchworm contigs into sets of connected components that comprise alternative splice forms or closely related paralogs and then generates and expands de Bruijn graphs, and finally outputs the transcripts as well as isoforms. The process starts by decomposing the reads into small overlapping k-mers and extending them by coverage. Alternative splicing is determined by finding the common sections of the intermediate transcripts, and those transcripts are re-grouped. The de Bruijn graphs are generated by integration of isoforms that are similar except one base. And finally, by finding and expanding the common section of transcripts and representing the most compact path on the graph, Trinity delivers the fully assembled transcriptome64. Subsequently, the high quality reads from 96 hpf larvae samples (slick oil treatments and controls) were mapped to the Trinity assembled de novo mahi-mahi transcriptome to estimate the transcript/unigene abundance using RSEM (RNA-Seq by Expectation-Maximization) (version 1.2.21)65 with the default aligner Bowtie (version 1.1.1)66. The expression level of each transcript/Unigene was measured with FPKM (fragments per kilobase per million reads). A script ‘PtR’ in Trinity was used to compare the biological replicates for each of the samples and generate a correlation matrix and Principal Component Analysis (PCA) plot. Subsequent analyses in RSEM were conducted using a selection of scripts provided as part of the Trinity package (version 2.2.0). Statistical differences in gene expression levels between 96 hpf mahi larvae exposed to slick oil and controls were calculated using DEseq2. Genes were considered differentially expressed when false discovery rate (FDR) < 0.05 (Benjamini−Hochberg correction). Trans-decoder (version 3.0.0) was used for coding sequence prediction, and Trinotate (2.0.2) for functional annotation. Trinotate is a comprehensive annotation suite specifically designed for automatic functional annotation of de novo assembled transcriptomes of non-model organisms, including homology search to known sequence data (BLAST + /SwissProt), protein domain identification (HMMER/PFAM), protein signal peptide and transmembrane domain prediction (signalP/tmHMM), and leveraging various annotation databases (eggNOG/GO/Kegg databases), reporting the best hits in the databases (http://trinotate.github.io/). All analysis was carried out on a local server running under the Institute for Integrative Genome Biology (IIGB)‘s Linux cluster, Biocluster, environment (http://manuals.bioinformatics.ucr.edu/home/hpc). The overall workflow is summarized graphically in Fig. 1.
A reference-transcriptome-guided analysis by OnRamp: OnRamp approach
A secondary analysis was carried out on an OnRamp Bioinformatics Genomics Research Platform (OnRamp Bioinformatics, San Diego, CA) as previously described10. OnRamp’s advanced Genomics Analysis Engine utilized an automated RNAseq workflow to map read alignment to the Takifugu rubripes transcriptome (FUGU4) using BLASTX: Basic Local Alignment Search Tool, generate gene-level count data, and differential expression analysis with DEseq2. Transcript count data from DESeq2 analysis of the samples were sorted according to their false discovery rate (FDR) at which a transcript is called significant. The protein FASTA sequences from Ensembl for Fugu were compared using Ensembl’s homology to create protein FASTA files that contained a human Entrez gene ID that mapped via Fugu to Mahi-mahi. A detailed description of these methods is presented in Xu10.
Gene ontology and Ingenuity Pathway Analyses
The two sorted transcript lists of mahi generated by both de novo and OnRamp approaches were mapped to human orthologs to generate HGNC (HUGO Gene Nomenclature Committee) gene symbols for downstream gene ontology (GO) term analysis, using Gorilla67 and ToppGene Suite68. This approach has been demonstrated to improve functional analysis of fish genes with a more sensitive systems level interrogation, by providing access to the best-annotated databases and tools for human/mouse/rat models10,69,70, while limitations of the mapping due to the extra genome duplication events in teleost fish and species differences in gene functions still exist. GO terms for molecular function, molecular component, biological process, pathway and phenotype were considered significantly enriched when p < 0.05. Considering gene duplication in fish species and the possible functionally different homologues between fish and human/mouse/rat71,72, the same set of the de novo transcript lists were also examined using the enrichment of GO terms against the zebrafish (Danio rerio) reference using DAVID Bioinformatics Resources 6.873. The analysis was performed on categories for biological process (GOTERM_BP_FAT), cellular component (GOTERM_CC_FAT) and molecular function (GOTERM_MF_FAT) using the functional annotation tool with a modified Fisher exact p-value (EASE score) < 0.01.
