Abstract
Background: Carnitine palmitoyltransferase IA (CPT-IA) deficiency is an inherited disorder of the carnitine cycle (MIM #255120). Patients affected by this deficiency might be missed easily because of lack of specific and sensitive biochemical markers. In this study, sensitivity and specificity of plasma free carnitine (C0) and long-chain acylcarnitines (lc-ac: C16:0-, C16:1-, C18:0-, C18:1- and C18:2-ac) was evaluated, including the sum of lc-ac (∑lc-ac) and the molar ratios C0/(C16:0-ac+C18:0-ac) and C0/∑lc-ac.
Methods: Nine plasma acylcarnitine profiles of 4 CPT-IA deficient patients were compared with profiles of 2,190 subjects suspected of or diagnosed with an inherited disorder of metabolism. Age-dependent reference values were calculated based on the patient population without a definite diagnosis of an inborn error of metabolism (n = 1,600). Sensitivity, specificity, and Receiver Operating Characteristic (ROC) curves were calculated based on samples of the whole patient population.
Results: Concentrations of C0 in plasma were normal in all CPT-IA deficient patient samples. ROC analyses showed highest diagnostic values for C18:0-ac, C18:1-ac, and ∑lc-ac (AUC 1.000) and lowest for C0 (AUC 0.738). Combining two markers, i.e., a plasma C18:1-ac concentration <0.05 μmol/L and a molar ratio of C0/(C16:0-ac+C18:0-ac) >587, specificity to diagnose CPT-IA deficiency increased to 99.3% compared with either C18:1-ac (97.4%) or C0/(C16:0-ac+C18:0-ac) (96.9%) alone, all at a sensitivity of 100%.
Conclusions: Combination of a low concentration of C18:1-ac with a high molar ratio of C0/(C16:0-ac+C18:0-ac) ratio in plasma has high diagnostic value for CPT-IA deficiency. Patients with a clinical suspicion of CPT-IA deficiency can be diagnosed with this test combination.
Keywords: Carnitine palmitoyltransferase type IA deficiency, Free carnitine, Acylcarnitines, Plasma, Sensitivity, Specificity
Background
Carnitine palmitoyltransferase IA (CPTIA, EC# 2.3.1.21) deficiency is a rare autosomal recessively inherited disorder of the carnitine cycle (MIM #255120) (Bonnefont et al. 2004). This enzyme is essential for transport of long-chain fatty acyl-CoA esters into mitochondria for subsequent beta-oxidation. It is located in the outer mitochondrial membrane and catalyzes the conversion of cytosolic long-chain acyl-CoA esters into their respective acylcarnitine esters. Acylcarnitines are subsequently transported across the inner mitochondrial membrane and reconverted to acyl-CoA thioesters by the actions of carnitine/acylcarnitine translocase (CACT, SLC25A20) and carnitine palmitoyltransferase II (CPTII, EC# 2.3.1.21). Loss of CPT-IA activity diminishes the intra-mitochondrial substrate levels for fatty acid beta-oxidation, thereby impairing energy generation (Bonnefont et al. 2004). Clinical symptoms often appear during periods of prolonged fasting or illness. Life-threatening hypoketotic hypoglycemia, hepatic encephalopathy, hepatomegaly with or without acute liver failure, seizures, and coma are prominent presenting clinical features. Mild cardiomegaly and heart beat disorders may be part of the clinical spectrum, as well as renal tubular acidosis (Bonnefont et al. 2004).
Classically, high plasma concentrations of free carnitine (C0) were considered diagnostic (Bonnefont et al. 2004; Stanley et al. 1992). It became, however, increasingly clear that this diagnostic parameter is problematic and that the diagnosis could be easily overlooked since quite often the concentration of C0 in plasma was found to be normal (Bergman 1994; Bonnefont et al. 2004; Innes et al. 2000; Primassin and Spiekerkoetter 2010; de Sain-van der Velden et al. 2013). It has been suggested that a high concentration of C0 in blood spot is more sensitive than its concentration in plasma (Primassin and Spiekerkoetter 2010; de Sain-van der Velden et al. 2013; Sim et al. 2001). This could be debated. Fingerhut et al. (2001) investigated the concentrations of C0 and acylcarnitines in 6 blood spot samples of 3 patients diagnosed with CPT-IA deficiency (2 days to 12 months old) and compared the results with those of 177,842 samples from the central Bavarian newborn-screening program. Blood spot C0 concentrations were normal in two of three patients. Diagnostic accuracy improved considerably in these patients when the molar ratio of C0/(C16:0-ac+C18:0-ac) was used (Fingerhut et al. 2001). In the most commonly used matrix, i.e., plasma, this ratio might have high sensitivity as well (de Sain-van der Velden et al. 2013). Unfortunately, the specificity of this marker is currently unknown.
