Abstract
Upon acquirement of pulmonary circulation, the ancestral heart may have been remodelled coincidently with, or accompanied by, the production and rearrangement of progenitor cells. However, the progenitor populations that give rise to the left ventricle (LV) and sinus venosus (SV) are still ambiguous. Here we show that the expression of Secreted frizzled-related protein Sfrp5 in the mouse identifies common progenitors for the outflow tract (OFT), LV, atrium and SV but not the right ventricle (RV). Sfrp5 expression begins at the lateral sides of the cardiac crescent, excluding early differentiating regions, and continues in the venous pole, which gives rise to the SV. Lineage-tracing analysis revealed that descendants of Sfrp5-expressing cells at E7.5 contribute not only to the SV but also to the LV, atria and OFT and are found also in the dorsal splanchnic mesoderm accompanied by the expression of the secondary heart field marker, Islet1. These findings provide insight into the arrangement of cardiac progenitors for systemic circulation.
It is unclear which progenitors define different regions of the heart. Here, the authors find Secreted frizzled-related protein 5 is expressed in murine progenitor cells for the outflow tract, first heart field, and sinus venosus, but not the right ventricle, and Wnt inhibition prevents progenitor proliferation.
Mammalian cardiac development begins with the cardiac crescent, which is composed of a bilateral cardiogenic splanchnic mesoderm, and the progressive differentiation of progenitors into cardiomyocytes to form the primary heart tube. During the successive development of the four-chambered heart, two independent pulmonary and systemic circulatory systems are established. In this process, the production and rearrangement of progenitor cells may account for remodelling of the ancestral heart into these two separate systems. Recent findings pertaining to another cardiac lineage called the secondary heart field (SHF) modified the classical view of cardiac development that the myocardium was derived from a single source. The SHF emerges in the splanchnic mesoderm to supply cells at both the arterial and venous poles of the heart tube, including the right ventricle (RV), most of the atria and outflow tract (OFT)1,2,3. However, the progenitor population that forms the first heart field (FHF), which gives rise to the left ventricle (LV) and the rest of the atria, and the sinus venosus (SV) are still ambiguous.
SHF progenitor cells, which transiently express Islet1 (Isl1), have been shown to express cardiac markers, including Fibroblast growth factor 10 (Fgf10), Myocyte enhancer factor 2c (Mef2c), T-box transcription factor 1 (Tbx-1), as well as other markers, and contribute to the RV, the OFT and most of the atria1,2,4,5,6,7,8,9. FHF progenitor cells have yet to be defined because the FHF lineage, which subsequently gives rise to the LV and a subset of the atria, is recognized only after the expression of differentiation markers such as Myosin light chain 2a (Mlc2a) and NK-2 transcription factor related, locus 5 (Nkx2-5) in the cardiac crescent3,10,11.
The SV is a cavity formed by a thin cardiac muscular wall, which drains blood from systemic veins and the coronary sinus into the right atrium, and gives rise to the sinoatrial node at the joint to the superior cardinal vein during embryogenesis. The origin of the SV has been previously addressed and mapped to the lateral rim of the heart fields in chickens by dye injection experiments12. Recently, the cell population in the lateral rim of the heart fields, which was defined by the expression of mutG5/hsp68/LacZ in mice, was found to contribute to the SV13. The venous pole was reported to be the precursor of the SV, which is marked by the expression of Tbx18 from E8.25 (refs 13, 14). Lineage-tracing experiments with Tbx18-Cre mice revealed that Tbx18-expressing cells form the SV and proepicardium15,16. Thus the origin of the SV has been suggested to be in the caudolateral regions of the cardiac crescent, although its relation to the FHF or SHF remains unclear.
Wnt signalling is one of the most important regulatory pathways in vertebrate cardiogenesis17,18,19,20,21. Wnt antagonism by Dkk1 or Crescent induces heart formation in vertebrates22,23,24, and Dkk1 is required for head induction in mammals25. Secreted frizzled-related protein (Sfrp) 1 and Sfrp2, which are also Wnt inhibitors, are expressed in the heart and are involved in cardiac tissue repair26,27, while the expression pattern and function of another subfamily member, Sfrp5, during cardiac development are unknown.
