Abstract
Previously, we reported that Alzheimer’s disease (AD) epitope vaccines (EVs) composed of N-terminal β-amyloid (Aβ42) B cell epitope fused with universal foreign T helper (Th) epitope(s) were immunogenic, potent, and safe in different amyloid precursor protein (APP) transgenic mice with early AD-like pathology. However, developing an effective therapeutic vaccine is much more challenging, especially when a self-antigen such as Aβ42 is a target. Here, we directly compare the efficacy of anti-Aβ42 antibodies in Tg2576 mice with low or high levels of AD-like pathology at the start of immunizations: 6–6.5 months for preventive vaccinations and 16–19 months for therapeutic vaccinations. EV in a preventive setting induced high levels of anti-Aβ antibodies, significantly reducing pathologic forms of Aβ in the brains of Tg2576 mice. When used therapeutically for immunesenescent Tg2576 mice, EV induced low levels of antibodies not sufficient for clearing of AD-like pathology. Separately, we demonstrated that EV was also not effective in 11–11.5-month-old Tg2576 mice with moderate AD-like pathology. However, we augmented the titers of anti-Aβ antibodies in transgenic (Tg) mice of the same age possessing the pre-existing memory Th cells and detected a significant decrease in diffuse and core plaques in cortical regions compared to control animals along with improved novel object recognition performance.
Keywords: Alzheimer’s disease, amyloid-β, epitope vaccine, preventive and therapeutic vaccines, immune responses, Aβ-pathology, inflammation, microhemorrhages, novel object recognition
Petrushina et al. show that Alzheimer’s disease epitope vaccine generated high titers of anti-Aβ antibodies and reduced brain pathology in young, but not middle-age and old, Tg2576 mice. Pre-existing memory Th cells augmented antibody titers in middle-age Tg2576 mice, decreasing Aβ pathology and improving novel object recognition performance.
Introduction
A critical goal of developing therapeutic interventions for Alzheimer’s disease (AD) has been the identification of suitable targets. During the last two decades, the predominant theory of the etiology of AD was that Aβ has a central role in the onset and progression of AD, as delineated in the amyloid cascade hypothesis.1, 2 According to this hypothesis, the accumulation of Aβ peptide, either by overproduction or aberrant clearance, results in deposition of Aβ in plaques, which leads to the formation of neurofibrillary tau tangles and cell death, resulting in dementia. Later on, the amyloid cascade hypothesis evolved to focus on oligomers and protofibrils of Aβ as instigators in the destruction of synaptic function.3, 4, 5 Support for the amyloid cascade hypothesis was spurred by the identification of mutations in amyloid precursor protein (APP) that are associated with familial AD6, 7, 8 or protection against amyloid pathology and age-related Alzheimer’s disease.9, 10, 11 In addition, it was discovered that overexpression of APP due to trisomy 21 in Downs disease was associated with a high incidence of AD in these individuals.12, 13, 14 Today it is clear that the pathological tau also plays a vital role in the development of AD pathology and progression of this disease,15 correlating better with the degree of dementia than Aβ plaques. Importantly, substantial data suggest that Aβ pathology emerges many years prior to tau pathology and accelerates formation of toxic tau aggregates,16, 17, 18 supporting the preventive rather than therapeutic potential of anti-amyloid therapies.
Immunotherapies targeting Aβ are aimed at reducing deposits of this peptide in the brain, and/or blocking the assembly of this peptide into pathological forms19, 20 that may disrupt cognitive function.21, 22, 23 Because immunotherapeutic strategies displayed great promise in animal models of AD, a strong effort was made by the industry to inhibit generation of toxic Aβ aggregates and remove soluble and aggregated Aβ deposited in the brains of AD patients by both active and passive anti-Aβ immunotherapy strategies.24, 25, 26, 27, 28, 29, 30, 31 Data from active vaccine trials indicate that, to be effective, anti-Aβ therapeutic should induce high titers of anti-Aβ antibodies without activation of autoreactive T cells.31, 32, 33, 34 On the other hand, published results from both active and passive Aβ-immunotherapy suggest that it should be initiated early in the disease, probably before toxic forms of this peptide accumulate in the brain.30, 35, 36 In fact, we recently pointed out that active Aβ-vaccines tested in clinical trials so far have produced ambivalent results due to their inadequate immunogenicity.31 To support our position and suggest an effective future clinical trials protocol, we decided for the first time to test the efficacy of our protein-based epitope vaccine (EV) composed of three copies of immunogenic N-terminal epitope of Aβ37, 38, 39, 40, 41 and two Th epitopes from tetanus toxoid (TT) in preventive and therapeutic settings in parallel. More specifically, to simulate disease stages in humans (i.e., prodromal, mild to moderate, and severe AD), we studied the immunogenicity and efficacy of EV (reduction of Aβ pathology and cognitive impairments) in Tg2576 mice that, at the start of vaccination, possess low, moderate, and high AD-like pathology.
Results
Comparison of Efficacy of Preventive and Therapeutic EV
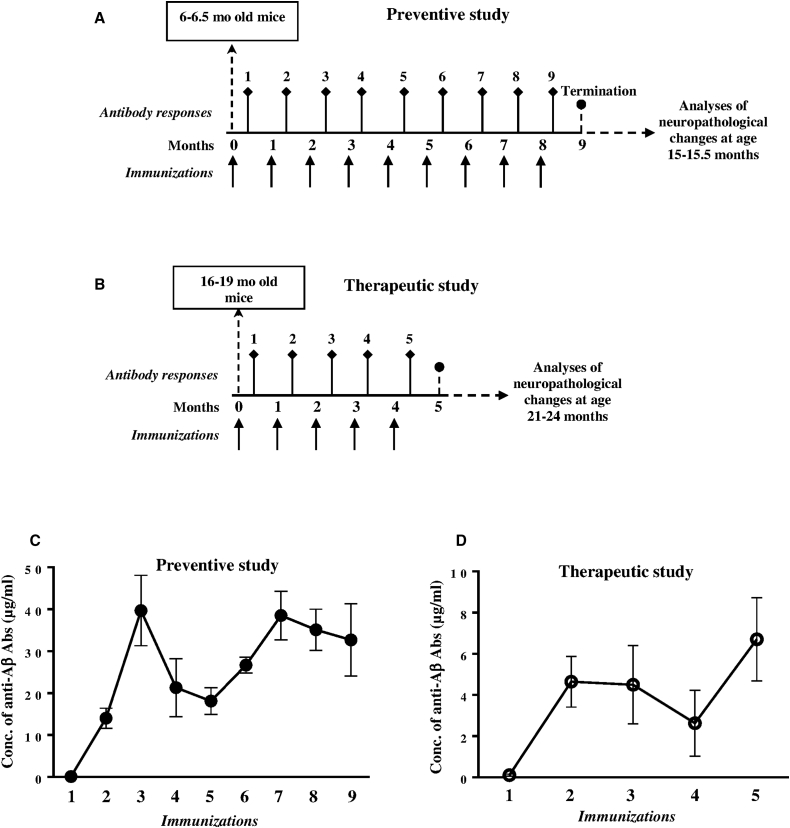
Previously, we reported that preventive EV induced anti-Aβ antibodies that inhibited accumulation of AD-like pathology in the brains of aged Tg2576 and 3×Tg-AD mice that, at the start of vaccination, possessed low levels of AD-like pathology.40, 42, 43 In this study, we decided to compare the efficacy of EV targeting the N terminus of Aβ in preventive and therapeutic settings in parallel. More specifically, we started immunizations of Tg2576 mice at a time when they possessed low (at 6–6.5 months old) and high (at 16–19 months old) levels of AD-like pathology, respectively (Figures 1A and 1B). As expected from our previous studies, in preventive settings, EV induced robust humoral immune responses; the anti-Aβ antibody concentrations in vaccinated mice reached the maximum titers after three immunizations (39.7 ± 8.4 μg/mL), declined after the fourth and fifth, and augmented again after subsequent immunizations (Figure 1C). The kinetics of humoral immune responses after therapeutic vaccinations were somewhat similar to the data obtained in the preventive setting, but the levels of anti-Aβ antibody concentrations were significantly less due to age-related immunosenescence and reached the maximum (6.7 ± 2.02 μg/mL) after the last immunization (Figure 1D).
Figure 1.
Immunization Design of Tg2576 Mice and Humoral Immune Responses Specific to Amyloid
(A) Preventive study: 6–6.5-month-old Tg2576 mice (n = 8) were immunized with adjuvanted EV. Control mice were injected with adjuvant alone (n = 6). (B) Therapeutic study: 16–19-month-old Tg2576 mice (n = 6) were immunized with adjuvanted EV. Control mice were injected with adjuvant alone (n = 5). (C and D) Concentrations of anti-Aβ antibodies in sera of EV immunized Tg2576 mice in preventive (C) and therapeutic (D) studies analyzed by ELISA.