Comparative toxicity pathway (tox-pathway) analysis was further conducted. If individual genes are not commonly regulated between methods/conditions, commonality may still exist on the level of pathway regulation. Comparative pathway analysis could also provide additional information on modes of action of toxicants. This comparative toxicity pathway analysis closely relates to the concept of adverse outcome pathways determining toxicity22. Ingenuity Pathway Analyses (IPA) (Ingenuity Systems Inc., Redwood City, CA, USA) was used to compare de novo and OnRamp approaches at different oil types and developmental time conditions to identify similarities and differences in canonical pathways and toxicity functions (IPA-Comparison analysis). IPA-Tox is an integral component of an integrated systems toxicology approach specifically for toxicity assessment. This component of IPA is taken from peer-review literature and manually reviewed by experts in the field of toxicology (both traditional and toxicogenomics), drawing on a library of hundreds of signaling and metabolic pathways. It provides pathway content as well as gene to phenotype associations from toxicity studies. By linking DEGs to their known role in specific tox endpoints, analysis of toxicogenomics data in IPA-Tox identifies subsets of DEGs that may be predictive of certain toxicity endpoints. IPA-Tox was used to examine the toxicity pathways and reveal biological pathways/mechanisms underlying toxicity-specific phenotypes, as well as to provide insights into candidate phenotype-specific biomarkers. Fisher’s exact test was used to calculate a p-value determining the probability that the association between the genes in the dataset and the IPA-Tox pathways as opposed to this occurring by chance alone.
Additional Information
How to cite this article: Xu, E. G. et al. Novel transcriptome assembly and comparative toxicity pathway analysis in mahi-mahi (Coryphaena hippurus) embryos and larvae exposed to Deepwater Horizon oil. Sci. Rep. 7, 44546; doi: 10.1038/srep44546 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
This research was made possible by a grant from The Gulf of Mexico Research Initiative. Grant No: SA-1520; Name: Relationship of Effects of Cardiac Outcomes in fish for Validation of Ecological Risk (RECOVER). M. Grosell holds a Maytag Chair of Ichthyology. The authors are grateful to the Gulf of Mexico Research Initiative Information and Data Cooperative (GRIIDC) for supporting data management system to store the data generated. GRIIDC DOI:10.7266/N7BG2M0. DOI: 10.7266/N7MW2F6S. J. G. Hardiman acknowledges Medical University of South Carolina College of Medicine start-up funds. This research was supported in part by the Genomics Shared Resource, Hollings Cancer Center.
Footnotes
The authors declare no competing financial interests.
Author Contributions E.G.X., E.M.M., M.G., G.H. & D.S. designed the experiment. E.M.M. and M.G. collected the animals. E.S.H. carried the RNA sequencing analysis. E.G.X. & E.S.H. carried out downstream data analysis. E.G.X. prepared all figures and tables and wrote the first draft of this manuscript, and D.S., G.H. & M.G. contributed substantially to the revisions of the manuscript.
