Abstract
Stroke is a leading cause of disability in the United States. Available treatments for stroke have only a modest effect on motor rehabilitation and about 50-60% of stroke patients remain with some degree of motor impairment after standard treatment. Non-invasive brain stimulation (NIBS) techniques have been proposed as adjuvant treatments to physical therapy for motor recovery after stroke. High frequency rTMS and anodal tDCS can be delivered over the affected motor cortex in order to increase cortical excitability and induce brain plasticity with the intention to enhance motor learning and achieve functional goals in stroke patients. Similarly, low frequency rTMS and cathodal tDCS can be delivered to the unaffected motor cortex to reduce interhemispheric inhibition and hinder maladaptive plasticity. The use of several drugs such as amphetamines, selective serotonin reuptake inhibitors (SSRIs), levodopa and cholinergic agents have been also proposed to enhance the motor function. Given that both NIBS and pharmacotherapy might provide some treatment effect independently for motor rehabilitation in stroke and with the rationale that they could work in a synergistic fashion, we believe that a combined therapy- NIBS plus pharmacotherapy- canlead to better outcomes than one or the other alone. In this paper we review the literature that support the potential use of a combined approach in stroke recovery and present the studies that have already investigated this idea.
1. Introduction
Stroke is the 4th cause of death and a leading cause of long term disability in the United States. Forty percent of stroke patients experience moderate functional impairments and 15% to 30% with severe disability [1]. The standard treatment for sub-acute and chronic stroke rehabilitation is a combination of physical, occupational and speech therapy, but still, after completing standard motor rehabilitation, about 50–60% of stroke patients experience some degree of motor deficits [2], highly impacting the quality of life of patients and their families. Thus there is a substantial need for developing novel therapies.
Novel therapies aiming at improving motor recovery have been investigated by many researchers. Several studies have demonstrated that noninvasive brain stimulation(NIBS) techniques such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS)can induce brain plasticity and have treatment effects in post-stroke motor recovery [3, 4]. However, the clinical significance of these outcomes is somewhat modest and, despite some promising results, two recent systematic reviews suggested that more information is needed to support the use of rTMS and tDCS in stroke recovery [5, 6]. With the same aim, amphetamines [7-9], levodopa [10, 11], cholinergic agents [12, 13] and SSRI's [14-16] have been tested for motor recovery post stroke and have shown to improve motor learning and enhance motor recovery, however, also modestly. Current research suggests that both interventions – NIBS and pharmacotherapy- have a small treatment effect. While not proven, it is possible that by combining these two approaches a large clinical effect could be achieved [17].
In this literature review we present the rationale behind this combined approach by reviewing both methods, NIBS and pharmacotherapy, independently and then we discuss the results of two available clinical studies that have already tested this combined therapy.
2. Stroke and cortical plasticity
Neuroplasticity is the capacity of the brain to change and adapt itself in response to different environmental stimuli. During the past decade, a large number of studies have demonstrated that the cerebral cortex interconnections are modifiable by behavioral manipulations and motor tasks learning. Animal models have shown that skill learning is accompanied by structural changes in cortical neurons, like dendritic growth and arborization, synapse formation, increased synapse strength and subsequent increase in the size of cortical motor representations and thickening of the motor cortex [18, 19].
After stroke, reorganization of the remaining healthy brain is the key for recovery to occur [20]. Several trials have indicated that the cerebral cortex undergoes functional and structural reorganization for weeks and months after injury with measurable compensatory changes [21]. Early recovery depends on the amount and time of resolution of the brain edema, the reperfusion of the ischemic penumbra and the impact that diaschisis had on different brain functions [20]. On the other hand, later recovery depends on the central nervous system (CNS) reorganization and plasticity [22].
Molecular alterations following brain injury also have an important effect in post stroke recovery. Studies have shown that excitability changes are related with down-regulation of Gamma-Amino butyric Acid (GABA) receptors and enhancement of N-Methyl-D-Aspartame (NMDA) receptors, in both peri-lesional and remote areas [23]. These processes can extend for a longer period of time and are highly influenced by rehabilitation and other interventions. In fact, several studies have shown that interventions like pharmacotherapy and NIBS, when used independently, induce significant changes in cortical plasticity.
If both interventions have positive but limited effects in motor recovery, it is possible that by combining them, a summatory effect can be achieved. In the next sections we will describe the information supporting the use of these therapies, and then the rationale behind combing them to obtain greater effects in cortical excitability and clinical outcomes.
3. Non-invasive brain stimulation for the treatment of stroke
Non-invasive stimulation of the brain has the capacity to modulate cortical excitability and induce brain plasticity. The mechanisms underneath these processes are not fully understood, but it's known that in part, they are mediated by long-term potentiation (LTP) and depression (LTD). LTP is the phenomena observed when stimulation is delivered repeatedly to the brain and a robust neural excitability occurs, enhancing synaptic efficacy and postsynaptic excitability [24]. The mechanisms behind this include NMDA receptor activity, synaptogenesis and modulation of GABA activity [24]. In the counterpart, LTD occurs when synaptic efficacy becomes diminished. This is mediated by a reduction in NMDA receptor activity and an increase in GABA activity. These two mechanisms of synaptic plasticity can be used clinically with the purpose of incrementing or inhibiting brain activity in different areas of the motor pathways [25].