In this report, we show that Sfrp5 expression is detected in lateral and caudal regions of the cardiac crescent, is expressed in cells distinct from those positive for markers of differentiated cardiomyocytes and the SHF and continues to be expressed in the developing SV. However, descendants of Sfrp5-expressing cells notably contribute to cardiomyocytes in all chambers of the heart, except for the RV. These observations suggest that Sfrp5-expressing cells in the lateral region of the cardiac crescent are progenitor cells for the OFT, the LV, the left and right atria and the SV. Hyperplasia in the inflow tract (IFT) was observed in Sfrp1/2/5 triple knockout (TKO) mouse embryos, suggesting that Sfrp5 regulates the proliferation of cardiac progenitor cells in the venous pole.
Results
Sfrp5 is expressed in progenitor cells of the SV and epicardium
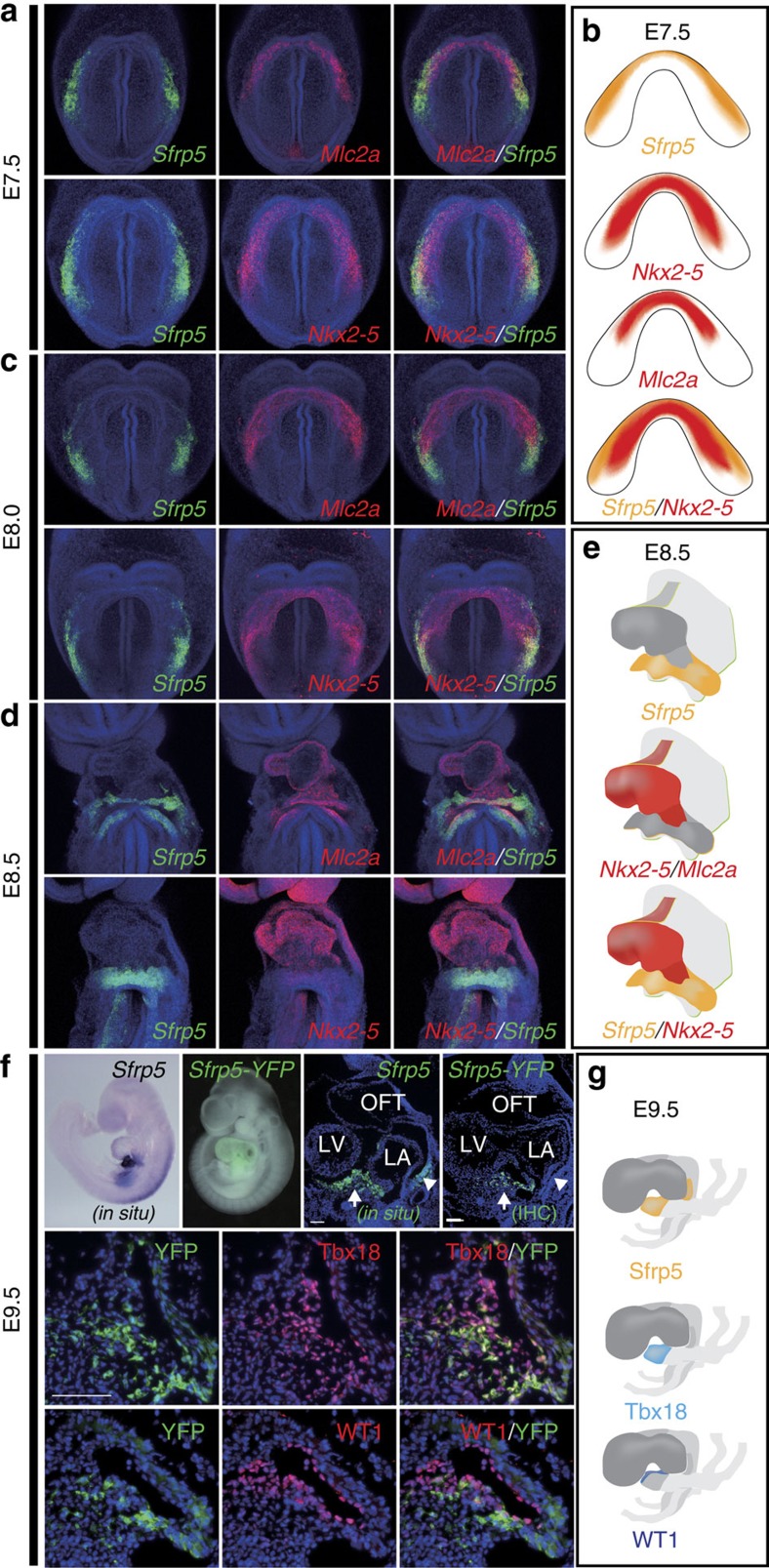
Wnt signalling is required for myocardial induction and is implicated in several steps of heart development17,18,19,20,21. The Sfrp subfamily genes (Sfrp1, 2 and 5) are known to encode secreted decoy receptors for Wnt signalling28 and to participate in the maintenance of adult cardiac function26,29. Among the three Sfrp genes, we found that Sfrp5 was specifically expressed in a particular part of the cardiac crescent at E7.5. Sfrp5 was detected in regions lateral and caudal to the region of Mlc2a and Nkx2-5 expression, which marks differentiating cardiomyocytes (Fig. 1a,b). The expression area of Sfrp5 was clearly distinct from those of Mlc2a and Nkx2-5 (Supplementary Fig. 1). During the heart tube looping stage (E8.0–E8.5), the expression of Sfrp5 was observed continuously in the venous pole of the heart (Fig. 1c–e), the pattern of which was distinct from Mlc2a and Nkx2-5, indicating that the Sfrp5-expressing area excludes differentiating cardiomyocytes. At E9.5, Sfrp5 expression was confined to the ventral venous pole, which is known to contain cardiac progenitors for the SV as well as the epicardium, suggesting that Sfrp5 might be expressed in such progenitors. To investigate this possibility, we performed double fluorescent immunohistochemistry using Tbx18 and WT1, specific markers for the SV and epicardium, respectively. To monitor the Sfrp5 expression, we used Sfrp5-venusYFP embryos. At E9.5, yellow fluorescent protein (YFP) was detected strongly in the ventral side and weakly in the dorsal side of the venous pole, similar to the pattern of Sfrp5 expression (Fig. 1f). The YFP signal highly overlapped with Tbx18 staining and moderately with WT1 staining (Fig. 1f,g), suggesting that Sfrp5 is expressed in progenitor cells of the SV and the epicardium.
Figure 1. Sfrp5 is expressed in the lateral cardiac crescent and subsequently in the SV.
(a,c,d) Double fluorescent whole-mount ISH of mouse embryos at E7.5 (a), E8.0 (c) and E8.5 (d) for detection of Sfrp5 (green) using Mlc2a or Nkx2-5 (red) as indicated in each panel. (b,e,g) Schematic drawings of gene expression (b,e) and protein distribution (g). (f) Whole-mount ISH (in situ), YFP image merged with bright field image (Sfrp5-YFP), section in situ with probe for Sfrp5 and single or double immunohistochemistry (IHC) of Sfrp5-venusYFP KI embryos using anti-GFP antibodies with anti-Tbx18 or anti-WT1 antibodies at E9.5, as indicated in each panel. Scale bars=Scale bars, 50 μm.
Cardiomyocytes in the SV show a unique gene expression profile, that is, TroponinT (TnT)- and Hcn4-positive but Nkx2-5-negative. This differs from the Nkx2-5- and TnT-positive chamber myocardium30. To further examine the expression profile of Sfrp5-venusYFP-expressing cells, we conducted marker expression analysis. At E9.5, Sfrp5-venusYFP-expressing cells were negative for Nkx2-5, TnT and Hcn4 (Supplementary Fig. 2a), whereas, at E10.5, these cells expressed TnT and Hcn4 but not Nkx2-5 (Supplementary Fig. 2b). These results indicate that Sfrp5-expressing cells in the ventral venous pole at E9.5 represent progenitors for the SV and that they differentiate into cardiomyocytes in the SV at E10.5. Thus the Sfrp5-expressing area at E7.5 may be the precursor of the SV.
