Abstract
Opioids are frequently used for the treatment of pain following spinal cord injury (SCI). Unfortunately, we have shown that morphine administered in the acute phase of SCI results in significant, adverse secondary consequences including compromised locomotor and sensory recovery. Similarly, we showed that selective activation of the κ-opioid receptor (KOR), even at a dose 32-fold lower than morphine, is sufficient to attenuate recovery of locomotor function. In the current study, we tested whether activation of the KOR is necessary to produce morphine's adverse effects using nor-Binaltorphimine (norBNI), a selective KOR antagonist. Rats received a moderate spinal contusion (T12) and 24 h later, baseline locomotor function and nociceptive reactivity were assessed. Rats were then administered norBNI (0, 0.02, 0.08, or 0.32 μmol) followed by morphine (0 or 0.32 μmol). Nociception was reassessed 30 min after drug treatment, and recovery was evaluated for 21 days. The effects of norBNI on morphine-induced attenuation of recovery were dose dependent. At higher doses, norBNI blocked the adverse effects of morphine on locomotor recovery, but analgesia was also significantly decreased. Conversely, at low doses, analgesia was maintained, but the adverse effects on recovery persisted. A moderate dose of norBNI, however, adequately protected against morphine's adverse effects without eliminating its analgesic efficacy. This suggests that activation of the KOR system plays a significant role in the morphine-induced attenuation of recovery. Our research suggests that morphine, and other opioid analgesics, may be contraindicated for the SCI population. Blocking KOR activity may be a viable strategy for improving the safety of clinical opioid use.
Keywords: : locomotor function, receptors, recovery, sensory function, SCI
Introduction
Despite the clinical utility of opioids, our previous studies have shown that morphine administered in the acute phase of spinal cord injury (SCI) reduces recovery of locomotor function, while increasing weight loss, tissue loss, mortality, and nociceptive reactivity even weeks after treatment.1–5 As opioids are currently among the most effective analgesics for the treatment of pain in the acute phase of SCI, it is essential that we identify the mechanisms underlying these adverse side effects and improve the safety of these medications.
Our data suggest that the detrimental effects of morphine may be mediated by κ-opioid receptors (KORs).6 Although morphine is a prototypical opioid agonist that binds with high affinity to the μ-opioid receptor (MOR), it also binds with lower affinity to the δ-opioid receptor (DOR) and KORs, as well as to non-classic receptors, such as the toll-like receptors (TLRs).7,8 Using selective agonists for each opioid receptor subtype, we found that only administration of GR89696 (the KOR agonist) was sufficient to undermine recovery after SCI.6 These data concur with previous literature. For example, Faden and colleagues showed that dynorphin, the endogenous ligand of the KOR, and dynorphin-related peptides induce paraplegia even when administered to the intact spinal cord.9,10 Prodynorphin mRNA expression and dynorphin immunoreactivity are also significantly elevated in the spinal cord following trauma, and correlate with injury severity and neurological dysfunction.11–15 Importantly, administration of intrathecal dynorphin exacerbates functional deficits after SCI, whereas treatment with dynorphin antiserum improves outcome after trauma.16 These data suggest that the adverse effects of morphine following SCI may be mediated by the KOR system.
There is controversy, however, regarding the opioid-receptor mediated and non-opioid actions of dynorphin. Supporting a KOR-mediated mechanism, administration of the highly selective KOR-antagonist nor-Binaltorphimine (norBNI) limits the paralytic effects of dynorphin,16 and has been shown to improve functional outcomes in rodent models of spinal 16,17 and brain18 injury. Conversely, although showing decreased potency, administration of Dyn A-(2–17), a prodynorphin product inactive at opioid receptors, replicates dynorphin's adverse effects on locomotor function.16 It has been posited that these pathophysiological effects may result from N-methyl-d-aspartate (NMDA)-mediated excitotoxicity. NMDA antagonists limit dynorphin-induced behavioral effects.19–21 Moreover, dynorphin administration increases extracellular levels of excitatory amino acids, including glutamate and aspartate, through a non-opioid mechanism.22 These data suggest that whereas KOR activation may contribute to decreased locomotor function after SCI, endogenous and exogenous opioids may also engage alternative mechanisms that compromise recovery from SCI.
The current study focused on the role of KORs in the morphine-induced attenuation of function after SCI. In the rodent model of SCI, we challenged morphine with norBNI pretreatment and monitored recovery for 21 days. We found that the effects of norBNI pretreatment were dose dependent. At higher doses, norBNI blocked the morphine-induced attenuation of recovery; however, the analgesic efficacy of morphine was also significantly decreased. Conversely, at low doses, analgesia was maintained, but the adverse effects of morphine on recovery persisted. A moderate dose of norBNI protected against morphine's effects without eliminating analgesic efficacy. These results suggest that blocking KOR activity may be a viable strategy for improving the safety of clinical opioid use.
Methods
Subjects
The subjects were male Sprague–Dawley rats (n = 100) obtained from Harlan (Houston, TX). They were ∼90–110 days old (300–350 g) and were individually housed in Plexiglas bins (45.7 [length] × 23.5 [width] × 20.3 [height] cm) with food and water continuously available. Following surgery, the rats were manually expressed in the morning (7:00–8:30 a.m.) and in the evening (4:30–6:00 p.m.) until they regained full bladder control (operationally defined as three consecutive days with an empty bladder at the time of expression). The rats were weighed daily, and checked for signs of autophagia and spastic hypertonia. A rat was classified as having spastic hypertonia if the limb was in an extended, fixed position and was resistant to movement. The rats were maintained on a 12 h light/dark cycle, and all behavioral testing was conducted during the light cycle. All of the experiments reported here were reviewed and approved by the Institutional Animal Care Committee at Texas A&M University and all National Institutes of Health (NIH) guidelines for the care and use of animal subjects were followed.
Surgery
Rats received a moderate contusion injury using the Infinite Horizon (IH) spinal cord impactor (PSI, Fairfax Station, VA). Briefly, rats were anesthetized with isoflurane (5%, gas), and after a stable level of anesthesia was reached, the concentration of isoflurane was lowered to 2–3%. The rat's back was shaved and disinfected with iodine, and a 5.0 cm incision was made over the spinal cord. Two incisions were made along the vertebral column, on each side of the dorsal spinous processes, extending ∼2 cm rostral and caudal to the T12 segment. Muscle and connective tissue were then dissected to expose the underlying vertebral segments. Musculature around the transverse processes was cleared to allow for clamping of the vertebral spinal column. Next, the dorsal spinous process at T12 was removed (laminectomy), and the spinal tissue exposed (∼L1-L3). The dura remained intact. The vertebral column was fixed within the IH device using two pairs of Adson forceps. A moderate injury was produced using an impact force of 150 kdyn and a 1 sec dwell time. After injury, a 15 cm long polyethylene (PE-10) cannula, fitted with a stainless steel guiding wire (P01008, Ernie Ball Inc., Coachella, CA), was threaded 2 cm under the vertebrae immediately caudal to the injury site. The tubing was inserted into the subarachnoid space. To prevent cannula movement, the exposed end of the tubing was secured to the vertebrae rostral to the injury using tissue adhesive (3M Vetbond Tissue Adhesive, 3M Animal Care Products, St Paul, MN). The wire was then pulled from the tubing and the wound was closed using Michel clips. To help prevent infection, rats were treated with 100 000 U/kg Pfizerpen (penicillin G potassium) immediately after surgery and again 2 days later. For the first 24 h after surgery, rats were placed in a recovery room maintained at 26.6°C. To compensate for fluid loss, rats were given 3 mL of saline after surgery. Michel clips were removed 14 days after surgery.
Drug administration
Drug administration took place on the day after surgery, following baseline tests of locomotion and thermal reactivity. Baseline Basso, Beattie, and Bresnahan (BBB) scores were balanced across all groups. Drugs were administered via an intrathecal route. First, rats were given a single dose of norBNI (Tocris Bioscience, Bristol, UK) dissolved in distilled water (doses ranged from 0 to 0.32 μmol). Two hours following pretreatment with norBNI, rats were administered 0 or 0.32 μmol of morphine (morphine sulfate was generously donated by the National Institute on Drug Abuse [NIDA] Drug Supply Program) also dissolved in distilled water. The 2 h waiting period between norBNI and morphine administration was chosen to account for the slow-onset of norBNI antagonistic action at the KOR, which reaches a plateau at 2 h in vivo.23 Filtered saline (0.9%) was used as the vehicle control, and to flush catheters following the injection of a drug.