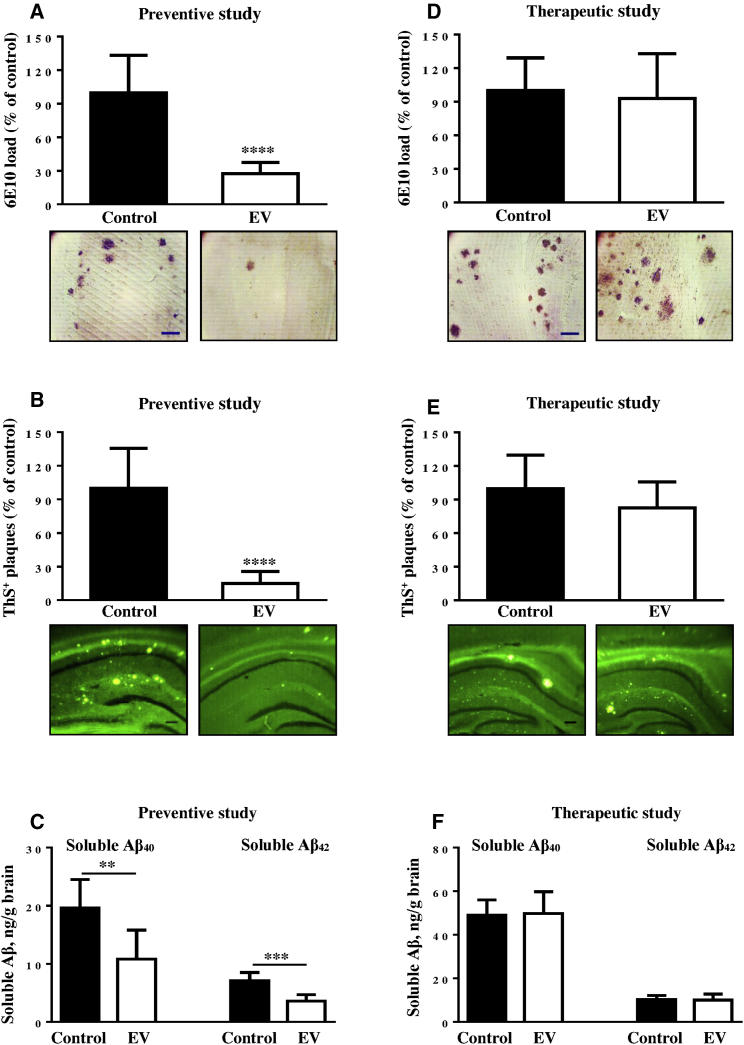
The differences in the levels of immunogenicity observed in preventive and therapeutic studies raised a question about the efficacy of EV in clearance of AD-like pathology in the brains of vaccinated Tg2576 mice used in these two studies. Accordingly, we studied neuropathological changes in the brains of 15–15.5-month-old and 21–24-month-old Tg2576 mice from preventive and therapeutic studies, respectively (Figure 2). As expected, the preventive vaccination significantly decreased the percent of diffuse and cored plaques detected by 6E10-immunostaining and ThS binding (Figures 2A and 2B). Current data suggest that soluble (or diffusible) Aβ oligomers mediate different toxic pathways in AD, such as tau hyperphosphorylation, synaptic dysfunction, impairment of memory, and neuronal death.5, 44, 45, 46, 47 We analyzed the levels of soluble Aβ40/42 peptides in the homogenates of brains of vaccinated and age-matched control Tg2576 mice. Data presented in Figure 2C revealed a significant reduction of both Aβ42 and Aβ40 soluble molecules in homogenates isolated from the brains of EV-immunized Tg2576 mice compared to that isolated from the brains of control animals injected with adjuvant alone.
Figure 2.
Anti-Aβ Antibody Induced by EV Inhibited AD-like Pathology in Mice from Preventive, but Not Therapeutic, Studies
(A, B, D, and E) Aβ load (A and D) and ThS-positive cored Aβ plaques (B and E) in the hemibrains of vaccinated (n = 8) and control mice (n = 6) in preventive (A and B) study and vaccinated (n = 6) and control (n = 5) mice in therapeutic study (D and E). (C and F) The levels of soluble Aβ40 and Aβ42 in the brain homogenates of mice used in preventive (C) and therapeutic (F) studies. Photomicrographs show representative images of hemibrain regions of immunized and control mice stained with 6E10 (scale bar, 100 μm) and ThS (scale bar, 200 μm). Error bars represent average ± SD (∗∗p < 0.01; ∗∗∗p < 0.001; ∗∗∗∗p < 0.0001).
In sharp contrast with preventive studies described above, data obtained in therapeutic studies demonstrated that the percent of 6E10-positive diffuse and ThS-positive cored plaques in the brains of vaccinated mice was equal to that in the same brain regions of age-matched control animals (Figures 2D and 2E). Next, we analyzed the possible effect of therapeutic vaccination on the levels of soluble Aβ40/42 deposits in the homogenates of brains of aged Tg2576 mice. Immunizations with EV did not change the levels of these soluble pathological molecules in the brains of vaccinated animals compared to the Aβ40/42 level in the brains of unvaccinated (adjuvant injected) control Tg2576 mice (Figure 2F).
In summary, a comparison of therapeutic and preventive vaccination strategies indicated that only the latter induced anti-Aβ antibodies at the levels sufficient for reducing Aβ pathology. In the therapeutic setting, no pathology was reduced, probably due to the low levels of anti-Aβ antibodies and/or more severe pathology that developed in the brains of old Tg2576 mice.
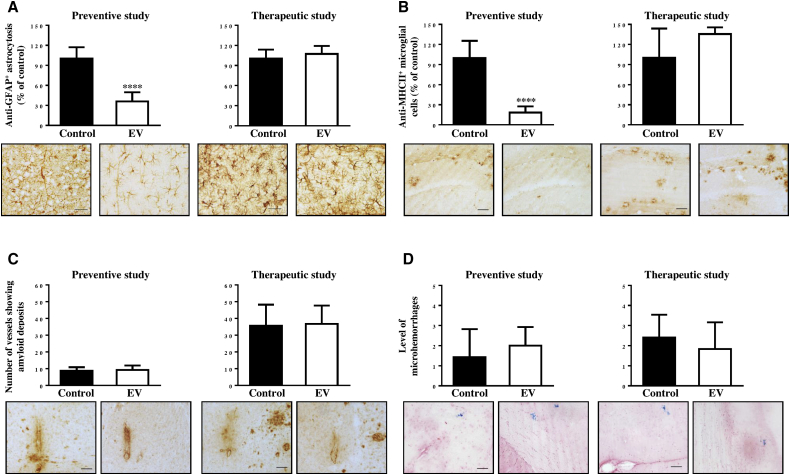
Vaccination with EV Reduced Glial Activation without Increase of Cerebral Amyloid Angiopathy
To compare the inflammation-related pathology in the brains of animals used for therapeutic versus preventive vaccinations studies, we examined astrocyte (glial fibrillary acidic protein, GFAP) and microglial (MHC class II) activation (Figures 3A and 3B ). Analysis of the astrocyte-specific marker in the brains of EV immunized mice showed the presence of fewer astrocytes as compared to the control group in mice from preventive, but not therapeutic, studies (Figure 3A). Quantitative image analysis of MHC class II-positive cells (I-Ab/I-Eb) also demonstrated significantly fewer activated (MHC class II+) microglial cells in Tg2576 mice used for preventive, but not therapeutic, studies (Figure 3B). Thus, preventive vaccination induced therapeutically effective concentrations of anti-Aβ antibodies that reduced AD-like pathology in the brains of 15–15.5-month-old Tg2576 mice, while therapeutic vaccination initiated in 16–19-month-old mice of the same strain was not effective at the time of termination at age 21–24 months.
Figure 3.
EV Vaccination Induced Less Activation of Astrocytes and Microglia in Young Tg2576 Mice (Preventive Study), but Not in Aged Tg2576 Mice (Therapeutic Study)
(A and B) Image analysis of hemibrains performed after immunostaining with anti-GFAP antibody (astrocytosis; A) and anti-MHC class II antibody (glial activation; B). (C) Number of vessels positive for amyloid deposits was calculated in the brains of experimental and control mice (scale bar, 50 μm). (D) Number of microhemorrhages in the hemibrains of vaccinated and control animals. Photomicrographs show representative images of hemibrain regions of immunized and control mice. Scale bars, 20 μm (A) and 50 μm (B–D). Error bars represent average ± SD (∗∗∗∗p < 0.0001).