There are several models on how brain plasticity changes the brain responses after stroke and how NIBS can modulate them. The most accepted model until the date is the interhemispheric competition model, which states that by suppressing the over-activity on the unaffected hemisphere, we can enhance recovery by reducing the interhemispheric inhibition over the stroke hemisphere. Following this model, there are two main strategies to induce brain plasticity via LTP and LTD: 1) to increase cortical excitability in the affected hemisphere using either excitatory anodal tDCS or high frequency rTMS, and 2) to decrease the over activity that occurs in the healthy hemisphere, which causes further inhibition of the lesioned one by the process of transcallosal inhibition, using inhibitory low frequency rTMS or cathodal tDCS. In the following section we will address these techniques, and previous experience of their use in healthy subjects and in stroke rehabilitation [26].
Another theory recently proposed by di Pino et al. suggests a bimodal balance–recovery model that combines the idea of inter hemispheric balancing and the structural recovery of the areas not directly affected by the stroke. This model suggests that, if NIBS is to be used to enhance motor recovery, then the technique and parameters should be defined according to the individual needs of patients depending on their lesion. This model, though very promising, still needs to be furthered investigated [27].
tDCS
tDCS has been widely tested in the last 2 decades for the treatment of neurological and psychiatric conditions. It is now known that tDCS can be used to increase or decrease cortical excitability in the area of stimulation; thus can guide brain plasticity for the recovery of several neurological conditions [28]. In healthy subjects, anodal tDCS over the motor cortex facilitates neuronal firing and induces cortical excitability measured by an increase in the motor evoked potential (MEP) amplitude. In an opposite way, cathodal tDCS decreases neuronal excitability. The lasting effect of the modulation induced by tDCS and the occurrence of LTP or LTD depends on the parameters of the stimulation and the amount of stimulation sessions [29]. Taking these changes in cortical excitability to a clinical scenario, anodal tDCS was proven to enhance motor performance [30] and learning when administered over the primary motor cortex. In the other hand, cathodal tDCS was found to have no effect [31, 32].
In stroke patients, several studies have shown that increasing the excitability of the motor cortex using tDCS can improve the motor performance and hand motor tasks [33]; and if administered for 7 days, its effects would be prolonged [34]. Most recent studies using functional magnetic resonance imaging (fMRI) also reported an increase in motor related activity and enhanced motor function after anodal tDCS over M1 in the lesioned hemisphere [35]. Furthermore, several studies have reported that inhibiting the contralesional hemisphere using cathodal tDCS on M1 can improve the motor recovery post stroke [36]. A recent study demonstrated that reducing the excitability of the intact hemisphere significantly enhanced motor learning in the paretic hand in stroke patients for up to 24 hours [37]. These results are consistent with another study that showed a significant enhancement of movement induced fMRI activity in the ipsi- lesionalhemisphere after cathodal tDCS in the intact hemisphere [35].
It should be noted that, despite the positive studies mentioned, a recently published systematic review concluded that the evidence supporting the use of tDCS in stroke recovery is of low quality and that further research is needed.
rTMS
Transcranial magnetic stimulation (TMS) delivered in a repetitive fashion has been seen to modulate neuronal firing and either excite or depress brain activity. In healthy volunteers, high frequency rTMS increased cortical excitability measured by a decrease in the motor threshold (MT) and an increase in the MEP amplitude and low frequency rTMS has been seen to depress cortical excitability and have the opposite effects in the MT and MEP [38-40]. The significance of these effects has also been demonstrated functionally in healthy subjects. In healthy volunteers, high frequency rTMS over M1 can improve motor learning in the contralateral upper limbbut decrease motor performance in the ipsilateral extremity [41]. Following a similar mechanism, low frequency rTMS was found to improve motor function in the contralateral hand [42].
In stroke patients, several studies reported improvement in motor function [43] and significant improvement in disability ratings that lasted for up to 10 days after stimulation with rTMS [40]. More recent studies have also shown modest improvement in upper limb functionality, especially grip strength, range of motion and pegboard performance [44]. rTMS can also be delivered in the contralesional hemisphere in the intent of reestablishing the inter-hemispheric activation/inhibition balance. In stoke patients, several randomized clinical trials have been conducted and demonstrated that inhibiting the motor cortex in the contralesional hemisphere improves pinch grip, reduces simple and complex reaction times and improves the scores in several motor function assessments in patients with upper limb dysfunction [45-47].
4. Pharmacotherapy for treatment of stroke
In the interest of finding alternatives for motor recovery after stroke, several pharmacological interventions have also been studied and found beneficial. Animal and human studies indicate that functional reorganization of brain connections may be pharmacologically influenced and that different substances can enhance motor rehabilitation in the sub-acute and chronic stages after stroke. The principle behind this concept is that the modulation of specific neurotransmitters or neurotrophic systems may facilitate neuronal plasticity and long term potentiation. The drugs that have been tested in this field are mainly amphetamines, dopaminergic agents, SSRI's, cholinergic substances and selective nor-epinephrine re-uptake inhibitors.