Assessments of sensory reactivity
To assess morphine's efficacy, the tail-flick test24–26 of thermal reactivity was used immediately before norBNI pretreatment, and 30 min after morphine administration. Rats were placed in restraining tubes and allowed to acclimate to the tail-flick apparatus (IITC Life Science Inc., Woodland Hills, CA) and testing room (maintained at 26.5°C) for 15 min. Prior to testing, the temperature of the light, focused on the tail, was set to elicit a baseline tail-flick response in approximately 4 sec in an intact rat. This preset temperature was then maintained across all rats. During testing, the latency to flick the tail away from the radiant heat source (light) was recorded. If a rat failed to respond, the test trial was automatically terminated after 8 sec of heat exposure. Two tests occurred at 2 min intervals, and the last tail-flick latency was recorded.
To evaluate the long-term recovery of sensory function, reactivity thresholds were also assessed after day 21 post-injury. Thermal reactivity was evaluated using the tail-flick test, as described. Mechanical reactivity was tested by applying von Frey filaments (Semmes-Weinstein Anesthesiometer, Stoelting Co., Chicago, IL) to the plantar surface of the hindpaws, in a manner previously described by others.25–27 Briefly, stimuli were presented every 2 sec until rats exhibited a motor (hindpaw withdrawal) and vocal response. The intensity of the stimuli that produced a response was reported using the formula provided by Semmes-Weinstein: Intensity = log10 (10,000 × g force). If one or both responses (motor and vocal) were not observed, testing was terminated at a force of 300 g. Each rat was tested twice on each hindpaw in a counterbalanced ABBA order (A = left paw, B = right paw).
Tactile reactivity was also assessed at the level of injury using the girdle test.28 For this test, the girdle region was shaved and a grid map of the girdle zone for allodynic responding was made on the rats using an indelible marker (44 squares). To ensure that the rats remained calm for testing, they were handled for 5 min immediately prior to beginning the girdle test. A von Frey filament with bending force of 204.14 mN (26 g force) was then applied to each point on the grid, and vocalization responses were recorded and mapped onto a grid map of that rat. Because rats do not normally vocalize to this stimulus, a vocalization response indicated that a noxious stimulus was experienced. In mapping the area of response, the number of vocalizations were recorded (Nv) and normalized by the following formula: (Nv × 100)/total number of applications (44), indicating the percent vocalizations out of the total number of applications.
Locomotor recovery
Locomotor behavior was assessed for 21 days post-injury, using the Basso, Beattie and Bresnahan (BBB) scale26,29 in an open enclosure (a blue children's wading pool, 99 cm in diameter, 23 cm deep). Baseline motor function was assessed on the day following injury and prior to drug treatment. Locomotor behavior was then scored once per day for 1 week (days 2–7). Rats were scored every other day from day 9 to day 15 and every 3rd day on days 18 and 21. Because rodents often remain motionless (freeze) when first introduced to a new apparatus, rats were acclimated to the observation fields for 5 min per day for 3 days prior to surgery. Each rat was placed in the open field and observed for 4 min. Care was taken to ensure that all investigators' scoring behavior had high intra- and interobserver reliability (all rs > 0.89) and that they were blind to experimental conditions.
Additional measures of motor recovery were obtained at the end of the 21 day recovery period using tapered beam and ladder walk tasks.26 Prior to testing, rats were habituated to the experimental context for 3 days (8 min per day). During this period of familiarization, they were trained to traverse a wide beam (48.3 cm) and enter a black box positioned at the end of the beam runway. The beginning of the runway was brightly lit, motivating rats to move toward the dark box. They were left in the box for 2 min after they had traversed the beam. Rats were then tested on the tapered beam and ladder. The beam walk test30,31 provides a comparative index of the postural stability of the rats, as well as a gross measure of paw placement abilities. The ledged, tapered beam was 17.15 cm wide at the start, and 0.95 cm wide at the end. We recorded the width at which each hindpaw failed to plantar place on the beam. The average width across the two legs was used as an index of beam walk performance. The ladder task26,32 provides a measure of the extent to which experimental manipulations affect the fine motor abilities of the hindpaws. The rats were required to cross a horizontal ladder (20 cm wide; 37 rungs at 2.5 cm spacing) in order to reach the black box. Using post-hoc frame-by-frame video analyses, we then recorded how many times the rats did not successfully place their hindpaws (their paws slipped between the rungs). Each hindpaw was scored according to the number of errors made, with lower scores indicating better ladder walk performance (a maximum of 11 errors was recorded if no plantar placement was observed per hindpaw).
Histology
At the end of behavioral testing, rats were deeply anesthetized (100 mg/kg of Beuthanasia, i.p.) and perfused intracardially with 4% paraformaldehyde. A 1 cm long segment of the spinal cord, which included the lesion center, was taken and prepared for cryostat sectioning. The tissue was sectioned coronally (20 μm) and every 10th slice was preserved for staining. All sections were stained with cresyl violet for Nissl substance and Luxol fast blue for myelin.33,34
The total cross-sectional area of the cord and spared tissue was assessed at the lesion center using Neurolucida software (MFB Bioscience, Williston, VT). Sections ±600, 1200, 1800, and 2400 μm from the lesion center (rostral and caudal) were also traced and analyzed. Four indices of lesion magnitude were derived: lesion, residual gray matter, residual white matter, and width. To determine the area of lesion, an observer who was blind to the experimental treatments traced around the boundaries of cystic formations and areas of dense gliosis.29 Nissl-stained areas that contained neurons and glia of approximately normal densities denoted residual gray matter. White matter was judged spared in myelin-stained areas lacking dense gliosis and swollen fibers. The total area of each cross-section was derived by summing the areas of damage, and gray and white matter. Width was determined from the most lateral points across the transverse plane. These analyses yielded six parameters for each section: white matter area, gray matter area, spared tissue (white + gray), damaged tissue area, net area (white + gray + damage), and section width.
To control for variability in section area across rats, we applied a correction factor derived from standard undamaged cord sections taken from age-matched controls. This correction factor is based on section widths and is multiplied by all area measurements to standardize area across analyses.35 By standardizing area across sections we were able to estimate the degree to which tissue is “missing” (i.e., tissue loss from atrophy, necrosis, or apoptosis). An accurate assessment of the degree to which a treatment has impacted, or lesioned, the cord includes both the remaining ‘‘damaged’’ tissue as well as resolved lesioned areas. When we sum the amount of ‘‘missing’’ tissue and the measured ‘‘damaged’’ area, we can derive an index of the relative lesion (% relative lesion) in each section that is comparable across sections. We can also compute the relative percent of gray and white matter remaining in each section, relative to intact controls. These measures are highly correlated with various measures of behavioral performance including BBB locomotor scores and recovery of bladder function.35
Statistical analysis
In this experiment, four norBNI doses were tested (0, 0.02, 0.08, and 0.32 μmol). For each dose, half of the rats were subsequently treated with morphine and the other half with vehicle. This 4 × 2 experimental design (eight groups) used a total of 100 rats (n = 12 for doses 0.02, 0.08, 0.32 μmol norBNI with or without morphine; n = 14 for dose 0 μmol norBNI with or without morphine).
The results were analyzed using a two factor analysis of variance (ANOVA). When main effects were found to be significant, the Bonferroni test was used for post-hoc analyses. Planned comparisons were also included in our assessment of the data, as we anticipated dose-dependent effects of norBNI on morphine's efficacy, even in the absence of overall main effects. This allowed for independent comparisons between morphine and vehicle-treated groups at each dose of norBNI. Lastly, in experiments with a continuous independent variable (e.g., recovery of locomotor function across days and histology), mixed-design ANOVAs were used.
Locomotor scores using the BBB scale were transformed, as described by Ferguson and coworkers,36 to help assure that the data were amendable to parametric analyses. Briefly, this transformation removes a discontinuity in the scale, which justifies the use of parametric statistical analyses, and increases statistical power. Additional statistical power was also achieved by obtaining a measure of locomotor performance 24 h after injury, prior to norBNI or morphine treatment. By using day 1 as a covariate in an analysis of covariance (ANCOVA), we substantially reduced unexplained variance and thereby increased statistical power.
