Significance
As an essential micronutrient, Gram-negative bacteria must concentrate Mn2+ into the cytosol via active transport systems to meet cellular demands. Whereas inner membrane Mn2+ transporters have been characterized, an active transporter for translocation of Mn2+ across the outer membrane has not been described. Here we report a Mn2+-scavenging pathway consisting of a newly identified TonB-dependent outer membrane manganese transporter, MnoT, and a type VI secretion system (T6SS)-secreted Mn2+-binding protein, TseM. Traditionally, T6SS is recognized as a contact-dependent nanomachine to inject effectors into eukaryotic or prokaryotic cells for virulence or for interbacterial competition. The contact-independent functions of T6SS for metal acquisition and bacteria–bacteria competition, reported here, suggest that T6SS may have been retrofitted by some bacteria to gain additional adaptive functions during evolution.
Keywords: type VI secretion, outer membrane transporter, ion uptake, oxidative stress, Burkholderia
Abstract
Type VI secretion system (T6SS) is a versatile protein export machinery widely distributed in Gram-negative bacteria. Known to translocate protein substrates to eukaryotic and prokaryotic target cells to cause cellular damage, the T6SS has been primarily recognized as a contact-dependent bacterial weapon for microbe–host and microbial interspecies competition. Here we report contact-independent functions of the T6SS for metal acquisition, bacteria competition, and resistance to oxidative stress. We demonstrate that the T6SS-4 in Burkholderia thailandensis is critical for survival under oxidative stress and is regulated by OxyR, a conserved oxidative stress regulator. The T6SS-4 is important for intracellular accumulation of manganese (Mn2+) under oxidative stress. Next, we identified a T6SS-4–dependent Mn2+-binding effector TseM, and its interacting partner MnoT, a Mn2+-specific TonB-dependent outer membrane transporter. Similar to the T6SS-4 genes, expression of mnoT is regulated by OxyR and is induced under oxidative stress and low Mn2+ conditions. Both TseM and MnoT are required for efficient uptake of Mn2+ across the outer membrane under Mn2+-limited and -oxidative stress conditions. The TseM–MnoT-mediated active Mn2+ transport system is also involved in contact-independent bacteria–bacteria competition and bacterial virulence. This finding provides a perspective for understanding the mechanisms of metal ion uptake and the roles of T6SS in bacteria–bacteria competition.
Manganese (Mn2+) is an essential micronutrient transition metal required for many cellular processes, including intermediary metabolism, transcriptional regulation, and particularly resistance to oxidative stress (1, 2). Manganese mitigates oxidative stress by serving as a cofactor for reactive oxygen species (ROS)-detoxifying enzymes (i.e., the superoxide dismutase SodA and the catalase KatN) and through the formation of nonproteinaceous manganese antioxidants in a large variety of organisms (1–4). Manganese can also enhance oxidative stress resistance by replacing the more reactive iron cofactor in certain iron-containing enzymes susceptible to oxidative attack (5). Given the essential role of Mn2+ in bacterial physiology, it is not surprising that restriction of this micronutrient is an important innate defense strategy. Indeed, Mn2+ is strictly sequestered by a defense mechanism termed nutritional immunity in mammalian hosts (6–8). To acquire sufficient Mn2+ concentrations for adaptation to environmental stress and pathogenesis within the host niche, bacteria have developed a number of Mn2+ transporters. The import of Mn2+ across the inner membrane is primarily mediated by either the ATP-binding cassette (ABC) family transporter exemplified by SitABCD (9, 10), or the natural resistance-associated macrophage protein (NRAMP) family transporter exemplified by MntH (11–13). Recently, Hohle et al. (14) described a Mn2+-selective channel protein, MnoP, which facilitates passive transport of free Mn2+ across the outer membrane of Bradyrhizobium japonicum. However, to date, an active transporter for translocation of Mn2+ across the outer membrane has not been described.
The type VI secretion system (T6SS) is a widespread protein export apparatus used by many Gram-negative bacteria to translocate effector proteins into eukaryotic or prokaryotic cells in a contact-dependent manner (15–18). Many species possess multiple evolutionarily distinct T6SSs, which are found to play versatile physiological roles in areas such as acute and chronic infections, interbacterial interactions, biofilm formation, and stress response (15, 19–23). A well-established function of the T6SSs is to compete against rival bacteria in polymicrobial environments by delivering “antibacterial” toxins such as cell-wall–degrading enzymes, nucleases, and membrane-targeting enzymes, into target competitor bacterial cells (24–27). Moreover, some T6SSs associated with pathogens have been reported to be involved in bacterial pathogenesis by translocating “antieukaryotic” effectors into eukaryotic cells to modulate host immunity and inflammation (28–31). The well-characterized antieukaryotic effectors are several “evolved” VgrG proteins and non-VgrG phospholipases and deamidases (28–31). Recently, we reported that the T6SS-4 from Yersinia pseudotuberculosis was involved in zinc transport via secretion of the zinc-chelating effector YezP into medium (32). Although the underlying mechanisms remain unknown, this finding uncovers a function of T6SS in increasing bacterial fitness by competition for essential nutrients and reducing ROS. This finding also raises the question of whether T6SS can transport other metal ions such as Mn2+, which plays crucial roles in detoxification of ROS in microorganisms.
Burkholderia thailandensis, a soil bacterium nonpathogenic to humans, is often used as a model organism for Burkholderia pseudomallei, the causative agent of the severe disease melioidosis (33, 34). B. pseudomallei is highly resistant to numerous antibiotics and is listed as a category B priority pathogen and a tier 1 select agent for its potential use as a biological weapon. Whereas the genomes of many bacteria harbor one to two T6SS gene clusters (22, 35), the closely related B. thailandensis and B. pseudomallei contain five and six such clusters, respectively, suggesting multiple functions or specificities for particular niches or hosts. In Burkholderia thailandensis, T6SS-5 was found to play a critical role in the virulence of the organism in a murine melioidosis model, and T6SS-1 was found to be required for contact-dependent interbacterial competition (22). However, the function of other such transporters remains enigmatic.
We here demonstrate that OxyR, a conserved oxidative stress regulator, regulates the T6SS-4 in B. thailandensis, which in turn facilitates the uptake of Mn2+ to mitigate oxidative stress through secreting a Mn2+-binding effector TseM. Uptake of TseM-bound Mn2+ requires a TonB-dependent outer membrane transporter (TBDT), MnoT, for active transport across the outer membrane under Mn2+-limited and oxidative stress conditions. Because TseM is the first reported proteinaceous nonhemopore metallophore recognized by TBDT, this finding greatly expands our understanding of active metal ion transport in bacteria. Metal assimilation and oxidative stress resistance mediated by the T6SS confers a contact-independent competitive advantage distinct from those well-studied T6SS-mediated contact-dependent functions, suggesting its diverse physiological functions have yet to be fully appreciated in Gram-negative bacteria.
Results
OxyR Negatively Regulates T6SS-4 Expression in B. thailandensis.
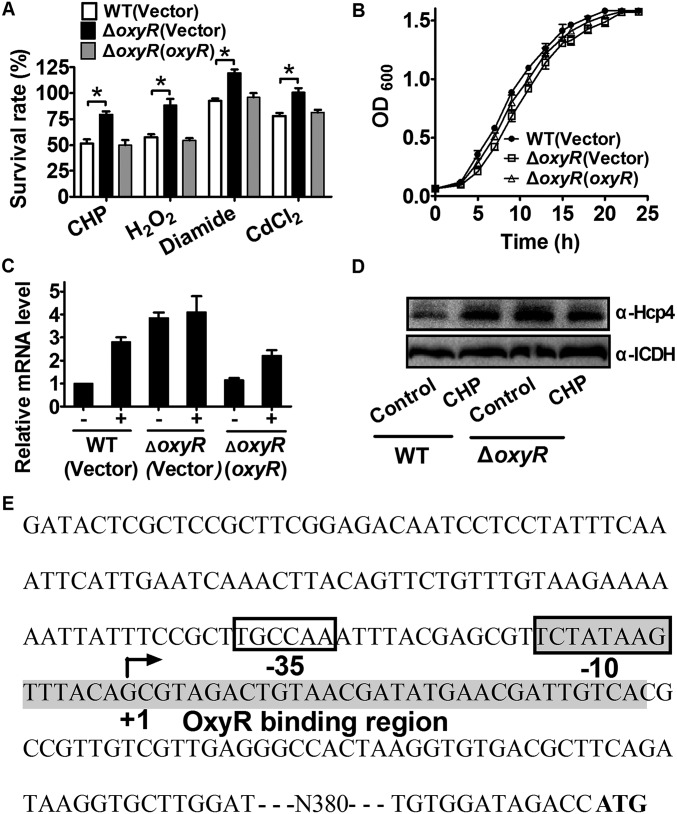
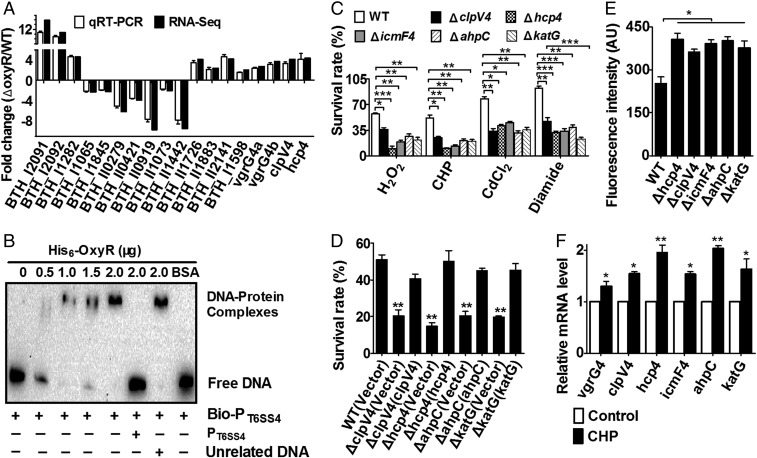
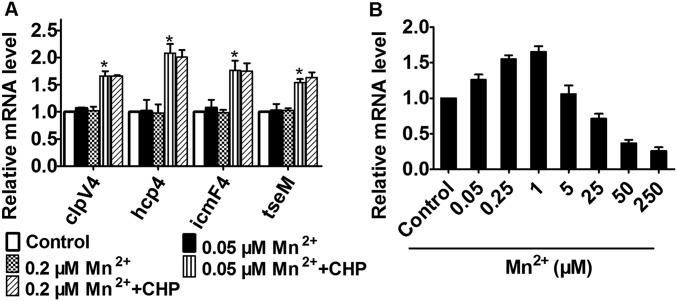
OxyR is a known regulator for oxidative stress in many bacterial species but its role in B. thailandensis has not been characterized. Here we found that the B. thailandensis ΔoxyR mutant showed increased resistance compared with the wild-type and the complement strain upon cumene hydroperoxide (CHP), H2O2, CdCl2, and diamide challenge (Fig. S1A). However, deletion of oxyR did not affect bacterial growth under normal condition (Fig. S1B). This finding indicates that B. thailandensis OxyR negatively regulates cellular defense genes against oxidative stress. To systematically identify OxyR-controlled genes, we performed RNA sequencing (RNA-seq)-based transcriptomic analysis and identified 673 differentially expressed genes either induced or repressed at least 1.6-fold (Dataset S1). We then validated the transcriptomic data using quantitative reverse transcriptase PCR (qRT-PCR) analysis on 14 representative genes (Fig. 1A). Interestingly, similar to the known target genes of OxyR, including ahpC (bth_I2092) and katG (bth_I1282), most genes encoding the major components of T6SS-4 showed significantly enhanced transcription in the ΔoxyR mutant compared with that in the wild type (Dataset S1). Negative regulation of T6SS-4 component genes (vgrG4a, vgrG4b, clpV4, and hcp4) by OxyR was also confirmed by qRT-PCR analysis (Fig. 1A). The negative regulation of T6SS-4 by OxyR was derepressed upon CHP challenge (Fig. S1 C and D). The results indicate that OxyR functions as a general transcriptional repressor similar to the OxyR homologs in B. pseudomallei (36), Neisseria gonorrheae (37), and Corynebacterium glutamicum (38).
Fig. S1.
OxyR regulates the expression of T6SS-4. (A) The ΔoxyR mutant was highly resistant to oxidative stress. The survival rate was measured by viability assay. Mean values with SDs (error bars) from at least three repeats are shown. *P < 0.05. (B) Growth curves of the wild-type (WT), ΔoxyR mutant, and the complemented strain ΔoxyR(oxyR) under normal condition. The growth of the indicated strains in LB was monitored by measuring OD600 at indicated time points. (C) The levels of hcp4 mRNAs in exponentially growing B. thailandensis cells with (+) or without (−) 20-min exposure to 0.15 mM CHP was determined by quantitative RT-PCR. The mRNA levels are presented relative to the value obtained from WT cells without CHP treatment. (D) The protein level of Hcp4 in differently treated WT and ΔoxyR mutant strains. Lysates from bacteria with or without (control) 30-min exposure to 0.15 mM CHP were resolved by SDS/PAGE, and Hcp4 was detected by immunoblotting using a specific anti-Hcp4 antibody. For the pellet fraction, isocitrate dehydrogenase (ICDH) was used as a loading control. (E) Identification of the OxyR binding site in the promoter region of T6SS-4. Putative OxyR binding site identified by the online software Virtual Footprint (www.prodoric.de/vfp) was indicated by shading. The ATG start codon of the first ORF of the T6SS-4 operon was marked in boldface, and the –35 and –10 elements of the T6SS-4 promoter are boxed. +1 denotes the transcription start point.
Fig. 1.
