Abstract
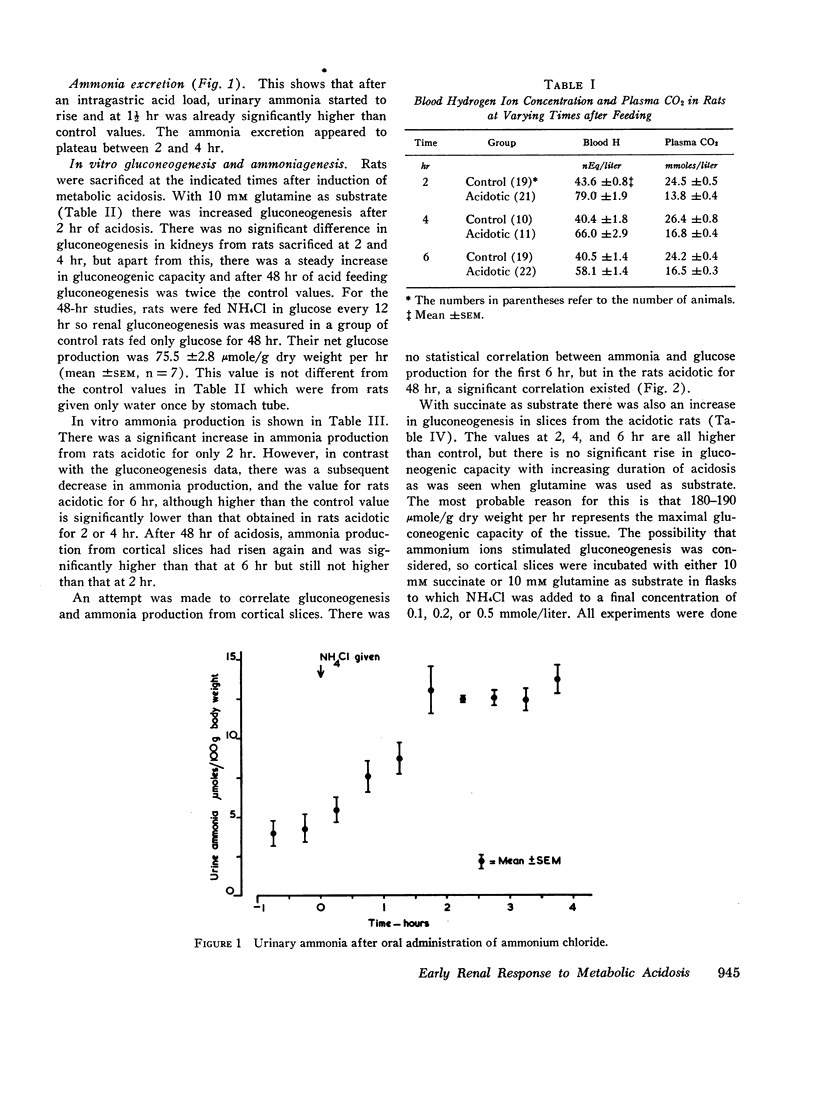
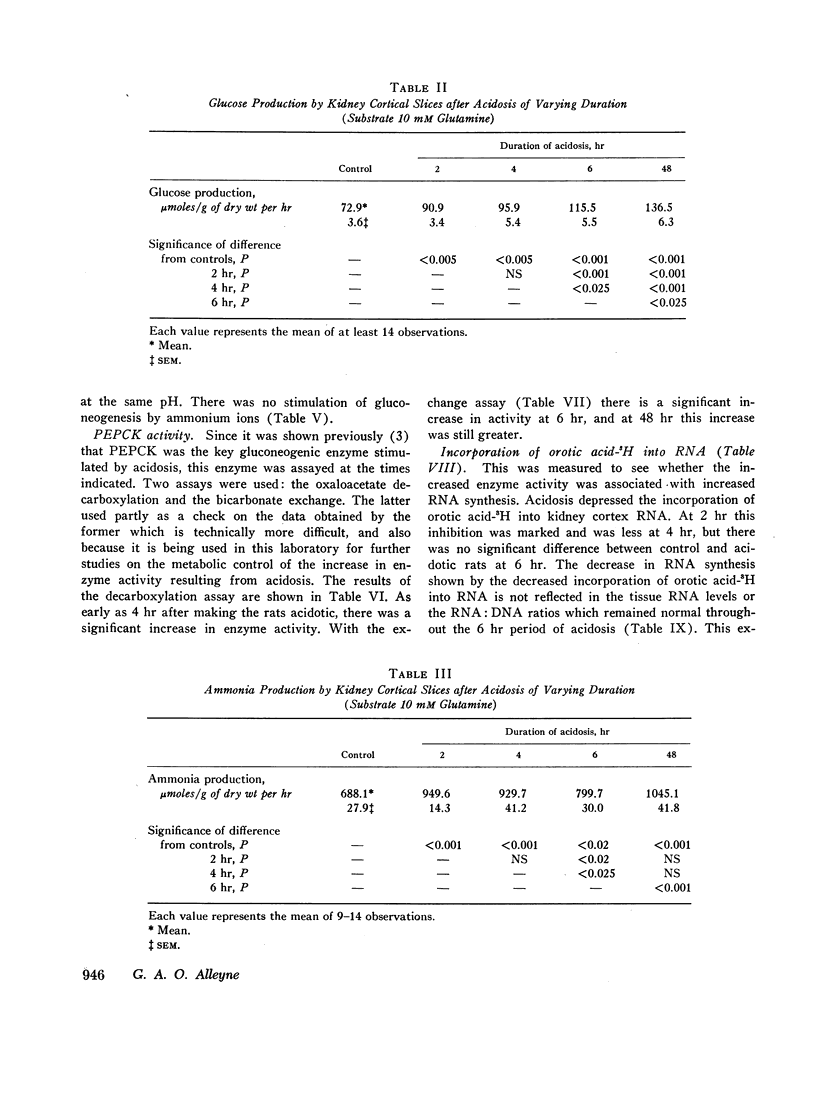
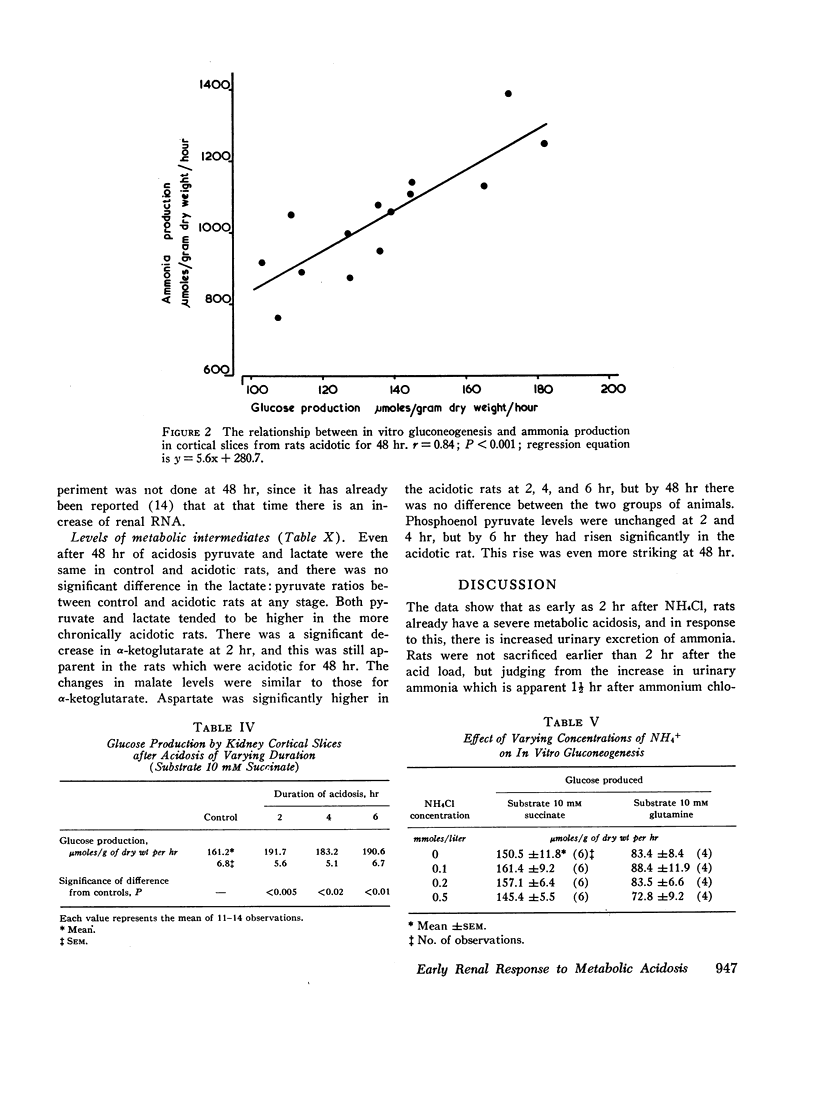
The early renal metabolic response was studied in rats made acidotic by oral feeding of ammonium chloride. 2 hr after feeding of ammonium chloride there was already significant acidosis. Urinary ammonia also increased after ammonium chloride ingestion and at 1½ hr was significantly elevated. In vitro gluconeogenesis by renal cortical slices was increased at 2 hr and thereafter increased steadily. Ammonia production by the same slices was also increased at 2 hr, but thereafter fell and at 6 hr had decreased to levels which, although higher than those of the control, were lower than those obtained from the rats acidotic for only 2 hr. There was no correlation between in vitro gluconeogenesis and ammonia production by kidney slices from rats during the first 6 hr of acidosis, but after 48 hr of ammonium chloride feeding, these two processes were significantly correlated. The early increase in renal gluconeogenesis was demonstrable with both glutamine and succinate as substrates.