Studies in healthy volunteers demonstrated that amphetamines enhance the effects of motor training dependent plasticity and methylphenidate (MP) has been seen to increase the MEP amplitude [48]. Clinical trials conducted in patients with hemi-paretic stroke haveshowed significant improvement in motor performance [7]. These results were later confirmed by et al, and Sonde et al, in different studies [8, 9]. However, more recent studies show mixed results [49]. It needs to be considered the disadvantage of amphetamines related to their side-effects. Patients treated by amphetamines can potentially show increase blood pressure and cardiac arrhythmias. Even though, some studies have shown positive results, the evidence is not enough to support the routine use of amphetamines for motor recovery after stroke.
It has been determined that dopamine has the ability of modulating motor function. In a TMS study levodopa was seen to increase LTP-like plasticity in magnitude and duration [50]. In another study, dopamine agonists had no effect on cortical plasticity, but haloperidol, a dopamine antagonist, suppressed LTP-like plasticity measured by the MEP amplitude [51]. In healthy humans, levodopa administered prior to motor training was found to have an enhancing effect in motor memory and hand activities of daily living [11, 52]. A study results showed that motor function was significantly better in stroke patients who received levodopa and physiotherapy [10], however, a recent study showed just a trend for improvement in patients with stroke who received levodopa [53]. In the other hand, dopamine antagonists were seem to delay recovery [54]. These results were consistent in patients with sub-acute and chronic stroke [11]. More studies are needed in order to assess the efficacy of levodopa in post stroke recovery.
Cholinergic agents have also been proposed to have an excitatory effect on the motor function. In healthy subjects the cholinesterase inhibitor rivastigmine, strongly increased the magnitude and duration of LTP- mediated plasticity [50]. This system has also been tested in several designs in stroke patients. It was demonstrated that motor memory formation with training was improved by tacrine (ACE- Inhibitor) but reduced with muscarinic antagonists [12, 52]. A case study in a stroke patient showed dramatic improvement in the lower limb motor function as a result of treatment with donepezil and a subsequent trial showed gains in motor function scales in patients taking this drug [13]. More information that supports the positive effects of cholinergic drugs in post stroke recovery is needed.
Serotonin has a well-known role in behavior and motor control. Serotonin modulating drugs have been tested in healthy volunteers and were seen to have an excitatory effect on cortical neurons, demonstrated by an increase in the MEP amplitude [15, 55]. Several SSRI's have been tested for motor recovery obtaining mixed results. Paroxetine showed an improvement in motor performance in healthy subjects and in patients with chronic stroke andthe use of fluoxetine resulted in an increased activation of the motor cortex and better motor skills of the affected hand [14, 15]. In the same way, fluoxetine was proven to be effective in conjunction with physical therapy for 3 months in the recovery on motor function post stroke, in independence and quality of life [16, 56]. Despite these positive results, more information and larger sample sizes are needed to support the use of SSRIs in the clinical practice.
5. Combined Therapy
The combination of non-invasive brain stimulation techniques with pharmacotherapy has been widely tested for the treatment of depression demonstrating its safety and obtaining positive results [57-60]. In the case of stroke recovery there is little evidence. With the knowledge summarized in this article, we propose a therapeutic approach for motor recovery in stroke which can potentiate the mechanisms of plasticity: a combined therapy using drugs inducing neural plasticity in combination with noninvasive brain stimulation (NIBS) techniques over the primary motor cortex in the affected hemisphere (high frequency rTMS or anodal tDCS) or inhibitory brain stimulation over the unaffected hemisphere (low frequency rTMS or cathodal tDCS), in order to facilitate motor recovery in patients within the sub-acute to chronic stages post stroke.
To understand the possible beneficial effects that pharmacotherapy might have in motor recovery when combined with NIBS as facilitators of plasticity, we would like to present the biological model of SSRIs in motor recovery as an example of this combined approach. Motor potentiation has been proved by pharmacological agents such as serotonin in both animals and humans [16]. Its neuro-modulatory role influences directly and indirectly the brain motor activity, as observed in imaging studies, where a single low dose of serotonin was able to modified motor activation maps in healthy subjects [14]. SSRIs enhance 5-hydroxytryptamine (5-HT) neurotransmission by increasing serotonin availability in the extracellular space. Neurophysiologically, there is a relationship between the level of motor activity and the serotoninergic neuronal action across the raphe nuclei with modulation of the sleep-wake-arousal cycle, demonstrating the importance of this system in the onset/offset of motor behavior. Moreover, modulation of 5-HT neuron activity is closely linked to the glutamatergic and GABAergic inputs, thus, the serotoninergic systems facilitates the motor output. The 5-HT system coordinates the autonomic and neuroendocrine functions in the presence of motor demands by inhibiting irrelevant sensory information; consequently, transcranial neuromodulation may increase the activity of serotoninergic pathways. In addition, serotonin may enhance the activation of genes and cell second messengers in order to induce long-term facilitation and synaptogenesis between sensory and motor neurons and facilitation of synaptic strength [61].