OxyR-regulated T6SS-4 is involved in oxidative stress resistance. (A) Genes differentially transcribed in the B. thailandensis oxyR mutant compared with those in the wild type were detected by transcriptomic and qRT-PCR analyses. Fourteen representative genes were chosen to validate the RNA-seq data by qRT-PCR. The white bars represent the mean values obtained for the reference wild type and three biological replicates. Error bars indicate the SD. Black bars represent RNA-seq data. (B) Binding of OxyR to the T6SS-4 promoter. Interaction of OxyR with a biotin-labeled probe was detected using streptavidin-conjugated HRP and a chemiluminescent substrate. An unlabeled promoter was added to determine the binding specificity of OxyR. Bio-PT6SS-4, biotin-labeled T6SS-4 promoter. (C and D) The indicated strains grown to the stationary phase were exposed to diverse stress for 40 min and the viability of the cells was determined. Data shown are the average and SD from three independent experiments. (E) Deletion of T6SS-4 led to accumulation of intracellular ROS under oxidative conditions. Intracellular ROS in stationary phase bacterial strains exposed to CHP was determined with the H2DCFDA probe. Data shown are the average and SD from three independent experiments. (F) Oxidative stress induced the expression of T6SS-4. B. thailandensis strains were treated with the indicated amounts of CHP and the expressions of the major components of T6SS-4 were measured by qRT-PCR. Data shown are the average and SD from three independent experiments. ***P ≤ 0.001; **P ≤ 0.01; *P ≤ 0.05.
To determine whether OxyR regulates T6SS-4 expression directly, we examined the interaction between OxyR and the T6SS-4 promoter using an electrophoretic mobility shift assay (EMSA). Incubation of a probe containing the T6SS-4 promoter (PT6SS-4) sequence (−536 to −186 relative to the ATG start codon of the first ORF of the T6SS-4 operon) with His6-OxyR led to the formation of DNA–protein complexes, and addition of excessive unlabeled probe abolished the formation of the protein–DNA complex (Fig. 1B). Consistently, a DNA element highly similar to the known recognition site for OxyR was identified in the T6SS-4 promoter region (Fig. S1E). Thus, OxyR negatively regulates T6SS-4 expression by specifically recognizing an operator within the T6SS-4 promoter.
The direct regulation of T6SS-4 by OxyR prompted us to examine whether the T6SS-4 plays a role in protection against oxidative stress. Thus, we determined the viability of B. thailandensis T6SS-4 mutants challenged with diverse oxidative stressors such as CHP, H2O2, CdCl2, and diamide. Similar to the ΔkatG and ΔahpC mutants, all mutants lacking conserved T6SS-4 structural genes were significantly more sensitive to these stressors than wild-type bacteria (Fig. 1C). Moreover, the sensitivity of the ΔclpV4 and Δhcp4 mutants to CHP was almost completely alleviated by complementation of the clpV4 and hcp4 genes, respectively (Fig. 1D), further supporting the role of T6SS-4 in combating oxidative stresses. For simplicity, we used CHP only as the oxidative stressor in the following experiments.
Oxidative stress induces the production of harmful ROS. To examine the effect of T6SS-4 on ROS reduction upon oxidative stress, we assessed the intracellular ROS levels in B. thailandensis wild-type and mutant strains challenged with CHP by using the fluorogenic probe 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA). As shown in Fig. 1E, mutants lacking essential components of T6SS-4 had significantly higher ROS levels than the wild type after exposure to CHP, indicating that T6SS-4 is critical in reducing ROS accumulated in B. thailandensis under stress conditions (Fig. 1E). In agreement with these observations, the expression of T6SS-4 component genes was also induced by CHP along with katG and ahpC (Fig. 1F). Altogether, these data indicate that the T6SS-4 is induced and important for survival under oxidative stress.
T6SS-4 Combats Oxidative Stress by Importing Mn2+.
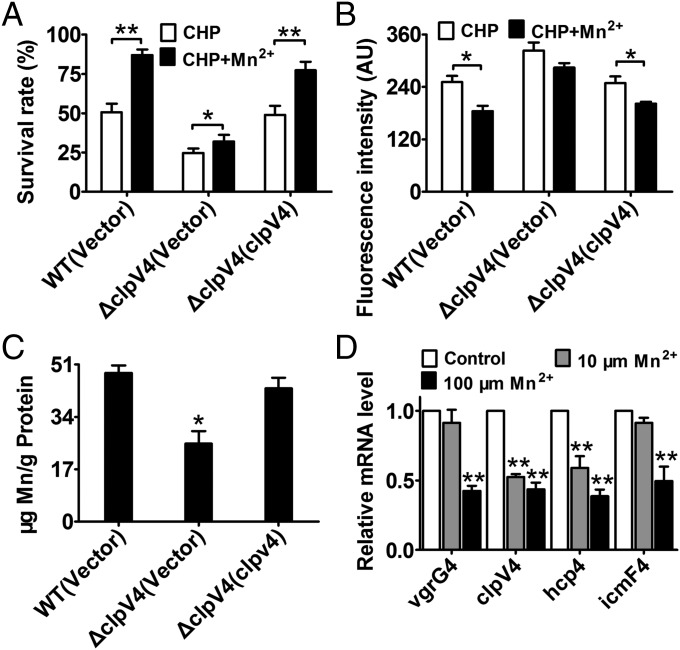
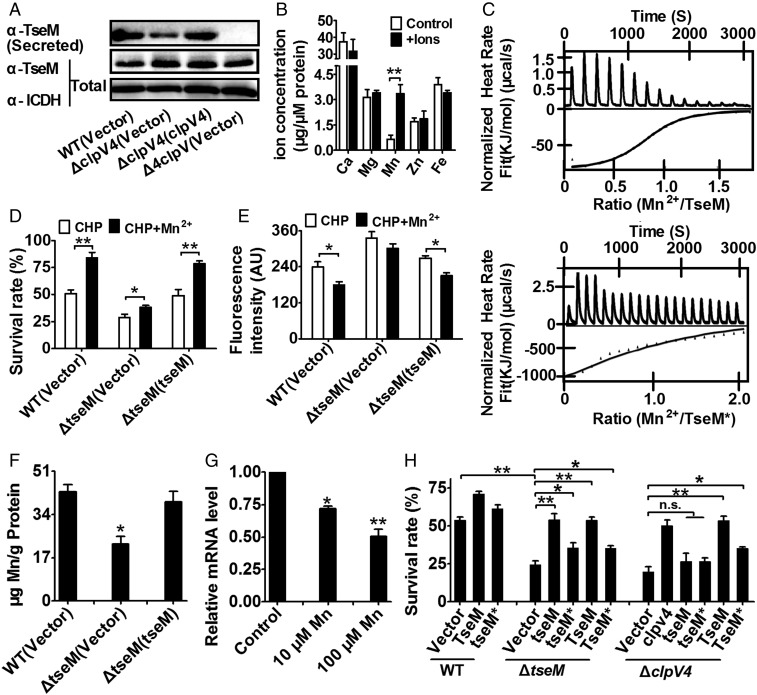
Manganese is known to play crucial roles in protection against oxidative damage (1–5). Our observation that B. thailandensis T6SS-4 has antioxidant function prompted us to examine whether it is involved in Mn2+ acquisition for oxidative stress survival. As shown in Fig. 2A, whereas exogenous Mn2+ (0.25 μM) markedly increased the survival rate of the wild-type and the complemented strain ΔclpV4(clpV4) under CHP challenge, the protective effect of exogenous Mn2+ was largely abolished in the ΔclpV4 mutant. In addition, exogenous Mn2+ significantly reduced intracellular ROS levels in the wild-type and the complemented strain, whereas a lesser effect was observed in the ΔclpV4 mutant (Fig. 2B). These results suggest that T6SS-4 plays a role in the transport of Mn2+ for survival under oxidative stress.
Fig. 2.
T6SS-4 is important for the accumulation of intracellular Mn2+ under oxidative stress conditions. (A) The alleviation of the sensitivity of B. thailandensis strains to CHP by exogenous Mn2+ (0.25 μM) required T6SS-4. Relevant stationary phase bacterial strains were exposed to 0.25 mM CHP in M9 medium with or without exogenously provided Mn2+ (0.25 μM) for 40 min and the viability of the cells was determined. The mean values and SDs from at least three repeats are shown. (B) Reduction of intracellular ROS in CHP-treated B. thailandensis strains by exogenous Mn2+ (0.25 μM) required T6SS-4. The mean values and SD from at least three repeats are shown. (C) Mn2+ uptake required T6SS-4 under oxidative stress conditions. Stationary phase B. thailandensis strains were exposed to 0.25 mM CHP for 20 min in PBS containing 0.25 μM MnSO4. Mn2+ associated with bacterial cells was measured by inductively coupled plasmon resonance atomic absorption spectrometry (ICP-MS). (D) T6SS-4 expression is inhibited under high Mn2+ conditions. B. thailandensis wild type was grown in LB containing 10 or 100 μM Mn2+, and the expression of the major T6SS-4 genes was measured by qRT-PCR. Data shown are the average of three independent experiments and error bars indicate the SD from three independent experiments. **P ≤ 0.01; *P ≤ 0.05.
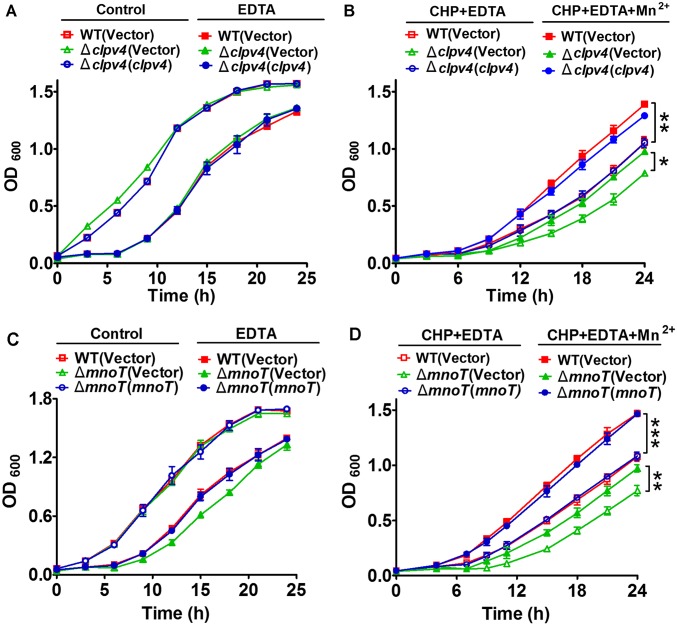
We then postulated that the growth of the ΔclpV4 mutant might be affected by Mn2+ starvation under oxidative conditions. This prediction was confirmed by comparing the growth of the B. thailandensis wild type, the ΔclpV4 mutant, and the complemented strain ΔclpV4(clpV4) in the presence of ethylene diamine tetraacetic acid (EDTA) under CHP stress (Fig. S2 A and B). Whereas the levels of growth of all tested strains were nearly identical in lysogeny broth (LB) medium and LB containing EDTA (Fig. S2A), the growth of the ΔclpV4 mutant was severely impaired in comparison with that of the wild-type in LB medium containing EDTA under CHP treatment (Fig. S2B). The plasmid-borne expression of clpV4 completely rescued the sensitivity of the ΔclpV4 mutant to CHP (Fig. S2B). Moreover, although the growth of all strains was improved by the addition of excessive Mn2+ (250 μM) under CHP treatment, the level of increase was less in the ΔclpV4 mutant than in the wild-type and the complemented strain ΔclpV4(clpV4) (Fig. S2B).
Fig. S2.
T6SS-4 and MnoT are required for optimal growth under oxidative stress. Saturated bacterial cultures were diluted to an OD600 of 0.05 in LB medium (A and C); LB medium with 250 μM EDTA (A and C); LB medium with 240 μM CHP and 250 μM EDTA (B and D); and LB medium with 240 μM CHP, 250 μM EDTA, and 250 μM Mn2+ (B and D). The growth of the cultures was monitored by measuring OD600 at indicated time points. Data shown were the average of three independent experiments; error bars indicate SD from three independent experiments. ***P < 0.001; **P < 0.01; *P < 0.05.
To test whether the increased T6SS-4–dependent survival is due to Mn2+ acquisition, we measured the total metal content in bacterial cells treated with CHP using inductively coupled plasmon resonance atomic absorption spectrometry (ICP-MS). Our results revealed that deletion of the clpV4 significantly lowered intracellular Mn2+ levels and that the complementation of clpV4 restored such defects (Fig. 2C). Consistent with its role in Mn2+ acquisition, the expression of T6SS-4 genes was repressed by exogenous Mn2+ in a dose-dependent manner (Fig. 2D). Altogether, these data demonstrate that the T6SS-4 mitigates oxidative stress through the uptake of antioxidant manganese.
T6SS-4 Secretes a Mn2+-Binding Protein Substrate.