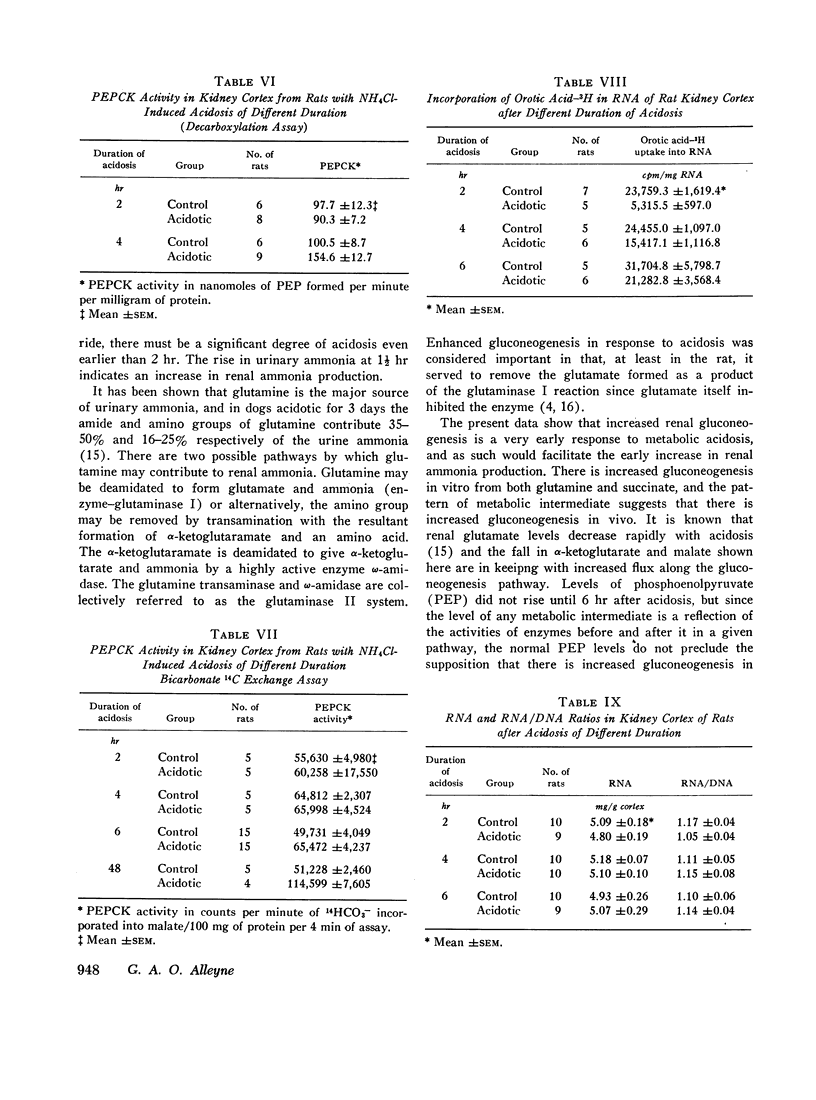
The activity of the enzyme phosphoenolpyruvate carboxykinase was increased after 4-6 hr of acidosis. During this time there was a decrease in renal RNA synthesis as shown by decreased uptake of orotic acid-5H into RNA.