An aspect that needs to be discussed when using a combined approach is the question of: who primes who? One possibility is that drugs, when given in combination with NIBS, are the ones who prime the effects that brain stimulation has on plasticity and cortical excitability, increasing the distant effects further away from the area of stimulation and spreading the effects to deeper structures of the brain. In the counterpart, another possibility is that NIBS is the one priming the effects of the drug administered. In this case we will see that by inducing neurotransmitter and receptor modulation, the effects of NIBS will become stronger in the localized network area where either rTMS or tDCS was delivered and modulating cortical excitability and plasticity (Figure 1.). The possibility of a mutually enhancing effect, and the discussion of ‘who primes who’, is still a matter of debate and more information is needed to determine what is the real process behind the possible summatory effect these two approaches may have when combined.
Figure 1.
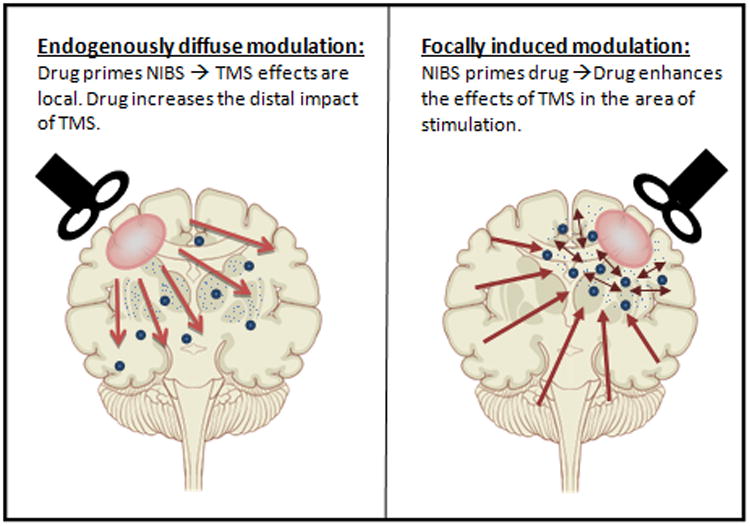
Who primes who dilemma. A) Long-distance NIBS effects can be induced by the action of the drug in deeper neural structures or B) Local network enhancement via drug interaction with the focal modulation offered by NIBS.
6. Existing research findings
With the objective of supporting this rationale, we conducted a search in Pubmed, Cochrane and EMBASE databases for studies that assessed the effects of the combination of NIBS with pharmacotherapy for the motor recovery in stroke patients. We restricted the search to studies in humans and published in English. We did not restrict the search by the study design. We found two studies that met this criterion and their results are described below (Table 1.).
Table 1. Research Studies: Combined therapy for stroke - NIBS plus.
| Study | N of patients | Age | Sex (%female) | NIBS technique | Drug used | Outcome |
|---|---|---|---|---|---|---|
| Wang Q. M., et al 2014 | 9 | 52.8 | 22% | tDCS | MP | Purdue pegboard (PTT) and TMS |
| Kakuda et al., 2011 | 5 | 61 | 40% | Low Freq. rTMS | Levodopa | FMS and WMFT |
A randomized controlled trial by Wang QM. et al.(2014) assessed the effects of MP in combination with tDCS in upper limb motor recovery in patients with sub-acute stroke. 9 patients within a month of an ischemic or hemorrhagic event were randomized to one of three groups: active tDCS- placebo MP, sham tDCS – MP or active tDCS- MP. The interventions were either sham or active tDCS at 1mA for 20 minutes over the primary motor cortex in combination with real or placebo 1 time oral dose of 20 mg of MP. The two main outcomes were cortical excitability measured by TMS and hand motor function assessed by the Purdue pegboard test. Hand motor function, measured before and after treatment, showed to be significantly different amongst the groups and a post-hoc analysis indicated significantly better results in the group of the combined therapy than tDCS or MP alone. In contrast there was no significant difference in cortical excitability tested by TMS amongst any of the groups (Table 2.)[62].
Table 2.
Post hoc multiple comparisons of the results in the performance on the Purdue pegboard test. RCT by Wang Q.M. et al. on tDCS in combination with MP for motor recovery post stroke.