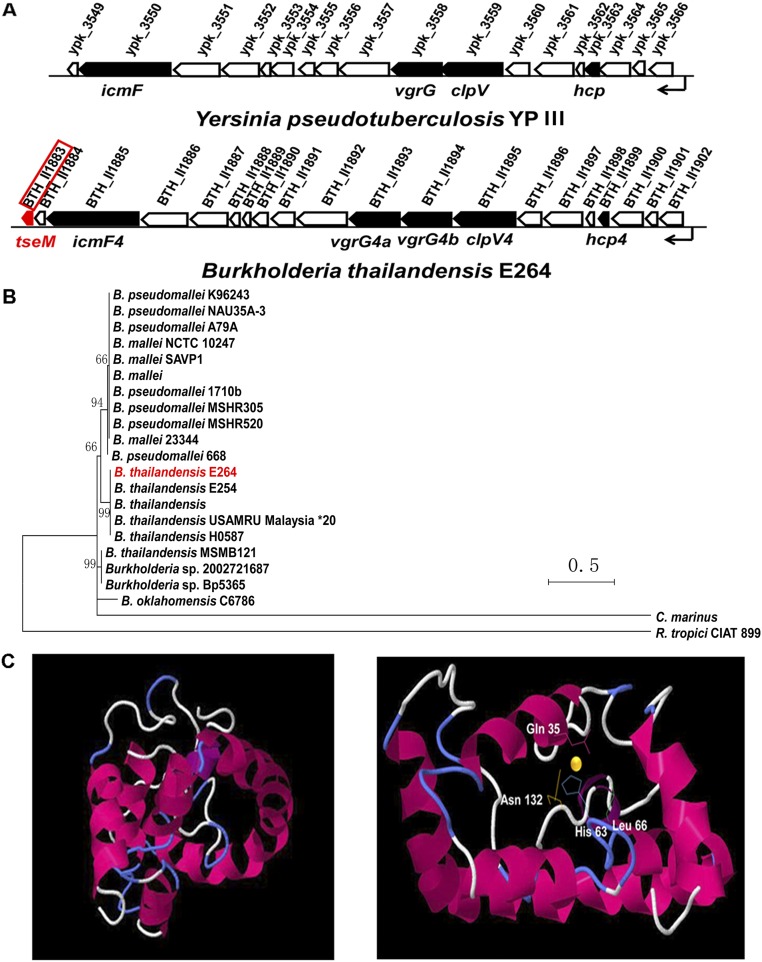
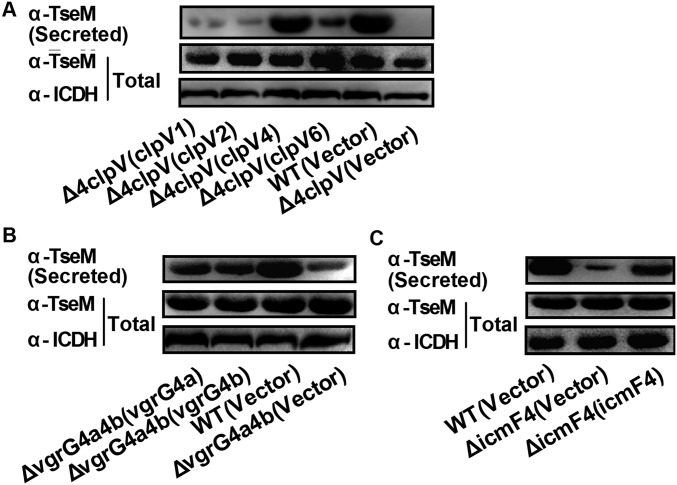
Previously, we showed that zinc transport by Y. pseudotuberculosis T6SS-4 can be achieved by secreting a zinc scavenger YezP located at the end of the T6SS-4 gene cluster (32). Here we identified a 154-residue protein (BTH_II1883) encoded by a gene also located at the end of the B. thailandensis T6SS-4 gene cluster (Fig. S3A). Moreover, no putative promoter was identified upstream of the BTH_II1883 ORF, suggesting that this gene is part of the T6SS-4 operon. This finding prompted us to examine whether BTH_II1883 is a secreted substrate of T6SS-4. As shown in Fig. 3A, significant amounts of BTH_II1883 were readily detected in the culture supernatant of wild-type bacteria recognized by a specific anti-BTH_II1883 rabbit polyclonal antibody (Fig. 3A). Deletion of clpV4 abrogated the secretion of BTH_II1883 to a large extent. The secretion of BTH_II1883 was completely abrogated in a mutant (Δ4clpV) lacking the clpV genes of the four T6SSs (T6SS-1, T6SS-2, T6SS-4, and T6SS-6) in B. thailandensis (Fig. 3A). Although complementation of the ΔclpV4 (Fig. 3A) or Δ4clpV mutant (Fig. S4A) with clpV4 restored BTH_II1883 secretion to the wild-type level, complementation of the Δ4clpV mutant with clpV1, clpV2, or clpV6 has only minor effects on recovery of BTH_II1883 secretion (Fig. S4A). These data suggest that BTH_II1883 is a substrate primarily secreted by T6SS-4 although there might be substrate cross-recognition among different T6SSs. The T6SS-4 cluster has two vgrG genes encoding VgrG4a and VgrG4b. Similarly, the ΔvgrG4aΔvgrG4b double mutant exhibited largely attenuated BTH_II1883 secretion, and complementation of either vgrG4a or vgrG4b partially restored BTH_II1883 secretion, indicating that the two VgrGs may act cooperatively as carriers in facilitation of TseM secretion (39) (Fig. S4B). In addition, TseM secretion was also impaired in the deletion mutant of icmF4, encoding a key T6SS structural protein, and restored in the complemented strain (Fig. S4C), further supporting that TseM secretion is mediated by the T6SS-4.
Fig. S3.
Identification of a putative T6SS effector in the B. thailandensis T6SS-4 gene cluster. (A) The structure of B. thailandensis T6SS-4 gene cluster is similar to the T6SS-4 gene cluster from Y. pseudotuberculosis. The tseM gene (bth_II1883, indicated in red) localizes in the end of the T6SS-4 operon. (B) Phylogenetic relationship of B. thailandensis TseM with homologous proteins in other relative Burkholderia. Different protein sequences were obtained from the SwissProt database. The phylogenetic tree was constructed using MEGA 6.0 by the neighbor-joining method and multiple sequence alignment was performed using CLUSTAL W. The scale bar indicates percentage of divergence (distance). Accession numbers are as follows: B. thailandensis E264 (ABC35934); B. thailandensis E254 (AIT22423); B. thailandensis (AIP65980); B. thailandensis USAMRU Malaysia (AIC89753); B. thailandensis MSMB121 (AGK49752); B. mallei (AIO54172); B. mallei SAVP1 (ABM48775); B. mallei NCTC 10247 (AIS27138); B. pseudomallei 668 (ABN88025); B. pseudomallei A79A (AIV92858); B. pseudomallei K96243 (YP_110555); B. pseudomallei NAU35A-3 (AIS91265); B. oklahomensis C6786 (AJX34588); Burkholderia sp. 2002721687 (AJY39125); Burkholderia sp. Bp5365 (ALX46218); Celeribacter marinus IMCC12053_1024 (ALI54972.1); and Rhizobium tropici CIAT 899 (AGB70070.1). (C) Side and top 3D model of TseM-ion binding built with phyre2. The ion is illustrated as a yellow sphere.
Fig. 3.
A Mn2+-binding protein translocated by T6SS-4 resisted oxidative stress. (A) TseM is a secreted substrate of T6SS-4. Proteins in culture supernatant of the relevant B. thailandensis strains were probed using specific anti-TseM antibody. For the pellet fraction, isocitrate dehydrogenase (ICDH) was used as a loading control. (B) The binding of divalent ions by TseM was detected by atomic absorption spectrometry. (C) The binding of Mn2+ by TseM. Mn2+-free TseM (Upper) or TseM* (TseMQ35R/H63A/N132R) (Lower) was used to evaluate Mn2+-binding activity by isothermal titration calorimetry (ITC). Data were analyzed using the NanoAnalyze software (TA Instruments). (D) The alleviation of the sensitivity of B. thailandensis strains to CHP by exogenous Mn2+ (0.25 μM) required the TseM protein. The viability of stationary phase B. thailandensis strains was determined after exposure to CHP, or CHP and 0.25 μM Mn2+ for 40 min. (E) Deletion of the tseM gene led to an accumulation of intracellular ROS. The intracellular levels of ROS were determined with the H2DCFDA probe after stationary phase B. thailandensis strains were exposed to CHP, or CHP with 0.25 μM Mn2+ for 40 min. (F) TseM is involved in Mn2+ acquisition. Stationary phase B. thailandensis strains were exposed to 0.25 mM CHP for 20 min in PBS containing 0.25 μM MnSO4. Mn2+ associated with bacterial cells was determined by ICP-MS. (G) TseM expression is inhibited by high Mn2+ conditions. B. thailandensis wild-type cells were grown in LB medium with 10 and 100 μM Mn2+, and the expression of tseM was measured using qRT-PCR. (H) The rescue of the tseM or clpV4 mutant using recombinant TseM protein. Recombinant TseM or TseM* (TseMQ35R/H63A/N132R) protein at 1 μM was added to bacterial survival experiments in M9 medium before viability assessment. Mutants complemented with the corresponding gene were used as controls. The mean values and SDs from at least three repeats are shown. **P ≤ 0.01; *P ≤ 0.05; n.s., not significant.
Fig. S4.
Effects of different clpVs and vgrGs on TseM secretion. (A) Secretion of TseM in the Δ4clpV mutant complemented with clpV1, clpV2, clpV4, and clpV6, respectively. (B) Secretion of TseM in the ΔvgrG4a4b mutant complemented with vgrG4a and vgrG4b, respectively. (C) Secretion of TseM in the ΔicmF4 mutant and the complemented strain. Proteins in culture supernatant were probed using specific anti-TseM antibody. For the pellet fraction, isocitrate dehydrogenase (ICDH) was used as a loading control.
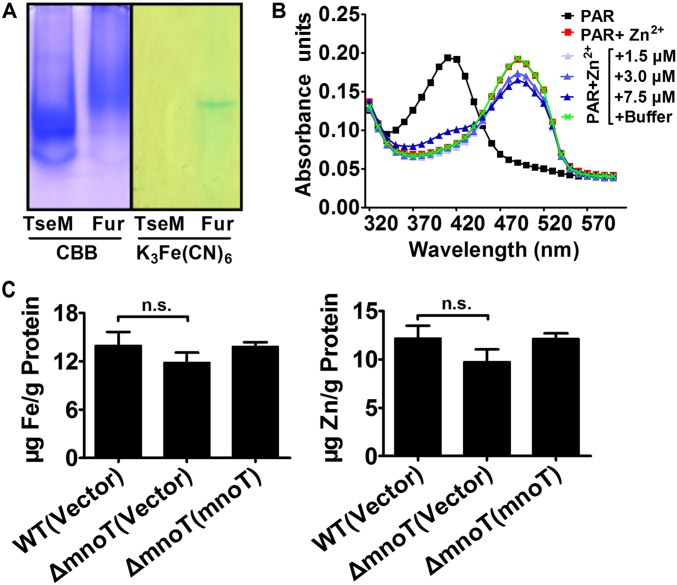
Analysis with atomic absorption spectrometry revealed that BTH_II1883 can specifically bind Mn2+ (Fig. 3B), and a binding Kd of 2.87 ± 0.32 μM was obtained by isothermal titration calorimetry (ITC) analysis (Fig. 3C, Upper). In addition, BTH_II1883 was not able to bind Ca2+, Mg2+, Zn2+, and Fe3+ (Fig. 3B). The inability of BTH_II1883 to bind iron and zinc ions was also confirmed using the potassium ferricyanide assay (Fig. S5A) and the 4-(2-pyridylazo) resorcinol (PAR) assay, respectively (Fig. S5B). Thus, this protein is named type VI secretion system effector for Mn2+ binding (TseM). TseM is conserved in multiple Burkholderia species such as B. pseudomallei, Burkholderia mallei, and Burkholderia oklahomensis (Fig. S3B). Three dimensional structure simulation predicted several ion-binding residues (Gln35, His63, and Asp132) in TseM (Fig. S3C). Mutation of Gln35, His63, and Asn132 (TseMQ35R/H63A/N132R) dramatically reduced (Kd = 219.4 ± 11.5 μM) but did not completely abrogate its affinity to Mn2+ (Fig. 3C, Lower), indicating the existence of other sites involved in Mn2+ binding.
Fig. S5.
TseM and MnoT are not involved in iron and zinc accumulation. (A) TseM (BTH_II1883) does not bind iron. Iron binding analysis by 15% native PAGE. Lanes 1 and 2 show the gel stained with Coomassie bright blue. Lanes 3 and 4 show the same gel stained for iron by the potassium ferricynaide method. His6-Fur was used as a positive control. Lanes 1 and 3 show TseM (BTH_II1883). Lanes 2 and 4 show His6-Fur. (B) TseM (BTH_II1883) does not bind Zn2+. Spectral scans of solutions containing 10 µM PAR without Zn2+ (black), with Zn2+ (red), or with Zn2+ and increasing concentrations of recombinant TseM (BTH_II1883) and control buffer (different color) are shown. (C) MnoT is not involved in iron and zinc accumulation in B. thailandensis. Stationary phase B. thailandensis strains were exposed to 0.25 mM CHP for 20 min in PBS containing 1 μM FeCl3 or 1 μM ZnSO4. Iron and zinc ions associated with bacterial cells were measured using ICP-MS. Data shown are the average and SD from three independent experiments. n.s., not significant.
Because of the Mn2+-dependent T6SS-4 protection in oxidative stress resistance and the Mn2+ binding of TseM, we hypothesized that TseM is important for oxidative stress resistance. Indeed, in the presence of exogenous Mn2+ (0.25 μM), the ΔtseM mutant exhibited increased sensitivity to CHP treatment (Fig. 3D), elevated ROS levels (Fig. 3E), and reduced intracellular Mn2+ (Fig. 3F), resembling the T6SS-4 mutants. Complementation of tseM restores these phenotypes to wild-type levels (Fig. 3 D–F). Similar to T6SS-4 component genes, the expression of tseM was repressed by exogenous Mn2+ in a dose-dependent manner (Fig. 3G) and was induced by low Mn2+ (0.05 μM and 0.2 μM) under oxidative stress (Fig. S6A).
Fig. S6.
Effects of Mn2+ and CHP on T6SS-4 and mnoT expression. (A) B. thailandensis wild type was grown in M9 medium containing different concentrations of Mn2+ with or without CHP (100 µM ), and the expression of clpV4, hcp4, icmF4, and tseM was measured by qRT-PCR. (B) The expression of mnoT is regulated by Mn2+. B. thailandensis wild type was grown in M9 medium containing different concentrations of Mn2+, and the expression of mnoT was measured by qRT-PCR. Data shown were the average of three independent experiments; error bars indicate SD from three independent experiments. *P < 0.05.