Additionally, we assessed the effect of preventive and therapeutic vaccinations with EV on vascular deposition of Aβ and microhemorrhages in the brain sections of Tg2576 mice. As expected from published reports,48 we observed an increase in cerebral amyloid angiopathy (CAA), and slightly more microhemorrhages occurred with aging in the brains of mice from therapeutic (21–24 months old) versus preventive (16–19 months old) studies. Importantly, no differences in CAA and microhemorrhages were observed in the brains of vaccinated animals compared to age-matched control mice in both studies (Figures 3C and 3D). Thus, immunization with EV did not exacerbate CAA in either the preventive or therapeutic studies, but it had a positive impact on glial activation in mice that, at the start of vaccination, possessed early (low levels) AD-like pathology, but not mice that had late (established or high levels) AD-like pathology.
The Role of Pre-existing Memory Th Cells in Immunological and Therapeutic Efficacy of AD Vaccine
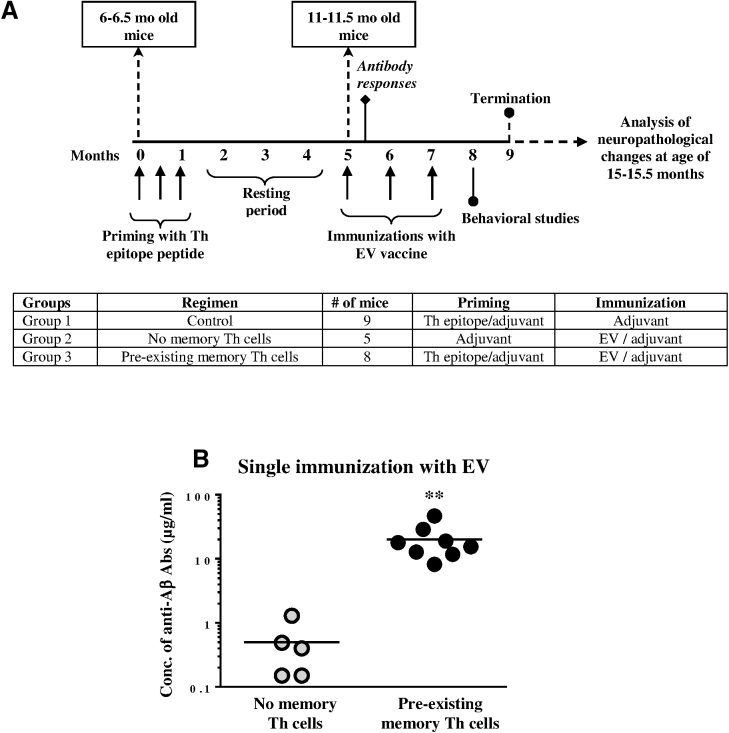
Data from the active and passive clinical trials somewhat suggested that an anti-Aβ immunotherapy could be effective if it was initiated before the accumulation of high levels of toxic forms of the Aβ peptide in the brain and the vaccine was sufficiently immunogenic and able to induce high anti-Aβ antibody titers.30, 31, 49, 50 Here we decided to test the efficacy of EV in Tg2576 mice with pre-existing memory Th cells, which should have helped to generate higher titers of antibodies quickly after the start of vaccinations (Figure 4A). In order to generate pre-existing memory Th cells, Tg2576 experimental and control mice possessing early AD-like pathology were immunized three times with an adjuvant-formulated peptide representing the Th epitope only (priming). Another control group of mice was injected with adjuvant only. To show the role of pre-existing memory Th cells in generation of anti-Aβ antibodies after 3 months of resting period, experimental (primed) and non-primed control groups of mice were immunized with EV formulated in adjuvant, while the primed control group of mice continued to receive injections of adjuvant only (Figure 4A). Of note, at the time of EV vaccination, mice were at middle age (11–11.5 months old) and already possessed moderate AD-like pathology. Detection of antibodies in sera after the first immunization with EV demonstrated that a single immunization of experimental mice with this vaccine induced robust anti-Aβ antibody responses. The vaccinated mice generated high concentrations of anti-Aβ antibodies (20.07 ± 12.43 μg/mL) that were significantly higher (p < 0.05) than concentrations of antibodies detected in vaccinated, but not primed, control mice (Figure 4B). Of course, another control group of mice injected only with adjuvant did not generate anti-Aβ antibodies at all (data not shown).
Figure 4.
Immunization Design for Tg2576 Mice and Humoral Immune Responses Specific to β-Amyloid in Mice with or without Pre-existing Memory T Cells
(A) 6–6.5month-old Tg2576 mice were primed with adjuvanted Th epitope peptide (group 1; n = 9 and group 3; n = 8) or injected with adjuvant (group 2; n = 5) three times. After 3 months of resting, mice from groups 2 and 3 were immunized with EV, and group 1 (control group) was injected with adjuvant only. (B) Antibody responses were measured after the first immunization with EV. Lines indicate the average (∗∗p < 0.01).
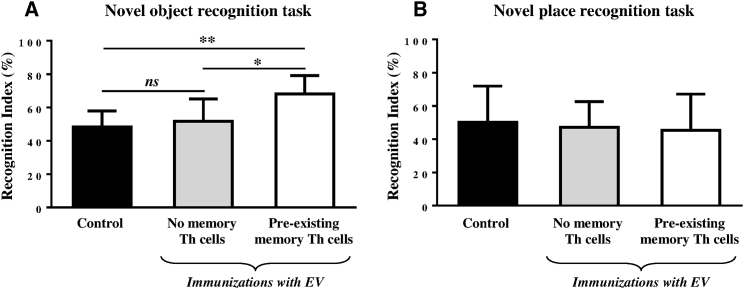
To study how the immunization with EV affects cognition in the presence or absence of pre-existing memory Th cells, Tg2576 mice received two additional vaccinations and were tested in novel object and novel place recognition tasks, the behavior tests that depend on certain areas of the bran typically affected by AD pathology.51, 52, 53 The data in Figure 5A show that priming with Th epitope peptide 4 months before the immunization with EV resulted in a significant increase in the exploration of a non-familiar object as indicated by the increased recognition index compared with mice immunized with EV alone or control mice (p < 0.05 and p < 0.001, respectively). On the contrary, immunization with EV resulted in no significant changes in novel place recognition task performance, even in the presence of the pre-existing memory T cells (Figure 5B).
Figure 5.
Cognitive Behavior of 15–15.5-Month-Old Tg2576 Mice with or without Pre-existing Memory Th Cells after Immunization with EV Vaccine
(A) Mice with pre-existing memory Th cells (n = 8) showed significantly increased recognition of novel object in novel object recognition task after immunization with EV compared to mice without memory Th cells (∗p < 0.05, n = 5) and control mice (∗∗p < 0.01, n = 9). (B) No significant changes in novel place recognition task performance were demonstrated regardless of presence or absence of pre-existing memory Th cells.
It is believed that the novel object and novel place recognition performances are primarily dependent on cortex and hippocampus, though these two regions are differentially involved in recognition memory consolidation.54, 55, 56, 57
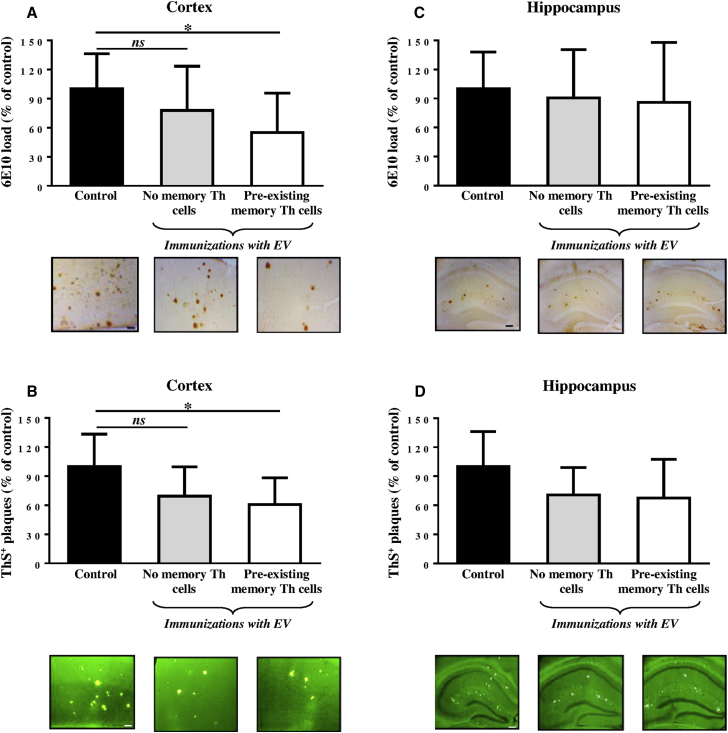
Accordingly, as a pre-clinical test of the therapeutic efficacy of EV vaccination, we studied neuropathological changes in the brains of aged Tg2576 mice (average 15–15.5 months old) possessing or not possessing the pre-existing memory Th cells prior to vaccinations. We tested numbers of 6E10 (diffuse and cored)- and ThS (cored)-positive plaques in the cortex and hippocampus of vaccinated mice with or without memory Th cells and the control adjuvant-only injected mice (Figure 6). We separately compared the Aβ loads in the hippocampal area known for its involvement in the novel place recognition versus the cortical areas, namely frontal and parahippocampal, the latter known for its importance for novel object recognition ability. We detected a slight, but significant, decrease in both 6E10 and ThS-positive plaques in cortical regions of hemibrains of experimental mice possessing the pre-existing memory Th cells at the start of immunization with EV containing the Aβ B cell epitope compared to control vaccinated animals (Figures 6A and 6B). No differences between numbers of diffuse or cored Aβ-plaques were detected between the two control groups, one was vaccinated with EV and another was injected with adjuvant only (Figures 6A and 6B). Importantly, our data showed no reduction in diffuse and cored Aβ-plaques in the hippocampal regions of the brains obtained from both groups of vaccinated mice versus brains isolated from control Tg2576 mice (Figures 6C and 6D). Hence, EV vaccination reduced the levels of 6E10 and ThS-positive Aβ-plaques only in cortical, but not in hippocampal, brain regions of vaccinated mice, and these results supported behavioral data presented in Figure 5.