Sfrp5 is expressed in progenitors of cardiac components except for RV
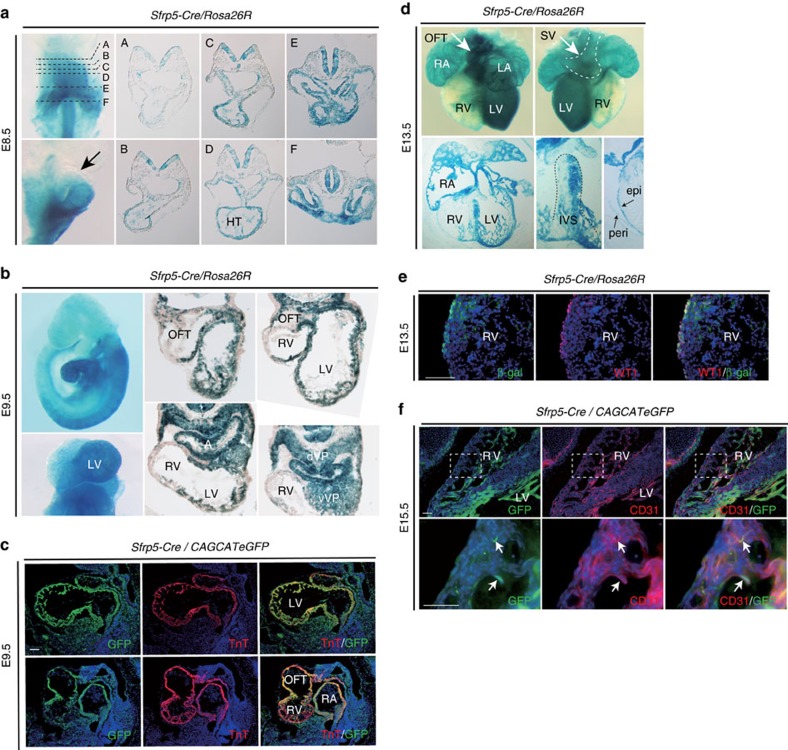
We next determined whether progenitors of the SV correspond to Sfrp5-expressing cells in the cardiac crescent. To test this possibility, we generated a Sfrp5-Cre line (Supplementary Fig. 3) and investigated the lineages of Sfrp5-expressing cells by crossing this line with Rosa26R or CAG-floxed-CAT-eGFP reporter mice31. At E8.5, β-galactosidase (LacZ) staining was unexpectedly observed in the entire heart tube, except for the most rostral portion (Fig. 2a). At E9.5, LacZ staining was observed in the OFT, LV, atria and venous pole but not in the RV (Fig. 2b). Labelled cells overlapped with TnT in the OFT, LV and atria, suggesting that descendants of Sfrp5-expressing cells differentiated into chamber myocardium in addition to the SV (Fig. 2c). At E13.5, LacZ-stained cells mostly occupied not only the LV but were also found in the WT1-expressing pericardium (Fig. 2d,e) and the cluster of differentiation 31 (CD31)- or platelet endothelial cell adhesion molecule-expressing endocardium (Fig. 2f). Labelled cells were found in part of the interventricular septum and rarely in the RV (Fig. 2b,d) and those that were found in the RV tended to localize in the trabecular myocardium (Fig. 2f). These observations suggest that Sfrp5 is expressed in progenitors of the SV, pericardium, epicardium and endocardium, as well as in all chamber myocardium except for the RV. Given that Isl1 lineages contribute to all myocardium except for the LV32,33, the fate of Isl1 lineages to the RV or Sfrp5 lineages to the LV may be exclusively determined during the earliest stage of cardiogenesis.
Figure 2. Descendants of Sfrp5-expressing cells contribute to the SV and all chamber myocardium except for the RV.
(a,b,d) Whole-mount and section LacZ staining of Sfrp5-Cre/Rosa26R embryos at E8.5 (a), E9.5 (b) and E13.5 (d). HT: heart tube; dVP: dorsal venous pole; vVP: ventral venous pole; epi: epicardium; peri: pericardium, IVS; interventricular septum. (c,e,f) Double immunohistochemistry of Sfrp5-Cre/CAG-floxed-CAT-eGFP (c,f) or Sfrp5-Cre/Rosa26R (e) embryos at E 9.5 (c), E 13.5 (e) and E15.5 (f), using anti-GFP or anti-β-gal (green) antibodies with anti-TnT, anti-Isl1, anti-WT1 or anti-CD31 antibodies (red). Rectangles in upper panels (f) are magnified in lower panels. Arrows indicate cells co-expressing YFP and CD31 (f). Scale bars=50 μm.
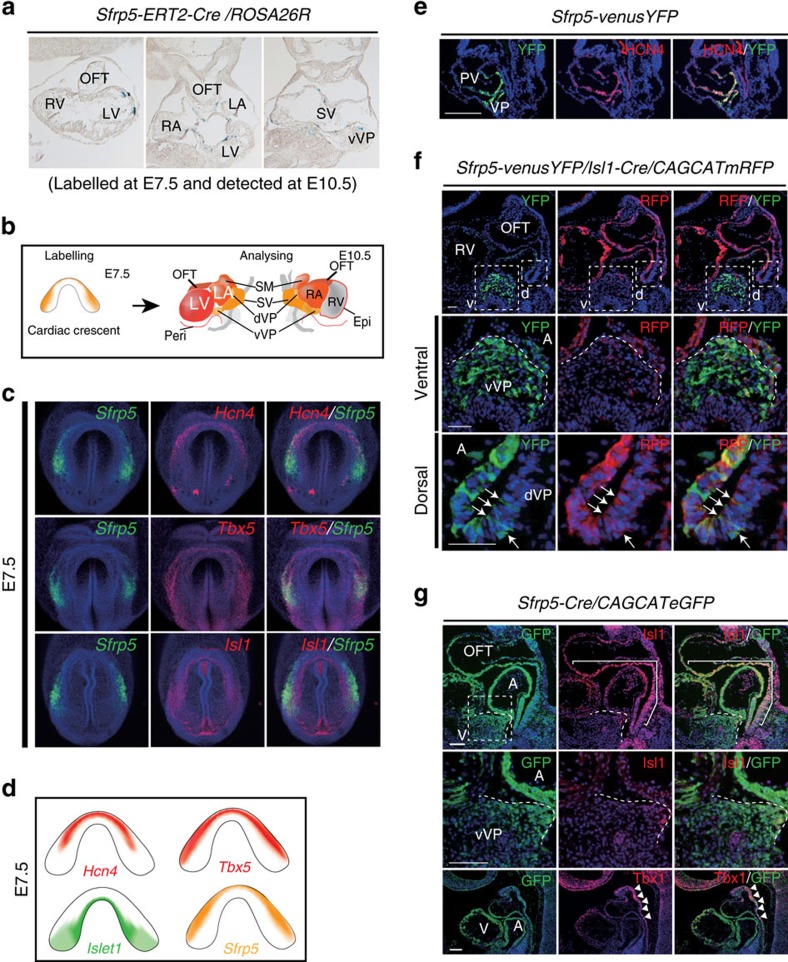
Since Sfrp5 expression is confined to the venous pole of the heart at E8.5, which does not include chamber myocardium, we speculated that cells expressing Sfrp5 prior to E8.5 might include progenitors of the FHF. To evaluate the fate of Sfrp5-expressing cells at specific developmental stages, we generated a tamoxifen-inducible (Estrogen Receptor T2 (ERT2)) Cre mouse line (Supplementary Fig. 3). After crossing Sfrp5-ERT2Cre mice with reporter mice, Sfrp5-expressing cells were labelled at E7.5 via tamoxifen injection and analysed at E10.5. LacZ staining revealed that cells labelled at E7.5 were found in the OFT, LV, atria, SV and venous pole but not in the RV (Fig. 3a,b). Interestingly, labelled cells were still found in the atria at E8.5 and E9.5 (Supplementary Fig. 4), indicating that Sfrp5-expressing cells at the venous pole may supply cardiomyocytes for the IFT. These data suggest that Sfrp5-expressing cells at E7.5 include common progenitors for the SV, OFT and all chamber myocardium except for the RV. There is therefore a strong possibility that Sfrp5 expression marks progenitors of the FHF.
Figure 3. Sfrp5-expressing cells at E7.5 contribute to the SV and all chamber myocardium except for the RV.