| Intervention | Mean difference | SE | p value | 95% confidence interval |
|---|---|---|---|---|
| tDCS+MP vs tDCS | 0.89 | 0.22 | 0.017 | 0.21 – 1.56 |
| tDCS+MP vs MP | 1.0 | 0.22 | 0.010 | 0.32 – 1.68 |
| tDCS vs MP | 0.1 | 0.22 | 0.874 | -0.57 – 0.79 |
Kakuda et al (2011) performed an open label pilot study to assess the safety and the efficacy of combining levodopa with inhibitory rTMS in the contralesional hemisphere with occupational therapy for the enhancement of motor recovery in patients with chronic stroke. Five patients with ischemic or hemorrhagic stroke were admitted to the hospital to receive 15 days of the combined therapy. The intervention consisted in 100 mg/day of levodopa started 1 week before admission and 22 treatments sessions of rTMS and occupational therapy. Low frequency rTMS was administered to the contralesional M1 for 20 minutes in the morning and 20 minutes in the afternoon in combination with 2 hours of motor training in the morning and other 2 hours in the afternoon for 15 days. The treatment with levodopa continued for 4 weeks after discharge. The main outcomes in this study were the Fugl-Meyer Assessment score (FMA) and the Wolf-Motor Function Test (WMFT). The results were positive for the five patients, obtaining significantly increased scores in the FMA scores that were maintained for up to 4 weeks in four of the patients. In the same way, the sum of performance times for 15 tasks in the Wolf-Motor function test shortened over the treatment period and remained that way in all the patients (Table 3.)[63].
Table 3.
Comparison of pre, post and 4 weeks after the combined treatment of levodopa, occupational therapy and rTMS. Open label trial by Kakuda et al. *
| Test | Pre-treatment (SD) | Post-treatment (SD) | 4 weeks after treatment (SD) |
|---|---|---|---|
| FMA (points) | 40.4 (11.71) | 43 (11.55) | 44.4 (9.96) |
| Total performance time of 15 WMFT tasks (sec) | 663.8 (467.81) | 482 (403.91) | 468 (397.30) |
| Total score of FAS of 15 WMFT tasks (points) | 37.6 (9.60) | 44.4 (8.23) | 43.2 (9.12) |
There was no statistical analysis performed in this study.
Despite the fact that both studies described show positive results supporting the novel idea of a combined therapy for motor recovery in stroke, there are strong limitations that need to be mentioned. In the study by Wang et al., data suggested that hand function may improve significantly with tDCS in combination with MP compared with wither therapies alone. Even though justified by the exploratory nature of this study, the short duration of the intervention, only one session of tDCS and one dose of MP, is an important limitation of this study. Also the sample size would need to be increased in order to assess the real effects of this intervention. The work by Kakuda et al. also has several limitations to be mentioned. The small number of patients included and the open label nature of the study does not allow a real comparison to assess the efficacy of this treatment in comparison to other therapies. Also there is no statistical analysis performed to allow us to know if their results are statistically significant. The results of both of these studies need to be confirmed by larger randomized control studies.
7. Conclusions
After decades of research in the field, there is enough evidence to suggest that brain plasticity after stroke is the basis for function recovery and rehabilitation. Strategies like NIBS and several pharmacological approaches have been seen to induce brain plasticity and modulate brain activity in order to achieve functional goals. rTMS and tDCS have both been widely tested and have shown to have an effect in the enhancement of the effects of physical therapy for stroke motor rehabilitation. In the same way, pharmacological treatments such as SSRI's, cholinomimetics, amphetamines and dopamine agonists have induced improvement in this population in experimental setups. It is not completely clear if NIBS could potentiate the action of CNS acting drugs or vice versa, but we can predict a summatory effect leading to functional improvement. New approaches are being addressed at the moment and hopefully new strategies will come up in the next few years. There is still the need of evidence that a combined therapy could induce brain plasticity more than one or the other alone and that this approach could have a positive effect on motor recovery compared to the conventional treatments.
Acknowledgments
This work was partially supported by a NIH grant (1R21HD079048-01A1)
References
- 1.Thom T, et al. Heart disease and stroke statistics--2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113(6):e85–151. doi: 10.1161/CIRCULATIONAHA.105.171600. [DOI] [PubMed] [Google Scholar]