Figure 6.
AD-like Pathology in the Hemibrain Cortical or Hippocampal Regions of 15–15.5-Month-Old Tg2576 Mice with or without Pre-existing Memory Th Cells Vaccinated with EV
(A and C) Aβ load in the hemibrain cortical (A) and hippocampal (C) regions of vaccinated mice with pre-existing memory Th cells (n = 8) and age-matched control animals (n = 9), and vaccinated mice without pre-existing memory Th cells (n = 5). (B and D) ThS-positive core Aβ plaques in the hemibrain cortical (B) and hippocampal (D) regions of vaccinated mice with pre-existing memory Th cells (n = 8) and age-matched control animals (n = 9), and vaccinated mice without pre-existing memory Th cells (n = 5). Photomicrographs show representative images of hemibrain regions of immunized and control mice. Scale bars, 100 μm (A and B) and 200 μm (C and D). Error bars represent average ± SD (∗p < 0.05).
Discussion
Previous studies in transgenic mouse models of AD-like amyloidosis have demonstrated that both passive and active immunotherapeutic approaches can inhibit the formation of Aβ-containing plaques and reduce behavioral deficits.58, 59, 60, 61 Not surprisingly, active and passive immunotherapy targeting Aβ have been actively pursued as a possible treatment for AD patients.31, 62, 63, 64
More than a decade ago we proposed an AD vaccine strategy37 based on the concept of an EV composed of several copies of an immunodominant self B cell epitope of Aβ combined with universal foreign Th epitopes to provide T cell support for robust antibody production. The various EV designs all induced high levels of anti-Aβ antibodies without activation of potentially harmful autoreactive Th cells in mice, rabbits, and monkeys.40, 65, 66, 67, 68 Importantly, immunizations of mouse models of AD with EV induced therapeutically potent anti-Aβ antibodies that inhibited accumulation of AD-like plaque pathology in the brains of older mice, reduced glial activation, and prevented the development of behavioral deficits in aged animals without increasing the incidence of microhemorrhages.40, 42, 43 In this study, we decided to compare the efficacy of our EV strategy in preventive and therapeutic settings in parallel. Results from these combined studies demonstrated that the preventive, but not therapeutic, vaccination strategy was effective in reduction of AD-like pathology in the brains of aged Tg2576 mice (Figures 2, 3A, and 3B) without increasing the incidence of microhemorrhages and CAA (Figures 3C and 3D). It is likely that the therapeutic strategy was ineffective due to (1) low immunogenicity of EV in older animals with immunosenescence; (2) complexity of the reduction of severe established AD-like pathology, which perhaps is also irreversible in very old mice; or (3) both. Accordingly, we decided to also test the efficacy of EV in middle-aged Tg2576 mice (11–11.5 months old) with moderate AD-like pathology.69 The single injection of these mice with EV did not induce high titers of antibodies, and subsequent injections neither reduced AD-like pathology in the brains nor improved the behavior of vaccinated mice at the age of 15–15.5 months (Figures 4B, 5, and 6). On the contrary, a single vaccination of Tg2576 mice of the same age with pre-existing memory Th cells led to the induction of robust anti-Aβ antibody responses: concentrations of anti-Aβ antibodies were significantly higher (p < 0.001) than that in vaccinated control mice (Figure 4B). Moreover, EV was effective in improving the novel object recognition performance in experimental mice with pre-existing memory Th cells, but not in control vaccinated or non-vaccinated animals (Figure 5). On the other hand, all groups performed poorly on the novel place recognition task. The novel object and novel place recognition tasks are believed to be primarily dependent on both cortex and hippocampus, although the contributions of these two regions are distinct and dissociable.54, 55, 56, 57 Therefore, we analyzed AD-like pathology in the cortical and hippocampal regions of vaccinated mice with and without memory Th cells and control non-vaccinated mice. We observed that immunization with EV was effective in reducing AD-like pathology in the cortex but not in the hippocampus, and this effect depended on pre-existing Th cells (Figure 6).
These data suggest that, at least in these conditions, the object recognition memory is impaired by cortical pathology, and the reduction of cortical-cored and diffuse Aβ plaques could improve the cognitive function in novel object recognition task, whereas hippocampus is responsible for object-in-place discrimination, which shows no improvement. In addition, it is also possible that improved novel object recognition index when hippocampal plaque loads remain unchanged is associated with the reduction of soluble and insoluble oligomeric forms of Aβ.
These observations may have important implications for future immunotherapy clinical trials because active Aβ vaccines have yet to be tested in preclinical or prodromal AD subjects. For example, a recent passive immunotherapy clinical trial with a human anti-Aβ antibody, aducanumab, selective for oligomeric and fibrillar forms of Aβ, found that, in patients with prodromal or mild AD, 1 year of monthly intravenous infusions of aducanumab reduced brain Aβ in a dose- and time-dependent manner.70 Although this was a phase 1 study not powered to show clinical benefit, there was some slowing of clinical decline measured by Clinical Dementia Rating-Sum of Boxes and Mini Mental State Examination scores. These results suggest that, to be effective, Aβ epitope vaccines should be (1) initiated as early as possible in prodromal AD subjects, people with AD risk, or at least in mild AD patients and (2) able to induce therapeutically potent titers of antibodies.31
Analysis of data from multiple immunotherapy studies in APP transgenic mouse models have been informative, although not definitive, with regard to the antibody-mediated mechanism(s) involved in inhibiting cerebral Aβ deposition or clearing pre-existing amyloid plaques.71 Two main hypotheses were proposed to account for clearance of Aβ from the brain by anti-Aβ antibodies. The first, “microglial-mediated hypothesis,” was dependent on the entry of the anti-Aβ antibodies into the central nervous system (CNS), where microglia would facilitate antibody-mediated clearance of CNS amyloid.59 The second hypothesis did not rely on anti-Aβ antibodies crossing the blood brain barrier. Rather, the “peripheral sink hypothesis” proposed by DeMattos et al.72, 73, 74 involved the sequestering of Aβ in an immune complex in the blood, thereby lowering the level of free Aβ, which contributed to a net efflux of Aβ from the brain. They also suggested that therapeutically relevant concentrations of anti-Aβ antibodies do not cross the blood brain barrier (BBB), as suggested by the theoretical calculations of the levels of both anti-Aβ antibodies and Aβ in the serum and CNS.75 However, the “peripheral sink” pathway was dependent on Aβ being exported from the CNS. The principle molecule involved in the transport of Aβ out of the brain is the low-density lipoprotein receptor-protein-1 (LRP-1),76, 77, 78 which, as suggested, binds to Aβ at the abluminal side of the endothelium and transports this peptide across the BBB and into the bloodstream.77 Interestingly, LRP-1 expression in the endothelial cells of the BBB appears to decrease significantly with normal aging and in AD, and this may also be the case in aging rodents.78 This may be one explanation for why the EV in the present study was ineffective in the older Tg2576 mice with substantial preexisting amyloid plaque deposition. This is not the first time that immunotherapy in APP transgenic mice with existing amyloid plaques did not reduce the amyloid load in the brain. Previously, it was shown that older APP/Tg mice (16–18 months old) with significant amyloid plaque deposition did not appear to respond to active immunization with Aβ, even though they produced equal levels of antibodies.79, 80
Another possibility for the lack of plaque clearance in older mice is that older plaques are refractory to degradation and clearance due to their increased fibrillar composition as well as multiple post-assembly modifications, such as cross-linking, glycation, and deposition of other plaque-associated factors, which suggests that early intervention in the disease process is highly desirable. Thus, age-related changes in the BBB and amyloid deposits may have profound implications for therapeutic approaches that rely on the entry of antibodies or an efflux of Aβ out of the brain to promote the clearance of Aβ. More recently, a new avenue for antibody-mediated clearance of Aβ from the CNS was proposed by Deane et al.,81 where they showed that anti-Aβ antibodies could lower the level of endogenous Aβ in wild-type mice and that mice deficient in neonatal Fc receptor (FcRn) had attenuated clearance. They concluded that immune complexes formed between Aβ and anti-Aβ antibodies were efficiently exported from the CNS by the FcRn and that this provided a mechanism for clearing the brain of Aβ-antibody-immune complexes that were not dependent on CNS microglia.81 Thus, several mechanisms may contribute to antibody-mediated CNS clearance of Aβ, whereas age- and disease-related changes in these clearance pathways might attenuate the effectiveness of the Aβ clearance mechanisms.