Next, we determined whether exogenous purified TseM restores the ability of relevant B. thailandensis mutants to survive oxidative challenge. Inclusion of metal-free TseM (1 μM) in cultures of the ΔtseM mutant fully restored its resistance to CHP, whereas inclusion of the Mn2+ binding attenuated the TseM* variant (TseMQ35R/H63A/N132R), or complementation of the tseM* (tseMQ35R/H63A/N132R) gene only slightly recovered its resistance (Fig. 3H). Moreover, exogenously provided TseM protein, but not TseM protein, intracellularly produced by providing a plasmid encoding TseM, also protected the T6SS-4 mutant ΔclpV4 from CHP-induced toxicity (Fig. 3H). This finding suggests that, after T6SS-4–mediated translocation, Mn2+ uptake by TseM occurred independently of the secretion system. Altogether, these data indicate that the Mn2+-binding effector TseM is required for T6SS-4–dependent Mn2+ acquisition under oxidative stress.
TseM Interacts with a Mn2+-Specific TonB-Dependent Outer Membrane Transporter.
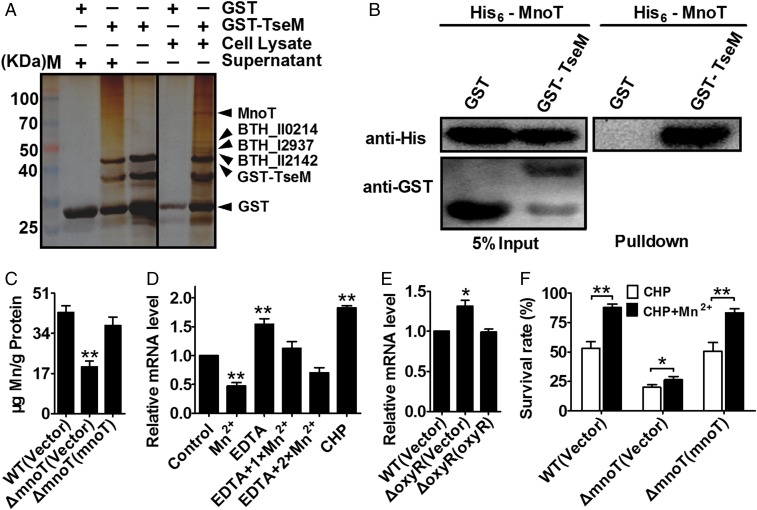
To reveal how TseM transports Mn2+ into the cell, we first attempted to identify bacterial components that interact with TseM. A GST pull-down assay was performed by incubating GST-Bind beads coated with GST-TseM or GST with cell lysates and supernatants of B. thailandensis wild type treated with CHP. After washing with PBS, proteins retained on the beads were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS/PAGE) and then visualized by silver staining. Several proteins in the cell lysate with molecular weights ranging from 40 to 100 kDa were specifically retained by beads coated with GST-TseM, but not by beads coated with GST (Fig. 4A). Mass spectrometry analysis identified the 85-kDa band as a member of the TBDT (BTH_I1598); the 60-kDa band was the ATP-dependent RNA helicase DbpA (BTH_II0214); the band around 50 kDa was identified as a chaperone protein (BTH_I2937); and the band around 44 kDa was an iron complex transport system permease (BTH_II2142) (Fig. 4A).
Fig. 4.
TseM interacts with a TBDT family transporter involved in Mn2+ transport. (A) MnoT was retained by agarose beads coated with GST-TseM. GST-Bind beads coated with GST-TseM (lanes 2, 3, and 5) or GST (lanes 1 and 4) were incubated with CHP-treated B. thailandensis supernatant (lanes 1 and 2) or cell lysates (lanes 4 and 5). After washing with PBS, the proteins resolved by SDS/PAGE were visualized using silver staining, and bands that specifically retained by the GST-TseM–coated beads were identified by mass spectrometry. (B) Direct binding of TseM to MnoT. His6-MnoT was incubated with GST-TseM or GST, and the protein complexes captured with glutathione beads were detected by Western blotting. (C) MnoT is involved in Mn2+ acquisition in B. thailandensis. Stationary phase B. thailandensis strains were exposed to 0.25 mM CHP for 20 min in PBS containing 0.25 μM MnSO4. Mn2+ associated with bacterial cells was determined by ICP-MS. Data shown are the average and SD from three independent experiments. (D) MnoT expression was inhibited by high Mn2+ conditions and induced by CHP and EDTA. Cells of B. thailandensis wild type were grown in LB medium with 100 μM Mn2+, 100 μM CHP, 100 μM EDTA, or 100 μM EDTA together with 100 μM Mn2+ (EDTA + 1× Mn2+), and 100 μM EDTA together with 200 μM Mn2+ (EDTA + 2× Mn2+). The expression of mnoT was measured by qRT-PCR. Data shown are the average and SD from three independent experiments. **P ≤ 0.01; *P ≤ 0.05. (E) The expression of MnoT was negatively regulated by OxyR. Cells of relevant B. thailandensis strains were grown in LB medium and the expression of mnoT was measured by qRT-PCR. Data shown are the average and SD from three independent experiments. (F) Alleviation of the sensitivity of B. thailandensis strains to CHP by exogenous Mn2+ (0.25 μM) required MnoT. The viability of relevant stationary B. thailandensis strains was determined after exposure to CHP or CHP with 0.25 μM Mn2+ for 40 min. The mean values and SD from at least three repeats are shown. **P ≤ 0.01; *P ≤ 0.05.
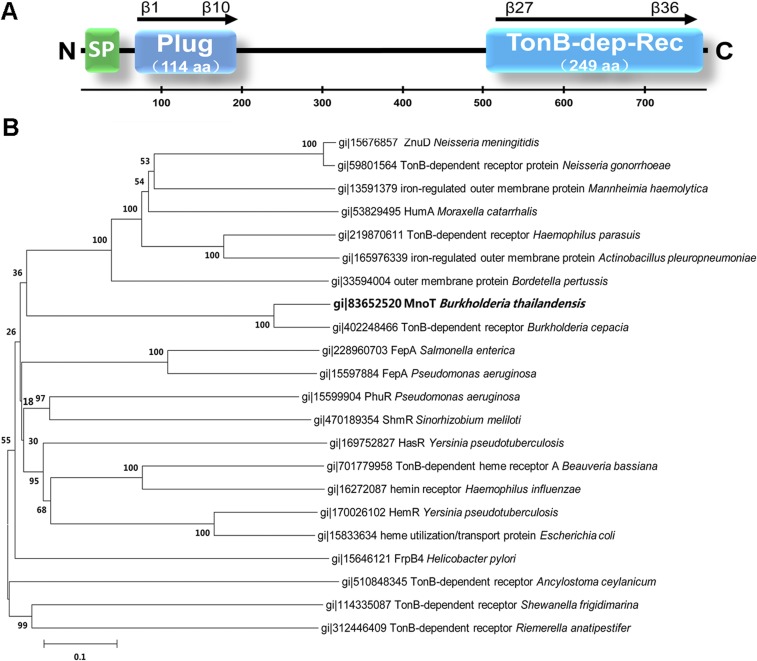
The identification of a TonB-dependent outer membrane transporter prompted us to hypothesize that TseM engages receptor proteins on the bacterial surface to deliver Mn2+ into the cell. Indeed, the specific interaction between TseM and BTH_I1598 was confirmed by an in vitro binding assay with purified GST-BTH_I1598 and His6-TseM proteins (Fig. 4B). Sequence analysis of BTH_I1598 predicted a 114-residue-long N-terminal TonB-plug domain (residues 68–181) and a TonB-dependent receptor domain from residue 529 to the C terminus of the protein (Fig. S7A). TBDTs are bacterial outer membrane transporters that mediate the active uptake of iron siderophores, vitamin B12, nickel, and zinc (40–42). According to phylogenetic tree analysis, BTH_I1598 formed an independent cluster evolutionarily distant from the known outer membrane zinc transporter ZnuD of Neisseria meningitidis (43) and nickel transporter FrpB4 in Helicobacter mustelae (44) (Fig. S7B). This result suggests that BTH_I1598 may be involved in transportation of other metal ions such as Mn2+.
Fig. S7.
Sequence analysis of MnoT. (A) Pertinent secondary structure elements of MnoT. Plug, the TonB-plug domain (residues 68–181); TonB-dep-Rec, the TonB-dependent receptor domain (residues 529–777). (B) Phylogenetic relationship of TonB-dependent outer membrane receptors. Different protein sequences were obtained from the SwissProt database. The phylogenetic tree was constructed using MEGA 6.0 by the neighbor-joining method and multiple sequence alignment was performed using CLUSTAL W. The scale bar indicates percentage of divergence (distance). SwissProt accession nos. of proteins from species are as follows: B. thailandensis BTH_I1598 (gi:83652520); N. meningitidis ZnuD (gi:15676857); Neisseria gonorrheae TonB-dependent receptor protein (gi:59801564); Mannheimia haemolytica iron-regulated outer membrane protein (gi:13591379); Moraxella catarrhalis HumA (gi:53829495); Haemophilus parasuis TonB-dependent receptor (gi:219870611); Actinobacillus pleuropneumoniae iron-regulated outer membrane protein (gi:165976339); Bordetella pertussis outer membrane protein (gi:33594004); Burkholderia cepacia TonB-dependent receptor (gi:402248466); Salmonella enterica FepA (gi:228960703); Pseudomonas aeruginosa FepA (gi:15597884); Pseudomonas aeruginosa PhuR (gi:15599904); Sinorhizobium meliloti ShmR (gi:470189354); Y. pseudotuberculosis HasR (gi:169752827); Beauveria bassiana TonB-dependent heme receptor A (gi:701779958); Haemophilus influenzae hemin receptor (gi:16272087); Y. pseudotuberculosis HemR (gi:170026102); E. coli heme utilization/transport protein (gi:15833634); Helicobacter pylori FrpB4 (gi:15646121); Ancylostoma ceylanicum TonB-dependent receptor (gi:510848345); Shewanella frigidimarina TonB-dependent receptor (gi:114335087); and Riemerella anatipestifer TonB-dependent receptor (gi:312446409).
To test the role of BTH_I1598 in Mn2+ transport, we measured the total metal content of bacterial cells using ICP-MS. Our results revealed that deletion of BTH_I1598 significantly reduced intracellular Mn2+ levels compared with those of the wild type, and complementation of the gene restored to wild-type levels (Fig. 4C). By contrast, deletion of the bth_I1598 gene had little effect on the accumulation of Fe and Zn ions (Fig. S5C). Consistent with its specific role in Mn2+ transport, the expression of BTH_I1598 was induced by low, but repressed by high, extracellular Mn2+ (Fig. 4D and Fig. S6B). Thus, we designated it Mn2+-specific outer membrane transporter (MnoT).
Interestingly, similar to the T6SS-4 genes, the expression of mnoT was repressed by OxyR in our transcriptomic analysis (Dataset S1), and the repression by OxyR was confirmed by qRT-PCR analysis (Fig. 4E). The expression of mnoT was also induced by CHP (Fig. 4D). Thus, MnoT might be involved in the oxidative stress response in B. thailandensis. Indeed, the ΔmnoT mutant exhibited higher sensitivity to CHP than the wild-type and the complemented strain (Fig. 4F). Importantly, although exogenous Mn2+ (0.25 μM) enhanced the survival rate of the wild-type and complemented strain upon CHP challenge, the effect of exogenous Mn2+ was largely abolished in the ΔmnoT mutant (Fig. 4F). Similar results were obtained by comparing the growth of the B. thailandensis wild-type, the ΔmnoT mutant, and the complemented strain in the presence of EDTA under CHP stress (Fig. S2 C and D). These results suggest that MnoT is an outer membrane active Mn2+ transporter and important in oxidative stress resistance.
The Mn2+ Transport Activity of T6SS is MnoT Dependent.
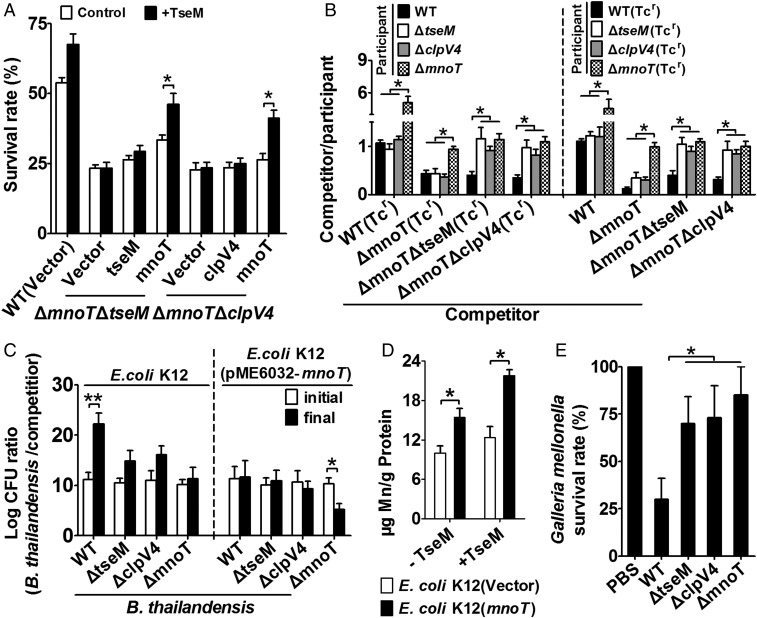
To investigate whether MnoT mediates the Mn2+ transport activity of TseM and T6SS-4, we constructed ΔmnoTΔtseM and ΔmnoTΔclpV4 double mutants. Both mutants showed highly reduced survival under CHP challenge compared with the wild type (Fig. 5A). Complementation of the tseM gene completely rescued the sensitivity of the ΔtseM mutant to CHP (Fig. 3D) but had marginal effect on rescue of the sensitivity of the ΔmnoTΔtseM double mutant (Fig. 5A). Moreover, although exogenous addition of metal-free apo-TseM (1 μM) significantly increased the survival of both the wild type and the ΔtseM mutant under CHP challenge (Fig. 3H), this effect was completely abolished in the ΔmnoTΔtseM(vector) and ΔmnoTΔtseM(tseM) strains (Fig. 5A). However, exogenous TseM protein efficiently protected the ΔmnoTΔtseM mutant from CHP toxicity when the mnoT gene was complemented (Fig. 5A). Similarly, complementation of clpV4 failed to rescue the sensitivity of the ΔmnoTΔclpV4 double mutant to CHP, and exogenous apo-TseM protected ΔmnoTΔclpV4(mnoT) but not ΔmnoTΔclpV4(vector) or ΔmnoTΔclpV4(clpV4) from CHP toxicity (Fig. 5A). These results demonstrate that the antioxidant activity and related Mn2+ transport activity of T6SS-4 and TseM are mediated by MnoT.