(a) Representative photographs of LacZ-stained sections from Sfrp5-ERT2Cre/Rosa-lacZ embryos at E10.5, labelled by tamoxifen at E7.5. (b) Schematic drawing for the expression of Sfrp5 (orange) and the cardiac contribution of its lineages (red) when cells were labelled at E7.5 and analysed at E10.5. (c) Double fluorescent whole-mount ISH of mouse embryos at E7.5 for detection of Sfrp5 (green) and Hcn4, Tbx5 or Isl1 (red), as indicated in each panel. (d) Schematic drawings of gene expression of Sfrp5, Isl1, Hcn4 and Tbx5. (e) Double immunohistochemistry of Sfrp5-venusYFP at E8.5 using anti-GFP (green) and anti-HCN4 (red) antibodies. Co-expression with HCN4 was found in the VP. Weak expression of YFP also co-distributed with HCN4-positive cells in the IFT and part of the primitive ventricle (PV). (f,g) Double immunohistochemistry of Sfrp5-venusYFP (e), Sfrp5-venusYFP/Isl1-Cre/CAG-(floxed)-CAT-mRFP (f) or Sfrp5-Cre/CAG-(floxed)-CAT-eGFP (g) embryos at E9.5 using anti-GFP (green) antibodies and anti-HCN4, anti-RFP, anti-Isl1 or anti-Tbx1 (red) antibodies. Rectangles in upper panels are magnified in lower panels (v: ventral; d: dorsal). Co-expression with Isl1 was detected (arrows in panel (f) and parenthesis in panel (g)). Boundaries for the VP from the atrium are indicated by white dotted lines (f,g). Signal for Tbx1, but not GFP, was observed in the anterior splanchnic mesoderm and anterior OFT (arrowheads in panel (g)). A, atrium.
Sfrp5 derivatives contribute to the FHF and part of the SHF lineages
To test this possibility, the expression of Sfrp5 was compared with that of the FHF marker genes, Hcn4 and Tbx5. Hcn4 is expressed in the prospective LV at E7.5, with expression shifting towards the SV30,34. Tbx5 is expressed in the FHF lineage at E6.5–E7.5 (refs 35, 36, 37). The expression of Hcn4 was observed in the anteromedial region compared with Sfrp5, while region marked by Tbx5 overlapped with and extended anteriorly from that marked by Sfrp5 (Fig. 3c,d). Since the expression of Hcn4 was restricted to the anterior portion of the Tbx5 expression area, we concluded that Hcn4 might mark only the early differentiated FHF lineage. Conversely, Tbx5 marks both undifferentiated and differentiating FHF progenitors, among which Sfrp5 expression is restricted to undifferentiated FHF progenitors existing in the lateral sides of the cardiac crescent. Moreover, cells weakly expressing YFP in the primitive ventricle and IFT were co-distributed with HCN4 in the Sfrp5-venusYFP mouse embryo at E8.5 (Fig. 3e), suggesting that descendants of Sfrp5-expressing cells contribute to the FHF lineage.
Lineage analysis also revealed that Sfrp5-expressing cells at E7.5 contributed to the OFT and atria, indicating that these cells also include progenitors of the SHF. However, at E7.5, the expression pattern of Sfrp5 was distinct from that of Isl1, an SHF marker, although some overlap was found in the expression in the caudal region (Fig. 3c,d). At E9.5, Isl1 transcripts and protein were expressed in the splanchnic mesoderm, including the dorsal venous pole, but were rarely expressed in the ventral venous pole, which contains Sfrp5-expressing cells (Supplementary Figs 5 and 6). To clarify whether the expression of Sfrp5 precedes that of Isl1 in the splanchnic mesoderm, we conducted lineage-tracing analysis using Isl1-Cre and CAG-floxed-CAT-mRFP reporter/Sfrp5-venusYFP mice. We found that the YFP signal in the ventral venous pole rarely overlapped with derivatives of Isl1-expressing cells (Fig. 3f). In Sfrp5-Cre/CAG-floxed-CAT-eGFP-reporter mice, however, GFP-labelled cells expressed Isl1 and Fgf10 in the dorsal splanchnic mesoderm and part of the OFT (Fig. 3g and Supplementary Fig. 7). These findings suggest that derivatives of Sfrp5-expressing cells also contributed to part of the SHF lineage, which may give rise to the OFT and atria. Notably, these GFP-labelled cells were rarely found in the anterior splanchnic mesoderm, where Tbx1, which is known to also be expressed in the SHF38,39, was distributed (Fig. 3g).
Sfrp5-YFP-expressing ES cells differentiate into cardiac components
We next tested whether Sfrp5-venusYFP-expressing embryonic stem (ES) cells could differentiate into cardiac components, such as endothelial, smooth muscle and myocardial cells. On day 2, the peak of YFP expression was found to be ahead of those of other differentiation markers (Supplementary Fig. 8). On day 8, CD31-, α-smooth muscle actin- and TnT-positive cells were found in the cell mass (Supplementary Fig. 8), suggesting that Sfrp5 could also serve as a marker gene of precursor cells for cardiac components in an in vitro differentiation system.
Sfrps may not be involved in cardiac differentiation
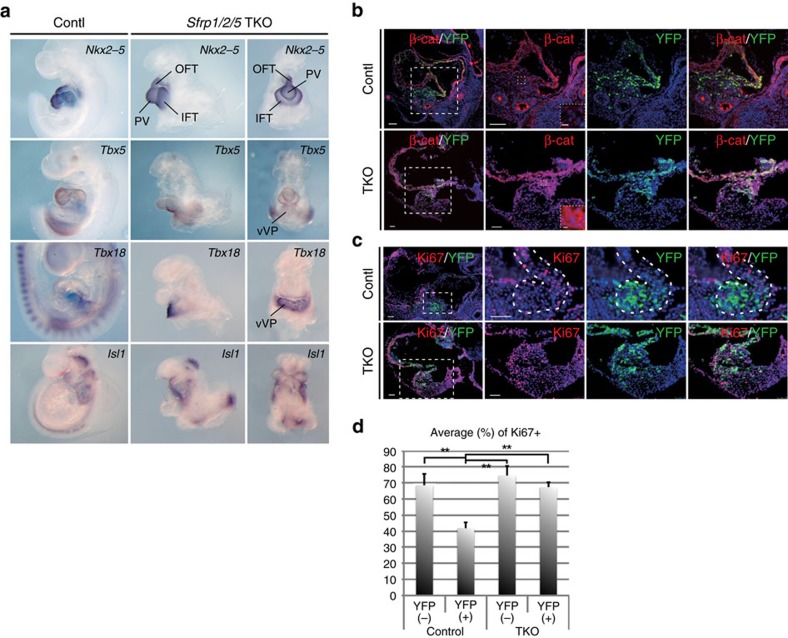
Our results imply that Sfrp5 is expressed and retained in cardiac progenitors but lost upon their differentiation, indicating that the loss of Sfrp5 might enhance the differentiation of these cardiac progenitors. To test this possibility, we generated Sfrp1/2/5 TKO mice. As reported previously, only TKO mice show severe growth retardation due to the compensatory functions of Sfrp1 and Sfrp2 (ref. 40). We focussed our analysis on the cardiac phenotype and analysed several lineage markers via in situ hybridization (ISH) at E9.5, the latest stage at which TKO embryos can be collected. Similar to controls, Nkx2-5 was present in the heart tube but not in the venous pole in TKO embryos (Fig. 4a). This indicates that the differentiation of cardiomyocytes occurred normally until this stage. The expression of Tbx5, Isl1 and Tbx18 was normal (Fig. 4a), suggesting that formation of the FHF, SHF and SV was not affected. These observations suggest that Sfrps may not affect differentiation.