To gain insight in the sensitivity ánd specificity of biochemical markers of CPT-IA deficiency, acylcarnitines in plasma of patients with proven CPT-IA deficiency were compared with those of patients in our historical dataset.
Methods
(Acyl-) carnitines in nine plasma samples of four patients with enzymatically and genetically confirmed CPT-IA deficiency were analyzed in three hospital laboratories, using comparable analytical methods. Blood spot acylcarnitines were available for three patients. The plasma results were compared with the historical dataset of 2,190 subjects of whom samples were analyzed in one of our laboratories between 2006 and 2012, (2,194 including the samples of 4 CPT-IA deficient patients). Only analyses of the first sample presented to our laboratories were included. The need for formal ethical review was waived by the local ethics committee. The study design was in accordance with the current revision of the Helsinki Declaration.
Samples were prepared according to local standard operating procedures. In short, plasma was deproteinized with acetonitrile. Subsequently, 6 deuterium-labeled internal standards in methanol/H2O were added and samples were analyzed by flow-injection tandem-mass spectrometry (API-3000, AB Sciex, Framingham, USA). Concentrations of C0 and the individual acylcarnitines were quantitated by multiple reaction monitoring. C0 by an m/z transition of 162->103 and acylcarnitines by selected precursor ions with m/z 85 as the common product ion, as described earlier by Derks et al. (2008). For free carnitine, a daughter-mass with an m/z 103 was chosen because it had a higher intensity than the m/z 85 fragment. Data evaluation was performed with Chemoview® 3.0 software (AB Sciex, Framingham, USA).
Reference values were calculated using a reference population, consisting of 1,600 subjects out of the total population of 2,194 subjects. The other 594 samples derived from subjects diagnosed with an inborn error of metabolism, mainly fatty acid oxidation disorders, organic acidurias, phenylketonuria, glycogen storage diseases, and urea cycle defect, or originated from subjects who underwent a function test, i.e., fasting, glucose tolerance, or exercise tests. Concentrations of C18:1-ac and C16:0-ac are relatively high compared to the other long-chain acylcarnitines (lc-ac) (see, e.g., Table 1). It was decided to study C0, lc-ac (C16:0-, C16:1-, C18:0-, C18:1-, and C18:2-ac), the sum of lc-ac (∑lc-ac), and the molar ratios C0/(C16:0-ac+C18:0-ac) and C0/∑lc-ac. Age-dependency of these biochemical markers was investigated in the reference population by Spearman rank tests (significant p < 0.05). For each established age category, 95% reference intervals were based on 2.5 and 97.5% percentiles (non-Gaussian distributed metabolites). Normality was assessed using Shapiro–Wilk tests (significant p < 0.05). Receiver Operating Characteristic (ROC) curves were generated from results of patients with CPT-IA deficiency versus all (age-matched) subjects in the total cohort. The values of the biochemical markers to diagnose CPT-IA deficiency were compared using the areas under the curve (AUC). Sensitivity and specificity were calculated at 95% reference limits; sensitivity = true positives (TP)/(TP+false negatives (FN)) and specificity = true negatives (TN)/(TN+false positives (FP)). Analyses were done using Excel 2007 and Analyse-it 2008 (Analyse-it software, Ltd).
Table 1.