Statement of ethics
We certify that all applicable institutional and governmental regulations concerning the ethical use of animals were followed during the course of this research.
Results
Analgesic efficacy
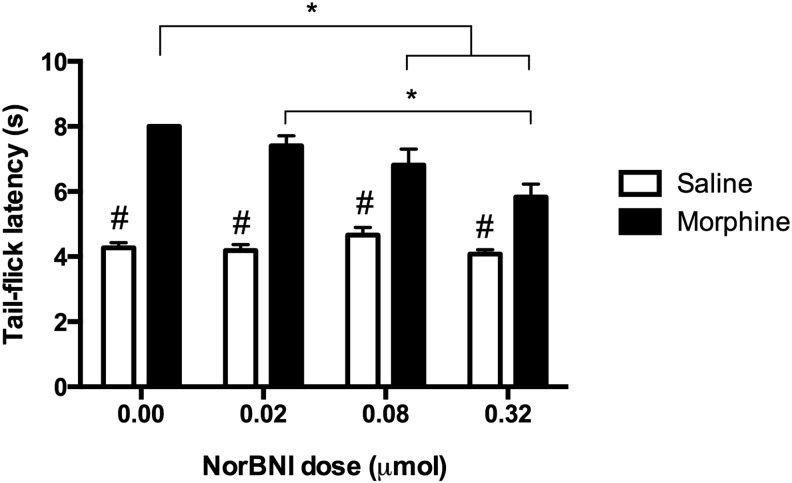
Sensory function was evaluated using the tail-flick test to establish baseline thermal reactivity thresholds. Prior to treatment, tail-flick scores (± S.E.M.) on day 1 were as follows: for rats treated with vehicle, 4.27 ± 0.16 sec for the 0 μmol dose, 4.31 ± 0.20 sec for the 0.02 μmol dose, 4.05 ± 0.24 sec for the 0.08 μmol dose, and 4.27 ± 0.09 sec for the 0.32 μmol dose; for rats treated with morphine, 3.79 ± 0.21 sec for the 0 μmol dose, 4.27 ± 0.21 sec for the 0.02 μmol dose, 4.39 ± 0.17 sec for the 0.08 μmol dose, and 4.29 ± 0.18 sec for the 0.32 μmol dose. Statistical analyses showed that there were no significant differences among the groups at this time point (F [7, 91] = 1.04, p > 0.05; data not shown). A two factor ANOVA on post-treatment scores (30 min after morphine administration), however, revealed significant main effects of morphine (F [1, 91] = 193.97, p < 0.05) and norBNI treatment (F [3, 91] = 5.52, p < 0.05), and a significant interaction (F [3, 91] = 6.57, p < 0.05) on tail-flick latency. As shown in Figure 1, all groups that received morphine had increased tail-flick latencies relative to controls. NorBNI, however, decreased the analgesic effects of morphine, with a significant linear reduction in efficacy across increasing doses (F [1, 46] = 21.03, p < 0.05). Whereas rats that received morphine alone displayed maximal latencies (the test was automatically terminated at 8 sec to prevent tissue injury), those pretreated with the highest dose of norBNI (0.32 μmol) had a mean tail-flick latency of 5.83 ± 0.40 sec.
FIG. 1.
The analgesic efficacy of morphine challenged with nor-Binaltorphimine (norBNI) pretreatment is depicted for the test of thermal reactivity. All groups that received morphine had significantly increased tail-flick latencies relative to vehicle-treated controls. However, morphine's analgesic effects decreased with increasing doses of norBNI. Results shown as mean ± SEM. *p < 0.05 for post-hoc tests; # p < 0.05 for planned comparisons.
Recovery of locomotor function
In addition to the effects on analgesia, we wanted to assess whether KOR activation was necessary to produce the morphine-induced attenuation of locomotor recovery.1–5 To address this, norBNI was administered as an adjuvant to morphine treatment 24 h following SCI, and locomotor recovery was monitored for a 21 day period. Locomotor scores collected before treatment on day 1 did not differ significantly across groups (F [7, 92] = 0.01, p > 0.05). Mean converted BBB scores (± S.E.M.) on day 1 ranged from 1.61 ± 0.26 to 1.88 ± 0.33. A mixed design ANCOVA using day 1 scores as a covariate did not find significant main effects of morphine (F [1, 91] = 1.85, p < 0.05) or norBNI treatment (F [3, 91] = 1.83, p > 0.05). An interaction approached, but did not reach, significance (F [3, 91] = 2.30, p = 0.083). However, as shown in Figure 2A, planned comparisons revealed that control rats (0 μmol of norBNI) treated with morphine showed significantly reduced locomotor recovery when compared with their vehicle-treated counterparts (F [1, 25] = 4.96, p < 0.05), replicating our previous studies.2,4 Morphine and vehicle groups also differed significantly at the lowest dose (0.02 μmol) (Figure 2B) of norBNI (F [1, 21] = 5.78, p < 0.05). In contrast, there were no differences between morphine- and vehicle-treated rats at the 0.08 μmol (F [1, 21] = 0.46, p > 0.05) or 0.32 μmol (F [1, 21] = 0.17, p > 0.05) doses of norBNI (Fig. 2C and D, respectively). This suggests that, although morphine and vehicle groups recovered equally when higher doses (0.08 and 0.32 μmol) of norBNI were used, lower doses of norBNI were not able to block the morphine-induced attenuation of locomotor recovery.
FIG. 2.
Nor-Binaltorphimine (norBNI) pretreatment blocked the adverse effects of morphine on long-term recovery of locomotor function. Morphine undermined recovery of locomotor function in the absence of norBNI (A) and at a dose of 0.02 μmol norBNI (B). Pretreatment with 0.08 (C) or 0.32 (D) μmol norBNI, however, prevented the morphine-induced attenuation of locomotor recovery. Results shown as mean ± SEM. # p < 0.05 for planned comparisons.
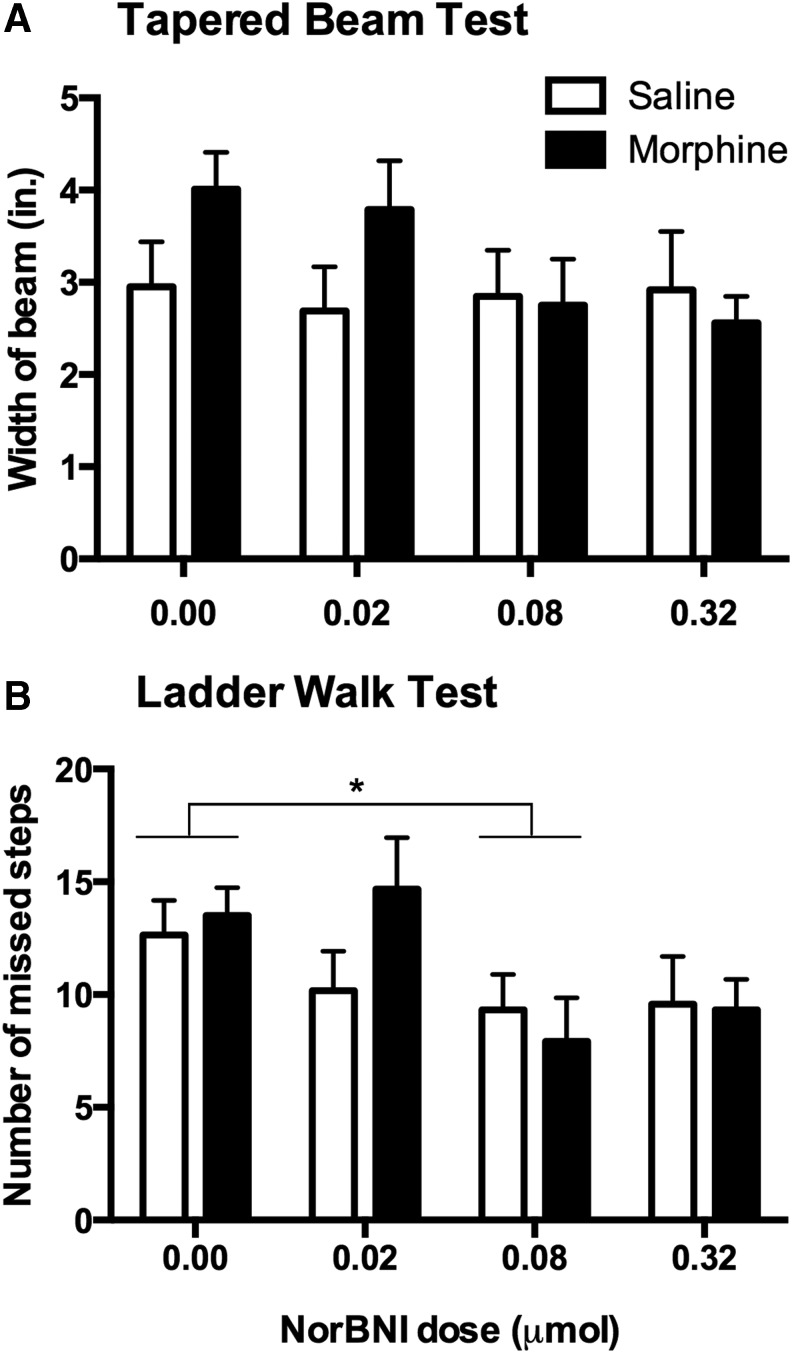
Motor recovery was further evaluated at the end of the 21 day recovery period using the tapered beam and ladder walk tests. Although we observed that morphine-treated rats at the higher doses of norBNI showed better performance on the tapered beam test than those receiving the lower doses (Fig. 3A), statistical analysis did not confirm significant main effects of norBNI (F [3, 92] = 1.17, p > 0.05) or morphine (F [1, 92] = 1.62, p > 0.05) treatment, and no interaction (F [3, 92] = 1.31, p > 0.05). In the ladder walk test (Fig. 3B), we found a significant main effect of norBNI (F [3, 92] = 3.39, p < 0.05). Post-hoc analysis showed that rats in the 0.08 μmol dose performed better than rats not treated with norBNI. However, we did not find an effect of morphine treatment (F [1, 92] = 0.60, p > 0.05) or a significant interaction (F [3, 92] = 1.11, p > 0.05).