Figure 4. Sfrp1/2/5 TKO embryos exhibit hyperplasia of the IFT accompanied by activated canonical Wnt signalling.
(a) ISH of control and TKO embryos with probes for Nkx2-5, Tbx5, Tbx18 and Isl1 genes. PV: primitive ventricle. (b,c) Double immunohistochemistry using anti-β-catenin (b) or anti-Ki67 (c) antibodies in control and TKO embryos. Rectangles from the left end panels are magnified in the right panels. Further magnified photos, located in the right bottom window, show that β-catenin accumulated in YFP-expressing cells in the ventral venous pole in TKO embryos. (d) Percentage of Ki67 positive (+) cells in Sfrp5-YFP-expressing (+) or negative (−) cells in control and TKO embryos (n=3). Data are presented as mean±s.d.; **P<0.01, as compared with control samples. Scale bars=50 μm (5 μm in dotted rectangles).
Sfrps prevent proliferation by inhibiting canonical Wnt signalling
TKO embryos appeared to have an enlarged IFT, as compared with the OFT. To determine whether Sfrps might regulate the proliferation of cardiac progenitors, we next assessed whether Wnt/β-catenin signalling and progenitor cell proliferation in the venous pole were affected in TKO embryos. In control embryos, β-catenin accumulated in the ventricles and atria, but less β-catenin was observed in YFP-positive cardiac progenitor cells in the ventral venous pole (Fig. 4b). In TKO embryos, however, β-catenin accumulated even in YFP-positive cardiac progenitor cells (Fig. 4b). In control mice, Ki67 was more highly expressed in YFP-negative cells than in YFP-positive cells, suggesting that proliferation may be inhibited in Sfrp5-expressing cardiac progenitors (Fig. 4c,d). In contrast, the percentage of Ki67-positive cells in TKO embryos increased in YFP-positive cells, as compared with the percentage in control samples (Fig. 4c,d). These results indicate that Sfrps prevent the proliferation of cardiac progenitors by inhibiting canonical Wnt signalling.
Discussion
Based on our findings, we propose a model for the contribution of Sfrp5-expressing cardiac precursors to heart development, except for the RV (Supplementary Fig. 9). We found that Sfrp5-expressing cells at E7.5 contributed not only to the OFT, LV and atria and also to the SV by fate mapping experiments using Sfrp5-Cre and -ERT2Cre mice. This observation gives rise to the possibility that the FHF lineage and the SV might share a common origin. This idea has been implied previously by the expression and lineage-tracing experiments of Hcn4, because Hcn4-expressing cells ultimately yield most of the LV, portions of the atria and the cardiac conduction system30,41,42,43,44. We found that the expression of Sfrp5 rarely overlapped with that of Hcn4, while cells weakly expressing YFP in the PV and IFT at E8.5 largely overlapped with Hcn4. The expression of Sfrp5 also largely overlapped with that of Tbx5 at E7.5, and early labelling of Tbx5-expressing cells at E6.5–E7.5 are reported to mark cells that are predominantly restricted to the LV and atria36,37, suggesting that Sfrp5-expressing cells include progenitors for the FHF. Moreover, Sfrp5 was continuously expressed in cells giving rise to the SV, suggesting that cells that transiently express Sfrp5 contribute to the FHF. Sfrp5-expressing cells start to express Tbx18 at around E8.0, and forced expression of Tbx18 reportedly promotes differentiation of cells into the SV, including the conduction system45, and represses a subset of myocardial genes46. Thus Sfrp5-expressing cells could maintain the potential of cells to differentiate into both the SV and FHF until Tbx18 expression initiates.
Alternatively, the early Sfrp5-expressing progenitor pool might already be heterogeneous in the commitment to each differentiation program. Since Tbx5-expressing cells at E6.5–E7.5 would not contribute to the OFT (personal communication from Dr Yashiro), a precise comparison of expression domains and lineages of Sfrp5 and Tbx5 may discriminate Sfrp5-expressing progenitors that contribute to the OFT from those that contribute to other regions in the cardiac crescent. However, the expression domain of Sfrp5 determined by ISH could be more restricted than it actually is at early stages, given the possibility of Cre recombination occurring in Sfrp5-expressing progenitors at levels undetectable by ISH. Thus the timing for cell fate determination and the potential for Sfrp5-expressing progenitor cells to differentiate should be addressed in future.
Another observation was that a portion of Sfrp5-expressing cells later expressed Isl1 and contributed to the OFT and atria (Fig. 5). This suggests that the Isl1-expressing SHF progenitor population has at least two distinct origins, one of which is derived from Sfrp5-expressing cells. We also found that Tbx1 is expressed in the anterior splanchnic mesoderm in which descendants of Sfrp5-expressing cells did not appear, indicating that Tbx1 could mark the subpopulation of Isl1-expressing cells in the SHF. Moreover, lineages of Sfrp5-expressing cells did not contribute to the RV. It was previously reported that mesodermal cells of the first branchial arch are involved in RV formation and that Tbx1 is expressed in the first branchial arch and descendants of Tbx1-expressing cells contribute to the RV38,39,47,48,49. These findings suggest a distinct origin of the RV relative to that of the other chambers. Given that septated ventricles have been evolutionally acquired to separate systemic and pulmonary circulation50, our results provide new insight into the arrangement of cardiac progenitors before the evolutionary emergence of the RV.
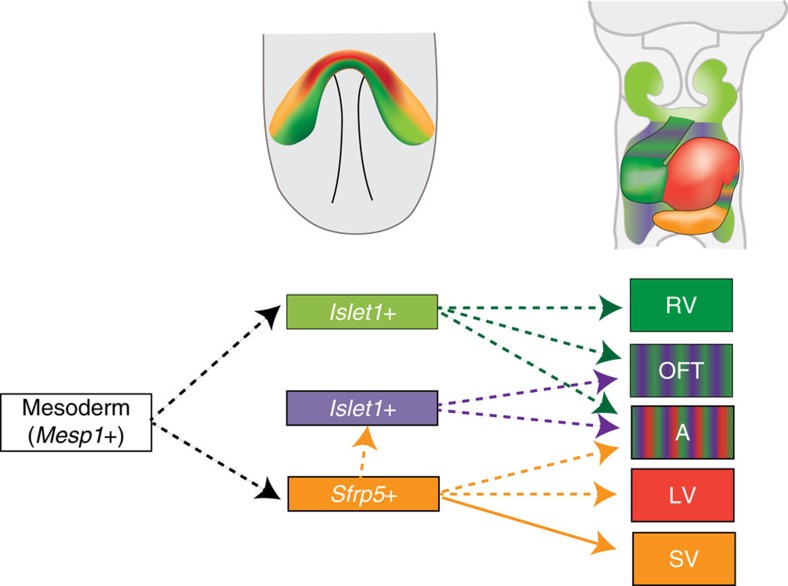
Figure 5. Model for the contribution of Sfrp5-expressing cardiac precursors to heart development.