Materials and Methods
Mice
Female Tg2576 mice (H-2bxs haplotype) were bred at the animal facility of the University of California at Irvine. All animals were housed in a temperature and light-cycle controlled facility, and their care was under the guidelines of the NIH and an approved Institutional Animal Care and Use Committee (IACUC) protocol at University of California, Irvine.
Experimental Protocols
Tg2576 mice in this study were chosen from two different cohorts. Cohort I was used in the first experimental set (preventive study versus therapeutic study), and cohort II was used in the second experimental set (analysis of β-amyloid in mice with or without pre-existing memory T cells). In each experimental set, all mice were allocated randomly among experimental and control groups.
Experimental Set I
In preventive studies, two groups of 6–6.5-month-old mice were injected nine times with 100 μg/mouse adjuvanted EV (n = 8) or adjuvant alone (n = 6) as we described previously (Figure 1A).37, 38, 40 In parallel, we conducted therapeutic studies by immunization of 16–19-month-old Tg2576 mice (n = 6). Control animals of the same age were injected with adjuvant alone (n = 5). Blood was collected before injections (pre-bleed) or 10 days after each immunization. Mice were terminated at the ages of 15–15.5 months for preventive and 21–24 months for therapeutic studies, and the neuropathological changes were analyzed in their brains (Figure 1B).
Experimental Set II
To generate mice with pre-existing Th memory cells, the first group of 6–6.5-month-old Tg2576 mice (group 3, n = 8) was injected with carrier Th epitopes as we previously reported.40 Mice from control groups were injected with the same Th epitopes (group 1, n = 9) or adjuvant only (group 2, n = 5). After 4 months of resting, period mice from group 1 were injected with adjuvant only and mice from groups 2 and 3 were immunized with adjuvanted EV (Figure 4A). Blood was collected before priming and boosting (pre-bleeds) or 10 days after each boost. The behavioral study was performed after the third boost, when mice were 14–14.5 months old. At the age of 15–15.5 months, mice were terminated and the neuropathological changes were analyzed in the brains.
Detection of Anti-Aβ Antibodies by ELISA
Anti-Aβ antibodies were detected using ELISA as described previously.40, 43, 65, 66 Anti-Aβ antibody concentrations in mice were calculated using a calibration curve generated with 6E10 monoclonal antibody (Covance).
Behavioral Tests
Novel object and novel place recognition tests were used to evaluate cognition as previously described.82 Briefly, each mouse was habituated to an empty arena for 5 min 24 hr prior to testing, and, on the first day of novel object testing, mice were exposed to two identical objects placed at opposite ends of the arena for 5 min. Twenty-four hours later, the mouse was returned to the arena, this time with one familiar object and one novel object. The time spent exploring the objects was recorded for 5 min. For the place recognition test, each mouse was placed in the arena with two identical objects evenly spaced and allowed to explore for 5 min. After 24 hr, mice were placed in the arena for 5 min with one object displaced to a new location, while the other object was not moved. The recognition index represents the percentage of the time that mice spend exploring the novel or moved object. Objects used in this task were carefully selected to prevent preference or phobic behavior.
Brain Collection
At the end of each experiment, vaccinated and control mice were sacrificed for neuropathological analysis as described previously.40, 42 The left hemisphere was snap frozen and reserved for measurements of both soluble Aβ40 and Aβ42 levels by ELISA. The right hemisphere was fixed in 4% paraformaldehyde in PBS at +4°C for 24 hr for further sectioning.
Immunohistochemistry and Histostaining
50 μm-thick coronal sections of fixed hemibrains were cut using the vibratome. To assess the extent of neuropathology, several sets of equally spaced sections were selected for each mouse brain, and the following antibodies were used. Aβ cored and defused deposits were detected with 6E10 (1:1000, Covance), as described by Davtyan, Movsesyan, and Petrushina and colleagues.40, 42, 43 Activated microglia were stained with the anti-I-A/I-E (marker of MHC II alloantigens, 1:100, BD PharMingen), and astrocytes were labeled with anti-GFAP (glial fibriliary acidic protein, 1:500, Dako) antibody as we described.40, 42, 43 Fibrillar Aβ deposits were visualized using Thioflavin S (ThS; Sigma-Aldrich), and hemosiderin deposits were detected via Prussian Blue staining. Both assays were performed as described by Davtyan, Movsesyan, and Petrushina and colleagues.40, 42, 43
Quantitative Image Analysis
The numbers of 6E10- or ThS-positive plaques were analyzed throughout the whole hemisphere as well as in the cortical and hippocampal regions. Measurements for the number of blood vessels showing amyloid depositions characteristic of cerebral amyloid angiopathy (CAA) were done in 6E10-stained brain sections; activated microglial and hemosiderin-positive profiles were counted through the whole hemisphere as well. The mean semiquantitative scores per hemisphere or per neuroanatomical region in those assays were determined based on visual microscopic inspection of sets, including five coronal sections equally spaced between −0.80 and −2.92 mm with respect to Bregma, performed by three independent observers blindly for the treatment conditions.
NIH ImageJ 1.45 s software was used to analyze 6E10-positive Aβ load and the number of GFAP-positive astrocytes. For every animal, a total of 19 images (802 × 650 um each) of cortex, striatum, and hippocampus were selected at approximately the same planes (3 sections between −0.82 and −2.75 mm with respect to Bregma). The images were captured using a MD700 video camera (AmScope) and 10× objective.
Biochemical Analysis
To determine the levels of both soluble Aβ40 and Aβ42, brain homogenates were prepared at JSW Life Sciences as described previously.40 The supernatants were collected as the source of soluble aggregates for analysis of soluble Aβ40 and Aβ42 using human β-amyloid ELISA kits (Invitrogen) according to the manufacturer’s recommendations. The final values of soluble Aβ were expressed as nanograms per gram wet weight of a hemibrain.
Statistical Analysis
All statistical parameters (mean, SD, significant difference, etc.) used in the experiments were calculated using Prism 6 software (GraphPad Software). Statistically significant differences were examined using ANOVA, and post hoc comparisons were done using Tukey’s test (p value < 0.05 was considered statistically different).
Author Contributions
I.P. and H.D. designed the experiments, performed the immunizations of mice, analyzed humoral and cellular immune responses, conducted IHC studies, analyzed and interpreted data and made conclusions, and drafted the manuscript. A.D. participated in immunizations of mice and analyses of humoral and cellular immune responses. A.H. collected the brains and participated in IHC analyses of brains. G.F.P. conducted and analyzed behavioral tests. D.H.C. and A.G. participated in the design of studies and interpretation of data. M.G.A. designed the studies, mentored primary authors, regularly analyzed all data with authors, conceived the study, and made conclusions. M.G.A. and A.G. prepared the final version of the manuscript and gave it to all authors. All authors have read and approved the final manuscript for publication.
Acknowledgments
This work was supported by funding from the NIH (NS-50895 and AG-20241) and the Alzheimer's Association (IIRG-12-239626 and NIRG-13-281227). The Aβ42 peptide was provided by the University of California Alzheimer’s Disease Research Center (UCI-ADRC), which is funded by NIH/NIA Grant P50 AG16573.
Contributor Information
Anahit Ghochikyan, Email: aghochikyan@immed.org.
Michael G. Agadjanyan, Email: magadjanyan@immed.org.