FIG. 3.
Nor-Binaltorphimine (norBNI) improved motor performance on the ladder walk test conducted 21 days after injury. Although there were no significant effects of morphine or norBNI on the tapered beam test (A), norBNI significantly improved performance on the ladder walk test (B). Results shown as mean ± SEM. *p < 0.05 for post-hoc tests.
Recovery of sensory function
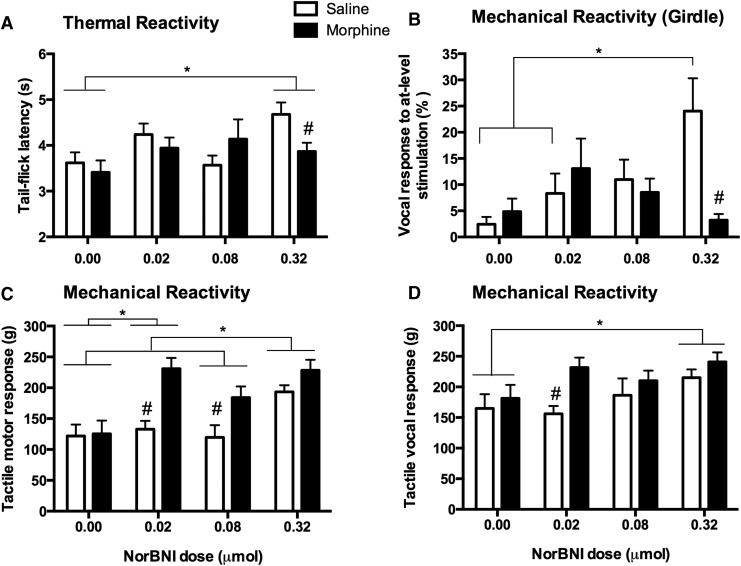
Long-term effects on sensory function were assessed at the end of the 21 day recovery period using the tail-flick, von Frey, and girdle tests. In the tail-flick test, mean latencies (± S.E.M.) ranged from 3.41 ± 0.26 to 4.68 ± 0.26 sec. Although we did not find an effect of morphine (F [1, 92] = 0.95, p > 0.05) or an interaction (F [3, 92] = 2.05, p > 0.05), statistical analyses revealed a significant main effect of norBNI treatment (F [3, 91] = 2.95, p < 0.05) on thermal reactivity after day 21. As shown in Figure 4A, norBNI improved tail-flick latency. In effect, post-hoc tests showed that rats treated with the highest dose of norBNI (0.32 μmol) had significantly higher reactivity thresholds than those that did not receive norBNI treatment (0.00 μmol).
FIG. 4.
There were significant effects of nor-Binaltorphimine (norBNI) pretreatment on the long-term recovery of sensory function. At the end of the 21 day recovery period, there was a significant main effect of norBNI on thermal reactivity (A). NorBNI decreased thermal reactivity relative to subjects that were not given norBNI. By contrast, on the girdle test of at-level allodynia, there was a significant interaction between norBNI and morphine treatment (B); norBNI appeared to increase reactivity in subjects that were not treated with morphine. As found on the test of thermal reactivity, both morphine and norBNI decreased motor (C) and vocal (D) responses to tactile stimulation at day 21 post-injury. Results shown as mean ± SEM. *p < 0.05 for post-hoc tests; # p < 0.05 for planned comparisons.
Mechanical reactivity of the hindpaws tested at the end of the recovery period appeared to decrease with higher doses of norBNI. A two factor ANOVA on motor thresholds (Fig. 4C) uncovered significant main effects of morphine (F [1, 91] = 14.82, p < 0.05) and norBNI (F [3, 91] = 8.58, p < 0.05), but an interaction only approached significance (F [3, 91] = 2.47, p = 0.067). Across norBNI groups, rats treated with higher doses of norBNI showed increased mechanical thresholds. Rats that did not receive norBNI showed the most reactivity to tactile stimulation, and significantly differed from those in the 0.02 μmol and 0.32 μmol groups. On the other hand, rats treated with the highest dose of norBNI (0.32 μmol) showed the least reactivity in this test. Surprisingly, overall, vehicle-treated rats had significantly lower motor thresholds than morphine-treated animals (p < 0.05). Similarly, a significant main effect of morphine treatment on vocal thresholds (Fig. 4D) to von Frey stimulation (F [1, 91] = 6.10, p < 0.05) supported the observation that vehicle-treated rats exhibited more symptoms of mechanical hyperalgesia in the long term. An effect of norBNI treatment approached but did not reach significance (F [3, 92] = 2.55, p = 0.06), and no interaction was observed (F [3, 92] = 0.88, p > 0.05). Together, the motor and vocal responses obtained in the von Frey test of mechanical reactivity suggest that morphine treatment in combination with norBNI may reduce pain and prevent the development of allodynia following SCI.
Lastly, in the girdle test of at-level allodynia, we did not find main effects of norBNI (F [3, 92] = 2.48, p > 0.05) or morphine (F [1, 92] = 2.15, p > 0.05), but there was a significant interaction between the two treatments (F [3, 91] = 4.37, p < 0.05). As depicted in Figure 4B, vocal responses to at-level stimulation decreased with escalating norBNI doses for morphine-treated rats. On the other hand, higher doses of norBNI appeared to exacerbate allodynia in vehicle-treated controls, shown by a steady increase in vocalization. At the highest dose tested (0.32 μmol), vehicle-treated rats vocalized ∼24% of the time, compared with 3% of morphine-treated rats.
Assessment of general health
During the recovery period, weight was also monitored as an index of general health. To control for the variability observed in starting weight within each group, a difference score was calculated by subtracting the starting weight (weight at the day of surgery) from daily weight across recovery. All rats lost the most weight the first week post-SCI, and then slowly regained weight over the subsequent weeks. Mean weight loss across recovery (Fig. 5) for rats treated with vehicle was 27.53 ± 3.78 g for the 0.00 μmol dose, 25.37 ± 3.49 g for the 0.02 μmol dose, 28.92 ± 3.97 g for the 0.08 μmol dose, and 25.42 ± 4.20 g for the 0.32 μmol dose. For morphine-treated rats, mean weight loss was as follows, 34.17 ± 3.32 g for the 0.00 μmol dose, 22.95 ± 4.00 g for the 0.02 μmol dose, 28.07 ± 3.96 g for the 0.08 μmol dose, and 28.02 ± 4.57 g for the 0.32 μmol dose. Statistical analyses showed that there were no effects of norBNI (F [3, 89] = 2.46, p > 0.05) or morphine (F [1, 89] = 0.29, p > 0.05) administration, and no interaction between the two (F [3, 89] = 1.15, p > 0.05). Overall, no differences on weight change across recovery were observed between the groups.