- 2.Hendricks HT, et al. Systematic review for the early prediction of motor and functional outcome after stroke by using motor-evoked potentials. Arch Phys Med Rehabil. 2002;83(9):1303–8. doi: 10.1053/apmr.2002.34284. [DOI] [PubMed] [Google Scholar]
- 3.Hsu WY, et al. Effects of repetitive transcranial magnetic stimulation on motor functions in patients with stroke: a meta-analysis. Stroke. 2012;43(7):1849–57. doi: 10.1161/STROKEAHA.111.649756. [DOI] [PubMed] [Google Scholar]
- 4.Marquez J, et al. Transcranial direct current stimulation (tDCS): Does it have merit in stroke rehabilitation? A systematic review. Int J Stroke. 2013 doi: 10.1111/ijs.12169. [DOI] [PubMed] [Google Scholar]
- 5.Elsner B, et al. Transcranial direct current stimulation (tDCS) for improving function and activities of daily living in patients after stroke. Cochrane Database Syst Rev. 2013;11:CD009645. doi: 10.1002/14651858.CD009645.pub2. [DOI] [PubMed] [Google Scholar]
- 6.Hao Z, et al. Repetitive transcranial magnetic stimulation for improving function after stroke. Cochrane Database Syst Rev. 2013;5:CD008862. doi: 10.1002/14651858.CD008862.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Walker-Batson D, et al. Amphetamine paired with physical therapy accelerates motor recovery after stroke. Further evidence. Stroke. 1995;26(12):2254–9. doi: 10.1161/01.str.26.12.2254. [DOI] [PubMed] [Google Scholar]
- 8.Crisostomo EA, et al. Evidence that amphetamine with physical therapy promotes recovery of motor function in stroke patients. Ann Neurol. 1988;23(1):94–7. doi: 10.1002/ana.410230117. [DOI] [PubMed] [Google Scholar]
- 9.Sonde L, et al. A double-blind placebo-controlled study of the effects of amphetamine and physiotherapy after stroke. Cerebrovasc Dis. 2001;12(3):253–7. doi: 10.1159/000047712. [DOI] [PubMed] [Google Scholar]
- 10.Scheidtmann K, et al. Effect of levodopa in combination with physiotherapy on functional motor recovery after stroke: a prospective, randomised, double-blind study. Lancet. 2001;358(9284):787–90. doi: 10.1016/S0140-6736(01)05966-9. [DOI] [PubMed] [Google Scholar]
- 11.Floel A, et al. Dopaminergic effects on encoding of a motor memory in chronic stroke. Neurology. 2005;65(3):472–4. doi: 10.1212/01.wnl.0000172340.56307.5e. [DOI] [PubMed] [Google Scholar]
- 12.Sawaki L, et al. Cholinergic influences on use-dependent plasticity. J Neurophysiol. 2002;87(1):166–71. doi: 10.1152/jn.00279.2001. [DOI] [PubMed] [Google Scholar]
- 13.Nadeau SE, et al. Donepezil as an adjuvant to constraint-induced therapy for upper-limb dysfunction after stroke: an exploratory randomized clinical trial. J Rehabil Res Dev. 2004;41(4):525–34. doi: 10.1682/jrrd.2003.07.0108. [DOI] [PubMed] [Google Scholar]
- 14.Loubinoux I, et al. A single dose of the serotonin neurotransmission agonist paroxetine enhances motor output: double-blind, placebo-controlled, fMRI study in healthy subjects. Neuroimage. 2002;15(1):26–36. doi: 10.1006/nimg.2001.0957. [DOI] [PubMed] [Google Scholar]
- 15.Gerdelat-Mas A, et al. Chronic administration of selective serotonin reuptake inhibitor (SSRI) paroxetine modulates human motor cortex excitability in healthy subjects. Neuroimage. 2005;27(2):314–22. doi: 10.1016/j.neuroimage.2005.05.009. [DOI] [PubMed] [Google Scholar]
- 16.Chollet F, et al. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): a randomised placebo-controlled trial. Lancet Neurol. 2011;10(2):123–30. doi: 10.1016/S1474-4422(10)70314-8. [DOI] [PubMed] [Google Scholar]
- 17.Floel A, Cohen LG. Recovery of function in humans: cortical stimulation and pharmacological treatments after stroke. Neurobiol Dis. 2010;37(2):243–51. doi: 10.1016/j.nbd.2009.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nudo RJ, Plautz EJ, Frost SB. Role of adaptive plasticity in recovery of function after damage to motor cortex. Muscle Nerve. 2001;24(8):1000–19. doi: 10.1002/mus.1104. [DOI] [PubMed] [Google Scholar]
- 19.Nudo RJ. Adaptive plasticity in motor cortex: implications for rehabilitation after brain injury. J Rehabil Med. 2003;(41 Suppl):7–10. doi: 10.1080/16501960310010070. [DOI] [PubMed] [Google Scholar]
- 20.Teasell R, Bayona NA, Bitensky J. Plasticity and reorganization of the brain post stroke. Top Stroke Rehabil. 2005;12(3):11–26. doi: 10.1310/6AUM-ETYW-Q8XV-8XAC. [DOI] [PubMed] [Google Scholar]
- 21.Green JB. Brain reorganization after stroke. Top Stroke Rehabil. 2003;10(3):1–20. doi: 10.1310/H65X-23HW-QL1G-KTNQ. [DOI] [PubMed] [Google Scholar]
- 22.Nudo RJ. Functional and structural plasticity in motor cortex: implications for stroke recovery. Phys Med Rehabil Clin N Am. 2003;14(1 Suppl):S57–76. doi: 10.1016/s1047-9651(02)00054-2. [DOI] [PubMed] [Google Scholar]