Upper panels show the expression area of Sfrp5 (orange) and Islet1 (light green) and the distribution of descendants of Sfrp5-expressing (red) and Islet1-expressing (green) cells at E7.5 (right) and E9.5 (left). The lower panel shows the lineage contribution to each part of the heart. Mesodermal cells, which transiently express Mesp1, may become cardiac precursor cells expressing Islet1 (light green) or Sfrp5 (orange). A portion of Sfrp5-expressing cells later begins to express Islet1 (purple). Transient Islet1-expressing cells that do not express Sfrp5 contribute to the RV, OFT and atrium (green), and cells that express Sfrp5 contribute to the OFT and atrium (purple) but not to the RV. Cells that transiently express Sfrp5 contribute to the LV, atrium and OFT (orange), and cells that continuously express Sfrp5 form the SV.
The role of Wnt signalling in cardiac development has been extensively reported17. The hyperactivation of Wnt/β-catenin signalling in cardiac precursors leads to enhanced proliferation and inhibition of further differentiation51,52. In our study, increased proliferation was observed in TKO embryos, but differentiation into primitive cardiomyocytes was not impaired. We speculate that artificial activation and lack of inhibition of Wnt/β-catenin signalling may result in different outcomes during myocardial differentiation, and a primary role of Wnt/β-catenin signalling in this context may be the regulation of proliferation rather than differentiation. Since Sfrp1/2/5 has been reported to regulate planar cell polarity pathways through the non-canonical Wnt pathway40, tissue- or cell-specific functions of Sfrps may exist. Further understanding of the molecular basis of cardiac progenitor differentiation into various types of cardiomyocytes could provide new therapeutic insights for regenerative medicine.
Methods
Mice
To generate Sfrp5-Cre recombinase (Cre) and Sfrp5-modified ERT2 conjugated with Cre knock-in mouse lines, a strategy similar to that used to generate the Sfrp5-venusYFP mouse line was adopted40. Targeting vectors were constructed by using a cassette containing Cre- or ERT2Cre-recombinase genes with frt-PGK-neomycin (neo)-bpA-frt, which was ligated with KpnI-SmaI and SpeI-XbaI fragments for long and short homologous arms, respectively. A MC1-DTA-negative selection marker cassette was also added to the outside of the short homologous arm to enrich for homologous recombinants (Supplementary Fig. 3). For recombination of the targeting vector for ERT2Cre, the CRISPR-Cas9 system was used. A 5′-GGCCAGGAGTACGACTACTACGG-3′ sequence was inserted into the pX330 plasmid (Addgene) using the BbsI site and was then co-transfected with the targeting vector. Homologous recombination was confirmed by southern hybridization for the Sfrp5-Cre line, using XbaI-EcoRI fragments for the probe, and by PCR for the Sfrp5-ERT2Cre line, using the following primer set: Neo6: 5′-CGACCACCAAGCGAAACATC-3′ and SAcontRR9: 5′-CCCAGGGAAGGGAATTGTTCTAG-3′. Deletion of Neo by crossing with the ROSA-Flp mouse line was confirmed by PCR, using the following primer set: DeltaNeoF1: 5′-CCTACATGCGCCCACTAGCCGT-3′ and DeltaNeoR2: 5′-CCTAGCGACCCCAAGCATCC-3′. TKO embryos were obtained by crossing with Sfrp1/2/5; −/−, +/−, −/− or −/−, +/−, venus (v)/v40. Isl1-Cre mouse lines have been previously generated using gene-targeting techniques7. The ROSA26R mouse line was a gift from Dr Soriano31. These lines were maintained in a 129 and C57BL/6 mixed background. A CAG-floxed-CAT-lyn-mRFP mouse line was newly and CAG-floxed-CAT-eGFP mouse line was previously established using transgenic techniques using C3HeJ × C57BL/6J F1 hybrid mice53.
Tamoxifen injection
Tamoxifen (Sigma) was injected peritoneally into 8–20-week-old pregnant female mice at a dosage of 35 mg kg−1 at E7.5–E9.5. Embryos were dissected on E10.5 or E13.5 and subjected to lacZ staining.
Whole-mount double fluorescent ISH
Embryos were dissected between E7.5 and E9.5 and fixed with 4% paraformaldehyde in PBS for durations of 1 h to overnight. After dehydration using a methanol series, embryos were rehydrated with 0.1% Tween 20 in PBS (PBST), bleached with 6% H2O2 in PBS, treated with Protease K and then hybridized with digoxygenin (DIG)- and/or dinitrophenyl-labelled probes at 68 °C overnight. They were then washed with 5 × and 2 × SSC/50% formamide, PBST and blocking reagent for 1 h. For detection of DIG-labelled probes, embryos were incubated with anti-DIG antibodies and detected using a TSA-Alexa555 detection system according to the manufacturer's instructions. For detection of dinitrophenyl, TSA-Alexa488 was used. Embryos were analysed by confocal laser microscopy (Leica LSM 5 PASCAL system).
Immunohistochemistry and LacZ-staining
Embryos were embedded with optimum cutting temperature compound after fixation and were sectioned at 10–14 μm increments. After washing with PBS and 1 h of preincubation with 1% BSA in PBS for blocking, sections were incubated with 500 × diluted primary antibodies (anti-GFP: Nakalai GF090R or Abcam ab290; anti-Tbx1: Santa Cruz sc-17874; anti-Tbx18: Santa Cruz SC17869; anti-Wt1: Abcam ab10670; anti-Nkx2-5: Abcam ab35842; anti-TnT: Thermo MS-295-P0; anti-Hcn4: Alomone Labs APC052; anti-RFP: MBL PM005; anti-β-catenin: Millipore 05-665; anti-Ki67: Abcam ab15580) at 4 °C overnight. The sections were then incubated with 500 × diluted secondary antibodies conjugated with Alexa Fluor 488 or 555 (Molecular Probes). Nuclei were counterstained with 4,6-diamidino-2-phenylindole. For LacZ-staining, frozen sections were incubated with X-gal solution at room temperature overnight after washing with PBS.
Cardiac induction of ES cells
ES cells were isolated from blastocysts of Sfrp5-venusYFP mice and maintained in Leukemia Inhibitory Factor-containing (1,000 U ml−1) ES culture medium (DMEM with 10% fetal bovine serum, 1 × Non-Essential Amino Acids Solution, 1 mM sodium pyruvate, and 10−4 M 2-mercaptoethanol). For cardiac induction, embryoid bodies, which were formed by the hanging-drop culture method for 48 h, were plated in 2-well chamber slides or 98-well plates (Nunc) and cultured for 8 days in ES culture medium without Leukemia Inhibitory Factor.