Reference values and ROC analyses for biochemical markers of CPT-IA deficiency
| Reference values | ROC analysis | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0–7 days (n = 169) | 8–30 days (n = 106) | 31 days and older (n = 1,325) | 31 days and older (n = 1,803 subjects + patients) | |||||||
| P2.5 | P97.5 | P2.5 | P97.5 | P2.5 | P97.5 | AUC (95% Cl) | Cut-off | Se | Sp | |
| Free camitine (C0) | 6.0 | 33.0 | 7.3 | 41.8 | 13.2 | 55.0 | 0.738 (0.513–0.964) | >55.0 | 25.0% | 96.1% |
| C16:0-ac | 0.04 | 0.40 | 0.03 | 0.24 | 0.04 | 0.22 | 0.992 (0.977–1.000) | <0.04 | 100.0% | 96.7% |
| C16:1-ac | 0.01 | 0.15 | 0.01 | 0.05 | 0.01 | 0.10 | 0.919 (0.827–1.000) | <0.01 | 50.0% | 97.0% |
| C18:0-ac | 0.01 | 0.10 | 0.01 | 0.10 | 0.02 | 0.08 | 1.000 (0.999–1.000) | <0.01 | 100.0% | 97.2% |
| C18:1-ac | 0.03 | 0.33 | 0.04 | 0.38 | 0.05 | 0.43 | 1.000 (1.000–1.000) | <0.05 | 100.0% | 97.4% |
| C18:2-ac | 0.01 | 0.17 | 0.01 | 0.16 | 0.02 | 0.19 | 0.994 (0.983–1.000) | <0.02 | 88.9% | 97.1% |
| Σlc-ac | 0.11 | 1.06 | 0.12 | 0.96 | 0.16 | 0.99 | 1.000 (0.999–1.000) | <0.16 | 100.0% | 96.9% |
| C0/(C16:0-ac+C18:0-ac) | 15 | 306 | 63 | 578 | 63 | 587 | 0.997 (0.990–1.000) | >587 | 100.0% | 96.9% |
| C0/Σlc-ac | 8 | 138 | 21 | 229 | 22 | 216 | 0.999 (0.998–1.000) | >216 | 100.0% | 96.7% |
Reference and corresponding cut-off values were based on the historical subject population, see text. Concentrations of C0, individual long-chain acylcarnitines (lc-ac), and the sum of lc-ac (∑lc-ac) are in μmol/L. Ratios are in mol/mol. All biochemical markers, except C18:1-ac, were significantly related to age (see text). The areas under the curve (AUC) for Receiver Operating Characteristic (ROC) curves and the sensitivity and specificity at 95% cut-off values are presented for subjects and patients ≥31 days of age only (see text)
Results
Reference Values and ROC Analysis of Biochemical Markers
Except for C18:1-ac, all markers were significantly age-dependent (Spearman rank P < 0.001), with correlation coefficients (R) between (−) 0.05 for ∑lc-ac and (+) 0.34 for C0. This relation was most significant in the first 30 days of life. Even though age-dependency remained to be significant after 30 days for C0 and C18:0-ac, their relation with age was considered too small to have a relevant effect on calculated reference values (+0.07 μmol/L per year for C0, and +0.0001 μmol/L per year for C18:0-ac (linear regression analyses)). Reference values were calculated for three age-categories: 0–6 days (n = 169), 7–30 days (n = 106), and ≥31 days (n = 1,325) (Table 1). All biomarkers in these subgroups had non-Gaussian distributions (Shapiro–Wilk p < 0.05).
Plasma and blood spot acylcarnitines concentrations and ratios of patients with CPT-IA deficiency are presented in Table 2. All nine plasma samples had normal concentrations of C0, whereas all had low C18:0-ac, C18:1-ac, and ∑lc-ac concentrations, and high molar ratios. The ROC curves, and the sensitivity and specificity using 95% reference limits are also presented in Table 1. Because all samples of CPT-IA deficient patients were obtained after 30 days of age, only subjects and patients ≥31 days of age were included (n = 1,803/2,194). AUC were highest for C18:1-ac, C18:0-ac, and ∑lc-ac, closely followed by C0/∑lc-ac. Using 95% reference limits, C18:1-ac <0.05 μmol/L had the highest diagnostic value with a sensitivity of 100% and a specificity of 97.4%. Values were slightly less for other lc-ac and ratios, see Table 1. The ratio C0/(C16:0-ac+C18:0-ac) had slightly better specificity at these cut-off values compared to C0/∑lc-ac.
Table 2.