Fig. 5.
The Mn2+ transport activity of TseM is dependent on MnoT. (A) The indicated bacterial strains grown to the stationary phase were exposed to CHP (0.25 mM) for 40 min in M9 medium containing 25 nM Mn2+, and the viability of the cells was determined. (B) Intrabacterial growth competition assays between the indicated competitor strains (x-axis) following incubation with participant strains at 37 °C for 12 h in M9 medium containing 20 μM CHP. The competitive index result is calculated as the final cfu ratio (competitor/participant) divided by the initial ratio. (C) Interbacterial growth competition assays between B. thailandensis and E. coli K12. Quantification of cfu before (initial) and after (final) growth competition assays between the indicated organisms. The cfu ratio of the relevant B. thailandensis strains versus the competitors is plotted. (D) MnoT enhanced Mn2+ accumulation in E. coli K12. Stationary phase E. coli K12(pME6032) and E. coli K12(pME6032-mnoT) strains were cultivated in M9 medium containing 50 μM CHP, 25 nM Mn2+, with (+) or without (−) 1 μM TseM, and intracellular Mn2+ was measured by ICP-MS. (E) Virulence survival of relative B. thailandensis strains in G. mellonella larvae. Ordinate represented the mean percentage survival rate of G. mellonella infected with different strains after 16 h. Error bars represent the SD from three independent experiments. **P ≤ 0.01; *P ≤ 0.05.
To reveal more about the mechanisms of Mn2+ transport mediated by MnoT and T6SS-4, we performed intrabacterial growth competition assays between different B. thailandensis strains under CHP challenge in liquid M9 medium containing 25 nM Mn2+. As shown in Fig. 5B, the wild type displayed a growth advantage when competing with ΔmnoT but not ΔtseM and ΔclpV4. Indeed, the ΔmnoT mutant displayed a 2.5- to 3.5-fold growth disadvantage when competing with not only the wild type, but also the ΔtseM and ΔclpV4 mutants. However, introducing the ΔmnoT mutation to the ΔtseM or the ΔclpV4 mutants abolished their competitive advantage over the ΔmnoT mutant. This result suggests that MnoT can indiscriminately transport Mn2+-bound TseM secreted by itself or other neighboring cells.
The importance of MnoT in mediation of the Mn2+ transport activity of T6SS-4 was further confirmed by interbacterial growth competition assays between B. thailandensis strains and Escherichia coli K12. Phylogenetic analysis shows that, whereas MnoT homologs are widely distributed in Burkholderia species, it is absent in E. coli K12 (Fig. S8). As shown in Fig. 5C, the B. thailandensis wild type was highly competitive against the E. coli K12 competitor under 50 μM CHP challenge in liquid M9 medium containing 25 nM Mn2+. However, the competitive advantage of B. thailandensis wild type over E. coli K12 was largely abolished in the ΔtseM, ΔclpV4, and ΔmnoT mutants (Fig. 5C). As a TonB-dependent outer membrane transporter, we reasoned that heterologous expression of MnoT in E. coli K12 will improve its capability in Mn2+ acquisition and growth fitness. As expected, expression of MnoT increased intracellular Mn2+ accumulation in E. coli K12 under oxidative stress condition especially in the presence of exogenously provided TseM protein (Fig. 5D). Moreover, expression of MnoT abrogated the competition advantage of the B. thailandensis wild type to the E. coli K12 competitor (Fig. 5C). In addition, whereas the E. coli K12(pME6032-mnoT) competitor showed equivalent competitive ability to the B. thailandensis ΔtseM and ΔclpV4 mutants, it outcompeted the B. thailandensis ΔmnoT mutant (Fig. 5C). Altogether, these data indicate the essential role of MnoT for the T6SS-4–dependent Mn2+ uptake that confers a contact-independent competitive advantage.
Fig. S8.
MnoT homologs are widely distributed in Burkholderia species. The phylogenetic tree generated by the minimum-evolution algorithm in MEGA 6.0 illustrates that MnoT is highly conserved among the vast majority of Burkholderia. The B. thailandensis E264 MnoT was indicated in red. The bar represents the genetic distance.
B. thailandensis Mutants Lacking clpV4, tseM, or mnoT Are Defective in Virulence in the Galleria mellonella Larvae Infection Model.
Mn2+ uptake systems have been reported to be required for full virulence of multiple bacterial pathogens (2, 7). To investigate the role of T6SS-4-dependent Mn2+ transport system in pathogenesis, G. mellonella (wax moth) larvae were infected with different B. thailandensis strains to determine their abilities to kill the larvae. The G. mellonella larvae infection model has been widely used to assess the virulence of various Gram-negative and Gram-positive bacterial species (45, 46). Infection with B. thailandensis wild type exhibited the lowest survival rate at an average of 27%, whereas challenge with mutants lacking clpV4, tseM, or mnoT resulted in the mean survival rates of ∼70%, 73%, and 85% of larvae, respectively (Fig. 5E). These data suggest that the T6SS-4–dependent Mn2+ transport system is important for virulence.
Discussion
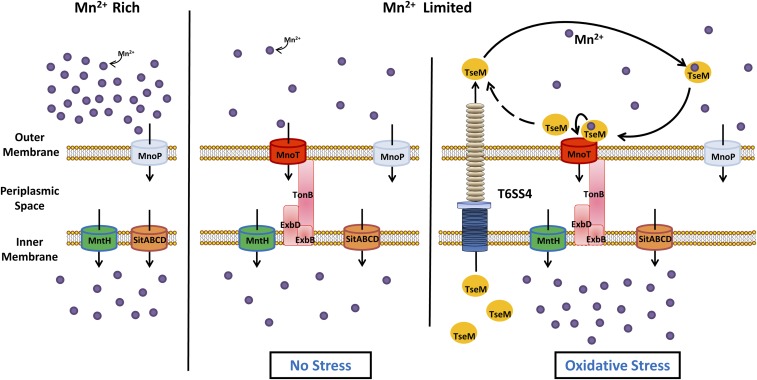
Although metal ions can traverse across the outer membrane via passive diffusion, Gram-negative bacteria must effectively concentrate scarce metal ions into the cytosol via active transport systems to meet cellular demands (47). To date, no active transporter for the translocation of Mn2+ across the outer membrane has been described. Here we propose a model of active transport of Mn2+ across the outer membrane mediated by the TonB-dependent outer membrane transporter MnoT and the T6SS effector TseM. Under high Mn2+ conditions, passive diffusion of Mn2+ through porins such as MnoP followed by transport across the inner membrane via either MntH or SitABCD fulfills cellular Mn2+ requirements. Low Mn2+ triggers the induction of the TonB-dependent outer membrane transporter MnoT for the active transport of Mn2+ across the outer membrane. When the bacterium encounters an oxidative stress challenge, T6SS-4 is induced to secrete the TseM manganeseophore into the extracellular milieu. Secreted TseM scavenges extracellular Mn2+ and delivers its Mn2+ load via direct interaction with MnoT. This active transport of Mn2+, mediated by TseM and MnoT, fulfills the increased cellular demand for Mn2+ under oxidative stress (Fig. 6). This T6SS-mediated Mn2+ uptake model not only expands our current understanding of the diverse T6SS functions but also provides insights for the interaction between specialized protein secretion systems and metal acquisition.
Fig. 6.
Model of T6SS-4–mediated Mn2+ transport and oxidative resistance in B. thailandensis.
Several lines of evidence indicate that MnoT functions in Mn2+ uptake. First, the ΔmnoT mutant was strongly deficient in Mn2+ accumulation under Mn2+ limitation (Fig. 4C). Next, MnoT was necessary to support bacterial growth under Mn2+-limited and oxidative stress conditions (Fig. S2 C and D). Moreover, deletion of mnoT abolished the protective effect of exogenous Mn2+ on oxidative stress resistance (Fig. 4F). Finally, as a Mn2+ transporter, the expression of mnoT was induced by low concentrations but repressed by high concentrations of Mn2+ (Fig. 4D and Fig. S6B). Consistent with its role in oxidative stress resistance, the expression of mnoT was also controlled by OxyR (Fig. 4E). These results indicate that mnoT encodes a novel Mn2+ uptake system that facilitates the uptake of Mn2+ at low concentrations and plays a crucial role in resistance to oxidative stress. To the best of our knowledge, an active transporter for translocation of Mn2+ across the outer membrane has not been previously described.
Most TBDTs studied to date are involved in the acquisition of iron by means of siderophore substrates (42, 48, 49). The siderophore substrates range in complexity from simple small molecules such as citrates to large proteins such as hemophores (48, 50). Bacterial hemophores are secreted proteins that scavenge heme in the external medium and bring it back to their specific outer membrane receptors (51, 52). Although proteinaceous metallophores such as nickelophore and zincophore have been proposed to be involved in TBDT-mediated nickel and zinc transport, none of these has been experimentally verified (43, 44). In the present study, we provide the following evidence that TseM facilitates MnoT-mediated Mn2+ transport under oxidative stress by acting as a manganeseophore: (i) recombinant TseM exhibited Mn2+ binding capacity (Fig. 3 B and C); (ii) the tseM mutant was strongly deficient in Mn2+ accumulation under oxidative stress conditions (Fig. 3F); (iii) deletion of tseM dramatically abolished the effect of exogenous Mn2+ on oxidative stress resistance (Fig. 3 D and E); (iv) the expression of tseM was repressed by high concentrations of Mn2+ (Fig. 3G); (v) TseM directly interacts with MnoT (Fig. 4 A and B); and (vi) the function of TseM on Mn2+ transport and oxidative stress resistance is highly dependent on the presence of MnoT (Fig. 5 A–D). We thus tentatively suggest the term “manganeseophore” for secreted Mn2+ binding proteins, which sequester this metal from the environment and interact with the outer membrane transporter for improved Mn2+ uptake.
We present TseM as a description of a proteinaceous metallophore other than hemophores for scavenging heme iron. Indeed, the TseM–MnoT manganese transport system shares some functional similarities with the bacterial HasA hemophore system. In the HasA system, the heme-binding protein HasA secreted by the type I secretion system scavenges heme from the host and presents it to a specific TBDT outer membrane transporter, HasR, whereby it is internalized by a TonB-dependent process (51, 52). Similarly, the T6SS secreted TseM scavenges manganese in the extracellular medium and reassociates with the TBDT transporter, MnoT, to facilitate bacterial manganese acquisition. Whether such metallophore–TBDT systems exist for the transport of other metal ions such as zinc and nickel require further investigation.
Together with the report that T6SS is involved in Zn2+ uptake in Y. pseudotuberculosis (32), our finding of the involvement of T6SS in Mn2+ uptake significantly expands the range of known functions of this specialized protein secretion system. Recently, Chen et al. (53) reported that the T6SS in Pseudomonas taiwanensis is involved in the secretion of iron chelator pyoverdine. Although the secretion mechanism of pyoverdine remains unidentified, this study implies the role of T6SS in iron uptake. Moreover, previous reports have shown that the expression of some T6SSs was regulated by iron and zinc in B. mallei and B. pseudomallei (35), and by the ferric uptake regulator Fur in Pseudomonas aeruginosa (54), further supporting the role of T6SS in metal ion acquisition. Therefore, we postulate that the “metal transporting” T6SSs may represent a novel type of T6SS that secretes metal scavenging proteins into the extracellular milieu for transport of various metal ions.
Antibacterial activities of the T6SS can induce ROS generation in target cells, which contribute to target cell death (55). In contrast, the metal transporting T6SSs can reduce intracellular ROS levels by taking up antioxidative Mn2+ (Fig. 2) and Zn2+ (32). Indeed, the B. thailandensis T6SS-4 reported here conferred bacteria a contact-independent competitive advantage in both interspecies and intraspecies bacterial competition (Fig. 5 B and C) through transporting Mn2+ under oxidative stress conditions, which is distinct from the contact-dependent competitive advantage conferred by those well-described antibacterial T6SSs (22, 24, 56). Thus, T6SSs play crucial roles in shaping the composition of a microbial population in the host or environmental niche, either by direct killing of competing cells via contact-dependent translocation of toxins, or by competition for essential nutrients via contact-independent secretion of proteinaceous metallophore. It will be interesting to investigate whether the metal transporting T6SS can puncture and inject toxins into target cells, or whether somehow this system has lost any puncturing ability of target cells and has been redesigned exclusively for extracellular release.
Because manganese is an essential micronutrient for bacterial growth, bacterial pathogens have evolved efficient mechanisms to acquire Mn2+ from the nutrient-restricted host environment to cause disease (2, 7). Indeed, Mn2+ uptake systems were reported to be required for full virulence of multiple bacterial pathogens such as Yersinia pestis (57), Y. pseudotuberculosis (13), Streptococcus mutans (58), Salmonella enterica serovar Typhimurium (59), Borrelia burgdorferi (60), and Brucella abortus (61) in different animal models. Consistently, we found the B. thailandensis ΔmnoT, ΔtseM, and ΔclpV4 mutants were attenuated in virulence in the G. mellonella larvae infection model (Fig. 5E), suggesting the importance of the T6SS-4–mediated Mn2+ transport system in the resistance to host nutritional immunity. Similar results were reported for the T6SS involved in Zn2+ uptake in Y. pseudotuberculosis in a mouse infection model (32). Thus, the metal transporting T6SSs may represent a promising target for therapeutic development of new antimicrobials.
Materials and Methods
Bacterial Strains and Growth Conditions.