Real-time PCR
Total RNA was isolated using TRIzol (Invitrogen). Reverse transcription was performed with the ReverTra Ace qPCR RT Kit (TOYOBO). Real-time PCR was conducted using SYBR Premix Ex Taq II (Takara Bio Inc. or Kapa Biosystems Inc.). Intensities of PCR products were measured and analysed using Mini-Opticon (Bio-Rad). The following primers were used: YFP: forward 5′-CCTGGTCGAG CTGGACGGCGAC-3′ and reverse 5′-TCACGAACTCCAGCAGGACCATG-3′, Nkx2-5: forward 5′-GCTACAAGTGCAAGCGACAG-3′ and reverse 5′-GGGTAGGCGTTGTAGCCATA-3′, β-MHC: forward 5′-TCCACGGGGAAGAGCATCCAT-3′ and reverse 5′-CAGACTCTGGAGGCTCTTCACT, Hcn4: forward 5′-CTCAACTCAGGCGTCTTCAAC-3′ and reverse 5′-CCACCGAAGTAGTAGCAGCAG-3′, and G3pdh: forward 5′-ACCACAGTCCATGCCATCAC-3′ and reverse 5′-TCCACCACCCTGTTGCTGTA -3′. Amplification conditions were 5 s at 95 °C, 20 s at 60 °C and 15 s at 72 °C for 49 cycles. G3pdh was amplified as an internal control.
Statistical analysis
A parametric multiple comparison test, one-way analysis of variance with Tukey–Kramer post hoc analysis, was mainly used (unless otherwise indicated) when Bartlett's test (P<0.05) showed homogeneity of variances with the Ekuseru–Toukei 2012 software (Social Survey Research Information Co., Ltd.). All data are expressed as mean±s.d. P<0.05 was considered statistically significant.
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and its Supplementary Information files or available on reasonable request from the corresponding authors.
Additional information
How to cite this article: Fujii, M. et al. Sfrp5 identifies murine cardiac progenitors for all myocardial structures except for the right ventricle. Nat. Commun. 8, 14664 doi: 10.1038/ncomms14664 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Supplementary Figures, Supplementary reference
Acknowledgments
We thank Mr Hidemichi Miyahara and Mr Makoto Kiso for their technical assistance, Dr Rieko Asai, Dr Hiroki Kurihara and Dr Hiroyuki Yamagishi for sharing probes for in situ hybridization, Dr Kenta Yashiro for helpful discussions and suggestions and all members of Saga's and Yoshizumi's laboratories. We also thank the Natural Science Centre for Basic Research and Development (N-BARD) at Hiroshima University for allowing the use of their facility. This study was supported in part by a Grant-in-Aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (22590183 and 16K08443). This work was also supported by NIG-JOINT (2012-B, 2013-B and 2016-A1).
Footnotes
The authors declare no competing financial interests.
Author contributions M.F. and A.S. performed the experiments. R.K., M.N., U.K., S.M.E., M.Y., A.S., Y.S. and H.K supplied the experimental apparatus and animals. All authors contributed to the analyses of the experimental data. M.Y, A.S., Y.S. and H.K. designed the research. M.F., A.S., Y.S and H.K. wrote the manuscript.
References
- Moorman A. F., Christoffels V. M., Anderson R. H. & van den Hoff M. J. The heart-forming fields: one or multiple? Philos. Trans. R. Soc. Lond. B Biol. Sci. 362, 1257–1265 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckingham M., Meilhac S. & Zaffran S. Building the mammalian heart from two sources of myocardial cells. Nature Rev. Genet. 6, 826–835 (2005). [DOI] [PubMed] [Google Scholar]
- Abu-Issa R. & Kirby M. L. Heart field: from mesoderm to heart tube. Annu. Rev. Cell Dev. Biol. 23, 45–68 (2007). [DOI] [PubMed] [Google Scholar]
- Viragh S. & Challice C. E. Origin and differentiation of cardiac muscle cells in the mouse. J. Ultrastruct. Res. 42, 1–24 (1973). [DOI] [PubMed] [Google Scholar]
- Kelly R. G., Brown N. A. & Buckingham M. E. The arterial pole of the mouse heart forms from Fgf10-expressing cells in pharyngeal mesoderm. Dev. Cell 1, 435–440 (2001). [DOI] [PubMed] [Google Scholar]
- Mjaatvedt C. H. et al. The outflow tract of the heart is recruited from a novel heart-forming field. Dev. Biol 238, 97–109 (2001). [DOI] [PubMed] [Google Scholar]
- Cai C. L. et al. Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev. Cell 5, 877–889 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y. et al. Islet 1 is expressed in distinct cardiovascular lineages, including pacemaker and coronary vascular cells. Dev. Biol. 304, 286–296 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prall O. W. et al. An Nkx2-5/Bmp2/Smad1 negative feedback loop controls heart progenitor specification and proliferation. Cell 128, 947–959 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snarr B. S., Kern C. B. & Wessels A. Origin and fate of cardiac mesenchyme. Dev. Dynam. 237, 2804–2819 (2008). [DOI] [PubMed] [Google Scholar]
- Evans S. M., Yelon D., Conlon F. L. & Kirby M. L. Myocardial lineage development. Circ. Res. 107, 1428–1444 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stalsberg H. & DeHaan R. L. The precardiac areas and formation of the tubular heart in the chick embryo. Dev. Biol. 19, 128–159 (1969). [DOI] [PubMed] [Google Scholar]
- Mommersteeg M. T. et al. The sinus venosus progenitors separate and diversify from the first and second heart fields early in development. Cardiovasc. Res. 87, 92–101 (2010). [DOI] [PubMed] [Google Scholar]
- Christoffels V. M. et al. Formation of the venous pole of the heart from an Nkx2-5-negative precursor population requires Tbx18. Circ. Res. 98, 1555–1563 (2006). [DOI] [PubMed] [Google Scholar]
- Cai C. L. et al. A myocardial lineage derives from Tbx18 epicardial cells. Nature 454, 104–108 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christoffels V. M. et al. Tbx18 and the fate of epicardial progenitors. Nature 458, E8–E9 discussion E9–E10 (2009). [DOI] [PubMed] [Google Scholar]
- Brade T., Manner J. & Kuhl M. The role of Wnt signalling in cardiac development and tissue remodelling in the mature heart. Cardiovasc. Res. 72, 198–209 (2006). [DOI] [PubMed] [Google Scholar]
- Cohen E. D., Tian Y. & Morrisey E. E. Wnt signaling: an essential regulator of cardiovascular differentiation, morphogenesis and progenitor self-renewal. Development 135, 789–798 (2008). [DOI] [PubMed] [Google Scholar]
- Gessert S. & Kuhl M. The multiple phases and faces of Wnt signaling during cardiac differentiation and development. Circ. Res. 107, 186–199 (2010). [DOI] [PubMed] [Google Scholar]