- 23.Redecker C, et al. Differential downregulation of GABAA receptor subunits in widespread brain regions in the freeze-lesion model of focal cortical malformations. J Neurosci. 2000;20(13):5045–53. doi: 10.1523/JNEUROSCI.20-13-05045.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hess G. Synaptic plasticity of local connections in rat motor cortex. Acta Neurobiol Exp (Wars) 2004;64(2):271–6. doi: 10.55782/ane-2004-1511. [DOI] [PubMed] [Google Scholar]
- 25.Hess G, Donoghue JP. Long-term potentiation of horizontal connections provides a mechanism to reorganize cortical motor maps. J Neurophysiol. 1994;71(6):2543–7. doi: 10.1152/jn.1994.71.6.2543. [DOI] [PubMed] [Google Scholar]
- 26.Takeuchi N, Izumi S. Noninvasive brain stimulation for motor recovery after stroke: mechanisms and future views. Stroke Res Treat. 2012;2012:584727. doi: 10.1155/2012/584727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Di Pino G, et al. Modulation of brain plasticity in stroke: a novel model for neurorehabilitation. Nat Rev Neurol. 2014;10(10):597–608. doi: 10.1038/nrneurol.2014.162. [DOI] [PubMed] [Google Scholar]
- 28.Hummel FC, Cohen LG. Non-invasive brain stimulation: a new strategy to improve neurorehabilitation after stroke? Lancet Neurol. 2006;5(8):708–12. doi: 10.1016/S1474-4422(06)70525-7. [DOI] [PubMed] [Google Scholar]
- 29.Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 2000;527 Pt 3:633–9. doi: 10.1111/j.1469-7793.2000.t01-1-00633.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hummel FC, et al. Facilitating skilled right hand motor function in older subjects by anodal polarization over the left primary motor cortex. Neurobiol Aging. 2010;31(12):2160–8. doi: 10.1016/j.neurobiolaging.2008.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nitsche MA, et al. Facilitation of implicit motor learning by weak transcranial direct current stimulation of the primary motor cortex in the human. J Cogn Neurosci. 2003;15(4):619–26. doi: 10.1162/089892903321662994. [DOI] [PubMed] [Google Scholar]
- 32.Reis J, et al. Noninvasive cortical stimulation enhances motor skill acquisition over multiple days through an effect on consolidation. Proc Natl Acad Sci U S A. 2009;106(5):1590–5. doi: 10.1073/pnas.0805413106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hummel F, Cohen LG. Improvement of motor function with noninvasive cortical stimulation in a patient with chronic stroke. Neurorehabil Neural Repair. 2005;19(1):14–9. doi: 10.1177/1545968304272698. [DOI] [PubMed] [Google Scholar]
- 34.Boggio PS, et al. Repeated sessions of noninvasive brain DC stimulation is associated with motor function improvement in stroke patients. Restor Neurol Neurosci. 2007;25(2):123–9. [PubMed] [Google Scholar]
- 35.Stagg CJ, et al. Cortical activation changes underlying stimulation-induced behavioural gains in chronic stroke. Brain. 2012;135(Pt 1):276–84. doi: 10.1093/brain/awr313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fregni F, et al. Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. Neuroreport. 2005;16(14):1551–5. doi: 10.1097/01.wnr.0000177010.44602.5e. [DOI] [PubMed] [Google Scholar]
- 37.Zimerman M, et al. Modulation of training by single-session transcranial direct current stimulation to the intact motor cortex enhances motor skill acquisition of the paretic hand. Stroke. 2012;43(8):2185–91. doi: 10.1161/STROKEAHA.111.645382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Romero JR, et al. Subthreshold low frequency repetitive transcranial magnetic stimulation selectively decreases facilitation in the motor cortex. Clin Neurophysiol. 2002;113(1):101–7. doi: 10.1016/s1388-2457(01)00693-9. [DOI] [PubMed] [Google Scholar]
- 39.Fitzgerald PB, et al. Intensity-dependent effects of 1 Hz rTMS on human corticospinal excitability. Clin Neurophysiol. 2002;113(7):1136–41. doi: 10.1016/s1388-2457(02)00145-1. [DOI] [PubMed] [Google Scholar]
- 40.Khedr EM, et al. Modulation of motor cortical excitability following rapid-rate transcranial magnetic stimulation. Clin Neurophysiol. 2007;118(1):140–5. doi: 10.1016/j.clinph.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 41.Kim YH, et al. Facilitative effect of high frequency subthreshold repetitive transcranial magnetic stimulation on complex sequential motor learning in humans. Neurosci Lett. 2004;367(2):181–5. doi: 10.1016/j.neulet.2004.05.113. [DOI] [PubMed] [Google Scholar]
- 42.Di Lorenzo C, et al. Influence of rTMS over the left primary motor cortex on initiation and performance of a simple movement executed with the contralateral arm in healthy volunteers. Exp Brain Res. 2013;224(3):383–92. doi: 10.1007/s00221-012-3318-y. [DOI] [PubMed] [Google Scholar]
- 43.Kim YH, et al. Repetitive transcranial magnetic stimulation-induced corticomotor excitability and associated motor skill acquisition in chronic stroke. Stroke. 2006;37(6):1471–6. doi: 10.1161/01.STR.0000221233.55497.51. [DOI] [PubMed] [Google Scholar]