FIG. 5.
Weight loss was unaffected by morphine or nor-Binaltorphimine (norBNI) treatment. Results shown as mean ± SEM.
We had very few instances of mortality, autophagia, and spasticity overall. In the entire experiment, only one rat had to be replaced because of death (treated with 0.02 μmol norBNI and morphine). Three rats exhibited mild autophagia, which resolved rapidly with treatment and did not require removal from the study (all were in the 0.02 μmol group; one treated with vehicle, and two with morphine). Lastly, four rats developed spastic hypertonia during the recovery period (two in the 0.08 μmol group, and one each in the 0 and 0.02 μmol groups, and all received morphine). No other complications were observed.
Histological analyses
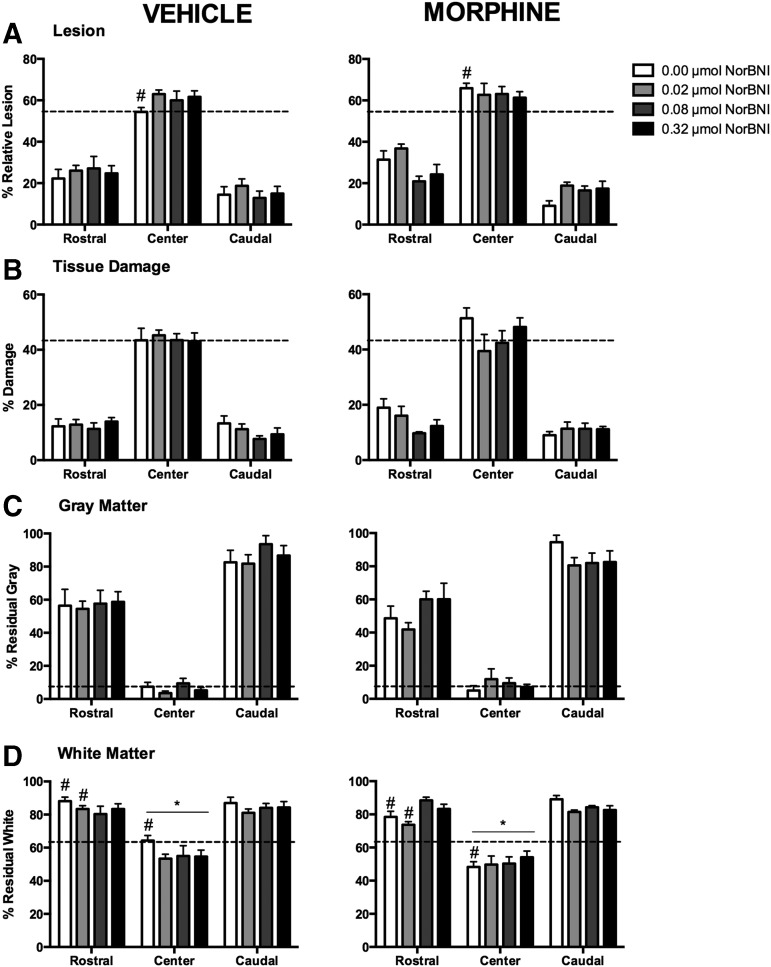
To assess whether the effects of the high doses of norBNI on locomotor function were the result of increased neuroprotection, we assessed the amount of tissue sparing in the spinal cord at the end of the recovery period. Spinal tissue was subdivided into rostral (2400–600 μm), center (0 μm), and caudal (−600 to −2400 μm) segments. Four measures were analyzed: relative lesion size (damage + missing tissue), tissue damage, residual gray matter, and residual white matter.
There were no significant main effects of norBNI or morphine treatment on relative lesion size across the extent of the spinal cord (Fig. 6A). Planned comparisons, however, revealed a significant difference between vehicle and morphine-treated rats without norBNI pretreatment at the center of the lesion (p < 0.05). Morphine-treated rats had increased damage at the injury center, with a mean lesion size (± S.E.M.) of 3.12 ± 0.26 mm2 compared with 2.39 ± 0.23 mm2 for vehicle controls. Similarly, no main effects on damage were observed, although rostral to the injury, planned comparisons showed that a difference between vehicle and morphine rats at the 0 μmol dose of norBNI approached significance (p = 0.066) (Fig. 6B).
FIG. 6.
As found in previous studies, morphine increased the percent relative lesion at the injury center (0 μm) relative to vehicle treated controls. This effect was not present in morphine-treated subjects that were pretreated with nor-Binaltorphimine (norBNI). There were no main effects of morphine or norBNI on relative lesion size (A), tissue damage (B), or residual gray matter (C). However, residual white matter was significantly affected by morphine treatment at the center of the lesion, and there was a significant interaction between norBNI and morphine treatment rostral to the injury site (D). Morphine decreased the residual white matter at the center of the lesion, but in the rostral segment, norBNI pretreatment at higher doses blocked the adverse effects of morphine on white matter sparing. Results shown as mean ± SEM. *p < 0.05 for post-hoc tests; # p < 0.05 for planned comparisons.
None of the groups differed on the percentage of residual gray matter at any level of the spinal cord (Fig. 6C). However, for residual white matter, the treatment groups showed extensive differences in the center and rostral segments (Fig. 6D). We found a significant main effect of morphine treatment at the center of the lesion (F [1, 56] = 3.94, p < 0.05), with morphine-treated rats showing less residual white matter than their vehicle-treated counterparts, irrespective of norBNI. Planned comparisons also uncovered a significant difference between vehicle and morphine controls (0 μmol norBNI) at this level (2.48 ± 0.12 mm2 and 1.86 ± 0.12 mm2 respectively). Rostral to the injury site, the effects on residual white matter appeared to be the result of a significant interaction between norBNI and morphine treatment (F [3, 56] = 3.75, p < 0.05). At the lower doses of norBNI (0 and 0.02 μmol), morphine-treated rats lost more white matter than vehicle-treated rats, and these differences reached statistical significance (p < 0.05). At the 0 μmol dose, residual white matter was 3.39 ± 0.10 mm2 for the vehicle group, and 3.02 ± 0.21 mm2 for the morphine group. At 0.02 μmol, residual white matter was 3.21 ± 0.20 mm2 for vehicle, and 2.84 ± 0.25 mm2 for morphine. Lastly, at the 0.08 μmol dose, a difference between morphine and vehicle groups approached, but did not reach significance (p = 0.073), and no differences were found at 0.32 μmol dose (p = 0.98).
Discussion
The results of the current study suggest that the KOR plays a critical role in the morphine-induced attenuation of locomotor recovery after SCI. In the absence of norBNI, morphine-treated rats showed decreased locomotor recovery relative to vehicle-treated controls, replicating our previous studies.2,4 Moderate to high doses of norBNI (0.08 and 0.32 μmol respectively), however, blocked the effects of morphine on locomotor recovery. Importantly, at these doses, morphine administration still produced significant analgesia. A KOR antagonist may be a viable clinical adjuvant to morphine.
A KOR antagonist may prevent the morphine-induced attenuation of locomotor recovery by reducing the extent of cell death at the site of injury. Our histological analyses showed that higher doses of norBNI increased the amount of residual white matter at the center of, and rostral to, the injury site. Although our previous work implicated activation of the KOR in reduced gray matter sparing at the lesion center,6 others have suggested that supporting cells may also be vulnerable to the effects of KOR activation following SCI.37 For example, Adjan and coworkers measured caspase-3 activity at the injury site in wild-type and prodynorphin knockout mice following an experimental spinal contusion.37 They found that not only was caspase-3 significantly decreased in tissue homogenates from prodynorphin knockout mice, but also that astrocytes and oligodendrocytes in these subjects expressed significantly lower levels of active caspase-3 hours after injury. Commensurate with our histological findings, these results suggest that norBNI may provide beneficial effects following SCI by protecting glia from the synergistic effects of endogenous (dynorphin) and exogenous (morphine) KOR activation.