- Martin L. K. et al. Canonical WNT signaling enhances stem cell expression in the developing heart without a corresponding inhibition of cardiogenic differentiation. Stem Cells Dev. 20, 1973–1983 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueno S. et al. Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proc. Natl Acad. Sci. USA 104, 9685–9690 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marvin M. J., Di Rocco G., Gardiner A., Bush S. M. & Lassar A. B. Inhibition of Wnt activity induces heart formation from posterior mesoderm. Genes Dev. 15, 316–327 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider V. A. & Mercola M. Wnt antagonism initiates cardiogenesis in Xenopus laevis. Genes Dev. 15, 304–315 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzahor E. & Lassar A. B. Wnt signals from the neural tube block ectopic cardiogenesis. Genes Dev. 15, 255–260 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukhopadhyay M. et al. Dickkopf1 is required for embryonic head induction and limb morphogenesis in the mouse. Dev. Cell 1, 423–434 (2001). [DOI] [PubMed] [Google Scholar]
- Kobayashi K. et al. Secreted Frizzled-related protein 2 is a procollagen C proteinase enhancer with a role in fibrosis associated with myocardial infarction. Nature Cell Biol. 11, 46–55 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He W. et al. Exogenously administered secreted frizzled related protein 2 (Sfrp2) reduces fibrosis and improves cardiac function in a rat model of myocardial infarction. Proc. Natl Acad. Sci. USA 107, 21110–21115 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawano Y. & Kypta R. Secreted antagonists of the Wnt signalling pathway. J. Cell Sci. 116, 2627–2634 (2003). [DOI] [PubMed] [Google Scholar]
- Bergmann M. W. WNT signaling in adult cardiac hypertrophy and remodeling: lessons learned from cardiac development. Circ. Res. 107, 1198–1208 (2010). [DOI] [PubMed] [Google Scholar]
- Liang X. et al. HCN4 dynamically marks the first heart field and conduction system precursors. Circ. Res. 113, 399–407 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soriano P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nature Genet. 21, 70–71 (1999). [DOI] [PubMed] [Google Scholar]
- Laugwitz K. L., Moretti A., Caron L., Nakano A. & Chien K. R. Islet1 cardiovascular progenitors: a single source for heart lineages? Development 135, 193–205 (2008). [DOI] [PubMed] [Google Scholar]
- Moretti A. et al. Multipotent embryonic Isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 (2006). [DOI] [PubMed] [Google Scholar]
- Spater D. et al. A HCN4+ cardiomyogenic progenitor derived from the first heart field and human pluripotent stem cells. Nature Cell Biol. 15, 1098–1106 (2013). [DOI] [PubMed] [Google Scholar]
- Bruneau B. G. et al. A murine model of Holt-Oram syndrome defines roles of the T-box transcription factor Tbx5 in cardiogenesis and disease. Cell 106, 709–721 (2001). [DOI] [PubMed] [Google Scholar]
- Devine W. P., Wythe J. D., George M., Koshiba-Takeuchi K. & Bruneau B. G. Early patterning and specification of cardiac progenitors in gastrulating mesoderm. eLife 3, e03848 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokkinopoulos I. et al. Single-cell expression profiling reveals a dynamic state of cardiac precursor cells in the early mouse embryo. PLoS ONE 10, e0140831 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamagishi H. et al. Tbx1 is regulated by tissue-specific forkhead proteins through a common Sonic hedgehog-responsive enhancer. Genes Dev. 17, 269–281 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huynh T., Chen L., Terrell P. & Baldini A. A fate map of Tbx1 expressing cells reveals heterogeneity in the second cardiac field. Genesis 45, 470–475 (2007). [DOI] [PubMed] [Google Scholar]
- Satoh W., Matsuyama M., Takemura H., Aizawa S. & Shimono A. Sfrp1, Sfrp2, and Sfrp5 regulate the Wnt/beta-catenin and the planar cell polarity pathways during early trunk formation in mouse. Genesis 46, 92–103 (2008). [DOI] [PubMed] [Google Scholar]
- Stevens S. M. & Pu W. T. HCN4 charges up the first heart field. Circ. Res. 113, 350–351 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikawa T. & Gourdie R. G. Pericardial mesoderm generates a population of coronary smooth muscle cells migrating into the heart along with ingrowth of the epicardial organ. Dev. Biol. 174, 221–232 (1996). [DOI] [PubMed] [Google Scholar]
- Pennisi D. J., Rentschler S., Gourdie R. G., Fishman G. I. & Mikawa T. Induction and patterning of the cardiac conduction system. Int. J. Dev. Biol. 46, 765–775 (2002). [PubMed] [Google Scholar]
- Asai R. et al. Endothelin receptor type A expression defines a distinct cardiac subdomain within the heart field and is later implicated in chamber myocardium formation. Development 137, 3823–3833 (2010). [DOI] [PubMed] [Google Scholar]
- Kapoor N., Liang W., Marban E. & Cho H. C. Direct conversion of quiescent cardiomyocytes to pacemaker cells by expression of Tbx18. Nature Biotechnol. 31, 54–62 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greulich F. et al. Misexpression of Tbx18 in cardiac chambers of fetal mice interferes with chamber-specific developmental programs but does not induce a pacemaker-like gene signature. J. Mol. Cell. Cardiol. 97, 140–149 (2016). [DOI] [PubMed] [Google Scholar]
- Lescroart F. et al. Clonal analysis reveals common lineage relationships between head muscles and second heart field derivatives in the mouse embryo. Development 137, 3269–3279 (2010). [DOI] [PubMed] [Google Scholar]
- Zaffran S., Kelly R. G., Meilhac S. M., Buckingham M. E. & Brown N. A. Right ventricular myocardium derives from the anterior heart field. Circ. Res. 95, 261–268 (2004). [DOI] [PubMed] [Google Scholar]
- Kelly R. G. Building the right ventricle. Circ. Res. 100, 943–945 (2007). [DOI] [PubMed] [Google Scholar]
- Jensen B., Moorman A. F. & Wang T. Structure and function of the hearts of lizards and snakes. Biol. Rev. Camb. Philos. Soc. 89, 302–336 (2014). [DOI] [PubMed] [Google Scholar]
- Klaus A., Saga Y., Taketo M. M., Tzahor E. & Birchmeier W. Distinct roles of Wnt/beta-catenin and Bmp signaling during early cardiogenesis. Proc. Natl Acad. Sci. USA 104, 18531–18536 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norden J., Greulich F., Rudat C., Taketo M. M. & Kispert A. Wnt/beta-catenin signaling maintains the mesenchymal precursor pool for murine sinus horn formation. Circ. Res. 109, e42–e50 (2011). [DOI] [PubMed] [Google Scholar]
- Kawamoto S. et al. A novel reporter mouse strain that expresses enhanced green fluorescent protein upon Cre-mediated recombination. FEBS Lett. 470, 263–268 (2000). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figures, Supplementary reference
Data Availability Statement
The authors declare that all data supporting the findings of this study are available within the paper and its Supplementary Information files or available on reasonable request from the corresponding authors.