Biochemical marker concentrations of 4 CPT-IA deficient patients
| Case | 1 | 1 | 1 | 1 | 1 | 2 | 3 | 3 | 4 |
|---|---|---|---|---|---|---|---|---|---|
| Gender | m | v | m | v | |||||
| Age (years, days) | 1y, 206d | 1y, 266d | 1y, 268d | 1y, 271d | 1y, 278d | 6y, 22d | 16y, 273d | 19y, 21d | 13y, 53d |
| Plasma (μmol/L) | |||||||||
| C0 | 55.6 | 51.6 | 33.5 | 15.9 | 49.1 | 48.2 | 34.7 | 52.4 | 31.6 |
| C16:0-ac | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.04 | 0.02 | 0.01 |
| C16:1-ac | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.01 | 0.01 | 0.00 |
| C18:0-ac | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.00 | 0.00 |
| C18:1-ac | 0.01 | 0.00 | 0.00 | 0.01 | 0.01 | 0.02 | 0.02 | 0.02 | 0.02 |
| C18:2-ac | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.01 | 0.01 | 0.02 |
| Σlc-ac | 0.04 | 0.01 | 0.01 | 0.02 | 0.02 | 0.05 | 0.09 | 0.06 | 0.05 |
| C0/(C16:0-ac+C18:0-ac) | 3,087 | 5,160 | 3,350 | 1,590 | 4,910 | 4,818 | 694 | 2,618 | 3,160 |
| C0/(Σlc-ac) | 1,592 | 5,160 | 3,350 | 795 | 2,455 | 964 | 385 | 873 | 632 |
| Blood spot (μmol/L) | |||||||||
| C0 | >100 | 197 | 270 | 228 | |||||
| C16:0-ac | 0.00 | 0.10 | 0.10 | ||||||
| C16:1-ac | 0.00 | 0.00 | 0.00 | ||||||
| C18:0-ac | 0.00 | 0.00 | 0.00 | ||||||
| C18:1-ac | 0.00 | 0.00 | 0.00 | ||||||
| C18:2-ac | 0.00 | 0.10 | 0.00 | ||||||
| C0/(C16:0-ac+C18:0-ac) | >5,000 | 1,350 | 2,280 | ||||||
| C0/(Σlc-ac) | >5,000 | 1,350 | 2,280 | ||||||
| Enzyme Activity (nmol min−1 mg−1 protein)a | 0.08 | 0.08 | 0.15 | 0.15 | |||||
| DNA mutations | c.1241C>T | c.1318G>A | c.1318G>A | c.1318G>A | |||||
| (A414V) | (A440T) | (A440T) | (A440T) | ||||||
This table shows concentrations of C0, individual long-chain acylcarnitines (lc-ac), the sum of lc-ac (∑lc-ac), and ratios for 9 samples of 4 CPT-IA deficient patients in plasma and blood spots together with lymphocyte CPT-IA enzyme activities and results of DNA mutation analysis. Two plasma samples of case 1 had non-detectable concentrations of C18:1-ac. In Fig. 1, these samples were presented at a C18:1-ac concentration of 0.005 μmol/L. The blood spot C0 concentration of case 1 was obtained from the Dutch neonatal screening program (see text). Patients were homozygous for mentioned DNA mutations
aReference values lymphocyte CPT-IA activity: 0.2–0.8 nmol min−1 mg−1 protein
Using a combination of two markers, i.e., a concentration in plasma of C18:1-ac <0.05 μmol/L and a molar ratio of C0/(C16:0-ac+C18:0-ac) >587, specificity to diagnose CPT-IA deficiency increased to 99.3% compared with either C18:1-ac (97.4%) or C0/(C16:0-ac+C18:0-ac) (96.9%) alone, all at a sensitivity of 100%.
Characteristics of Subjects with False-Positives Test Results
Figure 1 shows the values of the 2 biochemical markers: C18:1-ac and C0/(C16:0-ac+C18:0-ac) for all subjects and patients ≥31 days of age, including 5 follow-up samples of CPT-IA deficient patients. Forty-eight subjects had false-positive test results for C18:1-ac. Ten of them were diagnosed with an inborn error of metabolism other than CPT-IA deficiency, i.e., phenylketonuria (n = 4), medium chain acyl-CoA dehydrogenase (MCAD) deficiency (n = 4), biotinidase deficiency (n = 1), and molybdenum cofactor deficiency (n = 1). Two out of four patients with MCAD deficiency and the patient with biotinidase deficiency had low concentrations of the sum of all carnitine-containing species in plasma (total carnitine). Six patients without a diagnosis also had low total carnitine concentrations. None of the 48 subjects had a high total carnitine concentration.