Bacterial strains, plasmids, and primers used in this study are listed in Dataset S2. E. coli and B. thailandensis strains were cultured in LB aerobically on a rotary shaker (220 rpm) or on LB plates at 37 °C. The B. thailandensis E264 strain was the parent of all derivatives used in this study (62). M9 medium containing different levels of Mn2+ was used for survival assay and bacterial competition assay.
Protein Secretion Assay.
Secretion assays for TseM (BTH_II1883) were performed according to described methods (63). Briefly, strains were inoculated into 200 mL LB and incubated with continuous shaking until OD600 reached 1.6 at 37 °C. A 2-mL culture was centrifuged and the cell pellet was resuspended in 100 μL SDS-loading buffer; this whole-cell lysate sample was defined as TseMIN. A total of 180 mL of the culture was centrifuged, and the supernatant was filtered through a 0.22-μm filter (Millipore) and secreted proteins in the supernatant were collected by filtration over a nitrocellulose filter (BA85) (Whatman) three times. The filter was soaked in 100 μL SDS sample buffer for 15 min at 65 °C to recover the proteins present, and the sample was defined as TseMOUT. All samples were normalized to the OD600 of the culture and volume used in preparation.
Detailed protocols for these experiments and additional procedures are described in detail in SI Materials and Methods.
SI Materials and Methods
Antibiotics and Chemicals.
All chemicals were of Analytical Reagent Grade purity or higher. Antibiotics were added at the following concentrations: Chloramphenicol, 34 μg⋅ml−1 for E. coli and 50 μg⋅ml−1 for B. thailandensis; kanamycin, 50 μg⋅ml−1 for E. coli; streptomycin, 100 μg⋅ml−1 for B. thailandensis; ampicillin, 100 μg⋅ml−1 for E. coli; tetracycline, 25 μg⋅ml−1 for E. coli, and 50 μg⋅ml−1 for B. thailandensis.
Plasmid Construction.
For obtaining expression plasmids, the genes encoding B. thailandensis TseM (BTH_II1883), OxyR (BTH_I1281), and MnoT (BTH_I1598) were amplified by PCR. The obtained DNA fragments were digested and cloned into similar digested pGEX6p-1, pET28a, and pET15b, yielding corresponding plasmid derivatives. To prepare the ΔclpV4 in-frame deletion mutant, the suicide plasmid pDM4-pheS (62) was used to construct pDM4-pheS-ΔclpV4 (BTH_II1895). Briefly, the 819-bp upstream fragment and the 830-bp downstream fragment of clpV4 were amplified with primer pairs DclpV4-F1/DclpV4-R1 and DclpV4-F2/DclpV4-R2, respectively. The upstream and downstream PCR fragments were fused together with the primer pair DclpV4-F1//DclpV4-R2 by overlap PCR (23). The resulting PCR products were digested with SpeI and BglII and inserted into similar digested pDM4-pheS to create pDM4-pheS-ΔclpV4. The knockout plasmids pDM4-pheS-ΔicmF4 (BTH_II1885), pDM4-pheS-Δhcp4 (BTH_II1899), pDM4-pheS-ΔclpV1 (BTH_I2958), pDM4-pheS-ΔclpV2 (BTH_II0140), pDM4-pheS-ΔclpV6 (BTH_II0264), pDM4-pheS-ΔoxyR (BTH_I1281), pDM4-pheS-ΔkatG (BTH_I1282), pDM4-pheS-ΔahpC (BTH_I2092), pDM4-pheS-ΔmnoT (BTH_I1598), pDM4-pheS-ΔvgrG4a4b (BTH_ II1893-1894), and pDM4-pheS-ΔtseM (BTH_II1883) were constructed in similar manners by using primers listed in Dataset S2.
To complement the clpV4 mutant, primers clpV4-F/clpV4-R were used to amplify the clpV4 gene fragment from B. thailandensis genomic DNA. The amplified DNA fragments were digested and then cloned into similar digested pME6032 plasmid, obtaining plasmid pME6032-clpV4. The complementary plasmids pME6032-oxyR, pME6032-mnoT, pME6032-tseM, pME6032-vgrG4a, pME6032-vgrG4b, pME6032-icmF4, pME6032-clpV1, pME6032-clpV2, pME6032-clpV6, pME6032-aphC, and pME6032-katG were constructed in similar manners as described above with primers listed in Dataset S2.
Site-directed mutagenesis was carried out by overlap PCR to substitute the histidine residue at position 63 of TseM into an alanine (TseMH63A). The tseMH63A DNA fragment was obtained by two rounds of PCR. Primer pairs tseM-F1/tseMH63A-R and tseMH63A-F/tseM-R1 were used to amplify segments 1 and 2, respectively. The second round of PCR was carried out by using tseM-F1/tseM-R1 as primer pair, whereas segment 1 and segment 2 together were used as templates to obtain the tseMH63A fragment. The tseMH63A DNA fragment was digested by BamHI/XhoI and cloned into similar digested pGEX6p-1 to produce pGEX6p-1-tseMH63A. The primer pairs pGEX6p-1-F/tseMN132R-R and tseMN132R-F/pGEX6p-1-R were used to amplify the tseMH63A/N132R fragments by using the pGEX6p-1-tseMH63A DNA as template in the first round PCR, and similarly cloned into pGEX6p-1 to produce pGEX6p-1-tseMH63A/N132R by using tseM-F1/tseM-R1 primers in the second round PCR. Next, the pGEX6p-1-tseMH63A/N132R DNA served as template to amplify the tseMQ35R/H63A/N132R fragments with primer pairs pGEX6p-1-F/tseMQ35R-R and tseMQ35R-F/pGEX6p-1-R, and tseM-F1/tseM-R1 or tseZ-F3/tseZ-R3 primer pairs were used in the second round PCR to produce the pGEX6p-1-tseMQ35R/H63A/N132R or pME6032-tseMQ35R/H63A/N132R plasmid, respectively. All constructs were validated by DNA sequencing.
In-Frame Deletion and Complementation in B. thailandensis.
For constructing in-frame deletion mutants, the pDM4-pheS derivatives were transformed into relevant B. thailandensis strains through E. coli SM10(λpir)-mediated conjugational mating to carry out single crossover. The transconjugants were selected on LB agar medium containing chloramphenicol and streptomycin. Counter selection for markerless in-frame deletion was performed on M9 minimal medium agar plates with 0.4% glucose as a carbon source and 0.1% (wt/vol) p-chlorophenylalanine (62). For complementation, the pME6032 derivatives were transformed into relevant B. thailandensis strains by electroporation and the expression in B. thailandensis was induced by addition of 1 mM isopropyl β-d-1-thiogalactopyranoside (IPTG).
Overexpression and Purification of Recombinant Protein.
To express and purify soluble GST- and His6-tagged recombinant proteins, the pGEX6p-1, pET28a, and pET15b derivatives were transformed into E. coli XL1Blue, BL21(DE3), and transB(DE3) host strains, respectively. Bacteria were cultured at 37 °C in LB medium to an OD600 of 0.5, shifted to 22 °C, induced with 0.5 mM IPTG, and then cultivated for an additional 12 h at 22 °C. Harvested cells were sonicated and proteins were purified with the His-Bind Ni-NTA resin or the GST-Bind resin (Novagen) according to manufacturer’s instructions. Eluted recombinant proteins were dialyzed against PBS at 4 °C. Cleavage of the His6 tag was performed by adding 10 units of Enterokinase-Max (Invitrogen) and incubation at 22 °C overnight. Ni-NTA agarose was used to remove the cleaved tag and uncleaved protein from the tag-free protein.
For purification of the insoluble recombinant MnoT, E. coli BL21(DE3) containing pET15b-mnoT was grown in LB to an optical density at 600 nm of 0.6 after which 1 mM IPTG was added and growth was continued for 8 h at 26 °C. Recombinant MnoT accumulated in inclusion bodies were isolated as described (43). Briefly, the inclusion bodies were dissolved in 20 mM Tris⋅HCl, 100 mM glycine, 6 M urea (pH 8.3), and residual membranes were removed by centrifugation for 1 h at 200,000 × g. The protein was then refolded into its native conformation by diluting this stock solution 20-fold in refolding buffer containing 55 mM Tris⋅HCl, 0.21 mM sodium chloride, 0.88 mM potassium chloride, 880 mM l-arginine, and 0.5% 3-dimethyldodecylammoniopropane-sulfonate (SB-12) (Fluka), pH 7.0. After refolding overnight, the sample was dialyzed with 55 mM Tris⋅HCl (pH 6.5) containing 0.21 mM sodium chloride, 10 mM l-arginine, and 0.5% SB-12. The protein solution was filtered and stored at 4 °C. Proper folding was monitored by semi-native SDS/PAGE where the folded protein has a higher electrophoretic mobility than the denatured protein. The resulting proteins were stored at −80 °C until use. Protein concentrations were determined using the Bradford assay according to the manufacturer’s instructions (Bio-Rad) with BSA as standard.
Fluorescence Dye-Based Intracellular ROS Detection.
To detect intracellular ROS, the fluorescent reporter dye 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA) (Invitrogen) was used as previously described (32). Briefly, 1-mL samples were collected, washed with PBS, and then resuspended in 1 mL of PBS containing 10 μM H2DCFDA. Samples were incubated in the dark for 20 min at 28 °C. The cells were then pelleted, the supernatant was removed, and they were resuspended in 1 mL M9 medium with 0.4% glucose containing 0.25 mM CHP. After a 30-min treatment at 37 °C, the cells were pelleted, washed with PBS, resuspended in 1 mL of PBS, and then 200 μL of the resultant cell suspension was transferred to a dark 96-well plate. Fluorescence signals were measured using a SpectraMax M2 Plate Reader (Molecular Devices) with excitation/emission wavelengths of 495/520 nm. The results shown represented the mean of one representative assay performed in triplicate, and error bars represent SD. Statistical analysis was carried out with Student’s t test.
Determination of Intracellular Ion Content.
Intracellular ion content was determined as described previously (5, 13). Briefly, cells were grown in LB until stationary phase. After 20-mL culture solutions were collected and washed with PBS two times, the pellets were resuspended in 20 mL PBS buffer containing 0.4% glucose, 0.25 mM CHP, and 0.25 μM Mn2+, and then incubated further for 20 min. These cultures were centrifuged at 1,575 × g for 10 min. The wet cell pellet weight was measured and bacteria were chemically lysed using Bugbuster (Novagen) according to the manufacturer’s instructions. Bacteria were resuspended in Bugbuster solution by pipetting and incubation on a rotating mixer at a slow setting for 10 h. Total protein for each sample was measured by using NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies) according to the manufacturer’s instructions. Each sample was diluted 100-fold in 2% molecular grade nitric acid to a total volume of 5 mL. Samples were analyzed by inductively coupled plasma mass spectrometry (ICP-MS) (Varian 802-MS), and the results were corrected using the appropriate buffers for reference and dilution factors. Triplicate cultures of each strain were analyzed during a single experiment and the experiment was repeated at least three times.
Sensitivity Assays.
Stationary phase B. thailandensis strains grown in LB medium were collected, washed, and diluted 30-fold into M9 medium containing 25 nM Mn2+ (or as indicated), and treated with CHP (0.25 mM), H2O2 (1.0 mM), CdCl2 (0.1 mM), or diamide (0.35 mM), respectively, at 37 °C for 40 min. After treatment, the cultures were serially diluted and plated onto LB agar plates, and colonies were counted after 36-h growth at 37 °C. Percentage survival was calculated by dividing number of cfu of stressed cells by number of cfu of cells without stress (32). All these assays were performed in triplicate at least three times.
Quantitative Real-Time PCR.
Bacteria were harvested during the midexponential phase and RNA was extracted using the RNAprep Pure Cell/Bacteria Kit and treated with RNase-free DNase (Tiangen). The purity and concentration of the RNA were determined by gel electrophoresis and spectrophotometer (NanoDrop, Thermo Scientific). First-strand cDNA was reverse transcribed from 1 µg of total RNA with the TransScript First-Strand cDNA Synthesis SuperMix (TransGen Biotech). Quantitative real-time PCR (qRT-PCR) was performed in CFX96 Real-Time PCR Detection System (Bio-Rad) with TransStart Green qPCR SuperMix (TransGen Biotech). For all primer sets (Dataset S2), the following cycling parameters were used: 95 °C for 30 s followed by 40 cycles of 94 °C for 15 s and 50 °C for 30 s. For standardization of results, the relative abundance of 16S rRNA was used as the internal standard.
Western Blot Analysis.
Western blot analysis was performed as previously described (23). Samples were resolved by SDS/PAGE and transferred onto PVDF membranes (Millipore). The membrane was blocked in 5% (wt/vol) nonfat milk powder for 4 h at room temperature and incubated with primary antibodies at 4 °C overnight: anti-TseM (BTH_II1883) rabbit polyclonal antibody, 1:1,000; anti-ICDH, 1:6,000; anti-His (Millipore), 1:1,000; and anti-GST (Zhongshan Gold Bridge), 1:1,000. The membrane was washed three times in TBST buffer (50 mM Tris, 150 mM NaCl, 0.05% Tween 20, pH 7.4) and incubated with 1:5,000 dilution of horseradish peroxidase-conjugated secondary antibodies (Shanghai Genomics) for 1 h. Signals were detected using the ECL Plus Kit (GE Healthcare) following the manufacturer's specified protocol. The ICDH antisera were made in our previous studies (64). The purified His-tagged TseM and Hcp4 were used to generate rabbit anti-TseM and anti-Hcp4 polyclonal antibodies and the resulting antiserum was affinity purified against the same proteins.
EMSA.