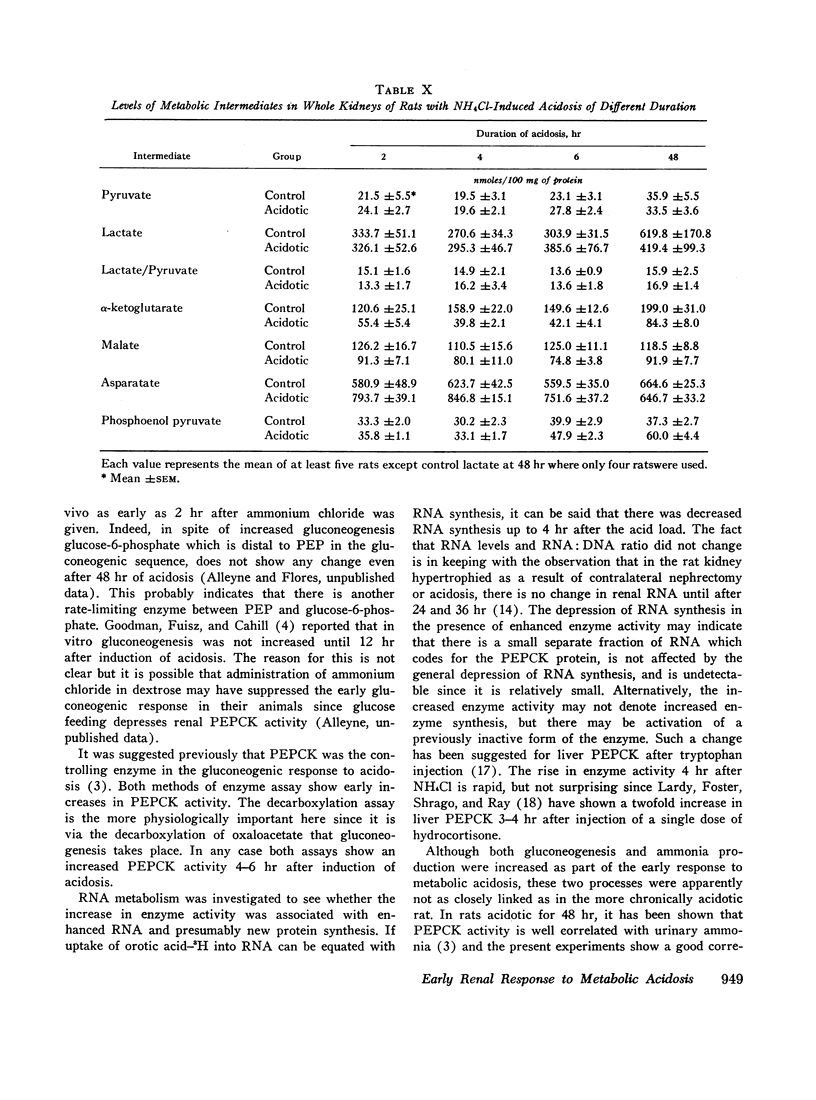
Metabolic intermediates were also measured in quick-frozen kidneys at varying times after induction of acidosis. There was an immediate rise in aspartate and a fall in α-ketoglutarate and malate levels. There was never any difference in pyruvate or lactate levels or lactate:pyruvate ratios between control and acidotic rats. Phosphoenolpyruvate rose significantly after 6 hr of acidosis.
All the data indicate that increased gluconeogenesis is an early response to metabolic acidosis and will facilitate ammonia production by utilization of glutamate which inhibits the glutaminase I enzyme. The pattern of change in metabolic intermediates can also be interpreted as showing that there is not only enhanced gluconeogenesis, but also that there may be significant increase of activity of glutaminase II as part of the very early response to metabolic acidosis.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alleyne G. A. Concentrations of metabolic intermediates in kidneys of rats with metabolic acidosis. Nature. 1968 Mar 2;217(5131):847–848. doi: 10.1038/217847a0. [DOI] [PubMed] [Google Scholar]
- Alleyne G. A., Scullard G. H. Renal metabolic response to acid base changes. I. Enzymatic control of ammoniagenesis in the rat. J Clin Invest. 1969 Feb;48(2):364–370. doi: 10.1172/JCI105993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bignall M. C., Elebute O., Lotspeich W. D. Renal protein and ammonia biochemistry in NH4Cl acidosis and after uninephrectomy. Am J Physiol. 1968 Aug;215(2):289–295. doi: 10.1152/ajplegacy.1968.215.2.289. [DOI] [PubMed] [Google Scholar]
- Chang H. C., Lane M. D. The enzymatic carboxylation of phosphoenolpyruvate. II. Purification and properties of liver mitochondrial phosphoenolpyruvate carboxykinase. J Biol Chem. 1966 May 25;241(10):2413–2420. [PubMed] [Google Scholar]
- DE DUVE C., WATTIAUX R., BAUDHUIN P. Distribution of enzymes between subcellular fractions in animal tissues. Adv Enzymol Relat Subj Biochem. 1962;24:291–358. doi: 10.1002/9780470124888.ch6. [DOI] [PubMed] [Google Scholar]
- Foster D. O., Lardy H. A., Ray P. D., Johnston J. B. Alteration of rat liver phosphoenolpyruvate carboxykinase activity by L-tryptophan in vivo and metals in vitro. Biochemistry. 1967 Jul;6(7):2120–2128. doi: 10.1021/bi00859a033. [DOI] [PubMed] [Google Scholar]
- GOLDSTEIN L., COPENHAVER J. H., Jr Relation of glutaminase I activity to glutamic acid concentration in the rat kidney. Am J Physiol. 1960 Feb;198:227–229. doi: 10.1152/ajplegacy.1960.198.2.227. [DOI] [PubMed] [Google Scholar]
- Goodman A. D., Fuisz R. E., Cahill G. F., Jr Renal gluconeogenesis in acidosis, alkalosis, and potassium deficiency: its possible role in regulation of renal ammonia production. J Clin Invest. 1966 Apr;45(4):612–619. doi: 10.1172/JCI105375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goorno W. E., Rector F. C., Jr, Seldin D. W. Relation of renal gluconeogenesis to ammonia production in the dog and rat. Am J Physiol. 1967 Oct;213(4):969–974. doi: 10.1152/ajplegacy.1967.213.4.969. [DOI] [PubMed] [Google Scholar]