Aside from directly engaging apoptotic mechanisms following SCI, KOR signaling may also contribute to secondary injury through aberrant glial activation. A role for the KOR in glial activation and proliferation following trauma is supported by evidence from other injury models. Following partial sciatic nerve ligation, KOR immunoreactivity is significantly increased in dorsal horn GABAergic neurons and astrocytes, ipsilateral and contralateral to the injury.38 Importantly, KOR activation appears to play a direct role in the astrocytic response to injury. As reported by Xu and coworkers, mice with a dynorphin knockout, a KOR knockout, or pretreated with norBNI, lacked the marked increase in glial fibrillary acidic protein (GFAP) immunoreactivity observed 1 week post-nerve injury in wild-type controls.39 In vitro, cultured astrocytes treated with the KOR agonist U50,488 showed a significantly increased proliferation rate, and this effect was blocked by norBNI.39 Glial proliferation is also characteristic of SCI. Microglia and astrocytes are among the many supporting cells that become activated in response to injury.40 These cells release a variety of toxic factors, including pro-inflammatory cytokines, which have been implicated in secondary injury. In our model, norBNI pretreatment may block the overproduction of neurotoxic factors by preventing KOR-mediated glial activation and proliferation, countering the adverse consequences of morphine treatment after SCI.
Similarly, by inhibiting aberrant glial activation, norBNI may also block the development of pathological pain following SCI.41–43 In the current study, treatment with norBNI decreased signs of pain in the chronic phase of injury. Assessed at the end of the 21 day recovery period, rats treated with the highest dose of norBNI (0.32 μmol) showed significantly higher thresholds than controls (0 μmol norBNI) in tests of thermal and tactile (hindpaw) reactivity. NorBNI may prevent the development of pain by blocking downstream targets of the KOR. Evidence suggests that the KOR can activate mitogen-activated protein kinases (MAPKs) in a variety of cells, including neurons and astrocytes, via an arrestin-mediated mechanism.39,44,45 Specifically, the KOR has been shown to activate p38 MAPK,39,44 a pathway that is usually engaged in response to physical and chemical stresses, and which plays a critical role in normal immune function and the inflammatory response.46 Importantly, p38 MAPK modulates neuronal apoptosis following SCI,47 and elevated levels of spinal p38 have been strongly implicated in the generation and maintenance of neuropathic pain in models of nerve and spinal cord injury.48–51 Evidence suggests that these neurotoxic and hyperalgesic effects may result from p38-mediated glial activation and inflammation.52 Treatment with p38 inhibitors prevents glial proliferation in the spinal cord and decreases the expression of neuropathic pain symptoms after injury.39,48,49,51 NorBNI may decrease KOR-mediated p38 signaling, preventing aberrant glial activation. As morphine administration also results in increased expression of many pro-inflammatory cytokines, including interleukin (IL)-1β and IL-6,2,53–56 using norBNI as an adjuvant may also reduce the development of paradoxical pain observed with prolonged morphine administration.54–57 Although not tested here, reducing glial proliferation and cytokine secretion by blocking KOR/p38 activation may be a viable therapy for SCI-related pain, and should be further investigated.
Surprisingly, in this study, norBNI had differential effects on at-level pain that depended on morphine treatment. In the girdle test, norBNI appeared to decrease allodynia in morphine-treated rats. The opposite was observed with vehicle-treated controls, which showed increased vocalization to at-level stimulation with escalating doses of norBNI. This may reflect maladaptive alterations in spinal circuitry, resulting from the inadequate mitigation of nociceptive transmission caused by reduced opioid activity. Spinal KORs significantly contribute to endogenous and exogenous pain modulation. For example, we have previously shown that the analgesic efficacy of the selective KOR agonist GR89696 is commensurate with DAMGO and morphine in tests of thermal and mechanical reactivity following SCI.6 Further, in the absence of mu opioid receptors, intrathecal administration of morphine produces analgesia by acting through spinal KORs.58 The contribution of KORs to the analgesic effect of morphine is further supported by the significant decrease in tail-flick latency that we observed with increasing doses of norBNI in the current study (Fig. 1). In our SCI rats treated with morphine, MOR and DOR activation may have compensated for the effects of norBNI. However, in vehicle controls, no additional mechanism was employed to counteract the loss of endogenous KOR antinociception. Thus, unchecked neuronal excitability in the acute phase of SCI may have contributed to at-level central sensitization and the development of chronic pain. Experimental evidence suggests that noxious stimulation undermines the plasticity of the spinal cord, and impairs sensorimotor recovery following SCI.35,59–61 Clinically, preemptive analgesia has also been used in an effort to prevent the establishment of central sensitization and postoperative pathological pain,62–64 although the effectiveness of this strategy remains controversial.
Conclusion
In sum, our results suggest that the KOR significantly contributes to morphine's analgesic efficacy. However, the KOR also appears to play an important role in the mechanisms leading to the adverse effects of morphine administration following SCI. We hypothesize that these effects are the result of an opioid-induced exacerbation of the inflammatory response intrinsic to SCI. In vitro and in vivo evidence suggests that opioid administration results in the activation of glial cells and the release of pro-inflammatory cytokines.53–56,65,66 Although non-classic opioid receptor signaling has been implicated in these opioid-immune interactions,8,67 our findings point to a previously overlooked role of KOR. Although not tested in this study, we posit that morphine's adverse effects may result from activation of KORs on glial cells.38,39,44,68 The synergistic effects of non-classic opioid receptor activation and KOR-mediated gliopathy could explain the decreased locomotor recovery, increased nociceptive reactivity, and decreased tissue sparing observed in our rodent model when morphine is administered following SCI.24 These results underscore the need for further studies targeting the cell-specific effects of opioid administration, and other medications, following SCI. Overall, however, these data suggest that KOR antagonists may be viable adjuvants to morphine, reducing the adverse long-term consequences of opioid administration in the acute phase of SCI.