References
- Peterson C. H. et al. Long-term ecosystem response to the Exxon Valdez oil spill. Science 302, 2082–2086 (2003). [DOI] [PubMed] [Google Scholar]
- Ji K. et al. Genotoxicity and endocrine-disruption potentials of sediment near an oil spill site: Two years after the Hebei Spirit oil spill. Environ. Sci. Technol. 45, 7481–7488 (2011). [DOI] [PubMed] [Google Scholar]
- Allan S. E., Smith B. W. & Anderson K. A. Impact of the Deepwater Horizon oil spill on bioavailable polycyclic aromatic hydrocarbons in Gulf of Mexico coastal waters. Environ. Sci. Technol. 46, 2033–2039 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Block B. A. et al. Electronic tagging and population structure of Atlantic bluefin tuna. Nature 434(7037), 1121−1127 (2005). [DOI] [PubMed] [Google Scholar]
- Muhling B. A. et al. Overlap between Atlantic bluefin tuna spawning grounds and observed Deepwater Horizon surface oil in the northern Gulf of Mexico. Mar. Pollut. Bull. 64(4), 679−687 (2012). [DOI] [PubMed] [Google Scholar]
- Rooker J. R. et al. Spatial, temporal, and habitat-related variation in abundance of pelagic fishes in the Gulf of Mexico: Potential implications of the Deepwater Horizon Oil Spill. PloS one 8(10), e76080 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hicken C. E. et al. Sublethal exposure to crude oil during embryonic development alters cardiac morphology and reduces aerobic capacity in adult fish. Proc. Natl. Acad. Sc. 108(17), 7086−7090 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Incardona J. P. et al. Exxon Valdez to Deepwater Horizon: Comparable toxicity of both crude oils to fish early life stages. Aquat. Toxicol. 142–143, 303–316 (2013). [DOI] [PubMed] [Google Scholar]
- Esbaugh A. J. et al. The effects of weathering and chemical dispersion on Deepwater Horizon crude oil toxicity to mahi-mahi (Coryphaena hippurus) early life stages. Sci. Total Environ. 543, 644–651 (2016). [DOI] [PubMed] [Google Scholar]
- Xu E. G. et al. Time-and oil-dependent transcriptomic and physiological responses to Deepwater Horizon oil in mahi-mahi (Coryphaena hippurus) embryos and larvae. Environ. Sci. Technol. 50(14), 7842–7851 (2016). [DOI] [PubMed] [Google Scholar]
- Incardona J. P., Collier T. K. & Scholz N. L. Defects in cardiac function precede morphological abnormalities in fish embryos exposed to polycyclic aromatic hydrocarbons. Toxicol. Appl. Pharmacol. 196, 191–205 (2004). [DOI] [PubMed] [Google Scholar]
- Huang L., Wang C., Zhang Y., Wu M. & Zuo Z. Phenanthrene causes ocular developmental toxicity in zebrafish embryos and the possible mechanisms involved. J. Hazard. Mater. 261, 172–180 (2013). [DOI] [PubMed] [Google Scholar]
- Mager E. M. et al. Acute embryonic juvenile exposure to deepwater horizon crude oil impairs swimming performance of mahi-mahi (Coryphaena hippurus). Environ. Sci. Tech. 48(12), 7053–706 (2014). [DOI] [PubMed] [Google Scholar]
- Heintz R. A. Chronic Exposure to Polynuclear Aromatic Hydrocarbons in Natal Habitats Leads to Decreased Equilibrium Size, Growth, and Stability of Pink Salmon Populations. Integr. Environ. Assess. Manag. 3, 351–363 (2007). [PubMed] [Google Scholar]
- Rice S. D. et al. Impacts to pink salmon following the Exxon Valdez oil spill: Persistence, toxicity, sensitivity, and controversy. Rev. Fish. Sci. 9, 165–211 (2001). [Google Scholar]
- Sørhus E. et al. Crude oil exposures reveal roles for intracellular calcium cycling in haddock craniofacial and cardiac development. Sci. Rep. 6 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edmunds R. C. et al. Corresponding morphological and molecular indicators of crude oil toxicity to the developing hearts of mahi mahi. Sci. Rep. 5, 17326 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collette B. et al. Coryphaena hippurus. The IUCN Red List of Threatened Species 2011: e.T154712A4614989. 10.2305/IUCN.UK.2011-2.RLTS.T154712A4614989.en. Downloaded on 26 September 2016 (2011). [DOI]