References
- 1.Hardy J., Selkoe D.J. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297:353–356. doi: 10.1126/science.1072994. [DOI] [PubMed] [Google Scholar]
- 2.Hardy J.A., Higgins G.A. Alzheimer’s disease: the amyloid cascade hypothesis. Science. 1992;256:184–185. doi: 10.1126/science.1566067. [DOI] [PubMed] [Google Scholar]
- 3.Sáez-Orellana F., Godoy P.A., Bastidas C.Y., Silva-Grecchi T., Guzmán L., Aguayo L.G., Fuentealba J. ATP leakage induces P2XR activation and contributes to acute synaptic excitotoxicity induced by soluble oligomers of β-amyloid peptide in hippocampal neurons. Neuropharmacology. 2016;100:116–123. doi: 10.1016/j.neuropharm.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 4.Cerpa W., Dinamarca M.C., Inestrosa N.C. Structure-function implications in Alzheimer’s disease: effect of Abeta oligomers at central synapses. Curr. Alzheimer Res. 2008;5:233–243. doi: 10.2174/156720508784533321. [DOI] [PubMed] [Google Scholar]
- 5.Haass C., Selkoe D.J. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat. Rev. Mol. Cell Biol. 2007;8:101–112. doi: 10.1038/nrm2101. [DOI] [PubMed] [Google Scholar]
- 6.Tanzi R.E. The genetics of Alzheimer disease. Cold Spring Harb. Perspect. Med. 2012;2:2. doi: 10.1101/cshperspect.a006296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nilsberth C., Westlind-Danielsson A., Eckman C.B., Condron M.M., Axelman K., Forsell C., Stenh C., Luthman J., Teplow D.B., Younkin S.G. The ‘Arctic’ APP mutation (E693G) causes Alzheimer’s disease by enhanced Abeta protofibril formation. Nat. Neurosci. 2001;4:887–893. doi: 10.1038/nn0901-887. [DOI] [PubMed] [Google Scholar]
- 8.Sahlin C., Lord A., Magnusson K., Englund H., Almeida C.G., Greengard P., Nyberg F., Gouras G.K., Lannfelt L., Nilsson L.N. The Arctic Alzheimer mutation favors intracellular amyloid-beta production by making amyloid precursor protein less available to alpha-secretase. J. Neurochem. 2007;101:854–862. doi: 10.1111/j.1471-4159.2006.04443.x. [DOI] [PubMed] [Google Scholar]
- 9.Jonsson T., Atwal J.K., Steinberg S., Snaedal J., Jonsson P.V., Bjornsson S., Stefansson H., Sulem P., Gudbjartsson D., Maloney J. A mutation in APP protects against Alzheimer’s disease and age-related cognitive decline. Nature. 2012;488:96–99. doi: 10.1038/nature11283. [DOI] [PubMed] [Google Scholar]
- 10.Maloney J.A., Bainbridge T., Gustafson A., Zhang S., Kyauk R., Steiner P., van der Brug M., Liu Y., Ernst J.A., Watts R.J., Atwal J.K. Molecular mechanisms of Alzheimer disease protection by the A673T allele of amyloid precursor protein. J. Biol. Chem. 2014;289:30990–31000. doi: 10.1074/jbc.M114.589069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zheng X., Liu D., Roychaudhuri R., Teplow D.B., Bowers M.T. Amyloid β-Protein Assembly: Differential Effects of the Protective A2T Mutation and Recessive A2V Familial Alzheimer’s Disease Mutation. ACS Chem. Neurosci. 2015;6:1732–1740. doi: 10.1021/acschemneuro.5b00171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang X., Huang T., Zhao Y., Zheng Q., Thompson R.C., Bu G., Zhang Y.W., Hong W., Xu H. Sorting nexin 27 regulates Aβ production through modulating γ-secretase activity. Cell Rep. 2014;9:1023–1033. doi: 10.1016/j.celrep.2014.09.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Granic A., Padmanabhan J., Norden M., Potter H. Alzheimer Abeta peptide induces chromosome mis-segregation and aneuploidy, including trisomy 21: requirement for tau and APP. Mol. Biol. Cell. 2010;21:511–520. doi: 10.1091/mbc.E09-10-0850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Potter H. Review and hypothesis: Alzheimer disease and Down syndrome--chromosome 21 nondisjunction may underlie both disorders. Am. J. Hum. Genet. 1991;48:1192–1200. [PMC free article] [PubMed] [Google Scholar]
- 15.Ballatore C., Lee V.M., Trojanowski J.Q. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat. Rev. Neurosci. 2007;8:663–672. doi: 10.1038/nrn2194. [DOI] [PubMed] [Google Scholar]
- 16.Stancu I.C., Vasconcelos B., Terwel D., Dewachter I. Models of β-amyloid induced Tau-pathology: the long and “folded” road to understand the mechanism. Mol. Neurodegener. 2014;9:51. doi: 10.1186/1750-1326-9-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jack C.R., Jr., Knopman D.S., Jagust W.J., Petersen R.C., Weiner M.W., Aisen P.S., Shaw L.M., Vemuri P., Wiste H.J., Weigand S.D. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013;12:207–216. doi: 10.1016/S1474-4422(12)70291-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nelson P.T., Alafuzoff I., Bigio E.H., Bouras C., Braak H., Cairns N.J., Castellani R.J., Crain B.J., Davies P., Del Tredici K. Correlation of Alzheimer disease neuropathologic changes with cognitive status: a review of the literature. J. Neuropathol. Exp. Neurol. 2012;71:362–381. doi: 10.1097/NEN.0b013e31825018f7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Solomon B., Koppel R., Frankel D., Hanan-Aharon E. Disaggregation of Alzheimer beta-amyloid by site-directed mAb. Proc. Natl. Acad. Sci. USA. 1997;94:4109–4112. doi: 10.1073/pnas.94.8.4109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Helmuth L. New therapies. New Alzheimer’s treatments that may ease the mind. Science. 2002;297:1260–1262. doi: 10.1126/science.297.5585.1260. [DOI] [PubMed] [Google Scholar]
- 21.Klyubin I., Walsh D.M., Lemere C.A., Cullen W.K., Shankar G.M., Betts V., Spooner E.T., Jiang L., Anwyl R., Selkoe D.J., Rowan M.J. Amyloid beta protein immunotherapy neutralizes Abeta oligomers that disrupt synaptic plasticity in vivo. Nat. Med. 2005;11:556–561. doi: 10.1038/nm1234. [DOI] [PubMed] [Google Scholar]
- 22.Salomone S., Caraci F., Leggio G.M., Fedotova J., Drago F. New pharmacological strategies for treatment of Alzheimer’s disease: focus on disease modifying drugs. Br. J. Clin. Pharmacol. 2012;73:504–517. doi: 10.1111/j.1365-2125.2011.04134.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gadad B.S., Britton G.B., Rao K.S. Targeting oligomers in neurodegenerative disorders: lessons from α-synuclein, tau, and amyloid-β peptide. J. Alzheimers Dis. 2011;24(Suppl 2):223–232. doi: 10.3233/JAD-2011-110182. [DOI] [PubMed] [Google Scholar]
- 24.Panza F., Solfrizzi V., Imbimbo B.P., Tortelli R., Santamato A., Logroscino G. Amyloid-based immunotherapy for Alzheimer’s disease in the time of prevention trials: the way forward. Expert Rev. Clin. Immunol. 2014;10:405–419. doi: 10.1586/1744666X.2014.883921. [DOI] [PubMed] [Google Scholar]
- 25.Delrieu J., Ousset P.J., Caillaud C., Vellas B. ‘Clinical trials in Alzheimer’s disease’: immunotherapy approaches. J. Neurochem. 2012;120(Suppl 1):186–193. doi: 10.1111/j.1471-4159.2011.07458.x. [DOI] [PubMed] [Google Scholar]
- 26.Lemere C.A., Masliah E. Can Alzheimer disease be prevented by amyloid-beta immunotherapy? Nat. Rev. Neurol. 2010;6:108–119. doi: 10.1038/nrneurol.2009.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lobello K., Ryan J.M., Liu E., Rippon G., Black R. Targeting Beta amyloid: a clinical review of immunotherapeutic approaches in Alzheimer’s disease. Int. J. Alzheimers Dis. 2012;2012:628070. doi: 10.1155/2012/628070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ghochikyan, A., and Agadjanyan, M.G. (2012). CAD-106, a beta-amyloid-based immunotherapeutic for Alzheimer’s disease. Thompson Reuter. https://partnering.thomson-pharma.com.