- 44.Yozbatiran N, et al. Safety and behavioral effects of high-frequency repetitive transcranial magnetic stimulation in stroke. Stroke. 2009;40(1):309–12. doi: 10.1161/STROKEAHA.108.522144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Takeuchi N, et al. Repetitive transcranial magnetic stimulation of contralesional primary motor cortex improves hand function after stroke. Stroke. 2005;36(12):2681–6. doi: 10.1161/01.STR.0000189658.51972.34. [DOI] [PubMed] [Google Scholar]
- 46.Mansur CG, et al. A sham stimulation-controlled trial of rTMS of the unaffected hemisphere in stroke patients. Neurology. 2005;64(10):1802–4. doi: 10.1212/01.WNL.0000161839.38079.92. [DOI] [PubMed] [Google Scholar]
- 47.Fregni F, et al. A sham-controlled trial of a 5-day course of repetitive transcranial magnetic stimulation of the unaffected hemisphere in stroke patients. Stroke. 2006;37(8):2115–22. doi: 10.1161/01.STR.0000231390.58967.6b. [DOI] [PubMed] [Google Scholar]
- 48.Ilic TV, Korchounov A, Ziemann U. Methylphenidate facilitates and disinhibits the motor cortex in intact humans. Neuroreport. 2003;14(5):773–6. doi: 10.1097/00001756-200304150-00023. [DOI] [PubMed] [Google Scholar]
- 49.Long D, Young J. Dexamphetamine treatment in stroke. QJM. 2003;96(9):673–85. doi: 10.1093/qjmed/hcg113. [DOI] [PubMed] [Google Scholar]
- 50.Kuo MF, Paulus W, Nitsche MA. Boosting focally-induced brain plasticity by dopamine. Cereb Cortex. 2008;18(3):648–51. doi: 10.1093/cercor/bhm098. [DOI] [PubMed] [Google Scholar]
- 51.Korchounov A, Ziemann U. Neuromodulatory neurotransmitters influence LTP-like plasticity in human cortex: a pharmaco-TMS study. Neuropsychopharmacology. 2011;36(9):1894–902. doi: 10.1038/npp.2011.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Meintzschel F, Ziemann U. Modification of practice-dependent plasticity in human motor cortex by neuromodulators. Cereb Cortex. 2006;16(8):1106–15. doi: 10.1093/cercor/bhj052. [DOI] [PubMed] [Google Scholar]
- 53.Sonde L, Lokk J. Effects of amphetamine and/or L-dopa and physiotherapy after stroke - a blinded randomized study. Acta Neurol Scand. 2007;115(1):55–9. doi: 10.1111/j.1600-0404.2006.00728.x. [DOI] [PubMed] [Google Scholar]
- 54.Goldstein LB. Influence of common drugs and related factors on stroke outcome. Curr Opin Neurol. 1997;10(1):52–7. doi: 10.1097/00019052-199702000-00011. [DOI] [PubMed] [Google Scholar]
- 55.Ilic TV, Korchounov A, Ziemann U. Complex modulation of human motor cortex excitability by the specific serotonin re-uptake inhibitor sertraline. Neurosci Lett. 2002;319(2):116–20. doi: 10.1016/s0304-3940(01)02563-0. [DOI] [PubMed] [Google Scholar]
- 56.Ziemann U. Pharmaco-transcranial magnetic stimulation studies of motor excitability. Handb Clin Neurol. 2013;116:387–97. doi: 10.1016/B978-0-444-53497-2.00032-2. [DOI] [PubMed] [Google Scholar]
- 57.Rossini D, et al. Does rTMS hasten the response to escitalopram, sertraline, or venlafaxine in patients with major depressive disorder? A double-blind, randomized, sham-controlled trial. J Clin Psychiatry. 2005;66(12):1569–75. doi: 10.4088/jcp.v66n1212. [DOI] [PubMed] [Google Scholar]
- 58.Rumi DO, et al. Transcranial magnetic stimulation accelerates the antidepressant effect of amitriptyline in severe depression: a double-blind placebo-controlled study. Biol Psychiatry. 2005;57(2):162–6. doi: 10.1016/j.biopsych.2004.10.029. [DOI] [PubMed] [Google Scholar]
- 59.Herwig U, et al. Antidepressant effects of augmentative transcranial magnetic stimulation: randomised multicentre trial. Br J Psychiatry. 2007;191:441–8. doi: 10.1192/bjp.bp.106.034371. [DOI] [PubMed] [Google Scholar]
- 60.Brunoni AR, et al. The sertraline vs. electrical current therapy for treating depression clinical study: results from a factorial, randomized, controlled trial. JAMA Psychiatry. 2013;70(4):383–91. doi: 10.1001/2013.jamapsychiatry.32. [DOI] [PubMed] [Google Scholar]
- 61.Michael D, et al. Repeated pulses of serotonin required for long-term facilitation activate mitogen-activated protein kinase in sensory neurons of Aplysia. Proc Natl Acad Sci U S A. 1998;95(4):1864–9. doi: 10.1073/pnas.95.4.1864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang QM, et al. Combination of transcranial direct current stimulation and methylphenidate in subacute stroke. Neurosci Lett. 2014;569:6–11. doi: 10.1016/j.neulet.2014.03.011. [DOI] [PubMed] [Google Scholar]
- 63.Kakuda W, et al. Combination treatment of low-frequency rTMS and occupational therapy with levodopa administration: an intensive neurorehabilitative approach for upper limb hemiparesis after stroke. Int J Neurosci. 2011;121(7):373–8. doi: 10.3109/00207454.2011.560314. [DOI] [PubMed] [Google Scholar]