- Yang L. et al. Transcriptional profiling reveals barcode-like toxicogenomic responses in the zebrafish embryo. Genome Biol. 8(10), p.1 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Dartel D. A. M., Pennings J. L. A., Robinson J. F., Kleinjans J. C. S. & Piersma A. H. Discriminating classes of developmental toxicants using gene expression profiling in the embryonic stem cell test. Toxicol. Lett. 201, 143–151 (2011). [DOI] [PubMed] [Google Scholar]
- Hermsen S. A., Pronk T. E., van den Brandhof E. J., van der Ven L. T. & Piersma A. H. Transcriptomic analysis in the developing zebrafish embryo after compound exposure: individual gene expression and pathway regulation. Toxicol. Appl. Pharmacol. 272(1), 161–171 (2013). [DOI] [PubMed] [Google Scholar]
- Ankley G. T. et al. Adverse outcome pathways: a conceptual framework to support ecotoxicology research and risk assessment. Environ. Toxicol. Chem. 29(3), 730–741 (2010). [DOI] [PubMed] [Google Scholar]
- Mehendale H. M. Tissue repair: an important determinant of final outcome of toxicant-induced injury. Toxicol. Pathol. 33(1), 41–51 (2005). [DOI] [PubMed] [Google Scholar]
- Wislocki P. G. Tumorigenicity of nitrated derivatives of pyrene, benz [a] anthracene, chrysene and benzo [a] pyrene in the newborn mouse assay. Carcinogenesis, 7(8), 1317–1322 (1986). [DOI] [PubMed] [Google Scholar]
- Ortiz L., Nakamura B., Li X., Blumberg B. & Luderer U. Reprint of “In utero exposure to benzo[a]pyrene increases adiposity and causes hepatic steatosis in female mice, and glutathione deficiency is protective”. Toxicol. Lett. 230, 314–321 (2014). [DOI] [PubMed] [Google Scholar]
- Olsvik P. A., Lie K. K., Nordtug T. & Hansen B. H. Is chemically dispersed oil more toxic to Atlantic cod (Gadus morhua) larvae than mechanically dispersed oil? A transcriptional evaluation. BMC genomics, 13(1), p.1 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michalopoulos G. K. Principles of liver regeneration and growth homeostasis. Compr. Physiol. 485–513 (2013). [DOI] [PubMed] [Google Scholar]
- Li J. et al. Neutralization of chemokine CXCL14 (BRAK) expression reduces CCl4 induced liver injury and steatosis in mice. Eur. J. Pharmacol. 671(1-3), 120–7 (2011). [DOI] [PubMed] [Google Scholar]
- Ding X. et al. The roles of leptin and adiponectin: a novel paradigm in adipocytokine regulation of liver fibrosis and stellate cell biology. Am. J. Pathol. 166(6), 1655–69 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan W. et al. Ligand activation of peroxisome proliferator-activated receptor beta/delta (PPARbeta/delta) attenuates carbon tetrachloride hepatotoxicity by downregulating proinflammatory gene expression. Toxicol. Sci. 105(2), 418–28 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He X. et al. PIK3IP1, a negative regulator of PI3K, suppresses the development of hepatocellular carcinoma. Cancer Res. 68(14), 5591–8 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Do N. et al. BMP4 is a novel paracrine inhibitor of liver regeneration. Am. J. Physiol. Gastrointest. Liver Physiol. 303(11), G1220–7 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Field H. A., Ober E. A., Roeser T. & Stainier D. Y. Formation of the digestive system in zebrafish. I. Liver morphogenesis. Dev. Biol. 253(2), 279–290 (2003). [DOI] [PubMed] [Google Scholar]
- Robichon C. & Dugail I. De novo cholesterol synthesis at the crossroads of adaptive response to extracellular stress through SREBP. Biochimie 89(2), 260–264 (2007). [DOI] [PubMed] [Google Scholar]
- Spear D. H., Kutsunai S. Y., Correll C. C. & Edwards P. A. Molecular cloning and promoter analysis of the rat liver farnesyl diphosphate synthase gene. J. Biol. Chem. 267(20), 14462–9 (1992). [PubMed] [Google Scholar]