Acknowledgments
The authors thank Kiralyn Brakel and Mabel N. Terminel for their comments on a previous version of this manuscript, and the NIDA Drug Supply Program for their generous contribution. This work was funded by grant DA31197 to M.A. Hook.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Woller S.A., Moreno G.L., Hart N., Wellman P.J., Grau J.W., and Hook M.A. (2012). Analgesia or addiction? implications for morphine use after spinal cord injury. J. Neurotrauma 29, 1650–1662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hook M.A., Washburn S.N., Moreno G., Woller S.A., Puga D., Lee K.H., and Grau J.W. (2011). An IL-1 receptor antagonist blocks a morphine-induced attenuation of locomotor recovery after spinal cord injury. Brain Behav. Immun. 25, 349–359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hook M.A., Liu G.T., Washburn S.N., Ferguson A.R., Bopp A.C., Huie J.R., and Grau J.W. (2007). The impact of morphine after a spinal cord injury. Behav. Brain Res. 179, 281–293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hook M.A., Moreno G., Woller S., Puga D., Hoy K., Jr., Balden R., and Grau J.W. (2009). Intrathecal morphine attenuates recovery of function after a spinal cord injury. J. Neurotrauma 26, 741–752 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Woller S.A., Malik J.S., Aceves M., and Hook M.A. (2014). Morphine self-administration following spinal cord injury. J. Neurotrauma 31, 1570–1583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aceves M., Mathai B., and Hook M. (2016). Evaluation of the effects of specific opioid receptor agonists in a rodent model of spinal cord injury. Spinal Cord 54, 767–777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kristensen K., Christensen C.B., and Christrup L.L. (1994). The mu 1, mu 2, delta, kappa opioid receptor binding profiles of methadone stereoisomers and morphine. Life Sci. 56, 45–50 [DOI] [PubMed] [Google Scholar]
- 8.Wang X., Loram L.C., Ramos K., de Jesus A.J., Thomas J., Cheng K., Reddy A., Somogyi A.A., Hutchinson M.R., and Watkins L.R. (2012). Morphine activates neuroinflammation in a manner parallel to endotoxin. Proc. Natl. Acad. Sci. U.S.A. 109, 6325–6330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Faden A.I., and Jacobs T.P. (1983). Dynorphin induces partially reversible paraplegia in the rat. Eur. J. Pharmacol. 91, 321–324 [DOI] [PubMed] [Google Scholar]
- 10.Herman B.H., and Goldstein A. (1985). Antinociception and paralysis induced by intrathecal dynorphin A. J. Pharmacol. Exp. Ther. 232, 27–32 [PubMed] [Google Scholar]
- 11.Cox B.M., Molineaux C., Jacobs T., Rosenberger J., and Faden A.I. (1985). Effects of traumatic injury on dynorphin immunoreactivity in spinal cord. Neuropeptides 5, 571–574 [DOI] [PubMed] [Google Scholar]
- 12.Faden A.I., Molioneaux C.J., Rosenberger J.G., Jacobs T.P., and Cox B.M. (1985). Endogenous opioid immunoreactivity in rat spinal cord following traumatic injury. Ann. Neurol. 17, 386–390 [DOI] [PubMed] [Google Scholar]
- 13.Faden A., Molineaux C., Rosenberger J., Jacobs T., and Cox B. (1985). Increased dynorphin immunoreactivity in spinal cord after traumatic injury. Regul. Pept. 11, 35–41 [DOI] [PubMed] [Google Scholar]
- 14.Przewłocki R., Haarmann I., Nikolarakis K., Herz A., and Höllt V. (1988). Prodynorphin gene expression in spinal cord is enhanced after traumatic injury in the rat. Brain Res. 4, 37–41 [DOI] [PubMed] [Google Scholar]
- 15.Tachibana T., Miki K., Fukuoka T., Arakawa A., Taniguchi M., Maruo S., and Noguchi K. (1998). Dynorphin mRNA expression in dorsal horn neurons after traumatic spinal cord injury: temporal and spatial analysis using in situ hybridization. J. Neurotrauma 15, 485–494 [DOI] [PubMed] [Google Scholar]
- 16.Faden A.I. (1990). Opioid and nonopioid mechanisms may contribute to dynorphin's pathophysiological actions in spinal cord injury. Ann. Neurol. 27, 67–74 [DOI] [PubMed] [Google Scholar]
- 17.Faden A.I., Takemori A., and Portoghese P. (1987). κ-Selective opiate antagonist nor-binaltorphimine improves outcome after traumatic spinal cord injury in rats. Cent. Nerv. Syst. Trauma 4, 227–234 [DOI] [PubMed] [Google Scholar]
- 18.Vink R., Portoghese P., and Faden A. (1991). Kappa-opioid antagonist improves cellular bioenergetics and recovery after traumatic brain injury. Am. J. Physiol. Regul. Integr. Comp. Physiol. 261, R1527–R1532 [DOI] [PubMed] [Google Scholar]
- 19.Bakshi R. and Faden A.I. (1990). Competitive and non-competitive NMDA antagonists limit dynorphin A-induced rat hindlimb paralysis. Brain Res. 507, 1–5 [DOI] [PubMed] [Google Scholar]
- 20.Bakshi R., and Faden A.I. (1990). Blockade of the glycine modulatory site of NMDA receptors modifies dynorphin-induced behavioral effects. Neurosci. Lett. 110, 113–117 [DOI] [PubMed] [Google Scholar]
- 21.Caudle R. and Isaac L. (1988). A novel interaction between dynorphin (1–13) and an N-methyl-D-aspartate site. Brain Res. 443, 329–332 [DOI] [PubMed] [Google Scholar]
- 22.Faden A.I. (1992). Dynorphin increases extracellular levels of excitatory amino acids in the brain through a non-opioid mechanism. J. Neurosci. 12, 425–429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Endoh T., Matsuura H., Tanaka C., and Nagase H. (1991). Nor-binaltorphimine: a potent and selective kappa-opioid receptor antagonist with long-lasting activity in vivo. Arch. Int. Pharmacodyn. Ther. 316, 30–42 [PubMed] [Google Scholar]
- 24.D'amour F.E., and Smith D.L. (1941). A method for determining loss of pain sensation. J. Pharmacol. Exp. Ther. 72, 74–79 [Google Scholar]
- 25.Jeffrey S.M., Sonya G.W., and You W. (2001). Assessing nociception in murine subjects. in: Methods in Pain Research. CRC Press, Boca Raton, FL, pp. 11–39 [Google Scholar]
- 26.Šedý J., Urdzíková L., Jendelová P., and Syková E. (2008). Methods for behavioral testing of spinal cord injured rats. Neurosci. Biobehav. Rev. 32, 550–580 [DOI] [PubMed] [Google Scholar]
- 27.Chaplan S., Bach F., Pogrel J., Chung J., and Yaksh T. (1994). Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 [DOI] [PubMed] [Google Scholar]
- 28.Christensen M.D. and Hulsebosch C.E. (1997). Chronic central pain after spinal cord injury. J. Neurotrauma 14, 517–537 [DOI] [PubMed] [Google Scholar]
- 29.Basso D.M., Beattie M.S. and Bresnahan J.C. (1995). A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 12, 1–21 [DOI] [PubMed] [Google Scholar]
- 30.Schallert T. Woodlee M.T., and Fleming S.M. (2002). Disentangling multiple types of recovery from brain injury. In Kriegelstein J. and Klumpp S. (eds.), Pharmacology of Cerebral Ischemia, Medpharm Scientific, Stuttgart, Germany, pp. 201–216 [Google Scholar]
- 31.Schallert T., and Woodlee M.T. (2005). Orienting and placing. In Whishaw I. and Kolb B. (eds.). The Behavior of the Laboratory Rat: A Handbook with Tests. Oxford University Press, Oxford, pp. 129–140 [Google Scholar]
- 32.Metz G.A., and Whishaw I.Q. (2002). Cortical and subcortical lesions impair skilled walking in the ladder rung walking test: a new task to evaluate fore-and hindlimb stepping, placing, and co-ordination. J. Neurosci. Methods 115, 169–179 [DOI] [PubMed] [Google Scholar]
- 33.Beattie M.S. (1992). Anatomic and behavioral outcome after spinal cord injury produced by a displacement controlled impact device. J. Neurotrauma 9, 157–160 [DOI] [PubMed] [Google Scholar]
- 34.Behrmann D.L., Bresnahan J.C., Beattie M.S., and Shah B.R. (1992). Spinal cord injury produced by consistent mechanical displacement of the cord in rats: behavioral and histologic analysis. J. Neurotrauma 9, 197–217 [DOI] [PubMed] [Google Scholar]
- 35.Grau J.W., Washburn S.N., Hook M.A., Ferguson A.R., Crown E.D., Garcia G., Bolding K.A., and Miranda R.C. (2004). Uncontrollable stimulation undermines recovery after spinal cord injury. J. Neurotrauma 21, 1795–1817 [DOI] [PubMed] [Google Scholar]
- 36.Ferguson A.R., Hook M.A., Garcia G., Bresnahan J.C., Beattie M.S., and Grau J.W. (2004). A simple post hoc transformation that improves the metric properties of the BBB scale for rats with moderate to severe spinal cord injury. J. Neurotrauma 21, 1601–1613 [DOI] [PubMed] [Google Scholar]
- 37.Adjan V., Hauser K., Bakalkin G., Yakovleva T., Gharibyan A., Scheff S., and Knapp P. (2007). Caspase-3 activity is reduced after spinal cord injury in mice lacking dynorphin: differential effects on glia and neurons. Neuroscience 148, 724–736 [DOI] [PubMed] [Google Scholar]