Fig. 1.
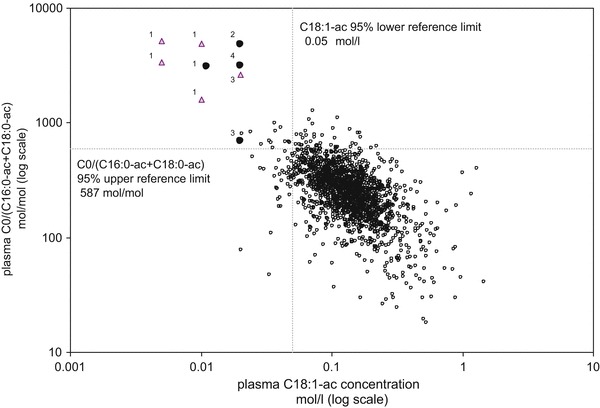
Presents the results of C18:1-ac versus C0/(C16:0-ac+C18:0-ac) of all subjects and patients ≥31 days of age, including the first (circles) and follow-up (triangles) samples of four patients with CPT-IA deficiency [n = 1,808 (1,803 + 5 follow-up samples)]. The numbers correspond to the case numbers presented in Table 2. The dashed lines indicate the 95% cut-off limits for C18:1-ac (0.05 μmol/L) and C0/(C16:0-ac+C18:0-ac) (587). The quadrant limited by C18:1-ac <0.05 μmol/L and C0/(C16:0-ac+C18:0-ac) >587 had a sensitivity of 100% and a specificity of 99.3% for diagnosis of CPT-IA deficiency in our study population
Fifty-six subjects out of the total population had a false-positive test results for the molar ratio C0/(C16:0-ac+C18:0-ac). Nineteen of them were diagnosed with an inborn error of metabolism other than CPT-IA deficiency, i.e., phenylketonuria (n = 11), glycogen storage disease 1A (n = 2), multiple acyl-CoA dehydrogenase deficiency (n = 1), MCAD deficiency (n = 1), molybdenum cofactor deficiency (n = 1), Smith–Lemli–Opitz syndrome (n = 1), pyruvate dehydrogenase deficiency (n = 1), and OCTN2 deficiency (n = 1). The MCAD deficient patient and ten subjects without a diagnosis had plasma total carnitine concentrations above the 95% percentile of their reference age group. None of the 56 subjects had a low total carnitine concentration in plasma.
Twelve patients had a false-positive test result when using the combination of a low C18:1-ac and a high molar ratio of C0/(C16:0-ac+C18:0-ac), of whom three had phenylketonuria and one had molybdenum cofactor deficiency. Of the other eight subjects, one was clinically suspected to suffer from CPT-IA deficiency. This was ruled out by CPT-IA enzyme activity measurements. All patients and subjects had a normal concentration of total carnitine.
Biochemical Markers of CPT-IA Deficiency in Neonatal Blood Spot
Acylcarnitine profiles were analyzed in blood spots obtained at the time of presentation in three of our patients with CPT-IA deficiency (cases 2–4). In these patients, C0 concentrations were above 197 μmol/L, whereas all lc-ac concentrations were below 0.02 μmol/L (Table 2). For case 1 the neonatal blood spot was requested at the Dutch neonatal screening authority. In this bloodspot, the concentration of C0 was found to be >100 μmol/L. In controls, C0 concentrations are lower and lc-ac concentrations are higher in blood spots compared to plasma (de Sain-van der Velden et al. 2013). This implies that these values are highly abnormal, even when applying plasma reference values.