- Nagashima S. et al. Plasma cholesterol-lowering and transient liver dysfunction in mice lacking squalene synthase in the liver. J. Lipid Res. 56(5), 998–1005 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Björkhem I., Leoni V. & Meaney S. Genetic connections between neurological disorders and cholesterol metabolism. J. Lipid Res. 51(9), 2489–503 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akadam-Teker B. et al. The effects of age and gender on the relationship between HMGCR promoter-911 SNP (rs33761740) and serum lipids in patients with coronary heart disease. Gene. 528(2), 93–8 (2013). [DOI] [PubMed] [Google Scholar]
- Moebius F. F., Fitzky B. U., Lee J. N., Paik Y. K. & Glossmann H. Molecular cloning and expression of the human delta7-sterol reductase. Proc. Natl. Acad. Sci. 95(4), 1899–902 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter C. J. Convergence of genes implicated in Alzheimer’s disease on the cerebral cholesterol shuttle: APP, cholesterol, lipoproteins, and atherosclerosis. Neurochem. Int. 50(1), 12–38 (2007). [DOI] [PubMed] [Google Scholar]
- Orsoni A. et al. Impact of LDL apheresis on atheroprotective reverse cholesterol transport pathway in familial hypercholesterolemia. J. Lipid Res. 53(4), 767–75 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y. et al. Regulation of cholesterologenesis by the oxysterol receptor, LXRalpha. J. Biol. Chem. 283(39), 26332–9 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin D. J., Plateroti M., Samarut J. & Osborne T. F. Two uniquely arranged thyroid hormone response elements in the far upstream 5′ flanking region confer direct thyroid hormone regulation to the murine cholesterol 7alpha hydroxylase gene. Nucleic. Acids Res. 34(14), 3853–61 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanos R. et al. Aryl hydrocarbon receptor regulates the cholesterol biosynthetic pathway in a dioxin response element‐independent manner. Hepatology 55(6), 1994–2004 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beard N. A., Laver D. R. & Dulhunty A. F. Calsequestrin and the calcium release channel of skeletal and cardiac muscle. Prog. Biophys. Mol. Biol. 85(1), 33–69 (2004). [DOI] [PubMed] [Google Scholar]
- Sun B. et al. Bone morphogenetic protein-4 mediates cardiac hypertrophy, apoptosis, and fibrosis in experimentally pathological cardiac hypertrophy. Hypertension 61(2), 352–360 (2013). [DOI] [PubMed] [Google Scholar]
- Metzger J. M. & Westfall M. V. Covalent and noncovalent modification of thin filament action: the essential role of troponin in cardiac muscle regulation. Circ. Res. 94(2), 146–158 (2004). [DOI] [PubMed] [Google Scholar]
- Simonides W. S. et al. Characterization of the promoter of the rat sarcoplasmic endoplasmic reticulum Ca2+-ATPase 1 gene and analysis of thyroid hormone responsiveness. J. Biol. Chem. 271(50), 32048–56 (1996). [DOI] [PubMed] [Google Scholar]
- Brette F. et al. Crude oil impairs cardiac excitation-contraction coupling in fish. Science, 343(6172), 772–776 (2014). [DOI] [PubMed] [Google Scholar]
- Hohendanner F. et al. Inositol-1,4,5-trisphosphate induced Ca2+ release and excitation-contraction coupling in atrial myocytes from normal and failing hearts. J. Physiol.-London 593, 1459–1477 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubansky B., Whitehead A., Miller J. T., Rice C. D. & Galvez F. Multitissue molecular, genomic, and developmental effects of the Deepwater Horizon oil spill on resident Gulf killifish (Fundulus grandis). Environ. Sci. Technol. 47(10), 5074–5082 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das M., Garg K., Singh G. B. & Khanna S. K. Attenuation of benzanthrone toxicity by ascorbic acid in guinea pigs. Toxicol. Sci. 22(3), 447–456 (1994). [DOI] [PubMed] [Google Scholar]
- Incardona J. P. & Scholz N. L. The influence of heart developmental anatomy on cardiotoxicity-based adverse outcome pathways in fish. Aquatic Toxicology, 177, 515–525 (2016). [DOI] [PubMed] [Google Scholar]