- 29.Cribbs D.H., Agadjanyan M. Active and passive Abeta-immunotherapy: pre-clinical and clinical studies and future directions: part II. CNS Neurol Disord Drug Targets. 2009;8:1–6. doi: 10.2174/187152709787601849. [DOI] [PubMed] [Google Scholar]
- 30.Wisniewski T. AD vaccines: conclusions and future directions. CNS Neurol. Disord. Drug Targets. 2009;8:160–166. doi: 10.2174/187152709787847289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Agadjanyan M.G., Petrovsky N., Ghochikyan A. A fresh perspective from immunologists and vaccine researchers: active vaccination strategies to prevent and reverse Alzheimer’s disease. Alzheimers Dement. 2015;11:1246–1259. doi: 10.1016/j.jalz.2015.06.1884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nicoll J.A., Wilkinson D., Holmes C., Steart P., Markham H., Weller R.O. Neuropathology of human Alzheimer disease after immunization with amyloid-beta peptide: a case report. Nat. Med. 2003;9:448–452. doi: 10.1038/nm840. [DOI] [PubMed] [Google Scholar]
- 33.Ferrer I., Boada Rovira M., Sánchez Guerra M.L., Rey M.J., Costa-Jussá F. Neuropathology and pathogenesis of encephalitis following amyloid-beta immunization in Alzheimer’s disease. Brain Pathol. 2004;14:11–20. doi: 10.1111/j.1750-3639.2004.tb00493.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Masliah E., Hansen L., Adame A., Crews L., Bard F., Lee C., Seubert P., Games D., Kirby L., Schenk D. Abeta vaccination effects on plaque pathology in the absence of encephalitis in Alzheimer disease. Neurology. 2005;64:129–131. doi: 10.1212/01.WNL.0000148590.39911.DF. [DOI] [PubMed] [Google Scholar]
- 35.Ghochikyan A. Rationale for peptide and DNA based epitope vaccines for Alzheimer’s disease immunotherapy. CNS Neurol. Disord. Drug Targets. 2009;8:128–143. doi: 10.2174/187152709787847298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Doody R.S., Thomas R.G., Farlow M., Iwatsubo T., Vellas B., Joffe S., Kieburtz K., Raman R., Sun X., Aisen P.S., Alzheimer’s Disease Cooperative Study Steering Committee. Solanezumab Study Group Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 2014;370:311–321. doi: 10.1056/NEJMoa1312889. [DOI] [PubMed] [Google Scholar]
- 37.Agadjanyan M.G., Ghochikyan A., Petrushina I., Vasilevko V., Movsesyan N., Mkrtichyan M., Saing T., Cribbs D.H. Prototype Alzheimer’s disease vaccine using the immunodominant B cell epitope from beta-amyloid and promiscuous T cell epitope pan HLA DR-binding peptide. J. Immunol. 2005;174:1580–1586. doi: 10.4049/jimmunol.174.3.1580. [DOI] [PubMed] [Google Scholar]
- 38.Cribbs D.H., Ghochikyan A., Vasilevko V., Tran M., Petrushina I., Sadzikava N., Babikyan D., Kesslak P., Kieber-Emmons T., Cotman C.W., Agadjanyan M.G. Adjuvant-dependent modulation of Th1 and Th2 responses to immunization with beta-amyloid. Int. Immunol. 2003;15:505–514. doi: 10.1093/intimm/dxg049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Petrushina I., Tran M., Sadzikava N., Ghochikyan A., Vasilevko V., Agadjanyan M.G., Cribbs D.H. Importance of IgG2c isotype in the immune response to b-amyloid in APP/Tg mice. Neurosci. Lett. 2003;338:5–8. doi: 10.1016/s0304-3940(02)01357-5. [DOI] [PubMed] [Google Scholar]
- 40.Davtyan H., Ghochikyan A., Petrushina I., Hovakimyan A., Davtyan A., Poghosyan A., Marleau A.M., Movsesyan N., Kiyatkin A., Rasool S. Immunogenicity, efficacy, safety, and mechanism of action of epitope vaccine (Lu AF20513) for Alzheimer’s disease: prelude to a clinical trial. J. Neurosci. 2013;33:4923–4934. doi: 10.1523/JNEUROSCI.4672-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Davtyan H., Ghochikyan A., Hovakimyan A., Petrushina I., Yu J., Flyer D., Madsen P.J., Pedersen L.O., Cribbs D.H., Agadjanyan M.G. Immunostimulant patches containing Escherichia coli LT enhance immune responses to DNA- and recombinant protein-based Alzheimer’s disease vaccines. J. Neuroimmunol. 2014;268:50–57. doi: 10.1016/j.jneuroim.2014.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Movsesyan N., Ghochikyan A., Mkrtichyan M., Petrushina I., Davtyan H., Olkhanud P.B., Head E., Biragyn A., Cribbs D.H., Agadjanyan M.G. Reducing AD-like pathology in 3xTg-AD mouse model by DNA epitope vaccine - a novel immunotherapeutic strategy. PLoS ONE. 2008;3:e2124. doi: 10.1371/journal.pone.0002124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Petrushina I., Ghochikyan A., Mktrichyan M., Mamikonyan G., Movsesyan N., Davtyan H., Patel A., Head E., Cribbs D.H., Agadjanyan M.G. Alzheimer’s disease peptide epitope vaccine reduces insoluble but not soluble/oligomeric Abeta species in amyloid precursor protein transgenic mice. J. Neurosci. 2007;27:12721–12731. doi: 10.1523/JNEUROSCI.3201-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Carrillo-Mora P., Luna R., Colín-Barenque L. Amyloid beta: multiple mechanisms of toxicity and only some protective effects? Oxid. Med. Cell. Longev. 2014;2014:795375. doi: 10.1155/2014/795375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.De Felice F.G., Wu D., Lambert M.P., Fernandez S.J., Velasco P.T., Lacor P.N., Bigio E.H., Jerecic J., Acton P.J., Shughrue P.J. Alzheimer’s disease-type neuronal tau hyperphosphorylation induced by A beta oligomers. Neurobiol. Aging. 2008;29:1334–1347. doi: 10.1016/j.neurobiolaging.2007.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lesné S., Koh M.T., Kotilinek L., Kayed R., Glabe C.G., Yang A., Gallagher M., Ashe K.H. A specific amyloid-beta protein assembly in the brain impairs memory. Nature. 2006;440:352–357. doi: 10.1038/nature04533. [DOI] [PubMed] [Google Scholar]
- 47.Shankar G.M., Li S., Mehta T.H., Garcia-Munoz A., Shepardson N.E., Smith I., Brett F.M., Farrell M.A., Rowan M.J., Lemere C.A. Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat. Med. 2008;14:837–842. doi: 10.1038/nm1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wilcock D.M., Rojiani A., Rosenthal A., Subbarao S., Freeman M.J., Gordon M.N., Morgan D. Passive immunotherapy against Abeta in aged APP-transgenic mice reverses cognitive deficits and depletes parenchymal amyloid deposits in spite of increased vascular amyloid and microhemorrhage. J. Neuroinflammation. 2004;1:24. doi: 10.1186/1742-2094-1-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tarawneh R., Holtzman D.M. Critical issues for successful immunotherapy in Alzheimer’s disease: development of biomarkers and methods for early detection and intervention. CNS Neurol. Disord. Drug Targets. 2009;8:144–159. doi: 10.2174/187152709787847324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kokjohn T.A., Roher A.E. Antibody responses, amyloid-beta peptide remnants and clinical effects of AN-1792 immunization in patients with AD in an interrupted trial. CNS Neurol. Disord. Drug Targets. 2009;8:88–97. doi: 10.2174/187152709787847315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hsiao K., Chapman P., Nilsen S., Eckman C., Harigaya Y., Younkin S., Yang F., Cole G. Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice. Science. 1996;274:99–102. doi: 10.1126/science.274.5284.99. [DOI] [PubMed] [Google Scholar]
- 52.Mouri A., Noda Y., Hara H., Mizoguchi H., Tabira T., Nabeshima T. Oral vaccination with a viral vector containing Abeta cDNA attenuates age-related Abeta accumulation and memory deficits without causing inflammation in a mouse Alzheimer model. FASEB J. 2007;21:2135–2148. doi: 10.1096/fj.06-7685com. [DOI] [PubMed] [Google Scholar]
- 53.Frye C.A., Walf A.A. Effects of progesterone administration and APPswe+PSEN1Deltae9 mutation for cognitive performance of mid-aged mice. Neurobiol. Learn. Mem. 2008;89:17–26. doi: 10.1016/j.nlm.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 54.Cohen S.J., Stackman R.W., Jr. Assessing rodent hippocampal involvement in the novel object recognition task. A review. Behav. Brain Res. 2015;285:105–117. doi: 10.1016/j.bbr.2014.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Guzmán-Ramos K., Moreno-Castilla P., Castro-Cruz M., McGaugh J.L., Martínez-Coria H., LaFerla F.M., Bermúdez-Rattoni F. Restoration of dopamine release deficits during object recognition memory acquisition attenuates cognitive impairment in a triple transgenic mice model of Alzheimer’s disease. Learn. Mem. 2012;19:453–460. doi: 10.1101/lm.026070.112. [DOI] [PubMed] [Google Scholar]