- HUGGETT A. S., NIXON D. A. Use of glucose oxidase, peroxidase, and O-dianisidine in determination of blood and urinary glucose. Lancet. 1957 Aug 24;273(6991):368–370. doi: 10.1016/s0140-6736(57)92595-3. [DOI] [PubMed] [Google Scholar]
- Kamm D. E., Fuisz R. E., Goodman A. D., Cahill G. F., Jr Acid-base alterations and renal gluconeogenesis: effect of pH, bicarbonate concentration, and PCO2. J Clin Invest. 1967 Jul;46(7):1172–1177. doi: 10.1172/JCI105610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lardy H. A., Foster D. O., Shrago E., Ray P. D. Metabolic and hormonal regulation of phosphopyruvate synthesis. Adv Enzyme Regul. 1964;2:39–47. doi: 10.1016/s0065-2571(64)80004-2. [DOI] [PubMed] [Google Scholar]
- McCullough H. The determination of ammonia in whole blood by a direct colorimetric method. Clin Chim Acta. 1967 Aug;17(2):297–304. doi: 10.1016/0009-8981(67)90133-7. [DOI] [PubMed] [Google Scholar]
- NORDLIE R. C., LARDY H. A. Mammalian liver phosphoneolpyruvate carboxykinase activities. J Biol Chem. 1963 Jul;238:2259–2263. [PubMed] [Google Scholar]
- PITTS R. F., PILKINGTON L. A., DEHAAS J. C. N15 TRACER STUDIES ON THE ORIGIN OF URINARY AMMONIA IN THE ACIDOTIC DOG, WITH NOTES ON THE ENZYMATIC SYNTHESIS OF LABELED CLUTAMIC ACID AND GLUTAMINES. J Clin Invest. 1965 May;44:731–745. doi: 10.1172/JCI105186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagliara A. S., Goodman A. D. Effect of adenosine 3',5'-monophosphate on production of glucose and ammonia by renal cortex. J Clin Invest. 1969 Aug;48(8):1408–1412. doi: 10.1172/JCI106106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preuss H. G. Pyridine nucleotides in renal ammonia metabolism. J Lab Clin Med. 1968 Sep;72(3):370–382. [PubMed] [Google Scholar]
- Sartorius O. W., Roemmelt J. C., Pitts R. F., Calhoon D., Miner P. THE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. IV. THE NATURE OF THE RENAL COMPENSATIONS IN AMMONIUM CHLORIDE ACIDOSIS. J Clin Invest. 1949 May;28(3):423–439. doi: 10.1172/JCI102087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson D. P., Sherrard D. J. Regulation of glutamine metabolism in vitro by bicarbonate ion and pH. J Clin Invest. 1969 Jun;48(6):1088–1096. doi: 10.1172/JCI106065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone W. J., Pitts R. F. Pathways of ammonia metabolism in the intact functioning kidney of the dog. J Clin Invest. 1967 Jul;46(7):1141–1150. doi: 10.1172/JCI105607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson D. H., Lund P., Krebs H. A. The redox state of free nicotinamide-adenine dinucleotide in the cytoplasm and mitochondria of rat liver. Biochem J. 1967 May;103(2):514–527. doi: 10.1042/bj1030514. [DOI] [PMC free article] [PubMed] [Google Scholar]