Electrophoretic mobility shift assay was performed using biotin 5′-end-labeled promoter probes. Bio-PT6SS-4 was amplified from B. thailandensis genomic DNA with primers T6p-oxyR-F-5′ biotin/T6p-oxyR-R-5′ biotin. The unlabeled PT6SS-4 competitor DNA was amplified from B. thailandensis genomic DNA with primers T6p-oxyR-F/T6p-oxyR-R. All PCR fragments were purified by EasyPure Quick Gel Extraction Kit (TransGen Biotech). Each 20-µL EMSA reaction solution was prepared by adding the following components according to the manufacturer’s protocol (LightShift Chemiluminescent EMSA Kit; Thermo Fisher Scientific): 1× binding buffer, 50 ng poly (dI-dC), 2.5% glycerol, 0.05% Nonidet P-40, 5 mM MgCl2, 3 ng biotin-DNA, 1 ng unlabeled DNA as competitor, and different concentrations of proteins. Reaction solutions were incubated for 20 min at room temperature. The protein-probes mixture was separated in a 6% polyacrylamide native gel and transferred to a Biodyne B nylon membrane (Thermo Fisher Scientific). Migration of biotin-labeled probes was detected by streptavidin-horseradish peroxidase conjugates that bind to biotin and chemiluminescent substrate according to the manufacturer’s protocol.
G. mellonella Infection Model.
The G. mellonella larvae infection model was used to evaluate the virulence of B. thailandensis mutants as described (45, 46). G. mellonella were purchased from Livefood JiaYing Ltd (TianJin) and maintained in the dark at 25 °C until use. Bacteria grown to OD600 of 1.6 in LB at 37 °C were harvested, washed, and resuspended in phosphate-buffered saline (PBS) to give a final concentration of 105 bacteria. Twenty G. mellonella larvae were injected with a 50-μL dose of 105 cfu in the secondary right proleg using a Hamilton H syringe and then incubated statically at 37 °C for 16 h before determining survival rates. A total of 50 μL PBS was injected as control to measure any potential lethal effects of the injection process, or larvae were not injected to measure the effects of the incubation procedure. Each experiment was performed in triplicate.
Metal-Free apo-TseM Preparation and Metal Ion Binding Assays.
Metal-free apo-TseM was prepared as described previously (65). Briefly, proteins were dialyzed overnight at 4 °C against 250 μM EDTA and 5 mM o-phenanthroline in 50 mM Hepes (pH 8.0), 150 mM NaCl, and 10% (vol/vol) glycerol, followed by three dialysis steps in 50 mM Hepes (pH 8.0), 150 mM NaCl, and 10% (vol/vol) glycerol to remove EDTA and o-phenanthroline.
Mn2+ binding was measured using isothermal titration calorimetry (ITC) at 25 °C with a NANO-ITC 2G microcalorimeter (TA Instruments) (66). The 1 mM MnSO4 solution used for titration was prepared with the apo-TseM dialysis buffer. The protein concentration in the sample solution was 50 μM. All protein and metal solutions were extensively degassed before titration. After a stable baseline had been achieved, the MnSO4 titration was performed with a total of 25 injections of 5 μL into the protein solutions (volume = 1.5 mL) until the protein sample was saturated with Mn2+. A control experiment in the absence of protein was performed to measure the heat generated due to Mn2+ dilution in the buffer. Blank titrations of the MnSO4 solution into the dialysis buffer were performed to correct for the dilution heat of the zinc solution. Data reduction and analyses were performed with the Nano Analyze software (TA Instruments), and an independent binding model was used. All ITC experiments were performed in triplicate.
Mn2+ binding was also detected using the metal reconstitution assay as previously described (67). Briefly, for removing as much of the ions as possible, purified TseM protein (100 μM) was added to the solution containing 25 mM Tris, 25 mM diethylene triamine pentaacetic acid, and 10% glycerol at pH 7.5 and put on ice. After 1 h, the protein solution was dialyzed with buffer (25 mM Tris, 10% glycerol, pH 7.5) at 4 °C. For reconstitution with metal ions, the resulting TseM protein (10 μM) was added to 25 μM of the desired divalent-metal ions (Fe3+, Mn2+, Mg2+, Cd2+, and Zn2+) and put on ice for 30 min, with Milli-Q water for preparing ions solution as the control. These solutions were dialyzed again (25 mM Tris, 10% glycerol, pH 7.5) as mentioned above to remove unbound metal ions and the metal ions bound to the protein were analyzed using atomic absorption spectroscopy (ZEEnit 650P; Analytik Jena).
Zn2+ binding of proteins was detected using the Zn2+-binding dye 4-(2-pyridylazo)-resorcinol (PAR) as previously reported (68). An iron-binding assay was performed with ferrous sulfate as previously described (69).
Intrabacterial and Interbacterial Growth Competition Assays.
Intrabacterial competition assays were conducted as described previously with minor modifications (70). In brief, overnight-grown competitor and participant strains were washed with M9 medium before mixing for competition. The initial competitor–participant ratio was 1:1 (OD600 of 2.0 for each strain) and the bacteria were incubated for 12 h at 37 °C in liquid M9 medium containing 20 μM CHP and 25 nM Mn2+. The competitor strains contained pME6032, conferring tetracycline resistance for selection. After competition, the competitor and participant colonies were counted on LB plates supplemented with tetracycline and streptomycin or streptomycin alone, and changes in the competitor/participant ratios were determined. Data from all competitions were analyzed using the Student’s t test.
Interbacterial competition assays were conducted as described (22, 71) with minor modifications. Briefly, overnight cultures of relevant B. thailandensis (streptomycin resistance) and the E. coli K12 competitors containing pME6032 vector or pME6032-mnoT (tetracycline resistance) were washed three times with M9 medium, adjusted to OD600 of 1.6, and then mixed in 10:1 (vol/vol) of relevant B. thailandensis versus the competitor E. coli K12. To calculate the initial cfu ratio of relevant B. thailandensis and competitor, 100 μL of the mixture was taken out, serially diluted, spread on LB plates containing different antibiotic, and incubated at 37 °C for 36 h. For competition assays, CHP (final concentration 50 μM) was added to the above residual mixture (2 mL) and incubated at 37 °C 100 rpm. After 12 h, the mixture was serially diluted, spread on LB plates containing different antibiotics, and the final cfu ratio was determined.
GST Pull-Down Assay.
The GST pull-down assay was performed as previously described with minor modifications (64, 72). Briefly, 0.5 mg purified GST fusion protein was mixed with cleared cell lysates collected from CHP-treated B. thailandensis culture (200 mL) on a rotator for 3 h at 4 °C, and 100 μL prewashed glutathione beads were added to the reactions. After another 2 h of incubation at 4 °C, the beads were washed five times with PBS. Proteins associated with beads were treated with SDS sample buffer, resolved by SDS/PAGE, and visualized by silver staining (Bio-Rad). Individual protein gel bands were excised, digested with trypsin, and analyzed by matrix-assisted laser desorption/ionization/mass spectrometry (Voyager-DESTR, Applied Biosystems). To analyze protein interactions with purified proteins, purified GST-TseM was mixed with His6-MnoT in PBS on a rotator for 2 h at 4 °C, and GST was used as a negative control. After adding 40 μL of a prewashed glutathione beads slurry, binding was allowed to proceed for another 2 h at 4 °C. The beads were then washed five times with TEN buffer [100 mM Tris-Cl (pH 8.0), 10 mM EDTA, 500 mM NaCl]. Retained proteins were detected by immunoblot after SDS/PAGE using the anti-His antibody (Millipore).
Amino Acid Sequence Alignment and 3D Modeling Prediction.
Sequence alignment and database searches were carried out using BLAST programs at the BLAST server of the National Center for Biotechnology Information (NCBI) website (https://www.ncbi.nlm.nih.gov/) and visualized by using BioEdit (www.mbio.ncsu.edu/bioedit/bioedit.html). The construction of phylogenetic tree was made with MEGA 6.0 program. The 3D models of TseM were generated using Phyre2 (www.sbg.bio.ic.ac.uk/∼phyre2/html/page.cgi?id=index).
RNA-Seq Experiment.
Total RNA was extracted from B. thailandensis wild type and the ΔoxyR mutant (three biological replicates) grown in LB at 37 °C with shaking (220 rpm) to a final optical density of ∼1.6, using bacteria total RNA isolation kit (Tiangen). RNA degradation and contamination was monitored on 1% agarose gels; RNA purity was checked using the NanoPhotometer spectrophotometer (Implen) and RNA integrity was assessed using the Bioanalyzer 2100 system (Agilent Technologies). A total of 3 μg RNA per sample was used as input material in RNA sample preparations for subsequent cDNA library construction. All six samples had RIN values above 7.0. Sequencing libraries were generated using Illumina HiSeq 2000 RNA Sample Preparation Kit (Illumina) following manufacturer’s recommendations and four index codes were added to attribute sequences to each sample. Differential expression analysis was performed using the NOIseq method (Sonia Tarazona 2100). P values were adjusted using the Benjamini–Hochberg method. Corrected P value of 0.05 and log2 (fold change) of 0.8 were set as the threshold for significantly differential expression. Gene Ontology (GO) enrichment analysis of differentially expressed genes was implemented by the GOseq R package, in which gene length bias was corrected. GO terms with corrected P values of less than 0.05 were considered significantly enriched by differential expressed genes.
Statistical Analysis.
Statistical analyses of survival assay, intracellular ion content determination, ROS determination, and expression data were performed using paired two-tailed Student’s t test. Statistical analyses were performed using GraphPad Prism Software (GraphPad Software).
Supplementary Material
Acknowledgments
We thank Professor Zhao-Qing Luo (Purdue University) for valuable discussions and critical reading of the manuscript and Professor Feng Shao and Dr. Qiuhe Lu (National Institute of Biological Sciences, Beijing) for kindly providing strains and plasmids. This work was supported by the National Natural Science Foundation of China (Grants 31670053, 31370150, and 31500087) and Fundamental Research Funds for the Central Universities, Northwest A&F University (Grant 2452015100). T.G.D. was supported by a Government of Canada Research Chair award and Canadian Foundation for Innovation Grant CFI-JELF. B.B. was supported by an Alberta Innovates Health Solutions postdoctoral fellowship and a Canadian Natural Sciences and Engineering Research Council fellowship.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1614902114/-/DCSupplemental.