- 38.Xu M., Petraschka M., McLaughlin J.P., Westenbroek R.E., Caron M.G., Lefkowitz R.J., Czyzyk T.A., Pintar J.E., Terman G.W., and Chavkin C. (2004). Neuropathic pain activates the endogenous κ opioid system in mouse spinal cord and induces opioid receptor tolerance. J. Neuroscience 24, 4576–4584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu M., Bruchas M.R., Ippolito D.L., Gendron L., and Chavkin C. (2007). Sciatic nerve ligation-induced proliferation of spinal cord astrocytes is mediated by κ opioid activation of p38 mitogen-activated protein kinase. J. Neurosci. 27, 2570–2581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Popovich P.G., Wei P., and Stokes B.T. (1997). Cellular inflammatory response after spinal cord injury in Sprague‐Dawley and Lewis rats. J. Comp. Neurol. 377, 443–464 [DOI] [PubMed] [Google Scholar]
- 41.Hulsebosch C.E. (2008). Gliopathy ensures persistent inflammation and chronic pain after spinal cord injury. Exp. Neurol. 214, 6–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gwak Y.S., and Hulsebosch C.E. (2009). Remote astrocytic and microglial activation modulates neuronal hyperexcitability and below-level neuropathic pain after spinal injury in rat. Neuroscience 161, 895–903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gwak Y.S., Kang J., Unabia G.C., and Hulsebosch C.E. (2012). Spatial and temporal activation of spinal glial cells: role of gliopathy in central neuropathic pain following spinal cord injury in rats. Exp. Neurol. 234, 362–372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bruchas M.R., Macey T.A., Lowe J.D., and Chavkin C. (2006). Kappa opioid receptor activation of p38 MAPK is GRK3-and arrestin-dependent in neurons and astrocytes. J. Biol. Chem. 281, 18,081–18,089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bruchas M.R., and Chavkin C. (2010). Kinase cascades and ligand-directed signaling at the kappa opioid receptor. Psychopharmacology (Berl.) 210, 137–147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Roux P.P., and Blenis J. (2004). ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol. Mol. Biol. Rev. 68, 320–344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang X.-j., Kong K.-m., Qi W.-l., Ye W.-l., and Song P.-s. (2005). Interleukin-1 beta induction of neuron apoptosis depends on p38 mitogen-activated protein kinase activity after spinal cord injury. Acta Pharmacol. Sin. 26, 934. [DOI] [PubMed] [Google Scholar]
- 48.Tsuda M., Mizokoshi A., Shigemoto‐Mogami Y., Koizumi S., and Inoue K. (2004). Activation of p38 mitogen‐activated protein kinase in spinal hyperactive microglia contributes to pain hypersensitivity following peripheral nerve injury. Glia 45, 89–95 [DOI] [PubMed] [Google Scholar]
- 49.Jin S.-X., Zhuang Z.-Y., Woolf C.J., and Ji R.-R. (2003). p38 mitogen-activated protein kinase is activated after a spinal nerve ligation in spinal cord microglia and dorsal root ganglion neurons and contributes to the generation of neuropathic pain. J. Neurosci. 23, 4017–4022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Crown E.D., Ye Z., Johnson K.M., Xu G.-Y., McAdoo D.J., and Hulsebosch C.E. (2006). Increases in the activated forms of ERK 1/2, p38 MAPK, and CREB are correlated with the expression of at-level mechanical allodynia following spinal cord injury. Exp. Neurol. 199, 397–407 [DOI] [PubMed] [Google Scholar]
- 51.Crown E.D., Gwak Y.S., Ye Z., Johnson K.M., and Hulsebosch C.E. (2008). Activation of p38 MAP kinase is involved in central neuropathic pain following spinal cord injury. Exp. Neurol. 213, 257–267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Svensson C.I., Marsala M., Westerlund A., Calcutt N.A., Campana W.M., Freshwater J.D., Catalano R., Feng Y., Protter A.A., and Scott B. (2003). Activation of p38 mitogen‐activated protein kinase in spinal microglia is a critical link in inflammation‐induced spinal pain processing. J. Neurochem. 86, 1534–1544 [DOI] [PubMed] [Google Scholar]
- 53.Hutchinson M.R., Coats B.D., Lewis S.S., Zhang Y., Sprunger D.B., Rezvani N., Baker E.M., Jekich B.M., Wieseler J.L., and Somogyi A.A. (2008). Proinflammatory cytokines oppose opioid-induced acute and chronic analgesia. Brain. Behav. Immun. 22, 1178–1189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Johnston I.N., Milligan E.D., Wieseler–Frank J., Frank M.G., Zapata V., Campisi J., Langer S., Martin D., Green P., and Fleshner M. (2004). A role for proinflammatory cytokines and fractalkine in analgesia, tolerance, and subsequent pain facilitation induced by chronic intrathecal morphine. J. Neurosci. 24, 7353–7365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Raghavendra V., Tanga F.Y., and DeLeo J.A. (2004). Attenuation of morphine tolerance, withdrawal-induced hyperalgesia, and associated spinal inflammatory immune responses by propentofylline in rats. Neuropsychopharmacology 29, 327–334 [DOI] [PubMed] [Google Scholar]
- 56.Raghavendra V., Rutkowski M.D., and DeLeo J.A. (2002). The role of spinal neuroimmune activation in morphine tolerance/hyperalgesia in neuropathic and sham-operated rats. J. Neurosci. 22, 9980–9989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mao J., Sung B., Ji R.-R. and Lim G. (2002). Chronic morphine induces downregulation of spinal glutamate transporters: implications in morphine tolerance and abnormal pain sensitivity. J. Neurosci. 22, 8312–8323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yamada H., Shimoyama N., Sora I., Uhl G.R., Fukuda Y., Moriya H., and Shimoyama M. (2006). Morphine can produce analgesia via spinal kappa opioid receptors in the absence of mu opioid receptors. Brain Res. 1083, 61–69 [DOI] [PubMed] [Google Scholar]
- 59.Garraway S.M., Turtle J.D., Huie J.R., Lee K.H., Hook M.A., Woller S.A., and Grau J.W. (2011). Intermittent noxious stimulation following spinal cord contusion injury impairs locomotor recovery and reduces spinal brain-derived neurotrophic factor–tropomyosin-receptor kinase signaling in adult rats. Neuroscience 199, 86–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Garraway S.M., Woller S.A., Huie J.R., Hartman J.J., Hook M.A., Miranda R.C., Huang Y.-J., Ferguson A.R., and Grau J.W. (2014). Peripheral noxious stimulation reduces withdrawal threshold to mechanical stimuli after spinal cord injury: role of tumor necrosis factor alpha and apoptosis. Pain 155, 2344–2359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Joynes R.L., Ferguson A.R., Crown E.D., Patton B.C., and Grau J.W. (2003). Instrumental learning within the spinal cord: V. Evidence the behavioral deficit observed after noncontingent nociceptive stimulation reflects an intraspinal modification. Behav. Brain Res. 141, 159–170 [DOI] [PubMed] [Google Scholar]
- 62.Katz J., Kavanagh B.P., Sandler A.N., Nierenberg H., Boylan J.F., Friedlander M., and Shaw B.F. (1992). Preemptive analgesia. Clinical evidence of neuroplasticity contributing to postoperative pain. Anesthesiology 77, 439–446 [DOI] [PubMed] [Google Scholar]
- 63.Woolf C.J., and Chong M.-S. (1993). Preemptive analgesia-treating postoperative pain by preventing the establishment of central sensitization. Anesth. Analg. 77, 362–379 [DOI] [PubMed] [Google Scholar]
- 64.Møiniche S., Kehlet H. and Dahl J.B. (2002). A qualitative and quantitative systemactic review of preemptive analgesia for postoperative pain relief: the role of timing of analgesia. J. Am. Soc. Anesthesiol. 96, 725–741 [DOI] [PubMed] [Google Scholar]
- 65.Song P., and Zhao Z.-Q. (2001). The involvement of glial cells in the development of morphine tolerance. Neurosci. Res. 39, 281–286 [DOI] [PubMed] [Google Scholar]
- 66.Cui Y., Chen Y., Zhi J.-L., Guo R.-X., Feng J.-Q., and Chen P.-X. (2006). Activation of p38 mitogen-activated protein kinase in spinal microglia mediates morphine antinociceptive tolerance. Brain Res. 1069, 235–243 [DOI] [PubMed] [Google Scholar]
- 67.Hutchinson M.R., Zhang Y., Shridhar M., Evans J.H., Buchanan M.M., Zhao T.X., Slivka P.F., Coats B.D., Rezvani N., and Wieseler J. (2010). Evidence that opioids may have toll-like receptor 4 and MD-2 effects. Brain. Behav. Immun. 24, 83–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.McLennan G.P., Kiss A., Miyatake M., Belcheva M.M., Chambers K.T., Pozek J.J., Mohabbat Y., Moyer R.A., Bohn L.M., and Coscia C.J. (2008). Kappa opioids promote the proliferation of astrocytes via Gβγ and β‐arrestin 2‐dependent MAPK‐mediated pathways. J. Neurochem. 107, 1753–1765 [DOI] [PMC free article] [PubMed] [Google Scholar]