- 56.Balderas I., Rodriguez-Ortiz C.J., Salgado-Tonda P., Chavez-Hurtado J., McGaugh J.L., Bermudez-Rattoni F. The consolidation of object and context recognition memory involve different regions of the temporal lobe. Learn. Mem. 2008;15:618–624. doi: 10.1101/lm.1028008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Dere E., Huston J.P., De Souza Silva M.A. The pharmacology, neuroanatomy and neurogenetics of one-trial object recognition in rodents. Neurosci. Biobehav. Rev. 2007;31:673–704. doi: 10.1016/j.neubiorev.2007.01.005. [DOI] [PubMed] [Google Scholar]
- 58.Bard F., Cannon C., Barbour R., Burke R.L., Games D., Grajeda H., Guido T., Hu K., Huang J., Johnson-Wood K. Peripherally administered antibodies against amyloid beta-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat. Med. 2000;6:916–919. doi: 10.1038/78682. [DOI] [PubMed] [Google Scholar]
- 59.Schenk D., Barbour R., Dunn W., Gordon G., Grajeda H., Guido T., Hu K., Huang J., Johnson-Wood K., Khan K. Immunization with amyloid-beta attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature. 1999;400:173–177. doi: 10.1038/22124. [DOI] [PubMed] [Google Scholar]
- 60.Morgan D., Diamond D.M., Gottschall P.E., Ugen K.E., Dickey C., Hardy J., Duff K., Jantzen P., DiCarlo G., Wilcock D. A beta peptide vaccination prevents memory loss in an animal model of Alzheimer’s disease. Nature. 2000;408:982–985. doi: 10.1038/35050116. [DOI] [PubMed] [Google Scholar]
- 61.Janus C., Pearson J., McLaurin J., Mathews P.M., Jiang Y., Schmidt S.D., Chishti M.A., Horne P., Heslin D., French J. A beta peptide immunization reduces behavioural impairment and plaques in a model of Alzheimer’s disease. Nature. 2000;408:979–982. doi: 10.1038/35050110. [DOI] [PubMed] [Google Scholar]
- 62.Wisniewski T., Goñi F. Immunotherapy for Alzheimer’s disease. Biochem. Pharmacol. 2014;88:499–507. doi: 10.1016/j.bcp.2013.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lannfelt L., Relkin N.R., Siemers E.R. Amyloid-ß-directed immunotherapy for Alzheimer’s disease. J. Intern. Med. 2014;275:284–295. doi: 10.1111/joim.12168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Godyń J., Jończyk J., Panek D., Malawska B. Therapeutic strategies for Alzheimer’s disease in clinical trials. Pharmacol. Rep. 2016;68:127–138. doi: 10.1016/j.pharep.2015.07.006. [DOI] [PubMed] [Google Scholar]
- 65.Ghochikyan A., Mkrtichyan M., Petrushina I., Movsesyan N., Karapetyan A., Cribbs D.H., Agadjanyan M.G. Prototype Alzheimer’s disease epitope vaccine induced strong Th2-type anti-Abeta antibody response with Alum to Quil A adjuvant switch. Vaccine. 2006;24:2275–2282. doi: 10.1016/j.vaccine.2005.11.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Davtyan H., Mkrtichyan M., Movsesyan N., Petrushina I., Mamikonyan G., Cribbs D.H., Agadjanyan M.G., Ghochikyan A. DNA prime-protein boost increased the titer, avidity and persistence of anti-Abeta antibodies in wild-type mice. Gene Ther. 2010;17:261–271. doi: 10.1038/gt.2009.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Evans C.F., Davtyan H., Petrushina I., Hovakimyan A., Davtyan A., Hannaman D., Cribbs D.H., Agadjanyan M.G., Ghochikyan A. Epitope-based DNA vaccine for Alzheimer’s disease: translational study in macaques. Alzheimers Dement. 2014;10:284–295. doi: 10.1016/j.jalz.2013.04.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Davtyan H., Ghochikyan A., Petrushina I., Hovakimyan A., Davtyan A., Cribbs D.H., Agadjanyan M.G. The MultiTEP platform-based Alzheimer’s disease epitope vaccine activates a broad repertoire of T helper cells in nonhuman primates. Alzheimers Dement. 2014;10:271–283. doi: 10.1016/j.jalz.2013.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kawarabayashi T., Younkin L.H., Saido T.C., Shoji M., Ashe K.H., Younkin S.G. Age-dependent changes in brain, CSF, and plasma amyloid (beta) protein in the Tg2576 transgenic mouse model of Alzheimer’s disease. J. Neurosci. 2001;21:372–381. doi: 10.1523/JNEUROSCI.21-02-00372.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sevigny J., Chiao P., Bussière T., Weinreb P.H., Williams L., Maier M., Dunstan R., Salloway S., Chen T., Ling Y. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537:50–56. doi: 10.1038/nature19323. [DOI] [PubMed] [Google Scholar]
- 71.Vasilevko V., Cribbs D.H. Novel approaches for immunotherapeutic intervention in Alzheimer’s disease. Neurochem. Int. 2006;49:113–126. doi: 10.1016/j.neuint.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 72.DeMattos R.B., Bales K.R., Cummins D.J., Dodart J.C., Paul S.M., Holtzman D.M. Peripheral anti-A beta antibody alters CNS and plasma A beta clearance and decreases brain A beta burden in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA. 2001;98:8850–8855. doi: 10.1073/pnas.151261398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.DeMattos R.B., Bales K.R., Parsadanian M., O’Dell M.A., Foss E.M., Paul S.M., Holtzman D.M. Plaque-associated disruption of CSF and plasma amyloid-beta (Abeta) equilibrium in a mouse model of Alzheimer’s disease. J. Neurochem. 2002;81:229–236. doi: 10.1046/j.1471-4159.2002.00889.x. [DOI] [PubMed] [Google Scholar]
- 74.DeMattos R.B., Bales K.R., Cummins D.J., Paul S.M., Holtzman D.M. Brain to plasma amyloid-beta efflux: a measure of brain amyloid burden in a mouse model of Alzheimer’s disease. Science. 2002;295:2264–2267. doi: 10.1126/science.1067568. [DOI] [PubMed] [Google Scholar]
- 75.Holtzman D.M., Bales K.R., Paul S.M., DeMattos R.B. Abeta immunization and anti-Abeta antibodies: potential therapies for the prevention and treatment of Alzheimer’s disease. Adv. Drug Deliv. Rev. 2002;54:1603–1613. doi: 10.1016/s0169-409x(02)00158-8. [DOI] [PubMed] [Google Scholar]
- 76.Shibata M., Yamada S., Kumar S.R., Calero M., Bading J., Frangione B., Holtzman D.M., Miller C.A., Strickland D.K., Ghiso J., Zlokovic B.V. Clearance of Alzheimer’s amyloid-ss(1-40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. J. Clin. Invest. 2000;106:1489–1499. doi: 10.1172/JCI10498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zlokovic B.V. Clearing amyloid through the blood-brain barrier. J. Neurochem. 2004;89:807–811. doi: 10.1111/j.1471-4159.2004.02385.x. [DOI] [PubMed] [Google Scholar]
- 78.Deane R., Wu Z., Sagare A., Davis J., Du Yan S., Hamm K., Xu F., Parisi M., LaRue B., Hu H.W. LRP/amyloid beta-peptide interaction mediates differential brain efflux of Abeta isoforms. Neuron. 2004;43:333–344. doi: 10.1016/j.neuron.2004.07.017. [DOI] [PubMed] [Google Scholar]
- 79.Das P., Murphy M.P., Younkin L.H., Younkin S.G., Golde T.E. Reduced effectiveness of Abeta1-42 immunization in APP transgenic mice with significant amyloid deposition. Neurobiol. Aging. 2001;22:721–727. doi: 10.1016/s0197-4580(01)00245-7. [DOI] [PubMed] [Google Scholar]
- 80.Zhou J., Fonseca M.I., Kayed R., Hernandez I., Webster S.D., Yazan O., Cribbs D.H., Glabe C.G., Tenner A.J. Novel Abeta peptide immunogens modulate plaque pathology and inflammation in a murine model of Alzheimer’s disease. J. Neuroinflammation. 2005;2:28. doi: 10.1186/1742-2094-2-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Deane R., Sagare A., Hamm K., Parisi M., LaRue B., Guo H., Wu Z., Holtzman D.M., Zlokovic B.V. IgG-assisted age-dependent clearance of Alzheimer’s amyloid beta peptide by the blood-brain barrier neonatal Fc receptor. J. Neurosci. 2005;25:11495–11503. doi: 10.1523/JNEUROSCI.3697-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Passos G.F., Medeiros R., Cheng D., Vasilevko V., Laferla F.M., Cribbs D.H. The bradykinin B1 receptor regulates Aβ deposition and neuroinflammation in Tg-SwDI mice. Am. J. Pathol. 2013;182:1740–1749. doi: 10.1016/j.ajpath.2013.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]