References
- 1.Aguirre JD, Culotta VC. Battles with iron: Manganese in oxidative stress protection. J Biol Chem. 2012;287(17):13541–13548. doi: 10.1074/jbc.R111.312181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lisher JP, Giedroc DP. Manganese acquisition and homeostasis at the host-pathogen interface. Front Cell Infect Microbiol. 2013;3:91. doi: 10.3389/fcimb.2013.00091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anjem A, Varghese S, Imlay JA. Manganese import is a key element of the OxyR response to hydrogen peroxide in Escherichia coli. Mol Microbiol. 2009;72(4):844–858. doi: 10.1111/j.1365-2958.2009.06699.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barnese K, Gralla EB, Valentine JS, Cabelli DE. Biologically relevant mechanism for catalytic superoxide removal by simple manganese compounds. Proc Natl Acad Sci USA. 2012;109(18):6892–6897. doi: 10.1073/pnas.1203051109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Puri S, Hohle TH, O’Brian MR. Control of bacterial iron homeostasis by manganese. Proc Natl Acad Sci USA. 2010;107(23):10691–10695. doi: 10.1073/pnas.1002342107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hood MI, Skaar EP. Nutritional immunity: Transition metals at the pathogen-host interface. Nat Rev Microbiol. 2012;10(8):525–537. doi: 10.1038/nrmicro2836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Juttukonda LJ, Skaar EP. Manganese homeostasis and utilization in pathogenic bacteria. Mol Microbiol. 2015;97(2):216–228. doi: 10.1111/mmi.13034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Diaz-Ochoa VE, et al. Salmonella mitigates oxidative stress and thrives in the inflamed gut by evading calprotectin-mediated manganese sequestration. Cell Host Microbe. 2016;19(6):814–825. doi: 10.1016/j.chom.2016.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kehres DG, Janakiraman A, Slauch JM, Maguire ME. SitABCD is the alkaline Mn(2+) transporter of Salmonella enterica serovar Typhimurium. J Bacteriol. 2002;184(12):3159–3166. doi: 10.1128/JB.184.12.3159-3166.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sabri M, et al. Contribution of the SitABCD, MntH, and FeoB metal transporters to the virulence of avian pathogenic Escherichia coli O78 strain χ7122. Infect Immun. 2008;76(2):601–611. doi: 10.1128/IAI.00789-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kehres DG, Zaharik ML, Finlay BB, Maguire ME. The NRAMP proteins of Salmonella typhimurium and Escherichia coli are selective manganese transporters involved in the response to reactive oxygen. Mol Microbiol. 2000;36(5):1085–1100. doi: 10.1046/j.1365-2958.2000.01922.x. [DOI] [PubMed] [Google Scholar]
- 12.Patzer SI, Hantke K. Dual repression by Fe(2+)-Fur and Mn(2+)-MntR of the mntH gene, encoding an NRAMP-like Mn(2+) transporter in Escherichia coli. J Bacteriol. 2001;183(16):4806–4813. doi: 10.1128/JB.183.16.4806-4813.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Champion OL, et al. Yersinia pseudotuberculosis mntH functions in intracellular manganese accumulation, which is essential for virulence and survival in cells expressing functional Nramp1. Microbiology. 2011;157(Pt 4):1115–1122. doi: 10.1099/mic.0.045807-0. [DOI] [PubMed] [Google Scholar]
- 14.Hohle TH, Franck WL, Stacey G, O’Brian MR. Bacterial outer membrane channel for divalent metal ion acquisition. Proc Natl Acad Sci USA. 2011;108(37):15390–15395. doi: 10.1073/pnas.1110137108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Durand E, Cambillau C, Cascales E, Journet L. VgrG, Tae, Tle, and beyond: The versatile arsenal of Type VI secretion effectors. Trends Microbiol. 2014;22(9):498–507. doi: 10.1016/j.tim.2014.06.004. [DOI] [PubMed] [Google Scholar]
- 16.Russell AB, Peterson SB, Mougous JD. Type VI secretion system effectors: Poisons with a purpose. Nat Rev Microbiol. 2014;12(2):137–148. doi: 10.1038/nrmicro3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ho BT, Dong TG, Mekalanos JJ. A view to a kill: The bacterial type VI secretion system. Cell Host Microbe. 2014;15(1):9–21. doi: 10.1016/j.chom.2013.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Basler M. Type VI secretion system: Secretion by a contractile nanomachine. Philos Trans R Soc Lond B Biol Sci. 2015;370(1679) doi: 10.1098/rstb.2015.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cascales E. The type VI secretion toolkit. EMBO Rep. 2008;9(8):735–741. doi: 10.1038/embor.2008.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pukatzki S, McAuley SB, Miyata ST. The type VI secretion system: Translocation of effectors and effector-domains. Curr Opin Microbiol. 2009;12(1):11–17. doi: 10.1016/j.mib.2008.11.010. [DOI] [PubMed] [Google Scholar]
- 21.Hood RD, et al. A type VI secretion system of Pseudomonas aeruginosa targets a toxin to bacteria. Cell Host Microbe. 2010;7(1):25–37. doi: 10.1016/j.chom.2009.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schwarz S, et al. Burkholderia type VI secretion systems have distinct roles in eukaryotic and bacterial cell interactions. PLoS Pathog. 2010;6(8):e1001068. doi: 10.1371/journal.ppat.1001068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang W, et al. A type VI secretion system regulated by OmpR in Yersinia pseudotuberculosis functions to maintain intracellular pH homeostasis. Environ Microbiol. 2013;15(2):557–569. doi: 10.1111/1462-2920.12005. [DOI] [PubMed] [Google Scholar]
- 24.Russell AB, et al. Type VI secretion delivers bacteriolytic effectors to target cells. Nature. 2011;475(7356):343–347. doi: 10.1038/nature10244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Russell AB, et al. Diverse type VI secretion phospholipases are functionally plastic antibacterial effectors. Nature. 2013;496(7446):508–512. doi: 10.1038/nature12074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dong TG, Ho BT, Yoder-Himes DR, Mekalanos JJ. Identification of T6SS-dependent effector and immunity proteins by Tn-seq in Vibrio cholerae. Proc Natl Acad Sci USA. 2013;110(7):2623–2628. doi: 10.1073/pnas.1222783110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ma LS, Hachani A, Lin JS, Filloux A, Lai EM. Agrobacterium tumefaciens deploys a superfamily of type VI secretion DNase effectors as weapons for interbacterial competition in planta. Cell Host Microbe. 2014;16(1):94–104. doi: 10.1016/j.chom.2014.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aubert DF, et al. A Burkholderia type VI rffector feamidates Rho GTPases to activate the pyrin inflammasome and trigger inflammation. Cell Host Microbe. 2016;19(5):664–674. doi: 10.1016/j.chom.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 29.Jiang F, et al. The Pseudomonas aeruginosa type VI secretion PGAP1-like effector induces host autophagy by activating endoplasmic reticulum stress. Cell Reports. 2016;16(6):1502–1509. doi: 10.1016/j.celrep.2016.07.012. [DOI] [PubMed] [Google Scholar]
- 30.Pukatzki S, Ma AT, Revel AT, Sturtevant D, Mekalanos JJ. Type VI secretion system translocates a phage tail spike-like protein into target cells where it cross-links actin. Proc Natl Acad Sci USA. 2007;104(39):15508–15513. doi: 10.1073/pnas.0706532104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hachani A, Wood TE, Filloux A. Type VI secretion and anti-host effectors. Curr Opin Microbiol. 2016;29:81–93. doi: 10.1016/j.mib.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 32.Wang T, et al. Type VI secretion system transports Zn2+ to combat multiple stresses and host immunity. PLoS Pathog. 2015;11(7):e1005020. doi: 10.1371/journal.ppat.1005020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haraga A, West TE, Brittnacher MJ, Skerrett SJ, Miller SI. Burkholderia thailandensis as a model system for the study of the virulence-associated type III secretion system of Burkholderia pseudomallei. Infect Immun. 2008;76(11):5402–5411. doi: 10.1128/IAI.00626-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garcia EC, Perault AI, Marlatt SA, Cotter PA. Interbacterial signaling via Burkholderia contact-dependent growth inhibition system proteins. Proc Natl Acad Sci USA. 2016;113(29):8296–8301. doi: 10.1073/pnas.1606323113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Burtnick MN, Brett PJ. Burkholderia mallei and Burkholderia pseudomallei cluster 1 type VI secretion system gene expression is negatively regulated by iron and zinc. PLoS One. 2013;8(10):e76767. doi: 10.1371/journal.pone.0076767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Loprasert S, Whangsuk W, Sallabhan R, Mongkolsuk S. Regulation of the katG-dpsA operon and the importance of KatG in survival of Burkholderia pseudomallei exposed to oxidative stress. FEBS Lett. 2003;542(1–3):17–21. doi: 10.1016/s0014-5793(03)00328-4. [DOI] [PubMed] [Google Scholar]
- 37.Tseng HJ, McEwan AG, Apicella MA, Jennings MP. OxyR acts as a repressor of catalase expression in Neisseria gonorrhoeae. Infect Immun. 2003;71(1):550–556. doi: 10.1128/IAI.71.1.550-556.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Teramoto H, Inui M, Yukawa H. OxyR acts as a transcriptional repressor of hydrogen peroxide-inducible antioxidant genes in Corynebacterium glutamicum R. FEBS J. 2013;280(14):3298–3312. doi: 10.1111/febs.12312. [DOI] [PubMed] [Google Scholar]
- 39.Hachani A, Allsopp LP, Oduko Y, Filloux A. The VgrG proteins are “à la carte” delivery systems for bacterial type VI effectors. J Biol Chem. 2014;289(25):17872–17884. doi: 10.1074/jbc.M114.563429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nikaido H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol Mol Biol Rev. 2003;67(4):593–656. doi: 10.1128/MMBR.67.4.593-656.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ferguson AD, Deisenhofer J. Metal import through microbial membranes. Cell. 2004;116(1):15–24. doi: 10.1016/s0092-8674(03)01030-4. [DOI] [PubMed] [Google Scholar]
- 42.Noinaj N, Guillier M, Barnard TJ, Buchanan SK. TonB-dependent transporters: Regulation, structure, and function. Annu Rev Microbiol. 2010;64:43–60. doi: 10.1146/annurev.micro.112408.134247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Stork M, et al. An outer membrane receptor of Neisseria meningitidis involved in zinc acquisition with vaccine potential. PLoS Pathog. 2010;6:e1000969. doi: 10.1371/journal.ppat.1000969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Schauer K, Gouget B, Carrière M, Labigne A, de Reuse H. Novel nickel transport mechanism across the bacterial outer membrane energized by the TonB/ExbB/ExbD machinery. Mol Microbiol. 2007;63(4):1054–1068. doi: 10.1111/j.1365-2958.2006.05578.x. [DOI] [PubMed] [Google Scholar]
- 45.Wand ME, Müller CM, Titball RW, Michell SL. Macrophage and Galleria mellonella infection models reflect the virulence of naturally occurring isolates of B. pseudomallei, B. thailandensis and B. oklahomensis. BMC Microbiol. 2011;11(1):11. doi: 10.1186/1471-2180-11-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ramarao N, Nielsen-Leroux C, Lereclus D. The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J Vis Exp. 2012;70(70):e4392. doi: 10.3791/4392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Finney LA, O’Halloran TV. Transition metal speciation in the cell: Insights from the chemistry of metal ion receptors. Science. 2003;300(5621):931–936. doi: 10.1126/science.1085049. [DOI] [PubMed] [Google Scholar]
- 48.Cassat JE, Skaar EP. Iron in infection and immunity. Cell Host Microbe. 2013;13(5):509–519. doi: 10.1016/j.chom.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Porcheron G, Garénaux A, Proulx J, Sabri M, Dozois CM. Iron, copper, zinc, and manganese transport and regulation in pathogenic Enterobacteria: correlations between strains, site of infection and the relative importance of the different metal transport systems for virulence. Front Cell Infect Microbiol. 2013;3:90. doi: 10.3389/fcimb.2013.00090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Maresso AW, Garufi G, Schneewind O. Bacillus anthracis secretes proteins that mediate heme acquisition from hemoglobin. PLoS Pathog. 2008;4(8):e1000132. doi: 10.1371/journal.ppat.1000132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Contreras H, Chim N, Credali A, Goulding CW. Heme uptake in bacterial pathogens. Curr Opin Chem Biol. 2014;19:34–41. doi: 10.1016/j.cbpa.2013.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cescau S, et al. Heme acquisition by hemophores. Biometals. 2007;20(3–4):603–613. doi: 10.1007/s10534-006-9050-y. [DOI] [PubMed] [Google Scholar]
- 53.Chen WJ, et al. Involvement of type VI secretion system in secretion of iron chelator pyoverdine in Pseudomonas taiwanensis. Sci Rep. 2016;6:32950. doi: 10.1038/srep32950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sana TG, et al. The second type VI secretion system of Pseudomonas aeruginosa strain PAO1 is regulated by quorum sensing and Fur and modulates internalization in epithelial cells. J Biol Chem. 2012;287(32):27095–27105. doi: 10.1074/jbc.M112.376368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dong TG, et al. Generation of reactive oxygen species by lethal attacks from competing microbes. Proc Natl Acad Sci USA. 2015;112(7):2181–2186. doi: 10.1073/pnas.1425007112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.MacIntyre DL, Miyata ST, Kitaoka M, Pukatzki S. The Vibrio cholerae type VI secretion system displays antimicrobial properties. Proc Natl Acad Sci USA. 2010;107(45):19520–19524. doi: 10.1073/pnas.1012931107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Perry RD, et al. Manganese transporters Yfe and MntH are Fur-regulated and important for the virulence of Yersinia pestis. Microbiology. 2012;158(Pt 3):804–815. doi: 10.1099/mic.0.053710-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Paik S, Brown A, Munro CL, Cornelissen CN, Kitten T. The sloABCR operon of Streptococcus mutans encodes an Mn and Fe transport system required for endocarditis virulence and its Mn-dependent repressor. J Bacteriol. 2003;185(20):5967–5975. doi: 10.1128/JB.185.20.5967-5975.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Boyer E, Bergevin I, Malo D, Gros P, Cellier MF. Acquisition of Mn(II) in addition to Fe(II) is required for full virulence of Salmonella enterica serovar Typhimurium. Infect Immun. 2002;70(11):6032–6042. doi: 10.1128/IAI.70.11.6032-6042.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ouyang Z, He M, Oman T, Yang XF, Norgard MV. A manganese transporter, BB0219 (BmtA), is required for virulence by the Lyme disease spirochete, Borrelia burgdorferi. Proc Natl Acad Sci USA. 2009;106(9):3449–3454. doi: 10.1073/pnas.0812999106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Anderson ES, et al. The manganese transporter MntH is a critical virulence determinant for Brucella abortus 2308 in experimentally infected mice. Infect Immun. 2009;77(8):3466–3474. doi: 10.1128/IAI.00444-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhao Y, Shao F. The NAIP-NLRC4 inflammasome in innate immune detection of bacterial flagellin and type III secretion apparatus. Immunol Rev. 2015;265(1):85–102. doi: 10.1111/imr.12293. [DOI] [PubMed] [Google Scholar]
- 63.Xu S, et al. FliS modulates FlgM activity by acting as a non-canonical chaperone to control late flagellar gene expression, motility and biofilm formation in Yersinia pseudotuberculosis. Environ Microbiol. 2014;16(4):1090–1104. doi: 10.1111/1462-2920.12222. [DOI] [PubMed] [Google Scholar]
- 64.Xu L, et al. Inhibition of host vacuolar H+-ATPase activity by a Legionella pneumophila effector. PLoS Pathog. 2010;6(3):e1000822. doi: 10.1371/journal.ppat.1000822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Vogel A, Schilling O, Niecke M, Bettmer J, Meyer-Klaucke W. ElaC encodes a novel binuclear zinc phosphodiesterase. J Biol Chem. 2002;277(32):29078–29085. doi: 10.1074/jbc.M112047200. [DOI] [PubMed] [Google Scholar]
- 66.Schilling O, et al. Zinc- and iron-dependent cytosolic metallo-beta-lactamase domain proteins exhibit similar zinc-binding affinities, independent of an atypical glutamate at the metal-binding site. Biochem J. 2005;385(Pt 1):145–153. doi: 10.1042/BJ20040773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huang X, Kocabas E, Hernick M. The activity and cofactor preferences of N-acetyl-1-D-myo-inosityl-2-amino-2-deoxy-α-D-glucopyranoside deacetylase (MshB) change depending on environmental conditions. J Biol Chem. 2011;286(23):20275–20282. doi: 10.1074/jbc.M111.234229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hunt JB, Neece SH, Schachman HK, Ginsburg A. Mercurial-promoted Zn2+ release from Escherichia coli aspartate transcarbamoylase. J Biol Chem. 1984;259(23):14793–14803. [PubMed] [Google Scholar]
- 69.Saraswathi R, Pait Chowdhury R, Williams SM, Ghatak P, Chatterji D. The mycobacterial MsDps2 protein is a nucleoid-forming DNA binding protein regulated by sigma factors sigma and sigma. PLoS One. 2009;4(11):e8017. doi: 10.1371/journal.pone.0008017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jiang F, Waterfield NR, Yang J, Yang G, Jin Q. A Pseudomonas aeruginosa type VI secretion phospholipase D effector targets both prokaryotic and eukaryotic cells. Cell Host Microbe. 2014;15(5):600–610. doi: 10.1016/j.chom.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 71.Russell AB, et al. A type VI secretion-related pathway in Bacteroidetes mediates interbacterial antagonism. Cell Host Microbe. 2014;16(2):227–236. doi: 10.1016/j.chom.2014.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hisabori T, et al. Thioredoxin affinity chromatography: A useful method for further understanding the thioredoxin network. J Exp Bot. 2005;56(416):1463–1468. doi: 10.1093/jxb/eri170. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.