- Schlenk D. & Benson W. H. eds Target organ toxicity in marine and freshwater teleosts: Organs. CRC press (2003). [Google Scholar]
- Charette S. J., Lavoie J. N., Lambert H. & Landry J. Inhibition of Daxx-mediated apoptosis by heat shock protein 27. Mol. Cell. Biol. 20(20), 7602–12 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm S., Stanger B. Z. & Leder P. RIP and FADD: two “death domain”-containing proteins can induce apoptosis by convergent, but dissociable, pathways. Proc. Natl. Acad. Sci. 93(20), 10923–7 (1996). [DOI] [PMC free article] [PubMed]
- Oka T., Schmitt A. P. & Sudol M. Opposing roles of angiomotin-like-1 and zona occludens-2 on pro-apoptotic function of YAP. Oncogene 31(1), 128–34 (2012). [DOI] [PubMed] [Google Scholar]
- Rider L., Shatrova A., Feener E. P., Webb L. & Diakonova M. JAK2 tyrosine kinase phosphorylates PAK1 and regulates PAK1 activity and functions. J. Biol. Chem. 282(42), 30985–96 (2007). [DOI] [PubMed] [Google Scholar]
- Luo Y. et al. Metabolic regulator betaKlotho interacts with fibroblast growth factor receptor 4 (FGFR4) to induce apoptosis and inhibit tumor cell proliferation. J. Biol. Chem. 285(39), 30069–78 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieberthal W. & Levine J. S. Mechanisms of apoptosis and its potential role in renal tubular epithelial cell injury. Am. J. Physiol Renal Physiol. 271(3), pp F477–F488 (1996). [DOI] [PubMed] [Google Scholar]
- Sanz A. B., Santamaría B., Ruiz-Ortega M., Egido J. & Ortiz A. Mechanisms of renal apoptosis in health and disease. J. Am. Soc. of Nephrol. 19(9), 1634–1642 (2008). [DOI] [PubMed] [Google Scholar]
- Stieglitz J. D. et al. Environmentally conditioned, year-round volitional spawning of cobia (Rachycentron canadum) in broodstock maturation systems. Aquacult. Res. 43, 1557−1566 (2012). [Google Scholar]
- Bolger A. M., Lohse M. & Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics, p.btu170 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grabherr M. G. et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nature Biotechnol. 29(7), 644–652 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li B. & Dewey C. N. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC bioinformatics, 12(1), p.1 (2011) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langmead B., Trapnell C., Pop M. & Salzberg S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10(3), p.1 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eden E., Navon R., Steinfeld I., Lipson D. & Yakhini Z. GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC bioinformatics, 10(1), p.1 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J., Bardes E. E., Aronow B. J. & Jegga A. G. ToppGene Suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res. 37(suppl 2), pp.W305–W311 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yadetie F. et al. Global transcriptome analysis of Atlantic cod (Gadus morhua) liver after in vivo methylmercury exposure suggests effects on energy metabolism pathways. Aquat. Toxicol. 126, 314–25 (2013). [DOI] [PubMed] [Google Scholar]
- Król E. et al. Differential responses of the gut transcriptome to plant protein diets in farmed Atlantic salmon. BMC genomics, 17(1), p.1 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wittbrodt J., Meyer A. & Schartl M. More genes in fish? BioEssays, 20(6), pp.511–515 (1998). [Google Scholar]
- Taylor J. S., Braasch I., Frickey T., Meyer A. & Van de Peer Y. Genome duplication, a trait shared by 22,000 species of ray-finned fish. Genome Res. 13(3), pp.382–390 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang D. W., Sherman B. T. & Lempicki R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4(1), 44–57 (2009). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.