Discussion
The results clearly show that CPT-IA deficiency can easily be overlooked when using the classical biomarker, i.e., high concentrations of C0 in plasma, as the sole criterion for diagnosis, as previously reported (Bergman 1994; Bonnefont et al. 2004; Innes et al. 2000; Primassin and Spiekerkoetter 2010; de Sain-van der Velden et al. 2013). Blood spot acylcarnitines, in particular the ratio of C0/(C16:0-ac+C18:0-ac), seem to have high diagnostic value for CPT-IA deficiency (Fingerhut et al. 2001; McHugh et al. 2011; Primassin and Spiekerkoetter 2010; de Sain-van der Velden et al. 2013; Sim et al. 2001). Even though our data support this suggestion, the present study also demonstrates that plasma, the more commonly used matrix, can be used for this purpose as well. The current study confirmed the high sensitivity of the molar ratio of C0/(C16:0-ac+C18:0-ac) in plasma, as proposed earlier (de Sain-van der Velden et al. 2013). Sensitivity of low plasma lc-ac concentrations was, however, equally high. When taking specificity under consideration, the diagnostic value became much better when using the combination of a high molar ratio of C0/(C16:0-ac+C18:0-ac) >587 mol/mol ánd a low C18:1-ac <0.05 μmol/L (cut-offs for patients ≥31 days of age). For low prevalent diseases such as CPT-IA deficiency, the increase of specificity from 97 to 99% has a dramatic effect on the positive predictive value of a test. In our selected population with a prevalence of 0.0018 (4/2,194), the positive predicted value increases from 7 to 25%.
ROC curve analysis is a well-known tool to compare diagnostic values of biomarkers. Using this analysis (Table 1), plasma C18:0-ac, C18:1-ac, and ∑lc-ac had highest and similar diagnostic values (AUC 1.000), closely followed by C0/∑lc-ac (AUC 0.999). To reduce the chance of missing a CPT-IA deficient patient, fixed cut-off values based on 95% reference values were evaluated. Using these cut-offs, the molar ratio C0/∑lc-ac was equally sensitive, but slightly less specific than the originally proposed molar ratio, i.e., C0/(C16:0-ac+C18:0-ac). False-positive results for C0/(C16:0-ac + C18:0-ac) were explained by high total carnitine concentrations in 11 of 56 patients and subjects. On the other hand, low C18:1-ac concentrations were associated with low total carnitine concentrations in 9 of 48 patients. Combining both markers significantly diminished the number of false positives, partly due to low or high carnitine concentrations. False-positive results were not clearly associated with other (known) inborn errors of metabolism. Three patients with phenylketonuria (3/100 in this cohort) had low C18:1-ac concentrations in combination with high C0/(C16:0-ac+C18:0-ac) ratios. After reviewing other acylcarnitine profiles of the three patients, only one sample, i.e., the first sample, had this remarkable result. The reason for this is unknown.
An important limitation of this study is the relatively low number of patients with CPT-IA deficiency. Plasma acylcarnitines have high intra-individual biological variation, depending on nutritional status (fasting), exercise, and clinical condition. The large biological variation may impair the diagnostic value of the markers. This is illustrated by the results of case 3. The analysis of the first sample gave near-normal values of the molar ratios of C0/(C16:0-ac+C18:0-ac) and C0/∑lc-ac, whereas the analysis of the second sample gave clearly abnormal values, see Table 2. When reviewing all nine plasma samples of our CPT-IA deficient patients (see Table 2 and Fig. 1), it is clear that the suggested cut-off values are highly sensitive and that the diagnostic value of the (combination of) markers is not dependent on the clinical condition and treatment regimens of our patients. When reviewing six plasma C0 concentrations and C0/(C16:0-ac+C18:0-ac) ratios of the 2 CPT-IA deficient patients presented in the study of de Sain-van der Velden et al. (2013), all the samples of these patients would probably also have been identified using the proposed marker combination. It is nevertheless important to investigate these markers in a larger cohort of CPT-IA deficient patients.
Conclusion
CPT-IA deficiency is rare and can easily be overlooked by relying on the classical marker, i.e., high plasma C0 concentrations. It has therefore been suggested to use blood spots acylcarnitines for diagnosis of CPT-IA deficiency. This study, however, demonstrates that CPT-IA deficiency can be diagnosed reliably with high sensitivity and specificity in plasma by a combination of a low C18:1-ac concentration and a high molar ratio of C0/(C16:0-ac+C18:0-ac). It is advised to further validate these markers in a larger cohort of patients with CPT-IA deficiency.
Acknowledgements
We gratefully acknowledge mw A.J. van Assen-Bolt, A. Gerding, and mw K. Boer for acylcarnitine analyses. Moreover, we thank mw A.J. van Assen-Bolt for her help in creating the acylcarnitine database.
Authors’ Contributions
MR Heiner-Fokkema was responsible for the study design, statistical analyses, and writing of this manuscript. FJ van Spronsen and D-J Reijngoud made significant contributions to the conception and design of this study. Data from CPT-I deficient patients were supplied by MR Heiner-Fokkema, FM Vaz, R Maatman, and LAJ Kluijtmans. All authors critically read the manuscript and gave final approval for this version.
Conflicts of Interest
MR Heiner-Fokkema, FM Vaz, R Maatman, LAJ Kluijtmans, FJ van Spronsen, and D-J Reijngoud declare that they have no conflicts of interest regarding the topic in this manuscript.
Take-Home Message
Carnitine palmitoyltransferase IA deficiency can be diagnosed reliably by plasma acylcarnitine analysis.
Compliance with Ethics Guidelines
All procedures followed were in accordance with the ethical standards of the medical ethical committee on human experimentation (institutional and national) and with the Helsinki.
Footnotes
Competing interests: None declared
Contributor Information
M. Rebecca Heiner-Fokkema, Email: m.r.heiner@umcg.nl.
Collaborators: Matthias R. Baumgartner, Marc Patterson, Shamima Rahman, Verena Peters, Eva Morava, and Johannes Zschocke
References
- Bergman AJ, Donckerwolcke RA, Duran M et al (1994) Rate-dependent distal renal tubular acidosis and carnitine palmitoyltransferase I deficiency. Pediatr Res 36:582–588 [DOI] [PubMed]
- Bonnefont JP, Djouadi F, Prip-Buus C, Gobin S, Munnich A, Bastin J. Carnitine palmitoyltransferases 1 and 2: biochemical, molecular and medical aspects. Mol Aspects Med. 2004;5:495–520. doi: 10.1016/j.mam.2004.06.004. [DOI] [PubMed] [Google Scholar]
- Derks TG, Boer TS, van Assen A, et al. Neonatal screening for medium-chain acyl-CoA dehydrogenase (MCAD) deficiency in The Netherlands: the importance of enzyme analysis to ascertain true MCAD deficiency. J Inherit Metab Dis. 2008;31:88–96. doi: 10.1007/s10545-007-0492-3. [DOI] [PubMed] [Google Scholar]
- de Sain-van der Velden MG, Diekman EF, Jans JJ, et al. Differences between acylcarnitine profiles in plasma and blood spots. Mol Genet Metab. 2013;110:116–121. doi: 10.1016/j.ymgme.2013.04.008. [DOI] [PubMed] [Google Scholar]
- Fingerhut R, Röschinger W, Muntau AC, et al. Hepatic carnitine palmitoyltransferase I deficiency: acylcarnitine profiles in blood spots are highly specific. Clin Chem. 2001;47:1763–1768. [PubMed] [Google Scholar]
- Innes AM, Seargeant LE, Balachandra K, et al. Hepatic carnitine palmitoyltransferase I deficiency presenting as maternal illness in pregnancy as maternal illness in pregnancy. Pediatr Res. 2000;47:43–45. doi: 10.1203/00006450-200001000-00010. [DOI] [PubMed] [Google Scholar]
- McHugh D, Cameron CA, Abdenur JE, et al. Clinical validation of cutoff target ranges in newborn screening of metabolic disorders by tandem mass spectrometry: a worldwide collaborative project. Genet Med. 2011;13:230–254. doi: 10.1097/GIM.0b013e31820d5e67. [DOI] [PubMed] [Google Scholar]
- Primassin S, Spiekerkoetter U. ESI-MS/MS measurement of free carnitine and its precursor gamma-butyrobetaine in plasma and dried blood spots from patients with organic acidurias and fatty acid oxidation disorders. Mol Genet Metab. 2010;101:141–145. doi: 10.1016/j.ymgme.2010.06.012. [DOI] [PubMed] [Google Scholar]
- Sim KG, Wiley V, Carpenter K, Wilcken B. Carnitine palmitoyltransferase I deficiency in neonate identified by dried blood spot free carnitine and acylcarnitine profile. J Inherit Metab Dis. 2001;24:51–59. doi: 10.1023/A:1005606805951. [DOI] [PubMed] [Google Scholar]
- Stanley CA, Sunaryo F, Hale DE, Bonnefont JP, Demaugre F, Saudubray JM. Elevated plasma carnitine in the hepatic form of carnitine palmitoyltransferase-1 deficiency. J Inherit Metab Dis. 1992;15:785–789. doi: 10.1007/BF01800021. [DOI] [PubMed] [Google Scholar]


